Abstract
Integrins are transmembrane receptors that possess distinct ligand-binding specificities in the extracellular domain and signaling properties in the cytoplasmic domain. While most integrins have a short cytoplasmic tail, integrin β4 has a long cytoplasmic tail that can indirectly interact with the actin cytoskeleton. Additionally, 'inside-out' signals can induce integrins to adopt a high-affinity extended conformation for their appropriate ligands. These properties enable integrins to transmit bidirectional cellular signals, making it a critical regulator of various biological processes.
Integrin expression and function are tightly linked to various aspects of tumor progression, including initiation, angiogenesis, cell motility, invasion, and metastasis. Certain integrins have been shown to drive tumorigenesis or amplify oncogenic signals by interacting with corresponding receptors, while others have marginal or even suppressive effects. Additionally, different α/β subtypes of integrins can exhibit opposite effects. Integrin-mediated signaling pathways including Ras- and Rho-GTPase, TGFβ, Hippo, Wnt, Notch, and sonic hedgehog (Shh) are involved in various stages of tumorigenesis. Therefore, understanding the complex regulatory mechanisms and molecular specificities of integrins are crucial to delaying cancer progression and suppressing tumorigenesis. Furthermore, the development of integrin-based therapeutics for cancer are of great importance.
This review provides an overview of integrin-dependent bidirectional signaling mechanisms in cancer that can either support or oppose tumorigenesis by interacting with various signaling pathways. Finally, we focus on the future opportunities for emergent therapeutics based on integrin agonists.
Video Abstract
Similar content being viewed by others
Introduction
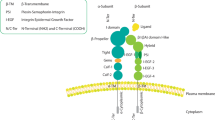
The first integrin is identified in 1986, as an integral membrane complex protein that plays a critical role in the association between the extracellular matrix (ECM) and the cytoskeleton [1, 2]. Integrins are a family of 24 heterodimeric receptors composed of stable covalently linked 18 α-subunits and 8 β-subunits, named according to their α/β-subunit compositions [3]. The α/β-subunits contain approximately 1000/750 amino acids, respectively [4]. Each subunit has a transmembrane helix and a cytoplasmic tail, which forms a ‘head’ (extracellular segment) supported by two α/β-subunit ‘legs’ (membrane-spanning regions and cytoplasmic tails) (Fig. 1). The head consists of Ig-like thigh and calf domains (C1 and C2)/Ig-like hybrid, EGF-like, and b-terminal domains (bTD), respectively [3,4,5]. Most integrins can mediate cell adhesion to various ECM proteins or ligands secreted by other cells and cellular counter receptors, such as intercellular adhesion molecule (ICAM) and vascular cell adhesion protein (VCAM) [6]. Correspondingly, many ECM and cell surface adhesion proteins can bind to a wide variety of integrin subtypes [4]. According to the characteristics of interaction, the contact between integrin and its ligand can be divided into four categories. Category I integrins, such as αV, α5β1, α8β1 and αIIbβ3, recognize vast ECM and soluble vascular ligands which contain RGD tripeptide active site [7, 8]. Category II integrins composed of α4β1, α4β7, α9β1, αEβ7 and β2, majorly identify fibronectin that have LDV peptide with RGD ligands related sequence [4]. Category III integrins comprising α1β1, α2β1, α10β1, and α11β1 are known to identify laminin or collagen that have collagenous GFOGER motif [9]; Category IV integrins like α3β1, α6β1, α7β1 and α6β4 recognize laminin, but the binding site is still unclear [4].
During the development process, integrin exerts an important role in cell rearrangement, migration and differentiation [10]. In myoblast and adipocyte differentiation, integrin α6 linking to laminin expression increases with a concurrent decrease in integrin α5 linking to fibronectin expression. However, when the dynamic balance is biased towards integrin α5, the cells may remain in the proliferation phase [11]. Integrin α6β4 induces epithelial cell proliferation and cooperation with its ligand laminin 5 regulates Ras-mediated keratinocytes differentiation in normal skin [12]. Gonadogenesis in C. elegans requires integrin (two α and one β subunit, αina-1/βpat-3)-mediated distal tip cell (DTC) migration. A dominant-negative form of integrin exchanges αina-1/βpat-3 and αpat-2/βpat-3 pairs cause DCT migration disorders. The integrin complementary expression is sufficient for the developmental process without ECM regulation [10, 11]. However, for muscle function, primordial germ cell (PGC) and neuronal cell migration, the regulation of ECM and surrounding cells is necessary for integrin-depended migration and development [11].
In addition, crosstalk between integrins and growth factor receptors (GFRs) is also necessary for the normal development process. For example, the phosphorylation of vascular endothelial growth factor receptor-2 (VEGFR-2) is regulated by integrin αvβ3 to recruit bone marrow cells in angiogenic sites [13]. Meanwhile, in human umbilical vein endothelial cells, VEGF2 activates integrin αvβ3 to regulate adhesion and migration [14]. Skin homeostasis needs the epidermal growth factor receptor (EGFR) association with integrin αvβ3 in human endothelial cells. Integrin-Src signaling-mediated adhesion to the basement membrane extracellular matrix leads to EGFR signaling activation by controlling four tyrosine phosphorylation [15]. Integrin αvβ6 and αvβ8 could activate TGFβ by promoting cleavage of latency-associated peptides (LAPs) to induce a conformational change [16, 17]. Consequently, the TGFβ up-regulates integrin α5β1 and αIIbβ3 in keratinocytes [18]. Of note, although GFRs and integrins co-localize at the cell membrane, they do not interact directly with each other, which needs to be explored further.
Integrins are an overarching regulator of pathophysiological progress, such as wound healing, tissue inflammation, tissue fibrosis, autoimmunity and metabolic disorders, in multicellular contexts of numerous diseases [19]. Integrin β1, as a subunit, comprises many heterodimers and plays a crucial role in wound healing due to its expression on various basement membrane cells and connective tissue cells [20]. Integrin αvβ1 can facilitate the differentiation of fibroblasts into myofibroblasts which may lead the wound closure, granulation tissue formation [21] and tissue fibrosis [22]. Integrin α5β1 connects with fibronectin to mediate keratinocyte proliferation which contributes to matrix adhesion during the invasion progress of connective tissue cells into the wound clot [23]. Integrin α9β1 is associated with wound re-epithelization [24] and integrin α11β1 is involved in the collagen remodeling of granulation tissue [25]. Integrin αv, the major regulator of TGFβ, associates with various β subunits for different roles. For instance, integrin αvβ1 induces ECM remodeling to regulate immune cell functions [26]. More so, integrin αvβ6 [27] and integrin αvβ8 [28] regulate immune response through activating TGFβ. Increasing persuasive research indicates that integrin αvβ3 is involved in inflammation response induced by macrophage activation, osteoclast development and inflammatory arthropathies [29,30,31]. Integrin αvβ3-mediated inflammatory process contributes to the pathogenesis of rheumatoid arthritis and the progress of related arthropathies [32]. Studies have shown that integrin regulates tissue fibrosis via binding to ECM to induce cell–cell and cell–matrix interactions [33]. Correspondingly, integrin αvβ5 [34], αvβ6 [35] and αvβ8 [36] mainly expressed in epithelial cells and fibroblasts, induce lung fibrosis through activating TGFβ. Moreover, integrin α1/5/6 [37], integrin β1 [38] and integrin β6 [39] are associated with fibrosis in the liver diseases such as chronic hepatitis B/C. Integrin β6 is also associated with primary sclerosing cholangitis (PSC) [39]. Integrin β6 [40], α3 [41] and α11β1 [42] are linked to human kidney fibrosis through inducing neovascularization and fibroblast differentiation. Given the contribution of integrin in multiple cell functions of both normal and diseased tissue, it is imminent to explore the involvement of integrin signaling in cancer growth and metastasis [1]. Here, we mainly review the newly discovered functions of integrin signaling in cancer and the emergent therapeutic opportunities dependent on mechanisms.
Integrin signaling in cancer
Integrin signaling in cancer initiation and tumor growth
Dysfunction of normal cells acclimates the initiation and progression of malignancy [43]. Integrin binding with ECM is necessary for cancer initiating cells to sense and respond to the tumor microenvironment [44]. Studies indicate that integrins function as cell surface markers, as well as functional regulators of cancer stem cells, during cancer initiation [44]. Integrin α6 also called CD49f, a laminin-binding receptor, is the richest and most common cancer stem cell marker, expressed highly in many cancers including colorectal cancer [45], breast cancer [46], skin squamous cell carcinoma [47] and glioblastoma [48]. In glioblastoma stem-like cells (GSCs), integrin α6 regulates adverse stem-associated features according to the different molecular subtypes. Integrin α6 plays a crucial role in maintaining cancer stemness in proneural GSCs. However, in mesenchymal GSCs, integrin α6 does not have an impact on stemness and self-renewal. Silencing of integrin α6 affects DNA damage repair machinery and cell cycle thereby increasing mesenchymal GSCs radiosensitivity to ionizing radiation [49]. In triple-negative breast cancer (TNBC), integrin α6 high or low populations are isolated from TgMFT121, Brca1f/f p53f/f and TgWAP-Cre mice tumors by FACS and shown to activate focal adhesion kinase (FAK), but more significantly by high integrin α6 population cell. The activation of FAK induces the expression of Polycomb complex protein BMI, a stem cell factor, which contributes to the initiation of TNBC [50]. Notably, integrin β4, a combination of integrin α6, has been reported to be involved in lung development and normal lung stem cells. It has been used as a marker to isolate epithelial stem cells from the mouse lung tissue [51]. Indeed, studies have linked integrin β4 to self-renewal and proliferation of lung cancer stem cells during the lung cancer progression [43].
Integrins control most cell survival, proliferation and differentiation. They act through mediating cytoskeletal linkage between cell adhesion to ECM and nuclear envelope as well as mechanotransduction. Given its fundamental function, integrins play positive or negative roles in numerous diseases [52]. Different α/β integrin combinations mediate specificities in cancer [2, 53]. As type I transmembrane glycoproteins, the stable flux of integrins between plasma membrane and intracellular pools drive cells to bind to extracellular ligands and transmit intracellular signals [53]. The cooperation between integrins and receptor tyrosine kinases (RTKs) has been shown to drive intracellular signalings that promote cancer cell proliferation. For instance, integrin α6β4 amplifies oncogenic signaling via cooperating with EGFR, ErbB2 and Met RTKs [54]; integrin αvβ3 interacts with platelet derived growth factor (PDGF) receptor to enhance the growth of PDGF-oversecreting gliomas [55]. While the crosstalk between these integrins and RTKs amplifies the signaling output, other integrin subtypes work in parallel with RTKs-dependent ones to either promote or suppress tumorigenesis [1]. Integrin αvβ5, αvβ6, and αvβ8 activate TGFβ to induce growth inhibiting effect and mediate migration and invasion in tumor cells [1, 56]. However, since TGFβ itself plays a dual role in tumorigenesis and progression, and its function in tumor and stromal cells is not identical [52], the integrin and TGFβ crosstalk in tumor growth still needs to be explored deeply.
Integrin signaling in cancer adhesion, invasion and metastasis
Increasing evidence indicates that integrin-mediated RTK signaling pathways are also implicated in tumor migration and invasion through cell–cell adhesion and cell motility. The activation of integrins were initiated through binding interactions involving integrin α/β subunits, extracellular ligands, FAK, Src, and associated activation components like Crk-associated substrate (CAS), paxillin, and talin (Fig. 2). Two important regulatory mechanisms induced by integrins have an imperative impact on this process. Initially, FAK signaling involved in RTKs induces tyrosine phosphorylation of E-cadherin-β-catenin complex. FAK acts as an integrin-regulated scaffold protein to recruit src-family kinases (SFKs) for focal adhesions which is crucial for cell invasion in cancers [12]. Additionally, integrin-linked kinase (ILK)-mediated epithelial-mesenchymal transition (EMT) exerts great contribution to increase cancer cell adhesion and invasion [57]. This process is majorly associated with Snail/Slug-reduced transcription and expression of E-cadherin [12] as well as AP1-induced matrix metalloproteinase 9 (MMP9) expression [57]. Integrin αvβ3 activates MMP2 specifically to facilitate cancer cell migration and invasion by leading the basement membrane degradation [58]. Simultaneously, this degradation develops new migration binding sites for other ones. The β4 tail of integrin α6β4 functions as an invasion signal adaptor and amplifier, promoting tumor invasion and inhibiting apoptosis. Therefore, the dysfunction of α6β4-RTKs signaling disrupts hemidesmosomes (adhesion complexes which regulate stable cell–matrix adhesion in the basement membrane by connecting intracellular keratin filaments with extracellular matrix) and leads to tumor invasion [12].
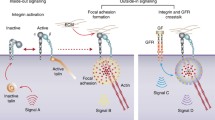
Besides, the engagement of integrin and ECM protein activates Rho- and Ras-GTPase which in turn controls cell adhesion, migration and invasion by regulating the dynamics of the actin cytoskeleton (outside-in signaling) [59]. Accordingly, both Rho and Ras superfamily proteins influence the interaction of integrin and ECM ligand (inside-out signaling) [60].
-
(1)
Outside-in signaling: This signaling mediates cellular responses induced by ligand binding to integrins. Integrin-induced adhesion is dependent on the relative activities of RhoA and Rac1. In the area of small nascent adhesions, Rac1 activation is accompanied by RhoA suppression. However, with large focal adhesion formation, RhoA but not Rac1 plays a major role. Activated integrin initiates two major downstream signaling pathways by activating FAK and Src kinases, gathered within the intracellular tails of β integrins. The FAK-Src complex in turn activates adhesion-associated adaptor proteins, such as paxillin and p130Cas subsequently binding and activation downstream adaptors [59]. What’s more, in the regulation of Rac-dependent adhesion by integrin engagement, ILK plays a circular role. ILK localizes to integrin β1 and β3 to form a scaffolding complex, which can activate PINCH1/2 and α/β parvin adaptor proteins [61].
-
(2)
Inside-out signaling: This signaling activates the ligand binding function of integrins. Rho- and Ras-GTPase encourage the binding of integrins to ligands by converting to a high affinity state. Ras, R-Ras and Rap1-GTPase may cause integrin activation which regulate cytoskeleton remodeling and integrin-dependent adhesion and migration on collagen [60]. Crosstalk and balance between Rho, Rac and Cdc42-GTPase also play a critical role in integrin-dependent invasion.
Cancer metastasis is the leading cause of cancer mortality. Metastasis is a cascading process including degrading the basement membrane and escaping from the primary tumor, accessing and surviving in the circulatory system, colonizing and proliferating in the parenchyma of the target organ [12, 43]. The properties of integrins suggest that they are crucial for the cascade process of cancer cell metastasis. Firstly, the interaction between integrins and ligands contributes to the degrading or remodeling of the ECM which is necessary for tumor cell escape [12]. Secondly, integrin binds various VEGFs and their receptors to form a complex which is required for active angiogenesis [62]. Finally, integrin-RTK signals regulate cell response to metastatic sites and initiate the metastatic cell survival, colonization and infinite proliferation in the targeting organ [63]. Notably, the metastatic process of several cancers presents extremely high levels of certain integrin forms. For example, hypoxia-inducibe factor (HIF) increases the expression of integrin α5β1 to accelerate metastasis of breast cancer towards lymph nodes and lung [64]. There is also research showing that integrin α5β1-mediated by kindlin-1 is involved in the early steps of breast cancer metastasis [65]. The crosstalk between integrin α4β1 and VCAM-1 induce melanoma and lymphoma metastasis to lung or spleen [66]. And as a pro-metastatic factor, L1-CAM interacts with integrin α5β1 and αVβ1 to promote numerous cancer metastasis, such as pancreatic ductal adenocarcinoma (PDAC), colorectal cancer as well as ovarian and endometrial cancers [67].
Integrin-mediated downstream signaling pathways in cancer
As a transmembrane receptor, integrin senses the content and stiffness of the surrounding ECM and biochemical signaling to mediate intracellular signal transduction, which occurs from adhesion sites or endosomes. The binding of ECM ligand (outside-in signaling) and specific cytoplasmic activators (inside-out signaling) to integrin lead to integrin-induced non-receptor tyrosine kinases activation. Studies show that the role of integrin in mechanotransduction and malignancy is bound to FAK-Src signaling activation [1]. FAK is the first kinase activated by integrin clustering via interaction between its C-terminal domain and integrin-containing components such as paxillin and talin. The activated FAK manifests as autophosphorylation at Y397, which leads to SFKs activation [68]. Well, the individual integrin subtypes including integrin β3 and integrin α4β1 directly stimulate the activation of Src by binding to its SH3 domain independent of FAK. Meanwhile, integrin α1β1, α5β1 and αvβ3 also activate the adaptor protein SHC through a palmitoylated SFK, such as Fyn or Yes. Numerous signalings, triggered by FAK-Src activation, such as Rho GTPase, TGFβ, Hippo, Wnt/β-Catenin and metabolism, require integrin-mediated ‘outside-in’ signals to drive diverse cellular functions [1]. As downstream pathway initiated by integrin, the FAK-Src or ILK signaling triggers uncontrolled mitogenic and survival or uncontrolled actin assembly to regulate mitogenic signaling as well as related gene expression, which subsequently regulates a series of cellular processes and biological events, including cell survival, proliferation, migration, self-renewal, EMT, and cell stemness maintenance [69] (Fig. 3).
Integrin signaling network in cancer. Integrin interaction with RTKs mediates uncontrolled mitogenic and survival or uncontrolled actin assembly to regulate mitogenic signaling as well as related gene expression via FAK-Src (integrin/RTKs) or ILK (integrin) signaling. Activated integrin signaling controls cancer initiation and tumor growth, cancer cell adhesion and adhesion-induced migration, invasion and metastasis. It is associated with incidence and mortality of cancers
Integrin-mediated Rho GTPase signaling
Rho GTPase, a family of small G proteins, plays a crucial role in modulating cytoskeleton dynamics, associated with cell polarity, motility, growth, proliferation and survival. In human, 20 Rho GTPase subfamilies have been verified and Rho (RhoA/B/C), Rac (Rac1/2/3/G) and Cdc42 (Cdc42, RhoQ/J) are well-studied Rho GTPase [70]. Rho GTPase signaling is mainly activated by extracellular signals such as cytokines, ECM protein or mechanical signal such as integrin. The function of these Rho GTPase in cell contractility and polarization is tightly regulated by integrin-induced tyrosine phosphorylation of FAK [71]. Integrin triggers FAK-Src signaling and subsequently leads Rho, Rac and Cdc42 activation. The activated Rho GTPase controls extension of lamellipodia and filopodia to form focal adhesions and actin stress fibers, which induces tumorigenesis and maligant progression, such as cell migration and invasion in cancer (Fig. 4). In breast cancer cells, integrin regulates epithelial adhesion, polarity and invasion by inducing mechanotransduction and Rho activation [72]. Meanwhile, FAK-induced Rho signaling regulates cell proliferation and self-renewal which is associated with YAP/TAZ and AP-1 activation [73]. In cancer-associated fibroblasts (CAFs), integrin-induced Rho activation triggers CAFs to acquire a contractile phenotype to regulate tumor stromal remodeling, contributing to cell migration, invasion and metastasis [74]. In PDAC, integrin α6β1 regulates Rac1 and Rho activation to modulate cell migratory and TGFβ1 activation, respectively, via an Eps8/Abi1/Sos1 axis [75]. More so, integrin β1 concerts with Rac1 during peripheral regeneration to regulate fibroblast-derived TNC (tenascin-C)-mediated Schwann cell migration [76]. Notably, integrin-dependent cell adhesion and aggregation are also regulated by Rho GTPase. For instance, integrin αIIbβ3-induced platelet aggregation is suppressed by botulinum C3 exoenzyme, an inhibitor of Rho GTPase. This exoenzyme can also inhibit integrin αLβ3, αLβ2, α4β1 and α5β1, known to regulate lymphocyte and fibroblast activation [77]. Furthermore, FAK modulates Rho activation, in turn, this activation simultaneously increases cell contractility and disassembly of focal adhesion.
Integrin-mediated Rho GTPase signaling. Integrin associate with RTKs to mediate Rho GTPase subfamilies (RhoA, Rac and Cdc42) signaling via FAK-Src (integrin/RTKs) or ILK (integrin). Activation Rho GTPase signaling controls extension of lamellipodia and filopodia to form focal adhesions and actin stress fibers. Integrin association with the actin cytoskeleton by Rho GTPase is important for tumorigenesis and migration
Integrin-mediated TGFβ signaling
Transforming growth factor beta (TGFβ) plays crucial homeostatic roles in the pathogenesis of inflammation and fibrosis [78]. There are three isomers of TGFβ (TGFβ1, TGFβ2 and TGFβ3) and high homologous and bind to the same TGFβ receptor (type I and type II). Growing evidence indicates that TGFβ spatiotemporal activation controlled by integrin has emerged as an important mechanism during a series of pathophysiological processes, including immunity, inflammation and fibrosis [19, 78]. Integrin αvβ6 is exclusively expressed in epithelial cells as a fibronectin receptor. Integrin αvβ6 perceives the surrounding signals and subsequently activates TGFβ in its LAPs. In normal epithelial cells, the crosstalk between integrin αvβ6 and TGFβ is critical for maintaining the tooth amelogenesis and periodontal health, hair follicle stem cell quiescence and suppression of intestinal epithelial cell inflammation to form epithelial barrier. On the other hand, in invasive cancers, the initiation of integrin αvβ6-mediated TGFβ signaling is associated with aggressive cancer and poor patient survival [79]. Other integrin-TGFβ complexes, besides integrin αvβ6, also exert a dual role that changes depending on the stage of cancer progression. In the early stage, integrin-TGFβ complex plays a cancer-suppressing role via activation of the anti-proliferative cytokines [79], while it acts a cancer promoter in the advanced stage of epithelial cancers [80].
During the wound healing of oral mucosa and skin epidermis, integrin αvβ6-TGFβ crosstalk is specifically increased. It is consistent with basement membrane regeneration, granulation formation and connective tissue remodeling [79]. During the development and genesis of fibrosis, integrin αvβ6-TGFβ complex drives fibrosis via Smad-dependent or non-Smad signaling, exerted by kinases and Rho GTPases [19]. These two different signaling pathways are crucial for the severity of fibrosis. However, not all TGFβ-mediated fibrosis is associated with integrin αvβ6. There is growing evidence that blocking integrin αvβ1, α3β1, αvβ3, αvβ5 and αvβ8 can be effective in the context of fibrosis. It works by controlling the differentiation of fibroblasts into myofibroblasts or the process of EMT in pulmonary fibrosis diseases [80]. In the immune system, integrin αvβ6 and αvβ8 are pivotal regulators to release TGFβ for either encouraging or inhibiting immune responses. Activated TGFβ triggers canonical (Smads) and non-canonical (Rho GTPase and MAPKs) signaling pathways to induce EMT and malignant progression in cancer (Fig. 5). Futhermore, modulation of activated TGFβ not only hampers the function of innate immune cells, but also exerts control over the recruitment, retention, and activation of immune cells, potentially culminating in the onset of severe autoimmunity [81].
Integrin-mediated mechanical cues: Hippo signaling and Wnt/β-catenin signaling
As a stiffness-sensor molecule, integrin not only acts as the physical scaffold between the extracellular stiffness and intracellular actin cytoskeleton, but also transduces the extracellular stimulation into cell to mediate mechanotransduction [82]. Several specific integrin subtypes are extremely involved in mechanical cues-mediated Hippo and Wnt/β-catenin signaling activation.
(1) Integrin-mediated Hippo signalling
The Hippo signaling is first discovered in Drosophila. Due to the high conservation of the Hippo pathway in mammals, an analogous Hippo core kinase cascade exists which consists of Mst1 and Mst2 (Hpo homolog), WW45 or Sav1 (Sav homolog), Last1 and Lats2 (Wts homolog) and Mob1 (Mats homolog). The kinase cascade phosphorylates YAP (Yki homolog) and leads to YAP/TAZ co-activator inactivation and this inactivation inhibits its nuclear-cytoplasmic translocation [83]. Several reports have demonstrated that YAP/TAZ is an overarching regulator for stretching forces, epithelial sheet shape and surrounding ECM stiffness in multicellular environment. For instance, integrin β5 regulates ECM-mediated macrophage polarization via FAK-ERK1/2 pathway [84]. Integrin β1-Src complex interacts with the basal extracellular stiffness in skin cancer, and induces YAP/TAZ nuclear translocation in basal layer cells. Integrin β1 skin-conditional knockout phenotype is similar to YAP/TAZ skin-specific loss [85]. During the vascular remodeling, thrombospondin-1 (Thbs1) responds to the cyclic stretch and acts as a matrix sensor to induce YAP nuclear location via activating integrin αvβ1-mediated Rap2-dependent manner [86]. In Ewing sarcoma, EWS-FLI1-mediated tenascin-C promotes progression through integrin α5β1-induced YAP activation [87]. In gastric cancer, annexin A6 in extracellular vesicles from cancer-associated fibroblasts induces drug resistance through integrin β1-FAK-YAP signaling [88]. In atherosclerosis, integrin β3 directly senses the shear forces followed by activating YAP/TAZ-JNK cascade to mediate atheroprotective effect [89]. In the epidermal squamous cell carcinoma cancer stem cells, transglutaminase2 (TG2) controls ΔNp63α and interacts with integrin α6β4. The axis subsequently regulates FAK/Src and PI3K/PDK1 signaling which increases YAP and ΔNp63α feedback accumulation [47]. In colon cancer, ILK suppresses Hippo signaling by inhibiting its upstream molecule Merlin via regulation MYPT1-PP1 (a myosin light-chain phosphatase) [90]. In addition, Rho GTPases, particularly RhoA contributes to YAP/TAZ dephosphorylation via regulating actin cytoskeleton dynamics, Notably, this process might occur independently of the Hippo core kinase cascade (Fig. 6).
Integrin-mediated Hippo signaling. Integrin senses the extracellular mechanical cues to regulate the on/off switch of Hippo signaling. The Hippo core kinase cascade may be dispensable in the process of YAP/TAZ nuclear translocation by integrin activation. However, Rho GTPases, particularly RhoA contributes to YAP/TAZ dephosphorylation via regulating actin cytoskeleton dynamics
(2) Integrin-mediated Wnt/β-catenin signalling
Wnt is an evolutionarily conserved signaling pathway involved in embryo development, tissue homeostasis and a variety of common diseases [91]. Wnt could be divided into non-canonical and canonical pathways. The canonical Wnt activation requires the cell membrane receptors binding to extracellular Wnt ligands, such as ECM, which immediately induces β-catenin nuclear translocation, known as Wnt/β-catenin signaling [92]. Integrin induces mechanotransduction through sensing shear stress, strain and ECM stiffness from extracellular cues. The extracellular mechanical cues play a crucial role in Wnt/β-catenin signaling activation during mechanosensing-mediate diseases [93]. The combination of integrin and Wnt signaling triggers phosphatidylinositol 3 kinase (PI3K) activation, subsequently this activations regulate glycogen synthase kinase 3β (GSK3β), ubiquitin (Ub)-induced β-catenin degradation, nuclear location and Wnt targeting gene expression (Fig. 7). Previous reports have indicated that integrin β1-dependent Wnt/β-catenin pathway participates in osteoblast differentiation, osteoblastogenesis and ostegenetic cell repair [93]. And this integrin β1/Wnt axis is also regarded as a potential therapeutic target in several cancers, such as colorectal and ovarian cancer [94, 95]. ECM-stimulating activated integrin β5 promotes hepatocellular carcinoma tumorigenesis through upregulating β-catenin level [96]. In TNBC, nanoparticles targeted integrin α5 inhibits β-catenin level to suppress tumor cell stemness and metastasis [97]. Notably, nanoparticles can also deliver integrin α9 to induce similar regulatory mechanism in TNBC [98]. ILK, as a mediator of cell–matrix interaction, is also associated with Wnt/β-catenin signaling in vascular smooth muscle cells as well as endothelial cell-related diseases [99].
Integrin-mediated metabolism in cancer
Cellular metabolism under both physiological and malignant condition is extremely associated with extracellular matrix remodeling and integrin traffic, especially in epithelial and endothelial cells with fibroblasts [100, 101]. Cancer cells directly acquire nutrients from surrounding microenvironment via integrin-induced endocytosis of ECM [102]. Furthermore, cancer cells secrete exosomes to regulate normal cell metabolism and angiogenesis, which is important in biological functions of immune and cancer cells. The metabolic crosstalk, in turn, induces ECM remodeling and vascular barrier impairment [53]. Integrin regulates mTOR signaling by activating PI3K/AKT or taking part of amino acid transport. mTOR activation by integrin directly regulates aerobic glycolysis, glycolysis or influences HIF-1α to induce mitochondrial respiration. Besides, HIF-1α, metabolic stress or the AMPK-activating kinase (LKB1) also increase AMPK activity to promote glycolysis and regulate FAK activation (Fig. 8). In TNBC, integrin β4 is in a persistently high expression state and this mediates the metabolic reprogramming of CAFs. The integrin β4, via exosomes, initiates BNIP3L-dependent mitophagy and lactic acid accumulation-induced glucose metabolism [103]. In prostate cancer, exosomes transmitted by integrin αvβ3 [104] and integrin αvβ6 [105] arouse cancer cell aggression. In neutrophil-mediated inflammatory response, integrin β2 participates in the adhesion process between exosomes from neutrophils and endothelium [106]. There is also a report indicating that in neutrophil, integrin β2-dependent adhesion modulates cell energy metabolism through a small GTPase Arf6-induced pathway [107]. Integrin α4β1 is considered as a biomarker of the malignant transformation during the hematopoietic stem cell-renewal and differentiation [108]. During the atherosclerosis and cancer in small intestine, integrin β7 expressed in the gut intraepithelial T cells controls inflammation and metabolism [109]. In cervical cancer, high expression of PDL1 promotes glycolysis and lymph node metastasis through integrin β4-SNAL1/SIRT3 signaling pathway [110]. Chemoresistance, such as cisplatin resistance, is exaggerated when integrin β4 binds to paxillin (PXN) and FAK to form a focal adhesion complex in lung cancer. This complex increases Bcl-2 expression and dynamin-related protein 1 (DRP1) to modulate cisplatin resistance [111].
Potential therapeutic target of integrins
In clinic, integrin is considered as a double-edged sword. On the one hand, integrin signaling activation promotes the resistance of chemotherapy and radiotherapy. However, since integrin is transmembrane protein sensitive to pharmacological treatment, disease therapeutic strategies targeting integrin may achieve considerable clinical success. Three classes of drug can target integrin: monoclonal antibodies, peptides and small molecule inhibitors [112]. To date, only few integrin targeted drugs including abciximab, eptifibatide, tirofiban, natalizumab, vedolizumab, lifitegrast (SAR-1118) and carotegrast (AJM-300) have been successfully marketed. These drugs mainly target αIIbβ3, αVβ3, α4β1, α4β7 and αLβ2 [113]. Recently, a comprehensive overview of integrin-targeting drugs undergoing clinical trials is presented in Table 1. Among these candidates, roughly twenty-five percent are currently in Phase I trials, while approximately sixteen percent have progressed to Phase III. Concurrently, some of the drugs are in the process of patient recruitment, while others have encountered challenges in advancing through the trial stages. Compared with monoclonal antibodies and peptides, small molecule inhibitors constitute the predominant portion of the ongoing clinical trials, due to their cost-effectiveness, safety profile, pharmacokinetic characteristics, and route of administration.
Targeting integrins beyond the ligand binding site (specifically the allosteric site), holds the potential to hinder integrin activation by either obstructing the orthosteric site or by maintaining the conformation in a low-affinity state. Within this context, monoclonal antibodies have been categorized into three distinct groups: inhibitory antibodies, activation-specific antibodies, and non-functional antibodies. The primary function of monoclonal antibodies lies in their capacity as competitive inhibitors, with a significant portion exerting allosteric inhibitory effects. These antibodies exhibit specificity for discrete regions within the integrin ectodomain, selectively targeting specific subunits or conformations.
Integrin αIIbβ3 is specifically expressed in platelets and megakaryocytes, aggravating cardiovascular and autoimmune diseases. Integrin αIIbβ3 antagonist, Abciximab, is the first drug used in thrombosis-associated disease. Due to the remarkable efficacy of abciximab, eptifibatide and tirofiban are developed immediately afterward [149, 150]. They inhibit platelet aggregation via binding to fibrinogen and other ligands that induce integrin αIIbβ3 activation in angiogenesis related disease [112, 150]. Talin, kindlin and other relatively uncommon proteins, such as ILK, β3 endonexin and vinculin are involved in integrin αIIbβ3 activation [151]. While other proteins including calcium and integrin binding protein 1 (CIB1) [152], docking protein 1 (Dok1) [153] and filamin [154] mediate integrin αIIbβ3 inactivation via binging either integrin αIIb or integrin β3 tail in the cytoplasmic region.
Integrin αVβ3 is mainly expressed in vascular smooth muscle cells and macrophages. Integrin αVβ3 mediates cell-dependent inflammatory angiogenesis, which is essential for the pathology rheumatoid arthritis and related arthropathies. Emerging evidence suggests that as an RGD-binding subfamily, integrin αVβ3 is also associated with ophthalmology and osteoporosis [150]. Vitaxin, a monoclonal antibody used in treatment of rheumatoid arthritis, antagonizes integrin αVβ3 by binding both integrin αV and integrin β3 tails. This disrupts the crosstalk of integrin αVβ3 and osteopontin and vitronectin. Integrin αV and β3 alone cannot be recognized by vitaxin [155].
Integrin α4β1, α4β7 and αLβ2 are leukocyte-specific protein, which contribute to immune response in inflammatory bowel disease (IBD), multiple sclerosis (MS) and dry eye disease (DED). Integrin α4β1 binding to VCAM-1 controls leukocyte diapedesis [156]. In addition, a fraction of integrin α4β1, expressed in endothelium, is also required to induce immune cell adhesion via VACM1, which plays a potential role in the infiltration of immune cells into central nervous system [150]. Integrin α4β7 is a leukocyte gut-homing receptor that binds mucosal vascular address in CAM1 to induce T-cell homing [157]. Integrin αLβ2 and α4β1 modulate T cell activation and adhesion to facilitate T cells infiltration into tumor cells [150]. Natalizumab is an extensive integrin α4 subtype inhibitor used in MS [158] and Crohn’s disease [159]. However, its limitation, including tendency to promote progressive multifocal leukoencephalopathy, and poor drug delivery system, makes it necessary to develop the gentler and more effective drugs [150]. Currently, a specific integrin α4β7 inhibitor, vedolizumab delivered subcutaneously has already been approved for clinical use in IBD, such as Crohn’s disease [160]. Meanwhile, drugs targeting integrin β7, like etrolizumab, is already in clinical study and is only effective against integrin β7, which has no effect on integrin α4β7. This specific feature likely provides unexpected benefits [157]. Another integrin antagonist, lifitegrast, prevents lymphocyte adhesion resulting in hindering T cell activation, releasing inflammatory factors and subsequently reducing T cell-induced inflammation by blocking the interaction of integrin αLβ2 and ICAM-1 in DED [161].
Integrin-ECM interaction triggers cell adhesion-induced drug resistance to chemotherapy, radiotherapy and targeted therapy. This unexpected drug resistance is due to the change of drug targets, substitutability of anti-apoptotic events and invalidation of cell death, especially in the treatment of cancer [43]. In breast cancer, integrin α6 mediates tamoxifen resistance via integrin α6/Src/AKT signaling [162]. Integrin β1 participates in lapatinib and transtuzumab resistance by activating human epidermal growth factor receptor 2 (HER2) and PI3K pathway which promotes breast cancer progression [163, 164]. In addition, the doxorubicin resistance is caused by the interaction between integrin β1 and galectin 1 [165]; and the cisplatin resistance is also triggered by integrin β1 recruitment and functioning [166]. In lung cancer, integrin β4/PXN/FAK complex mediates cisplatin resistance through regulating ubiquitin specific peptidase 1 (USP1) and voltage-dependent anion channel 1 (VDAC1), which are associated with mitochondrial function and maintaining genomic stability [111]. Integrin β1 also induces erlotinib resistance by regulating the canonical Src signaling. The phenomena indicates that integrin β1 plays an important role in the drug resistance to EGFR-targeted strategy [43].
At present, there are emerging clinic trials focusing on integrin-related nanoparticles for DNA/RNA therapeutics, expected to bring detection and therapeutic promise in the biomedical fields. Integrin α4β7 binding to VCAM-1 and MadCAM-1 induces homing to different tissues. MadCAM-1 is crucial for leukocytes adhesion to intestinal endothelium [167]. A recent study shows that intestinal endothelium generates a recombinant protein containing two domains of MadCAM-1 which has great affinity for integrin α4β7. This study silences interferon γ via lipid nanoparticles targeting the integrin α4β7-MadCAM-1 high affinity conformation and achieves an exciting treatment effect in the experimental colitis [168]. Increasing studies have indicated that nanoparticles carrier RGD peptide efficiently overcome the barriers of DNA transit to target cells. The complex containing high RGD content exerts huge therapeutic effect. Surprisingly, in uterine leiomyoma cells, peptide-based nanoparticles for integrin αvβ3 targeted DNA delivery expand ganciclovir treatment induced cell death [169]. In colorectal carcinoma cells, nanoparticles carrier RGD peptide as well as derivatives of PLGA-tetrac targeting integrin αvβ3 contribute to efficiency of resveratrol treatment on cancer growth and metastasis [170]. These non-viral vehicles-based chemotherapeutic agents combined treatment is of high potential in the clinical translation. Additionally, in PET/CT imaging and photothermal ablation therapy, targeting integrin αvβ3 by copper sulfide nanoparticles carrier RGD peptide effectively improves the side effects associated with the route of administration [171]. Meanwhile, other assembled forms of integrin targeting drugs are also tried in the clinic and preclinical studies, example silica loaded monoclonal antibodies against integrin α2β1 nanoparticles used in macropinocytosis-like mechanism [172], lip ECO-based nanoparticles delivering integrin β3 siRNA used in TNBC [173] and so on.
Natural product compounds targeting integrin and its regulatory components are also proposed as high efficiency, low toxicity and fewer side effects strategy for therapy. Curcumin is a traditional herbal medicine derived from Curcuma longa. Curcumin has been verified as an anti-inflammatory, antiproliferative, antioxidant and antitumor agent used in clinics [174]. In non-small cell lung cancer (NSCLC) and a series of fibrosis-related diseases, curcumin mainly acts on the integrin β1 pathway to suppress proliferation and malignancy [175, 176]. Curcuma also targets integrin α6β4 by regulating AKT/ENPP2 signaling to suppress migration and invasion in breast cancer [177]. Curcumin in combination with resveratrol has been reported to exert an antiangiogenic effect via reducing integrin β3 expression and the inhibitory effect is amplified by the combined form [178]. In addition, resveratrol targeting integrin β1 plays a crucial anti-proliferation and anti-invasion roles in the colorectal cancer microenvironment [179, 180]. An integrin β1/mTOR axis is required for fibroblast differentiation in corneal blindness [181]. Phloretin, a natural product found in apples and strawberries [182], suppresses integrin αvβ3/Src signaling to regulate the actin cytoskeleton during the invasion process of osteosarcoma [183]. Meanwhile, phloretin has been considered as a glucose transporter inhibitor that induces cell death in osteosarcoma cells. It suggests that phloretin treatment enhances the apoptotic sensitivity and cytotoxic effect of chemotherapeutic drugs via mediating integrin αvβ3 and MAPK pathway [184]. Phloretin targeting integrin β3 is also involved in the interaction between leukocyte and endothelial triggered by thrombin during thrombosis and atherosclerosis [185]. Ouabain is the major ingredient of Strophanthus gratus seeds. Ouabain-induced neutrophil migration inhibition is associated with an integrin β2 chain molecule of CD18 and chemokine receptor CXCR2 inflammatory response [186]. Artemisinin, a traditional medicine, is a sesquiterpene lactone compound extracted from Artemisia that acts mainly against malaria. Additionally, artemisinin acts as an antitumor, anti-angiogenic and pro-apoptotic function [187]. Several studies have demonstrated that artemisinin inhibits integrin β3 as well as other receptor-coupled signalings, such as interleukin 1, tumor necrosis factor α, and toll-like receptors in inflammatory and autoimmune diseases, osteoclastogenesis and melanoma [188, 189].
Besides, approximately 260 other drugs targeting various integrin subtypes have already been studied preclinically in academic as well as industry clinical trials [160]. Focusing on integrin in combination with other radiation therapy and chemotherapy treatment strategies may enhance drug sensitivity rather than single-agent treatment. Research on drugs targeting integrin holds promise for treating related diseases.
Conclusions and future perspectives
Integrin, as a crucial transmembrane receptor, with ‘outside-in’ ligand-binding specificities and ‘inside-in’ signaling properties is not yet fully explored. Abnormal activation of the ‘outside-in’ and the ‘inside-out’ bidirectional signaling machinery of integrin is closely associated with common diseases, such as cancer, chronic inflammation and thrombosis. Although studies on integrin are rapidly expanding, given the number of various integrin subtypes and the complexity of integrin-mediating signals, investigation of how integrin traffics and functions in the development and the course of diseases is still rewarding.
It is already confirmed that the recruitment of talin and probably kindlin are essential for integrin binding to various ligands and activation, which establishes its mechanical sensitivity. Talin and kindlin induce integrin-ligand complex via binding the tail of integrin β, thereby disrupting the α/β ectodomain. Noteworthy, mechanical forces play a similar indispensable role in integrin regulating adhesion-related mechanotransduction. While at this point, talin and kindlin may not cause integrin heterodimer disruption-induced inside signaling but deliver mechanical forces to the integrin-ligand complex. However, whether kindlin plays a similarly crucial role to talin or exerts an extra potential role in integrin stabilization is still unclear and worth to be explored sequentially. When bound to ligands, integrin aggregates and subsequently participates in the actin skeleton network. The integrin-actin axis regulates several intracellular signalings, such as FAK/Src, Rho GTPase, Ras-ERK and Hippo pathway, as well as cellular behaviors, such as proliferation, migration, invasion, apoptosis, survival and cell stemness.
Although numerous studies on integrin have been conducted clinically, however, the approved drugs and therapies are unsatisfactory. Currently, monoclonal antibodies, peptides and small molecules are applied in clinical trials, however, most of the target is on the integrin-ligand binding site or the ligand itself which brings little benefit. Hence, paying more attention to the crosstalk between integrin and other pathways is valuable and will bring unexpected novel opportunities for therapeutics. While the crosstalk between the integrative function of integrins and other pathways, such as TGFβ, Hippo-YAP/TAZ, Wnt-β/catenin and metabolism-related signaling, is initially obscure and complicated as the study progressed, it has become exquisitely clearer. Integrin-dependent regulation of TGFβ is involved in IBD and other immune responses, which could be considered as a therapeutic target. Vedolizumab, as an integrin α4β7 inhibitor, has been used in IBD therapy in clinics. It indicates that integrin-TGFβ signaling will bring increased therapeutic effects. YAP/TAZ nuclear translocation is controlled by integrin β1-Src signaling in skin cancer, basal layer cells as well as other cancer-associated fibroblasts. Dasatinib, as an Src inhibitor, retains YAP/TAZ in the cytoplasm by suppressing integrin β1-Src signaling. Nanoparticles targeting integrin α5 become a novel and effective strategy in metastatic TNBC via modulating Wnt-β/catenin signaling. Besides the synthetic drugs, natural plant compounds as well as nanoparticle-based delivery and RNA-interfered technology have already been used in clinics and enrolled in clinical trials probably due to their high efficiency, low toxicity and fewer side effects.
The tumor microenvironment has been the leitmotif during the research on various cancers. Integrin-mediated mechanosensitivity is unignorable for mechanical cues transmission. In the future, studies on integrin should pay attention to the dependency of integrin and the translational biomarkers to ensure clinical efficacy. To be successful, we should take advantage of genetically engineered models to develop effective diagnostic and therapeutic technologies.
Availability of data and materials
Not applicable.
References
Cooper J, Giancotti FG. Integrin signaling in cancer: mechanotransduction, stemness, epithelial plasticity, and therapeutic resistance. Cancer Cell. 2019;35(3):347–67.
Moreno-Layseca P, et al. Integrin trafficking in cells and tissues. Nat Cell Biol. 2019;21(2):122–32.
Humphries MJ, et al. Integrin structure: heady advances in ligand binding, but activation still makes the knees wobble. Trends Biochem Sci. 2003;28(6):313–20.
Campbell ID, Humphries MJ. Integrin structure, activation, and interactions. Cold Spring Harb Perspect Biol. 2011;3(3):a004994.
Staunton DE, et al. Targeting integrin structure and function in disease. Adv Immunol. 2006;91:111–57.
Sun Z, Costell M, Fassler R. Integrin activation by talin, kindlin and mechanical forces. Nat Cell Biol. 2019;21(1):25–31.
Xiao T, et al. Structural basis for allostery in integrins and binding to fibrinogen-mimetic therapeutics. Nature. 2004;432(7013):59–67.
Xiong JP, et al. Crystal structure of the extracellular segment of integrin alpha Vbeta3 in complex with an Arg-Gly-Asp ligand. Science. 2002;296(5565):151–5.
Emsley J, et al. Structural basis of collagen recognition by integrin alpha2beta1. Cell. 2000;101(1):47–56.
Bökel C, Brown NH. Integrins in development: moving on, responding to, and sticking to the extracellular matrix. Dev Cell. 2002;3(3):311–21.
Meighan CM, Schwarzbauer JE. Temporal and spatial regulation of integrins during development. Curr Opin Cell Biol. 2008;20(5):520–4.
Guo W, Giancotti FG. Integrin signalling during tumour progression. Nat Rev Mol Cell Biol. 2004;5(10):816–26.
Koch S, Claesson-Welsh L. Signal transduction by vascular endothelial growth factor receptors. Cold Spring Harb Perspect Med. 2012;2(7): a006502.
Casali BC, et al. The effects of alphavbeta3 integrin blockage in breast tumor and endothelial cells under Hypoxia in vitro. Int J Mol Sci. 2022;23(3):1745.
Elbediwy A, et al. Integrin signalling regulates YAP and TAZ to control skin homeostasis. Development. 2016;143(10):1674–87.
Thomas JR, Paul NR, Morgan MR. Adhesion and growth factor receptor crosstalk mechanisms controlling cell migration. Essays Biochem. 2019;63(5):553–67.
Gringhuis SI, et al. Fungal sensing by dectin-1 directs the non-pathogenic polarization of T(H)17 cells through balanced type I IFN responses in human DCs. Nat Immunol. 2022;23(12):1735–48.
Gimeno-Lluch I, et al. The role of the fibronectin synergy site for skin wound healing. Cells. 2022;11(13):2100.
Nolte M, Margadant C. Controlling immunity and inflammation through integrin-dependent regulation of TGF-beta. Trends Cell Biol. 2020;30(1):49–59.
Schnittert J, et al. Integrins in wound healing, fibrosis and tumor stroma: High potential targets for therapeutics and drug delivery. Adv Drug Deliv Rev. 2018;129:37–53.
Liu S, et al. Expression of integrin beta1 by fibroblasts is required for tissue repair in vivo. J Cell Sci. 2010;123(Pt 21):3674–82.
Reed NI, et al. The alphavbeta1 integrin plays a critical in vivo role in tissue fibrosis. Sci Transl Med. 2015;7(288):288ra79.
Koivisto L, et al. Integrins in wound healing. Adv Wound Care (New Rochelle). 2014;3(12):762–83.
Russo JD, et al. Integrin α5β1 nano- presentation regulates collective keratinocyte migration independent of substrate rigidity. Elife. 2021;10: e69861.
Schulz JN, et al. Reduced granulation tissue and wound strength in the absence of a11b1Integrin. J Invest Dermatol. 2015;135(5):1435–44.
Lin SN, et al. Human intestinal myofibroblasts deposited collagen VI enhances adhesiveness for T cells - A novel mechanism for maintenance of intestinal inflammation. Matrix Biol. 2022;113:1–21.
Busenhart P, et al. Inhibition of integrin alphavbeta6 sparks T-cell antitumor response and enhances immune checkpoint blockade therapy in colorectal cancer. J Immunother Cancer. 2022;10(2):e003465.
Laine A, et al. Regulatory T cells promote cancer immune-escape through integrin alphavbeta8-mediated TGF-beta activation. Nat Commun. 2021;12(1):6228.
Deng C, et al. Targeted apoptosis of macrophages and osteoclasts in arthritic joints is effective against advanced inflammatory arthritis. Nature Commun. 2021;12(1):2174.
Wang Q, et al. Dysregulated integrin alphaVbeta3 and CD47 signaling promotes joint inflammation, cartilage breakdown, and progression of osteoarthritis. JCI Insight. 2019;4(18):e128616.
Terry SYA, et al. Monitoring therapy response of experimental arthritis with radiolabeled tracers targeting fibroblasts, macrophages, or integrin αvβ3. J Nucl Med. 2016;57(3):467–72.
Wilder RL. Integrin alpha V beta 3 as a target for treatment of rheumatoid arthritis and related rheumatic diseases. Ann Rheum Dis. 2002;2(Suppl 2):ii96-99.
Henderson NC, Sheppard D. Integrin-mediated regulation of TGFbeta in fibrosis. Biochim Biophys Acta. 2013;1832(7):891–6.
Berghoff AS, et al. alphavbeta3, alphavbeta5 and alphavbeta6 integrins in brain metastases of lung cancer. Clin Exp Metastasis. 2014;31(7):841–51.
Madala SK, et al. Inhibition of the alphavbeta6 integrin leads to limited alteration of TGF-alpha-induced pulmonary fibrosis. Am J Physiol Lung Cell Mol Physiol. 2014;306(8):L726-735.
Minagawa S, et al. Selective targeting of TGF-β activation to treat fibroinflammatory airway disease. Sci Transl Med. 2014;6(241):241ra79.
Nejjari M, et al. Integrin up-regulation in chronic liver disease: relationship with inflammation and fibrosis in chronic hepatitis C. J Pathol. 2001;195(4):473–81.
Kuwashiro T, et al. Regulation of interferon signaling and HCV-RNA replication by extracellular matrix. Int J Mol Med. 2018;42(2):957–65.
Popov Y, et al. Integrin alphavbeta6 is a marker of the progression of biliary and portal liver fibrosis and a novel target for antifibrotic therapies. J Hepatol. 2008;48(3):453–64.
Han R, et al. C3a and suPAR drive versican V1 expression in tubular cells of focal segmental glomerulosclerosis. JCI Insight. 2019;4(7):e122912.
Zheng G, et al. alpha3 integrin of cell-cell contact mediates kidney fibrosis by integrin-linked kinase in proximal tubular E-cadherin deficient mice. Am J Pathol. 2016;186(7):1847–60.
Bansal R, et al. Integrin alpha 11 in the regulation of the myofibroblast phenotype: implications for fibrotic diseases. Exp Mol Med. 2017;49(11): e396.
Seguin L, et al. Integrins and cancer: regulators of cancer stemness, metastasis, and drug resistance. Trends Cell Biol. 2015;25(4):234–40.
Xiong J, et al. Integrins regulate stemness in solid tumor: an emerging therapeutic target. J Hematol Oncol. 2021;14(1):177.
Beaulieu JF. Integrin alpha6beta4 in colorectal cancer: expression, regulation, functional alterations and use as a biomarker. Cancers (Basel). 2019;12(1):41.
Parker KA, et al. lncRNA BORG:TRIM28 complexes drive metastatic progression by inducing alpha6 integrin/CD49f expression in breast cancer stem cells. Mol Cancer Res. 2021;19(12):2068–80.
Fisher ML, et al. Transglutaminase interaction with α6/β4-integrin stimulates YAP1-dependent ΔNp63α stabilization and leads to enhanced cancer stem cell survival and tumor formation. Cancer Res. 2016;76(24):7265–76.
Kowalski-Chauvel A, et al. Alpha6-Integrin regulates FGFR1 expression through the ZEB1/YAP1 transcription complex in glioblastoma stem cells resulting in enhanced proliferation and stemness. Cancers (Basel). 2019;11(3):406.
Stanzani E, et al. Dual role of integrin alpha-6 in glioblastoma: supporting stemness in proneural stem-like cells while inducing radioresistance in mesenchymal stem-like cells. Cancers (Basel). 2021;13(12):3055.
Fernandez-Zapico ME. GLI1 finds a new role in cancer stem cell biology. EMBO Mol Med. 2013;5(4):483–5.
Zheng Y, et al. A rare population of CD24(+)ITGB4(+)Notch(hi) cells drives tumor propagation in NSCLC and requires Notch3 for self-renewal. Cancer Cell. 2013;24(1):59–74.
Xiong J, Balcioglu HE, Danen EH. Integrin signaling in control of tumor growth and progression. Int J Biochem Cell Biol. 2013;45(5):1012–5.
Soe ZY, Park EJ, Shimaoka M. Integrin regulation in immunological and cancerous cells and exosomes. Int J Mol Sci. 2021;22(4):2139.
Guo W, et al. β4 integrin amplifies ErbB2 signaling to promote mammary tumorigenesis. Cell. 2006;126(3):489–502.
Sturm D, et al. Paediatric and adult glioblastoma: multiform (epi)genomic culprits emerge. Nat Rev Cancer. 2014;14(2):92–107.
Rys JP, et al. Discrete spatial organization of TGFbeta receptors couples receptor multimerization and signaling to cellular tension. Elife. 2015;4: e09300.
Hannigan G, Troussard AA, Dedhar S. Integrin-linked kinase: a cancer therapeutic target unique among its ILK. Nat Rev Cancer. 2005;5(1):51–63.
Sternlicht MD, Werb Z. How matrix metalloproteinases regulate cell behavior. Annu Rev Cell Dev Biol. 2001;17:463–516.
Lawson CD, Burridge K. The on-off relationship of Rho and Rac during integrin-mediated adhesion and cell migration. Small GTPases. 2014;5: e27958.
Parise LV, Lee J, Juliano RL. New aspects of integrin signaling in cancer. Semin Cancer Biol. 2000;10(6):407–14.
Tu Y, et al. A new focal adhesion protein that interacts with integrin-linked kinase and regulates cell adhesion and spreading. J Cell Biol. 2001;153(3):585–98.
Duro-Castano A, et al. Modulating angiogenesis with integrin-targeted nanomedicines. Adv Drug Deliv Rev. 2017;119:101–19.
Desgrosellier JS, Cheresh DA. Integrins in cancer: biological implications and therapeutic opportunities. Nat Rev Cancer. 2010;10(1):9–22.
Yousefi H, et al. Understanding the role of integrins in breast cancer invasion, metastasis, angiogenesis, and drug resistance. Oncogene. 2021;40(6):1043–63.
Bonin F, et al. Kindlin-1 drives early steps of breast cancer metastasis. Cancer Commun (Lond). 2022;42(10):1036–40.
Schlesinger M, Bendas G. Vascular cell adhesion molecule-1 (VCAM-1)–an increasing insight into its role in tumorigenicity and metastasis. Int J Cancer. 2015;136(11):2504–14.
Sokeland G, Schumacher U. The functional role of integrins during intra- and extravasation within the metastatic cascade. Mol Cancer. 2019;18(1):12.
Alanko J, Ivaska J. Endosomes: emerging platforms for integrin-mediated FAK signalling. Trends Cell Biol. 2016;26(6):391–8.
Rigiracciolo DC, et al. Focal adhesion kinase fine tunes multifaced signals toward breast cancer progression. Cancers (Basel). 2021;13(4):645.
Crosas-Molist E, et al. Rho GTPase signaling in cancer progression and dissemination. Physiol Rev. 2022;102(1):455–510.
Lee BY, et al. FAK signaling in human cancer as a target for therapeutics. Pharmacol Ther. 2015;146:132–49.
Paszek MJ, et al. Tensional homeostasis and the malignant phenotype. Cancer Cell. 2005;8(3):241–54.
Zanconato F, et al. Genome-wide association between YAP/TAZ/TEAD and AP-1 at enhancers drives oncogenic growth. Nat Cell Biol. 2015;17(9):1218–27.
Gaggioli C, et al. Fibroblast-led collective invasion of carcinoma cells with differing roles for RhoGTPases in leading and following cells. Nat Cell Biol. 2007;9(12):1392–400.
Tod J, et al. Pro-migratory and TGF-β-activating functions of αvβ6 integrin in pancreatic cancer are differentially regulated via an Eps8-dependent GTPase switch. J Pathol. 2017;243(1):37–50.
Zhang Z, et al. Fibroblast-derived tenascin-C promotes Schwann cell migration through beta1-integrin dependent pathway during peripheral nerve regeneration. Glia. 2016;64(3):374–85.
Schwartz MA, Shattil SJ. Signaling networks linking integrins and rho family GTPases. Trends Biochem Sci. 2000;25(8):388–91.
Ong CH, et al. TGF-beta-induced fibrosis: A review on the underlying mechanism and potential therapeutic strategies. Eur J Pharmacol. 2021;911: 174510.
Koivisto L, et al. Integrin alphavbeta6: Structure, function and role in health and disease. Int J Biochem Cell Biol. 2018;99:186–96.
Margadant C, Sonnenberg A. Integrin-TGF-beta crosstalk in fibrosis, cancer and wound healing. EMBO Rep. 2010;11(2):97–105.
Aluwihare P, et al. Mice that lack activity of alphavbeta6- and alphavbeta8-integrins reproduce the abnormalities of Tgfb1- and Tgfb3-null mice. J Cell Sci. 2009;122(Pt 2):227–32.
Wang Y, et al. Matrix stiffness regulates macrophage polarization in atherosclerosis. Pharmacol Res. 2022;179: 106236.
Pan D. The hippo signaling pathway in development and cancer. Dev Cell. 2010;19(4):491–505.
Xing X, et al. Matrix stiffness-mediated effects on macrophages polarization and their LOXL2 expression. FEBS J. 2021;288(11):3465–77.
Elbediwy A, Thompson BJ. Evolution of mechanotransduction via YAP/TAZ in animal epithelia. Curr Opin Cell Biol. 2018;51:117–23.
Yamashiro Y, et al. Matrix mechanotransduction mediated by thrombospondin-1/integrin/YAP in the vascular remodeling. Proc Natl Acad Sci U S A. 2020;117(18):9896–905.
He S, et al. EWS-FLI1-mediated tenascin-C expression promotes tumour progression by targeting MALAT1 through integrin alpha5beta1-mediated YAP activation in Ewing sarcoma. Br J Cancer. 2019;121(11):922–33.
Uchihara T, et al. Extracellular vesicles from cancer-associated fibroblasts containing annexin A6 induces FAK-YAP activation by stabilizing beta1 integrin enhancing drug resistance. Cancer Res. 2020;80(16):3222–35.
Wang L, et al. Integrin-YAP/TAZ-JNK cascade mediates atheroprotective effect of unidirectional shear flow. Nature. 2016;540(7634):579–82.
van Soldt BJ, Cardoso WV. Hippo-Yap/Taz signaling: Complex network interactions and impact in epithelial cell behavior. Wiley Interdiscip Rev Dev Biol. 2020;9(3): e371.
Yu F, et al. Wnt/beta-catenin signaling in cancers and targeted therapies. Signal Transduct Target Ther. 2021;6(1):307.
Liu J, et al. Wnt/beta-catenin signalling: function, biological mechanisms, and therapeutic opportunities. Signal Transduct Target Ther. 2022;7(1):3.
Astudillo P. Extracellular matrix stiffness and Wnt/beta-catenin signaling in physiology and disease. Biochem Soc Trans. 2020;48(3):1187–98.
Zhao H, et al. Wnt signaling in colorectal cancer: pathogenic role and therapeutic target. Mol Cancer. 2022;21(1):144.
Condello S, et al. Tissue tranglutaminase regulates interactions between ovarian cancer stem cells and the tumor niche. Cancer Res. 2018;78(11):2990–3001.
Lin Z, et al. Integrin-beta5, a miR-185-targeted gene, promotes hepatocellular carcinoma tumorigenesis by regulating beta-catenin stability. J Exp Clin Cancer Res. 2018;37(1):17.
Li Y, et al. In vivo β-catenin attenuation by the integrin α5-targeting nano-delivery strategy suppresses triple negative breast cancer stemness and metastasis. Biomaterials. 2019;188:160–72.
Wang Z, et al. Integrin alpha9 depletion promotes beta-catenin degradation to suppress triple-negative breast cancer tumor growth and metastasis. Int J Cancer. 2019;145(10):2767–80.
Park H, et al. Integrin-linked kinase controls retinal angiogenesis and is linked to Wnt signaling and exudative vitreoretinopathy. Nat Commun. 2019;10(1):5243.
Ata R, Antonescu CN. Integrins and cell metabolism: an intimate relationship impacting cancer. Int J Mol Sci. 2017;18(1):189.
Nazemi M, Rainero E. Cross-talk between the tumor microenvironment, extracellular matrix, and cell metabolism in cancer. Front Oncol. 2020;10:239.
Mana G, Valdembri D, Serini G. Conformationally active integrin endocytosis and traffic: why, where, when and how? Biochem Soc Trans. 2020;48(1):83–93.
Sung JS, et al. ITGB4-mediated metabolic reprogramming of cancer-associated fibroblasts. Oncogene. 2020;39(3):664–76.
Krishn SR, et al. Prostate cancer sheds the alphavbeta3 integrin in vivo through exosomes. Matrix Biol. 2019;77:41–57.
Lu H, et al. Exosomal alphavbeta6 integrin is required for monocyte M2 polarization in prostate cancer. Matrix Biol. 2018;70:20–35.
Subramanian BC, et al. The LTB4-BLT1 axis regulates actomyosin and beta2-integrin dynamics during neutrophil extravasation. J Cell Biol. 2020;219(10):e201910215.
Gamara J, et al. Arf6 regulates energy metabolism in neutrophils. Free Radic Biol Med. 2021;172:550–61.
Ashok D, et al. Insights into bone marrow niche stability: an adhesion and metabolism route. Front Cell Dev Biol. 2021;9: 798604.
Mukherjee P, Chattopadhyay A, Fogelman AM. The role of the small intestine in modulating metabolism and inflammation in atherosclerosis and cancer. Curr Opin Lipidol. 2019;30(5):383–7.
Wang S, et al. Programmed death ligand 1 promotes lymph node metastasis and glucose metabolism in cervical cancer by activating integrin β4/SNAI1/SIRT3 signaling pathway. Oncogene. 2018;37(30):4164–80.
Mohanty A, et al. A non-genetic mechanism involving the integrin beta4/Paxillin axis contributes to chemoresistance in lung cancer. iScience. 2020;23(9):101496.
Ley K, et al. Integrin-based therapeutics: biological basis, clinical use and new drugs. Nat Rev Drug Discov. 2016;15(3):173–83.
Pang X, et al. Targeting integrin pathways: mechanisms and advances in therapy. Signal Transduct Target Ther. 2023;8(1):1.
Karpusas M, et al. Crystal structure of the a1b1 integrin I domain in complex with an antibody fab fragment. J Mol Biol. 2003;327(5):1031–41.
Breuer J, et al. VLA-2 blockade in vivo by vatelizumab induces CD4+FoxP3+ regulatory T cells. Int Immunol. 2019;31(6):407–12.
Nwagwu CD, et al. Convection-enhanced delivery of a first-in-class anti-beta1 integrin antibody for the treatment of high-grade glioma utilizing real-time imaging. Pharmaceutics. 2020;13(1):40.
Besse B, et al. Phase Ib safety and pharmacokinetic study of volociximab, an anti-alpha5beta1 integrin antibody, in combination with carboplatin and paclitaxel in advanced non-small-cell lung cancer. Ann Oncol. 2013;24(1):90–6.
Bell-McGuinn KM, et al. A phase II, single-arm study of the anti-alpha5beta1 integrin antibody volociximab as monotherapy in patients with platinum-resistant advanced epithelial ovarian or primary peritoneal cancer. Gynecol Oncol. 2011;121(2):273–9.
Hersey P, et al. A randomized phase 2 study of etaracizumab, a monoclonal antibody against integrin alpha(v)beta(3), + or - dacarbazine in patients with stage IV metastatic melanoma. Cancer. 2010;116(6):1526–34.
Chu FM, et al. A phase 1, multicenter, open-label study of the safety of two dose levels of a human monoclonal antibody to human alpha(v) integrins, intetumumab, in combination with docetaxel and prednisone in patients with castrate-resistant metastatic prostate cancer. Invest New Drugs. 2011;29(4):674–9.
Heidenreich A, et al. A randomized, double-blind, multicenter, phase 2 study of a human monoclonal antibody to human alphanu integrins (intetumumab) in combination with docetaxel and prednisone for the first-line treatment of patients with metastatic castration-resistant prostate cancer. Ann Oncol. 2013;24(2):329–36.
Elez E, et al. Abituzumab combined with cetuximab plus irinotecan versus cetuximab plus irinotecan alone for patients with KRAS wild-type metastatic colorectal cancer: the randomised phase I/II POSEIDON trial. Ann Oncol. 2015;26(1):132–40.
O’Day S, et al. A randomised, phase II study of intetumumab, an anti-alphav-integrin mAb, alone and with dacarbazine in stage IV melanoma. Br J Cancer. 2011;105(3):346–52.
Sandborn WJ, et al. Efficacy and safety of Abrilumab in a randomized, placebo-controlled trial for moderate-to-severe ulcerative colitis. Gastroenterology. 2019;156(4):946-957 e18.
Hibi T, et al. Efficacy and safety of abrilumab, an alpha4beta7 integrin inhibitor, in Japanese patients with moderate-to-severe ulcerative colitis: a phase II study. Intest Res. 2019;17(3):375–86.
Takeuchi T, et al. ASP5094, a humanized monoclonal antibody against integrin alpha-9, did not show efficacy in patients with rheumatoid arthritis refractory to methotrexate: results from a phase 2a, randomized, double-blind, placebo-controlled trial. Arthr Res Ther. 2020;22(1):252.
Feagan BG, et al. Treatment of ulcerative colitis with a humanized antibody to the a 4 b 7 integrin. N Engl J Med. 2005;352(24):2449–507.
Vermeire S, et al. Etrolizumab as induction therapy for ulcerative colitis: a randomised, controlled, phase 2 trial. Lancet. 2014;384(9940):309–18.
Gubatan J, et al. Anti-Integrins for the treatment of inflammatory bowel disease: current evidence and perspectives. Clin Exp Gastroenterol. 2021;14:333–42.
Danese S, et al. Etrolizumab versus infliximab for the treatment of moderately to severely active ulcerative colitis (GARDENIA): a randomised, double-blind, double-dummy, phase 3 study. Lancet Gastroenterol Hepatol. 2022;7(2):118–27.
Lima ESR, et al. Anti-angiogenic collagen IV-derived peptide target engagement with alpha(v)beta(3) and alpha(5)beta(1) in ocular neovascularization models. iScience. 2023;26(2):106078.
Sandborn WJ, et al. PTG-100, an oral alpha4beta7 antagonist peptide: preclinical development and phase 1 and 2a studies in ulcerative colitis. Gastroenterology. 2021;161(6):1853-1864 e10.
Stoeltzing O, et al. Inhibition of integrin alpha5beta1 function with a small peptide (ATN-161) plus continuous 5-FU infusion reduces colorectal liver metastases and improves survival in mice. Int J Cancer. 2003;104(4):496–503.
Beddingfield BJ, et al. The integrin binding peptide, ATN-161, as a novel therapy for SARS-CoV-2 infection. JACC Basic Transl Sci. 2021;6(1):1–8.
Stupp R, et al. Cilengitide combined with standard treatment for patients with newly diagnosed glioblastoma with methylated MGMT promoter (CENTRIC EORTC 26071–22072 study): a multicentre, randomised, open-label, phase 3 trial. Lancet Oncol. 2014;15(10):1100–8.
Rosenthal MA, et al. Evaluation of the safety, pharmacokinetics and treatment effects of an alpha(nu)beta(3) integrin inhibitor on bone turnover and disease activity in men with hormone-refractory prostate cancer and bone metastases. Asia Pac J Clin Oncol. 2010;6(1):42–8.
Hickman A, et al. LFA-1 activation enriches tumor-specific T cells in a cold tumor model and synergizes with CTLA-4 blockade. J Clin Invest. 2022;132(13):e154152.
Cirkel GA, et al. A dose escalating phase I study of GLPG0187, a broad spectrum integrin receptor antagonist, in adult patients with progressive high-grade glioma and other advanced solid malignancies. Invest New Drugs. 2016;34(2):184–92.
John AE, et al. Translational pharmacology of an inhaled small molecule αvβ6 integrin inhibitor for idiopathic pulmonary fibrosis. Nature Commun. 2020;11(1):4659.
Miller DH, et al. Firategrast for relapsing remitting multiple sclerosis: a phase 2, randomised, double-blind, placebo-controlled trial. Lancet Neurol. 2012;11(2):131–9.
Decaris ML, et al. Dual inhibition of alpha(v)beta(6) and alpha(v)beta(1) reduces fibrogenesis in lung tissue explants from patients with IPF. Respir Res. 2021;22(1):265.
Yang L, Venneti S, Nagrath D. Glutaminolysis: a hallmark of cancer metabolism. Annu Rev Biomed Eng. 2017;19:163–94.
Semba CP, et al. A phase 2 randomized, double-masked, placebo-controlled study of a novel integrin antagonist (SAR 1118) for the treatment of dry eye. Am J Ophthalmol. 2012;153(6):1050-60 e1.
Diamant Z, et al. Effect of a very late antigen-4 receptor antagonist on allergen-induced airway responses and inflammation in asthma. Clin Exp Allergy. 2005;35(8):1080–7.
Hijazi Y, et al. Evaluation of the effect of multiple-dose administration of R411, a dual alpha4beta1-alpha4beta7 integrin antagonist on the major CYP isoform activities in healthy volunteers. Eur J Clin Pharmacol. 2006;62(1):83–5.
Rikken S, et al. Prehospital treatment with zalunfiban (RUC-4) in patients with ST- elevation myocardial infarction undergoing primary percutaneous coronary intervention: Rationale and design of the CELEBRATE trial. Am Heart J. 2023;258:119–28.
Lucaciu LA, Seicean R, Seicean A. Small molecule drugs in the treatment of inflammatory bowel diseases: which one, when and why? - a systematic review. Eur J Gastroenterol Hepatol. 2020;32(6):669–77.
Nicholson NS, et al. Orbofiban: an orally active GPIIb/IIIa platelet receptor antagonist. Med Res Rev. 2001;21(3):211–6.
Bledzka K, Smyth SS, Plow EF. Integrin alphaIIbbeta3: from discovery to efficacious therapeutic target. Circ Res. 2013;112(8):1189–200.
Slack RJ, et al. Emerging therapeutic opportunities for integrin inhibitors. Nat Rev Drug Discov. 2022;21(1):60–78.
Huang J, et al. Platelet integrin alphaIIbbeta3: signal transduction, regulation, and its therapeutic targeting. J Hematol Oncol. 2019;12(1):26.
Yuan W, et al. CIB1 is an endogenous inhibitor of agonist-induced integrin alphaIIbbeta3 activation. J Cell Biol. 2006;172(2):169–75.
Oxley CL, et al. An integrin phosphorylation switch: the effect of beta3 integrin tail phosphorylation on Dok1 and talin binding. J Biol Chem. 2008;283(9):5420–6.
Kiema T, et al. The molecular basis of filamin binding to integrins and competition with talin. Mol Cell. 2006;21(3):337–47.
Wilder RL, et al. Integrin alpha V beta 3 as a target for treatment of rheumatoid arthritis and related rheumatic diseases. Ann Rheum Dis. 2002;61(2):96–9.
Sandborn WJ, Yednock TA. Novel approaches to treating inflammatory bowel disease: targeting alpha-4 integrin. Am J Gastroenterol. 2003;98(11):2372–82.
Tang MT, et al. Review article: nonclinical and clinical pharmacology, pharmacokinetics and pharmacodynamics of etrolizumab, an anti-beta7 integrin therapy for inflammatory bowel disease. Aliment Pharmacol Ther. 2018;47(11):1440–52.
Mazdeh M, et al. The effect of natalizumab on disability score and relapse rate of multiple sclerosis patients: a prospective cohort study. Clin Transl Med. 2018;7(1):38.
Yu Y, Schurpf T, Springer TA. How natalizumab binds and antagonizes alpha4 integrins. J Biol Chem. 2013;288(45):32314–25.
Goodman SL, Picard M. Integrins as therapeutic targets. Trends Pharmacol Sci. 2012;33(7):405–12.
Abidi A, et al. Lifitegrast: A novel drug for treatment of dry eye disease. J Pharmacol Pharmacother. 2016;7(4):194–8.
Campbell PS, et al. AhR ligand aminoflavone suppresses alpha6-integrin-Src-Akt signaling to attenuate tamoxifen resistance in breast cancer cells. J Cell Physiol. 2018;234(1):108–21.
Huang C, et al. b1 integrin mediates an alternative survival pathway in breast cancer cells resistant to lapatinib. Breast Cancer Res. 2011;13(4):R84.
Hanker AB, et al. Extracellular Matrix/Integrin signaling promotes resistance to combined inhibition of HER2 and PI3K in HER2þ breast cancer. Can Res. 2017;77(12):3280–92.
Nam K, et al. Binding of galectin-1 to integrin β1 potentiates drug resistance by promoting survivin expression in breast cancer cells. Oncotarget. 2017;8(22):35804–23.
Yin HL, et al. beta1 integrin as a prognostic and predictive marker in triple-negative breast cancer. Int J Mol Sci. 2016;17(9):1432.
Sun H, et al. Distinct chemokine signaling regulates integrin ligand specificity to dictate tissue-specific lymphocyte homing. Dev Cell. 2014;30(1):61–70.
Dammes N, et al. Conformation-sensitive targeting of lipid nanoparticles for RNA therapeutics. Nat Nanotechnol. 2021;16(9):1030–8.
Egorova A, et al. Peptide-based nanoparticles for alphavbeta3 integrin-targeted DNA delivery to cancer and uterine leiomyoma cells. Molecules. 2022;27(23):8363.
Cheng TM, et al. Nano-strategies targeting the integrin alphavbeta3 network for cancer therapy. Cells. 2021;10(7):1684.
Cui L, et al. Integrin alphavbeta3-Targeted [(64)Cu]CuS Nanoparticles for PET/CT imaging and photothermal ablation therapy. Bioconjug Chem. 2018;29(12):4062–71.
Kankaanpää P, et al. Cellular recognition and macropinocytosis-like internalization of nanoparticles targeted to integrin α2β1. Nanoscale. 2015;7(14):17889–901.
Parvani JG, et al. Silencing beta3 integrin by targeted ECO/siRNA nanoparticles inhibits EMT and metastasis of triple-negative breast cancer. Cancer Res. 2015;75(11):2316–25.
Tsuda T. Curcumin as a functional food-derived factor: degradation products, metabolites, bioactivity, and future perspectives. Food Funct. 2018;9(2):705–14.
Xu X, et al. Curcumin suppresses the malignancy of non-small cell lung cancer by modulating the circ-PRKCA/miR-384/ITGB1 pathway. Biomed Pharmacother. 2021;138: 111439.
Tavakol S, et al. The impact of the particle size of curcumin nanocarriers and the ethanol on beta_1-integrin overexpression in fibroblasts: a regenerative pharmaceutical approach in skin repair and anti-aging formulations. DARU J Pharma Sci. 2019;27(1):159–68.
Kim HI, et al. Curcumin inhibition of integrin (α6β4)-Dependent breast cancer cell motility and invasion. Cancer Prev Res (Phila). 2008;1(5):385–91.
Garcia-Quiroz J, et al. Synergistic antitumorigenic activity of calcitriol with curcumin or resveratrol is mediated by angiogenesis inhibition in triple negative breast cancer xenografts. Cancers (Basel). 2019;11(11):1739.
Brockmueller A, Shayan P, Shakibaei M. Evidence that beta1-Integrin is required for the anti-viability and anti-proliferative effect of resveratrol in CRC cells. Int J Mol Sci. 2022;23(9):4714.
Brockmueller A, et al. beta1-Integrin plays a major role in resveratrol-mediated anti-invasion effects in the CRC microenvironment. Front Pharmacol. 2022;13: 978625.
Tomasello L, et al. PFN1 and integrin-beta1/mTOR axis involvement in cornea differentiation of fibroblast limbal stem cells. J Cell Mol Med. 2019;23(11):7210–21.
Aksorn N, Chanvorachote P. Integrin as a molecular target for anti-cancer approaches in lung cancer. Anticancer Res. 2019;39(2):541–8.
Lee EJ, et al. Inhibition of osteoclast activation by phloretin through disturbing alphavbeta3 integrin-c-Src pathway. Biomed Res Int. 2015;2015: 680145.
Hsieh IS, Yang RS, Fu WM. Osteopontin upregulates the expression of glucose transporters in osteosarcoma cells. PLoS ONE. 2014;9(10): e109550.
Kim MS, et al. Phloretin suppresses thrombin-mediated leukocyte-platelet-endothelial interactions. Mol Nutr Food Res. 2014;58(4):698–708.
Cavalcante-Silva LHA, et al. Ouabain reduces the expression of the adhesion molecule CD18 in neutrophils. Inflammopharmacology. 2020;28(3):787–93.
Buommino E, et al. Artemisinin reduces human melanoma cell migration by down-regulating alpha V beta 3 integrin and reducing metalloproteinase 2 production. Invest New Drugs. 2009;27(5):412–8.
Efferth T, Oesch F. The immunosuppressive activity of artemisinin-type drugs towards inflammatory and autoimmune diseases. Med Res Rev. 2021;41(6):3023–61.
Zeng XZ, et al. Artesunate attenuates LPS-induced osteoclastogenesis by suppressing TLR4/TRAF6 and PLCgamma1-Ca(2+)-NFATc1 signaling pathway. Acta Pharmacol Sin. 2020;41(2):229–36.
Funding
This study is supported by National Natural Science Foundation of China grants (No.82103462, No.81972625, No.82073286), Liaoning Revitalization Talents Program (XLYC2002035); Natural Science Foundation of Liaoning Province (No.2021-BS-043); Shenyang Support Program for Young and Middle-aged Science and Technology Innovation Talents (No.RC210461).
Author information
Authors and Affiliations
Contributions
Writing-review and editing: Siyi Li, Hai-long Piao, Hong-Xu Liu; Supervision and supplement: Chibuzo Sampson, Changhao Liu. Siyi Li and, Hai-long Piao, illustrated the figures for the manuscript. All authors approved the final manuscript.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
All authors agree to submit the article for publication.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Li, S., Sampson, C., Liu, C. et al. Integrin signaling in cancer: bidirectional mechanisms and therapeutic opportunities. Cell Commun Signal 21, 266 (2023). https://doi.org/10.1186/s12964-023-01264-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12964-023-01264-4