Abstract
Background/objective
The genetic architecture of extreme non-syndromic obesity in adults remains to be elucidated. A range of genes are known to cause monogenic obesity, but even when pathogenic mutations are present, there may be variable penetrance.
Methods
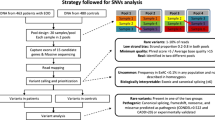
Whole-exome sequencing (WES) was carried out on a 15-year-old male proband of Pakistani ancestry who had severe obesity. This was followed by family segregation analysis, using Sanger sequencing. We also undertook re-analysis of WES data from 91 unrelated adults with severe obesity (86% white European ancestry) from the Personalised Medicine for Morbid Obesity (PMMO) cohort, recruited from the UK National Health Service.
Results
We identified an oligogenic mode of inheritance of obesity in the proband’s family—this provided the impetus to reanalyze existing sequence data in a separate dataset. Analysis of PMMO participant data revealed two further patients who carried more than one rare, predicted-deleterious mutation in a known monogenic obesity gene. In all three cases, the genes involved had known autosomal dominant inheritance, with incomplete penetrance.
Conclusion
Oligogenic inheritance may explain some of the variable penetrance in Mendelian forms of obesity. We caution clinicians and researchers to avoid confining sequence analysis to individual genes and, in particular, not to stop looking when the first potentially-causative mutation is found.
Similar content being viewed by others
Introduction
Obesity is a genetically heterogeneous disorder that is known to occur in multifactorial, monogenic, or syndromic forms. The increasing availability of whole-exome sequencing (sequencing of the genomic regions encoding proteins and closely linked regulatory regions), has improved our understanding of those sub-forms of obesity that are inherited in a simple Mendelian fashion. However, we are still some way from appreciating the full complexity of its genetic architecture, including explanations for the variable penetrance of known pathogenic mutations (whether or not a carrier of a harmful mutation expresses the expected trait) [1, 2]. It is possible that, at least in some cases, more than one genetic deficit may be required for production of a severe phenotype. The concept of inheritance patterns more complex than simple Mendelian dominance or recessivity has been suggested previously in obesity syndromes and other conditions, but the implications of this in people ascertained only for severe obesity remains unclear [3,4,5,6,7,8].
Here, we present results suggesting oligogenic inheritance of obesity (whereby a small number of mutations act together to cause the phenotype), first in a family trio where the proband has obesity and learning difficulties, and subsequently in unrelated adults with severe obesity, but without known learning disability or dysmorphism.
Results
Family segregation analysis: proband 1
Proband 1, a 22-year-old male of South Asian origin with obesity, attention deficit hyperactivity disorder (ADHD), depression, and sleep apnoea, underwent whole-exome sequencing (WES) for diagnostic purposes. A heterozygous deleterious mutation in the sarcoma (Src) homology 2 B adaptor protein 1 (SH2B1) gene c.539 C > T:p.(Ser180Phe), with a CADD score of 23.6 (Table 1 and Fig. 1A) was detected in initial analysis. The identified variant is located close to the site of a variant (A175N) previously reported in individuals with severe early-onset obesity which was shown to disrupt the function of NGF-induced neuronal differentiation. Subsequent characterisation of the proband’s parents by Sanger sequencing revealed that the father without obesity was carrying the same mutation (Fig. 1B), raising questions about penetrance or pathogenicity of this variant. Re-evaluation of the original exome sequence data revealed two further deleterious variants, that had previously been overlooked on the assumption that the SH2B1 mutation was causative for the phenotype: c.3539 A > T:p.(Asp1180Val) in MBD5, and c.3280 C > T:p.(Leu1094Phe) in POGZ, with CADD scores of 22.9 and 24.2 respectively (Fig. 1B, C). All mutations were rare with ≤0.1% minor allele frequency (MAF) in gnomAD, as shown in Table 1. Considered individually, none of the three variants showed co-segregation with the phenotype, but the proband had inherited all three.
A Chromatograms of Sanger sequencing demonstrating heterozygosity for SH2B1:c.539 C > T in the proband and his healthy parents. B Chromatogram of Sanger sequencing demonstrating heterozygosity for POGZ:c.3280 C > T in the proband and his healthy father. C Chromatogram of Sanger sequencing demonstrating heterozygosity for MBD5:c.3539 A > T in the proband and his healthy mother. D Pedigree of the family and the putatively deleterious variants in each individual.
Oligogenic obesity in severe adult obesity without learning disability or dysmorphism
After identifying the initial case with potentially oligogenic mode of inheritance, re-analysis was performed of WES data from 91 unrelated adults (86% white European ancestry) with severe obesity from the PMMO cohort, recruited from UK bariatric services [9]. In this analysis, two further cases of apparent oligogenic inheritance of obesity were identified. The identified oligogenic events affected pairs of genes SH2B1/RAI1 and SETD2/POGZ, as summarised in Table 2. Each variant was a missense variant in heterozygous state (which matched the known autosomal dominant mode of inheritance) and had a CADD score greater than 20. Although these variants affect evolutionarily-conserved residues; this is the first report that they are potentially causative of obesity. Notably, for each of these pairs, one of the affected genes was among those mutated in proband 1.
Discussion
To our knowledge, this is the first report of apparent oligogenic inheritance of severe (non-syndromic) obesity, and is potentially of clinical importance in explaining the observed variable penetrance of known pathogenic mutations.
The initial finding of apparent oligogenic inheritance was in a proband (proband 1) with obesity and learning difficulties. Exome sequencing revealed heterozygous predicted-deleterious variants in three separate obesity-relevant genes: SH2B1, POGZ and MBD5. However, each gene is reportedly autosomal dominant, and one of these variants was present in the healthy mother and two in the healthy father. This suggests that an oligogenic inheritance model might be responsible for the phenotype of the proband.
The first identified variant in proband 1 was in a known obesity gene, SH2B1. The variant is located close to a variant (p.A175N) previously reported in individuals with severe early-onset obesity, which was shown to disrupt the function of NGF-induced neuronal differentiation [10]. Mutations in the POGZ gene cause White-Sutton syndrome, which has a highly variable phenotype including obesity, developmental delay, language and speech delay, motor delay, microcephaly, and non-specific vision problems [11,12,13]. POGZ encodes a zinc finger protein believed to have an important role in mitotic progression and possibly in neuronal proliferation [14, 15]. The third predicted-deleterious variant of interest in this proband is in MBD5, which encodes a member of the MBD family (which also includes the MECP2 gene, a causal gene for Rett syndrome) [16]. The encoded protein is highly expressed in brain, oocytes and testis and is thought to have a role in heterochromatin and epigenetic reprogramming [16]. The MBD5 gene has been identified as a potential causative locus contributing to 2q23.1 microdeletion syndrome: The phenotypic effects of genomic disruption of MBD5 (ranging from point mutation to deletion or duplication) have considerable overlap with the clinical phenotype of 2q23.1 deletion syndrome, and have been termed MBD5-associated neurodevelopmental disorder (MAND) [17, 18]. Haploinsufficiency of MBD5 causes diverse phenotypes which include hyperphagia, behavioural problems, craniofacial abnormalities, language impairment, microcephaly, development and motor delay, short stature, sleep disturbance, epilepsy, hyperphagia, obesity, and seizures [17,18,19,20,21,22,23,24,25,26]. The severity and complexity of the phenotypic spectrum of MBD5 gene disruption varies from that of the high-penetrance 2q23.1 microdeletion [17,18,19], and incomplete penetrance for MBD5 mutations has been identified in several previous report [17, 19]. Missense variants in MBD5 (which perhaps have reduced penetrance) are associated with risk of autism spectrum disorders and may also contribute to schizophrenia and depression [26]. Obesity and hyperphagia are considered one of the most frequent clinical features (>50%) of 2q23.1 deletion syndrome [18, 21]. In addition, in a recent study of individuals with early-onset obesity, three copy number variants affecting this gene were identified [27].
Prompted by these findings, two further instances of possible oligogenic obesity were identified by re-analysis of WES data from 91 unrelated adults with severe obesity from the PMMO cohort (with severe obesity, but without identified learning disability). We note that in these two subsequent cases, variants affecting the SH2B1 and POGZ genes are again involved. Further analysis in larger clinical cohorts will be required to determine whether this is a stochastic effect, or reflects some aspect of the functional implication of mutations in these genes: i.e. that a “second hit” (either genetic or environmental) may be required for penetrance.
Consistent with this, there is evidence that SH2B1 mutations have incomplete or variable penetrance, supporting a possible requirement for other genetic and/or environmental factors to manifest the disease [28]. Individuals with variants in SH2B1 exhibit a wide range of phenotypes including obesity, insulin resistance, and neurodevelopmental problems, while variants in other related obesity genes result in a diverse phenotypic spectrum which includes obesity.
We suggest that defects in more than one gene may be acting in concert to produce the observed phenotype in the individuals presented here. Recently, several diseases have been reported to show oligogenic inheritance, such as cardiovascular disease, autism, and Bardet-Biedl syndrome [3, 5,6,7].
There remain a number of obstacles to the use of mutational analysis in clinical practice for obesity, the most pressing being assessment of the clinical implications of variants of unknown significance (VUS). Here, several of the variants presented have not been previously reported and/or investigated for their functional effects, so would formally be reported as VUS. A further limitation of these analyses is that, since the PMMO participants are unrelated adults, DNA was not available for family segregation analysis. In this report, we used CADD scores, coupled with low MAF values in a large database of exome sequence data (gnomAD v2.1.1), to filter variants for likely pathogenicity. While the two cases identified here were of European ancestry, for whom ancestry-matched gnomAD allele frequencies were available, this approach currently has potential limitations for patients of non-European ancestry where less data on “normal” genetic variation is available. The genetics community awaits the development of methodologies for high throughput functional analyses of VUS to allow full interpretation of results, as well as large-scale analysis of sequenced and carefully phenotyped individuals of different ancestries [8]. Given the very low population-specific MAFs of the variants detected, however, it seems unlikely that these combinations are simply the result of chance. Additionally, further work will be required to ascertain whether some genes are more frequently involved in oligogenic obesity than other genes or whether this is a chance finding.
In summary, we present results suggesting that some cases of extreme obesity are due to a combination of mutations in more than one gene. This phenomenon could easily be missed in analysing data from candidate genes where it might be tempting to rely on a single explanatory mutation of known effect, without considering other (possibly heterozygous) VUS. As a result of our experience, we urge caution in interpretation of sequencing results from individual candidate genes which, in the instance of the family of proband 1, could have given rise to inaccurate genetic counselling. Utilisation of new tools that are being developed to explore the possibility of oligogenic inheritance may uncover additional examples in other conditions [29, 30]. WES or whole genome sequencing (WGS) allows exploration of a wider range of genes, and allows re-analysis as new causative genes are discovered, and where resources allow, should be the approach of choice.
Materials and Methods
Study participants and clinical information
Proband 1 and parents
A 22-year-old male proband of Pakistani heritage and his parents were seen at the Imperial College London Diabetes Centre in Abu Dhabi (Research Ethics Committee (IREC029)), for diagnostic purposes. Blood samples were collected from the patient and his parents. The proband has history of progressive weight gain starting in his mid-teens. He started to have problems with school performance at around 15 years of age. There was no family history of note and the parents were not consanguinous. On examination at age 10, he was noted to have generalised obesity (BMI 38.6 kg/m2) and prominent acanthosis nigricans. He had normal blood pressure and no Cushingoid features. Biochemical and hormonal investigations were normal, apart from profound hyperinsulinaemia. The patient was commenced on metformin with no response in terms of his weight. He was also seen by psychiatrists and was treated with various drugs, including antidepressants. The proband and his parents were enrolled in a clinical research study at the Imperial College London Diabetes Centre (ICLDC) in Abu Dhabi. WES was performed for the proband, followed by Sanger sequencing of the proband and his parents to confirm the presence of the variant and to check the segregation of the identified variant within the family.
PMMO cohort
The PMMO cohort (https://classic.clinicaltrials.gov/ct2/show/NCT01365416) is an observational research study of individuals with severe obesity (BMI > 40, or BM > 35 with at least one co-morbidity) who were ascertained from family doctors and/or undergoing bariatric surgery in a UK hospital-based service. The research has ethical approval from the NRES Committee London Riverside (Reference: 11/LO/0935/principal investigator: Professor Alexandra Blakemore) and informed consent was obtained from each subject [9]. A sub-group of 91 PMMO participants with BMI > 50 and no known syndromic features, were selected for a pilot WES study.
Whole-exome sequencing and variant annotation
WES was performed using DNA from whole blood samples by the Genomics Service, MRC Clinical Sciences Centre, Imperial College London, UK. For this, an enriched library was prepared using SureSelectXT Human All Exon V4+UTRs and sequencing was run on a HiSeq25000 platform, generating 100 bp paired-end reads. FastQC version 0.10.0. was used to assess the quality of sequencing. Sequencing reads were mapped to the hg19 (GRCh37) reference genome through using BWA mem version 0.7.2. To refine the alignment Picard software (version 1.85) was used to remove duplicate reads and reduce false positive using BWA. For recalibration, realignments and variants calling the Genome Analysis Toolkit (GATK) were used. The annotated file was created using ANNOVAR.
For variant identification and analysis, variants were filtered and prioritised based on standard filtration and evaluation steps for WES data analysis as described previously in Alsters et al. [31]. This includes absence or low minor allele frequency in a public database (MAF < 1%), risk prediction by at least two out of four in silico prediction programs (Combined Annotation Dependent Depletion (CADD), Polyphen, Sorting Intolerant from Tolerant (SIFT) and, Proavean), and matching the relevant mode of inheritance for each gene [31, 32]. Subsequently, variants were re-screened against a list of monogenic obesity and syndromic obesity genes (see Supplementary Table 1). Sanger sequencing was carried out to confirm the presence of variants identified by WES, and to check for familial segregation in the parents of proband 1.
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Loos RJF, Yeo GSH. The genetics of obesity: from discovery to biology. Nat Rev Genet. 2022;23:120–33.
Bouchard C. Genetics of obesity: what we have learned over decades of research. Obesity. 2021;29:802–20.
Manara E, Paolacci S, D’Esposito F, Abeshi A, Ziccardi L, Falsini B, et al. Mutation profile of BBS genes in patients with Bardet–Biedl syndrome: an Italian study. Ital J Pediatr. 2019;45:72.
Mukherjee S, Cogan JD, Newman JH, Phillips JA 3rd, Hamid R, Undiagnosed Diseases Network, et al. Identifying digenic disease genes via machine learning in the Undiagnosed Diseases Network. Am J Hum Genet. 2021;108:1946–63.
Gifford CA, Ranade SS, Samarakoon R, Salunga HT, de Soysa T, Huang Y, et al. Oligogenic inheritance of a human heart disease involving a genetic modifier. Science. 2019;364:865–70.
Schaaf CP, Sabo A, Sakai Y, Crosby J, Muzny D, Hawes A, et al. Oligogenic heterozygosity in individuals with high-functioning autism spectrum disorders. Hum Mol Genet. 2011;20:3366–75.
Hoefele J, Wolf MT, O’Toole JF, Otto EA, Schultheiss U, Dêschenes G, et al. Evidence of oligogenic inheritance in nephronophthisis. J Am Soc Nephrol. 2007;18:2789–95.
Heyne HO, Karjalainen J, Karczewski KJ, Lemmelä SM, Zhou W, FinnGen, Havulinna AS, et al. Mono- and biallelic variant effects on disease at biobank scale. Nature. 2023;613:519–25.
NIHR. Personalise Medicine for Morbid Obesity. UK Clinical Trials Gateway. 2012.
Doche ME, Bochukova EG, Su HW, Pearce LR, Keogh J, Henning E, et al. Human SH2B1 mutations are associated with maladaptive behaviors and obesity. J Clin Invest. 2012;122:4732–6.
Stessman HAF, Willemsen MH, Fenckova M, Penn O, Hoischen A, Xiong B, et al. Disruption of POGZ is associated with intellectual disability and autism spectrum disorders. Am J Hum Genet. 2016;98:541–52.
White J, Beck CR, Harel T, Posey JE, Jhangiani S, Tang S, et al. POGZ truncating alleles cause syndromic intellectual disability. Genome Med. 2016;8:3.
Fitzgerald TW, Gerety SS, Jones WD, van Kogelenberg M, King DA, McRae J, et al. Large-scale discovery of novel genetic causes of developmental disorders. Nature. 2014;519:223–8.
Nozawa RS, Nagao K, Masuda HT, Iwasaki O, Hirota T, Nozaki N, et al. Human POGZ modulates dissociation of HP1α from mitotic chromosome arms through Aurora B activation. Nature Cell Biology. 2010;12:719–27.
Ye Y, Cho MT, Retterer K, Alexander N, Ben-Omran T, Al-Mureikhi M, et al. De novo POGZ mutations are associated with neurodevelopmental disorders and microcephaly. Cold Spring Harb Mol Case Stud. 2015;1:a000455.
Laget S, Joulie M, Le Masson F, Sasai N, Christians E, Pradhan S, et al. The human proteins MBD5 and MBD6 associate with heterochromatin but they do not bind methylated DNA. PLoS One. 2010;5:e11982.
Talkowski ME, Mullegama SV, Rosenfeld JA, van Bon BW, Shen Y, Repnikova E, et al. Assessment of 2q23.1 microdeletion syndrome implicates MBD5 as a single causal locus of intellectual disability, epilepsy, and autism spectrum disorder. Am J Hum Genet. 2011;89:551–63.
Mullegama SV, Elsea SH. Clinical and molecular aspects of MBD5-associated neurodevelopmental disorder (MAND). Eur J Hum Genet. 2016;24:1235–43.
Hodge JC, Mitchell E, Pillalamarri V, Toler TL, Bartel F, Kearney HM, et al. Disruption of MBD5 contributes to a spectrum of psychopathology and neurodevelopmental abnormalities. Mol Psychiatry. 2014;19:368–79.
Mullegama SV, Pugliesi L, Burns B, Shah Z, Tahir R, Gu Y, et al. MBD5 haploinsufficiency is associated with sleep disturbance and disrupts circadian pathways common to Smith-Magenis and fragile X syndromes. Eur J Hum Genet. 2015;23:781–9.
Shichiji M, Ito Y, Shimojima K, Nakamu H, Oguni H, Osawa M, et al. A cryptic microdeletion including MBD5 occurring within the breakpoint of a reciprocal translocation between chromosomes 2 and 5 in a patient with developmental delay and obesity. Am J Med Genet A. 2013;161A:850–5.
Marco EJ, Aitken AB, Nair VP, da Gente G, Gerdes M, Bologlu L, et al. Burden of de novo mutations and inherited rare single nucleotide variants in children with sensory processing dysfunction. BMC Med Genomics. 2018;11:50.
Han JY, Jang W, Park J, Kim M, Kim Y, Lee I, et al. Diagnostic approach with genetic tests for global developmental delay and/or intellectual disability: single tertiary center experience. Ann Hum Genet. 2018;83:115–23.
Tadros S, Wang R, Waters JJ, Waterman C, Collins A, Collinson M, et al. Inherited 2q23.1 microdeletions involving the MBD5 locus. Mol Genet Genomic Med. 2017;5:608–13.
Woodbury-Smith M, Nicolson R, Zarrei M, Yuen RKC, Walker S, Howe J, et al. Variable phenotype expression in a family segregating microdeletions of the NRXN1 and MBD5 autism spectrum disorder susceptibility genes. NPJ Genom Med. 2017;2:17.
Ishizuka K, Kimura H, Yoshimi A, Banno M, Kushima I, Uno Y, et al. Investigation of single-nucleotide variants in MBD5 associated with autism spectrum disorders and schizophrenia phenotypes. Nagoya J Med Sci. 2016;78:465–74.
Pettersson M, Viljakainen H, Loid P, Mustila T, Pekkinen M, Armenio M, et al. Copy number variants are enriched in individuals with early-onset obesity and highlight novel pathogenic pathways. J Clin Endocrinol Metab. 2017;102:3029–39.
Pearce LR, Joe R, Doche ME, Su HW, Keogh J, Henning E, et al. Functional characterization of obesity-associated variants involving the alpha and beta isoforms of human SH2B1. Endocrinology. 2014;155:3219–26.
Papadimitriou S, Gazzo A, Versbraegen N, Nachtegael C, Aerts J, et al. Predicting disease-causing variant combinations. Proc Natl Acad Sci USA. 2019;116:11878–87.
Renaux A, Papadimitriou S, Versbraegen N, Nachtegael C, Boutry S, Nowé A, et al. ORVAL: a novel platform for the prediction and exploration of disease-causing oligogenic variant combinations. Nucleic Acids Res. 2019;47:W93–W8.
Alsters SI, Goldstone AP, Buxton JL, Zekavati A, Sosinsky A, Yiorkas A, et al. Truncating homozygous mutation of carboxypeptidase E (CPE) in a morbidly obese female with type 2 diabetes mellitus, intellectual disability and hypogonadotrophic hypogonadism. PLoS ONE. 2015;10:e0131417.
An JY, Cristino AS, Zhao Q, Edson J, Williams SM, Ravine D, et al. Towards a molecular characterization of autism spectrum disorders: an exome sequencing and systems approach. Transl Psychiatry. 2014;4:e394.
Author information
Authors and Affiliations
Contributions
SA was responsible for conducting the research, analysing data, and writing the article. SIA focused on recruiting participants for the PMMO cohort, generating data, and performing the initial analysis. Both AMY and NANH contributed in bioinformatics processing of extracting and generating data. RGW provided expert advice on data analysis and results interpretation. HS, SP and NL offered clinical expertise and advice. NL also involved in recruiting participants from ICLDC. The principal investigator, AIFB, designed and initiated the study, providing overall direction and supervision of the research.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Almansoori, S., Alsters, S.I., Yiorkas, A.M. et al. Oligogenic inheritance in severe adult obesity. Int J Obes 48, 815–820 (2024). https://doi.org/10.1038/s41366-024-01476-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41366-024-01476-9
- Springer Nature Limited