Abstract
Introduction
Dimethyl fumarate (DMF) showed favorable benefit-risk in patients with relapsing-remitting multiple sclerosis (MS) in phase 3 DEFINE and CONFIRM trials and in the ENDORSE extension study. Disease activity can differ in younger patients with MS compared with the overall population.
Methods
Randomized patients received DMF 240 mg twice daily or placebo (PBO; years 0–2 DEFINE/CONFIRM), then DMF (years 3–10; continuous DMF/DMF or PBO/DMF; ENDORSE); maximum follow-up (combined studies) was 13 years. This integrated post hoc analysis evaluated safety and efficacy of DMF in a subgroup of young adults aged 18–29 years.
Results
Of 1736 patients enrolled in ENDORSE, 125 were young adults, 86 treated continuously with DMF (DMF/DMF) and 39 received delayed DMF (PBO/DMF) in DEFINE/CONFIRM. Most (n = 116 [93%]) young adults completed DMF treatment in DEFINE/CONFIRM. Median (range) follow-up time in ENDORSE was 6.5 (2.0–10.0) years. Young adults entering ENDORSE who had been treated with DMF in DEFINE/CONFIRM had a model-based Annualized Relapse Rate (ARR; 95% CI) of 0.24 (0.16–0.35) vs. 0.56 (0.35–0.88) in PBO patients. ARR remained low in ENDORSE: 0.07 (0.01–0.47) at years 9–10 (DMF/DMF group). At year 10 of ENDORSE, EDSS scores were low in young adults: DMF/DMF, 1.9 (1.4); PBO/DMF, 2.4 (1.6). At ~ 7 years, the proportion of young adults with no confirmed disability progresion was 81% for DMF/DMF and 72% for PBO/DMF. Patient-reported outcomes (PROs) (SF-36 and EQ-5D) generally remained stable during ENDORSE. The most common adverse events (AEs) in young adults during ENDORSE were MS relapse (n = 53 [42%]). Most AEs were mild (n = 20 [23.3%], n = 7 [17.9%]) to moderate (n = 45 [52.3%], n = 23 [59.0%]) in the DMF/DMF and PBO/DMF groups, respectively. The most common serious AE (SAE) was MS relapse (n = 19 [15%]).
Conclusion
The data support a favorable benefit-risk profile of DMF in young adults, as evidenced by well-characterized safety, sustained efficacy, and stable PROs.
Clinical Trial Information
Clinical trials.gov, DEFINE (NCT00420212), CONFIRM (NCT00451451), and ENDORSE (NCT00835770).
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Why carry out this study? |
Patients with early onset MS are reported to relapse more frequently and have significant disability at an earlier age; therefore, disease-modifying therapy initiated early in the disease course is critical for preventing disease worsening |
This study investigates the relationship between MS disease activity and effect of DMF treatment on efficacy and safety outcomes in young adults aged between 18 and 29 years compared with the overall ENDORSE population (ages 18–55) |
What was learned from the study? |
This study supports a favorable benefit-risk profile of DMF in young adults, with incidence of AEs and SAEs consistent with those observed in the adult population and most AEs being mild to moderate in severity |
Young adults taking DMF during DEFINE/CONFIRM showed reductions in ARR and number of Gd+ lesions relative to PBO patients and consistent with the overall population |
DMF demonstrated sustained efficacy in the young adult population in the ENDORSE extension study: ARR remained stable and low over 10 years, with most patients not experiencing a relapse, and PROs remained stable |
Introduction
Multiple sclerosis (MS) is a chronic, demyelinating, inflammatory disease typically diagnosed at age 20–40 years [1, 2]. In young adults, MS is a major cause of non-traumatic disability [2], placing a substantial burden on individuals and society due to the long-term loss of productivity, physical and cognitive disability, fatigue, comorbidity, assistance with daily living, and multidisciplinary healthcare requirements [3]. Patients with early-onset MS are reported to relapse more frequently, with higher disease activity compared with their adult counterparts; although disability accumulation occurs at a slower rate, younger patients eventually have significant disability at an earlier age [4,5,6,7]. These features suggest that younger patients with MS experience a more inflammatory disease course than older patients [6, 8]. As a heterogeneous chronic disease, treatment goals for MS among younger patients therefore include prompt intervention for prevention of relapses and disability accumulation [2, 9]. A disease-modifying therapy (DMT), initiated early in the disease course [10, 11], is critical for preventing confirmed disability worsening (CDW) and maintenance of low relapse rates to meet these goals.
Dimethyl fumarate (DMF; Tecfidera, BG00012) is approved worldwide for the treatment of relapsing forms of MS [12]. As of December 31, 2022, > 585,000 patients have received DMF, representing > 1,333,000 patient-years of exposure. In two pivotal phase 3 studies, DEFINE (NCT00420212) and CONFIRM (NCT00451451), and the completed long-term extension study, ENDORSE (NCT00835770), DMF demonstrated strong efficacy on clinical and radiologic measures and a favorable benefit-risk profile in patients with relapsing-remitting MS [13,14,15]. Real-world trial data have also been consistent with the favorable efficacy and safety profile of DMF demonstrated in phase 3 trials [16, 17], in addition to demonstrating improvements on patient-reported outcomes (PROs) [18,19,20].
We report the results of an integrated post hoc analysis of the two pivotal phase 3 studies, DEFINE and CONFIRM, and their long-term extension study, ENDORSE, investigating the relationship between MS disease activity and effect of DMF treatment on efficacy and safety outcomes in young adults aged between 18 and 29 years compared with the overall ENDORSE population (ages 19–58) [21].
Methods
Patients
Patients entered ENDORSE [21] following completion of DEFINE [15] or CONFIRM [14]. Patient details for DEFINE and CONFIRM were reported previously [21]. In this report, young adult patients were defined as adults aged 18–29 years [6].
Study Design
ENDORSE [21] was an extension of DEFINE [15] and CONFIRM [14], with a minimum of 10 years (480 weeks) of planned follow-up (2 years in DEFINE/CONFIRM, plus ≥ 8 years in ENDORSE). Patients randomized in DEFINE/CONFIRM to DMF 240 mg twice daily (BID) or three times daily (TID) continued on the same dosage at the start of ENDORSE; patients randomized in the parent studies to placebo (PBO) or glatiramer acetate (GA; in CONFIRM only) were re-randomized 1:1 to DMF BID or TID. Following the 2013 market authorization of DMF, patients receiving DMF TID switched to DMF BID (approved dose) at next study visit. Details of ENDORSE were reported previously [13]. TID-treated patients were excluded from this analysis. The study was approved by local or central ethics committees and conducted in accordance with International Conference on Harmonization Guidelines for Good Clinical Practice and the Declaration of Helsinki. All patients provided written informed consent.
Safety and Hematology Assessments
Patients who received at least one dose of DMF (BID) in ENDORSE were included in the safety analysis (Fig. 1). Data on adverse events (AEs) were collected throughout the study. Laboratory assessments included blood and urine samples at baseline (every 4 weeks until week 24, then every 12 weeks thereafter) and hematologic parameters, including absolute lymphocyte count (ALC) at baseline and every ≥ 12 weeks thereafter. All ALC analyses utilized the integrated analysis of the DEFINE/CONFIRM/ENDORSE trials and were based on first exposure to DMF. Rate of ALC reconstitution in patients discontinuing DMF was assessed by linear mixed-model analysis in the integrated dataset stated above.
Efficacy Assessments
Annualized relapse rate (ARR) (relapse confirmed by an independent Neurologic Evaluation Committee)—defined as new or recurrent neurologic symptoms lasting ≥ 24 h, accompanied by new objective neurologic findings—was assessed for years 0–2 (DEFINE/CONFIRM) and 3–10 (ENDORSE). The proportion of patients relapsed at 10 years (8 years in ENDORSE) was evaluated. Confirmed disability progression (CDP) was measured every 24 weeks on the Expanded Disability Status Scale (EDSS). Time to 24-week CDW was defined as a ≥ 1.0-point increase from a baseline EDSS score ≥ 1 confirmed for 24 weeks or a ≥ 1.5-point increase from a baseline EDSS score of 0 confirmed for 24 weeks. An EDSS score of 4 has been used as a milestone marker for onset of ambulation impairment [22, 23]. In addition, confirmed disability improvement (CDI) at 24 weeks (defined as an EDSS score decrease ≥ 1.0-point in patients with a baseline score of ≥ 2.0 EDSS; confirmation 24 weeks later required) was assessed. Gadolinium-enhancing (Gd+), T2-hyperintense (T2), and T1-hypointense (T1) lesions were measured by MRI at 2 years in DEFINE/CONFIRM and at 8 years in ENDORSE. Similarly, no evidence of disease activity (NEDA-3), defined as no clinical or MRI worsening (clinical NEDA defined as no relapses or CDW; MRI NEDA defined as no Gd+ lesions or new/enlarging T2 lesions), was measured at 2 years in DEFINE/CONFIRM and at 8 years in ENDORSE. Patient-reported outcomes (PROs) were assessed using the 36-item Short Form Health Survey (SF-36) and EuroQol-5D (EQ-5D).
Patients who received DMF TID in DEFINE/CONFIRM/ENDORSE or GA in CONFIRM were excluded to focus on the approved DMF BID dosage for this analysis.
Statistical Analysis
Safety parameters were summarized using descriptive statistics, and ALC over time was assessed. ALC was characterized using Common Terminology Criteria for Adverse Events: < 0.5 × 109/L (grade 3/4, severe lymphopenia), ≥ 0.5 to < 0.8 × 109/L (grade 2, moderate lymphopenia), and ≥ 0.8 to < 0.91 (lower limit of normal [LLN]) × 109/L (grade 1, mild lymphopenia) [12]. ARR was defined as the total number of relapses divided by the number of patient-years in the study. Adjusted ARR was obtained from a negative binomial regression model adjusted for number of relapses in the year prior to study entry, baseline EDSS score, and follow-up time. The proportion of patients relapsed at 10 years was based on the Kaplan-Meier product limit method. Cox proportional-hazards model assessed time to first CDP and CDI.
Results
Patients
Of 2079 patients who completed DEFINE/CONFIRM, 1736 entered ENDORSE and received ≥ 1 dose of DMF (ENDORSE intent-to-treat and safety population; Fig. 1) [21]. Overall, 501 patients were continuously treated with DMF BID (DMF/DMF), and 249 received delayed DMF BID treatment (PBO/DMF). The young adult population comprised 125 patients; 86 were continuously treated with DMF BID (DMF/DMF) and 39 received delayed DMF treatment (PBO/DMF) (Fig. 1).
Patient baseline demographics are shown in Table 1. Most young adults completed DMF treatment in DEFINE/CONFIRM (n = 116 [93%]). The median (range) total follow-up time from randomization in DEFINE/CONFIRM and ENDORSE for young adults was 6.5 (2.0–10.0) years. Thirty-nine young adults had 7 years of follow-up in ENDORSE. Common reasons for study treatment discontinuation in the young adult population were consent withdrawn (18%), investigator decision (6%), AEs (5%), lost to follow-up (3%), MS progression (2%), MS relapse (2%), subject non-compliance (2%), and other reasons (23%).
Clinical Efficacy in the Young Adult and Overall ENDORSE Patient Population
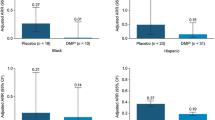
Young adults patients who entered ENDORSE following treatment with 240 mg DMF BID during DEFINE/CONFIRM had a model-based ARR (95% confidence interval [CI]) of 0.24 (0.16–0.35) compared with 0.56 (0.35–0.88) in patients treated with PBO during the same treatment period (Fig. 2a). This was in line with the overall population, where patients entering ENDORSE after treatment with DMF over years 0–2 had a model-based ARR (95% CI) of 0.19 (0.16–0.22) compared with 0.30 (0.26–0.34) in patients treated with PBO (Fig. 2b).
ARR during treatment with PBO or DMF (years 0–2 in DEFINE/CONFIRM) in a young adults and b the overall population. ARR annualized relapse rate, BID twice daily, CI confidence interval, DMF dimethyl fumarate, PBO placebo. Adjusted ARR and 95% CI are based on negative binomial regression, adjusting for Expanded Disability Status Scale scores at baseline and relapses in prior 12 months of study start. Relapse was defined as new or recurrent neurologic symptoms not associated with fever or infection, lasting ≥ 24 h accompanied by new objective neurologic findings upon examination by the treating neurologist
In young adult patients continuously treated with DMF BID (DMF/DMF) in ENDORSE, the ARR (95% CI) adjusted for baseline disease and demographic characteristics remained low, from 0.17 (0.10–0.29) in the first year to 0.07 (0.01–0.47) in years 9–10 (Fig. 3a). ARR (95% CI) remained consistent and low in the overall population, from 0.20 (0.16–0.25) in the first year to 0.11 (0.07–0.17) in years 9–10 (Fig. 3b). The estimated proportion of young adult patients with relapses at 2 years (96 weeks) was 30.2% for DMF/DMF and 59.0% for placebo (PBO/DMF) treatment groups (see Fig. S1 in the electronic supplementary material).
Adjusted ARR (objective relapses) by yearly interval for the DMF/DMF group in a the young adult patient DMF/DMF population and b the overall ENDORSE population over 10 years (ENDORSE 8 years). ARR annualized relapse rate, CI confidence interval, DMF dimethyl fumarate. Adjusted ARR and 95% CI are based on negative binomial regression, adjusting for Expanded Disability Status Scale scores at baseline and relapses in prior 12-month of study start. Relapse was defined as new or recurrent neurologic symptoms not associated with fever or infection, lasting ≥ 24 h accompanied by new objective neurologic findings upon examination by the treating neurologist
After ~ 8 years of DMF treatment in ENDORSE (year 10), mean (standard deviation [SD]) EDSS scores were low in both the young adult and overall populations: DMF/DMF, 1.9 (1.4); PBO/DMF, 2.4 (1.6) in the young adult subpopulation and DMF/DMF, 2.0 (1.4); PBO/DMF, 1.5 (0.0) in the overall population. Rates of CDP were low over the course of the study; in the young adult subpopulation, the proportion of patients with no 24-week CDP (combined DEFINE/CONFIRM and ENDORSE data) at 96 weeks was 96% and 78% for DMF/DMF and PBO/DMF, respectively. At 336 weeks (~ 7 years), the proportion of young adult patients with no CDP was 81% for DMF/DMF and 72% for PBO/DMF (Fig. 4). In the overall population, over 10 years, the proportion of patients with no CDP was 72% of DMF/DMF and 73% of PBO/DMF patients. In young adult patients, EDSS scores were ≤ 3.5 at year 2 and year 10, respectively, for 92.4% (85/92) and 92.9% (26/28) of DMF/DMF patients and 88.4% (38/43) and 90.9% (10/11) of PBO/DMF patients. In the overall population, EDSS scores were ≤ 3.5 at year 2 and year 10, respectively, for 86% (413/479) and 77% (173/226) of DMF/DMF patients and 82% (179/217) and 74% (67/90) of PBO/DMF patients. The proportion of patients with CDI at 96 weeks was 13% for DMF/DMF and 8% for PBO/DMF and 20% for DMF/DMF and 8% for PBO/DMF at 336 weeks (Fig. S2 in the supplementary material).
Kaplan-Meier estimated proportion of patients with 24-week sustained confirmed disability progressiona at 336 weeks (7 years). BL baseline, CI confidence interval, DMF dimethyl fumarate, PBO placebo. aSustained disability progression defined as ≥ 1.0-point increase on the Expanded Disability Status Scale (EDSS) from a baseline EDSS ≥ 1.0 sustained for 24 weeks or a ≥ 1.5-point increase on the EDSS from a baseline EDSS of 0 sustained for 24 weeks. Image cutoff at 336 weeks due to numbers at risk falling below 10 patients in both groups after this time point
Radiologic Efficacy in the Young Adult Patient Population
In DEFINE/CONFIRM, mean (SD) number of Gd+ lesions decreased from baseline to week 96 in DMF-treated young adult patients (3.8 [8.0] vs. 0.6 [1.6]), but remained unchanged in PBO-treated patients (1.5 [1.7] vs. 1.4 [1.6]). This is in line with what was observed in the overall population: mean (SD) number of Gd+ lesions decreased from baseline to week 96 in the DMF-treated group (1.9 [5.0] vs. 0.3 [1.3]) and remained unchanged in the PBO-treated patients (2.2 [5.9] vs. 1.8 [4.9]).
The estimated proportion of patients with NEDA-3 in DEFINE/CONFIRM who entered the ENDORSE study was 54.1% in the young adult DMF-treated patients and 26.3% in the PBO-treated patients at 48 weeks and 43.2% versus 15.8% at 96 weeks following randomization in DEFINE/CONFIRM, respectively. Similarly, the estimated proportion of patients in the overall population with NEDA-3 at 48 weeks was 65.9% in DMF-treated patients and 46.4% in PBO-treated patients and 50.4% and 36.6% at 96 weeks in the DMF and PBO groups, respectively.
Safety and Tolerability
Among young adult patients, AEs experienced by ≥ 2% patients in any treatment group and serious AEs (SAEs) experienced by ≥ 5 patients in any treatment group are summarized in Table 2. The most common AEs in the young adult population during ENDORSE were MS relapse (n = 53 [42%]), flushing (n = 31 [25%]), and nasopharyngitis (n = 30 [24%]). Most AEs were mild (n = 20 [23.3%], n = 7 [17.9%]) to moderate (n = 45 [52.3%], n = 23 [59.0%]) in the DMF/DMF and PBO/DMF groups, respectively. The most common SAE was MS relapse (n = 19 [15%]). Overall incidence and type of AEs and SAEs among young adults were consistent with those reported in DEFINE and CONFIRM (Table S2 in the supplementary material) and were generally similar across treatment groups.
In ENDORSE, gastrointestinal disorders were reported for 37% of young adult patients and were higher for PBO/DMF (41%) compared with DMF/DMF (35%) (Table 3). These differences were driven by higher rates of incidence in the first year of ENDORSE for the patients switching to DMF from placebo in the PBO/DMF group and are consistent with the timing of gastrointestinal AEs reported in DMF-treated patients in DEFINE and CONFIRM [14, 15]. In DEFINE/CONFIRM, gastrointestinal disorders were reported for 62% DMF/DMF young adult patients compared with 56% PBO/DMF patients. Similar differences were seen for flushing AEs between the DMF/DMF and PBO/DMF groups in ENDORSE (21% and 33%, respectively). Consistent with DEFINE/CONFIRM, microalbuminuria, proteinuria, and hematuria were the most common renal AEs reported (Table 3 and Table S2 in the supplementary material).
Absolute Lymphocyte Count
Post-baseline lymphocyte measurements were available in 81 and 38 young adult patients in the DMF/DMF and PBO/DMF groups, respectively. In the DMF/DMF group, ALC decreased over the first 48 weeks during DEFINE/CONFIRM (mean percent change from baseline ALC, − 21.2% at week 48) and remained generally stable for the duration of the study (Fig. 5a). In the ENDORSE follow-up study, the PBO/DMF mean percent change in ALC in young adult patients at 48 weeks after DMF initiation was − 19.1% (week 144 for combined follow-up). An analysis of ALCs in DEFINE/CONFIRM of the young adult patients on DMF showed that 90% of patients had ALC values > LLN, whereas 4 (4.4%) had mild lymphopenia (ALC between 0.80 and 0.91 × 109/L); 3 (3.9%) and 1 (1.3%) of young adult patients developed moderate or severe lymphopenia, respectively (Fig. 5b).
a Lymphocyte mean values over time for young adults continually treated with DMF BID (DMF/DMF) and in young adults whose DMF treatment was delayed (PBO/DMF); b severity of lymphopenia over time in young adults treated with DMF. ALC absolute lymphocyte count, BID twice daily, CI confidence interval, DMF dimethyl fumarate, PBO placebo. *t-test p < 0.05. aNo lymphopenia ≥ LLN (ALC ≥ 0.91 × 109/L). bMild lymphopenia grade 1, ALC = ≥ 0.8 to < 0.91 × 109/L. cModerate lymphopenia grade 2, ALC ≥ 0.5 to < 0.8 × 109/L. dSevere lymphopenia grade 3/4, ALC < 0.5 × 109/L.
In the overall population of ENDORSE, post-baseline lymphocyte measurements were available in 2222 of 2263 patients. Over the first 48 weeks, ALC decreased: at week 48, mean percentage change from baseline was − 27.7%. ALC remained generally stable for the duration of the study and remained ~ LLN (0.91 × 109/L) for the majority (59%) of patients. Of patients treated with DMF, 235 (10.6%) developed prolonged moderate lymphopenia and 53 (2.4%) developed prolonged severe lymphopenia, [13].
Patient-reported Outcomes
Among young adults, PROs (SF-36 and EQ-5D) generally remained stable during ENDORSE (Fig. S3 in the supplementary material), which was consistent with the overall ENDORSE population [13].
Discussion
This post hoc integrated analysis of the phase 3 studies DEFINE and CONFIRM and long-term extension study ENDORSE investigated the relationship between MS disease activity and effect of DMF treatment in young adults ages 18–29 years (mean [SD] age 24 [2.8] years) compared with the overall ENDORSE population (37.9 [9.1] years at randomization). The data support a favorable benefit-risk profile of DMF in young adults, as evidenced by well-characterized safety, sustained efficacy, and stable PROs.
The incidences of AEs and SAEs in young adults were consistent with those observed in the overall adult population in the DEFINE/CONFIRM and real-world datasets. MS relapse, flushing, and nasopharyngitis were most frequently reported, and most AEs were mild to moderate in severity. ALC decreased over the first 48 weeks in young adults treated with DMF, consistent with data from the full adult population of DMF-treated patients, and remained generally stable thereafter. DMF decreases absolute lymphocyte counts but does not affect all subsets uniformly. It particularly decreases the pro-inflammatory T-helper Th1 and Th17 subsets, creating a bias toward more anti-inflammatory Th2 and regulatory subsets [24, 25].
In patients continually treated with DMF BID in DEFINE/CONFIRM and ENDORSE, ARR remained stable and low over 10 years, and most patients in both the overall and young adult patient populations did not experience a relapse. Similarly, in both the overall and young adult populations in DEFINE/CONFIRM and ENDORSE, patients who had a delayed start to DMF treatment (PBO/DMF) experienced a significant decrease in ARR after starting DMF and maintained a low ARR thereafter. Similar to the overall ENDORSE population, disability scores among young adults remained stable across the study period, with most (93%) DMF BID-treated patients maintaining walking ability over 7 years. Our CDI findings in young adults, together with the observation of a shorter time since first symptoms/diagnosis compared with the overall population, are consistent with literature supporting the benefit of starting DMT early in younger patients with MS [26]. PROs assessed as part of this integrated analysis were stable with continuous DMF treatment.
There is evidence that disease activity (ARR and number of MRI lesions) is higher in pediatric onset MS [6, 27, 28] and, as it has been shown to decline with age [28], it follows that it would be higher in younger adults with MS than in older adults. Our baseline data are reflective of this finding in that younger adults had more relapses than the overall population in the 12 months prior to study entry, as well as a trend toward a higher number of Gd+ lesions. However, despite a seemingly greater baseline disease activity, the treatment effect of DMF in younger adults in terms of reductions in relapse rates, MRI lesion activity, and disability progression was comparable to that in the overall study population.
As expected in a long-term extension study, discontinuation rates were higher than in the DEFINE/CONFIRM 2-year studies. Reasons for discontinuations from long-term studies are varied and could be a result of patient’s pregnancy, moving, sites closing, countries dropping out of the study once the drug gains regulatory approval, or pregnancy.
Our findings are supported by other studies in the literature evaluating the effects of DMTs in young adults with MS. A post hoc analysis of phase 3 studies in the fingolimod program reported that ARRs were higher in younger patients (all p < 0.05) and significantly reduced with fingolimod versus placebo or interferon beta-1a (IFNβ-1a), with the youngest patients showing the highest reductions in ARR [29]. Furthermore, a retrospective analysis of 2477 patients with relapsing-remitting MS showed that relapse rates were age dependent and that patients aged < 40 years tended to benefit the most from DMTs [30]. In a meta-analysis of randomized, blinded MS clinical trials involving > 28,000 patients with MS [31], the efficacy of DMTs on MS disability was reported to depend greatly on age, with younger patients showing the most benefit. In addition, the results suggested that delaying DMT at an early stage of MS could lead to a decrease in the cumulative efficacy of DMT treatment when patients are older [31]. In pediatric patients with MS aged 10–17 years, DMF has been shown to be more effective than IFNβ-1a in that more patients were free of new or newly enlarging T2 lesions following 96 weeks of treatment with DMF compared with IFNβ-1a (16% vs. 5%, respectively) [32]. These studies provide further evidence of the beneficial effects of early and consistent treatment with DMF.
Limitations of these data include a relatively small sample size of the young adult population, especially among the PBO/DMF cohort (n = 39) in ENDORSE; however, we included pooled data with total number of young adults of 125 patients. As this was an exploratory post hoc analysis, these findings should be interpreted with caution. Like most MS studies, enrolling diverse patients and including more ethnicities would be desirable for future design.
Conclusion
Overall, these findings suggest a favorable benefit-risk profile for DMF in the treatment of relapsing-remitting MS in young adults. In particular, the reductions in relapse rates and number of Gd+ lesions following DMF treatment were comparable to those seen in the overall study population, despite evidence of higher disease activity in young adults versus the overall population at baseline.
References
Conradsson D, Ytterberg C, von Koch L, Johansson S. Changes in disability in people with multiple sclerosis: a 10-year prospective study. J Neurol. 2018;265(1):119–26. https://doi.org/10.1007/s00415-017-8676-8.
Filippi M, Bar-Or A, Piehl F, et al. Multiple sclerosis. Nat Rev Dis Prim. 2018;4(1):43. https://doi.org/10.1038/s41572-018-0041-4.
Kobelt G, Thompson A, Berg J, et al. New insights into the burden and costs of multiple sclerosis in Europe. Mult Scler. 2017;23(8):1123–36. https://doi.org/10.1177/1352458517694432.
Scalfari A, Lederer C, Daumer M, et al. The relationship of age with the clinical phenotype in multiple sclerosis. Mult Scler. 2016;22(13):1750–8. https://doi.org/10.1177/1352458516630396.
Tremlett H, Devonshire V. Is late-onset multiple sclerosis associated with a worse outcome? Neurology. 2006;67(6):954–9. https://doi.org/10.1212/01.wnl.0000237475.01655.9d.
Ziemssen T, Albrecht H, Haas J, et al. Descriptive analysis of real-world data on fingolimod long-term treatment of young adult RRMS patients. Front Neurol. 2021;12:637107. https://doi.org/10.3389/fneur.2021.637107.
Simone IL, Carrara D, Tortorella C, et al. Course and prognosis in early-onset MS: comparison with adult-onset forms. Neurology. 2002;59(12):1922–8. https://doi.org/10.1212/01.wnl.0000036907.37650.8e.
McKay KA, Hillert J, Manouchehrinia A. Long-term disability progression of pediatric-onset multiple sclerosis. Neurology. 2019;92(24):e2764–73. https://doi.org/10.1212/WNL.0000000000007647.
Montalban X, Gold R, Thompson AJ, et al. ECTRIMS/EAN guideline on the pharmacological treatment of people with multiple sclerosis. Mult Scler. 2018;24(2):96–120. https://doi.org/10.1177/1352458517751049.
Chalmer TA, Baggesen LM, Norgaard M, et al. Early versus later treatment start in multiple sclerosis: a register-based cohort study. Eur J Neurol. 2018;25(10):1262-e110. https://doi.org/10.1111/ene.13692.
Mills EA, Begay JA, Fisher C, Mao-Draayer Y. Impact of trial design and patient heterogeneity on the identification of clinically effective therapies for progressive MS. Mult Scler. 2018;24(14):1795–807. https://doi.org/10.1177/1352458518800800.
Biogen. Tecfidera prescribing information. 2022. https://www.tecfidera.com/content/dam/commercial/multiple-sclerosis/tecfidera/pat/en_us/pdf/full-prescribing-info.pdf. Accessed 6 July 2022.
Gold R, Arnold DL, Bar-Or A, et al. Long-term safety and efficacy of dimethyl fumarate for up to 13 years in patients with relapsing-remitting multiple sclerosis: final ENDORSE study results. Mult Scler. 2022;28(5):801–16. https://doi.org/10.1177/13524585211037909.
Fox RJ, Miller DH, Phillips JT, et al. Placebo-controlled phase 3 study of oral BG-12 or glatiramer in multiple sclerosis. N Engl J Med. 2012;367(12):1087–97. https://doi.org/10.1056/NEJMoa1206328.
Gold R, Kappos L, Arnold DL, et al. Placebo-controlled phase 3 study of oral BG-12 for relapsing multiple sclerosis. N Engl J Med. 2012;367(12):1098–107. https://doi.org/10.1056/NEJMoa1114287.
Cohan SL, Moses H, Calkwood J, et al. Clinical outcomes in patients with relapsing-remitting multiple sclerosis who switch from natalizumab to delayed-release dimethyl fumarate: a multicenter retrospective observational study (STRATEGY). Mult Scler Relat Disord. 2018;22:27–34. https://doi.org/10.1016/j.msard.2018.02.028.
Salter A, Lancia S, Cutter G, et al. Characterizing long-term disability progression and employment in NARCOMS registry participants with multiple sclerosis taking dimethyl fumarate. Int J MS Care. 2021;23(6):239–44. https://doi.org/10.7224/1537-2073.2020-109.
Berger T, Brochet B, Brambilla L, et al. Effectiveness of delayed-release dimethyl fumarate on patient-reported outcomes and clinical measures in patients with relapsing-remitting multiple sclerosis in a real-world clinical setting: PROTEC. Mult Scler J Exp Transl Clin. 2019;5(4):2055217319887191. https://doi.org/10.1177/2055217319887191.
Kresa-Reahl K, Repovic P, Robertson D, et al. Effectiveness of delayed-release dimethyl fumarate on clinical and patient-reported outcomes in patients with relapsing multiple sclerosis switching from glatiramer acetate: RESPOND, a prospective observational study. Clin Ther. 2018;40(12):2077–87. https://doi.org/10.1016/j.clinthera.2018.10.011.
Repovic P, Robertson D, Kresa-Reahl K, et al. Effectiveness of dimethyl fumarate in patients with relapsing multiple sclerosis switching after suboptimal response to glatiramer acetate, including patients with early multiple sclerosis: subgroup analysis of RESPOND. Neurol Ther. 2021;10(1):169–82. https://doi.org/10.1007/s40120-020-00223-2.
Gold R, Arnold DL, Bar-Or A, et al. Long-term effects of delayed-release dimethyl fumarate in multiple sclerosis: Interim analysis of ENDORSE, a randomized extension study. Mult Scler. 2017;23(2):253–65. https://doi.org/10.1177/1352458516649037.
Ford CC, Johnson KP, Lisak RP, et al. A prospective open-label study of glatiramer acetate: over a decade of continuous use in multiple sclerosis patients. Mult Scler. 2006;12(3):309–20. https://doi.org/10.1191/135248506ms1318oa.
Kurtzke JF. Rating neurologic impairment in multiple sclerosis: an expanded disability status scale (EDSS). Neurology. 1983;33(11):1444–52. https://doi.org/10.1212/wnl.33.11.1444.
Longbrake EE, Mao-Draayer Y, Cascione M, et al. Dimethyl fumarate treatment shifts the immune environment toward an anti-inflammatory cell profile while maintaining protective humoral immunity. Mult Scler. 2021;27(6):883–94. https://doi.org/10.1177/1352458520937282.
Wu Q, Wang Q, Mao G, et al. Dimethyl fumarate selectively reduces memory T cells and shifts the balance between Th1/Th17 and Th2 in multiple sclerosis patients. J Immunol. 2017;198(8):3069–80. https://doi.org/10.4049/jimmunol.1601532.
Kopp TI, Blinkenberg M, Petersen T, Sorensen PS, Magyari M. Long term effect of delayed treatment on disability in patients with paediatric onset multiple sclerosis: a prospective Danish cohort study. Mult Scler Relat Disord. 2020;40:101956. doi: https://doi.org/10.1016/j.msard.2020.101956.
Waubant E, Chabas D, Okuda DT, et al. Difference in disease burden and activity in pediatric patients on brain magnetic resonance imaging at time of multiple sclerosis onset vs adults. Arch Neurol. 2009;66(8):967–71. https://doi.org/10.1001/archneurol.2009.135.
Dahlke F, Arnold DL, Aarden P, et al. Characterisation of MS phenotypes across the age span using a novel data set integrating 34 clinical trials (NO.MS cohort): age is a key contributor to presentation. Mult Scler. 2021;27(13):2062–76. https://doi.org/10.1177/1352458520988637.
Gartner J, Chitnis T, Ghezzi A, et al. Relapse rate and MRI activity in young adult patients with multiple sclerosis: a post hoc analysis of phase 3 fingolimod trials. Mult Scler J Exp Transl Clin. 2018;4(2):2055217318778610. https://doi.org/10.1177/2055217318778610.
Tremlett H, Zhao Y, Joseph J, Devonshire V, Neurologists UC. Relapses in multiple sclerosis are age- and time-dependent. J Neurol Neurosurg Psychiatry. 2008;79(12):1368–74. https://doi.org/10.1136/jnnp.2008.145805.
Weideman AM, Tapia-Maltos MA, Johnson K, Greenwood M, Bielekova B. Meta-analysis of the age-dependent efficacy of multiple sclerosis treatments. Front Neurol. 2017;8:577. https://doi.org/10.3389/fneur.2017.00577.
Vermersch P SM, Levin S, Alroughani R, Deiva K, Pozzilli C, Lyons J, Mokliatchouk O, Pultz J, N'Dure F, Liu S, Badwan R, Branco F, Hood-Humphrey V, Franchimont N, Hanna J, Maghzi A-H. Effect of dimethyl fumarate versus interferon β-1a in patients with pediatric-onset multiple sclerosis: the CONNECT randomized clinical trial. JAMA Network Open. 2022 (in press).
Acknowledgements
We thank the participants of this study.
Funding
Biogen, Cambridge, MA, USA, provided funding for the study and the publication fee.
Medical Writing and/or Editorial Assistance
Biogen provided funding for medical writing support in the development of this paper. Karen Spach, PhD, from Excel Scientific Solutions, wrote the first draft of the manuscript based on input from authors. Alison Terry, Excel Scientific Solutions, copy edited and styled the manuscript per journal requirements.
Authorship
All named authors meet ICMJE criteria for authorship for this article, take responsibility for the integrity of the work as a whole, and have given their approval for this version to be published.
Authors’ Contributions
Lilyana Amezcua: Concept and design of study, data collection, data interpretation, drafting and critically revising the manuscript. Yang Mao-Draayer: Concept and design of study, data collection, data interpretation, drafting and critically revising the manuscript. Wendy S. Vargas: Concept and design of study, data collection, data interpretation, drafting and critically revising the manuscript. Rebecca Farber: Concept and design of study, data collection, data interpretation, drafting and critically revising the manuscript. Sara Schaefer: Concept and design of study, data collection, data interpretation, drafting and critically revising the manuscript. Filipe Branco: Concept and design of study, data interpretation, drafting and critically revising the manuscript. Sarah M. England: Concept and design of study, data interpretation, drafting and critically revising the manuscript. Nicholas Belviso: Concept and design of study, data collection, statistical analyses, data interpretation, drafting and critically revising the manuscript. James B. Lewin: Concept and design of study, data interpretation, drafting and critically revising the manuscript. Jason P. Mendoza: Concept and design of study, data interpretation, drafting and critically revising the manuscript. Sai L. Shankar: Concept and design of study, data collection, data interpretation, drafting and critically revising the manuscript.
Investigators
For principal investigator list, please see supplementary material, pages 10–13.
Disclosures
Lilyana Amezcua: advisory/consulting fees from EMD Serono, Genzyme, and Novartis; research support from Biogen and MedDay. Yang Mao-Draayer: consulting fees from Biogen, Celgene, EMD Serono, Genzyme, Novartis, Janssen, Horizon, and Roche-Genentech; contracted research for NIH NINDS R01-NS080821, NIAID Autoimmune Center of Excellence UM1-AI110557, Biogen, Chugai, Novartis, and Sanofi-Genzyme; speaker bureaus for Biogen and Teva. Wendy S. Vargas: advisory/consulting fees from Alexion, Biogen, Genentech, and Octapharma; research support from Teva. Rebecca Farber: Consulting fees for Alexion and Genentech; research grants from Novartis and Biogen; research support from NIH, grant number 2U19AI128949-06. Sara Schaefer: Consulting/advisory fees from Novartis, Biogen, and Genzyme; speaking fees from Biogen. Filipe Branco, Sarah M. England, Nicholas Belviso, James B. Lewin, Jason P. Mendoza, and Sai L. Shankar are employees and stockholders of Biogen.
Compliance with Ethics Guidelines
The study was approved by local or central ethics committees and conducted in accordance with International Conference on Harmonisation Guidelines for Good Clinical Practice and the Declaration of Helsinki. All patients provided written informed consent. For a full list of the central ethics committees for ENDORSE, please see Table S3 in the supplementary material.
Data Availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Author information
Authors and Affiliations
Consortia
Corresponding author
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Amezcua, L., Mao-Draayer, Y., Vargas, W.S. et al. Efficacy of Dimethyl Fumarate in Young Adults with Relapsing-Remitting Multiple Sclerosis: Analysis of the DEFINE, CONFIRM, and ENDORSE Studies. Neurol Ther 12, 883–897 (2023). https://doi.org/10.1007/s40120-023-00475-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40120-023-00475-8