Abstract
Introduction
Most studies have analyzed pre-arrest and resuscitation factors associated with mortality after cardiac arrest (CA) in children, but many patients that reach return of spontaneous circulation die within the next days or weeks. The objective of our study was to analyze post-return of spontaneous circulation factors associated with in-hospital mortality after cardiac arrest in children.
Methods
A prospective multicenter, multinational, observational study in 48 hospitals from 12 countries was performed. A total of 502 children aged between 1 month and 18 years with in-hospital cardiac arrest were analyzed. The primary endpoint was survival to hospital discharge. Univariate and multivariate logistic regression analyses were performed to assess the influence of each post-return of spontaneous circulation factor on mortality.
Results
Return of spontaneous circulation was achieved in 69.5% of patients; 39.2% survived to hospital discharge and 88.9% of survivors had good neurological outcome. In the univariate analysis, post- return of spontaneous circulation factors related with mortality were pH, base deficit, lactic acid, bicarbonate, FiO2, need for inotropic support, inotropic index, dose of dopamine and dobutamine at 1 hour and at 24 hours after return of spontaneous circulation as well as Pediatric Intensive Care Unit and total hospital length of stay. In the multivariate analysis factors associated with mortality at 1 hour after return of spontaneous circulation were PaCO2 < 30 mmHg and >50 mmHg, inotropic index >14 and lactic acid >5 mmol/L. Factors associated with mortality at 24 hours after return of spontaneous circulation were PaCO2 > 50 mmHg, inotropic index >14 and FiO2 ≥ 0.80.
Conclusions
Secondary in-hospital mortality among the initial survivors of CA is high. Hypoventilation, hyperventilation, FiO2 ≥ 0.80, the need for high doses of inotropic support, and high levels of lactic acid were the most important post-return of spontaneous circulation factors associated with in-hospital mortality in children in our population.
Similar content being viewed by others
Introduction
Most studies have analyzed pre-arrest and resuscitation factors associated with mortality after cardiac arrest (CA) in children [1]-[13]. Previous studies have shown that lower human development index of countries, characteristics of the hospital, CA that occurred out of hospital and out of the Pediatric Intensive Care Unit (PICU), oncohematologic disease, treatment with inotropic drugs at the time of the CA, CA due to neurological disease or sepsis, time to the initiation of resuscitation, asystole as the initial electrocardiographic (ECG) rhythm, need for adrenaline, bicarbonate or fluid expansion and the duration of cardiopulmonary resuscitation (CPR) are associated with higher mortality [1]-[13].
An important percentage of patients that reach return of spontaneous circulation (ROSC) die within the next days or weeks. However, there are no multicenter, multinational prospective studies on CA in children to have analyzed post-ROSC prognostic factors. In previous studies performed on the same prospective register we have analyzed the pre-arrest and resuscitation factors [11] and the ventilation and oxygenation factors associated with mortality [14]. The objective of the present study was to analyze the factors affecting mortality and neurological outcome of in-hospital CA in children. The hypothesis was that respiratory status and hemodynamic status are the most important prognosis factors after ROSC in children.
Methods
An open multicenter prospective study was designed and information and an invitation to participate were sent to the pediatric departments and PICUs of hospitals in Latin-American countries, Spain, Portugal, and Italy. The study was approved by local Institutional Review Boards (Additional file 1). Registration on the website [15] was necessary to participate in the study. Consent of parents of patients was not considered necessary because it was an observational study during and after CA and it is necessary to obtain data immediately.
A protocol was drawn up in accordance with the Utstein style [16],[17]. Children aged from 1 month to 18 years who suffered in-hospital CA between December 2007 and December 2009 were included. CA was defined by the presence of all the following signs: unresponsiveness, apnea, absence of signs of life and absence of a palpable central pulse or bradycardia with less than 60 beats per minute (bpm) with poor perfusion in infants requiring external cardiac compressions and assisted ventilation.
All data were entered via a secure, encrypted website and were electronically submitted to the coordinating center. That center performed a review of all records to ensure data quality, and site investigators were queried to complete missing data and resolve discrepancies.
Patient-related variables and arrest and life support-related parameters have been previously published [11] and also the relationship between ventilation and oxygenation with mortality [14]. In the present study we analyzed the influence on survival of several post-ROSC parameters, such as arterial gasometry and lactic acid at the first hour and 24 hours after ROSC, the need for mechanical ventilation, recovery of spontaneous breathing (although mechanical ventilation could be needed), the need for vasoactive drugs and doses of vasoactive drugs, vasoactive-inotropic index (VIS) [18], and ECG rhythm after ROSC. Hospital course and clinical and neurological status at hospital discharge according to the pediatric cerebral performance category [PCPC] were registered [19]. Variable definitions were based on Utstein-style guidelines [16],[17]. The primary endpoint was survival to hospital discharge. The secondary outcome measure was neurological status at hospital discharge; a good neurological status was defined as a PCPC score of 1 or 2 [19].
Statistical analyses were conducted using SPSS software version 18.1 (SPSS Inc, Chicago, IL, USA). Outcomes were compared between groups using the chi-square (χ 2) test or Fisher’s exact test for categorical variables. Univariate and multivariate logistic regression analysis was performed to assess the influence of each one of the factors on mortality. A logistic regression model was constructed for variables at 1 hour and at 24 hours after ROSC. All individual factors with statistical significance in the univariate analysis and P <0.1 were eligible for inclusion in the logistic regression model. Receiver operator characteristic (ROC) curves were used to decided cutoffs values for VIS and lactic acid. Ventilation and oxygenation cutoff values were chosen according to previous studies [20],[21] and normal limits of pH, ventilation and oxygenation. Finally a logistic regression model was constructed including patient-related variables, arrest- and life support-related parameters, and post-ROSC parameters. Adjusted odds ratios (OR) and 95% confidence intervals (CI) were calculated for each model. ROC curves were used to assess the predictive capacity of each model.
Results
Forty-eight hospitals from twelve countries participated in the study. The analysis included 563 episodes of in-hospital CA in 502 patients. CA occurred in the PICU in 50% of cases, in the emergency department in 26.8%, and in other hospital areas in 23.2%.
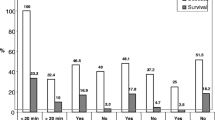
Return of spontaneous circulation (ROSC) for more than 20 minutes was achieved in 349 patients (69.5%), but 152 (30.3%) patients died later in hospital due to new CA (32.9%), multiple organ dysfunction (27%), limitation of medical therapy (25%) or brain death (15.1%): 197 patients (39.2%) survived to hospital discharge. Five patients were rescued with extracorporeal membrane oxygenation (ECMO) during CPR and four of them survived to hospital discharge. The characteristics of the 502 patients, pre-arrest factors, and cardiac arrest and resuscitation factors associated with mortality have been previously published [11].
Post-ROSC factors associated with mortality
Table 1 shows the comparison between survivors and non-survivors in post-ROSC factors. Non-survivors had lower pH, higher base excess, higher lactic acid levels and higher inspired oxygen fraction (FiO2) at 1 and 24 hours after ROSC than survivors. A greater percentage of non-survivors needed inotropic support, and they required higher doses of dopamine and dobutamine, and had a higher inotropic score than survivors did. Nevertheless, the percentage of patients receiving milrinone was lower in non-survivors than in survivors. When patients without inotropic support before CA were analyzed separately, children who required inotropic support after ROSC had 42.1% higher mortality than those who did not need it 26.9% (P =0.036). Finally, the length of PICU stay and total hospital stay was shorter in non-survivors.
Table 2 summarizes post-ROSC factors and their relationship with survival to hospital discharge in the univariate regression analysis. The need for inotropic support, an inotropic index greater than 14, the absence of spontaneous breathing, PaCO2 < 30 mmHg or >50 mmHg, FiO2 ≥ 0.80 and lactic acid levels >5 mmol/L at 1 hour after ROSC, as well as pH <7.30, PaCO2 > 50 mmHg and FiO2 > 50% at 24 hours after ROSC were significantly associated with higher mortality rates. There were no significant differences in post-ROSC PaO2 between survivors and non-survivors patients, even when the 24 patients with cyanotic heart disease and 6 patients on ECMO were excluded of analysis.
In the multivariate analysis, factors associated with mortality at 1 hour after ROSC were PaCO2 < 30 mmHg and >50 mmHg, inotropic index >14 and lactic acid >5 mmol/L (Table 3). Factors associated with mortality at 24 hours after ROSC were PaCO2 > 50 mmHg, inotropic index >14 and FiO2 ≥ 0.8 (Table 3).
The logistic regression model at 1 hour after ROSC had an AUC of 0.733 (CI 0.681 to 0.785; P =0.001). The logistic regression model at 24 hours after ROSC had an AUC of 0.769 (CI 0.720 to 0.819; P =0.001), (Figure 1).
Post-ROSC factors associated with neurological outcome
Neurological status at hospital discharge was assessed in 120 patients (60.9%), and 107 of them (89%) had a normal neurological status or showed mild disability (PCPC l or 2). PCPC before CA and at hospital discharge was compared. Only 2.8% of patients with PCPC 1 or 2 before CA presented a PCPC >2 at hospital discharge.
When comparing patients with good and bad neurological outcome (PCPC >2), at 1 hour after ROSC those with a bad outcome had significantly lower levels of bicarbonate (19.1 (7.4) mEq/L versus 21.8 (6.9) mEq/L; P =0.025), higher lactic acid levels (8.1 (6.5) mmol/L versus 5.8 (11.3) mmol/L; P =0.003) and higher base excess (BE) (-7.1 (9.4) mEq/L versus -3.4 (9.6) mEq/L; P =0.042) (Table 4). A higher percentage of patients with bad neurological outcome received dobutamine (40.9%) than those with good neurological outcome (19%) P =0.007 (Table 4).
The univariate analysis showed that dobutamine administration and lactic acid levels >5 mmol/L at 1 hour, and pH >7.50, PaCO2 > 50 mmHg and BE >4 mEq/L at 24 hours after ROSC were associated with poor neurological evolution (Table 5).
In the multivariate analysis the only factor associated with poor neurological outcome at 1 hour after ROSC was lactic acid above 5 mmol/L (OR 9.902, CI (1.992 to 51.008); P =0.006). None of the factors at 24 hours after ROSC showed statistical significance in the multivariate analysis.
Pre-arrest, resuscitation and post-ROSC factors
The multivariate analysis including pre-arrest factors, resuscitation factors and post-ROSC factors is shown in Table 6. Factors associated with in-hospital mortality were hemato-oncologic illness, neurologic cause of arrest, CA in the emergency department, treatment with inotropic drugs before CA, administration of sodium bicarbonate, PaCO2 < 30 mmHg 1 hour after ROSC, PaCO2 > 50 mmHg one hour after ROSC and FiO2 ≥ 0.80 24 hours after ROSC.
Discussion
To our knowledge, this is the first multicenter multinational study that analyzed the association of early post-ROSC factors with outcome of in-hospital cardiac arrest in children according to the Utstein style guidelines. Sustained ROSC was achieved in 69.5% patients but secondary in-hospital mortality among the initial survivors of CA was 43.5% and survival to hospital discharge was therefore 39.2%.
Oxygenation and ventilation parameters
Several studies, including our previous analysis, showed that alterations in ventilation and oxygenation during the first hours after ROSC are associated with prognosis [14],[20]-[24]. Our study shows that PaCO2 < 30 mmHg and >50 mmHg at 1 hour and PaCO2 > 50 mmHg at 24 hours after ROSC are mortality indicators [14]. Our results differ from those reported in a retrospective study in 195 children after CA, in which no relationship was found between ventilation and mortality [20]. Hyperventilation may increase mortality and brain damage by reducing cerebral blood flow and tissue perfusion resulting in ischemia [22]. On the other hand, hypoventilation may increase the risk of cerebral edema and intracranial hypertension due to cerebral vasodilation [23]. In addition, hypercapnia can impair myocardial function and induce vasoconstriction of the pulmonary vascular bed [23]. Our findings highlight the importance of monitoring ventilation using capnography and blood gas analysis in order to rapidly achieve an appropriate ventilation status after ROSC, although capnography values can be altered in patients with abnormal PaO2/FiO2.
PaO2 was not associated with mortality in the univariate or in the multivariate analysis at 1 hour and at 24 hours after ROSC. Two recent retrospective studies in children did not find this association between mortality and oxygenation either [20],[24]. Nevertheless, another retrospective study that analyzed 1,875 pediatric patients found a correlation between mortality in the PICU and hypoxia and, to a lesser extent, with hyperoxia. This study did not analyze the relationship between ventilation and mortality [21].
In our study, non-survivors had higher FiO2 than survivors, and the univariate analysis showed that FiO2 ≥ 0.80 was associated with mortality. The multivariate logistic regression study showed that high FiO2 could be considered a risk factor only at 24 hours after ROSC. Elevated FiO2 may cause cellular toxicity as shown in previous studies in neonates [25]. On the other hand, elevated FiO2 could also indicate a greater need for oxygen, as worse tissue oxygenation may exist. Nevertheless, no relationship was found between PaO2 or PaO2/FiO2 and mortality in the patients in our study, and it may be that these patients did not require such a high FiO2. On the other hand we did not find association between ventilation and oxygenation parameters and neurologic outcome. This may be because the number of patients with hypoxia and hyperoxia was insufficient to detect significant differences, or that only important alterations in oxygenation could influence neurologic outcome.
We think that it is possible that ventilation and oxygenation could influence the prognosis of children who suffer CA. However, multicenter controlled studies with a sufficient number of patients are needed because many other factors besides ventilation and oxygenation may influence outcome in CA patients.
Lactid acid
Lactic acid is one of the most commonly used parameters to assess and monitor hypoperfusion or tissue hypoxia in critically ill patients, as it has been demonstrated to have good prognostic capacity and it is easy and fast to measure [26]. Lactate levels in patients who have recovered from CA probably reflect the severity of the ischemia-reperfusion syndrome. Nevertheless, high lactic acid levels may exist without the presence of tissue hypoperfusion due to the administration of adrenaline or to the presence of hyperglycemia, which are very common after CA [26].
Several studies have found that lactate levels in the first 48 hours after CA is lower in survivors and in patients without neurological damage [27]-[29]. The levels of lactate after ROSC and 12 or 24 h later were significantly higher in non survivors adults and children after out-of-hospital and in-hospital CA [9],[30]-[33]. Lactate clearance within the first 24 hours (lactate after ROSC minus lactate 24 hours after ROSC) × 100/lactate after ROSC) is significantly higher in survivors than in non-survivors [30]-[32].
In our study, non-survivors had more acidosis both at 1 hour and at 24 hours after ROSC. Acidosis was mainly due to metabolic acidosis, with lower bicarbonate levels and higher base deficit in non-survivors than in survivors. Nevertheless, the only factor associated with mortality in the logistic regression analysis was lactic acid at 1 hour and at 24 hours after ROSC. Non-survivors presented significantly higher levels of lactic acid at 1 and 24 hours after ROSC, and lactic acid levels >5 mmol/L were associated with higher mortality in the univariate and multivariate analysis.
Although lactate at 1 and at 24 hours after ROSC was higher in non-survivors, no significant differences were found in lactate clearance, because lactate acid levels significantly decreased in the first 24 hours in both groups (from 16.9 to 5.8 mmol/L in non-survivors and from 6.7 to 3.7 mmol/L in survivors).
Vasoactive treatment
Hemodynamic alterations after ROSC are also late mortality risk factors. Cardiac rhythm after ROSC and need for vasoactive drugs in the first 24 hours were analyzed in order to assess hemodynamic alterations. Other hemodynamic parameters, such as heart rate, blood pressure or central venous pressure were not registered. The non-surviving group had a greater percentage of patients requiring vasoactive support and at higher doses (higher inotropic index) than the surviving group. The univariate and multivariate studies showed that vasoactive-inotropic score >14 was significantly associated with mortality.
Several studies have found that the need for pressors previous to CA is a mortality risk factor, both in adults [34]-[37] and in children [4],[6],[8],[11]. Our study shows that the need for pressors and at higher doses (vasoactive-inotropic score) after CA is associated with higher risk of mortality. This fact has also been found by Meert et al. [9]. This highlights the influence of early hemodynamic alterations on outcome in children after CA, and the importance of treating these alterations as they appear.
On the other hand, a large percentage of patients received more than one vasoactive agent after ROSC. That may be the cause for not finding a significant association between mortality and the administration or dosage of any specific vasoactive drug, but with the intensity of vasoactive treatment in general. Inotropic score has proved to be appropriate in assessing vasoactive support and its relationship with mortality in several studies in children in shock, after open heart surgery and after heart transplantation [18],[38],[39]. Rhodes et al. found that the inotropic score was higher in non-survivors than in survivors with CA after congenital heart surgery [39]. Our study also suggests that the vasoactive-inotropic score may be a useful prognostic indicator in children after CA.
A recent retrospective study in adults showed that the combination of elevated lactate levels and the need for vasoactive support had a good mortality predictive capacity in patients that recovered from CA [40]. Our results agree with those from the mentioned study, although in our study the dose of vasoactive support also proved to have mortality predictive capacity. Furthermore, PaCO2 levels, which were not registered in the study in adults, also proved to have prognostic capacity in our study. On the other hand, lactic acid levels >5 mmol/L at 1 hour after ROSC was the only factor that was associated with bad neurological outcome in our study.
Pre-arrest, resuscitation and post-ROSC multivariate analysis
Meert et al. [9] performed a multivariate analysis including pre-arrest, resuscitation and 12 h post-ROSC factors. In this study the only post-ROSC factor associated with survival al hospital discharge was the responsive pupils after ROSC. Conversely, our multivariate analysis, including pre-arrest, resuscitation and post-ROSC factors, showed that the post-ROSC factors associated with mortality were hypoventilation and hyperventilation 1 hour after ROSC and high FiO2 24 hours after ROSC, highlighting the importance of the control of ventilation and oxygenation after ROSC.
Limitations
Our study has several limitations. One of them is that hemodynamic variables such as heart rate, blood pressure and central venous pressure in the first 24 hours after ROSC were not registered, making it impossible to accurately assess the presence of shock in these patients. In a recent study hypotension after ROSC was related to bad prognosis [41]. Hypothermia or hyperthermia and the parameters of mechanical ventilation were not registered neither.
Pre-arrest values of lactate or vasoactive-inotropic score could influence post-ROSC values but we did not register pre-arrest lactate and VIS data in our patients. On the other hand, our study has only analyzed prognostic factors in the first 24 hours after ROSC, which may be the most important but not the only ones. Other factors that affect prognosis but may appear in the following days, such as nosocomial infections or multiple organ failure, were not analyzed. Actually, the median PICU stay for non-survivors was 8 days, and many of these patients died because of complications due to multi-organ failure.
Finally, only 61% of patients have neurologic outcome evaluation, although there were no differences in baseline characteristics between patients with and without a neurologic outcome measure. On the other hand, only a small number of patients had bad neurological outcomes. This is why the power of the statistical analysis in relationship with neurological outcome is poor, so results must be interpreted with caution. Therefore, more studies are needed to prospectively assess both early and late post-ROSC mortality and neurologic outcome risk factors in children after CA.
Conclusions
We conclude that secondary in-hospital mortality among the initial survivors of CA is high (43.5% in our study). The most important early mortality risk factors after ROSC in in-hospital CA in children are hyperventilation, hypoventilation, high FiO2 requirements need for high doses of inotropic drugs and high lactic acid levels. High lactic acid levels at 1 hour after ROSC were associated with bad neurological outcome.
Key messages
-
Secondary in-hospital mortality among the initial survivors of CA is high (43.5% in our study)
-
The most important early mortality risk factors after ROSC in-hospital CA in children were hyperventilation, hypoventilation, need for high doses of inotropic drugs, high lactic acid levels and high FiO2 requirements
-
High lactic acid levels at 1 hour after ROSC were associated with bad neurological outcome
Early treatment of hemodynamic and respiratory disturbances after ROSC could improve mortality in initial survivors of CA
Additional file
Abbreviations
- BE:
-
base excess
- bpm:
-
beats per minute
- CA:
-
cardiac arrest
- CPR:
-
cardiopulmonary resuscitation
- ECG:
-
electrocardiographic
- ECMO:
-
extracorporeal membrane oxygenation
- FiO2:
-
inspired oxygen fraction
- OR:
-
adjusted odds ratios
- PaO2:
-
arterial partial pressure of oxygen
- PCPC:
-
pediatric cerebral performance category
- PICU:
-
Pediatric Intensive Care Unit
- ROC:
-
receiver operator characteristic
- ROSC:
-
return of spontaneous circulation
- VIS:
-
vasoactive-inotropic index
References
Suominen P, Olkkola KT, Voipio V, Korpela R, Palo R, Rasanan J: Utstein style reporting of-in hospital paediatric cardiopulmonary resuscitation. Resuscitation. 2000, 45: 17-25. 10.1016/S0300-9572(00)00167-2.
Reis AG, Nadkarni V, Perondi MB, Grisi S, Berg RA: A prospective investigation into the epidemiology of in-hospital pediatric cardiopulmonary resuscitation using the International Utstein reporting style. Pediatrics. 2002, 109: 200-209. 10.1542/peds.109.2.200.
López-Herce J, Garcia C, Dominguez P, Carrillo A, Rodriguez-Nunez A, Calvo C, Delgado MA: Characteristics and outcome of cardiorespiratory arrest in children. Resuscitation. 2004, 63: 311-313. 10.1016/j.resuscitation.2004.06.008.
Rodriguez-Nunez A, Lopez-Herce J, Garcia C, Carrillo A, Dominguez P, Calvo C, Delgado MA: Effectiveness and long-term outcome of cardiopulmonary resuscitation in paediatric intensive care units in Spain. Resuscitation. 2006, 71: 301-309. 10.1016/j.resuscitation.2005.11.020.
Tibballs J, Kinney S: A prospective study of outcome of in-patient paediatric cardiopulmonary arrest. Resuscitation. 2006, 71: 310-318. 10.1016/j.resuscitation.2006.05.009.
De Mos N, van Litsenburg RR, McCrindle B, Bohn DJ, Parshuram CS: Pediatric in-intensive-care-unit cardiac arrest: incidence, survival, and predictive factors. Crit Care Med. 2006, 34: 1209-1215. 10.1097/01.CCM.0000208440.66756.C2.
Meaney PA, Nadkarni VM, Cook EF, Testa M, Helfaer M, Kaye W, Larkin GL, Berg RA: Higher survival rates among younger patients after pediatric intensive care unit cardiac arrest. Pediatrics. 2006, 118: 2424-2433. 10.1542/peds.2006-1724.
Wu ET, Li MJ, Huang SC, Wang CC, Liu YP, Lu FL, Ko WJ, Wang MJ, Wang JK, Wu MH: Survey of outcome of CPR in pediatric in-hospital cardiac arrest in a medical center in Taiwan. Resuscitation. 2009, 80: 443-448. 10.1016/j.resuscitation.2009.01.006.
Meert KL, Donaldson A, Nadkarni V, Tieves KS, Schleien CL, Brilli RJ, Clark RS, Shaffner DH, Levy F, Statler K, Dalton HJ, van der Jagt EW, Hackbarth R, Pretzlaff R, Hernan L, Dean JM, Moler FW: Multicenter cohort study of in-hospital pediatric cardiac arrest. Pediatr Crit Care Med. 2009, 10: 544-553. 10.1097/PCC.0b013e3181a7045c.
Berens RJ, Cassidy LD, Matchey J, Campbell D, Colpaert KD, Welch T, Lawson M, Peterson C, O'Flynn J, Dearth M, Tieves KS: Probability of survival based on etiology of cardiopulmonary arrest in pediatric patients. Paediatr Anaesth. 2011, 21: 834-840. 10.1111/j.1460-9592.2010.03479.x.
López-Herce J, Del Castillo J, Matamoros M, Cañadas S, Rodriguez-Calvo A, Cecchetti C, Rodriguez-Núñez A, Alvarez AC: Factors associated with mortality in pediatric in-hospital cardiac arrest: a prospective multicenter multinational observational study. Intensive Care Med. 2013, 39: 309-318. 10.1007/s00134-012-2709-7.
Moler FW, Meert K, Donaldson AE, Nadkarni V, Brilli RJ, Dalton HJ, Clark RS, Shaffner DH, Schleien CL, Statler K, Tieves KS, Hackbarth R, Pretzlaff R, van der Jagt EW, Levy F, Hernan L, Silverstein FS, Dean JM: In-hospital versus out-of-hospital pediatric cardiac arrest: a multicenter cohort study. Crit Care Med. 2009, 37: 2259-2267. 10.1097/CCM.0b013e3181a00a6a.
Donoghue AJ, Nadkarni VM, Elliott M, Durbin D: Effect of hospital characteristics on outcomes from pediatric cardiopulmonary resuscitation: a report from the national registry of cardiopulmonary resuscitation. Pediatrics. 2006, 118: 995-1001. 10.1542/peds.2006-0453.
Del Castillo J, López-Herce J, Matamoros M, Cañadas S, Rodriguez-Calvo A, Cechetti C, Rodriguez-Núñez A, Alvarez AC: Hyperoxia, hypocapnia and hypercapnia as outcome factors after cardiac arrest in children. Resuscitation. 2012, 83: 1456-1461. 10.1016/j.resuscitation.2012.07.019.
Iberoamerican study of cardiac arrest in children. [], ["http://www.pcrpediatrica.es]
Cummins R, Chamberlain D, Hazinski MF, Nadkarni V, Kloek W, Kramer E: Recommended guidelines for reviewing, reporting, and conducting research on in hospital resuscitation: the in-hospital "Utstein style". Resuscitation. 1997, 34: 151-183. 10.1016/S0300-9572(97)01112-X.
Jacobs I, Nadkarni V, Bahr J, Berg RA, Billi JE, Bossaert L, Cassan P, Coovadia A, D'Este K, Finn J, Halperin H, Handley A, Herlitz J, Hickey R, Idris A, Kloeck W, Larkin GL, Mancini ME, Mason P, Mears G, Monsieurs K, Montgomery W, Morley P, Nichol G, Nolan J, Okada K, Perlman J, Shuster M, Steen PA, Sterz F, et al: Cardiac arrest and cardiopulmonary resuscitation outcomes reports: update and simplification of the utstein templates for resuscitation registries: a statement for healthcare professionals from a task force of the international liaison committee on resuscitation (American heart association, European resuscitation council, Australian resuscitation council, New Zealand resuscitation council, heart and stroke foundation of Canada, InterAmerican heart foundation, resuscitation councils of southern Africa. Circulation. 2004, 110: 3385-3397. 10.1161/01.CIR.0000147236.85306.15.
Gaies MG, Gurney JG, Yen AH, Napoli ML, Gajarski RJ, Ohye RG, Charpie JR, Hirsch JC: Vasoactive-inotropic score as a predictor of morbidity and mortality in infants after cardiopulmonary bypass. Pediatr Crit Care Med. 2010, 11: 234-238. 10.1097/PCC.0b013e3181b806fc.
Fiser DH, Long N, Roberson PK, Heffley G, Zolten K, Brodie-Fowler M: Relationship of pediatric overall performance category and pediatric cerebral performance category scores at pediatric intensive care unit discharge with outcome measures collected at hospital discharge and 1- and 6-month follow-up assessments. Crit Care Med. 2000, 28: 2616-2620. 10.1097/00003246-200007000-00072.
Bennett KS, Clark AE, Meert KL, Topjian AA, Schleien CL, Shaffner DH, Dean JM, Moler FW: Early oxygenation and ventilation measurements after pediatric cardiac arrest: lack of association with outcome. Crit Care Med. 2013, 41: 1534-1542. 10.1097/CCM.0b013e318287f54c.
Ferguson LP, Durward A, Tibby SM: Relationship between arterial partial oxygen pressure after resuscitation from cardiac arrest and mortality in children. Circulation. 2012, 126: 335-342. 10.1161/CIRCULATIONAHA.111.085100.
Curley G, Kavanagh BP, Laffey JG: Hypocapnia and the injured brain: more harm than benefit. Crit Care Med. 2010, 38: 1348-1359.
Curley G, Laffey JG, Kavanagh BP: Bench-to-bedside review: carbon dioxide. Crit Care. 2010, 14: 220-10.1186/cc8926.
Guerra-Wallace MM, Casey FL, Bell MJ, Fink EL, Hickey RW: Hyperoxia and hypoxia in children resuscitated from cardiac arrest. Pediatr Crit Care Med. 2013, 14: e143-e148. 10.1097/PCC.0b013e3182720440.
Saugstad OD: Resuscitation of newborn infants: from oxygen to room air. Lancet. 2010, 376: 1970-1971. 10.1016/S0140-6736(10)60543-0.
Jansen TC, van Bommel J, Bakker J: Blood lactate monitoring in critically ill patients: a systematic health technology assessment. Crit Care Med. 2009, 37: 2827-2839. 10.1097/CCM.0b013e3181a98899.
Kliegel A, Losert H, Sterz F, Holzer M, Zeiner A, Havel C, Laggner AN: Serial lactate determinations for prediction of outcome after cardiac arrest. Medicine (Baltimore). 2004, 83: 274-279. 10.1097/01.md.0000141098.46118.4c.
Müllner M, Sterz F, Domanovits H, Behringer W, Binder M, Laggner AN: The association between blood lactate concentration on admission, duration of cardiac arrest, and functional neurological recovery in patients resuscitated from ventricular fibrillation. Intensive Care Med. 1997, 23: 1138-1143. 10.1007/s001340050470.
Shinozaki K, Oda S, Sadahiro T, Nakamura M, Hirayama Y, Watanabe E, Tateishi Y, Nakanishi K, Kitamura N, Sato Y, Hirasawa H: Blood ammonia and lactate levels on hospital arrival as a predictive biomarker in patients with out-of-hospital cardiac arrest. Resuscitation. 2011, 82: 404-409. 10.1016/j.resuscitation.2010.10.026.
Karagiannis C, Georgiou M, Kouskouni E, Iacovidou N, Santos T: Association of lactate levels with outcome after in-hospital cardiac arrest. Resuscitation. 2012, 83: e175-e176. 10.1016/j.resuscitation.2012.04.016.
Grossestreuer A, Perman S, Napoli A, Abella B, Becker L, Leary M, Gaieski D: Association between serum lactate levels, lactate clearance and survival to discharge in post-cardiac arrest patients. Resuscitation. 2012, 83: e2-10.1016/j.resuscitation.2012.08.008.
Starodoub R, Abella B, Grossstreuer A, Shofer FS, Perman SM, Leary M, Gaiesiki DF: Association of serum lactate levels and survival outcomes in patients undergoing therapeutic hypothermia after cardiac arrest. Resuscitation. 2012, 83: e2-
Topjian AA, Clark AE, Casper TC, Berger JT, Schleien CL, Dean JM, Moler FW: Early lactate elevations following resuscitation from pediatric cardiac arrest are associated with increased mortality. Pediatr Crit Care Med. 2013, 14: e380-e387. 10.1097/PCC.0b013e3182976402.
Tian J, Kaufman DA, Zarich S, Chan PS, Ong P, Amoateng-Adjepong Y, Manthous CA: Outcomes of critically ill patients who received cardiopulmonary resuscitation. Am J Respir Crit Care Med. 2010, 182: 501-506. 10.1164/rccm.200910-1639OC.
Grigoriyan A, Vazquez R, Palvinskaya T, Bindelglass G, Rishi A, Amoateng-Adjepong Y, Manthous CA: Outcomes of cardiopulmonary resuscitation for patients on vasopressors or inotropes: A pilot study. J Crit Care. 2009, 24: 415-418. 10.1016/j.jcrc.2009.02.005.
Levy PD, Ye H, Compton S, Chan PS, Larkin GL, Welch RD: Factors associated with neurologically intact survival for patients with acute heart failure and in- hospital cardiac arrest. Circ Heart Fail. 2009, 2: 572-581. 10.1161/CIRCHEARTFAILURE.108.828095.
Larkin GL, Copes WS, Nathanson BH, Kaye W: Pre-resuscitation factors associated with mortality in 49,130 cases of in-hospital cardiac arrest: a report from the national registry for cardiopulmonary resuscitation. Resuscitation. 2010, 81: 302-311. 10.1016/j.resuscitation.2009.11.021.
Davidson J, Tong S, Hancock H, Hauck A, da Cruz E, Kauffman J: Prospective validation of the vasoactive-inotropic score and correlation to short-term outcomes in neonates and infants after cardiothoracic surgery. Intensive Care Med. 2012, 38: 1184-1190. 10.1007/s00134-012-2544-x.
Rhodes JF, Blaufox AD, Seiden HS, Asnes JD, Gross RP, Rhodes JP, Griepp RB, Rossi AF: Cardiac arrest in infants after congenital heart surgery. Circulation. 1999, 100: II194-II199. 10.1161/01.CIR.100.suppl_2.II-194.
Cocchi MN, Miller J, Hunziker S, Carney E, Salciccioli J, Farris S, Joyce N, Zimetbaum P, Howell MD, Donnino MW: The association of lactate and vasopresssor need for mortality prediction in survivors of cardiac arrest. Minerva Anestesiol. 2011, 77: 1063-1071.
Topjian AA, French B, Sutton RM, Conlon T, Nadkarni VM, Moler FW, Dean JM, Berg RA: Early postresuscitation hypotension is associated with increased mortality following pediatric cardiac arrest. Crit Care Med. 2014, 42: 1518-1523. 10.1097/CCM.0000000000000216.
Acknowledgements
This study was supported in part by grant RT02377 from the Science and Technology for Development (CYTED) Program and by grant PI081167 from Carlos III Institute of Health, Spain. They did not participate in design, collection, analysis, interpretation of data, writing of the manuscript or the decision to submit the manuscript for publication. List of the investigators of the Iberoamerican Pediatric Cardiac Arrest Study Network: Jesús López-Herce, Jimena del Castillo, Javier Urbano, Angel Carrillo, Jose Maria Bellon, Sarah Fernández (Hospital General Universitario Gregorio Maranon, Madrid, Spain), Martha Matamoros, Roger Rodríguez, Allison Callejas, Douglas Carranza, Hilda Zerón (Hospital Escuela, Tegucigalpa, Honduras), Sonia Canadas, Pedro Dominguez (Hospital Valle de Hebron, Barcelona, Spain), Ana Rodriguez Calvo, Lorenzo Marcos (Hospital Nino Jesus, Tucuman, Argentina), Corrado Cechetti (Ospedale Bambinu Gesu, Roma, Italy), Marta Silva (Hospital San Joao, Porto, Portugal), Regina Grigolli Cesar (Irmandade da Santa Casa de Misericordia, Sao Paulo, Brasil), Javier Pilar Orive (Hospital de Cruces, Baracaldo, Spain), Ana Maria Nieva (Hospital de Ninos Ricardo Gutierrez, Buenos Aires, Argentina), Antonio Rodriguez-Nunez (Hospital Clinico Universitario, Santiago de Compostela, Spain), Marta Parada (Hospital Pediatrico, Coimbra, Portugal), Maria Angeles Garcia Teresa (Hospital Nino Jesus, Madrid, Spain), Di Prietro Pasquale (Ospedale Gaslini, Genova, Italy), Miguel Angel Delgado (Hospital Universitario La Paz, Madrid, Spain), Mauricio Fernandez, Hospital Pablo Tobon Uribe, Medellin, Colombia), Roxana Flavia Jaen (Hospital Britanico, Buenos Aires, Argentina), Juan Garbayo Solana (Hospital Reina Sofia, Cordoba, Spain), Raul Borrego Dominguez (Hospital Virgen de la Salud, Toledo, Spain), Victor Monreal (Hospital Roberto del Rio, Santiago de Chile, Chile), Cristina Molinos (Hospital de Cabueñes, Asturias, Spain), Iolster Thomas (Hospital Universitario Austral, Buenos Aires, Argentina), Ricardo Iramain (Hospital Nacional de Asunción, Asuncion, Paraguay), Juan Carlos de Carlos (Hospital Son Dureta, Palma de Mallorca, Spain), Corsino Rey Galán (Hospital Central de Asturias, Oviedo, Spain), Custodio Calvo Macías, Hospital Carlos Haya, Málaga, Spain), Olivia Pérez Quevedo (Hospital Materno Infantil de Las Palmas, Las Palmas de Gran Canaria, Spain), Adriana Koliski (Hospital da Clinicas da UFPR, Curitiba, Brasil), Santiago Campos (Hospital SOLCA, Quito, Ecuador), Alfredo Reparaz (Complexo Hospitalario Universitario de Vigo, Vigo, Spain), Sivia Sanchez Perez (Corporacion Parc Taul, Sabadell, Spain), Deolinda Matos (Hospital Garcia de Orta, Almada, Portugal), Claudia Carolina Benaroya Hospital Regional Rio Gallegos, Rio Gallegos, Argentina), Jessica Ortiz Rodríguez (Hospital San Juan de Dios, Barcelona, Spain), Pedro Pablo Oyagüez (Complejo Asistencial de Burgos, Spain), Juan Carlos de Carlos (Hospital Son Dureta, Palma de Mallorca, Spain), Mario José Sánchez Fernández (Hospital Josep Trueta, Gerona, Spain), Cristina Molinos (Hospital de Cabueñes, Asturias, Spain), Concepción Goñi Orayen (Hospital Virgen del Camino, Pamplona, Spain), Asunción Pino Vázquez (Hospital Clínico de Valladolid, Spain), Ma Elena May Llanas (Hospital Mutua de Tarrasa, Barcelona, Spain), Abián Montesdeoca Melián (Hospital Universitario de Canarias, Tenerife, Spain), Isabel Lucía Benítez Gómez (Hospital Virgen del Rocío, Sevilla, Spain), Antonio de Francisco (Hospital Germans Trias i Pujol, Barcelona, Spain). Santiago Hermógenes Esquivel (Hospital Oscar Alende, Lomas de Zamora; Vicente López y Planes, General Rodriguez, Argentina), Cecilia Andrea Chede (Hospital de Clínicas UBA, Caba, Argentina), Gabriel Cassaletti Bustillo (Clínica Shaio, Bogota, Colombia), Lourdes Marroquin Yanez (Hospital Infantil de México Federico Gómez, Mexico, Spain).
Author information
Authors and Affiliations
Consortia
Corresponding author
Additional information
Competing interests
All authors declare that they have no competing interests.
Authors’ contributions
JLH: conceived, designed and coordinated the study, analyzed data and drafted the manuscript. JC: participated in the design and coordination of the study, in the statistical analysis of data and helped to draft the manuscript. MM: participated in the coordination of the study, analysis of data and helped to draft the manuscript. SC: participated in the coordination of the study, analysis of data and helped to draft the manuscript. ARC: participated in the coordination of the study, analysis of data and helped to draft the manuscript. CC: participated in the coordination of the study, analysis of data and helped to draft the manuscript. ARN: participated in the design and coordination of the study and helped to draft the manuscript. AC: participated in the design and coordination of the study and helped to draft the manuscript. All authors read and approved the final manuscript.
Electronic supplementary material
Authors’ original submitted files for images
Below are the links to the authors’ original submitted files for images.
Rights and permissions
This article is published under an open access license. Please check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended use exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
About this article
Cite this article
López-Herce, J., del Castillo, J., Matamoros, M. et al. Post return of spontaneous circulation factors associated with mortality in pediatric in-hospital cardiac arrest: a prospective multicenter multinational observational study. Crit Care 18, 607 (2014). https://doi.org/10.1186/s13054-014-0607-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s13054-014-0607-9