Abstract
Background
The increase in the frequency and intensity of droughts is pointed out as one of the main factors altering biogeochemical cycles in the Amazon basin. An eco-nutritional approach using X-ray fluorescence micro-analysis (µXRF) is proposed to verify the long- and short-term effects of droughts on the growth and xylem nutrient concentrations of Cedrela fissilis Vell.
Methods
Fourteen radii were selected from a tree-ring width chronology and X-rayed by Itrax Multiscanner. Profiles of ring width, wood density, and concentrations of aluminum (Al), phosphorus (P), sulfur (S), calcium (Ca), potassium (K), manganese (Mn), iron (Fe) and strontium (Sr) together with Al/Ca, Ca/Mn, K/Ca, Sr/Ca and Mn/S ratios were constructed and correlated with precipitation, temperature, the difference between precipitation and potential evapotranspiration (P-PET) and standardized precipitation–evapotranspiration index (SPEI).
Results
During dry years, C. fissilis showed narrower, less dense rings, lower Al, P, S and Ca, and higher K and Fe concentrations (the opposite was found in wet years). Ring width decreased (together with Al, P, S, K, Ca, Mn, Fe, Sr, Al/Ca, K/Ca and Sr/Ca) and wood density increased (together with Ca/Mn and Mn/S), which was associated with an increase in evapotranspiration and temperature over time, mainly since 1990. Cedrela fissilis showed a tendency to increase its capacity for resistance, and a recovery and resilience in growth over time associated with responses in Al, Ca, P and S. However, it showed a risk in the capacity for recovery of the pre-drought density values, associated with unsatisfactory responses in Al, Ca, K, Fe and P.
Conclusions
This study is the first attempt to analyze tree-ring nutritional evidences of C. fissilis trees to climate sensitivity and resilience to drought, based on long-term data from seasonal moist tropical forests of the Amazon. Our data suggested that C. fissilis is undergoing alterations in the concentration, use and redistribution of nutrients associated with increasing wood density and decreasing growth over time, due to the increase of drought frequency in the southern Amazon.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Introduction
Drought in the Amazon region affects the growth and nutritional status of trees, conditioning the ecological processes by pushing the forest to critical physiological thresholds. The observations in the last decades and the forecasts for the coming years show the dangerous tipping point of recovery that the Amazon region can reach (Lovejoy and Nobre 2018) as a consequence (among others) of the increases in the frequency of droughts and floods (Phillips et al. 2009; Marengo et al. 2016; Marengo and Espinoza 2016), large-scale deforestation (Lejeune et al. 2015; Boers et al. 2017; Exbrayat et al. 2017; Marengo et al. 2018), and forest fires (Marengo and Espinoza 2016; Aragão et al. 2018; Marengo et al. 2018). These agents of Amazon forest alteration can have important implications for hydrological cycle variation (e.g., evapotranspiration, heat flux and water yield and discharge) (Fu and Li 2004; Li and Fu 2004; Marengo 2009; Coe et al. 2016; Smith et al. 2023), forest ecosystem dynamics (e.g., tree mortality) (Aleixo et al. 2019; Fauset et al. 2019; Locosselli et al. 2020) and biogeochemical cycling (e.g., carbon sink and nutrient imbalances) (Brienen et al. 2015; Buscardo et al. 2016; Grace 2016).
The major factors underlying patterns in biogeochemical cycles in ecosystems, including Amazon forests, are represented by climate, species composition, soil fertility, and successional status (Vitoussek and Sanford 1986). Disturbances in these factors generate an imbalance in the stocks, allocation and flows of nutrients (McGrath et al. 2001; Quesada et al. 2012; Buscardo et al. 2016; Malhi et al. 2021). Over recent decades, biogeochemistry studies in the Amazon region have focused on the carbon cycle (Brienen et al. 2015; Araújo et al. 2016; Gloor 2016; Grace 2016; Higuchi et al. 2016; Covey et al. 2021; Pinagé et al. 2023). The undisturbed Amazon forest acts as a substantial carbon sink that offsets carbon dioxide (CO2) emissions from fossil fuel combustion and land use change in the region (Brienen et al. 2015; Phillips et al. 2017). The increase in the frequency of droughts and the associated occurrence of fires and mortality of species, mainly in the central and southern Amazon region (Phillips et al. 2016; Lovejoy and Nobre 2018), aggravated the natural carbon loss (Espírito-Santo et al. 2014), turning the basin into a strong source of CO2 (Covey et al. 2021; Malhi et al. 2021). The interactions among other biogeochemical cycles, such as those of nitrogen (Figueiredo et al. 2019) and phosphorus (Fleischer et al. 2019), have also gained relevance due to their modulating role in the carbon cycle. The co-limitation by nitrogen (not deficient) and phosphorus (deficient in 90% of the Amazonian soils, Sanchez et al. 1982) is an important constraint to plant productivity (biomass) and CO2 fertilization responses of Amazon forest (Buscardo et al. 2016; Malhi et al. 2021; Cunha et al. 2022).
Other essential minerals for plant productivity, such as calcium, potassium, magnesium and sulfur (and their non-essential analog minerals, such as strontium) (Burger and Lichtscheidl 2019) are highly stocked by the canopy (Jordan et al. 1980; Jordan 1985), although they are poorly available in the most common Amazonian soils (75% classified as Ultisons and Oxisols) (Sanchez et al. 1982) due to the low cation exchange capacity of the soils (Sanchez et al. 1982; Cuevas and Medina 1986; Vitoussek and Sanford 1986; Laurance et al. 1999; Moreira et al. 2006; Buscardo et al. 2016). These soils are deep, well-drained with mostly favorable physical properties, but they present low fertility characterized by the predominance of iron and aluminum oxides (also manganese oxides in less drained areas) (Moreira et al. 2006; Mescouto et al. 2011; De Souza et al. 2018) and kaolinite clay minerals, low organic matter content and low pH values (acid soil, pH < 5.5) (Sanchez et al. 1982; Buscardo et al. 2016; Souza et al. 2018). The low fertility of these soils is also due to parent material (predominantly sedimentary rocks), heavy rainfall (promotes leaching), and high temperatures that result in rapid decomposition of organic matter (De Souza et al. 2018). Furthermore, under a regime of climate change (longer and more intense dry seasons) (Phillips et al. 2016), it is expected that in soils with high clay content, such as Ultisons and Oxisols, forests will suffer significant losses of biomass, specifically, increased vulnerability and substantial decline in late-successional tree abundance (Levine et al. 2016).
Tree vulnerability to drought varies according to the functional diversity of the species in the stand (Grossiord 2020), its role in forest dynamics (Esquivel-Muelbert et al. 2019) and its adaptive capacity to the local environment (Aguirre-Gutiérrez et al. 2020), which translates into a heterogeneous response of the species throughout the Amazon (Malhi et al. 2021). Ecohydrological theories of tree responses to drought (Anderegg et al. 2018; Wang et al. 2020; Wu et al. 2020) are consistent with xylematic responses suggesting that more resilient hydraulic architecture (e.g., greater embolism resistance of their water-transporting xylem) is less likely to succumb to drought (Lachenbruch and Mcculloh 2014; Rowland et al. 2015; Islam et al. 2018; Janssen et al. 2019; Powers et al. 2020). Furthermore, trade-offs between growth and wood density are expected, where, for example, species with a strong growth response to water availability averaged a low wood density, which may facilitate stem water storage (Mendivelso et al. 2013, 2014; Camarero et al. 2020). Other defense mechanisms of plants against drought are possible if there is an optimal balance in nutrient supply (Gessler et al. 2017; Hevia et al. 2019). However, in the event that the availability of soil nutrients is very low, the stock and reabsorption of nutrients in the xylem and phloem reservoirs will be low, altering the morphology of the plant (e.g., decreased growth and productivity, vessel size adjustments, trade-offs between sapwood area with leaf and root area) and physiology (e.g., decreased production of proline and enzyme synthesis) to withstand drought stress (St. Clair et al. 2008; Kreuzwieser and Gessler 2010; Gessler et al. 2017; Etienne et al. 2018; Salazar-Tortosa et al. 2018; Hevia et al. 2019; Durgante et al. 2023).
Regarding the vulnerability of Cedrela fissilis, the long-lived pioneer species (Peña-Claros et al. 2008) showed greater growth recovery, resilience and resistance to drought compared to other Cedrela species in the Peruvian Tropical Andean cloud forest (Rodríguez-Ramírez et al. 2022), providing xeric adaptation (Scholz et al. 2013). This characteristic may reinforce the idea of the species' ability to grow in marginal sites with suboptimal soil conditions and marked water deficit (e.g., seasonally dry forests) (Pereira et al. 2018), consistent with its wide distribution in Neotropical forests (Pennington and Muellner 2010; Barstow 2018; Siqueira et al. 2019). However, the species also shows poor hydraulic adjustment, which may suggest a higher risk of cavitation and/or embolism if the frequency or intensity of drought increases (Rodríguez-Ramírez et al. 2022). The hydraulic vulnerability of trees is also strongly influenced by maximum temperature and evapotranspiration (Rodríguez-Ramírez et al. 2022). Furthermore, in a population of C. fissilis in the southern Amazon forest, it was observed that warmer conditions during the wet season negatively affected xylem sulfur concentrations (Ortega Rodriguez et al. 2023a), nutrients that induce plant tolerance against high-temperature stress (Ihsan et al. 2019). However, to our knowledge, the degree of vulnerability to nutritional imbalances (modification of use and allocation of nutrients) associated with drought-induced Cedrela growth responses is unknown (see Villagra et al. 2013).
Here, we use dendrochemistry (Hietz et al. 2015) for an eco-nutritional approach. The inter-annual concentrations of nutrients in tree-rings of Cedrela fissilis Vell. (Meliaceae) of the southern Amazon region were used to assess how climate trends and droughts alter long- and short-term tree growth changes. We hypothesize that climate change and the increase in the frequency and intensity of extreme droughts alter long-term nutrient balances, affecting long-term growth patterns (Hevia et al. 2019). Specifically, we compared tree ring widths, wood density and nutrient concentrations in a drought-prone region of Amazonian forests (Agudelo et al. 2019). Our objectives were: (i) to assess long-term wood nutrient concentrations in C. fissilis; (ii) to investigate the relationships among wood traits (tree-ring width, wood density and nutrient concentrations) and climate inter-annual variability and trends; (iii) to investigate the effects of extreme drought, and the long- and short-term nutrient imbalances. Investigating the role of nutrient changes in the wood of trees from moist forest exposed to marked seasonality in water availability will improve our understanding of how tropical species will react to climate change and whether these forests and their productivity will be vulnerable to drought damage.
Methods
Study area
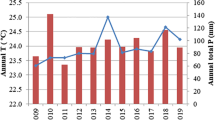
We sampled trees of C. fissilis in the Forest Management Unit (FMU) No. 12 (harvest plan 2019, area 2455.7 ha) of Jamari National Forest (JNF) under permanent forest management conducted by the logging firm AMATA. JNF is located in the southern Amazon basin, in the state of Rondônia (09°15′00″ S and 63°02′05″ W; MMA 2005). The area is classified as a moist forest with a well-defined dry season from June to August (with less than 50 mm of rain per month) and a wet season from September to May. The potential evapotranspiration exceeds precipitation from April to October. The average annual rainfall in this region is 2438 mm and the annual average temperature is 26 °C (Additional file 1: Fig. S1).
The soil is classified as Oxisol (Additional file 1: Fig. S1a) (Soil Survey Staff 2022), deep (1 to 2 m) to very deep (> 2 m) (MMA 2005), with a water retention capacity between 50% and 80% (Fengler et al. 2022). The soil has high acidity (pH < 4) and Al exchange (H + Al < 150 mmolc dm−3), and low natural fertility with a predominance of low base saturation (< 10%), cation exchange capacity (< 150 mmolc dm−3), Mg (< 1 mmolc dm−3), Ca (< 2 mmolc dm−3), K (< 0.8 mmolc dm−3) and P (< 8 mg dm−3) and soil organic matter (< 40 g kg−1) (Fengler et al. 2022).
Sampling and preparation
We selected 29 trees of C. fissilis felled and stored at log yards between April and May of 2019. Dominant trees > 30 cm diameter at breast height (DBH) were collected in a random sampling design. Wood discs of each tree were cut at the top level of the first log (6 m) (Granato-Souza et al. 2018). Four radial samples (10 cm width and 5 cm thickness) from each disc were cut, air dried and polished gradually with sandpaper (from 80 to 600 grains inch−2) for cross-dating and annual ring width (RW) measurement. Furthermore, a thin radial Sect. (2 cm width and 1.7 mm thickness) was cut transversely for each radial sample to analyze the ring wood density (RD, X-ray densitometry) and element content (micro X-ray fluorescence, μXRF).
Annual ring width measurements
Radial samples were cross-dated using skeleton plots and visual dating under the microscope (Stokes and Smiley 1996). Each cross section was scanned at 1200 dpi resolution with an Epson Expression 10000XL scanner, and the RW was measured with a resolution of 0.01 mm using CDendro and CooRecorder® software (Cybis Electronic 2013). The “Schulman Shift” convention (Schulman 1956) was not applied to the dating of the JNF series, which means that the tree growth of the period 2017/2018 was assigned to the calendar year 2018.
Wood density data collection
Thin radial sections from 29 trees were kept under constant temperature and humidity (20 °C and 60%, respectively) for 48 h in an atmosphere at 12% moisture content (Tomazello-Filho et al. 2008; Quintilhan et al. 2021). Wooden laths were then scanned, and X-rayed using an X-ray densitometry chamber (Faxitron MX20-DC12, Faxitron X-ray, Illinois, USA) with facilities at the Wood Anatomy and Tree-ring Laboratory, University of Sao Paulo, Brazil. X-ray images were then analyzed with WINDENDRO Density 2017a® software (Regent Instruments Inc., Quebec, Canada) to obtain the density profile with 15 μm intervals expressed in g cm−3.
Chemical data collection
The same thin radial sections of 14 trees used for density analyses were kept under controlled conditions (20 °C temperature and 50% humidity) and X-ray fluorescence micro-analyzed (μXRF) in an ItraxMultiscanner (Cox Analytical Systems, Mölndal, Sweden) at the laboratory of DendrOlavide, Universidad Pablo de Olavide (Seville, Spain).
Itrax was operated at 30 kV and 50 mA with a Cu-tube, and samples were exposed to the X-ray beam for 40 s at each measurement point in the radial direction (20-μm step size). Count rates of fluorescent photons of elements and a radiographic grayscale image at each measurement point of each sample were obtained. Peaks in the μXRF spectrum were assigned to specific elements using Q-spec software (Cox Analytical Systems, Mölndal, Sweden), producing relative concentrations (counts of fluorescent photons per second, cps) (Croudace et al. 2006) of those elements detected within the wood structure for every analyzed point. Radiographic images were synchronized with previously constructed ring width chronologies to corroborate the identification of the growth ring boundaries. Consequently, the element-specific count rates were assigned for each annual ring and the average annual time series for each radial section were produced using WinDendro™ (Regent Instruments Inc., Quebec, Canada).
In this study, we selected: (i) aluminum (Al), which is an indicator of soil acidification (Göransson and Eldhuset 1991; Cutter and Guyette 1993); (ii) phosphorus (P), an structural component of plant compounds and associated with the formation of younger xylem cells (Peterson and Anderson 1990); (iii) sulfur (S), essential to define the cellular architecture and play a role in the detoxification of potential toxic metals (Fairchild et al. 2009; Yadav 2010); (iv) potassium (K), involved in the cambial activity related to cell expansion (Fromm 2010); (v) calcium (Ca), involved in the cambial activity related to cell differentiation (Lautner and Fromm 2010); (vi) manganese (Mn), enzymatic function involved in amino acid and lignin synthesis (Pittman 2005) but important growth-limiting factor in acid soils (Kogelmann and Sharpe 2006) and competitive elements of Ca, K, P and Fe at the xylem cation exchange sites (McClenahen et al. 1989; Peterson and Anderson 1990); (vii) iron (Fe), essential for plant growth (meristematic cell division) and chlorophyll synthesis, although highly reactive and toxic under acid ambient conditions (Morrissey and Guerinot 2009); and (viii) strontium (Sr), despite not being an essential element, plants store it in the cell wall, because it behaves identically to Ca in many biogeochemical processes (Drouet and Herbauts 2008; Tagami and Uchida 2017; Burger and Lichtscheidl 2019). We also evaluated the ratios of aluminum to calcium (Al/Ca) (Kuang et al. 2008) and calcium to manganese (Ca/Mn) (Kogelmann and Sharpe 2006), reliable indicators of soil acidification; potassium to calcium (K/Ca), which is an indicator of cambial activity (Fromm 2010); strontium to calcium (Sr/Ca), associated with wood density (Silkin and Ekimova 2012) and manganese to sulfur (Mn/S), potential indicator of plant toxicity and stress associated to soil acidification (Neves et al. 2017).
Building tree-ring series
Cross-dating of the RW and RD series was evaluated using the COFECHA program (Holmes 1983) by comparing the consistency of the different ring width series using Pearson correlations at 30-year intervals. The annual nature of the tree rings and the robustness of the aligned mean series were verified and confirmed using radiocarbon “bomb-peak” dating (Santos et al. 2021). Fourteen radii, which presented the best correlation statistics with the mean site series of width and density, were selected to construct the chemical (Al, P, S, K, Ca Mn, Fe and Sr) chronologies.
Raw series were standardized and detrended to obtain comparable ring-width, -density and -chemical series of each tree and remove the age-, size-related trend and non-climatic signals using the ARSTAN program (Cook and Krusic 2005). A horizontal line function through the mean was initially applied only for the chemical series as an alternative for normalizing values. Then, ring-width, ring-density, and ring-chemical series were detrended by fitting a 30-year cubic spline function through the tree-ring series of each measured radius to retain high-frequency variability. We used a spline function, where the frequency response is 0.50 at a wavelength of 0.67 multiplied by the length of the series in years. Then, the ring data were divided by the fitted lines to obtain the standardized ring indices. All the wood trait chronologies were evaluated using dendrochronological statistics, including the mean correlation among all-time series (Rbar; Cook and Pederson 2011), the first-order autocorrelation (AC), the mean sensitivity (MS), and the Expressed Population Signal (EPS; Wigley et al. 1984).
Climate data set
Monthly precipitation and temperature data were obtained from (ANA, National Water Agency, Brazil; https://dadosabertos.ana.gov.br/) and INPE (National Institute of Space Research, Brazil; http://bancodedados.cptec.inpe.br/tabelaestacoes/faces/consultawmo.jsp). A regional mean precipitation (1975–2018) and temperature (1957–2018) series from five and six local meteorological stations, respectively, were created (Additional file 1: Table S1). The HOM and MET programs from Dendrochronology program library (DPL, Holmes 1994) were used to estimate missing data and calculate the regional time series. As a metric that expresses drought intensity, we used the difference between precipitation (P) and evapotranspiration (PET) (1975–2018) and the standardized precipitation evapotranspiration index (SPEI) (1950–2018). The PET and SPEI were calculated using the SPEI package (Vicente-Serrano et al. 2010) in R (R Core Team 2021).
Data analyses
To describe significant increasing or decreasing trends in raw series of RW, RD and z-scores of elements (i.e., the mean was subtracted from each value and divided by the standard deviation), we applied a linear regression t test for the period 1940–2018. To explore common long-term variance and associations among raw and detrended series of wood traits (RW, RD, elements and ratios), we applied principal component analysis (PCA) of the correlation matrix and Pearson's correlation.
We calculated Pearson correlation coefficients for the common periods 1975–2018 for local precipitation and P-PET and 1960–2018 for temperature and SPEI to quantify associations between wood traits and climate data. These correlations were calculated monthly from September of the previous year to August of the current year, which included the period of wood formation of C. fissilis close to the study region from prior September/October to current April/May, indicating a period of cambial dormancy from current June to August (period of leaf exchange) (Dünisch et al. 2003; Lobão 2011; Baker et al. 2017). We also calculated seasonal correlations considering the beginning of the rainy season (before September to November), middle of the rainy season (before December to current February), end of the rainy season (current March to May) and dry season (current June to August).
To compare the growth responses (RW and RD) and the chemical traces in tree-rings during dry and wet years, we used one-way ANOVA. The ten driest (1983, 1997, 2005, 2006, 2007, 2010, 2011, 2012, 2014, 2015) and wettest (1976, 1977, 1978, 1979, 1986, 1989, 1990, 2001, 2013, 2018) years were selected for the period 1975–2018 according to the ranking of normalized values of the P-PET (12 months, prior September to current September). The selected dry years are in accordance with the extreme drought events registered in the southern Amazon basin (Espinoza et al. 2019). To quantify the response of growth and the stability of element concentrations to extreme droughts during recent decades, we calculated the indices of resistance (Rt), recovery (Rc), resilience (Rs) and relative resilience (rRs = Rs–Rt) (Lloret et al. 2011). They were calculated using detrended data of wood traits (RW, RD, elements and ratios) instead of raw data to remove age and size effects and potential bias due to the eccentric growth of tropical species. The Rt index quantifies the difference between growth/chemical trace during the dry year and the preceding years, whereas the Rc index accounts for the growth/chemical trace reaction following the drought period. The Rs index quantifies the difference in growth/chemical traces before and after the dry year. Finally, the rRs index is the difference between Rs and Rt (cf. Sánchez-Salguero et al. 2013). To quantify these indices, we defined a period of 3 years before and after drought occurrence according to the previously applied criterion (e.g., Gazol et al. 2017). To calculate the pointer years and the resilience indices, we used the pointRes2.0 package (van der Maaten-Theunissen et al. 2021). Since extreme drought events in the Amazon region can last more than 1 year, the value of the pointer years was calculated through the average of the successive dry years. We selected the extreme drought events of 1981–1983, 1997–1998, 2005–2007, 2010–2011, and 2014–2015, which affected large areas of the southern Amazon basin (Ronchail et al. 2002; Williams et al. 2005; Marengo et al. 2008; Lovejoy and Nobre 2018; Espinoza et al. 2019; Caioni et al. 2020). Comparisons between wood trait responses during extreme droughts were assessed by one-way ANOVA and the increasing or decreasing trends of these responses over the years were evaluated by a linear regression t test.
Results
Growth and nutrient series and their associations
The descriptive statistics of all standardized chronologies are shown in Table 1. The RW and RD chronologies were based on 69% and 52% of all sampled C. fissilis trees, respectively. They showed a significant mean correlation among the series (Rbar = 0.31 and 0.29, respectively) and presented an EPS that was statistically reliable for the period 1901–2018 (EPS > 0.85). The chemical chronologies presented a significant mean correlation among the series based on 54% (Al, Rbar = 0.21), 45% (P, Rbar = 0.23), 63% (S, Rbar = 0.30), 45% (K, Rbar = 0.58), 36% (Ca, Rbar = 0.26), 54% (Mn, Rbar = 0.30), 45% (Fe, Rbar = 0.25), 45% (Sr, Rbar = 0.35), 36% (Al/Ca, Rbar = 0.28), 36% (Ca/Mn, Rbar = 0.33), 54% (K/Ca, Rbar = 0.25), 45% (Sr/Ca, Rbar = 0.32) and 45% (Mn/S, Rbar = 0.44) of all trees selected for chemical analysis. Only the K and Mn/S chronologies presented an EPS > 0.85 for the period 1901–2018.
The correlation between raw data of C. fissilis tree-ring parameters showed a significant positive and negative association of almost all the elements with the RW (Al, S, K, Ca, Mn and Sr; positive loading in PC1) and RD (Al, S, K, Ca, Mn, Sr, Al/Ca, K/Ca, Sr/Ca; negative loading in PC1), respectively. The raw data of Ca/Mn and Mn/S ratios (negative loading in PC1) showed a significant negative association with RW and all other chemical series but a positive association with RD (Fig. 1A, B). The first principal component for raw data explained 57% of the total variance, and the second axis explained an additional 12% (Fig. 1A). After detrending, a significant positive correlation between RW and RD was observed (positive loading in PC2). Significant positive correlations were maintained between K (negative loading in PC2) and Ca and K/Ca; Al/Ca (positive loading in PC1) and Al; P (positive loading in PC1) and Al/Ca, K/Ca and Sr/Ca; and Sr/Ca (positive loading in PC1) and Sr, Al/Ca and K/Ca. Significant negative correlations were maintained between Ca/Mn (negative loading in PC1) and P, Al/Ca, K/Ca and Sr/Ca (Fig. 1C, D). The first principal component for detrended data explained 18% of the total variance and 12% the second one (Fig. 1C).
a, c Principal component analysis of years (dots) and b, d Pearson’s correlation of raw (a, b) and standardized (c, d) tree-ring parameters of Cedrela fissilis. Raw data (tree-ring in mm, tree-ring density in g cm−3, and z-scores of elements) and residual index of tree-ring width (RW), wood density (RD) together with series of aluminum (Al), phosphorus (P), Sulfur (S), potassium (K), calcium (Ca), manganese (Mn), iron (Fe), strontium (Sr), aluminum to calcium (Al/Ca), calcium to manganese (Ca/Mn), potassium to calcium (K/Ca), strontium to calcium (Sr/Ca) and manganese to sulfur (Mn/S) are projected in a reduced dimension space defined by principal components PC1 (x axes) and PC2 (y axes). The PCA and correlation were computed over the 1940–2018 period
Long- and short-term patterns of growth and nutrients
Raw data of RW together with z-scores of all elements (Al, P, S, K, Ca, Mn, Fe and Sr) and the molar ratios Al/Ca, K/Ca and Sr/Ca, while RD together with the molar ratios Ca/Mn and Mn/S showed similar long-term trends (Fig. 2). According to the fitted linear regression model, the RW (and Al, P, S, K, Ca, Mn, Fe, Sr, Al/Ca, K/Ca and Sr/Ca) decreased, while the RD (and Ca/Mn and Mn/S) increased between 1940 and 2018. The long-term trends of tree-ring parameters are not affected by the sapwood–heartwood transition zone (2002 ± 6), although the Fe trend goes from increasing until 1984 and then decreasing until 2018, with a change occurring before the early to heartwood transition zone.
Annual values and fitted (red lines) long-term trends of ring width (RW), wood density (RD), z-scores of element concentration and molar ratios of C. fissilis trees. Z-scores of aluminum (Al), phosphorus (P), sulfur (S), potassium (K), calcium (Ca), manganese (Mn), iron (Fe), strontium (Sr) and molar ratios of aluminum to calcium (Al/Ca), calcium to manganese (Ca/Mn), potassium to calcium (K/Ca), strontium to calcium (Sr/Ca) and manganese to sulfur (Mn/S). Vertical light red boxes represent extreme drought events registered in the southern Amazon basin in 1981–1983, 1997–1998, 2005–2007, 2010–2011, and 2014–2015 (Espinoza et al. 2019). The vertical green dashed line and error interval indicate the average year of sapwood–heartwood transition (2002 ± 6)
Normalized P-PET showed an increase during the dry years of the recent decades, mainly from 2000 onward (Fig. 3). A significant (p < 0.05) decrease in the tree-ring width and wood density together with the concentrations of Al, S and Ca was observed in the dry years compared with the wet years. In dry years, the concentrations of P also decreased, and those of K and Fe increased (significant, p < 0.1) compared with the values in wet years (Fig. 3). The Mn, Sr and Al/Ca, Ca/Mn, K/Ca, Sr/Ca and Mn/S ratios did not show significant differences between dry and wet years (Fig. 3, Additional file 1: Fig. S2).
Mean index residual chronologies of tree-ring width (RW), wood density (RD) and element concentration of aluminum (Al), phosphorus (P), sulfur (S), potassium (K), calcium (Ca), manganese (Mn), iron (Fe), and strontium (Sr) from C. fissilis trees from southern Amazon and the total annual difference between precipitation and potential evapotranspiration (P-PET, time-scales from prior September to current September). Vertical light red boxes represent extreme drought events registered in the southern Amazon basin in 1981–1983, 1997–1998, 2005–2007, 2010–2011, 2014–2015 (Espinoza et al. 2019). On the right is the boxplot of normalized parameters in dry and wet years; a significant difference (SD) between means is also presented (p < 0.05)
Climate–growth–nutrient relationships
The correlation between C. fissilis tree-ring parameters and precipitation showed that the local rainy season positively affected tree growth and density (RW, r = 0.29; RD, r = 0.29), particularly in March. The rainy season also positively affected the element concentrations of Al (in September, r = 0.31), S (in October, r = 0.33), Ca (in November, r = 0.44) and Sr (in March, r = 0.34). Concentrations of Al, P and S (particularly in July) presented positive correlations (r > 0.29) with precipitation in the dry season (Fig. 4). Warmer temperatures during the rainy season (mainly between January and April) and the beginning of the dry season (mainly between June and July) negatively (r < − 0.28) affected the P and S concentrations. Warmer months during the rainy season positively affected growth (RW in October, r = 0.28), K concentration (in May, r = 0.27) and Ca/Mn (in February, r = 0.37). Warmer months during the dry season positively affected Ca (in July, r = 0.26) and Ca/Mn (in June, r = 0.26).
Correlation values between mean index residual chronologies of tree-ring width (RW), wood density (RD), element concentrations of aluminum (Al), phosphorus (P), sulfur (S), potassium (K), calcium (Ca), manganese (Mn), iron (Fe) and strontium (Sr) and molar ratios of aluminum to calcium (Al/Ca), calcium to manganese (Ca/Mn), potassium to calcium (K/Ca), strontium to calcium (Sr/Ca) and manganese to sulfur (Mn/S) of C. fissilis trees with monthly and seasonal precipitation (period 1975–2018), temperature (period 1960–2018), the difference between precipitation and potential evapotranspiration (P-PET, period 1975–2018) and the standardized precipitation–evapotranspiration index (SPEI, period 1960–2018) from the southern Amazon. The green shading includes the rainy months for the study region and gray shading shows the seasonal correlations
The correlations between P-PET and tree-ring parameters showed that months of low evapotranspiration in the rainy season positively affected tree growth (RW in March, r = 30) and concentrations of Al (in September, r = 0.31), P (in January, r = 0.30), S (in October, r = 0.31), Ca (in November, r = 0.43) and Sr (in March, r = 0.32), and negatively affected K (between March and May, r = − 0.29) and K/Ca (in March, r = − 0.47) (Fig. 4). Low evapotranspiration in the dry season positively affected Al, P and S (r > 0.30). Regarding the SPEI drought monitor, low evapotranspiration in the dry season positively affected P (in June, r = 0.24) and S (in August, r = 0.26). In contrast, low evapotranspiration in the rainy season negatively affected Mn, Sr (in February, r = − 0.24) and Fe (in October, r = − 0.28).
Drought–growth–nutrients resilience
Growth (RW) and wood density (RD) resistance for C. fissilis increased with time (from 1981–1983 to 2014–2015 droughts), and a similar increasing trend was observed for the resistance explained by the concentrations of Al, Ca, P, Ca/Mn and Mn/S, unlike S, K, Mn, Fe, Sr, Al/Ca, K/Ca, and Sr/Ca, which decreased (Fig. 5, Additional file 1: Figs. S3, S4). Growth and wood density resilience increased with time, and similar increasing trends of resilience were observed for Al, S, Ca, Mn, Sr and Ca/Mn, unlike K, P, Fe, Al/Ca, K/Ca, Sr/Ca and Mn/S, which decreased. A significant positive correlation between resistance and resilience of growth and wood density to years of droughts was observed. Furthermore, the resilience explained by Sr concentration presented a significant positive correlation with the resilience of growth and wood density (Additional file 1: Fig. S5). A significant positive correlation was also observed between the resistance of Mn and Sr and the resilience of Al/Ca and K/Ca. A significant negative correlation was observed between Ca/Mn and Sr/Ca resistance and the resilience of Mn and Fe, Mn and Mn/S, and S and Sr/Ca (Additional file 1: Fig. S5). The highest growth and wood density resistance and resilience (together with S- and Ca/Mn-resilience) were observed in the 2010–2011 drought. The lowest growth resistance was registered in the 2005–2007 drought, while the lowest wood density resistance and the lowest growth and wood density resilience (together with S- and Ca/Mn-resilience) were registered in the 1981–1983 drought (Fig. 5, Additional file 1: Figs. S3, S4).
Boxplots showing standardized ring width (RW), wood density (RD), concentrations of aluminum (Al), sulfur (S), potassium (K), calcium (Ca) and calcium to manganese ratio (Ca/Mn) stability during extreme droughts from C. fissilis trees from the southern Amazon. Different letters indicate significant differences (p < 0.05) between droughts. In the box plots, the boundary indicates the 25th percentile, a solid line within the box marks the median, an asterisk within the box marks the mean, and the upper boundary of the box indicates the 75th percentile. Whiskers above and below the box indicate the 10th and 90th percentiles. Blue lines show the relations between tree-ring parameters and years of extreme droughts, and Pearson’s correlation coefficient is presented at the bottom. Extreme drought events in 1981–1983, 1997–1998, 2005–2007, 2010–2011, and 2014–2015 in the southern Amazon basin were selected (Espinoza et al. 2019)
Growth (similar to S, Mn, Sr) recovery and relative resilience increased with time, unlike wood density (similar to Al, K, Ca, P, Fe, Ca/Mn, Al/Ca, K/Ca, Sr/Ca and Mn/S) recovery and relative resilience which decreased (Fig. 5, Additional file 1: Figs. S3, S4). The recovery and relative resilience of growth and wood density presented significant negative correlations with the recovery and the relative resilience explained by the P concentration and Sr concentration, respectively (Additional file 1: Fig. S5). A significant positive correlation was observed between the recovery of S and Mn and K and Sr/Ca and the relative resilience of S and Mn. A significant negative correlation was observed between recovery and the relative resilience of S and K/Ca, S and Sr/Ca, Mn and Al/Ca and Mn and K/Ca (Additional file 1: Fig. S5). The highest growth recovery and relative resilience were observed in the 2014–2015 drought, while the lowest growth recovery (together with Sr-recovery) and relative resilience (together with Sr-relative resilience) were observed in the 1981–1983 drought. The highest wood density recovery (together with Ca-, Al/Ca- and K/Ca-recovery) and relative resilience (together with Ca-, Al/Ca- and K/Ca-relative resilience) were observed in the 1981–1983 drought, while the lowest wood density recovery (together with K/Ca-recovery) and relative resilience (together with K/Ca-relative resilience) were observed in the 2010–2011 drought (Fig. 5, Additional file 1: Figs. S3, S4).
Discussion
Growth and nutrient changes
Significant long- and short-term growth and nutrient imbalances of C. fissilis were observed, linked to the increased frequency of dry years in recent decades (Figs. 2, 3, Additional file 1: Figs. S1, S2). The alterations in the soil water regime and nutrient bioavailability linked to hydroclimate trend alterations cause a reduction in the physiological responses of trees, inhibit photosynthesis and alter carbohydrate and amino acid metabolism (Gavrilescu 2021; Malhi et al. 2021). These physiological and metabolic alterations reduce tree productivity (in most cases) (Schlesinger et al. 2016) and produce a long-term trade-off between growth and wood density (Bergès et al. 2000; Bouriaud et al. 2004; Bontemps et al. 2013; Vannoppen et al. 2018), as was observed for C. fissilis, which presented density increases (r = 0.70, p < 0.05) and growth decreases (r = − 0.35, p < 0.05) since 1940 (Fig. 2, Ortega Rodriguez et al. 2023a).
The decrease in C. fissilis growth over time has been previously reported in trees from forests with marked climatic seasonality that suffer a historical increase in extreme drought (Feeley et al. 2007; Mendivelso et al. 2014, 2016; Islam et al. 2019). The severity of these droughts is related to an increase in the intensity of the dry season and a delay in the onset of the rainy season in the southern Amazon region (Marengo et al. 2018; Espinoza et al. 2019). This region also shows a significant negative trend in SPEI values (r = − 0.65, p < 0.01), P-PET values (r = − 0.39, p < 0.05), and precipitation (r = − 0.28, p < 0.05) and a significant positive trend in temperature since 1970 (r = 0.85, p < 0.001) (Additional file 1: Fig. S1), which reinforces the idea of changes in the water regime in recent decades.
Regarding the wood density trends, C. fissilis is a deciduous long-lived pioneer species (Peña-Claros et al. 2008), and its radial wood density gradient is expected to increase in mature stages of development to generate stem stability (Nock et al. 2009). This explanation could satisfy the 5.62% gradual increase in wood density of C. fissilis trees in the JNF between 1940 and 1999 (Fig. 2). However, between 2000 and 2018 (sapwood region), the trend of wood density began to increase gradually by 14.11%, coinciding with the increase in the frequency of dry years. The sapwood region of C. fissilis presents lower wood density values due to the higher number of empty spaces (active vessels) through which water is transported (Mendivelso et al. 2013). However, positive associations between the increase of drought events and the wood density of the species have been observed in recent decades (Mendivelso et al. 2016; Ortega Rodriguez et al. 2023b). Another possible explanation for this positive wood density trend over time could be linked to atmospheric CO2 increases (NOAA 2021); however, there is insufficient evidence that this relationship is cause and effect for all angiosperms (Yazaki et al. 2005; Arsić et al. 2021).
Contrary to our expectations, we did not observe an increase over time in the Ca xylem concentration trend (or analogous elements, such as Sr or Mn) despite the wood density (Ortega Rodriguez et al. 2022) or local temperature increase (Sánchez-Salguero et al. 2019). Even mobile elements such as P, K, and S were expected to increase in the sapwood and close to the bark of C. fissilis trees (Ortega Rodriguez et al. 2022) due to strong symplastic transport (through plasmodesmata) in cambial regions of active growth (Smith et al. 2014; Hietz et al. 2015). P, K, and S presented a decreasing trend in their concentrations associated with growth decrease in the pith to bark direction (Fig. 1A, B). Our results suggest that alterations in the local water regime directly influenced all nutrient concentrations in the xylem tissues, mainly since 1980 (p < 0.05: Ca, r = − 0.49; K, r = − 0.84; P, r = − 0.64; Mn, r = − 0.81; S, r = − 0.88; Sr, r = − 0.78, Al, r = − 0.54, and p < 0.10: Fe, r = − 0.12). When soil water reserves decline during prolonged drought, microbial activity is inhibited, and diffuse and mass flow uptake between roots and soil decreases; likewise, the total water potential gradient between root, xylem, and leaf cells results in lower nutrient concentrations in tree tissues (He and Dijkstra 2014; Schlesinger et al. 2016; Gessler et al. 2017; Gavrilescu 2021).
Regarding element ratios, drought may also modify cross-talk between nutrients (uptake and storage) and hence modify leaf demands with potential consequences in storage organs (Etienne et al. 2018). The Ca/Mn increases and Al/Ca decreases over time suggesting a decrease in soil acidity and an increase in the effective rates of cation mobilization (e.g., Ca, Mg, Fe, Mn and Zn) (Kogelmann and Sharpe 2006; Kuang et al. 2008). Increases in temperature and droughts are associated with increased pH in arid soils (Jiao et al. 2016), and possibly in acid soils. This facilitates the acropetal mobility of cations through the xylem, which is fast in Ca and slow in Mn (Etienne et al. 2018) and Al (Shen and Ma 2001). However, with extreme droughts, a deficit in water plus nutrient (soil solution) flow is generated, which appears first in the new leaves, inducing their senescence and remobilizing the nutrients from the senescent organs to the reserve organs (Etienne et al. 2018). In this process, Ca (available above pH 6.5) is not affected as much as Mn (Etienne et al. 2018) and Al (both available below pH 5.5), so its proportion will be higher in the respective ratios of xylem tissues.
An increase in Mn/S was also observed with time. Although the improvement in soil pH (with increases in temperature and drought) suggests improvements in S-soil availability (Etienne et al. 2018), the water deficit caused by this same stress decreases the basipetal leaf-xylem S mobility and xylem storage (Ihsan et al. 2019). These cellular spaces left by S could be occupied by Mn when acropetal mobility occurs and could generate toxicity due to the decrease in water-soluble nonprotein thiols (-SH, functional group), which become defense mechanisms when cellular Mn concentrations increase (Neves et al. 2017). This has been studied in Mn phytotoxicity experiments, although neither the water deficit nor the temperature were controlled (Neves et al. 2017).
Contrary to what we expected, the K/Ca and Sr/Ca ratios presented an opposite trend to density over time (Fig. 2). According to Fromm (2010), in situations of a low K regime (frequent dry periods), the secondary cell walls begin their formation earlier (fibrillary phase), apparently forming a cell wall with greater volume; in contrast, the process of cell division and formation of the primary cell wall (amorphous phase) decreases. Furthermore, according to Silkin and Ekimova (2012), Ca distributed in the primary cell wall and the middle lamella is proportional to the cell cross-sectional area, while Sr uniformly distributed along the cell wall is proportional to the cell wall cross-sectional area. Thus, the historical increase in density was expected to imply an increase in the K/Ca and Sr/Ca ratios. However, it is possible that, similar to Mn and Al cations, K and Sr fluxes in the tree are also affected more than Ca under water deficit. Acropetal and basipetal mobility (both in the xylem and phloem) of K (Carvalho et al. 2005) imply their frequent distribution from older to younger tissues, and their re-translocation to permanent organs. Furthermore, K and Ca reduce Sr uptake (Isermann 1981) in acropetal motility pathways (Feller et al. 2015). With water deficit, deficient Sr (Østmoe 2020) and K (Armstrong and Kirkby 1979) mobility occurs in the phloem, so its reallocation toward the xylem and the root will be less, while Ca is not affected.
During dry years (negative values of P-PET) of the period 1970–2018, C. fissilis showed narrower (RW) and less dense (RD) rings, lower Al, S, Ca (p < 0.05) and P (p < 0.10), and higher K (p < 0.10) concentrations (the opposite was found in wet years) (Figs. 1D, 3). The positive relationship between dry years and narrower rings has been previously reported for Cedrela species (Villalba et al. 1992; Dünisch et al. 2003; Dünisch 2005; Lobão 2011; Pereyra Espinoza et al. 2014; Paredes-Villanueva et al. 2016; Granato-Souza et al. 2018b, a; Pereira et al. 2018; Lisi et al. 2020; Humanes-Fuente et al. 2020; Rodríguez-Ramírez et al. 2022). The positive tree-ring width-wood density relationship of C. fissilis was also observed in other semiring porosity tree species in both tropical (Quintilhan et al. 2021) and temperate forests (Bergès et al. 2000; Toïgo et al. 2015; Vannoppen et al. 2018). The ring early- and latewood widths and their respective densities in C. fissilis also presented positive relationships (Ortega Rodriguez et al. 2023b).
The positive relationship between the ring width and density of C. fissilis suggests that the species quickly stops or strongly limits its radial growth together with the cessation of the wood densification process as soon as a soil water deficit occurs (Bouriaud et al. 2004). This compromises the hydraulic safety (risk of capitation or embolism) of C. fissilis, whose 52% of the ring width (earlywood) is formed mainly by the combination of large vessels and marginal parenchyma (Ortega Rodriguez et al. 2023b). In extremely dry situations, the species can buffer its vulnerability to cavitation by losing foliage (Mendivelso et al. 2016), developing vessels with smaller diameter size (Islam et al. 2019; Rodríguez-Ramírez et al. 2022) as a strategy to resist water flow, which reduces the amount of water lost through transpiration. However, as already observed by Rodríguez-Ramírez et al. (2022), this strategy may be compromised by the poor hydraulic adjustment of the species due to the predominance of large vessels.
In dry tropical tree taxa (such as Cedrela), only wood mass invested in fiber walls contributes to hydraulic security (Janssen et al. 2019). This invested mass in cell wall thickening can result in decreased cell division and enlargement (Bouriaud et al. 2004). Fiber wall thickening may help surrounding vessels resist partial implosion and prevent stretching or rupture of pit membranes and microfractures in vessel walls, thereby reducing the risk of embolism (Jacobsen et al. 2005). The thickening of fiber walls and the decrease in cell division may be related to the higher K and lower Ca concentrations in dry years, respectively. K has been shown to be a mandatory factor for the cell wall thickening of xylem cells in trees (Wind et al. 2004; Fromm 2010). High levels of K favor the osmotic potential of the cambium, widening it and increasing the fiber and vessel size in the xylem (Fromm 2010). The vessel-associated radial (and possibly axial) parenchyma plays an important role in K recycling within the stem (Fromm 2010) and facilitates K flux through the plasma membrane H+-ATPase into the vessels (Arend et al. 2004). On the other hand, Ca is mainly deposited in the apoplast, where it binds to various cell wall sites, offering rigidity to developing cell walls in the course of xylem differentiation (Lautner and Fromm 2010). Ca deficiency in the xylem not only causes a decrease in cambium activation and cell differentiation (Follet-Gueye et al. 1998; Lautner and Fromm 2010) but also decreases lignification and the crosslinking of carboxyl groups within the pectin layer, decreasing the hardness and elasticity of the wood (Wimmer and Lucas 1997).
Narrow rings of dry years also showed lower concentrations of macronutrients, such as P and S, and Al, which is not considered an essential element. P plays a role in photosynthetic, respiratory, nutrient synthesis, and plant growth processes (Vance et al. 2003). In some Amazonian Ultisols and Oxisols, symbiotic activity between roots and mycorrhizae increases P availability by making more P available than is fixed by iron and aluminum oxides (Alexander 1989; Hu and Schmidhalter 2005; Quesada and Lloyd 2016). However, under dry soil conditions, mycorrhizal activity decreases, decreasing the P uptake of trees (Hu and Schmidhalter 2005). By decreasing the water flow and P, the meristematic activity decreases, interrupting the formation of young leaves and apical and cambial growth (Kurita et al. 2022). Low P availability can also alter the re-translocation of the element from the meristematic regions to the phloem and xylem parenchyma cells in the senesce period (Kurita et al. 2022).
S plays a role in plant growth, development processes and redox regulation, as it is a component of amino acids (e.g., methionine, cystine and cysteine) in proteins (Herschbach and Rennenberg 2001; Abrol and Ahmad 2003; Fairchild et al. 2009). It is also a component of metabolites (e.g., glutathione and phytochelatins), cofactors of modulating enzymes of effective defense mechanisms to encounter plant stress (Khan et al. 2014; Ihsan et al. 2019). Trees acquire the required S mainly through the uptake of sulfate by nonmycorrhizal and mycorrhizal roots and some amount from sulfur dioxide in the atmosphere by photosynthetically active tissues (stomata) of mature leaves (Herschbach and Rennenberg 2001). Drought stress causes a decrease in S uptake by roots and its allocation to leaves (Herschbach and Rennenberg 2001; Abrol and Ahmad 2003). The leaf loss of C. fissilis as a strategy to overcome water stress (Mendivelso et al. 2016) decreases the leaf production of reduced S and simultaneously decreases its storage in the parenchyma cells of the phloem and xylem, as well as ray and pit cells (Herschbach and Rennenberg 2001).
Although Al is highly rhizotoxic and is a major stress factor to plants on acid tropical soils, many native trees are well-adapted to high Al conditions (Brunner and Sperisen 2013). Al-oxalate processed in root cells is converted to Al-citrate in the xylem and translocated to leaf cells for reconversion to Al-oxalate (Shen and Ma 2001). Al is mainly stored in the cell wall of the leaves and roots (Brunner and Sperisen 2013). The uptake of water and nutrients by plants under stress conditions (e.g., drought, high temperatures, salinity and nutrient deficiency) increases due to a greater root surface area induced by Al (Bojórquez-Quintal et al. 2017). Drought stress changes the porosity of the cell wall structure (reduce the space between cellulose, xyloglucan, pectin and structure protein) of roots and reduces the accumulation of Al and its effect on plants (Bojórquez-Quintal et al. 2017).
Climate–growth–nutrient relationships
Our results showed that the radial wood formation (RW, significant positive correlation in March) and wood density levels (RD, significant positive correlation between March and May) of C. fissilis starts at the beginning of the rainy season in September/October and is completed at the end of the rainy season in April/May, similar to other Cedrela species in the southern Amazon region (Dünisch et al. 2003; Brienen and Zuidema 2005; Lobão 2011). Furthermore, when C. fissilis begins to produce the marginal parenchyma between the end of the dry season and the beginning of the rainy season (Marcati et al. 2006), the initial promoter of root and shoot activity will be Al (mainly in September) (Ka et al. 2008; Bojórquez-Quintal et al. 2017), inducing the absorption of nutrients, such as P (Tomioka et al. 2005), Mg (Bose et al. 2013) K and Ca (Bojorquez-Quintal et al. 2014). Then, the formation of large vessels of the earlywood together with or even before the flushing of new leaves (Lobão 2011) will generate conductive pathways for the acropetal mobility of S, Ca and P (mainly in October, November and January, respectively) through the sap flux between the roots and the new leaves in the months of low evapotranspiration in the rainy season (Mcelrone et al. 2013). The phosphate and sulfate transport systems initiate coordinated operations for the physiological functioning of the tree (Smith 2001), promoting the formation of young xylem cells (Peterson and Anderson 1990) and defining its cellular architecture (Fairchild et al. 2009), respectively. Furthermore, Ca stimulates cambial reactivation and cell division at the beginning of the growing season, between September and November (Lautner and Fromm 2010).
Low evapotranspiration in the rainy season will also positively affect Sr storage (mainly in March), a nonessential element analogous to Ca (Burger and Lichtscheidl 2019) while negatively affecting the xylem storage of Mn and Fe (mainly in February and October, respectively). Mn and Fe, elements with low mobility, are mainly used in the leaf for respiratory processes (Glavac et al. 1990; Morrissey and Guerinot 2009) and, as in the case of Mn, can increase their levels in the xylem sap immediately before and after leaf emergence (Glavac et al. 1990). The months of low evapotranspiration in the rainy season also negatively affect the K concentrations and K/Ca (mainly between March and May). This apparently happens, because under normal conditions of water availability, C. fissilis prioritizes cambial activity based on cell division and vessel enlargement (positive relationship with Ca) and not thickening of the cell wall (negative relationship with K) (Fromm 2010).
The rainfall and low evapotranspiration during the dry period (between June and August) also influenced the Al, P and S storage. This can reinforce the idea of the high stimulation for nutrient uptake of roots by Al (Bojórquez-Quintal et al. 2017), as well as the activity of P and S in the transport systems and nutrient redistribution in the tree (Smith 2001). Furthermore, this positive relationship of P and S concentrations with precipitation in the dry period suggests that when the demand for these nutrients decreases in the young leaves, P (Kurita et al. 2022) and S (Hartmann et al. 2000; Herschbach and Rennenberg 2001) are re-translocated (basipetal mobility) to xylem storage tissues (marginal parenchyma, Ortega Rodriguez et al. 2022) to be used in the following growing season when sap flux increases again.
Our study also reported a strong negative association between P and S records and temperature during the wet seasons (mainly between January and April) and during June and July of the dry season (Fig. 4). It is possible that with high temperatures, there is an increase in S translocation from the xylem to the growing parts of the shoot and discharge on the leaves to play a defense function against heat stress (Herschbach and Rennenberg 2001; Ihsan et al. 2019). S in its glutathione (GSH) form is triggered by heat and drought stress and, upon oxidation, maintains optimal biochemical and redox cellular functions within plant cells (Ihsan et al. 2019). Regarding P, the leaves and young shoots are its main sink (Kavka and Polle 2016), and its xylem concentration is more affected than S by seasonal activity (Barrelet et al. 2006). During warmer temperatures, tree transpiration increases (Will et al. 2013), suggesting an acceleration in P plus water flux toward the tree apex (Kurita et al. 2022), decreasing xylem P storage. Under these conditions, the regulation of P transport will depend on the species adaptations of vascular structures (Raghothama 1999; Lin et al. 2014; Zavišić and Polle 2018).
Growth–nutrient resilience under extreme droughts
C. fissilis showed an increase in growth and wood density resistance and resilience over time (from 1981–1983 to 2014–2015 droughts) (Fig. 5) as a consequence of the delayed onset of the rainy season that increased the water deficit in the southern Amazon region (Marengo et al. 2018). The increase over time in the capacity of C. fissilis trees to absorb drought stress and continue growing during the dry years (resistance) was also associated with increases in resistance explained by the concentrations of Al, Ca, P, Ca/Mn and Mn/S and decreases in S, K, Mn, Fe, Sr, Al/Ca, K/Ca and Sr/Ca (Fig. 5, Additional file 1: Figs. S3, S4, S5). In the context of decreased soil acidity conditions (Ca/Mn increases and Al/Ca decreases), C. fissilis trees have an adaptation strategy improving their root system, potentiated by Al uptake, which facilitates the elongation of roots and proliferation of lateral roots both in the short and long term (Ghanati et al. 2005; Bojórquez-Quintal et al. 2017). The increases over time in the resistance associated with P and Ca uptake reinforce the idea of a cambial activity process prioritizing vascular regulation (Lautner and Fromm 2010) developing a greater number of smaller vessels (Rodríguez-Ramírez et al. 2022) and probably more elongated vessels (Scholz et al. 2013), which are necessary for the flow of phosphates that regulate carboxylation in photosynthesis (Raaimakers et al. 1995). Furthermore, the decreases over time in resistance associated with K uptake and K/Ca ratios reinforce the species' initial strategy of prioritizing the formation of many small vessels to the detriment of thickening its cell wall. On the other hand, elements related to the physiological activities of the leaf, such as S, which is involved in defense against heat stress (Ihsan et al. 2019), and Fe and Mn, which are involved in the respiration process (Glavac et al. 1990; Morrissey and Guerinot 2009), decrease over time. This may be associated with the leaf loss strategy of C. fissilis (Mendivelso et al. 2016).
Our results also showed that the recovery process in growth over time increases (associated with the recovery capacity explained by S, Mn and Sr) and the wood density decreases (associated with the recovery capacity explained by Al, K, Ca, P, Fe, Ca/Mn, Al/Ca, K/Ca, Sr/Ca, Mn/S). The increase over time in the capacity of trees to recover in terms of growth rates and wood density observed before the drought event (resilience) was also associated with increases in resilience explained by Al, S, Ca, Mn, Sr and Ca/Mn and decreases in K, P, Fe, Al/Ca, K/Ca, Sr/Ca and Mn/S (Figs. 5, Additional file 1: Figs. S3, S4, S5). This suggests that for recovery processes over time, both growth and density, requires sources of S. As previously mentioned, S seems to be one of the essential elements in containing the stress caused by dry conditions (Ihsan et al. 2019), and its storage and re-translocation to the regions of meristematic activity will guarantee the resumption of physiological processes toward normal conditions (Herschbach and Rennenberg 2001; Fairchild et al. 2009; Ihsan et al. 2019). However, there is a risk that the species will not return to the density values prior to drought, which is associated with a deficit in the functions performed by key elements in promoting tree nutrition such as Al (Bojórquez-Quintal et al. 2017), cambial activity such as K and Ca (Fromm 2010) and xylem cell formation such as P (Peterson and Anderson 1990) and Fe (Morrissey and Guerinot 2009). This risk may be even more serious, because C. fissilis records the effects of drought stress up to 24 months after the extreme event, making the species demand accelerated compensation strategies between growth and wood density (Tao et al. 2022; Ortega Rodriguez et al. 2023b).
Conclusions
The eco-nutritional evidence obtained in this study showed that C. fissilis is undergoing alterations in the concentration, use and redistribution of nutrients associated with the wood density increase and growth decrease over time. This is due to the decrease in the water availability generated by the increase in the frequency of drought spells in the southern Amazon. Alterations in the concentration of nutrients in the xylem pointed to adaptations in (i) the roots due to a decrease in acidity of soil and water flow (e.g., Al, Ca/Mn and Al/Ca); (ii) cambial activity and vascular regulation as part of the ring-width and density trade-off (Ca, K, P, S, Sr, K/Ca and Sr/Ca); (iii) the formation, function and senescence of shoots and leaves as part of the first response to tree stress (S, Fe, Mn and Mn/S). The species presents a tendency to increase its capacity for resistance, recovery and resilience in growth over time, associated with responses in the capacity to use Al (growth promoter and root function), Ca, P (xylematic growth activators) and S (essential for the reduction of drought stress). However, there is a risk in the capacity for recovery of the normal pre-drought density values, associated with unsatisfactory responses in the capacity to use Al (promoter of tree nutrition), Ca, K (essential for cambial activity and cell wall structure), Fe and P (essential in the formation and architecture of the xylem cells). This study is one of the first attempts to clarify aspects of climate sensitivity and drought resilience from a nutritional approach based on long-term annual data obtained by dendrochemical techniques in Amazon dry seasonal moist forests. Future works should relate the chemical traces in the xylem with the proportion of anatomical elements, hydraulic adjustments and vulnerability of the vessels to verify the nutritional demands related to the plasticity of anatomical wood traits of C. fissilis.
Availability of data and materials
The data from this article will be incorporated into the ITRDB BRA015 (https://www.ncei.noaa.gov/access/paleo-search/study/ 37,447) as a complement to the chronologies already registered. Analysis data will be available on request. Supplementary material to this article can be found in Additional file 1.
Abbreviations
- AC:
-
First-order autocorrelation
- cps:
-
Counts per second
- DBH:
-
Diameter at breast height
- EPS:
-
Expressed population signal
- GSH:
-
Glutathione
- H:
-
Tree height
- JNF:
-
Jamari National Forest
- MS:
-
Mean sensitivity
- P-PET:
-
Difference between precipitation and potential evapotranspiration
- PCA:
-
Principal component analysis
- RW:
-
Ring width
- RD:
-
Ring density
- SG:
-
Specific gravity
- SPEI:
-
Standardized precipitation–evapotranspiration index
- µXRF:
-
X-ray fluorescence micro-analysis
References
Abrol Y, Ahmad A (2003) Sulfur in plants. Kluwer Academic Publishers, Alphen aan den Rijn
Agudelo J, Arias PA, Vieira SC, Martínez JA (2019) Influence of longer dry seasons in the Southern Amazon on patterns of water vapor transport over northern South America and the Caribbean. Clim Dyn 52:2647–2665. https://doi.org/10.1007/s00382-018-4285-1
Aguirre-Gutiérrez J, Malhi Y, Lewis SL et al (2020) Long-term droughts may drive drier tropical forests towards increased functional, taxonomic and phylogenetic homogeneity. Nat Commun 11:3346. https://doi.org/10.1038/s41467-020-16973-4
Aleixo I, Norris D, Hemerik L et al (2019) Amazonian rainforest tree mortality driven by climate and functional traits. Nat Clim Chang 9:384–388
Alexander I (1989) Mycorrhizas in tropical forests. In: Proctor J (ed) Mineral Nutrients in Tropical Forest and Savanna Ecosystems. Oxford: Blackwell Scientific Publications, pp 169–188
Anderegg WRL, Konings AG, Trugman AT et al (2018) Hydraulic diversity of forests regulates ecosystem resilience during drought. Nature 561:538–541. https://doi.org/10.1038/s41586-018-0539-7
Aragão LEOC, Anderson LO, Fonseca MG et al (2018) 21st Century drought-related fires counteract the decline of Amazon deforestation carbon emissions. Nat Commun 9:536. https://doi.org/10.1038/s41467-017-02771-y
Araújo AC, von Randow C, Restrepo-Coupe N (2016) Ecosystem-atmosphere exchanges of CO2 in dense and open “Terra Firme” rainforests in Brazilian Amazonia. In: Nagy L, Forsberg B, Artaxo P (eds) Interactions between biosphere, atmosphere and human land use in the Amazon basin, vol 227. Ecological Studies. Springer, Berlin, pp 149–169
Arend M, Monshausen G, Wind C et al (2004) Effect of potassium deficiency on the plasma membrane H+-ATPASE of the wood ray parenchyma in poplar. Plant Cell Environ 27:1288–1296. https://doi.org/10.1111/j.1365-3040.2004.01236.x
Armstrong MJ, Kirkby EA (1979) Estimation of potassium recirculation in tomato plants by comparison of the rates of potassium and calcium accumulation in the tops with their fluxes in the xylem stream. Plant Physiol 63:1143–1148. https://doi.org/10.1104/pp.63.6.1143
Arsić J, Stojanović M, Petrovičová L et al (2021) Increased wood biomass growth is associated with lower wood density in Quercus petraea (Matt.) Liebl. saplings growing under elevated CO2. PLoS ONE 16:e0259054. https://doi.org/10.1371/journal.pone.0259054
Baker JCA, Santos GM, Gloor M, Brienen RJW (2017) Does Cedrela always form annual rings? Testing ring periodicity across South America using radiocarbon dating. Trees Struct Funct 31:1999–2009. https://doi.org/10.1007/s00468-017-1604-9
Barrelet T, Ulrich A, Rennenberg H, Krähenbühl U (2006) Seasonal profiles of sulphur, phosphorus, and potassium in Norway spruce wood. Plant Biol 8:462–469. https://doi.org/10.1055/s-2006-924044
Barstow M (2018) Cedrela fissilis. In: IUCN Red List Threat. Species 2018. https://doi.org/10.2305/IUCN.UK.2018-1.RLTS.T33928A68080477.en
Bergès L, Dupouey JL, Franc A (2000) Long-term changes in wood density and radial growth of Quercus petraea Liebl. in northern France since the middle of the nineteenth century. Trees Struct Funct 14:398–408. https://doi.org/10.1007/s004680000055
Boers N, Marwan N, Barbosa HMJ, Kurths J (2017) A deforestation-induced tipping point for the South American monsoon system. Sci Rep 7:41489. https://doi.org/10.1038/srep41489
Bojorquez-Quintal JE, Sanchez-Cach LA, Ku-Gonzalez A et al (2014) Differential effects of aluminum on in vitro primary root growth, nutrient content and phospholipase C activity in coffee seedlings (Coffea arabica). J Inorg Biochem 134:39–48
Bojórquez-Quintal E, Escalante-Magaña C, Echevarría-Machado I, Martínez-Estévez M (2017) Aluminum, a friend or foe of higher plants in acid soils. Front Plant Sci 8:1767. https://doi.org/10.3389/fpls.2017.01767
Bontemps JD, Gelhaye P, Nepveu G, Hervé JC (2013) When tree rings behave like foam: moderate historical decrease in the mean ring density of common beech paralleling a strong historical growth increase. Ann For Sci 70:329–343. https://doi.org/10.1007/s13595-013-0263-2
Bose J, Babourina O, Shabala S, Rengel Z (2013) Low-pH and aluminum resistance in arabidopsis correlates with high cytosolic magnesium content and increased magnesium uptake by plant roots. Plant Cell Physiol 54:1093–1104. https://doi.org/10.1093/pcp/pct064
Bouriaud O, Bréda N, Le Moguédec G, Nepveu G (2004) Modelling variability of wood density in beech as affected by ring age, radial growth and climate. Trees Struct Funct 18:264–276. https://doi.org/10.1007/s00468-003-0303-x
Brienen RJW, Zuidema PA (2005) Relating tree growth to rainfall in Bolivian rain forests: a test for six species using tree ring analysis. Oecologia 146:1–12. https://doi.org/10.1007/s00442-005-0160-y
Brienen RJW, Phillips OL, Feldpausch TR et al (2015) Long-term decline of the Amazon carbon sink. Nature 519:344–348. https://doi.org/10.1038/nature14283
Brunner I, Sperisen C (2013) Aluminum exclusion and aluminum tolerance in woody plants. Front Plant Sci 4:172. https://doi.org/10.3389/fpls.2013.00172
Burger A, Lichtscheidl I (2019) Strontium in the environment: Review about reactions of plants towards stable and radioactive strontium isotopes. Sci Total Environ 653:1458–1512. https://doi.org/10.1016/j.scitotenv.2018.10.312
Buscardo E, Nardoto G, Luizão F et al (2016) The biogeochemistry of the main forest vegetation types in Amazonia. Springer, Berlin, pp 225–266
Caioni C, Silvério DV, Macedo MN et al (2020) Droughts amplify differences between the energy balance components of Amazon forests and croplands. Remote Sens 12:525. https://doi.org/10.3390/rs12030525
Camarero JJ, Mendivelso HA, Sanchez-Salguero R (2020) How past and future climate and drought drive radial-growth variability of three tree species in a bolivian tropical dry forest. In: Pompa-García M, Camarero JJ (eds) Latin American dendroecology. Springer, Cham, pp 121–140
Carvalho C, Mosquera B, Veiga R, Anjos RM (2005) Radioecological investigations in Brazilian tropical plants. Braz J Phys 35:808–810. https://doi.org/10.1590/S0103-97332005000500023
Coe MT, Macedo MN, Brando PM et al (2016) The hydrology and energy balance of the Amazon Basin. Springer, Berlin, pp 35–53
Cook E, Krusic P (2005) Program ARSTAN: a tree-ring standardization program based on detrending and autoregressive time series modeling, with interactive graphics. Manuscr file, Tree-Ring Lab Lamont Doherty Earth Obs Columbia Univ Palisades
Cook ER, Pederson N (2011) Uncertainty, emergence, and statistics in dendrochronology. Springer, Dordrecht, pp 77–112
Covey K, Soper F, Pangala S et al (2021) Carbon and beyond: the biogeochemistry of climate in a rapidly changing Amazon. Front For Glob Chang 1:618401. https://doi.org/10.3389/ffgc.2021.618401
Croudace IW, Rindby A, Rothwell RG (2006) ITRAX: Description and evaluation of a new multi-function X-ray core scanner. Geol Soc Spec Publ 267:51–63. https://doi.org/10.1144/GSL.SP.2006.267.01.04
Cuevas E, Medina E (1986) Nutrient dynamics within amazonian forest ecosystems: I. Nutrient flux in fine litter fall and efficiency of nutrient utilization. Oecologia 68:466–472. https://doi.org/10.1007/BF01036756
Cunha HFV, Andersen KM, Lugli LF et al (2022) Direct evidence for phosphorus limitation on Amazon forest productivity. Nature 608:558–562. https://doi.org/10.1038/s41586-022-05085-2
Cutter BE, Guyette RP (1993) Anatomical, chemical, and ecological factors affecting tree species choice in dendrochemistry studies. J Environ Qual 22:611–619. https://doi.org/10.2134/jeq1993.00472425002200030028x
Cybis Electronic (2013) CDendro and CooRecorder V.7.7
de Pereira GA, Barbosa ACMC, Torbenson MCA et al (2018) The climate response of Cedrela fissilis annual ring width in the Rio São Francisco Basin, Brazil. Tree-Ring Res 74:162–171. https://doi.org/10.3959/1536-1098-74.2.162
De Souza ES, Fernandes AR, De Souza Braz AM et al (2018a) Physical, chemical, and mineralogical attributes of a representative group of soils from the eastern Amazon region in Brazil. Soil 4:195–212. https://doi.org/10.5194/soil-4-195-2018
Drouet T, Herbauts J (2008) Evaluation of the mobility and discrimination of Ca, Sr and Ba in forest ecosystems: consequence on the use of alkaline-earth element ratios as tracers of Ca. Plant Soil 302:105–124. https://doi.org/10.1007/s11104-007-9459-2
Dünisch O (2005) Influence of the El-niño southern oscillation on cambial growth of Cedrela fissilis Vell. in tropical and subtropical Brazil. J Appl Bot Food Qual 79:5–11
Dünisch O, Montóia VR, Bauch J (2003) Dendroecological investigations on Swietenia macrophylla King and Cedrela odorata L. (Meliaceae ) in the central Amazon. Trees Struct Funct 17:244–250. https://doi.org/10.1007/s00468-002-0230-2
Durgante FM, Higuchi N, Ohashi S et al (2023) Soil fertility and drought interact to determine large variations in wood production for a hyperdominant Amazonian tree species. Front For Glob Chang 5:1–11. https://doi.org/10.3389/ffgc.2022.1065645
EMBRAPA (2006) Sistema brasileiro de classificação de solos, 2nd edn. Empresa Brasileira de Pesquisa Agropecuária, Rio de Janeiro
Espinoza JC, Ronchail J, Marengo JA, Segura H (2019) Contrasting North-South changes in Amazon wet-day and dry-day frequency and related atmospheric features (1981–2017). Clim Dyn 52:5413–5430. https://doi.org/10.1007/s00382-018-4462-2
Espírito-Santo FDB, Gloor M, Keller M et al (2014) Size and frequency of natural forest disturbances and the Amazon forest carbon balance. Nat Commun 5:3434. https://doi.org/10.1038/ncomms4434
Esquivel-Muelbert A, Baker TR, Dexter KG et al (2019) Compositional response of Amazon forests to climate change. Glob Chang Biol 25:39–56. https://doi.org/10.1111/gcb.14413
Etienne P, Diquelou S, Prudent M et al (2018) Macro and micronutrient storage in plants and their remobilization when facing scarcity: the case of drought. Agriculture 8:14. https://doi.org/10.3390/agriculture8010014
Exbrayat J-F, Liu YY, Williams M (2017) Impact of deforestation and climate on the Amazon Basin’s above-ground biomass during 1993–2012. Sci Rep 7:15615. https://doi.org/10.1038/s41598-017-15788-6
Fairchild IJ, Loader NJ, Wynn PM et al (2009) Sulfur fixation in wood mapped by synchrotron X-ray studies: implications for environmental archives. Environ Sci Technol 43:1310–1315. https://doi.org/10.1021/es8029297
Fauset S, Gloor M, Fyllas NM et al (2019) Individual-based modeling of Amazon forests suggests that climate controls productivity while traits control demography. Front Earth Sci 7:83. https://doi.org/10.3389/feart.2019.00083
Feeley KJ, Joseph Wright S, Nur Supardi MN et al (2007) Decelerating growth in tropical forest trees. Ecol Lett 10:461–469. https://doi.org/10.1111/j.1461-0248.2007.01033.x
Feller U, Anders I, Wei S (2015) Effects of PEG-induced water deficit in Solanum nigrum on Zn and Ni uptake and translocation in split root systems. Plants 4:284–297. https://doi.org/10.3390/plants4020284
Fengler FH, Bressane A, Tchechen RS et al (2022) Post-mined areas monitoring in Brazilian Amazonia: a new perspective on soil restoration indicators in tropical forests. In: Poisson (eds) Meio Ambiente, Sustentabilidade e Tecnologia–Vol. 10, Poisson. Poisson, Belo Horizonte-MG
Figueiredo V, Enrich-Prast A, Rütting T (2019) Evolution of nitrogen cycling in regrowing Amazonian rainforest. Sci Rep 9:8538. https://doi.org/10.1038/s41598-019-43963-4
Fleischer K, Rammig A, De Kauwe MG et al (2019) Amazon forest response to CO2 fertilization dependent on plant phosphorus acquisition. Nat Geosci 12:736–741. https://doi.org/10.1038/s41561-019-0404-9
Follet-Gueye ML, Verdus MC, Demarty M et al (1998) Cambium pre-activation in beech correlates with a strong temporary increase of calcium in cambium and phloem but not in xylem cells. Cell Calcium 24:205–211. https://doi.org/10.1016/S0143-4160(98)90129-4
Fromm J (2010) Wood formation of trees in relation to potassium and calcium nutrition. Tree Physiol 30:1140–1147. https://doi.org/10.1093/treephys/tpq024
Fu R, Li W (2004) The influence of the land surface on the transition from dry to wet season in Amazonia. Theor Appl Climatol 78:97–110. https://doi.org/10.1007/s00704-004-0046-7
Gavrilescu M (2021) Water, soil, and plants interactions in a threatened environment. Water 13:2746. https://doi.org/10.3390/w13192746
Gazol A, Ribas M, Gutiérrez E, Camarero JJ (2017) Aleppo pine forests from across Spain show drought-induced growth decline and partial recovery. Agric For Meteorol 232:186–194. https://doi.org/10.1016/j.agrformet.2016.08.014
Gessler A, Schaub M, McDowell NG (2017) The role of nutrients in drought-induced tree mortality and recovery. New Phytol 214:513–520
Ghanati F, Morita A, Yokota H (2005) Effects of aluminum on the growth of tea plant and activation of antioxidant system. Plant Soil 276:133–141. https://doi.org/10.1007/s11104-005-3697-y
Glavac V, Koenies H, Ebben U (1990) Seasonal variation of calcium, magnesium, potassium, and manganese contents in xylem sap of beech (Fagus sylvatica L.) in a 35-year-old limestone beech forest stand. Trees-Struct Funct 4:75–80
Gloor E (2016) Climate and the Amazonian carbon balance. In: Nagy L, Forsberg B, Artaxo P (eds) Interactions between biosphere, atmosphere and human land use in the Amazon basin, vol 227. Ecological Studies. Springer, Berlin, pp 101–117. https://doi.org/10.1007/978-3-662-49902-3_6
Göransson A, Eldhuset TD (1991) Effects of aluminium on growth and nutrient uptake of small Picea abies and Pinus sylvestris plants. Trees-Struct Funct 5:136–142. https://doi.org/10.1007/BF00204334
Grace J (2016) The Amazon carbon balance: an evaluation of methods and results. Springer, Berlin, pp 79–100
Granato-Souza D, Stahle DW, Barbosa AC et al (2018) Tree rings and rainfall in the equatorial Amazon. Clim Dyn 52:1857–1869. https://doi.org/10.1007/s00382-018-4227-y
Grossiord C (2020) Having the right neighbors: how tree species diversity modulates drought impacts on forests. New Phytol 228:42–49
Hartmann T, Mult S, Suter M et al (2000) Leaf age-dependent differences in sulphur assimilation and allocation in poplar (Populus tremula × P. alba) leaves. J Exp Bot 51:1077–1088. https://doi.org/10.1093/jexbot/51.347.1077
He M, Dijkstra FA (2014) Drought effect on plant nitrogen and phosphorus: a meta-analysis. New Phytol 204:924–931. https://doi.org/10.1111/nph.12952
Herschbach C, Rennenberg H (2001) Sulfur nutrition of deciduous trees. Naturwissenschaften 88:25–36. https://doi.org/10.1007/s001140000200
Hevia A, Sánchez-Salguero R, Camarero JJ et al (2019) Long-term nutrient imbalances linked to drought-triggered forest dieback. Sci Total Environ 690:1254–1267. https://doi.org/10.1016/j.scitotenv.2019.06.515
Hietz P, Horsky M, Prohaska T et al (2015) High-resolution densitometry and elemental analysis of tropical wood. Trees-Struct Funct 29:487–497. https://doi.org/10.1007/s00468-014-1126-7
Higuchi N, Suwa R, Higuchi FG et al (2016) Overview of forest carbon stocks study in Amazonas State, Brazil. In: Nagy L, Forsberg B, Artaxo P (eds) Interactions between biosphere, atmosphere and human land use in the Amazon Basin, vol 227. Ecological Studies. Springer, Berlin, pp 171–187
Holmes R (1983) Computer-assisted quality control in tree-ring dating and measurement. Tree-Ring Bull 43:51–67
Holmes RL (1994) Dendrochronology program library: users manual. University of Arizona, Tucson
Hu Y, Schmidhalter U (2005) Drought and salinity: a comparison of their effects on mineral nutrition of plants. J Plant Nutr Soil Sci 168:541–549. https://doi.org/10.1002/jpln.200420516
Humanes-Fuente V, Ferrero ME, Muñoz AA et al (2020) Two centuries of hydroclimatic variability reconstructed from tree-ring records over the Amazonian Andes of Peru. J Geophys Res Atmos 125:e2020JD032565. https://doi.org/10.1029/2020JD032565
Ihsan MZ, Daur I, Alghabari F et al (2019) Heat stress and plant development: role of sulphur metabolites and management strategies. Acta Agric Scand Sect B Soil Plant Sci 69:332–342. https://doi.org/10.1080/09064710.2019.1569715
Isermann K (1981) Uptake of stable strontium by plants and effects on plant growth. In: Skoryna SC (ed) Handbook of stable strontium. Springer, Boston, pp 1–9
Islam M, Rahman M, Bräuning A (2018) Long-term hydraulic adjustment of three tropical moist forest tree species to changing climate. Front Plant Sci 9:1761. https://doi.org/10.3389/fpls.2018.01761
Islam M, Rahman M, Bräuning A (2019) Impact of extreme drought on tree-ring width and vessel anatomical features of Chukrasia tabularis. Dendrochronologia 53:63–72. https://doi.org/10.1016/J.DENDRO.2018.11.007
Jacobsen AL, Ewers FW, Pratt RB et al (2005) Do xylem fibers affect vessel cavitation resistance? Plant Physiol 139:546–556
Janssen TAJ, Hölttä T, Fleischer K et al (2019) Wood allocation trade-offs between fiber wall, fiber lumen, and axial parenchyma drive drought resistance in neotropical trees. Plant Cell Environ 43:965–980. https://doi.org/10.1111/pce.13687
Jiao F, Shi XR, Han FP, Yuan ZY (2016) Increasing aridity, temperature and soil pH induce soil C-N-P imbalance in grasslands. Sci Rep 6:19601. https://doi.org/10.1038/srep19601
Jordan CF (1985) Soils of the Amazon rainforest. In: Prance GT, Lovejoy TE (eds) Amazonia: Key Environments Series. Pergamon Press, Oxford, pp 83–94
Jordan C, Golley F, Hall J, Hall J (1980) Nutrient scavenging of rainfall by the canopy of an Amazonian rain forest. Biotropica 12:61–66. https://doi.org/10.2307/2387774
Ka FF, Carr HP, Zhang J, Ming HW (2008) Growth and nutrient uptake of tea under different aluminium concentrations. J Sci Food Agric 88:1582–1591. https://doi.org/10.1002/jsfa.3254
Kavka M, Polle A (2016) Phosphate uptake kinetics and tissue-specific transporter expression profiles in poplar (Populus × canescens) at different phosphorus availabilities. BMC Plant Biol 16:206. https://doi.org/10.1186/s12870-016-0892-3
Khan NA, Khan MIR, Asgher M et al (2014) Salinity tolerance in plants: revisiting the role of sulfur metabolites. J Plant Biochem Physiol 2:1–8. https://doi.org/10.4172/2329-9029.1000120
Kogelmann WJ, Sharpe WE (2006) Soil acidity and manganese in declining and nondeclining sugar maple stands in Pennsylvania. J Environ Qual 35:433–441. https://doi.org/10.2134/jeq2004.0347
Kreuzwieser J, Gessler A (2010) Global climate change and tree nutrition: influence of water availability. Tree Physiol 30:1221–1234
Kuang YW, Wen DZ, Zhou GY et al (2008) Reconstruction of soil pH by dendrochemistry of Masson pine at two forested sites in the Pearl River Delta, South China. Ann For Sci 65:804–804. https://doi.org/10.1051/forest:2008070
Kurita Y, Kanno S, Sugita R et al (2022) Visualization of phosphorus re-translocation and phosphate transporter expression profiles in a shortened annual cycle system of poplar. Plant Cell Environ 45:1749–1764. https://doi.org/10.1111/pce.14319
Lachenbruch B, Mcculloh KA (2014) Traits, properties, and performance: how woody plants combine hydraulic and mechanical functions in a cell, tissue, or whole plant. New Phytol 204:747–764. https://doi.org/10.1111/nph.13035
Laurance WF, Fearnside PM, Laurance SG et al (1999) Relationship between soils and Amazon forest biomass: a landscape-scale study. For Ecol Manage 118:127–138. https://doi.org/10.1016/S0378-1127(98)00494-0
Lautner S, Fromm J (2010) Calcium-dependent physiological processes in trees. Plant Biol 12:268–274. https://doi.org/10.1111/j.1438-8677.2009.00281.x
Lejeune Q, Davin EL, Guillod BP, Seneviratne SI (2015) Influence of Amazonian deforestation on the future evolution of regional surface fluxes, circulation, surface temperature and precipitation. Clim Dyn 44:2769–2786. https://doi.org/10.1007/s00382-014-2203-8
Levine NM, Zhang K, Longo M et al (2016) Ecosystem heterogeneity determines the ecological resilience of the Amazon to climate change. Proc Natl Acad Sci USA 113:793–797. https://doi.org/10.1073/pnas.1511344112
Li W, Fu R (2004) Transition of the large-scale atmospheric and land surface conditions from the dry to the wet season over Amazonia as diagnosed by the ECMWF re-analysis. J Clim 17:2637–2651. https://doi.org/10.1175/1520-0442(2004)017%3c2637:TOTLAA%3e2.0.CO;2
Lin WY, Huang TK, Leong SJ, Chiou TJ (2014) Long-distance call from phosphate: systemic regulation of phosphate starvation responses. J Exp Bot 65:1817–1827
Lisi CS, Pagotto MA, Anholetto CR et al (2020) Dendroecological studies with Cedrela odorata L., northeastern Brazil. In: Pompa-García M, Camarero J (eds) Latin American dendroecology. Springer, Berlin, pp 37–59
Lloret F, Keeling EG, Sala A (2011) Components of tree resilience: effects of successive low-growth episodes in old ponderosa pine forests. Oikos 120:1909–1920. https://doi.org/10.1111/j.1600-0706.2011.19372.x
Lobão MS (2011) Dendrocronologia, fenologia, atividade cambial e qualidade do lenho de árvores de Cedrela odorata L., Cedrela fissilis Vell. e Schizolobium parahyba var. amazonicum Hub. ex Ducke, no estado do Acre. Brasil. p 215
Locosselli GM, Brienen RJW, de Souza LM et al (2020) Global tree-ring analysis reveals rapid decrease in tropical tree longevity with temperature. Proc Natl Acad Sci USA 117:33358–33364. https://doi.org/10.1073/PNAS.2003873117
Lovejoy TE, Nobre C (2018) Amazon tipping point. Sci Adv 4:eaat2340. https://doi.org/10.1126/sciadv.aat2340
Malhi Y, Melack J, Gatti L V, et al (2021) Chapter 6: Biogeochemical cycles in the Amazon. In: Amazon assessment report 2021
Marcati CR, Angyalossy V, Evert RF (2006) Seasonal variation in wood formation of Cedrela fissilis (Meliaceae). IAWA J 27:199–211. https://doi.org/10.1163/22941932-90000149
Marengo JA (2009) Long-term trends and cycles in the hydrometeorology of the Amazon basin since the late 1920s. Hydrol Process 23:3236–3244. https://doi.org/10.1002/hyp
Marengo JA, Espinoza JC (2016) Extreme seasonal droughts and floods in Amazonia: causes, trends and impacts. Int J Climatol 36:1033–1050. https://doi.org/10.1002/JOC.4420
Marengo JA, Nobre CA, Tomasella J et al (2008) Hydro-climatic and ecological behaviour of the drought of Amazonia in 2005. Phil Trans R Soc B Biol Sci 363:1773–1778
Marengo JA, Williams ER, Alves LM et al (2016) Extreme seasonal climate variations in the Amazon Basin: droughts and floods. In: Nagy L, Forsberg BR, Artaxo P (eds) Interactions between biosphere, atmosphere and human land use in the Amazon Basin. Springer, Berlin, pp 55–78
Marengo JA, Souza CM, Thonicke K et al (2018) Changes in climate and land use over the Amazon region: current and future variability and trends. Front Earth Sci 6:228. https://doi.org/10.3389/feart.2018.00228
McClenahen JR, Vimmerstedt JP, Scherzer AJ (1989) Elemental concentrations in tree rings by PIXE: statistical variability, mobility, and effects of altered soil chemistry. Can J For Res 19:880–888
Mcelrone AJ, Choat B, Gambetta GA, Brodersen CR (2013) Water uptake and transport in vascular plants. Nat Educ Knowl 4:6
McGrath DA, Smith CK, Gholz HL, Oliveira FDA (2001) Effects of land-use change on soil nutrient dynamics in Amazônia. Ecosystems 4:625–645. https://doi.org/10.1007/s10021-001-0033-0
Mendivelso HA, Camarero JJ, Royo Obregón O et al (2013) Differential growth responses to water balance of coexisting deciduous tree species are linked to wood density in a Bolivian tropical dry forest. PLoS ONE 8:e73855. https://doi.org/10.1371/journal.pone.0073855
Mendivelso HA, Camarero JJ, Gutiérrez E, Zuidema PA (2014) Time-dependent effects of climate and drought on tree growth in a Neotropical dry forest: short-term tolerance vs. long-term sensitivity. Agric For Meteorol 188:13–23. https://doi.org/10.1016/j.agrformet.2013.12.010
Mendivelso HA, Camarero JJ, Gutiérrez E, Castaño-Naranjo A (2016) Climatic influences on leaf phenology, xylogenesis and radial stem changes at hourly to monthly scales in two tropical dry forests. Agric For Meteorol 216:20–36. https://doi.org/10.1016/j.agrformet.2015.09.014
Mescouto CST, Lemos VP, Filho HAD et al (2011) Distribution and availability of copper, iron, manganese and zinc in the archaeological black earth profile form the Amazon region. J Braz Chem Soc 22:1484–1492. https://doi.org/10.1590/s0103-50532011000800012
MMA-Ministério Do Meio Ambiente (2005) Plano de manejo da Floresta Nacional do Jamari, vol I. IBAMA, Brasilia
Moreira FMS, Siqueira JO, Brussard L (2006) Soil biodiversity in Amazonian and other Brazilian ecosystems. CABI Publishing, Wallingford
Morrissey J, Guerinot ML (2009) Iron uptake and transport in plants: the good, the bad, and the ionome. Chem Rev 109:4553–4567. https://doi.org/10.1021/cr900112r
Neves MI, Prajapati DH, Parmar S et al (2017) Manganese toxicity hardly affects sulfur metabolism in Brassica rapa. In: Sulfur Metabolism in Higher Plants: Fundamental, Environmental and Agricultural Aspects. Springer, Berlin, pp 155–162. https://doi.org/10.1007/978-3-319-56526-2_15
NOAA (2021) Global monitoring laboratory-carbon cycle greenhouse gases. https://gml.noaa.gov/ccgg/trends/global.html. Accessed 8 Nov 2022
Nock CA, Geihofer D, Grabner M et al (2009) Wood density and its radial variation in six canopy tree species differing in shade-tolerance in western Thailand. Ann Bot 104:297–306. https://doi.org/10.1093/aob/mcp118
Ortega Rodriguez DR, Hevia A, Sánchez-Salguero R et al (2022) Exploring wood anatomy, density and chemistry profiles to understand the tree-ring formation in Amazonian tree species. Dendrochronologia 71:125915. https://doi.org/10.1016/J.DENDRO.2021.125915
Ortega Rodriguez DR, Sánchez-Salguero R, Hevia A et al (2023a) Climate variability of the southern Amazon inferred by a multi-proxy tree-ring approach using Cedrela fissilis Vell. Sci Total Environ 871:162064. https://doi.org/10.1016/j.scitotenv.2023.162064
Ortega Rodriguez DR, Sánchez-Salguero R, Hevia A et al (2023b) Long- and short-term impacts of climate and dry-season on wood traits of Cedrela fissilis Vell. in southern Brazilian Amazon. Agric For Meteorol 333:109392. https://doi.org/10.1016/j.agrformet.2023.109392
Østmoe S (2020) Transfer of strontium in the potato plant, Solanum tuberosum L., following single foliar wet deposition: field experiments performed in Eastern Norway and at the West Coast of Norway. Norwegian University of Life Sciences
Paredes-Villanueva K, López L, Navarro Cerrillo RM (2016) Regional chronologies of Cedrela fissilis and Cedrela angustifolia in three forest types and their relation to climate. Trees-Struct Funct 30:1581–1593. https://doi.org/10.1007/s00468-016-1391-8
Peña-Claros M, Peters EM, Justiniano MJ et al (2008) Regeneration of commercial tree species following silvicultural treatments in a moist tropical forest. For Ecol Manage 255:1283–1293. https://doi.org/10.1016/j.foreco.2007.10.033
Pennington TD, Muellner AN (2010) A Monograph of Cedrela (Meliaceae). dh books, Milborne Port
Pereyra Espinoza MJ, Inga Guillen GJ, Santos Morales M, Rodríguez Arisméndiz R (2014) Potencialidad de Cedrela odorata (Meliaceae) para estudios dendrocronológicos en la selva central del Perú. Rev Biol Trop 62:783–793. https://doi.org/10.15517/rbt.v62i2.9835
Peterson DL, Anderson DR (1990) Content of chemical elements in tree rings of lodgepole pine and whitebark pine from a subalpine Sierra Nevada forest. Res Pap Pacific Southwest Res Station USDA For Serv 200. p 9
Phillips OL, Aragão LEOC, Lewis SL et al (2009) Drought sensitivity of the Amazon rainforest. Science 323:1344–1347. https://doi.org/10.1126/science.1164033
Phillips OL, Lewis SL, Higuchi N, Baker T (2016) Recent changes in Amazon forest biomass and dynamics. In: Nagy L, Forsberg B, Artaxo P (eds) Interactions between biosphere, atmosphere and human land use in the Amazon Basin, vol 227. Ecological Studies. Springer, Berlin, pp 191–224
Phillips OL, Brienen RJW, Gloor E et al (2017) Carbon uptake by mature Amazon forests has mitigated Amazon nations’ carbon emissions. Carbon Balance Manag 12:1. https://doi.org/10.1186/s13021-016-0069-2
Pinagé ER, Keller M, Peck C et al (2023) Effects of forest degradation classification on the uncertainty of aboveground carbon estimates in the Amazon. Carbon Balance Manag 18:2. https://doi.org/10.1186/s13021-023-00221-5
Pittman JK (2005) Managing the manganese: molecular mechanisms of manganese transport and homeostasis. New Phytol 167:733–742
Powers JS, Vargas GG, Brodribb TJ et al (2020) A catastrophic tropical drought kills hydraulically vulnerable tree species. Glob Chang Biol 26:3122–3133. https://doi.org/10.1111/gcb.15037
Quesada CA, Lloyd J (2016) Soil-vegetation interactions in Amazonia. In: Nagy L, Forsberg B, Artaxo P (eds) Interactions between biosphere, atmosphere and human land use in the Amazon basin, vol 227. Ecological Studies. Springer, Berlin, pp 267–299. https://doi.org/10.1007/978-3-662-49902-3_12
Quesada CA, Phillips OL, Schwarz M et al (2012) Basin-wide variations in Amazon forest structure and function are mediated by both soils and climate. Biogeosciences 9:2203–2246. https://doi.org/10.5194/bg-9-2203-2012
Quintilhan MT, Santini L, Ortega Rodriguez DR et al (2021) Growth-ring boundaries of tropical tree species: aiding delimitation by long histological sections and wood density profiles. Dendrochronologia 69:125878. https://doi.org/10.1016/J.DENDRO.2021.125878
R Core Team (2021) R: a language and environment for statistical computing. R Found Stat Comput. https://www.R-project.org. Accessed 20 Oct 2022
Raaimakers D, Boot RGA, Dijkstra P, Pot S (1995) Photosynthetic rates in relation to leaf phosphorus content in pioneer versus climax tropical rainforest trees. Oecologia 102:120–125. https://doi.org/10.1007/BF00333319
Raghothama KG (1999) Phosphate acquisition. Annu Rev Plant Biol 50:665–693. https://doi.org/10.1146/annurev.arplant.50.1.665
Rodríguez-Ramírez EC, Ferrero ME, Acevedo-Vega I et al (2022) Plastic adjustments in xylem vessel traits to drought events in three Cedrela species from Peruvian Tropical Andean forests. Sci Rep 12:21112. https://doi.org/10.1038/s41598-022-25645-w
Ronchail J, Cochonneau G, Molinier M et al (2002) Interannual rainfall variability in the Amazon basin and sea-surface temperatures in the equatorial Pacific and the tropical Atlantic Oceans. Int J Climatol 22:1663–1686. https://doi.org/10.1002/joc.815
Rowland L, Da Costa ACL, Galbraith DR et al (2015) Death from drought in tropical forests is triggered by hydraulics not carbon starvation. Nature 528:119–122. https://doi.org/10.1038/nature15539
Salazar-Tortosa D, Castro J, Villar-Salvador P et al (2018) The “isohydric trap”: a proposed feedback between water shortage, stomatal regulation, and nutrient acquisition drives differential growth and survival of European pines under climatic dryness. Glob Chang Biol 24:4069–4083. https://doi.org/10.1111/gcb.14311
Sanchez PA, Bandy DE, Villachica JH, Nicholaides JJ (1982) Amazon basin soils: management for continuous crop production. Science 216:821–827. https://doi.org/10.1126/science.216.4548.821
Sánchez-Salguero R, Camarero JJ, Dobbertin M et al (2013) Contrasting vulnerability and resilience to drought-induced decline of densely planted vs. natural rear-edge Pinus nigra forests. For Ecol Manage 310:956–967. https://doi.org/10.1016/j.foreco.2013.09.050
Sánchez-Salguero R, Camarero JJ, Hevia A et al (2019) Testing annual tree-ring chemistry by X-ray fluorescence for dendroclimatic studies in high-elevation forests from the Spanish Pyrenees. Quat Int 514:130–140. https://doi.org/10.1016/j.quaint.2018.09.007
Santos GM, Ortega Rodriguez DR, de Barreto N et al (2021) Growth assessment of native tree species from the southwestern Brazilian Amazonia by post-AD 1950 14C analysis: implications for tropical dendroclimatology studies and atmospheric 14C reconstructions. Forests 12:1177. https://doi.org/10.3390/f12091177
Schlesinger WH, Dietze MC, Jackson RB et al (2016) Forest biogeochemistry in response to drought. Glob Chang Biol 22:2318–2328. https://doi.org/10.1111/gcb.13105
Scholz A, Klepsch M, Karimi Z, Jansen S (2013) How to quantify conduits in wood? Front Plant Sci 4:56. https://doi.org/10.3389/fpls.2013.00056
Schulman E (1956) Dendroclimatic changes in semi-arid America. University of Arizona Press, Tucson
Shen R, Ma JF (2001) Distribution and mobility of aluminium in an Al-accumulating plant, Fagopyrum esculentum Moench. J Exp Bot 52:1683–1687. https://doi.org/10.1093/jxb/52.361.1683
Silkin PP, Ekimova NV (2012) Relationship of strontium and calcium concentrations with the parameters of cell structure in Siberian spruce and fir tree-rings. Dendrochronologia 30:189–194. https://doi.org/10.1016/j.dendro.2011.06.003
Siqueira SDF, Higuchi P, Da Silva AC (2019) Contemporary and future potential geographic distribution of Cedrela fissilis Vell. under climate change scenarios. Rev Árvore 43:e430306. https://doi.org/10.1590/1806-90882019000300006
Smith FW (2001) Sulphur and phosphorus transport systems in plants. Plant Soil 232:109–118. https://doi.org/10.1023/A:1010390120820
Smith KT, Balouet JC, Shortle WC et al (2014) Dendrochemical patterns of calcium, zinc, and potassium related to internal factors detected by energy dispersive X-ray fluorescence (EDXRF). Chemosphere 95:58–62. https://doi.org/10.1016/j.chemosphere.2013.08.017
Smith C, Baker JCA, Spracklen DV (2023) Tropical deforestation causes large reductions in observed precipitation. Nature 615:270–275. https://doi.org/10.1038/s41586-022-05690-1
Soil Survey Staff (2022) Keys to soil taxonomy, 13 edn. USDA-Natural Resources Conservation Service, Washington DC, pp 1–410
Souza JL, Fontes MP, Gilkes R et al (2018b) Geochemical signature of Amazon tropical rainforest soils. Artic Rev Bras Cienc Solo 42:170192–170193
St. Clair SB, Sharpe WE, Lynch JP (2008) Key interactions between nutrient limitation and climatic factors in temperate forests: a synthesis of the sugar maple literature. Can J For Res 38:401–414. https://doi.org/10.1139/X07-161
Stokes MA, Smiley TL (1996) An introduction to tree-ring dating. University of Arizona Press, Tucson
Tagami K, Uchida S (2017) Soil-to-crop transfer factors (TFs) of alkaline earth elements and comparison of TFs of stable Sr with those of global fallout 90Sr. In: Gupta DK, Walther C (eds) Behaviour of strontium in plants and the environment. Springer, Berlin, pp 75–92
Tao S, Chave J, Frison PL et al (2022) Increasing and widespread vulnerability of intact tropical rainforests to repeated droughts. Proc Natl Acad Sci USA. https://doi.org/10.1073/pnas.2116626119
Toïgo M, Vallet P, Tuilleras V et al (2015) Species mixture increases the effect of drought on tree ring density, but not on ring width, in Quercus petraea–Pinus sylvestris stands. For Ecol Manage 345:73–82. https://doi.org/10.1016/J.FORECO.2015.02.019
Tomazello-Filho M, Brazolin S, Chagas MP et al (2008) Application of X-ray technique in nondestructive evaluation of eucalypt wood. Maderas Cienc y Tecnol 10:139–149. https://doi.org/10.4067/S0718-221X2008000200006
Tomioka R, Oda A, Takenaka C (2005) Root growth enhancement by rhizospheric aluminum treatment in Quercus serrata Thunb. seedlings. J For Res 10:319–324. https://doi.org/10.1007/s10310-005-0152-0
van der Maaten-Theunissen M, Trouillier M, Schwarz J et al (2021) pointRes 2.0: new functions to describe tree resilience. Dendrochronologia 70:125899. https://doi.org/10.1016/J.DENDRO.2021.125899
Vance CP, Uhde-Stone C, Allan DL (2003) Phosphorus acquisition and use: critical adaptations by plants for securing a nonrenewable resource. New Phytol 157:423–447. https://doi.org/10.1046/j.1469-8137.2003.00695.x
Vannoppen A, Boeckx P, De Mil T et al (2018) Climate driven trends in tree biomass increment show asynchronous dependence on tree-ring width and wood density variation. Dendrochronologia 48:40–51. https://doi.org/10.1016/J.DENDRO.2018.02.001
Vicente-Serrano SM, Beguería S, López-Moreno JI (2010) A multiscalar drought index sensitive to global warming: The standardized precipitation evapotranspiration index. J Clim 23:1696–1718. https://doi.org/10.1175/2009JCLI2909.1
Villagra M, Campanello PI, Bucci SJ, Goldstein G (2013) Functional relationships between leaf hydraulics and leaf economic traits in response to nutrient addition in subtropical tree species. Tree Physiol 33:1308–1318. https://doi.org/10.1093/treephys/tpt098
Villalba R, Holmes RL, Boninsegna JA (1992) Spatial patterns of climate and tree growth variations in subtropical northwestern Argentina. J Biogeogr 19:631–649. https://doi.org/10.2307/2845706
Vitoussek PM, Sanford RL (1986) Nutrient cycling in moist tropical forest. Annu Rev Ecol Syst 17:137–167. https://doi.org/10.1146/annurev.es.17.110186.001033
Wang J, Yang D, Detto M et al (2020) Multi-scale integration of satellite remote sensing improves characterization of dry-season green-up in an Amazon tropical evergreen forest. Remote Sens Environ 246:111865. https://doi.org/10.1016/j.rse.2020.111865
Wigley TML, Briffa KR, Jones PD (1984) On the average value of correlated time series, with applications in dendroclimatology and hydrometeorology. J Clim Appl Meteorol 23:201–213
Will RE, Wilson SM, Zou CB, Hennessey TC (2013) Increased vapor pressure deficit due to higher temperature leads to greater transpiration and faster mortality during drought for tree seedlings common to the forest-grassland ecotone. New Phytol 200:366–374. https://doi.org/10.1111/nph.12321
Williams E, Dall’Antonia A, Dall’Antonia V et al (2005) The drought of the century in the Amazon Basin: an analysis of the regional variation of rainfall in South America in 1926. Acta Amaz 35:231–238. https://doi.org/10.1590/s0044-59672005000200013
Wimmer R, Lucas BN (1997) Comparing mechanical properties of secondary wall and cell corner middle lamella in spruce wood. IAWA J 18:77–88
Wind C, Arend M, Fromm J (2004) Potassium-dependent cambial growth in poplar. Plant Biol 6:30–37. https://doi.org/10.1055/s-2004-815738
Wu J, Serbin SP, Ely KS et al (2020) The response of stomatal conductance to seasonal drought in tropical forests. Glob Chang Biol 26:823–839. https://doi.org/10.1111/gcb.14820
Yadav SK (2010) Heavy metals toxicity in plants: an overview on the role of glutathione and phytochelatins in heavy metal stress tolerance of plants. South African J Bot 76:167–179
Yazaki K, Maruyama Y, Mori S et al (2005) Effects of elevated carbon dioxide concentration on wood structure and formation in trees. In: Omasa K, Nouchi I, De Kok LJ (eds) Plant responses to air pollution and global change. Springer, Berlin, pp 89–97
Zavišić A, Polle A (2018) Dynamics of phosphorus nutrition, allocation and growth of young beech (Fagus sylvatica L.) trees in P-rich and P-poor forest soil. Tree Physiol 38:37–51. https://doi.org/10.1093/treephys/tpx146
Acknowledgements
We thank the Wood Anatomy and Tree-Ring Laboratory (LAIM) (FAPESP project: 2009/53951-7), Department of Forest Sciences, Luiz de Queiroz College of Agriculture (ESALQ).
Funding
DROR was supported by a fellowship from the Fundação de Amparo à Pesquisa do Estado do São Paulo (FAPESP; grants # 2018/22914-8, 2020/04608-7). RSS was supported by VULBOS project (UPO-1263216), VURECLIM (P20_00813) and AH by PinCaR project (UHU-1266324) both from FEDER Funds, Andalusia Regional Government, Consejería de Economía, Conocimiento, Empresas y Universidad 2014–2020. RSS and AH were also supported by EQC2018-004821-P and IE19_074 UPO projects cofounded by Spanish “Plan Estatal de Investigación Científica y Técnica y de Innovación 2017–2020” and “Plan Andaluz de Invetigación, Desarrollo e Innovación (PAIDI 2020)”, respectively. RCB was supported from FAPESP (Grant # 2020/11502-0). MJF and MTF were supported from FAPESP–FAPEAM (# 01/2020) and CAPES (PROCAD/Amazônia, # 21/2018). This study was also partially funded by PIRE-project (FAPESP; Grant # 2017/50085-3).
Author information
Authors and Affiliations
Contributions
Conceptualization: DROR, RSS, FAR, MTF. Methodology: DROR, RSS, AH. Resources: RSS, AH, MTF. Sample collection and preparation: DROR. Sample analysis: DROR, RSS, AH, FAR. Data analysis: DROR, RSS, AH. Writing: DROR, RSS, AH, FAR, RCB, MJF, JHS, MTF. Reviewing: DROR, RSS, AH, FAR, RCB, MJF, JHS, MTF. Editing: DROR, FAR, JHS. Supervision: RSS, AH, FAR, MTF.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Additional file 1
: Fig. S1. a Map showing the location of Jamari National Forest, JNF (red triangle), Cedrela fissilis Vell. distribution (dark gray dots) and Amazon soil classification according to the Brazilian Soil Classification System (EMBRAPA 2006). Soils according to Soil taxonomy (Soil Survey Staff 2022): Ultisols (Argisols), Inceptisols (Cambisols), Entisols (Neosols, Gleysols), Oxisols (Latosols), Histosols (Organosols), Alfisols (Luvisols, Plinthosols), Spodosols (Spodosols). b Climate diagram of Walter and Lieth, based on regional meteorological and hydrological records from 1975 to 2018 (data source in Table S1), indicates dry months (solid orange area) and months with rainfall above 50 mm (solid blue areas). Green bars represent the estimated growing period of C. fissilis, which generally falls between September (previous year) and the end of May (current year) according to Lobão (2011). Phenology stages are represented by leaf icons based on C. fissilis data from Lobão (2011): open, gray filled and dark filled indicate periods of leaf fall, leaf flash and leaf fall/leaf flash (occurring simultaneously), respectively. c Anomalies (standardized values) of total annual precipitation, mean annual temperature, total annual difference between precipitation and potential evapotranspiration (P-PET) and total annual standardized precipitation evapotranspiration index (SPEI). Fig. S2. Mean index residual chronologies of molar ratios from C. fissilis trees from the southern Amazon and total annual difference between precipitation and potential evapotranspiration (P-PET, time-scales from prior September to current September). Vertical light red boxes represent extreme drought events registered in the southern Amazon basin in 1981–1983, 1997–1998, 2005–2007, 2010–2011, and 2014–2015 (Espinoza et al. 2019). On the right is the boxplot of normalized parameters in dry and wet years; a significant difference (SD) between means is also presented (p < 0.05). Fig. S3. Boxplots showing standardized ring width (RW), wood density (RD), concentrations of aluminum (Al), sulfur (S), potassium (K), calcium (Ca) and calcium to manganese ratio (Ca/Mn) stability during extreme droughts from C. fissilis trees from the southern Amazon. Fig. S4. Boxplots showing standardized ring width (RW), wood density (RD), concentrations of aluminum (Al), sulfur (S), potassium (K), calcium (Ca) and calcium to manganese ratio (Ca/Mn) stability during extreme droughts from C. fissilis trees from the southern Amazon. Fig. S5. Pearson´s correlation of stability of tree-ring parameters: width (RW), wood density (RD), elements (aluminum, Al; phosphorus, P; sulfur, S; potassium, K; calcium, Ca; manganese, Mn; iron, Fe; strontium, Sr) and ratios (aluminum to calcium, Al/Ca; calcium to manganese, Ca/Mn; potassium to calcium, K/Ca; strontium to calcium, Sr/Ca; manganese to sulfur, Mn/S) of C. fissilis. Statistical significance of Pearson´s correlation: *p < 0.05. Table S1. Information from meteorological stations for collected precipitation (P, 1975–2018) and temperature (T, 1957–2018) data.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ortega Rodriguez, D.R., Sánchez-Salguero, R., Hevia, A. et al. Does climate change alter the nutrient trends of Cedrela fissilis Vell. trees in the southern Brazilian Amazon?. Ecol Process 12, 58 (2023). https://doi.org/10.1186/s13717-023-00472-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s13717-023-00472-7