Abstract
Childhood stunting is associated with impaired cognitive development and increased risk of infections, morbidity, and mortality. The composition of the enteric microbiota may contribute to the pathogenesis of stunting. We systematically reviewed and synthesized data from studies using high-throughput genomic sequencing methods to characterize the gut microbiome in stunted versus non-stunted children under 5 years in LMICs. We included 14 studies from Asia, Africa, and South America. Most studies did not report any significant differences in the alpha diversity, while a significantly higher beta diversity was observed in stunted children in four out of seven studies that reported beta diversity. At the phylum level, inconsistent associations with stunting were observed for Bacillota, Pseudomonadota, and Bacteroidota phyla. No single genus was associated with stunted children across all 14 studies, and some associations were incongruent by specific genera. Nonetheless, stunting was associated with an abundance of pathobionts that could drive inflammation, such as Escherichia/Shigella and Campylobacter, and a reduction of butyrate producers, including Faecalibacterium, Megasphera, Blautia, and increased Ruminoccoccus. An abundance of taxa thought to originate in the oropharynx was also reported in duodenal and fecal samples of stunted children, while metabolic pathways, including purine and pyrimidine biosynthesis, vitamin B biosynthesis, and carbohydrate and amino acid degradation pathways, predicted linear growth. Current studies show that stunted children can have distinct microbial patterns compared to non-stunted children, which could contribute to the pathogenesis of stunting.
Similar content being viewed by others
Introduction
Stunting is the most prevalent form of childhood malnutrition, affecting approximately 22% or 148.1 million children under five years old in 20221. Defined by the World Health Organization (WHO) as impaired growth and development experienced in children due to inadequate nutrition and repeated infections, it is often reported as a height for age more than two standard deviations below the standard child growth median. Although the global prevalence of childhood stunting has decreased from 30.1 to 22.2% in the past twenty years, there have been minimal reductions in the highest-burden regions of Africa and South Asia, where 42% and 53%, respectively, of all stunted children, live1,2. As the population in low- and middle-income countries (LMICs) continues to grow, the number of children affected by stunting will likely increase. Stunting has a range of adverse outcomes, including increased susceptibility, incidence, and severity of infectious diseases (particularly pneumonia and diarrhea), poor cognitive development, and mortality3,4,5. The pathophysiology of childhood stunting is likely multifaceted. Poor sanitation, recurrent infections, poor nutrition, and genetic predisposition are traditionally implicated in stunting6,7,8. However, despite the epidemiological and statistical associations, the pathophysiology of stunting remains unclear. Recent studies have pointed to the role of the gut microbiome in normal childhood growth and that alterations in the gut microbiome composition are associated with stunting and may play a role in its development9,10.
The gut microbiome, a complex community of bacteria, eukaryotic and prokaryotic viruses, fungi, and archaea in the gastrointestinal tract, has a range of functions relevant to normal childhood growth and development. These include the metabolism of macro- and micro-nutrients, production of vitamins for growth and maintaining gut health, hormone regulation facilitating production and energy harvesting from nutrients, and helping maintain the intestinal mucosa’s structure and barrier function11,12,13,14,15. The gut microbiome also shapes the development of the innate and adaptive immune system and provides colonization resistance against exogenous pathogens and enteric infections16,17,18. The colonization of the intestinal microbiome overlaps with critical developmental periods of child growth. Young children, particularly in LMICs, are at high risk for microbiota perturbation secondary to frequent infectious illness episodes, antibiotic use, poor nutrition, and unsanitary living environments. However, how these perturbations, and the composition and function of the developing microbiome, may translate into growth outcomes and health is uncertain9,19,20,21.
Although multiple studies have evaluated correlations between intestinal microbiome composition and stunting, and several review articles have been written on this subject, no systematic reviews have systematically evaluated and synthesized the associations between microbiome composition and stunting outcomes. Given the therapeutic potential of the microbiome and the need to understand its potential etiologic role in the development of stunting, this systematic review aimed to summarize and evaluate the existing literature that correlates the composition of the gut microbiota and childhood stunting in LMICs.
Results
Study summary overview
Study selection
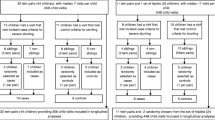
The literature search identified 935 studies (Medline-431, Embase-625, SCOPUS-120, and the Global Index Medicus-113). Three studies were identified by searching reference lists and grey literature. After removing duplicates and screening using titles and abstracts for inclusion and exclusion criteria, 39 articles were selected for full-text reading, and 14 articles met all the criteria and are included in this review. Figure 1 shows the PRISMA flow chart of article screening and selection stages.
Summary of included studies
Of the fourteen included studies, eight were conducted in Asia (Bangladesh22,23,24,25, India26,27, Indonesia28,29, five in Africa (Malawi25,30,31, Zimbabwe32 Madagascar, and the Central African Republic33 and two in Peru, South America34,35. Six studies assessed child-level microbiome characteristics at one time point: four were cross-sectional studies27,28,33, and two were case-control studies22,23. Eight studies evaluated the gut microbiome over a period of time: five24,25,26,29,30 assessed the microbiome in stunted children at baseline, and another followed up time point, while three studies31,34,35 followed healthy children and compared the gut microbiome in those who developed stunting with those who did not. The age of the children in the study population varied between studies from birth (0 days) to 5 years, while the number of study participants ranged from 20 to 404. A summary of the included studies is shown in Table 1.
The gut microbiome was assessed in fecal samples for all studies. In addition to fecal sampling, the gut microbiota was assessed using duodenal22,33 and gastric samples33. Except for two studies that used shotgun sequencing23,25, all studies evaluated the gut microbiome using 16S rRNA sequencing, although the region of amplification differed between the studies: V4 (n = 7), V3-V4 (n = 3), and V1-V3 (n = 1). Stunting in most studies was defined stunting using the WHO-standard definition of HAZ/LAZ z-score <−2. Three studies described stunting as poor growth velocity defined as a negative change in LAZ scores between two time points, specifically a difference of less than −0.3 between two growth measurement points30 and any negative change in HAZ/LAZ24,31.
Quality and Risk of Bias Assessment
All studies were evaluated for methodological quality using the Joanna Briggs Institute (JBI) critical appraisal tool. The largest risk of bias in almost all studies was related to unassessed potential confounding, as most studies did not adjust for confounders, such as diet and antibiotics use that could significantly influence the gut microbiome. Overall, the risk of bias for the studies was categorized as low or moderate (Supplementary Table 2).
Primary Outcomes
Alpha and beta diversity in stunted and non-stunted children
Of the included studies, 12 reported alpha diversity in stunted children using varying alpha diversity indices (Fig. 2). Only one study reported a higher alpha diversity in stunted children (using four diversity indices: Shannon, Pielou’s evenness, observed taxonomic units (OTUs), and Faith’s PD)28. Six studies reported a similar alpha diversity (Shannon) for stunted and non-stunted children24,26,27,30,31,35, and two studies using metagenomic sequencing methods reported significantly lower alpha diversity for Simpson’s25, Shannon’s and OTU’s23 indices. Four studies22,29,32,34 did not report alpha diversity.
Of the 14 studies included in this review, five studies23,26,30,33,34 had publicly available sequence and metadata available to re-assess for alpha diversity. We conducted a meta-analysis using the random effects model to test the association between stunting and Shannon diversity from publicly available raw reads. The forest plot shows that substantial heterogeneity was observed (I2=97%; p < 0.001), with no significant differences in Shannon diversity between stunted and non-stunted children (Fig. 3). We did not perform a metadata analysis due to the heterogeneity observed among the studies.
Similarly, the results of between-group comparisons for beta diversity were inconsistent among the studies (Fig. 2). Four studies reported a higher beta diversity in stunted children using weighted uniFrac distance26,27, unweighted uniFrac28, and Bray-Curtis dissimilarity28,35. No significant differences were observed between stunted and non-stunted children in four studies when using different measures: weighted uniFrac distance30, unweighted uniFrac27, and Bray-Curtis dissimilarity24,27,33. Seven studies did not report on beta diversity.
Differences in microbial composition at the phylum level
Six studies reported differences in microbial composition at the phylum level in stunted and non-stunted children; all were conducted in Asia (Table 2). Stunted children had a higher abundance of Bacillota28,29, Bacteroidota26,29, and Pseudomonoadota23,29. A lower abundance for Bacteroidota was reported in stunted children in28, while no significant differences were observed between stunted and non-stunted children for Pseudomonoadota24,26,28; Bacteroidota24; Bacillota23,24,26; and Actinomycetota23,24,26,28.
Differences in the microbial composition at the genus level
Twelve studies reported significant differences in the relative bacterial abundances of fecal samples in stunted vs. non-stunted children at the genus level (Fig. 4). They report an over-representation of genera from the Baccilota and Pseudomonadota phyla in stunted children. However, associations between these genera and stunting were not always consistent. A higher relative abundance was observed for Bacillota (Weisella, Veillonella, Allisonella, Acidaminococcus, Catenibacterium, and Streptococcus), Pseudomonadota (Escherichia/Shigella, Desulfovibrio, Aggregatibacter, Neisseria, Campylobacter and others). Taxa from other phyla in higher abundance in stunted children included Micrococcus and Rothia (Actinomycetota) and Porphyromonas (Bacteroidota). Taxa with a significantly lower relative abundance in stunted children included Bacillota (Megasphaera, Dorea, Lachnospira, Megamonas, Blautia, Bacteroides, Enterobacter, Anearococcus), Bacteroidota (Bacteroides, Parabacteroides, Coprobacter), Actinomycetota (Olsenella, Bifidobacterium) and Pseudomonadota (Enterobacter). Discrepant or inconsistent associations were reported for ten genera: Providencia, Prevotella, Ruminococcus, Lactobacillus, Klebsiella, Helicobacter, Faecalibacterium, Eubacterium, Dialister, and Clostridium. Two studies31,32 did not report any significant difference in the microbiome composition of stunted and non-stunted children. The complete list of genera identified and their associations with stunting are shown in Fig. 3. We also observed that only Campylobacter was reported to be significantly higher in stunted children across the three regions. Stunted children in Africa and Asia had more shared taxa, including an abundance of Escherichia/Shigella, Aggregatibacter, Veillonella, Streptococcus, and reduced Bifidobacterium, Blautia, and Megasphera (Supplementary Fig. 1).
Duodenal and gastric microbiota
The duodenal microbiota in stunted was reported in two studies from Asia (Bangladesh)22 and Africa (Madagascar and Central African Republic)33 (Fig. 5). In both studies, the duodenal samples from stunted children had an overrepresentation of Veillonella, Gemella, Neisseria, Actinomyces, Haemophilus, and Rothia species. We cross-referenced taxa from the duodenum with those from stool to assess taxa present in both sample types and found Rothia, Veillonella, Streptococcus, Gemella, Fusobacterium, Neisseria, and Haemophilus were abundant in both samples (Fig. 5). However, several taxa with a higher abundance in stunted children were only reported in duodenal samples, including Staphylococcus, Pastuerella, Johnsenella, Moraxella, and Kingella. We also observed that other than the two studies reporting on duodenal taxa, only one further study reported similar taxa in stool. The gastric microbiome was only reported in one study and had taxa similar to the duodenal taxa reported33.
The gut virome
In addition to bacteria, the gastrointestinal tract also hosts diverse eukaryotic and prokaryotic viruses that make up the virome. The virome is dominated by prokaryotic viruses or bacteriophages36. Two studies described associations between the eukaryotic gut virome30 and prokaryotic virome and stunting23,30. In Desai et al., no viral taxa were associated with growth (stunting) indices, and the viral diversity and abundance were not statistically different between children with adequate and poor growth velocities. They also reported an increase in bacteriophage richness with an increase in bacterial richness in children with adequate and moderate growth, which was not observed in children with poor growth. Similarly, in Mirzei et al., phage diversity and richness were higher in non-stunted children than in stunted children, and phage diversity, parallel with bacterial diversity, was higher in non-stunted children than in stunted children23.
Metabolic Pathways
Of the studies included, only one study32 reported on metagenomic pathways associated with linear growth. The study evaluated the microbiome, including associations with growth in HIV-exposed and non-exposed children in rural Zimbabwe. Although no significant taxonomic differences were observed in stunted and non-stunted children, a spectrum of metabolic pathways associated with and could predict linear growth. They show that pathways encoding lipid biosynthesis, B vitamins, and purine and pyrimidine biosynthesis were predictive of LAZ at 1 and 2 months, while at 3 and 6 months, pathways encoding fermentation and carbohydrate biosynthesis were better predictors of LAZ. At 18 months, carbohydrate and amino acid degradation pathways predicted linear growth.
Discussion
Childhood stunting has been associated with micronutrient deficiencies, repeated enteric infections that increase both nutrient loss and energy requirements, and helminthic infections37,38. We provide a synthesis of recent data from 14 studies that identify microbiota phenotypes associated with stunting across cohorts and geography to better understand how the microbiota may interact with the pathophysiology of stunting. Our results show that all but two studies31,32 identified associations between the gut microbiome and childhood stunting. We also show that while some bacterial taxa differed between stunted and non-stunted children, their associations were inconsistent across the studies at both phylum and genus levels. No one genus was associated with stunting across all studies. However, stunting was consistently associated with abundant putative pathobionts, including Escherichia/Shigella, Campylobacter, Desulfovibrio, and Neisseria. On the other hand, the duodenal microbiota in stunted children was abundant in taxa thought to normally reside in the nasopharyngeal tract. Specific taxonomic categories, including Veillonela, Neisseria, Streptococcus, and Haemophilus, were reported with an increased frequency in stunted children across geographies in both duodenal and fecal samples.
One possible explanation for observed differences between studies could be variability in the definition of stunting among studies. While the WHO growth standard using Z-scores (HAZ/LAZ<-2) is widely employed to define stunting, some studies defined stunting as a change in Z-scores between two time points. Ongoing discussion exists about the best approach to identify children with growth deficits and stunting. Some researchers argue that children described as ‘not stunted’ by WHO standards could have significant growth deficits and experience stunting even as they fit into the normal growth range and that there is no significant change in their risks and consequences of stunting simply by crossing the stunting cutoff. They suggest that a more sensitive definition of stunting should be based on a child’s growth between two time points20. This approach may be more sensitive than cross-sectional measurements of stunting as it takes into account the child’s growth trajectory over time and measures the magnitude of growth faltering.
Our review identified several associations of the gut microbiome that support mechanistic hypotheses about how the gut microbiome may impact childhood stunting. Stunting is a nutritional disorder within the spectrum of malnutrition characterized by distinct clinical and phenotypic sequelae and risk factors39. While this review focuses strictly on studies that measure stunting as an outcome, there are potential mechanistic overlaps with the extensive literature on microbiome and malnutrition40,41,42. Gut microbiome immaturity may also contribute to stunting. Several of the taxa identified in this review have also been reported in studies in Bangladesh and Malawi evaluating the microbiome in children suffering from severe acute malnutrition40,43,44. In these studies, gut microbiome immaturity was evaluated using machine-learning techniques on age-discriminatory bacterial species and reported as a ‘microbiota for age Z-score’ defining the degree to which a gut microbiota deviates from that of healthy age-matched children—an immature microbiome. The immature microbiome associating with severe malnutrition was characterized by a reduced microbial diversity, a depletion of Faecalibacterium, Bifidobacterium, Clostridium, and Ruminococcus, and an increase in pathogenic microorganisms at 18 months. The studies also report that diarrhea, formula-feeding and diarrheal events had significant effects on the relative microbiota maturation index. The authors hypothesize that microbiota immaturity can contribute to systemic inflammation, impaired nutrient synthesis, and absorption, exacerbating the malnutrition cycle.
Another shared mechanism is the gut microbiome’s role in maintaining the integrity of the intestinal barrier that holds back microorganisms, antigens, and toxins from the mucosal tissues. A disturbance of the gut microbiome can lead to increased intestinal permeability, which can cause intestinal inflammation. Inflammation, in turn, can alter the structure and function of the small intestinal villi, reducing the absorptive surface area for nutrient uptake and limiting the uptake of essential nutrients45,46. Species, particularly from the Pseudomonadota phylum, reported in this review to associate with stunting, have been shown to activate inflammatory cascades 47,48 and have been associated with dysbiosis, disease, and malnutrition in children49,50. Although only two studies23,29 in this review observed significantly higher abundance at the phylum level, at the genus level, consistent negative associations for stunting were observed for putative pathobionts such as Escherichia/Shigella23,24,27,33, Campylobacter26,33,34, Desulfovibrio, and Neisseria29,33 from this phylum. These taxa have also been reported in studies evaluating malnourished children in Bangladesh51 and children with Kwashiorkor and Marasmus, a severe form of malnutrition, from Niger and Senegal49,52 and The Gambia53. These results suggest that some microbiome characteristics are shared across the malnutrition spectrum, with these taxa associating with differing phenotypic/clinical outcomes such as stunting, moderate acute malnutrition (MAM), and more severe phenotypes such as Kwashiorkor and Marasmus. It is unclear if these bacteria result from other underlying processes or are causal in the stunting pathway. However, these pathobionts, particularly in a disturbed microbial ecosystem, have pathogenic potential, with the ability to secrete toxins, invade the intestinal epithelium, and trigger inflammatory responses47,48,54. Desulfovibrio, for example, plays a role in sulfate reduction by breaking down dietary sulfates, producing hydrogen sulfide, high concentrations of which may cause inflammation and damage to the gut lining55, while Escherichia/Shigella, which share many virulence strategies, can cause damage to enterocytes, cell death and tissue damage, igniting pro-inflammatory cascades56,57 Even without obvious symptoms, this can increase the risk of gastroenteritis and infections. It can also cause a redirection of the limited amino acids and energy needed for growth to immune function, which can worsen growth impairment58.
Our review also showed that stunted children often have a lower abundance of putative butyrate producers, including Faecalibacterium, Megasphera, Blautia, and Bifidobacterium, and an increased abundance of Ruminococcus. These taxa are known to produce microbial metabolites known as short-chain acids (SCFAs), which serve as an important source of energy for colonic cells, aid in metabolic processes, and maintain the health of the colonic mucosa by inhibiting the pathways that lead to the production of pro-inflammatory cytokines, thereby regulating mucosal inflammation59,60,61. They also induce innate immunity and play an important role in assimilating nutrients and gut maturation48,62,63. The importance of butyrate producers was demonstrated by Blanton et al. when growth and metabolic abnormalities were partially restored to undernourished mice after the addition of Ruminococcus gnavus and Clostridium symbiosum43.
Studies using direct biopsy sampling from the duodenum and the stomach provided further insight into the association between the infant gut microbiome and stunting in children. These studies are particularly important as metagenomic samples do not represent all microbial communities found along the gastrointestinal tract mucosa, and the duodenal microbiome would help better understand the ecology in the small intestine where absorption of nutrients occurs64,65,66. In both Vonaesch et al. and Chen et al., a significant overrepresentation of taxa thought to be ordinarily resident in the oropharyngeal tract was observed in the duodenum of stunted children (Fig. 5). Vonaesch et al. phrased their finding as “a decompartmentalization of the gastrointestinal tract,” where microbial taxa, usually resident in the oropharynx, are overrepresented along the gastrointestinal tract33, resulting in inflammation and damage of the small intestinal walls, characteristic of environmental enteric dysfunction, leading to stunting. We cross-referenced the duodenal and fecal taxa reported in other studies and observed that other than Chen et al. and Vonaesch et al., only one further study29 reported an over-representation of taxa usually found in the nasopharynx in stool samples. These taxa included Rothia, Veillonella, Streptococcus, Haemophilus, Gemella, and Neisseria (Fig. 5). However, as no control groups were used for the duodenal studies, it is impossible to tell if these taxa are part of the normal flora whose abundance was altered or if the sampling technique that involves intubation or capsulation could have introduced these bacteria from the oropharynx. Additionally, samples from the oropharyngeal sites were not collected to compare with duodenal samples to determine the similarity of the taxa. Including control groups in such studies remains a challenge, as collecting samples in otherwise healthy children is unethical. To circumvent this bottleneck, there is a need to develop less invasive sample collection techniques in children similar to those used by Shalon et al.67.
An analysis of metagenomic pathways suggests that the capacity of the gut microbiome to influence growth depends on the interactions of several metagenomic pathways, and some taxa present in early life may be functionally redundant32. B vitamin biosynthesis was consistently a top predictive feature of LAZ, contrasting with results from a large randomized controlled study of vitamin B12 supplementation, which showed that vitamin B-12 levels at baseline did not predict linear growth in those not taking B-12 supplements15. Vitamin B12 is both synthesized and utilized by bacteria in the gut microbiome. It may, therefore, simply be a marker of an underlying dysbiotic microbiome and not causal in the linear growth pathway. Conversely, supplementation of vitamin B12 alone in a dysbiotic gut may not redress underlying drivers of dysbiosis, such as inflammation and malabsorption68. However, there is very limited data in this area of research, and more data is required to arrive at robust conclusions. Other areas of research have reported promising outcomes for microbiota-directed therapies and can exemplify the methodology required to establish a direct causal link between the gut microbiome and growth outcomes. In their randomized feeding study, where they compared a ready-to-use supplementary food (RUSF) and a microbiota-directed complementary food prototype (MDCF-2) in young children, Gordon’s group was able to demonstrate that modulation of targeted microbiota components found in association studies altered growth outcomes in mouse models and human trials. Children receiving the MDCF-2 showed a change in 21 bacterial taxa significantly positively associated with weight for length zscore and 70 proteins, including mediators of bone growth and neurodevelopment69. Similarly, another randomized feeding study by Gehrig et al. 2019, identified an MDCF that was effective in repairing the microbiota, increasing biomarker levels of growth, immune function, and bone formation70. Such results support a causal link between microbial patterns and growth outcomes and demonstrate the potential of microbial-directed therapies to improve childhood stunting, and this review provides valuable data on potential target taxa.
Strengths and limitations
To the best of our knowledge, this is the first systematic review to perform an unbiased synthesis of research on associations between the gut microbiome and childhood stunting in LMICs where the burden of stunting is highest. We used a comprehensive search strategy with a rigorous methodology to limit bias and to provide a comprehensive overview of available data on the gut microbiome and childhood stunting.
Our review has methodological limitations that should be considered when interpreting the results. Firstly, even though all studies were carried out in developing countries, there was an overrepresentation of studies from the Asian region (9, 5, and 2 studies for Asia, Africa, and S. America). Geographic location, diet, sanitation and hygiene, healthcare systems, etc., could impact the synthesis of the results and associations observed across the studies.
In addition to the above-stated, we observed several limitations in the study designs of the included studies. Most studies used small sample sizes, as such, the results cannot be generalized to whole populations. Secondly, cross-sectional studies do not provide sufficient information on the genesis of stunting, while longitudinal studies that utilize sampling with wide time intervals between sample collection points are not ideal for documenting temporal variations and changes in the gut microbiota over time, as age significantly influences the infant gut microbiota71.
Third, the studies in this review used 16S rRNA and shotgun sequencing methods. 16S rRNA sequencing methods have been acknowledged to have limited sensitivity and resolution compared to metagenomics72. This could be a potential source of the observed discrepant associations, e.g., at the genus level, the alpha diversity (Shannon and Simpson) in this review was lower in studies using Shotgun metagenomics compared to 16S rRNA methods. The different regions of amplification (V4, V3, V1-V3, and V3-V4) and pipelines used (DADA2, QIIME, and Mothur) in the studies in this review could influence the observed results. Additionally, one study used pooled samples for analysis23.
Another key limitation was that most studies were observational and only showed associations with stunting but not causality and, as such, did not answer whether a specific gut microbiome composition is a cause or consequence of stunting. However, Chen et al., in their study, evaluated the causal relationship between their findings (that 14 bacterial taxa were associated with poor growth) and the pathogenesis of stunting, providing mechanistic animal and proof of principle data to establish the causal pathway. They did this by administering identified bacterial taxa into germ-free mice. Of the 23 bacterial strains they detected in mice, nine corresponded to the core duodenal bacterial taxa (taxa present in over 80% of duodenal aspirates), which reported a negative correlation with LAZ z-scores showing that stunting phenotypes can be transmitted via microbial taxa22. More mechanistic proof of principle, animal, and clinical studies are needed to establish the causal pathway.
We observed high heterogeneity among the studies, which precluded a meta-analysis. Variations in the age of participants (from birth to 5 years), the definition of the outcome, the study design, sequencing techniques used, and geographic location made pooling results difficult. As such, we only provide a narrative synthesis of the data, and caution should be taken when generalizing these findings.
Despite the limitations listed above, this review shows that stunted children can have distinct differences in microbial composition, suggesting a potential role of the gut microbiome in childhood stunting. These findings provide important groundwork for further research to (i) improve our understanding of the mechanisms underlying the gut microbiome’s influence on childhood stunting and (ii) develop targeted and effective interventions to prevent and improve stunting in children.
Impact, Future Direction, and Conclusion
Understanding the role of the gut microbiome in human health, particularly childhood stunting, has important implications for developing interventions aimed at and improving stunting outcomes. As such, using the gut microbiome to improve health outcomes is a promising area for future research. There is an urgent need for microbiome studies to have clinically relevant results. Our systematic review identified inconsistent phenotype association with stunting and future studies should move this field forward by exploring the mechanisms and functions the microbiome may play in predisposing young children to stunting. Studies should first identify microbial compositional or metabolic profiles that can be used as biomarkers for early diagnosis of stunting. Current studies in this review provide a descriptive characterization of the gut microbiome, which provides valuable information on the composition of bacterial taxa but does not fully capture the mechanistic and functional roles and interactions of these bacteria within the gut ecosystem. More studies combining advanced techniques such as metagenomics, metabolomics, and transcriptomics would provide a more comprehensive view of present organisms, their functions, metabolic pathways, and the changes they induce in the gut to contribute to child growth. Second, future research should identify mechanistic pathways that permit microbiome-based interventions to treat stunting more effectively. Therapies targeting the gut microbiome, such as microbiota-directed foods designed to promote the growth of taxa associated with healthy growth, have been developed as potential alternatives or complements to standard management of stunting69,70. However, these approaches need further research to determine their safety and effectiveness and must be easily accessible and affordable in resource-limited settings where the need is greatest.
In conclusion, this systematic review highlights associations between gut microbiome composition and childhood stunting. While there is growing interest in understanding the gut microbiome’s role in childhood stunting, current research remains limited. In this review we show both inconsistent and consistent changes in microbial composition across different studies between stunted and non-stunted children. Future research should employ improved study methodologies evaluating the function of the microbiome in stunted children. Such an evidence base would provide hope that a broader range of microbiome-based interventions may be employed to prevent and reverse the devastating impacts of stunting in children in low- and middle-income settings in the future.
Methods
We conducted a systematic literature search for studies examining any gut microbiota associations with early childhood growth. Our search strategy followed the Population, Intervention, Comparison, and Outcomes (PICO) and Preferred Reporting Items for Systematic reviews and Meta-analyses (PRISMA) guidelines for reporting systematic reviews73,74, and the protocol was registered on PROSPERO (#CRD42022307788). A meta-analysis on alpha diversity was conducted using available raw data to determine the heterogeneity of the studies.
Eligibility criteria
Studies were included if they used randomized clinical trials (RCTs) or observational study designs. Further inclusion criteria were: studies in under-five children residing in an LMIC who had both their gut microbiome profiled and stunting status or growth velocity documented; studies using high throughput sequencing methods such as 16S rRNA, Shotgun metagenomic sequencing methods, etc., to define the gut microbiome; studies reporting on the presence and abundance of microbial taxa in stunted and healthy children. Studies were excluded if they did not provide sufficient information to investigate the question of interest (i.e., missing sequencing methodology, age of the child, participant stunting status), if they were case reports on single participants, or if they reported on the microbiota after specific interventions without data on baseline status.
Data Sources and Search Strategy
A systematic literature review was conducted in Global Medicus Index, MEDLINE, and EMBASE to identify published articles and abstracts between 2005 and February 29, 2023. Searches using medical subject headings (MeSH) terms and keywords or combinations of free-text words for “gut microbiota,” “children,” “LMICs,” and stunting” were used in the search strategy Boolean operators (“AND,” “OR,” and “NOT”) were used to broaden or narrow the search results (Supplementary Table 1). Identified articles were exported to Rayyan, where duplicate records were identified and removed. Two reviewers (M.C and V.H) independently reviewed abstracts, and articles meeting inclusion criteria were selected to review the full-text article further. We also examined the reference lists of selected articles to identify additional relevant studies. Any disagreements on the studies to include were resolved through a discussion, and when no consensus was reached, a third reviewer (M.S) was consulted. The search strategy and results are shown in Supplementary Table 1.
Study selection and data extraction
Study selection was based on the inclusion and exclusion criteria. The first selection was based on titles, and the second was on abstracts of selected titles. Finally, full-text screening was conducted to decide the final articles to be included in the review. Abstracts, titles, and reviews of the full text of selected abstracts were independently done by two reviewers (M.C and V.H). Any disagreements on the studies to include were resolved by consensus. A third reviewer (C.C.L) resolved conflicts if an agreement on inclusion was not reached. Data extraction from the eligible studies was performed independently by M.C. and V.H. using an Excel spreadsheet and compared. The data extracted included the study title, author, year of publication, study objective, location, study design, inclusion and exclusion criteria, stunting definition, study duration, and analytical methods used to describe the gut microbiome, study population, and all reported results on bacterial diversity and richness and bacterial taxonomic composition. Any missing data were noted in the reports. The data extracted were screened for completeness and accuracy by M.C. and V.H.
Exposures and outcomes
The primary exposure of interest was the gut microbiota, specifically the alpha and beta-diversity and bacterial taxonomic composition (phylum and genus) and metagenomic pathways. The primary outcome for this review was childhood stunting, defined as either a length/height-for-age z-score (LAZ/HAZ) less than two standard deviations below the standard child growth median, or poor growth velocity, defined as a negative change in LAZ/HAZ between two growth measurement points. We therefore sought to compare the microbiome diversity, taxonomic composition, and metagenomic pathways of stunted versus non-stunted children across studies.
Quality assessment
Quality assessment was conducted on all the studies using the Joanna Briggs Institute (JBI) assessment checklist for case-control, cross-sectional, and cohort studies75 by two independent reviewers (M.C. and C.C.L.).
Data availability
All statistical analyses were carried out in R software R 4.1.0. All graphs were plotted using ggplot2 v3.2.1, cowplot v1.1.3, and forestplot v 3.1.3 packages. No customized codes were used in the analyses. The data used to create the figures is available on Figshare (10.6084/m9.figshare.25577259).
References
UNICEF, WHO. World Bank. Levels and Trends in Child Malnutrition: UNICEF-WHO-World Bank Joint Child Malnutrition Estimates [Internet]. Available from: https://www.who.int/publications/i/item/9789240073791. (2023).
WHO MULTICENTRE GROWTH REFERENCE STUDY GROUP. WHO Child Growth Standards: Length/height-for-age, weight-for-age, weight-for-length, weight-forheight and body mass index-for-age: methods and development. Geneva, Switzerland. Acta Paediatr. 450, 76–85 (2006).
Kossmann, J., Nestel, P., Herrera, M. G., el Amin, A. & Fawzi, W. W. Undernutrition in relation to childhood infections: a prospective study in the Sudan. Eur. J. Clin. Nutr. 54, 463–472 (2000).
Caulfield, L. E., de Onis, M., Blössner, M. & Black, R. E. Undernutrition as an underlying cause of child deaths associated with diarrhea, pneumonia, malaria, and measles. Am. J. Clin. Nutr. 80, 193–198 (2004).
Dewey, K. G. & Begum, K. Long-term consequences of stunting in early life. Matern Child Nutr. 7, 5–18 (2011).
Danaei, G. et al. Risk factors for childhood stunting in 137 developing countries: a comparative risk assessment analysis at global, regional, and Country levels. PLoS Med. 13, e1002164 (2016).
Checkley, W. et al. Multi-country analysis of the effects of diarrhoea on childhood stunting. Int J. Epidemiol. 37, 816–830 (2008).
Yatsunenko, T. et al. Human gut microbiome viewed across age and geography. Nature 486, 222–227 (2012).
Robertson, R. C., Manges, A. R., Finlay, B. B. & Prendergast, A. J. The Human Microbiome and Child Growth – First 1000 Days and Beyond. Trends Microbiol. p. 131–147. https://doi.org/10.1016/j.tim.2018.09.008 (2019).
Million, M., Diallo, A. & Raoult, D. Gut microbiota and malnutrition. Microb. Pathogen. 106, 127–138 (2017).
Turnbaugh, P. J. et al. An obesity-associated gut microbiome with increased capacity for energy harvest. Nature 444, 1027–1031 (2006).
Ley, R. E., Turnbaugh, P. J., Klein, S. & Gordon, J. I. Microbial ecology: Human gut microbes associated with obesity. Nature 444, 1022–1023 (2006).
Chambers, E. S., Morrison, D. J. & Frost, G. Control of appetite and energy intake by SCFA: What are the potential underlying mechanisms? Proc. Nutr. Soc. 74, 328–336 (2015).
Schroeder, B. O. & Bäckhed, F. Signals from the gut microbiota to distant organs in physiology and disease. Nat. Med [Internet] 22, 1079–1089 (2016).
Strand, T. A. et al. Vitamin B-12, folic acid, and growth in 6- to 30-month-old children: a randomized controlled trial. Pediatrics [Internet] 135, e918–e926 (2015).
Baümler, A. J. & Sperandio, V. Interactions between the microbiota and pathogenic bacteria in the gut. Nature 535, 85–93 (2016).
Gensollen, T., Iyer, S. S., Kasper, D. L. & Blumberg, R. S. How colonization by microbiota in early life shapes the immune system. Sci. (1979). 352, 539–544 (2016).
Kane, A. V., Dinh, D. M. & Ward, H. D. Childhood malnutrition and the intestinal microbiome. Pediatr. Res. 77, 256–262 (2015).
Arrieta, M. C. et al. Early infancy microbial and metabolic alterations affect risk of childhood asthma. Sci. Transl. Med. 7, 307ra152 (2015).
de Onis, M. & Branca, F. Childhood stunting: a global perspective. Matern Child Nutr. 12, 12–26 (2016).
Mameli, C., Mazzantini, S. & Zuccotti, G. V. Nutrition in the first 1000 days: the origin of childhood obesity. Int. J. Environ. Res. Public Health 13, 838 (2016).
Chen, R. Y. et al. Duodenal microbiota in stunted undernourished children with enteropathy. N. Engl. J. Med. 383, 321–333 (2020).
Khan Mirzaei, M. et al. Bacteriophages isolated from stunted children can regulate gut bacterial communities in an age-specific manner. Cell Host Microbe 27, 199–212.e5 (2020).
Perin, J. et al. A retrospective case-control study of the relationship between the gut microbiota, enteropathy, and child growth. Am. J. Tropical Med. Hyg. 103, 520–527 (2020).
Gough, E. K. et al. Linear growth faltering in infants is associated with Acidaminococcus sp. and community-level changes in the gut microbiota. Microbiome 3, 24 (2015).
Dinh, D. M. et al. Longitudinal analysis of the intestinal microbiota in persistently stunted young children in South India. PLoS ONE [Electron. Resour.] 11, e0155405 (2016).
Shivakumar, N. et al. Gut microbiota profiles of young South Indian children: child sex-specific relations with growth. PLoS ONE [Electron. Resour.] 16, e0251803 (2021).
Surono, I. S., Widiyanti, D., Kusumo, P. D. & Venema, K. Gut microbiota profile of Indonesian stunted children and children with normal nutritional status. PLoS ONE [Electron. Resour.] 16, e0245399 (2021).
Masrul, M. et al. Microbiota profile with stunting children in west sumatera province, indonesia. Open Access Maced. J. Med Sci. 8, 334–340 (2020).
Desai, C. et al. Growth velocity in children with environmental enteric dysfunction is associated with specific bacterial and viral taxa of the gastrointestinal tract in malawian children. PLoS Negl. Trop. Dis. 14, e0008387 (2020).
Kamng’ona, A. W. et al. The association of gut microbiota characteristics in Malawian infants with growth and inflammation. Sci. Rep. 9, 12893 (2019).
Robertson, R. C. et al. The gut microbiome and early-life growth in a population with high prevalence of stunting. Nat. Commun. 14, 654 (2023).
Vonaesch, P. et al. Stunted childhood growth is associated with decompartmentalization of the gastrointestinal tract and overgrowth of oropharyngeal taxa. Proc. Natl Acad. Sci. USA 115, E8489–E8498 (2018).
Rouhani, S. et al. Gut microbiota features associated with campylobacter burden and postnatal linear growth deficits in a Peruvian birth cohort. Clin. Infect. Dis. 71, 1000–1007 (2020).
Zambruni, M. et al. Stunting is preceded by intestinal mucosal damage and microbiome changes and is associated with systemic inflammation in a cohort of Peruvian infants. Am. J. Tropical Med. Hyg. 101, 1009–1017 (2019).
Shkoporov, A. N. et al. The human gut virome is highly diverse, stable, and individual specific. Cell Host Microbe 26, 527–541 (2019).
Stephenson, L. S., Latham, M. C. & Ottesen, E. A. Malnutrition and parasitic helminth infections. Parasitology [Internet]. 2001/06/15. Available from: https://www.cambridge.org/core/article/malnutrition-and-parasitic-helminth-infections/7B648AE4E9EBC6C006AC39272FFA7BE7121, S23–S38 (2000).
Guerrant, R. L., Deboer, M. D., Moore, S. R., Scharf, R. J. & Lima, A. A. M. The impoverished gut - A triple burden of diarrhoea, stunting and chronic disease. Nat. Rev. Gastroenterol. Hepatol. 10, 220–229 (2013).
Prendergast, A. J. & Humphrey, J. H. The stunting syndrome in developing countries. Paediatr Int Child Health [Internet]. 2014. Nov;34:250–265. Available from: https://pubmed.ncbi.nlm.nih.gov/25310000.
Smith, M. I. et al. Gut microbiomes of Malawian twin pairs discordant for kwashiorkor. Sci. (1979). 339, 548–554 (2013).
Ghosh, T. S. et al. Gut microbiomes of Indian children of varying nutritional status. PLoS ONE [Electron. Resour.] 9, e95547 (2014).
Raman, A. S. et al. A sparse covarying unit that describes healthy and impaired human gut microbiota development. Sci. (1979) [Internet]. 365, eaau4735 (2019).
Blanton, L. V. et al. Gut bacteria that prevent growth impairments transmitted by microbiota from malnourished children. Science. 2016 Feb;351: https://doi.org/10.1126/science.aad3311.
Subramanian, S. et al. Persistent gut microbiota immaturity in malnourished Bangladeshi children. Nature 510, 417–421 (2014).
Michielan, A. & D’Incà, R. Intestinal permeability in inflammatory bowel disease: pathogenesis, clinical evaluation, and therapy of leaky gut. Neuwirt H., editor. Mediators Inflamm [Internet] 2015, 628157 (2015).
Bischoff, S. C. et al. Intestinal permeability – a new target for disease prevention and therapy. BMC Gastroenterol. [Internet] 14, 189 (2014).
Strober, W. Impact of the gut microbiome on mucosal inflammation. Trends Immunol [Internet] 34, 423–430. Available from: https://www.sciencedirect.com/science/article/pii/S1471490613001087 (2013).
Belkaid, Y. & Hand, T. W. Role of the microbiota in immunity and inflammation. Cell 157, 121–141 (2014).
Pham, T. P. et al. Gut microbiota alteration is characterized by a proteobacteria and Fusobacteria bloom in Kwashiorkor and a bacteroidetes paucity in Marasmus. Sci. Rep. 9, 9084 (2019).
Lupp, C. et al. Host-mediated inflammation disrupts the intestinal microbiota and promotes the overgrowth of Enterobacteriaceae. Cell Host Microbe 2, 119–129 (2007).
Monira, S. et al. Gut microbiota of healthy and malnourished children in Bangladesh. Front Microbiol. 2, 228 (2011).
Million, M. & Raoult, D. Linking gut redox to human microbiome. Hum. Micro. J. 10, 27–32 (2018).
Nabwera, H. M. et al. Interactions between fecal gut microbiome, enteric pathogens, and energy regulating hormones among acutely malnourished rural Gambian children. EBioMedicine 73, 103644 (2021).
Yu, L. C. H. Microbiota dysbiosis and barrier dysfunction in inflammatory bowel disease and colorectal cancers: exploring a common ground hypothesis. J. Biomed. Sci. 25, 79 (2018).
Carbonero, F., Benefiel, A., Alizadeh-Ghamsari, A. & Gaskins, H. R. Microbial pathways in colonic sulfur metabolism and links with health and disease. Front. Physiol. 3. Available from: https://www.frontiersin.org/articles/10.3389/fphys.2012.00448 (2012).
Croxen, M. A. & Finlay, B. B. Molecular mechanisms of Escherichia coli pathogenicity. Nat. Rev. Microbiol. 8, 26–38 (2010).
Phalipon, A. & Sansonetti, P. J. Shigella’s ways of manipulating the host intestinal innate and adaptive immune system: a tool box for survival? Immunol. Cell Biol. 85, 119–129 (2007).
Rouhani, S. et al. Diarrhea as a potential cause and consequence of reduced gut microbial diversity among undernourished children in peru. Clin. Infect. Dis. 71, 989–999 (2020).
Canani, R. B. et al. Potential beneficial effects of butyrate in intestinal and extraintestinal diseases. World J. Gastroenterol. 17, 1519–1528 (2011).
Fluitman, K. S. et al. The intestinal microbiota, energy balance, and malnutrition: emphasis on the role of short-chain fatty acids. Expert Rev. Endocrinol. Metab. 12, 215–226 (2017).
Mowat, A. M. & Agace, W. W. Regional specialization within the intestinal immune system. Nat. Rev. Immunol. [Internet] 14, 667–685 (2014).
Mendez-Salazar, E. O., Ortiz-Lopez, M. G., Granados-Silvestre, M. D. L. A., Palacios-Gonzalez, B. & Menjivar, M. Altered gut microbiota and compositional changes in firmicutes and proteobacteria in mexican undernourished and obese children. Front Microbiol. 9, 2494 (2018).
Iddrisu, I. et al. Malnutrition and gut microbiota in children. Nutrients 13, 8 (2021).
Lkhagva, E. et al. The regional diversity of gut microbiome along the GI tract of male C57BL/6 mice. BMC Microbiol. 21, 44 (2021).
Hillman, E. T., Lu, H., Yao, T. & Nakatsu, C. H. Microbial ecology along the gastrointestinal tract. Microbes Environ. 32, 300–313 (2017).
Leite, G. G. S. et al. Mapping the segmental microbiomes in the human small bowel in comparison with stool: a REIMAGINE study. Dig. Dis. Sci. 65, 2595–2604 (2020).
Shalon, D. et al. Profiling the human intestinal environment under physiological conditions. Nature 617, 581–591 (2023).
Guetterman, H. M. et al. Vitamin B-12 and the gastrointestinal microbiome: a systematic review. 13, Advances in Nutrition. Oxford University Press, p. 530–558 (2022).
Chen, R. Y. et al. A microbiota-directed food intervention for undernourished children. N. Engl. J. Med. 384, 1517–1528 (2021).
Gehrig, J. L. et al. Effects of microbiota-directed foods in gnotobiotic animals and undernourished children. Sci. (1979). 365, 12 (2019).
Hopkins, M. J., Sharp, R. & Macfarlane, G. T. Variation in human intestinal microbiota with age. Digestive Liver Dis. 34, S12–S18 (2002).
Poretsky, R., Rodriguez-R, L. M., Luo, C., Tsementzi, D. & Konstantinidis, K. T. Strengths and limitations of 16S rRNA gene amplicon sequencing in revealing temporal microbial community dynamics. PLoS ONE 9, e93827 (2014).
Methley, A. M., Campbell, S., Chew-Graham, C., McNally, R. & Cheraghi-Sohi, S. P. I. C. O. PICOS and SPIDER: a comparison study of specificity and sensitivity in three search tools for qualitative systematic reviews. BMC Health Serv. Res. 14, 579 (2014).
Liberati A. et al. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate healthcare interventions: explanation and elaboration. BMJ [Internet]. Available from: https://www.bmj.com/content/339/bmj.b2700 (2009).
Porritt, K., Gomersall, J. & Lockwood, C. JBI’s systematic reviews: study selection and critical appraisal. AJN Am. J. Nurs. 114, 47–52 (2014).
Acknowledgements
This work was supported by the Schlumberger Foundation’s flagship program, Faculty for the Future (FFTF), through a post-graduate scholarship awarded to Mwelwa Chibuye and The European and Developing Countries Clinical Trials Partnership (EDCTPII) through the ShigaPlexIm consortium (grant ID-RIA2018V-2308). We also acknowledge the Enterics and Vaccines Research (EDVRU) and Analysis (AU) Units at The Center for Infectious Disease Research in Zambia (CIDRZ). This research was additionally supported by The Netherlands Organization for Health Research and Development (ZonMw, zonmwl.nl) VENI09150161810022 The Netherlands Organization for Health Research and Development (ZonMw,zonmwl.nl) 10430022010019 to V.C.H., WellcomeTrust (wellcome.org) 219775/Z/19/Z to V.C.H., Health Holland TKI (https://www.health-holland.com) and GLORIA fund to V.C.H.
Author information
Authors and Affiliations
Contributions
Conceptualization, M.C., C.S., R.C., and V.C.H.; methodology, D.R.M., M.C., V.C.H., and R.S., M.S.; formal analysis, M.C., D.R.M.; writing original draft preparation, M.C., D.R.M., and V.C.H.; review and editing V.C.H., C.S., R.C., S.B., M.S., C.C.L., D.R.M. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Chibuye, M., Mende, D.R., Spijker, R. et al. Systematic review of associations between gut microbiome composition and stunting in under-five children. npj Biofilms Microbiomes 10, 46 (2024). https://doi.org/10.1038/s41522-024-00517-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41522-024-00517-5
- Springer Nature Limited