Abstract
The kidney is a complex organ involved in the excretion of metabolic products as well as the regulation of body fluids, osmolarity, and homeostatic status. These functions are influenced in large part by alterations in the regional distribution of blood flow between the renal cortex and medulla. Renal perfusion is therefore a key determinant of glomerular filtration. Therefore the quantification of regional renal perfusion could provide important insights into renal function and renal (patho)physiology. Arterial spin labeling (ASL) based perfusion MRI techniques, can offer a noninvasive and reproducible way of measuring renal perfusion in animal models. This chapter addresses the basic concept of ASL-MRI.
This chapter is based upon work from the COST Action PARENCHIMA, a community-driven network funded by the European Cooperation in Science and Technology (COST) program of the European Union, which aims to improve the reproducibility and standardization of renal MRI biomarkers. This introduction chapter is complemented by two separate chapters describing the experimental procedure and data analysis.
You have full access to this open access chapter, Download protocol PDF
Similar content being viewed by others
Key words
1 Introduction
Kidneys are highly perfused organs in the body as they receive 20–25% blood of the resting cardiac output. High blood flow to the kidneys ensures adequate delivery of plasma to be filtered by the glomeruli, a physiological process necessary for the regulation of homeostasis. Therefore, the changes in renal blood flow (RBF) will also change the glomerular filtration rate (GFR) and, consequently, the volume and composition of body fluids. Thus, even a slight change in renal perfusion that exceeds renal autoregulation can have a significant impact on the kidney function. The complex interaction between RBF, renal oxygen delivery, renal oxygen consumption, and GFR made measuring of renal perfusion an attractive target to understand the regulation and homeostasis of renal tissue in health and disease.
Renal perfusion, defined as the blood flow that passes through a unit mass of renal tissue within a given time (mL/min/g) that is also a key determinant of glomerular filtration. A unique aspect of the renal perfusion is that the blood flow is regionally specific. Due to the lack of a reliable technic which allowed to precisely measure the regional RBF, it is roughly estimated that 80–85% of the total renal blood flow supplies renal cortex, 10–15% outer medulla and only 1–5% inner medulla [1].The cortical blood flow is tightly regulated by well-documented mechanisms. Conversely, medullary blood flow regulation is less understood and difficult to measure [2]. The capillary density and surface area is higher in the medulla than in the cortex at least in the mice. It is very challenging to measure the medulla renal perfusion because of the complexity of the microcirculation and its tight connection of arterioles from the cortex. Although inner renal medulla receives less than 1–5% of the total renal blood flow, but changes in this region can still have major effect on electros and water homeostasis and control of arterial blood pressure [3]. Regardless, to understand the physiological regulation of renal perfusion and the role of its dysregulation in separate regions will require methods that allow quantification of renal perfusion both at the whole organ level and at the local tissue level. Most of the imaging techniques for assessing renal perfusion that have been used in humans and animals have significant limitations (Table 1) and by far there is still no gold-standard technique available. For instance, renal perfusion can be measured with dynamic contrast-enhanced MRI (DCE-MRI). However, the major drawback of DCE-MRI is the involvement of contrast media for the measurement of perfusion. The contrast media are easily filtered by renal glomeruli and hence it is difficult to obtain absolute quantification. Other known side effects, such as the development of nephrogenic systemic fibrosis, also limit their application to patients with renal deficits.
A noninvasive, practical, robust, and reproducible method to measure renal perfusion is of great interest but remains elusive, both in the clinic and basic research. The Arterial spin labeling (ASL) MRI technique introduced three decades ago for quantifying tissue perfusion [6, 7], is potentially the only available technique to meet all these needs. ASL is a MRI method generating endogenous contrast specifically related to capillary blood flow. It is therefore free of exogenous contrast media. By magnetically labeling arterial blood proton spins with inversion or saturation as a freely diffusible endogenous tracer, ASL allows for the measurement of RBF. Originally developed for measuring cerebral perfusion, the use of endogenous blood signal to obtain tissue perfusion information is particularly relevant for multiple and longitudinal imaging of patients. This attribute is also important for preclinical imaging applications, as multiple tail-vein injections and/or catheterizations can cause local inflammation and necrosis resulting in reduced access to veins. The mature and reliable animal models combined with reproducible imaging techniques are an indispensable part of scientifically designed kidney disease studies, and play an important role in resolving the general bottleneck in renal disease treatment that is usually delayed due to difficulties in imaging disease progression.
The ASL approach has been rigorously studied in various preclinical applications including correlating the degree of acute rejection and renal cortical blood flow in rat allograft model [8], comparison of imaging results with histologic examination in ischemia-induced acute kidney injury (AKI) [9], early detection of antiangiogenic treatment responses in a mouse xenograft tumor model [10] and validation of the pathophysiological process of contrast-induced acute kidney injury in rats [11]. In this chapter, we address the basic concept and the developments of renal ASL with the emphasis on kidney disease models in rodents.
This introduction chapter is complemented by two separate chapters describing the experimental procedure and data analysis, which are part of this book.
This chapter is part of the book Pohlmann A, Niendorf T (eds) (2020) Preclinical MRI of the Kidney—Methods and Protocols. Springer, New York.
2 Measurement Concept
2.1 Basic Concept of ASL-MRI
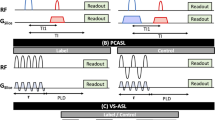
ASL provides quantitative parametric images of tissue perfusion. It uses a modified proton magnetization of arterial blood as an endogenous, freely diffusible contrast medium which determines the delivery rate of oxygen and nutrients to the capillary bed and is expressed as the volume of blood traveling through a gram of tissue per minute (mL 100 g−1 min−1). The basic ASL experiment collects two image types, referred to as the “label” (or tag) and “control” (or nonlabeled) image respectively. The label is applied using radiofrequency (RF) pulses to alter the longitudinal magnetization of protons in the arterial blood before it enters the desired imaging plane, and an image is collected after a time delay. The control image is acquired at the same time delay without labeling the arterial blood protons. Provided that the inverted magnetization of the inflowing blood is the only difference between the control and label images, a simple subtraction of the label from the control image yields a perfusion-weighted image in which signal intensity is proportional to perfusion. All ASL pulse sequences consist of two components: (1) a preparation module to magnetically label inflowing blood, and (2) an imaging readout scheme to generate paired images of the target tissue under “control” and “tagged” conditions. ASL methods may be classified as to how each of these modules is constructed (Fig. 1).
Left: All ASL pulse sequences consist of two modules: a preparation module to magnetically label flowing blood, and a readout module to generate paired images. Right: Schematic showing different labeling modules. A single or a train of radiofrequency pulse for labeling followed by a postlabel delay before the image is acquired within one TR.
2.2 Labeling Methods and Strategies for Kidney perfusion Measurement in Rodents
There are two main labeling strategies that have been developed: pulsed ASL (PASL) [12] and continuous ASL (CASL) (Fig. 1, left). Pulsed and continuous ASL labeling methods differ fundamentally in both the spatial extent and the duration of the labeling (Fig. 1, right), and these differences give rise to the strengths and weaknesses of each approach [13]. PASL uses a single short pulse or a limited number of pulses, to invert a thick slab of arterial water spins. It can be further divided into those that label spins asymmetrically with respect to the plane of imaging (Signal Targeting by Alternating Radiofrequency pulses; STAR [14] and Proximal Inversion with Control of Off-Resonance Effects; PICORE [15] or symmetrically (Flow-sensitive Alternating Inversion Recovery; FAIR) [16]. In contrast, CASL techniques use a continuous radiofrequency wave in a plane upstream of the imaging volume for generating a flow-driven inversion of arterial blood flowing through that plane. Later, a modified form called pseudo-continuous arterial spin labeling (pCASL) has been developed, that uses a long series of short RF-pulses together with a strong slice-selection gradient, also resulting in a flow-driven inversion of upstream blood proton spins. pCASL thus combines favorable features of CASL (high signal-to-noise) with those of PASL (lower energy deposition). Today most commercially available ASL products are based on pCASL or a PASL variant. Table 2 shows examples of preclinical studies using ASL for measuring RBF in animal models and list each of the labeling and readout methods.
2.2.1 Pulsed ASL (PASL)
The PASL variant (FAIR) is the major method used for measuring renal perfusion in rodents due to its ease of implementation. FAIR uses a single long-duration RF pulse which is applied to invert the arterial magnetization as blood flows through the labeling plane while a gradient is applied in the direction of arterial flow. In FAIR ASL the magnetization is usually prepared using a global inversion which is referred to as the “labeling” and a slice selective inversion which is referred to as “control.” The sequence begins with a spatially selective inversion pulse limited to a small region in and around the imaged slice. In the standard labeling scheme the selective inversion slice (control) is in the same plane as the imaging slice with a thickness of 1.5–5 times the thickness of the imaging slice. The inversion pulse is usually slightly wider than the imaging slice to minimize artifacts and ensure uniform inversion. For the “labeling,” the same inversion pulse is applied but without the slice-selection gradient. This causes an inversion of spins within the entire sensitive volume of the RF coil (Fig. 2a). By subtracting the control image from the labeling image, a perfusion-weighted image (PWI) is formed, which with the appropriate modeling can be quantified using a perfusion map in units of mL/100 g/min. In various preclinical MRI situations, for instance with dedicated small brain birdcage transmit/receive (TX/RX) resonators, the transmit coil used may not be long enough to provide a sufficiently global inversion for FAIR perfusion measurements.
Basic principle of arterial spin labeling (ASL) acquisition. It consists of a differential technique during which two acquisitions are carried out: one with the labeling of arterial protons (label image) and a control image. Arterial blood is labelled by inverting its magnetization. After an inflow time (inversion time), which allows the labeled blood to flow into the tissue of interest, an image is acquired with an appropriate readout sequence. (a) FAIR uses a nonselective inversion pulse for the labeled images that becomes selective with the addition of a slice selection gradient for the control. (b) The magnetic labeling of arterial protons is carried out upstream from the volume of interest, by radiofrequency pulses. The labeled protons then migrate via the arterial vessels toward the tissue where they pass from the capillary compartment to the extravascular compartment (example shown: pCASL)
2.2.2 Continuous ASL (CASL) and Pseudo-Continuous ASL (pCASL)
CASL uses a continuous flow-driven radiofrequency inversion of blood upstream of the imaging volume. The radiofrequency wave is transmitted either by a dedicated, separate labeling coil or by the volume transmit coil that is also used for imaging. The latter approach has the advantage that no special hardware components are required but the disadvantage of depositing high amounts of RF energy leading to magnetization transfer contrast that has to be properly corrected. One drawback of CASL labeling approach is the consequently high transmitter duty-cycle requirement.
The pCASL technique has evolved from the original CASL technique in both, brain and kidney applications [24]. pCASL uses series of short and high-power RF and gradient pulses instead of the long RF pulses used in CASL to generate a flow-driven inversion. Compared to FAIR, pseudo-continuous ASL (pCASL) provides significant advantages such as increased SNR and better definition of the labeling bolus. FAIR (pulsed ASL) may be attractive for renal imaging because it can label closer to the tissue, but pCASL offers the potential advantage of high SNR of the kidneys and the whole abdomen, which would be desirable in some specific applications (e.g., metastatic). An earlier study compared the performances of FAIR and pCASL in terms of sensitivity, absolute quantification, reproducibility, and flexibility of implementation using healthy mice [18]. It was reported that the application of pCASL at 11.75 T scanner had higher sensitivity compared to FAIR (> þ20%). Finally, since pCASL can be straightforwardly used in multislice mode and in any imaging orientation, it may be considered as a method of choice for mouse kidney perfusion studies.
2.3 Imaging Readout
A typical ASL MRI acquisition combines an ASL magnetization preparation module (labeling methods) followed by a subsequent rapid imaging module (readouts) to capture the blood flow–weighted contrast. These two aspects of the acquisition are independent of each other. It is important to note that the majority of the ASL imaging developments has focused on the optimization of preparation module of the ASL acquisition [20].
An important prerequisite for successful kidney imaging is the ability to get fast imaging to limit the sensitivity to motion [19]. As such, ASL has been implemented with a wide variety of fast image readout techniques, which in turn can be optimized according to a specific application. The fast image readout techniques can be broadly classified as two-dimensional (2D) or 3D acquisition schemes. Whereas 3D readouts are generally recommended for brain ASL, a consensus of the optimal readout module for renal applications has yet to be established. This consideration has been recently achieved to recommend a 2D readout in humans (unpublished by PARENCHIMA ASL expert panel).
With respect to 2D readouts, to determine the optimal readout for renal ASL, a number of factors should be considered. The optimal readout should have a short echo time (TE) in order to provide the highest image signal-to-noise ratio (SNR), and to reduce the amount of signal dephasing and distortion. At longer TEs, the short tissue intrinsic T2 and T2* in the abdomen leads to rapid signal drop and the loss of perfusion signal. The ideal readout should be collected in a short shot length to enable multiple slices through the kidney to be acquired prior to the decay of the ASL label, thus enabling whole-kidney perfusion assessment [25]. Many ASL MRI studies have utilized either echo-planar imaging (EPI) or true fast imaging with steady-state free precession (bSSFP; True FISP) readouts [20] though the readout of ASL can be measured with any method. Both EPI and bSSFP readouts are prone to off-resonance artifacts on high field MRI scanners [20] where the majority of animal studies are done by using such high field or ultra-high field (> = 7 T) scanners. Specifically, B0 inhomogeneity results in increased distortion / ghosting and banding artifacts from EPI and True FISP imaging readouts. These artifacts are particularly problematic for abdominal imaging applications such as kidney where cardiac and respiratory motions well as adipose tissue can make precise shimming difficult. In addition, the increase in T1 relaxation times which is proportional with the field strength can result in spoiled gradient echo images with a lower signal-to-noise ratio (SNR) relative to these other imaging readouts. The low SNR is specifically problematic for ASL MRI techniques as the differential blood flow signal is typically less than 10% of the mean tissue signal.
Though the optimization of EPI acquisitions allow for whole-kidney coverage but nevertheless provide sub-optimal and slice-dependent perfusion-weighted SNR and is susceptible to off-resonance conditions mentioned above. There is an urgent need to develop a rapid and robust ASL MRI imaging readout that is balanced on blood flow labeling from the ASL labeling module and also avoid B0 inhomogeneities and motion artifacts on high field preclinical MRI scanners.
While several groups independently optimize the bSSFP image readout and shown the advantage over EPI readouts [25], more advanced methods now provide alternative readouts, such as 3D gradient and spin echo (GRASE) [26, 27]. 3D GRASE has superior SNR compared to 2D acquisitions. An alternative to 3D GRASE is 3D rapid acquisition with relaxation enhancement (RARE) combined with a spiral readout trajectory (shorter echo times and superior SNR), but can introduce through-plane blurring due to long readout times.
3 Overview of Applications
3.1 ASL for Measuring Kidney Perfusion in Animal Models
The encouraging results using ASL for measuring human renal perfusion mandate further validation of renal ASL measurements using animal models. For example, in vivo interventional swine model has been used with FAIR-ASL technique to track cortical perfusion changes during pharmacologic and physiologic alterations in renal blood flow [28]. For measuring renal hemodynamics and oxygenation in X-ray contrast agent induced acute kidney injury (CIAKI), a rat model was used. Rats were injected with contrast media and followed by ASL measurements [29]. Other models, for instance ischemia induced AKI in mice, was done by transient unilateral clamping of the renal pedicle for either 35 min for moderate AKI or 45 min for severe AKI. Using ASL, renal perfusion impairment was found more pronounced in severe compared to moderate AKI, with severe AKI causing a 33% reduction in renal perfusion [9]. In addition, ASL can be useful for longitudinal assessment of renal perfusion impairment due to acute and chronic renal allograft rejection in translational mouse models [30]. By combining with the histological assessment, perfusion impairment can be validated and revealed its relationship to the severity of vascular changes and renal damage. Other applications such as exploring the renoprotective efficacy of drug treatment in ischemia-induced AKI or in renal allograft recipients revealed that the renoprotective effect of the drug was associated with improved renal perfusion [22]. Thus, noninvasive perfusion imaging might provide insights into the underlying disease mechanisms in acute and chronic renal allograft rejection and might facilitate monitoring of novel treatment strategies. Similar approach can be applied to monitor kidney function of most common kidney diseases, such as CIAKI, diabetic nephropathy, and kidney transplant. In addition, ASL techniques can also be used to monitor the therapeutic responses of treatments to common renal hypoxia disease. Figure 3 is an example of ASL measurement using FAIR for labeling and EPI for image readout in a healthy mouse. Table 2 lists all examples of preclinical studies using ASL for measuring RBF in rodent models.
References
Wentland AL, Artz NS, Fain SB, Grist TM, Djamali A, Sadowski EA (2012) MR measures of renal perfusion, oxygen bioavailability and total renal blood flow in a porcine model: noninvasive regional assessment of renal function. Nephrol Dial Transplant 27:128–135
Kennedy-Lydon TM, Crawford C, Wildman SS, Peppiatt-Wildman CM (2013) Renal pericytes: regulators of medullary blood flow. Acta Physiol 207:212–225
Cowley AW Jr, Mattson DL, Lu S, Roman RJ (1995) The renal medulla and hypertension. Hypertension 25:663–673
Schneider AG, Goodwin MD, Bellomo R (2013) Measurement of kidney perfusion in critically ill patients. Crit Care 17:220
Lemoine S, Papillard M, Belloi A, Rognant N, Fouque D, Laville M, Rouviere O, Juillard L (2011) Renal perfusion: noninvasive measurement with multidetector CT versus fluorescent microspheres in a pig model. Radiology 260:414–420
Detre JA, Leigh JS, Williams DS, Koretsky AP (1992) Perfusion imaging. Magn Reson Med 23:37–45
Williams DS, Detre JA, Leigh JS, Koretsky AP (1992) Magnetic resonance imaging of perfusion using spin inversion of arterial water. Proc Natl Acad Sci U S A 89:212–216
Wang JJ, Hendrich KS, Jackson EK, Ildstad ST, Williams DS, Ho C (1998) Perfusion quantitation in transplanted rat kidney by MRI with arterial spin labeling. Kidney Int 53:1783–1791
Hueper K, Gutberlet M, Rong S, Hartung D, Mengel M, Lu X, Haller H, Wacker F, Meier M, Gueler F (2014) Acute kidney injury: arterial spin labeling to monitor renal perfusion impairment in mice-comparison with histopathologic results and renal function. Radiology 270:117–124
Rajendran R, Huang W, Tang AM, Liang JM, Choo S, Reese T, Hentze H, van Boxtel S, Cliffe A, Rogers K, Henry B, Chuang KH (2014) Early detection of antiangiogenic treatment responses in a mouse xenograft tumor model using quantitative perfusion MRI. Cancer Med 3:47–60
Liang L, Chen WB, Chan KW, Li YG, Zhang B, Liang CH, Liu GS, Zhang SX (2016) Using intravoxel incoherent motion MR imaging to study the renal pathophysiological process of contrast-induced acute kidney injury in rats: comparison with conventional DWI and arterial spin labelling. Eur Radiol 26:1597–1605
Kwong KK, Belliveau JW, Chesler DA, Goldberg IE, Weisskoff RM, Poncelet BP, Kennedy DN, Hoppel BE, Cohen MS, Turner R et al (1992) Dynamic magnetic resonance imaging of human brain activity during primary sensory stimulation. Proc Natl Acad Sci U S A 89:5675–5679
Nery F, Gordon I, Thomas DL (2018) Non-invasive renal perfusion imaging using arterial spin labeling MRI: challenges and opportunities. Diagnostics 8:2
Edelman RR, Siewert B, Adamis M, Gaa J, Laub G, Wielopolski P (1994) Signal targeting with alternating radiofrequency (STAR) sequences: application to MR angiography. Magn Reson Med 31:233–238
Wong EC, Buxton RB, Frank LR (1997) Implementation of quantitative perfusion imaging techniques for functional brain mapping using pulsed arterial spin labeling. NMR Biomed 10:237–249
Kim SG (1995) Quantification of relative cerebral blood flow change by flow-sensitive alternating inversion recovery (FAIR) technique: application to functional mapping. Magn Reson Med 34:293–301
Rajendran R, Lew SK, Yong CX, Tan J, Wang DJ, Chuang KH (2013) Quantitative mouse renal perfusion using arterial spin labeling. NMR Biomed 26:1225–1232
Duhamel G, Prevost V, Girard OM, Callot V, Cozzone PJ (2014) High-resolution mouse kidney perfusion imaging by pseudo-continuous arterial spin labeling at 11.75T. Magn Reson Med 71:1186–1196
Prevost VH, Girard OM, Callot V, Cozzone PJ, Duhamel G (2015) Fast imaging strategies for mouse kidney perfusion measurement with pseudocontinuous arterial spin labeling (pCASL) at ultra high magnetic field (11.75 tesla). J Magn Reson Imaging 42:999–1008
Gao Y, Goodnough CL, Erokwu BO, Farr GW, Darrah R, Lu L, Dell KM, Yu X, Flask CA (2014) Arterial spin labeling-fast imaging with steady-state free precession (ASL-FISP): a rapid and quantitative perfusion technique for high-field MRI. NMR Biomed 27:996–1004
Zimmer F, Zollner FG, Hoeger S, Klotz S, Tsagogiorgas C, Kramer BK, Schad LR (2013) Quantitative renal perfusion measurements in a rat model of acute kidney injury at 3T: testing inter- and intramethodical significance of ASL and DCE-MRI. PLoS One 8:e53849
Klotz S, Pallavi P, Tsagogiorgas C, Zimmer F, Zollner FG, Binzen U, Greffrath W, Treede RD, Walter J, Harmsen MC, Kramer BK, Hafner M, Yard BA, Hoeger S (2016) N-octanoyl dopamine treatment exerts renoprotective properties in acute kidney injury but not in renal allograft recipients. Nephrol Dial Transplant 31:564–573
Tan H, Thacker J, Franklin T, Prasad PV (2015) Sensitivity of arterial spin labeling perfusion MRI to pharmacologically induced perfusion changes in rat kidneys. J Magn Reson Imaging 41:1124–1128
Roberts DA, Detre JA, Bolinger L, Insko EK, Lenkinski RE, Pentecost MJ, Leigh JS Jr (1995) Renal perfusion in humans: MR imaging with spin tagging of arterial water. Radiology 196:281–286
Buchanan CE, Cox EF, Francis ST (2018) Evaluation of 2D imaging schemes for pulsed arterial spin labeling of the human kidney cortex. Diagnostics 8:43
Robson PM, Madhuranthakam AJ, Smith MP, Sun MR, Dai W, Rofsky NM, Pedrosa I, Alsop DC (2016) Volumetric arterial spin-labeled perfusion imaging of the kidneys with a three-dimensional fast spin echo acquisition. Acad Radiol 23:144–154
Cutajar M, Thomas DL, Banks T, Clark CA, Golay X, Gordon I (2012) Repeatability of renal arterial spin labelling MRI in healthy subjects. MAGMA 25:145–153
Artz NS, Wentland AL, Sadowski EA, Djamali A, Grist TM, Seo S, Fain SB (2011) Comparing kidney perfusion using noncontrast arterial spin labeling MRI and microsphere methods in an interventional swine model. Investig Radiol 46:124–131
Chen WB, Liang L, Zhang B, Liu CL, Liu HJ, Luo HY, Zeng QX, Liang CH, Liu GS, Zhang SX (2015) To evaluate the damage of renal function in CIAKI rats at 3T: using ASL and BOLD MRI. Biomed Res Int 2015:593060
Hueper K, Schmidbauer M, Thorenz A, Brasen JH, Gutberlet M, Mengel M, Hartung D, Chen R, Meier M, Haller H, Wacker F, Rong S, Gueler F (2017) Longitudinal evaluation of perfusion changes in acute and chronic renal allograft rejection using arterial spin labeling in translational mouse models. J Magn Reson Imaging 46:1664–1672
Acknowledgments
This work was funded in part (Thoralf Niendorf, Sonia Waiczies, Andreas Pohlmann, and Joao Periquito) by the German Research Foundation (Gefoerdert durch die Deutsche Forschungsgemeinschaft (DFG), Projektnummer 394046635, SFB 1365, RENOPROTECTION. Funded by the Deutsche Forschungsgemeinschaft (DFG, German Research Foundation), Project number 394046635, SFB 1365, RENOPROTECTION).
This chapter is based upon work from COST Action PARENCHIMA, supported by European Cooperation in Science and Technology (COST). COST (www.cost.eu) is a funding agency for research and innovation networks. COST Actions help connect research initiatives across Europe and enable scientists to enrich their ideas by sharing them with their peers. This boosts their research, career, and innovation.
PARENCHIMA (renalmri.org) is a community-driven Action in the COST program of the European Union, which unites more than 200 experts in renal MRI from 30 countries with the aim to improve the reproducibility and standardization of renal MRI biomarkers.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Open Access This chapter is licensed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license and indicate if changes were made.
The images or other third party material in this chapter are included in the chapter's Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the chapter's Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder.
Copyright information
© 2021 The Author(s)
About this protocol
Cite this protocol
Ku, MC., Fernández-Seara, M.A., Kober, F., Niendorf, T. (2021). Noninvasive Renal Perfusion Measurement Using Arterial Spin Labeling (ASL) MRI: Basic Concept. In: Pohlmann, A., Niendorf, T. (eds) Preclinical MRI of the Kidney. Methods in Molecular Biology, vol 2216. Humana, New York, NY. https://doi.org/10.1007/978-1-0716-0978-1_13
Download citation
DOI: https://doi.org/10.1007/978-1-0716-0978-1_13
Published:
Publisher Name: Humana, New York, NY
Print ISBN: 978-1-0716-0977-4
Online ISBN: 978-1-0716-0978-1
eBook Packages: Springer Protocols