Abstract
Background
Forest ecosystems undergo significant transformations due to harvesting and climate fluctuations, emphasizing the critical role of seeding in natural regeneration and long-term structural preservation. Climate change further amplifies these dynamics, affecting phenology across species and regions. In Tierra del Fuego (Argentina), Nothofagus pumilio (lenga) forests represent the most important timber resource, and it is managed through different silvicultural strategies. This species demonstrates notable post-disturbance regeneration, yet seed fall exhibits significant variability, leading to variations in seed quality (e.g., viability). This study aims to assess fluctuations in N. pumilio seed quality, determine how it varies concerning forest management strategies, annual productivity, and the co-occurrence of climatic phenomena including El Niño-Southern Oscillation (ENSO) and the Southern Annular Mode (SAM).
Results
Viable seeds represented 18.4% of the total, notably higher in unharvested than in managed areas. Conversely, empty seeds were more prevalent in harvested areas (> 75%). Seed quality exhibited significant differences across silvicultural treatments, except for insect-predated seeds, which had similar proportions across all areas, though dispersed retention showed higher predation. When considering years with varying production levels, high-production years favoured full and viable seeds, particularly in unharvested forests and aggregated retention, while low-production years saw reduced viability across all treatments. Quadratic models revealed that viability increased with seed production, where unharvested forests achieved the highest values. Climate variability influenced seed proportions, where ENSO+/SAM+ promoting more full and viable seeds, while ENSO–/SAM+ favoured non-predated seeds, especially in unharvested stands.
Conclusions
Seed quality varies among treatments and years with different levels of seeding. Variations in seed quality, linked to climatic events, influence seed viability. Seed quality plays a critical role in forest regeneration, ensuring a seedling bank for harvested stands to face climate variability. These findings are relevant for forest management and ecosystem services, considering the increasing climate variability and extreme events. Understanding these influences is crucial for Nothofagus pumilio forests' sustainability and global forest adaptation strategies.
Similar content being viewed by others
Background
Forest ecosystems are intricately influenced by fluctuations in climate, with seeding emerging as a pivotal process crucial for forest natural regeneration and the preservation of its structure over extended timeframes (Martinez Pastur et al. 2016; Srur et al. 2018; LaMontagne et al. 2020). Particularly, within the backdrop of climate change (IPCC 2022), which results in widespread and ongoing shifts in phenology across many taxa and within varied geographic regions (Cleland et al. 2007). In this sense, the most important requirement is the production of viable seeds (Fenner 1985) for full density regeneration of disturbed areas.
In Tierra del Fuego, Argentina, Nothofagus pumilio (Poepp. & Endl.) Krasser (commonly named lenga) forests are the main resource for timber in the sawmill industry (Martínez Pastur et al. 2009) harvested and managed through different practices. It is a deciduous species naturally distributed across the geographical range of Argentina and Chile from 33º to 56º SL (Donoso 1975; Veblen et al. 1996). It evolves by giving rise to monospecific forests, ecosystems characterized by straightforward and predictable ecological dynamics driven by gap formation (Wardle 1970; Alan and Veblen 1993). Growth phases development are described for N. pumilio across diverse site quality gradients (Richter and Frangi 1992; Martínez Pastur et al. 2004), where forest management strategies for these monospecific forests are predominantly based on the natural regeneration of harvested stands (Martínez Pastur et al. 2019). Variable retention harvesting, which combines economic and conservation objectives, has emerged as a prominent strategy in N. pumilio forests during the twenty-first century, incorporating different levels of retention, such as aggregate and dispersed retention (Martínez Pastur et al. 2009, 2019). It guarantees forest regeneration due to both pre-harvesting saplings existence development and continuous seed fall supply. In this case, the first years after harvesting critically influence seed fall and seedling recruitment for the restoration of impacted forest structures (Martínez Pastur et al. 2011a,b; Rodríguez-Souilla et al. 2023a, b).
The species exhibits robust regeneration through seed germination following both natural and anthropogenic disturbances, such as harvesting. Notably, seed fall is characterized by intermittent occurrences and pronounced inter-annual variations (Herrera et al. 1998; Torres et al. 2015), at both stand and landscape levels (Martínez Pastur et al. 2013; Rodríguez-Souilla et al. 2023a). Consequently, seed fall emerges as a pivotal process to ensure the uninterrupted continuity of forest ecosystems (Rosenfeld et al. 2006; Cellini 2010; Martinez Pastur et al. 2011a) and the safeguarding of their inherent genetic resources (Marchelli and Gallo 1999; Premoli and Kitzberger 2005). Seeding cycles have been well-documented for other Nothofagus forest species (Alley et al. 1998; Monks and Kelly 2006). The intricate interplay of biological processes and environmental factors (Qiu et al. 2023), including topography, soil conditions, and forest structure (Övergaard et al. 2007; Soler et al. 2010; Bahamonde et al. 2013), significantly affects annual seed production (Hernández et al. 1992; Hacket-Pain et al. 2019; Koyejo et al. 2020; Kitayama et al. 2021). It also represents high differences according to retentions levels, as example of variable retention (Martínez Pastur et al. 2019; Rodríguez-Souilla et al. 2023a). Within the framework of cyclic seed production and variations influenced by the impacts of forest harvesting, it is imperative to recognize that this process also involves fluctuations in the quality of the fruits (Toro Manríquez et al. 2016). These variations include the potential for fruits to be predated by bird and insect activities, display instances of absent endosperm (empty fruits), exhibit instances of embryo-only abortions, and demonstrate viability or not. The proportions of each of these scenarios can be variable (Ledgard and Cath 1983; Martinez Pastur et al. 2008), primarily influenced by years of high and low seed production, along with climatic factors that alter tree physiology (Allen and Platt 1990) and in terms of the remaining forest structure after harvesting (Martinez Pastur et al. 2008). This process may represent a bottleneck that conditions the establishment of a new seedling cohort (Hofgaard 1993). In the context of southern Patagonia forests, harvesting practices play a transformative role, altering light availability and soil moisture and consequently stimulating seedling growth (Martínez Pastur et al. 2009, 2011b). The residual canopy overstory assumes the role of a seed source and mitigates microclimatic shifts within managed stands (Chen et al. 1993, 1995; Martínez Pastur et al. 2011a).
Moreover, the regional climate exerts substantial influence (Isagi et al. 1997; Liebhold et al. 2004; Bahamonde et al. 2011), particularly during the stages of primordia development and flowering (McKee and Richards 1996; Martinez Pastur et al. 2008; Torres et al. 2015) shaping seed fall cycles and masting events (Piovesan and Adams 2001; Schauber et al. 2002; Bogdziewicz et al. 2019; Qiu et al. 2023) within managed and unmanaged forests. The response of N. pumilio seed production and its quality to annual weather variations might be notable (Richardson et al. 2005) in Tierra del Fuego (Torres et al. 2015). This area is strongly affected by both the Atlantic and Pacific oceans, the Antarctic climate, the Andes (Berman et al. 2013; Garreaud et al. 2013), and by large-scale climatic modes such as El Niño-Southern Oscillation (ENSO, with its phases La Niña and El Niño) and the Southern Annular Mode (SAM, with its positive and negative phases) (Silvestri and Vera 2009). Particularly, the synergy of ENSO and SAM has the potential to magnify climate fluctuations in South America (due to teleconnection phenomena), especially during spring and summer (Silvestri and Vera 2003; Fogt and Bromwich 2006, 2011; Han et al. 2017; Lim et al. 2019). This intricate relationship underscores the importance of utilizing climatic indices such as ENSO and SAM to comprehend the close connections between climate and forest dynamics, particularly in regions with limited climate data access. Interestingly, this relationship between climatic events and seeding patterns has been demonstrated in New Zealand by Fletcher (2015), and Rodríguez Souilla et al. (2023b) employed these indices to assess regeneration height growth, while Srur et al. (2018) studied the establishment of the N. pumilio tree line using similar metrics. Although climatic factors have been associated with annual variations in seed production, scant attention has been given to the qualitative attributes of seeds. In this sense, frost days and heavy winds during flowering might harm seed production, as not favourable climate conditions may act as lack of available resources during seed filling (Goszka and Snell 2020).
Thus, this study aims to explore factors governing the quality fluctuations of Nothofagus pumilio seeds. Particularly, we determine how it varies concerning forest management strategies (comparing harvested and unharvested forests), annual seed production (comparing years with differing levels), and the co-occurrence of climatic phenomena (comparing years with varying ENSO and SAM effects). Two main hypotheses are presented: (i) Seed quality shows discernible changes as a function of forest retention level and annual seed production; this is based on the premise that the generated microclimatic conditions (e.g., light exposure and precipitation) operate as fundamental thresholds capable of negatively influencing seed quality. (ii) Climatic events exert a substantial impact on seed quality, by modulating tree phenology in a way that varies according to the degree of retention applied.
Methods
Study area
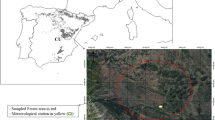
The study was conducted in monospecific Nothofagus pumilio forests located at Los Cerros ranch (54º 18ʹ SL, 67º 49ʹ WL) in the central area of Tierra del Fuego (Argentina). In these forests, variable retention harvesting was implemented for timber purposes. This harvesting method retains a percentage of the original forest structure by leaving aggregates (a circular patch of 30 m radius per ha) and dispersed dominant trees (10–15 m2 ha−1 of basal area) evenly distributed between the aggregates (Martínez Pastur et al. 2009). The studied forests have medium site quality (class II–III according to Martínez Pastur et al. 1997), with a dominant tree height of 22–24 m, which presented a range of 700–900 m3 ha−1 of total over-bark volume and 70–80 m2 ha−1 of basal area before harvesting. This study belongs to the long-term PEBANPA network (Parcelas de Ecología y Biodiversidad de Ambientes Naturales en Patagonia Austral, INTA-UNPA-CONICET, Argentina) (Peri et al. 2016).
Sampling design
Six stands (5–10 ha each) were selected for measurements: three stands were harvested with variable retention, while three stands of undisturbed forests were considered controls (unharvested forests, PF). Within the harvested stands, sampling areas were randomly located covering three different conditions according to the retention levels (see Martínez Pastur et al. 2011a): (i) within aggregate retention (AR), (ii) dispersed retention under the influence of AR (< 20 m from the aggregate edges) (DRI), and (iii) dispersed retention outside the influence of AR (DR). A total of 72 sampling areas (4 forest treatments, 3 stands and controls, 6 replicates) were selected, and monitored annually between 2011 and 2022. Forest structure corresponded to the same study area and time (2–17 YAH, equal to period studied) as Rodríguez-Souilla et al. (2023a, b) where forest treatments showed significant differences not only between treatments (Table 1), but also along the period studied, where stand basal area averaged 71.4 m2 ha−1 for PF, 60.4 m2 ha−1 for AR, 16.9 m2 ha−1 for DRI and 8.8 m2 ha−1 for DR, while crown cover averaged 86.6% for PF, 74.8% for AR, 48.9% for DRI and 40.2% for DR.
In the sampling areas, litter traps (50 cm long, 30 cm wide, 20 cm high hard plastic baskets) were randomly placed in control and harvested stands (4 treatments, 3 areas, 6 replicates = 72 litter traps), which were kept in their fixed sites throughout the monitored years (collecting the annual biomass production), protected with sticks around them to reduce the possibility of disturbance by livestock. Each trap was collected every May and manually sorted by seeds and leaves of N. pumilio trees; other components of the litter were discarded (e.g., small branches, fungi, epiphyte plants, miscellaneous, and other understory species). The methodology considered the outputs of Martínez Pastur et al. (2008), where most of the seeds fall between March and April and 90% of the leaves during April. Seeds were counted, and production at each sampling area was quantified (SP, million ha−1 yr−1). SP was classified into years of high (> 12 million ha−1 yr−1, n = 4), medium (4–12 million ha−1 yr−1, n = 4) and low production (< 4 million ha−1 yr−1, n = 4).
The collected seeds were classified as: predated (by birds or insects) and non-predated (sound, undamaged). From the total number of sound seeds, a representative sample was taken from each sample (n = 25, total of 12,389 seeds over period studied). The latter were differentiated into empty (without endosperm) and full. Full ones were classified as: aborted (only embryo inside); dead (black or grey endosperm). Immediately, for full and sound seeds, tetrazolium test (2,3,5-triphenyltetrazolium chloride) was conducted over the full seeds to determine seed viability. For this, embryos were incubated for 24 h in a water dilution of 0.1% solution in darkness at 25 °C (Cuevas 2000) and then classified by viable seeds or non-viable seeds according to the enzymatic activity (Moore 1985). The number of seeds in each category was counted, and the proportions were calculated relative to the total.
Regional climate data
In addition, the years were characterized using climate indices for the fruiting period of each year (ENSO and SAM averages between September and April), obtained from the websites: weather.gov/fwd/indices; stateoftheocean.osmc.noaa.gov/atm/sam.php. Each period was classified according to its prevalence of positive ( +) or negative (−) values of ENSO–SAM (ENSO+/SAM+ , ENSO+/SAM−, ENSO–/SAM+). ENSO−/SAM− situation was not registered between 2011 and 2022. Positive and negative ENSO values represent La Niña or El Niño events, for cold and warm phases at tropical latitudes, respectively. Positive SAM values represent westerly wind belt shifts southward or, analogously negative, contraction towards Antarctica.
Statistical analyses
For comparisons, different Kruskall–Wallis tests were performed because data did not meet the normality assumptions due to percentage values, using the Mann–Whitney U test to compare mean values: (i) 7 analyses for each seed quality category (insects, birds, empty, viable, not viable, deaths, abortion) considering forest treatments (PF, AR, DRI, DR) as main factors; (ii) 9 analyses for each seed quantity category (high, medium, low) and seed quality category (not predated, full, viable) considering forest treatments (PF, AR, DRI, DR) as main factors; (iii) 12 analyses for each seed quality category (not predated, full, viable) and forest treatment (PF, AR, DRI, DR) considering each ENSO–SAM combination (ENSO+/SAM+ , ENSO+/SAM−, ENSO−/SAM+) as main factors. In addition, non-linear regression models (quadratic polynomials) were fit for relations between SP and % Viable. Statistical analyses were performed using STATGRAPHICS Centurion XVI.I software (StatPoint Technologies Inc, US).
Results
The annual seed production exhibited variation between harvested and unharvested areas, with an average for the studied period of 9.35 million ha−1 yr−1 for PF, 7.16 million ha−1 yr−1 for AR, 2.25 million ha−1 yr−1 for DRI, and 1.08 million ha−1 yr−1 for DR. Viable seeds constituted 18.4% of the total seed population, with a notably higher proportion observed in PF and AR treatments. Conversely, empty seeds comprised the largest proportion (Fig. 1A), with higher proportions observed in harvested treatment areas. During the study period, the quality categories of seeds exhibited significant differences among the treatments, except for seeds predated by insects, where similar proportions were observed across all areas, although higher proportions were observed in DR.
A Average classification and percentages for the entire seed quality period studied. n = 12,389 seeds over period studied. B Percentage values of the final categories analyzed by Kruskall–Wallis nonparametric test, where different letters show significant differences by the Mann–Whitney U test (p < 0.05). H(p): Kruskal–Wallis statistic and associated probability. PF primary unharvested forests, AR aggregated retention, DRI dispersed retention under the influence of AR, DR dispersed retention without influence of AR
When examining seed quality across years characterized by varying production levels (Table 1), significant differences were observed, particularly with regard to full and viable seeds during high production years, which exhibited greater prevalence in PF and AR. For empty seeds, harvested areas presented 55 to 75%, lower for DRI. Furthermore, there was a consistent reduction in the proportion of full and viable seeds in correspondence with annual production levels across all treatments, with the lowest viability observed in years characterized by low seed production. Nevertheless, the not predated category displayed disparities only during years of low production, where it was also more prominent in PF.
Further, detailing seed viability for the treatments, the quadratic models developed reveal a goodness of fit ranging from 65.4% to 80.2% (see Fig. 2). These models showed that maximum viability is associated with a higher seed production. Notably, substantial distinctions are evident among treatments, particularly between harvested and unharvested areas. Specifically, AR, DRI, and DR treatments achieved comparable maximum proportion of viable seeds (39.1%, 33.0%, 37.4%, respectively), regardless of variations in annual seed production. In contrast, PF exhibited higher proportion of viable seeds, even in the context of maximum seed production (46.2%).
Quadratic polynomial fitted to each treatment with a coefficient of determination (r2) and significance level (*** P < 0.001). PF primary unharvested forests, AR aggregated retention, DRI dispersed retention under the influence of AR, DR dispersed retention without influence of AR. Each value represents the average number of seeds per year (million ha−1 yr−1) versus its corresponding viability (%) per treatment
When climate variability was integrated into the analyses (as presented in Fig. 3), the ENSO+/SAM+ combination exhibited elevated values and notable differences in full and viable seeds, particularly in PF and AR. Conversely, the ENSO−/SAM+ combination yielded higher proportions of non-predated seeds, with statistically significant differences observed in all treatments except for PF. It is worth noting that all treatments displayed similar patterns of seed proportions in response to different combinations of climate events, with these being most pronounced in unharvested areas.
Kruskall–Wallis non-parametric test for seed quality (%Not predated, %Full, %Viable) for each treatment (PF primary unharvested forests, AR aggregated retention, DRI dispersed retention under the influence of AR, DR dispersed retention without influence of AR) analysing ENSO/SAM combination occurrence. Different letters show significant differences (ab ≠ a, b) by the Mann–Whitney U test (p < 0.05)
Discussion
Variability in seed quality across different treatments and climatic events provides insights into the ecology of Nothofagus pumilio forests, where silvicultural interventions induce structural changes in the forest, particularly in terms of basal area and overstory canopy cover. Inter-annual variation in seed production is an inherent feature of Nothofagus forests (Ledgard and Cath 1983; Allen and Platt 1990; Monks and Kelly 2006), closely linked to annual litter-fall production (Rodríguez-Souilla et al. 2023a). In this sense, previous studies have attributed these fluctuations to physiological responses of trees to disturbances and climatic events (Isagi et al. 1997; Liebhold et al. 2004; Bahamonde et al. 2011; Torres et al. 2015). Our study focuses on how the quality of N. pumilio seeds varies in relation to annual production levels, silvicultural treatments, and the prevailing climatic conditions during flowering and fruit formation. Notably, on average, one out of every two healthy seeds is empty (highest proportion), with this ratio varying significantly according to high and low production years, a pattern partly explained by climatic events. For instance, in years of low seed production in unharvested forests (PF), only one out of every five seeds is full, and less than one out of every ten seeds is viable (Alley et al. 1998). This is in agreement with Allen and Platt (1990), where years of high seed production are accompanied by an increase in viability percentage (Veblen et al. 1996; Cellini 2010) up to a maximum level for Nothofagus solandri (as shown in Fig. 2), as for Nothofagus menziesii too (Burrows and Allen 1991). This represents that during high seed production years in PF, there are approximately 6.30 million ha−1 viable seeds, while in low production years, this value drops to 0.23 million ha−1 seeds (3.8%). Harvested areas consistently exhibit higher proportions of empty seeds and lower viability (Martinez Pastur et al. 2008; Cellini 2010), irrespective of annual seed production levels. This situation is possibly due to genetic incompatibilities (Donohue et al. 2005), different microclimatic conditions created post harvesting (Martinez Pastur et al. 2008; Cellini 2010) or the male-to-female flower ratio (Martinez Pastur et al. 2008; Soler et al. 2010), despite the high dispersion capacity of N. pumilio pollen (Kelly et al. 2001). This could potentially act as a bottleneck for post-disturbance regeneration, depending on the availability of high seed production years for cohort establishment (Rodríguez-Souilla et al. 2023b), and considering that these forests do not present a seed bank in the soil because seed viability is lost at the end of the growing season (Cuevas and Arroyo 1999). However, for our study site, annual seed input has proven sufficient to regenerate the forest to full density after logging (Rodríguez-Souilla et al. 2023b). In contrast, seed predation by insects and birds does not show variation according to annual seed production levels, except in harvested sited when ENSO+/SAM+ occurs, coinciding with conditions that lead to higher seed numbers and consequently a major food offer and increased annual growth of saplings (Rodríguez-Souilla et al. 2023b).
Climate variability appears to influence tree phenology, which, in turn, affects seed production (Richardson et al. 2005; Martínez Pastur et al. 2008; Allen et al. 2014). This suggests that synchronized flowering stages can result in a greater proportion of full and viable seeds (Koenig et al. 2015), even more so, in years of high seed production. While Nothofagus forests have been extensively studied in relation to climate, few studies in Tierra del Fuego (Argentina) have explored the connection between climate and the dynamics of native forests, including establishment, growth, mortality, and site quality variation (Torres et al. 2015; Rodríguez-Catón and Villalba 2018; Srur et al. 2018; Rodríguez-Souilla et al. 2023b). Positive and negative fluctuations in ENSO and SAM have discernible effects on local temperatures and precipitation (Rodríguez-Souilla et al. 2023b), acting as limiting factors in forest development for Nothofagus pumilio, as evidenced in our study. The influence of ENSO and SAM on physiological processes in different forests has been documented (Schauber et al. 2002; Hadad et al. 2021), with their effects being modified by the seasonality of temperature and rainfall in different regions. Moreover, the ongoing climate change process (IPCC 2022) and how climatic events, such as extreme ENSO or SAM, affect the region, along with the increasing frequency and persistence of these events (Rodríguez-Souilla et al. 2023b), must be taken into account. In our study, warmer springs (ENSO+/SAM+) tend to result in more consistent and abundant viable seeds, result of abundant flowering, enhancing pollination success (Allen and Platt 1990). Differences in magnitude and significance were registered for harvested and unharvested areas, denoting differences in their climatic resilience (Thompson et al. 2009). This, in turn, contributes to higher seed production, particularly during the critical phases of floral primordial development (Schauber et al. 2002; Martinez Pastur et al. 2007), provided that stress thresholds disrupting flowering and fruit formation are not reached: (i) frost damaged flowers may be incapable of being fertilized, (ii) low temperatures can negatively affect pollen viability and pollen tube growth, (iii) pollinator activity may be reduced in low temperatures (Tremblay et al. 2002; Qiu et al 2023), and (iv) strong winds can cause damage and massive fall of flowers or newly formed seeds. Furthermore, warmer spring and summer temperatures promote increased net carbon availability (Allen and Platt 1990; Richardson et al. 2005). This does not mean that climatic conditions will predict with absolute certainty the quality of seeds that will result in the seed fall quantity. Then, the need for individuals to recover from a year of high seed production and consequently high carbon demand must be taken into account (Allen and Platt 1990; Silvertown 2008). Furthermore, it is essential to recognize that the increased variability in climate and the occurrence of extreme events, which individuals are not accustomed to, can have adverse effects on the regular growth of trees, particularly concerning to seed viability (Goszka and Snell 2020). Besides, various ecological processes, including regeneration at the altitudinal limit of N. pumilio and ring width variations in Araucaria araucana (Hadad and Roig 2016), have been associated with positive SAM phases (in consonance with high variability in climate and occurrence of extreme events) in northern Patagonia (Srur et al. 2018). These processes underscore how higher temperatures, coupled with positive ENSO events, facilitate seedling establishment and growth at tree lines, ultimately driving maximum seed production (Fletcher 2015). Torres et al. (2015) also identified connections between climatic variables and annual seed production in Tierra del Fuego forests, suggesting that these climate events may shape the observed patterns, particularly rainfalls. The implications of these findings are significant for forest management and ecosystem service maintenance, providing valuable insights into forest dynamics related to seeding, a crucial process for forest regeneration (Rodríguez-Souilla et al. 2023b), especially in the context of high climate variability and forest vulnerability to disturbances.
Conclusions
Forest management of Nothofagus pumilio forests should consider not only annual seed production but also the effects of climate events on their quality, particularly viability. Seed quality exhibited differences across treatments, with empty seeds being more prevalent in harvested areas. The variation in seed quality observed, particularly during high and low production years, suggests that climatic events play a pivotal role in determining the viability of seeds. Furthermore, our findings highlight the significant influence of climatic events (ENSO and SAM), on seed production and quality. ENSO+/SAM+ combination tended to result in more consistent and abundant viable seeds, reflecting the importance of favourable climatic conditions during floral development and pollination. Our work emphasizes the importance of assessing seed quality in studying regeneration of N. pumilio forests. This aspect is crucial to ensuring the success of interventions, as it determines the creation of a seedling bank that will regenerate the harvested stands in a context of high climate variability and ongoing climate change. The implications of our findings extend beyond ecological understanding. They have direct relevance to forest management practices and the maintenance of ecosystem services. As climate variability increases and extreme events become more frequent, understanding how these factors influence seed quality is essential for ensuring the sustainability of N. pumilio forests. This knowledge can inform broader discussions about climate resilience and adaptation strategies for forests worldwide.
Availability of data and materials
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- SP:
-
Seed production
- YAH:
-
Years after harvesting
- ENSO:
-
El Niño-Southern Oscillation
- SAM:
-
Southern annular mode
- PF:
-
Primary unharvested forest
- AR:
-
Aggregate retention
- DRI:
-
Dispersed retention under the influence of AR
- DR:
-
Dispersed retention outside the influence of AR
References
Alan JR, Veblen TT (1993) Structure and tree-fall gap dynamics of old-growth Nothofagus forests in Tierra del Fuego, Argentiana. J Veg Sci 4(5):641–654. https://doi.org/10.2307/3236129
Allen RB, Platt KH (1990) Annual seedfall variation in Nothofagus solandri (Fagaceae), Canterbury, New Zealand. Oikos 57:199–206. https://doi.org/10.2307/3565940
Allen RB, Hurst JM, Portier J, Richardson SJ (2014) Elevation dependent response of tree mast seeding to climate change over 45 years. Ecol Evol 4:3525–3537. https://doi.org/10.1002/ece3.1210
Alley JC, Fitzgerald BM, Berben PH, Haslett SJ (1998) Annual and seasonal patterns of litter-fall of hard beech (Nothofagus truncata) and silver beech (Nothofagus menziesii) in relation to reproduction. NZ J Bot 36(3):453–464. https://doi.org/10.1080/0028825X.1998.9512583
Bahamonde HA, Peri PL, Monelos LH, Martínez Pastur G (2011) Aspectos ecológicos de la regeneración por semillas en bosques nativos de Nothofagus antarctica en Patagonia Sur, Argentina. Bosque 32(1):20–29. https://doi.org/10.4067/S0717-92002011000100003
Bahamonde HA, Peri PL, Monelos LH, Martínez Pastur G (2013) Regeneración por semillas en bosques nativos de Nothofagus antarctica bajo uso silvopastoril en Patagonia Sur, Argentina. Bosque 34(1):89–101. https://doi.org/10.4067/S0717-92002013000100011
Berman AL, Silvestri G, Compagnucci R (2013) On the variability of seasonal temperature in southern South America. Clim Dyn 40:1863–1878. https://doi.org/10.1007/s00382-012-1596-5
Bogdziewicz M, Szymkowiak J, Fernández-Martínez M, Peñuelas J, Espelta JM (2019) The effects of local climate on the correlation between weather and seed production differ in two species with contrasting masting habit. Agric For Meteorol 268:109–115. https://doi.org/10.1016/j.agrformet.2019.01.016
Burrows LE, Allen RB (1991) Silver beech (Nothofagus menziesii (Hook. F.) Oerst.) seedfall patterns in the Takitimu Range, South Island, New Zealand. NZ J Bot 29(4):361–365. https://doi.org/10.1080/0028825X.1991.10415489
Cellini JM (2010) Estructura y regeneración bajo distintas propuestas de manejo de bosques de Nothofagus pumilio (Poepp et. Endl) Krasser en Tierra del Fuego, Argentina. Doctoral Thesis. Universidad Nacional de La Plata. 157 pp. https://doi.org/10.35537/10915/42453
Chen J, Franklin JF, Spies TA (1993) Contrasting microclimates among clearcut, edge, and interior of old-growth Douglas-fir forest. Agric For Meteorol 63:219–237. https://doi.org/10.1016/0168-1923(93)90061-L
Chen J, Franklin JF, Spies TA (1995) Growing-season microclimate gradients from clearcut edges into old-growth Douglas-fir forests. Ecol Appl 5:74–86. https://doi.org/10.2307/1942053
Cleland EE, Chuine I, Menzel A, Mooney HA, Schwartz MD (2007) Shifting plant phenology in response to global change. Trends Ecol Evol 22(7):357–365. https://doi.org/10.1016/j.tree.2007.04.003
Cuevas J (2000) Tree recruitment at the Nothofagus pumilio alpine timberline in Tierra del Fuego, Chile. J Ecol 88:840–855. https://doi.org/10.1046/j.1365-2745.2000.00497.x
Cuevas J, Arroyo MK (1999) Ausencia de banco de semillas persistente en Nothofagus pumilio (Fagaceae) en Tierra del Fuego, Chile. Rev Chil Hist Nat 72:73–82
Donohue K, Polisetty CR, Wender NJ (2005) Genetic basis and consequences of niche construction: plasticity-induced genetic constraints on the evolution of seed dispersal in Arabidopsis thaliana. Am Nat 165:537–550. https://doi.org/10.1086/429162
Donoso C (1975) Distribución ecológica de las especies de Nothofagus en la zona mesomórfica. Boletín técnico nº 33, Facultad de Ciencias Forestales de la Universidad de Chile. 39 pp.
Fenner M (1985) Seed ecology. Chapman & Hall Ltd, London
Fletcher M (2015) Mast seeding and the El Niño-Southern Oscillation: a long-term relationship? Plant Ecol 216(4):527–533. https://doi.org/10.1007/s11258-015-0456-x
Fogt R, Bromwich D (2006) Decadal variability of the SAM teleconnection to the high-latitude South Pacific governed by coupling with the Southern Annular Mode. J Clim 19(6):979–997. https://doi.org/10.1175/JCLI3671.1
Garreaud R, Lopez P, Minvielle M, Rojas M (2013) Large-scale control on the Patagonian climate. J Clim 26:215–230. https://doi.org/10.1175/JCLI-D-12-00001.1
Goszka AR, Snell RS (2020) Seed quality and seed quantity in red maple depends on weather and individual tree characteristics. Ecol Evol 10(23):13109–13121. https://doi.org/10.1002/ece3.6900
Hacket-Pain A, Ascoli D, Berretti R, Mencuccini M, Motta R, Nola P, Piussi P, Ruffinatto F, Vacchiano G (2019) Temperature and masting control Norway spruce growth, but with high individual tree variability. For Ecol Manage 438:142–150. https://doi.org/10.1016/j.foreco.2019.02.014
Hadad MA, Roig Juñent FA (2016) Sex-related climate sensitivity of Araucaria araucana Patagonian forest-steppe ecotone. For Ecol Manage 362:130–141. https://doi.org/10.1016/j.foreco.2015.11.049
Hadad M, Roig FA, Arco Molina JG, Hacket-Pain A (2021) Growth of male and female Araucaria araucana trees respond differently to regional mast events, creating sex-specific patterns in their tree-ring chronologies. Ecol Indic 122:107245. https://doi.org/10.1016/j.ecolind.2020.107245
Han T, Wang H, Sun J (2017) Strengthened relationship between the Antarctic Oscillation and ENSO after the mid-1990s during austral spring. Adv Atmos Sci 34:54–65. https://doi.org/10.1007/s00376-016-6143-6
Hernández IM, Santa Regina I, Gallardo JF (1992) Dinámica de la descomposición de la hojarasca forestal en bosques de la Cuenca del Duero (Provincia de Zamora): modelización de la pérdida de peso. Arid Soil Res Rehab 6:339–355. https://doi.org/10.1080/15324989209381328
Herrera CM, Jordano P, Guitián J, Traveset A (1998) Annual variability in seed production by woody plants and the masting concept: reassessment of principles and relationship to pollination and seed dispersal. Am Nat 152:576–594. https://doi.org/10.1086/286191
Hofgaard A (1993) Seed rain quantity and quality, 1984–1992, in a high altitude old-growth spruce forest, northern Sweden. New Phytol 125(3):635–640. https://doi.org/10.1111/j.1469-8137.1993.tb03913.x
IPCC (2022) Climate Change 2022: impacts, adaptation, and vulnerability. Contribution of Working Group II to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change. In: Pörtner HO, Roberts DC, Tignor M, Poloczanska ES, Mintenbeck K, Alegría A, Craig M, Langsdorf S, Löschke S, Möller V, Okem A, Rama B (eds.). Cambridge University Press. Cambridge, UK and New York, USA. https://doi.org/10.1017/9781009325844.
Isagi Y, Kawahara T, Kamo K, Ito H (1997) Net production and carbon cycling in a bamboo Phyllostachys pubescens stand. Plant Ecol 130(1):41–52. https://doi.org/10.1023/A:1009711814070
Kelly D, Hart D, Allen R (2001) Evaluating the wind pollination benefits of masting seeding. Ecology 82:117–126. https://doi.org/10.1890/0012-9658(2001)082[0117:ETWPBO]2.0.CO;2
Kitayama K, Ushio M, Aiba SI (2021) Temperature is a dominant driver of distinct annual seasonality of leaf litter production of equatorial tropical rain forests. J Ecol 109(2):727–736. https://doi.org/10.1111/1365-2745.13500
Koenig WD, Knops JMH, Carmen WJ, Pearse IS (2015) What drives masting? The phenological synchrony hypothesis. Ecology 96(1):184–192. https://doi.org/10.1890/14-0819.1
Koyejo AO, Okpara DA, Onyeonagu CC, Eteng EU (2020) Effects of climatic variations on leaf litter production in A. floribunda agroforestry system in South-East Nigeria. Niger J Soil Sci 30(2):42–48
LaMontagne JM, Pearse IS, Greene DF, Koenig WD (2020) Mast seeding patterns are asynchronous at a continental scale. Nat Plants 6:460–465. https://doi.org/10.1038/s41477-020-0647-x
Ledgard N, Cath P (1983) Seed of New Zealand Nothofagus species: studies of seed weight, viability, shape and the effect of varying stratification periods. NZ J For 28:150–162
Liebhold AM, Koening WD, Bjornstad ON (2004) Spatial synchrony in population dynamics. Ann Rev Eol Evol Syst 35:467–490. https://doi.org/10.1146/annurev.ecolsys.34.011802.132516
Lim EP, Hendon HH, Hope P, Chung C, Delage F, McPhaden MJ (2019) Continuation of tropical Pacific Ocean temperature trend may weaken extreme El Niño and its linkage to the Southern Annular Mode. Sci Rep 9:e17044. https://doi.org/10.1038/s41598-019-53371-3
Marchelli P, Gallo LA (1999) Annual and geographic variation in seed traits of Argentinean populations of southern beech Nothofagus nervosa (Phil.) Dim. et Mil. For Ecol Manage 121:239–250. https://doi.org/10.1080/0028825X.2021.1920433
Martínez Pastur G, Peri PL, Vukasovic R, Vaccaro S, Piriz Carrillo V (1997) Site index equation for Nothofagus pumilio Patagonian forest. Phyton 6(1/2):55–60
Martínez Pastur G, Lencinas MV, Peri PL, Arena M (2007) Photosynthetic plasticity of Nothofagus pumilio seedlings to light intensity and soil moisture. Forest Ecology and Management 243(2):274–282. https://doi.org/10.1016/j.foreco.2007.03.034
Martínez Pastur G, Cellini JM, Peri PL, Lencinas MV, Gallo E, Soler RM (2009) Alternative silviculture with variable retention in timber management of South Patagonia. For Ecol Manage 258:436–443. https://doi.org/10.1016/j.foreco.2011.06.021
Martínez Pastur G, Cellini JM, Lencinas MV, Barrera MD, Peri PL (2011a) Environmental variables influencing regeneration of Nothofagus pumilio in a system with combined aggregated and dispersed retention. For Ecol Manage 261:178–186. https://doi.org/10.1016/j.foreco.2010.10.002
Martínez Pastur G, Peri PL, Cellini JM, Lencinas MV, Barrera MD, Ivancich H (2011b) Canopy structure analysis for estimating forest regeneration dynamics and growth in Nothofagus pumilio forests. Ann For Sci 68:587–594. https://doi.org/10.1007/s13595-011-0059-1
Martínez Pastur G, Soler R, Pulido F, Lencinas MV (2013) Variable retention harvesting influences biotic and abiotic drivers of regeneration in Nothofagus pumilio southern Patagonian forests. For Ecol Manage 289:106–114. https://doi.org/10.1016/j.foreco.2012.09.032
Martínez Pastur G, Peri PL, Lencinas MV, Soler R, Bahamonde HA, Valenzuela A, Cabello JL, Anderson CB (2016) Investigación socio-ecológica a largo plazo en la Patagonia Austral: Estrategias interdisciplinarias para lograr la conservación de los recursos naturales a través de un manejo sustentable bajo escenarios de cambio global. Ecosistemas 25(1):49–57. https://doi.org/10.7818/ECOS.2016.25-1.06
Martínez Pastur G, Rosas YM, Toro Manríquez M, Huertas Herrera A, Miller JA, Cellini JM, Barrera MD, Peri PL, Lencinas MV (2019) Knowledge arising from long-term research of variable retention harvesting in Tierra del Fuego: where do we go from here? Ecol Process 8:24. https://doi.org/10.1186/s13717-019-0177-5
Martinez Pastur G, Lencinas MV, Vukasovic R, Peri PL, Díaz B, Cellini JM (2004) Turno de corta y posibilidad de los bosques de lenga (Nothofagus pumilio) en Tierra del Fuego (Argentina). Bosque 25(1):29–42. https://doi.org/10.4067/S0717-92002004000100003
Martinez Pastur G, Lencinas MV, Peri PL, Cellini JM (2008) Flowering and seeding patterns in unmanaged and managed Nothofagus pumilio south Patagonian forests. Forstarchiv 79:60–65. https://doi.org/10.2376/0300-4112-79-60
McKee J, Richards AJ (1996) Variation in seed production and germinability in common reed (Phragmites australis) in Britain and France with respect to climate. New Phytol 133:233–243. https://doi.org/10.1111/j.1469-8137.1996.tb01890.x
Monks A, Kelly D (2006) Testing the resource-matching hypothesis in the mast seeding tree Nothofagus truncata (Fagaceae). Austral Ecol 31:366–375. https://doi.org/10.1111/j.1442-9993.2006.01565.x
Moore RP (1985) Handbook on tetrazolium testing. International Seed Testing Association, Zurich, Switzerland
Övergaard R, Gemmel P, Karlsson M (2007) Effects of weather conditions on mast year frequency in beech (Fagus sylvatica L.). Forestry 80(5):555–565. https://doi.org/10.1093/forestry/cpm020
Peri PL, Lencinas MV, Bousson J, Lasagno R, Soler R, Bahamonde HA, Martínez Pastur G (2016) Biodiversity and ecological long-term plots in southern Patagonia to support sustainable land management: the case of PEBANPA network. J Nat Conserv 34:51–64. https://doi.org/10.1016/j.jnc.2016.09.003
Piovesan G, Adams JM (2001) Masting behaviour in beech: linking reproduction and climatic variation. Can J Bot 79(9):1039–1047. https://doi.org/10.1139/b01-089
Premoli AC, Kitzberger T (2005) Regeneration mode affects spatial genetic structure of Nothofagus dombeyi forests. Mol Ecol 14(8):2319–2329. https://doi.org/10.1111/j.1365-294x.2005.02629.x
Qiu T, Aravena MC, Ascoli D, Bergeron Y, Bogdziewicz M, Boivin T et al (2023) Masting is uncommon in trees that depend on mutualist dispersers in the context of global climate and fertility gradients. Nat Plants 9:1044–1056. https://doi.org/10.1038/s41477-023-01446-5
Richardson SJ, Allen RB, Whitehead D, Carswell FE, Ruscoe WA, Platt KH (2005) Climate and net carbon availability determine temporal patterns of seed production by Nothofagus. Ecology 86:972–981. https://doi.org/10.1890/04-0863
Richter L, Frangi J (1992) Bases ecológicas para el manejo del bosque de Nothofagus pumilio de Tierra del Fuego. Revista Fac Agron 68:35–52
Rodríguez Catón M, Villalba R (2018) Indicadores del decaimiento en bosques de Nothofagus pumilio en el norte de la Patagonia Argentina. Madera y Bosques 24(2):e2421588. https://doi.org/10.21829/myb.2018.2421588
Rodríguez-Souilla J, Lencinas MV, Cellini JM, Chaves JE, Roig FA, Peri PL, Martínez Pastur G (2023a) Seed fall and leaf litter relationships in Nothofagus pumilio forests: changes according to retention levels and years after harvesting. Trees 37:583–597. https://doi.org/10.1007/s00468-022-02365-2
Rodríguez-Souilla J, Cellini JM, Lencinas MV, Roig FA, Chaves JE, Aravena Acuña MC, Peri PL, Martínez Pastur G (2023b) Variable retention harvesting and climate variations influence over natural regeneration dynamics in Nothofagus pumilio forests of Southern Patagonia. For Ecol Manage 544:e121221. https://doi.org/10.1016/j.foreco.2023.121221
Rosenfeld JM, Navarro Cerrillo R, Guzman Alvarez JR (2006) Regeneration of Nothofagus pumilio (Poepp. et Endl.) Krasser forests after five years of seed tree cutting. J Environ Manage 78(1):44–51. https://doi.org/10.1016/j.jenvman.2005.03.009
Schauber EM, Kelly D, Turchin P, Simon C, Lee WG, Allen RB, Payton IJ, Wilson PR, Cowan PE, Brockie RE (2002) Masting by eighteen New Zealand plant species: the role of temperature as a synchronizing cue. Ecology 83(5):1214–1225. https://doi.org/10.1890/0012-9658(2002)083[1214:MBENZP]2.0.CO;2
Silvertown JW (2008) The evolutionary ecology of mast seeding in trees. Biol J Linn Soc 14(2):235–250. https://doi.org/10.1111/j.1095-8312.1980.tb00107.x
Silvestri G, Vera C (2003) Antarctic Oscillation signal on precipitation anomalies over southeastern South America. Geophys Res Let 30(21):e2115. https://doi.org/10.1029/2003GL018277
Silvestri G, Vera C (2009) Nonstationary impacts of the Southern Annular Mode on Southern Hemisphere climate. J Clim 22(22):6142–6148. https://doi.org/10.1175/2009JCLI3036.1
Soler RM, Martínez Pastur G, Lencinas MV, Peri PL (2010) Flowering and seeding patterns in primary, secondary and managed Nothofagus antarctica South Patagonian forests. NZ J Bot 48:63–73. https://doi.org/10.1080/0028825X.2010.482959
Srur AM, Villalba R, Rodríguez-Catón M, Amoroso MM, Marcotti E (2018) Climate and Nothofagus pumilio establishment at upper treelines in the Patagonian Andes. Front Earth Sci 6:57. https://doi.org/10.3389/feart.2018.00057
Thompson I, Mackey B, McNulty S, Mosseler A (2009) Forest resilience, biodiversity, and climate change. In: A synthesis of the biodiversity/resilience/stability relationship in forest ecosystems. Technical Series. Secretariat of the Convention on Biological Diversity. Montreal, Canada
Toro Manríquez M, Mestre L, Lencinas MV, Promis Á, Martínez Pastur G, Soler RM (2016) Flowering and seeding patterns in pure and mixed Nothofagus forests in Southern Patagonia. Ecol Process 5(1):21. https://doi.org/10.1186/s13717-016-0065-1
Torres AD, Cellini JM, Lencinas MV, Barrera MD, Soler RM, Díaz-Delgado R, Martínez Pastur G (2015) Seed production and recruitment in primary and harvested Nothofagus pumilio forests: influence of regional climate and years after cuttings. For Syst 24(1):1–11. https://doi.org/10.5424/fs/2015241-06403
Tremblay MF, Bergeron Y, Lalonde D, Mauffette Y (2002) The potential effects of sexual reproduction and seedling recruitment on the maintenance of red maple (Acer rubrum L.) populations at the northern limit of the species range. J Biogeogr 29:365–373. https://doi.org/10.1046/j.1365-2699.2002.00665.x
Veblen TT, Donoso C, Kitzberger T, Rebertus AJ (1996) Ecology of southern Chilean and Argentinean Nothofagus forests. In: Veblen TT, Hill RS, Read J (eds) Ecology and biogeography of Nothofagus forests. Yale University Press, New Heaven, London
Wardle JA (1970) The ecology of Nothofagus solandri: 3. Regeneration. NZ J Bot 8:571–608. https://doi.org/10.1080/0028825X.1970.10430161
Acknowledgements
To the researchers, technicians, students and landowners (ranch and sawmill companies) that supported this research, without which it would have been impossible to obtain the valuable information used in this work.
Funding
This research was funded by the grant PIP 2021–2023 Nº 0871 “Resilience of managed Nothofagus pumilio forests against harvesting, climate change and extreme events in Tierra del Fuego” of CONICET (Argentina).
Author information
Authors and Affiliations
Contributions
GMP and PLP conceived and designed the experiments, MVL, JMC, JEC, and JRS collaborated in fieldwork and analysed the data, and FAR helped in writing the manuscript. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Rodríguez-Souilla, J., Chaves, J.E., Lencinas, M.V. et al. Quality evaluation of Nothofagus pumilio seeds linked to forest management and climatic events. Ecol Process 13, 7 (2024). https://doi.org/10.1186/s13717-024-00485-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s13717-024-00485-w