Abstract
Instream barriers can constrain dispersal of nonnative fishes, creating opportunities to test their impact on native communities above and below these barriers. Deposition of sediments in a river inflow to Lake Powell, USA resulted in creation of a large waterfall prohibiting upstream movement of fishes from the reservoir allowing us to evaluate the trophic niche of fishes above and below this barrier. We expected niche overlap among native and nonnative species would increase in local assemblages downstream of the barrier where nonnative fish diversity and abundance were higher. Fishes upstream of the barrier had more distinct isotopic niches and species exhibited a wider range in δ15N relative to downstream. In the reservoir, species were more constrained in δ15N and differed more in δ13C, representing a shorter, wider food web. Differences in energetic pathways and resource availability among habitats likely contributed to differences in isotopic niches. Endangered Razorback Sucker (Xyrauchen texanus) aggregate at some reservoir inflows in the Colorado River basin, and this is where we found the highest niche overlap among species. Whether isotopic niche overlap among adult native and nonnative species has negative consequences is unclear, because data on resource availability and use are lacking; however, these observations do indicate the potential for competition. Still, the impacts of diet overlap among trophic generalists, such as Razorback Sucker, are likely low, particularly in habitats with diverse and abundant food bases such as river-reservoir inflows.
Similar content being viewed by others
Introduction
Nonnative species have been widely introduced into freshwater habitats, presenting one of the most significant and persistent threats to native species1,2,3. Nonnative fishes, for example, can have wide ranging effects4, including altering energetic pathways and changing food web structure5,6. Gauging the impact of biological invasions is relatively straightforward for top predators that consume native species7, but assessing impacts occurring through indirect effects of competition are more difficult to identify. Evidence from stable isotope analysis suggests that native species often shift their trophic niche in the presence of nonnative species, presumably to less preferred, lower quality resources8,9,10. Competitive interactions between species with similar trophic niches occupying the same habitat might lead to exclusion unless species partition resources11,12,13, but direct evidence of competition in the wild is difficult to detect14,15. Still, food web studies can provide important information on potential species interactions and elucidate impacts of nonnative species on native assemblages10,15.
Fish assemblage and food web structure can change across habitat and trophic resource gradients that shift longitudinally in rivers16,17,18, but these gradients are often altered by river regulation19,20. Construction of dams and reservoirs have transformed riverscapes into a mosaic of lotic and lentic habitats21, frequently leading to changes in fish assemblage composition both downstream and upstream of dams22. Further, water development has resulted in discontinuities in resource gradients23,24,25, created new resource gradients21,26, and facilitated introduction and proliferation of nonnative species27. These alterations have changed natural habitat gradients (e.g., habitat templates28) and increased the number of potential competitors29,30,31. Habitat alterations have also led to reduction and homogenization of niche space resulting from decreased trophic diversity and shortening of food chains in some river systems32, while species introductions have caused lengthening of food chains in others9,33,34.
Our understanding of trophic resource use by some imperiled fishes and their overlap with nonnative fishes is limited. Predation and competition are the most often cited effects of nonnative species4,35,36, yet evidence of competition is often missing in research of freshwater systems, partially because of a lack of data on trophic resource use and availability. Moreover, because of their rarity and conservation status, it is often difficult to directly assess the diet of imperiled species37,38,39, limiting our understanding of impacts of nonnative species as resource competitors. Non-lethal and less intrusive techniques, such as stable isotope analysis, provide an alternative approach to compare overlap in trophic resource use of fishes9,10,40. For instance, δ13C reflects organic matter sources, such as autochthonous versus allochthonous primary production in streams41 or pelagic versus benthic primary production in lakes42, but the δ13C values of these sources can sometimes overlap43. Meanwhile, δ15N can provide an estimate of trophic position44,45, and several informative analytical metrics have been developed to quantify isotopic niche breadth (realized niche) and trophic overlap among species in an assemblage46,47,48.
Nonnative species introductions and habitat alteration are the leading hypothesized drivers for native fish imperilment in the Colorado River basin in the American Southwest35,49. Novel habitats in the basin, such as large water-storage reservoirs, serve as sources for nonnative species, but some native species also appear to be using river-reservoir inflow habitats50,51. Previous research identified overlap in resource use between younger life stages of native (e.g., Flannelmouth Sucker Catostomus latipinnis) and nonnative species (e.g., Common Carp Cyprinus carpio) in the upper Colorado basin9,52,53, but data on large-river, federally endangered species, such as Colorado Pikeminnow (Ptychocheilus lucius) and Razorback Sucker (Xyrauchen texanus) are more limited, likely because of their rarity in the system.
We characterized isotopic niches of species across a river-reservoir continuum in habitats differing in relative abundance of native and nonnative species. Deposition of sediments from the San Juan River at the inflow to Lake Powell on the Colorado River, USA and new channel formation following declines in reservoir water level resulted in the creation of a large waterfall that prohibits the upstream migration of fishes from the reservoir54,55. This created a rare opportunity to compare relative positions of species in trophic niche space in the river above this barrier, where the assemblage is dominated by native fishes, and in the river and reservoir below the barrier, where the assemblage is dominated by nonnative fishes. We had three objectives in this study. First, we identified differences in mean isotopic signatures of fishes within habitats, and expected species isotopic niches would reflect differences in resource use. Second, we quantified isotopic niche breadth of species within habitats, and predicted wider isotopic niche breadth for all species in the reservoir, because this habitat consists of areas with both detritus-based and algae-based food webs (e.g., reservoirs consist of lotic and lentic habitats26). Finally, we assessed niche overlap between pairs of native and nonnative fishes within habitats. We predicted trophic niche overlap between native and nonnative species would be greatest in the reservoir, because of the higher diversity and relative abundance of nonnative fish species (i.e., more potential competitors), and predicted overlap would decrease moving upstream into riverine habitats. We were primarily interested in assessing isotopic niche overlap among X. texanus and other benthic omnivores that might occupy a similar trophic position (e.g., fish from the same trophic guild are functionally similar56). We also included data on P. lucius because of its conservation status, and a lack of data on trophic resource use of this species in the wild53. Both species are the focus of conservation and recovery efforts and use riverine and reservoir inflow habitats in the basin50. Tissues from other nonnative species (fishes and quagga mussels Dreissena rostriformis bugensis) were collected, but in only one of the three habitats so we excluded these from analyses and present these data for reference in the Electronic Supplementary Material (Supplementary Table S1, Fig. S1).
Methods
Study area
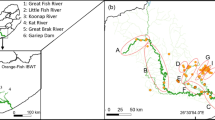
In 2018 and 2019, we sampled fishes and collected tissues for stable isotope analysis across 135 river kilometers (rkm) of the San Juan River, including the San Juan River arm of Lake Powell in southeastern Utah (Fig. 1). The San Juan River begins in the Rocky Mountains in southwestern Colorado and flows south and west across parts of Colorado, New Mexico, and Utah before its eventual confluence with the Colorado River, which is now inundated by Lake Powell. The historical confluence is approximately 30 rkm downstream of the lower extent of our study area. We sampled three habitats along the river-reservoir continuum (Fig. 1). The upstream habitat (hereafter river upstream; 67 rkm) occurred from Mexican Hat, Utah to approximately 15 rkm upstream of the Piute Farms Waterfall, a 6 m tall geomorphic barrier to upstream fish movement that has emerged on the San Juan River as a result of sediment deposition during higher water levels in Lake Powell, and subsequent superimposition processes through delta sediments55. This habitat was mainly canyon-bound and higher gradient, lotic habitat (mean gradient = 1.6 m km−1, measured from Google Earth). The middle habitat began directly below the Piute Farms Waterfall (hereafter river downstream) and included 21 rkm of the San Juan River and Lake Powell inflow. This habitat consisted of lower gradient (0.6 m km−1), slowing velocity, and turbid lotic habitats at the upper end of the reservoir (the riverine zone; sensu26). The last, and most downstream, habitat also occurred in the San Juan River–Lake Powell inflow across 15 rkm, and encompassed gradients of decreasing turbidity and increasing channel width and depth (here after reservoir)51.
Fish tissues for isotope analyses were collected over 135 river kilometers (rkm) from three habitats along the lower San Juan River and into Lake Powell in the Colorado River Basin, southeastern Utah, USA. Habitats were delineated in relation to the Piute Farms Waterfall (0 rkm) on the San Juan River and to the San Juan River inflow to Lake Powell. Colored bars represent the extent of sampling in each habitat. Reservoir habitat photograph by Douglas Turnquist, used with permission. River downstream and river upstream photograph by Casey Pennock.
Stable isotope tissue collection
We collected pectoral fin tissues from the distal end of the fin for isotope analysis from fishes captured in the river upstream and river downstream habitats in March (13–31), April (1–2), June (8–10), and September (22–28) in 2018 using raft electrofishing (ETS Electrofishing Systems, Madison, Wisconsin), and from reservoir fishes in April and May 2019 captured in trammel nets (45.2 m long × 1.2 m tall with 25 mm internal bar mesh and 305 mm outer bar mesh). Fin tissues typically have an isotopic half-life of several months for both δ13C and δ15N 57,58,59. In the river, we sampled fish in 0.1–3.2 km increments depending on fish densities to reduce handling stress on native fishes. We sampled fish in the reservoir by placing a trammel net in every 0.5 km segment of shoreline along approximately 20 km of the historic river channel51. The US Fish and Wildlife Service stocks PIT-tagged X. texanus every fall in the San Juan River, and therefore we removed individuals stocked the previous fall from analyses to avoid influence of residual hatchery diets on isotope signatures. All sampling was performed in accordance with the relevant guidelines and regulations, reviewed and approved by the Utah State University Institutional Animal Care and Use Committee Animal Care and Ethics Committee (#10,169), and permitting from the U.S. Fish and Wildlife Service (TE067729-6), Navajo Nation (Special Permit, Scientific Collecting 1187), and National Park Service (GLCA-2017-SCI-0003).
We either air-dried tissues or preserved them in table salt in the field. Following60, we prepared salt-preserved tissues by rinsing tissues with distilled water and soaking them in distilled water for 4 h. In the laboratory, we dried tissues (60 °C for ~ 8 h), homogenized tissues with a mortar and pestle, and packed samples into tin capsules, and weighed to the nearest 0.1 mg. We analyzed tissues for carbon and nitrogen stable isotope ratios. Tissues collected from the reservoir were analyzed at the Stable Isotope Mass Spectrometry Laboratory, Kansas State University, Manhattan, Kansas by continuous-flow, direct combustion, and mass spectrometry using an Elementar Vario Cube elemental analyzer coupled to an Elementar Vision mass spectrometer (Elementar Americas, Mt. Laurel, New Jersey). Tissues collected from the river upstream and river downstream habitats were analyzed at the Utah State University Department of Geology Stable Isotope Laboratory, using a Thermo Scientific Gasbench II or Costech ECS4010 elemental analyzer (Costech Analytical Technologies Inc., Valencia, California) coupled to a Thermo Scientific Delta V Advantage IRMS (Thermo Fisher Scientific, Walmath, Virginia). We report data on a per mille basis (‰) in delta (δ) notation. We calculated delta values as:
where R is equal to 13C/12C and 15N/14N. We used laboratory standards calibrated against international standards: Pee Dee Belemnite was the standard for carbon and atmospheric molecular nitrogen was the standard for nitrogen. Measurement error on routine analysis of laboratory standards was less than 0.50‰ for both δ13C and δ15N. For comparison, we also ran duplicate samples (n = 4) at both stable isotope laboratories and these differed on average by 1.4‰ (CV = 0.4) for δ13C and 0.58‰ (CV = 0.08) for δ15N.
Statistical analysis
Fish assemblage variation among habitats
To assess fish assemblage composition across our study area, we quantified variation in relative abundance of native species across our three habitats. We used species relative abundance because sampling methods differed between the river (raft electrofishing) and reservoir (trammel netting) habitats. We first transformed catch data into catch per unit effort (fish h−1), and then calculated relative abundance as the catch per unit effort of native species divided by the total catch per unit effort. Both capture techniques tend to target large-bodied fishes and are biased against smaller fishes61; therefore, we removed Red Shiner (Cyprinella lutrensis) and Western Mosquitofish (Gambusia affinis), two small-bodied species, from the dataset before calculating relative abundance. We tested for differences in native species relative abundance among habitats using a linear mixed effect model using the lme4 package62. We logit transformed proportional data prior to fitting the model, and included reach as a fixed factor and a random intercept of month nested within year. We used likelihood ratio tests with the car package63 to estimate statistical significance (α = 0.05).
Within reach isotopic variation
We tested for differences in species isotopic niche positions within habitats by separately comparing values of δ13C and δ15N of each species using linear mixed effects models. Statistical significance was assessed using likelihood ratio tests as described above. We used linear mixed effects models and included species as a fixed factor and fish length as a random effect. There was some variation among the seasonal representation of some species in the river upstream and river downstream habitats (Supplementary Table S2), but the majority of fishes were present across seasons. To account for temporal variation, we included a random effect of month of sample collection. However, the random effect of month had a variance of zero in the reservoir models and the random effect of fish length had a variance of zero in the river downstream models and the δ13C model in the river upstream. We used residual plots to check adherence to model assumptions and found no clear violations of normality or heteroskedasticity. We used Tukey’s HSD to make pairwise comparisons between species following a significant global model (α = 0.05). Finally, using the piecewiseSEM package64, we calculated variance explained by fixed factors using marginal R2 and fixed and random factors together using conditional R265.
Within reach isotopic niche breadth and overlap
We used standard ellipse area of δ13C and δ15N as a measure of isotopic niche breadth of each species in each reach. We calculated standard ellipse areas corrected for sample sizes (SEAc) using the SIBER package47. Standard ellipses represent the core trophic niche of a species and encompass roughly 40% of the samples in a group regardless of the sample size47. To estimate niche overlap between native and nonnative species pairs in each reach we used the nicheROVER package66. This analysis uses a Monte Carlo resampling routine (n = 10,000 draws) to randomly draw from the sampled population and calculates a mean and 95% credible interval of niche overlap probabilities. These values are directional in that they represent the estimated probability a randomly drawn individual of one species overlaps with the niche of another species in δ13C and δ15N bivariate space48. All analyses were conducted using the R statistical language version 3.5.367.
Results
Fish assemblage composition changed distinctly along our study area. As expected, nonnative species were more dominant towards the reservoir and relative abundance of native species declined from upstream to downstream (F2,112.87 = 21.37, P < 0.001; Fig. 2). Six nonnative species were only captured downstream of the waterfall in the river downstream and reservoir habitats, and most nonnative fish were in higher abundance downstream of the waterfall (Supplementary Fig. S1). For example, average catch per unit effort of C. carpio was 4× greater in the river downstream than the river upstream. The one exception was Channel Catfish (Ictalurus punctatus), which was 20× more abundant upstream of the waterfall relative to the river downstream in late March and early April.
Relative abundance of native species in fish assemblages across the San Juan River–Lake Powell continuum from the reservoir, to the river downstream, to the river upstream. Triangles represent marginal means from a linear mixed effects model including reach as a fixed factor and a random effect of month nested within year, the bold lines are medians, box edges are the 25th and 75th quartiles, and whiskers extend from box edges to the smallest and largest value no further than 1.5 × the interquartile range. Points represent 0.1–3.2 km river reaches sampled or individual trammel nets in the reservoir. A small amount of jitter was added to points to reduce overlap. The y-axis has been scaled with a square root transformation to better show small proportions.
Isotopic signatures indicated differences in trophic resource overlap across the river-reservoir continuum (Fig. 3). We collected tissues from 68 fishes in the river upstream, 108 in the river downstream, and 186 in the reservoir (Table 1). Means of δ13C were statistically different among some species within the river downstream and reservoir habitats (Table 2), but there were no statistical differences among species in the river upstream (P = 0.094). δ15N values, on average, differed significantly among some species in all habitats (Table 2). The fixed effect of species explained relatively little variation in δ13C in the river upstream and river downstream (marginal R2 = 0.09–0.16) compared to the reservoir (marginal R2 = 0.54). Conversely, the fixed effect of species contributed to a relatively low proportion of the explained variation in δ15N in the reservoir (marginal R2 = 0.26) compared to the river upstream (marginal R2 = 0.69) and river downstream (marginal R2 = 0.31; Table 2). In the reservoir, species were more constrained in δ15N, and only I. punctatus had a significantly different δ15N signature from other species based on post-hoc tests (Table 1). In sum, relative differences among species in the river upstream were driven more by differences in δ15N; in the reservoir differences were more pronounced in δ13C. Differences among species in the river downstream appeared somewhat intermediate between the river upstream and the reservoir (Fig. 3).
Isotopic niche breadth generally increased from the river upstream to the river downstream and the reservoir, somewhat matching our prediction that niche breadth would be wider in the Lake Powell inflow. Isotopic niche breadth for native and nonnative species varied over the three habitats (Table 1), and nonnative species showed relatively larger shifts compared to native species (Fig. 4). In the river upstream, both nonnative species exhibited relatively small isotopic niche breadths (2.24 and 3.69) based on SEAc, and the SEAc of native species (mean across species = 9.33) was 3× larger than nonnative species (Table 1). This pattern was not apparent in the river downstream of the waterfall, where SEAc of native species declined relative to the river upstream by 18% for X. texanus, 38% for C. latipinnis, and 55% for P. lucius. Conversely, SEAc of I. punctatus increased by 188% and C. carpio by 514% relative to the river upstream (Table 1). In the reservoir, SEAc of native C. latipinnis was 49% larger relative to the river upstream. The niche breadth of nonnative I. punctatus (130%) and C. carpio (325%) was still larger compared to the river upstream, but SEAc was 20% and 30% lower relative to the river downstream, respectively. The SEAc of Gizzard Shad (Dorosoma cepedianum) increased by 56% from the river downstream to the reservoir. Relative to all other species X. texanus had the most consistent niche breadth across habitats ranging from 7.96 in the river upstream to 6.46 in the river downstream to 7.39 in the reservoir (Table 1).
Isotope biplots from fish assemblages in the river upstream on the San Juan River, Utah (a), river downstream (b), and in Lake Powell reservoir (c). Ellipses are standard area ellipses corrected for samples size (SEAc; Jackson et al. 2011). Some species present in assemblages differ among panels (i.e., P. lucius and D. cepedianum). Filter-feeding Quagga mussels (Dreissena rostriformis bugensis) were only collected in the reservoir (dot represents the mean and lines are SE).
We predicted niche overlap would be highest in the reservoir, but similar to niche breadth niche overlap was actually highest in the river downstream. The probability of niche overlap between native fishes and other native and nonnative fishes varied among habitats, corresponding with increases in the number and relative abundances of nonnative species. Probability of niche overlap among species pairs tended to be highest in the river downstream (Table 3). For instance, the probability of C. latipinnis overlapping the niche of C. carpio and I. punctatus in the river upstream was ≤ 25% on average. In the river downstream, the probability of C. latipinnis overlapping the niche of those same two species was 88% on average (Table 3). We observed similar patterns for P. lucius and X. texanus, whereby the probability of overlap with other species was higher in the river downstream relative to the river upstream. In the reservoir, probabilities of overlap between native and nonnative species remained high relative to the river upstream and ranged from 30–68% and 41–83% for C. latipinnis and X. texanus, respectively. The highest overlap probabilities across all three habitats occurred between C. latipinnis and X. texanus in the reservoir (93%; Table 3).
Discussion
Variation in species overlap in isotopic space was apparent along the river-reservoir inflow gradient that coincided with changes in fish assemblage composition and likely variation in diversity of energetic pathways moving from lotic to lentic habitats. Along with increases in dominance of nonnative fishes, we observed increased isotopic niche overlap in the river and reservoir downstream of the waterfall compared to the river upstream where we observed lower nonnative fish relative abundance and occurrence. The Piute Farms Waterfall is a complete barrier to upstream fish movement55, and this coupled with nonnative fish removal efforts in the San Juan River has reduced abundance of some nonnative species such as C. carpio68 that likely move upstream from source populations in the reservoir. Compared to the river upstream, higher overlap in isotopic niche space among species in the river downstream of the waterfall and in the reservoir might indicate chances for competition are higher in these anthropogenic-derived habitats. However, this pattern might simply reflect that these habitats have higher diversity of nonnative fishes (i.e., more potential competitors).
We observed changes in the amount of isotopic overlap between species, and particularly, native and nonnative species among the three habitats. Overlap of species in isotopic niche space can be an indication of potential competition for food resources69,70,71. The two native suckers demonstrated the highest probability of overlap with other species across all habitats, likely due to a more generalist feeding strategy. In contrast, P. lucius, a presumed piscivore72, exhibited a relatively small probability of overlap with other species, but this was dependent on the habitat. Some nonnative species, such as I. punctatus, are hypothesized to limit populations of native fish in the Colorado River basin through predation73,74. Our results suggest relatively little overlap between P. lucius, the species with the highest δ15N in the river upstream and I. punctatus, and supports other observations of limited piscivory by I. punctatus in the San Juan River74. Although we observed more overlap among species in the two habitats downstream of the waterfall, we do not expect competition is a strong driver of assemblage dynamics as river-reservoir inflows are not likely resource-limited26,75,76. Because native fishes in the Colorado River basin tend to be trophic generalists77, they might be less susceptible to competitive exclusion by nonnative species despite overlap in isotopic niche space. Fishes in the Colorado River basin likely evolved to capitalize on resource availability that varied across space and time, including across lotic-lentic habitat gradients such as the Colorado River Delta78,79, expansive reaches of river impounded by lava dams80, and lentic habitats created by high water events81.
We only had data on baseline trophic levels from the reservoir, which limited our ability to isolate the cause of observed shifts in isotopic niches, which could be due to changes in baseline isotopic signatures, diet, or habitat use. The δ13C of filter-feeding quagga mussels in the reservoir was more depleted in samples collected farther in-reservoir (Supplementary Fig. S2), which we hypothesize is due to a shift in basal energy pathways with the food web influenced more by terrestrial inputs (e.g., detritus) in lotic habitat and more by autochthonous resources (e.g., phytoplankton) in lentic habitat. Lotic and lentic habitats differ in dominant energetic pathways of basal resources41,82,83, and it appears that, while some fishes had δ13C values that were similar in the two river habitats but markedly different from the reservoir (e.g., I. punctatus; Fig. 3), others had δ13C values that were similar in the reservoir and river downstream but generally different from the river upstream (e.g., X. texanus). Catostomus latipinnis was the only species that appeared to obtain resources from a mix of river and reservoir resources based on the intermediate δ13C values in the river downstream of the barrier. Some fish species could be partitioning habitat and garnering energy from more littoral or pelagic resources in the reservoir42. For instance, D. cepedianum and X. texanus might forage more in the littoral zone or shallower in the water column where food resources are less depleted84,85,86, which matches patterns observed in other food web and habitat use studies from Lake Powell87,88. The δ13C signature of X. texanus captured in the river downstream of the waterfall align more with that from the reservoir than the river upstream, which was not surprising since this species moves in the river for only a few weeks or months (M. Bogaard, unpublished data), spending most of their time in the reservoir87.
Many native fishes in the Colorado River Basin, as well as the nonnative species we assessed, are considered trophic generalists. All fishes, native and nonnative, appeared to be feeding on primary consumers and higher trophic levels (e.g., predatory macroinvertebrates, smaller fishes, zooplankton) rather than feeding directly on phytoplankton or benthic algae in the reservoir, assuming trophic fractionation of 3.4 ‰ in δ15N45. For instance, the δ15N of filter-feeding quagga mussels collected from the reservoir reach during our study had a δ15N of 6.3 ± 1.1 (mean ± SE) and much more variable δ13C (-33.1 ± 2.3). Previous research on isotopic niches of native fishes in the Colorado River basin have mainly used smaller-bodied individuals relative to those we used here (e.g.,53), limiting our ability to make direct comparisons of isotopic signatures. Whereas trophic position of fish tends to increase with size89; the random effect of fish length explained relatively little variation in our models. Nonetheless, our δ15N values for P. lucius (total length range: 222–526 mm), I. punctatus, and C. latipinnis are comparable to those reported previously from the San Juan River (range: 100–350 mm TL)90, although body sizes were not reported for the latter two species.
Niche breadth varied spatially among the three habitats for native and nonnative species, presumably as the major resource base shifted from allochthonous to autochthonous26,82,91. Somewhat surprisingly, on average, native fishes demonstrated consistent trophic niche breadth (based on SEAc) among the three habitats. All native fishes demonstrated substantially wider niches in the river upstream relative to nonnative fishes, and although the niche breadth of some nonnative species increased moving downstream towards the reservoir (i.e., C. carpio and I. punctatus), X. texanus maintained a relatively wide niche breadth across habitats. In all three habitats, niche breadth was generally influenced more by variation in δ13C among individuals within species (based on the ratio of SD of δ13C to SD of δ15N; most values > 1.4), potentially due to variation in basal resource (only measured in the reservoir) or habitat use. Only C. carpio in the river upstream and downstream and I. punctatus in the river downstream appeared to have niche breadth dually influenced by variation in δ13C and δ15N (ratio values = 0.8–1.1). Overall, species exhibited shifts towards more depleted carbon isotope sources moving from the river upstream (mean ± SD; − 22.5 ± 2.6) to the reservoir (− 27.5 ± 3.5), but the amount of overlap in isotopic niche space among species varied across habitats.
Human transformation of riverscapes has restructured fish assemblages with consequences for the conservation of native species. Artificial habitats, such as reservoirs and isolated river fragments, are now common92, and native and nonnative species co-occur in these habitats93, with nonnative species often thriving in more modified habitat51,94,95. In the Colorado River basin, native fish assemblages have endured habitat loss and degradation alongside the introduction and establishment of nonnative species, but recovery efforts are hindered by a full accounting of limiting factors, particularly the importance of biotic interactions. Although we observed greater niche overlap between native and nonnative fishes in habitats with a greater relative abundance of nonnative fishes (i.e., the river-reservoir inflow), it remains unclear whether this overlap has negative impacts on native populations. It is difficult to assess the importance of overlap between native fish and potential nonnative competitors because data on resource availability are lacking across the basin. This study adds to a growing body of research suggesting adult native fishes may be able to coexist with some nonnative fishes that also have an opportunistic feeding strategy. In addition, we demonstrate some native fishes might be able to successfully utilize highly modified river-reservoir inflow and reservoir habitat51,95,96, habitats similar to lotic-lentic habitats that have been present across their evolutionary history.
References
Dudgeon, D. et al. Freshwater biodiversity: importance, threats, status and conservation challenges. Biol. Rev. 81, 163–182. https://doi.org/10.1017/S1464793105006950 (2006).
Strayer, D. L. & Dudgeon, D. Freshwater biodiversity conservation: recent progress and future challenges. J. N. Am. Benthol. Soc. 29, 344–359. https://doi.org/10.1899/08-171.1 (2010).
Reid, A. J. et al. Emerging threats and persistent challenges for freshwater biodiversity. Biol. Rev. 94, 849–873. https://doi.org/10.1111/brv.12480 (2019).
Cucherousset, J. & Olden, J. D. Ecological impacts of nonnative freshwater fishes. Fisheries 36, 215–230. https://doi.org/10.1080/03632415.2011.574578 (2011).
Vander Zanden, M. J., Casselman, J. M. & Rasmussen, J. B. Stable isotope evidence for the food web consequences of species invasions in lakes. Nature 401, 464–467. https://doi.org/10.1038/46762 (1999).
Britton, J. R., Davies, G. D. & Harrod, C. Trophic interactions and consequent impacts of the invasive fish Psuedorasbora parva in a native aquatic food web: a field investigation in the UK. Biol. Invasions 12, 1533–1542. https://doi.org/10.1007/s10530-009-9566-5 (2010).
Cox, J. G. & Lima, S. L. Naiveté and an aquatic-terrestrial dichotomy in the effects of introduced predators. Trends Ecol. Evol. 21, 674–680. https://doi.org/10.1016/j.tree.2006.07.011 (2006).
Marks, J. C., Haden, G. A., O’Neil, M. & Pace, C. Effects of flow restoration and exotic species removal on recovery of native fish: Lessons from a dam decommissioning. Restor. Ecol. 18, 934–943. https://doi.org/10.1111/j.1526-100X.2009.00574.x (2010).
Walsworth, T. E., Budy, P. & Thiede, G. P. Longer food chains and crowded niche space: effects of multiple invaders on desert stream food web structure. Ecol. Freshw. Fish 22, 439–452. https://doi.org/10.1111/eff.12038 (2013).
Rogosch, J. S. & Olden, J. D. Invaders induce coordinated isotopic niche shifts in native fish species. Can. J. Fish. Aquat. Sci. 77, 1348–1358. https://doi.org/10.1139/cjfas-2019-0346 (2020).
Connell, J. H. The influence of interspecific competition and other factors on the distribution of the barnacle Chthamalus stellatus. Ecology 42, 710–723. https://doi.org/10.2307/1933500 (1961).
Zaret, T. M. & Rand, A. S. Competition in tropical stream fishes: Support for the competitive exclusion principle. Ecology 52, 336–342. https://doi.org/10.2307/1934593 (1971).
Britton, J. R., Ruiz-Navarro, A., Verreycken, H. & Amat-Trigo, F. Trophic consequences of introduced species: comparative impacts of increased interspecific versus intraspecific competitive interactions. Funct. Ecol. 32, 486–495. https://doi.org/10.1111/1365-2435.12978 (2018).
Connell, J. H. On the prevalence and relative importance of interspecific competition: evidence from field experiments. Am. Nat. 122, 661–696. https://doi.org/10.1086/284165 (1983).
David, P. et al. Impacts of invasive species on food webs: a review of empirical data. Adv. Ecol. Res. 56, 1–60. https://doi.org/10.1016/bs.aecr.2016.10.001 (2017).
Vannote, R. L., Wayne Minshall, G., Cummins, K. W., Sedell, J. R. & Cushing, C. E. The river continuum concept. Can. J. Fish. Aquat. Sci. 37, 130–137. https://doi.org/10.1139/f80-017 (1980).
Ibañez, C. et al. Convergence of temperate and tropical stream fish assemblages. Ecography 32, 658–670. https://doi.org/10.1111/j.1600-0587.2008.05591.x (2009).
Winemiller, K. O. et al. Stable isotope analysis reveals food web structure and watershed impacts along the fluvial gradient of a Mesoamerican coastal river. River Res. Appl. 27, 791–803. https://doi.org/10.1002/rra.1396 (2011).
Ward, J. V. & Stanford, J. A. The serial discontinuity concept: extending the model to floodplain rivers. River Res. Appl. 10, 159–168. https://doi.org/10.1002/rrr.3450100211 (1983).
Sabo, J. L. et al. Pulsed flows, tributary inputs and food-web structure in a highly regulated river. J. Appl. Ecol. 55, 1884–1895. https://doi.org/10.1111/1365-2664.13109 (2018).
Sabater, S. Alterations of the global water cycle and their effects on river structure, function and services. Freshw. Rev. 1, 75–89. https://doi.org/10.1608/FRH-1.1.5 (2008).
Arrantes, C. C., Fitzgerald, D. B., Hoeinghaus, D. J. & Winemiller, K. O. Impacts of hydroelectric dams on fishes and fisheries in tropical rivers through the lens of functional traits. Curr. Opin. Environ. Sustain. 37, 28–40. https://doi.org/10.1016/j.cosust.2019.04.009 (2019).
Cross, W. F. et al. Ecosystem ecology meets adaptive management: food web response to a controlled flood on the Colorado River, Glen Canyon. Ecol. Appl. 21, 2016–2033. https://doi.org/10.1890/10-1719.1 (2011).
Cross, W. F. et al. Food web dynamics in a large river discontinuum. Ecol. Monogr. 83, 311–337. https://doi.org/10.1890/12-1727.1 (2013).
Wellard Kelley, H. A. et al. Macroinvertebrate diets reflect tributary inputs and turbidity-driven changes in food availability in the Colorado River downstream of Glen Canyon Dam. Freshw. Sci. 32, 397–410. https://doi.org/10.1899/12-088.1 (2013).
Thornton, K. W., Kimmel, B. L. & Payne, F. E. Reservoir Limnology: Ecological Perspectives (John Wiley and Sons, 1990).
Havel, J. E., Lee, C. E. & Vander Zanden, J. M. Do reservoirs facilitate invasions into landscapes?. Bioscience 55, 518–525. https://doi.org/10.1641/0006-3568(2005)055[0518:DRFIIL]2.0.CO;2 (2005).
Southwood, T. R. E. Habitat, the templet for ecological strategies?. J. Anim. Ecol. 46, 337–365. https://doi.org/10.2307/3817 (1977).
Brook, B. W., Sodhi, N. S. & Bradshaw, C. J. A. Synergies among extinction drivers under global change. Trends Ecol. Evol. 23, 453–460. https://doi.org/10.1016/j.tree.2008.03.011 (2008).
Mercado-Silva, N., Helmus, M. R. & Vander Zanden, M. J. The effects of impoundment and non-native species on a river food web in Mexico’s central plateau. River Res. Appl. 25, 1090–1108. https://doi.org/10.1002/rra.1205 (2009).
Villéger, S., Blanchet, S., Beauchard, O., Oberdorff, T. & Brosse, S. Homogenization patterns of the world’s freshwater fish faunas. Proc. Natl. Acad. Sci. U. S. A. 108, 18003–18008. https://doi.org/10.1073/pnas.1107614108 (2011).
Delong, M. D., Thorp, J. H., Thoms, M. C. & McIntosh, L. M. Trophic niche dimensions of fish communities as a function of historical hydrological conditions in a Plains river. River Syst. 19, 177–187. https://doi.org/10.1127/1868-5749/2011/019-0036 (2011).
Pilger, T. J., Gido, K. B. & Propst, D. L. Diet and trophic niche overlap of native and nonnative fishes in the Gila River, USA: implications for native fish conservation. Ecol. Freshw. Fish 19, 300–321. https://doi.org/10.1111/j.1600-0633.2010.00415.x (2010).
Mor, J. R. et al. Dam regulation and riverine food-web structure in a Mediterranean river. Sci. Total Environ. 625, 301–310. https://doi.org/10.1016/j.scitotenv.2017.12.296 (2018).
Tyus, H. M. & Saunders, J. F. III. Nonnative fish control and endangered fish recovery: lessons from the Colorado River. Fisheries 25, 17–24. https://doi.org/10.1577/1548-8446(2000)025%3c0017:NFCAEF%3e2.0.CO;2 (2000).
Strayer, D. L. Alien species in fresh waters: ecological effects, interactions with other stressors, and prospects for the future. Freshw. Biol. 55, 152–174. https://doi.org/10.1111/j.1365-2427.2009.02380.x (2010).
Marks, J. C., Williamson, C. & Hendrickson, D. A. Coupling stable isotope studies with food web manipulations to predict the effects of exotic fish: lessons from Cuatro Ciénegas, Mexico. Aquat. Conserv. 21, 317–323. https://doi.org/10.1002/aqc.1199 (2011).
Cooke, S. J., Paukert, C. & Hogan, Z. Endangered river fish: factors hindering conservation and restoration. Endanger. Species Res. 17, 179–191. https://doi.org/10.3354/esr00426 (2012).
Pennock, C. A., Farrington, M. A. & Gido, K. B. Feeding ecology of early life stage Razorback Sucker relative to other sucker species in the San Juan River. Trans. Am. Fish. Soc. 148, 938–951. https://doi.org/10.1002/tafs.10188 (2019).
Cucherousset, J., Bouletreau, S., Martino, A., Roussel, J. M. & Santoul, F. Using stable isotope analyses to determine the ecological effects of non-native fishes. Fish. Mgmt. Ecol. 19, 111–119. https://doi.org/10.1111/j.1365-2400.2011.00824.x (2012).
Finlay, J. C. Stable-carbon-isotope ratios of river biota: Implications for energy flow in lotic food webs. Ecology 82, 1052–1064. https://doi.org/10.1890/0012-9658(2001)082[1052:SCIROR]2.0.CO;2 (2001).
France, R. L. Differentiation between littoral and pelagic food webs in lakes using stable carbon isotopes. Limnol. Oceanogr. 40, 1310–1313. https://doi.org/10.4319/lo.1995.40.7.1310 (1995).
Fry, B. Stable Isotope Ecology (Springer-Verlag, 2006).
Vander Zanden, M. J., Cabana, G. & Rasmussen, J. B. Comparing trophic position of freshwater fish calculated using stable nitrogen isotope ratios (δ15N) and literature dietary data. Can. J. Fish. Aquat. Sci. 54, 1142–1158. https://doi.org/10.1139/f97-016 (1997).
Post, D. M. Using stable isotopes to estimate trophic position: Models, methods, and assumptions. Ecology 83, 703–718. https://doi.org/10.1890/0012-9658(2002)083[0703:USITET]2.0.CO;2 (2002).
Layman, C. A., Arrington, D. A., Montaña, C. G. & Post, D. M. Can stable isotope ratios provide for community-wide measures of trophic structure?. Ecology 88, 42–48. https://doi.org/10.1890/0012-9658(2007)88[42:CSIRPF]2.0.CO;2 (2007).
Jackson, A. L., Inger, R., Parnell, A. C. & Bearhop, S. Comparing isotopic niche widths among and within communities: SIBER: stable Isotope Bayesian Ellipses in R. J. Anim. Ecol. 80, 595–602. https://doi.org/10.1111/j.1365-2656.2011.01806.x (2011).
Swanson, H. K. et al. A new probabilistic method for quantifying n-dimensional ecological niches and niche overlap. Ecology 96, 318–324. https://doi.org/10.1890/14-0235.1 (2015).
Minckley, W. L. & Deacon, J. E. Battle Against Extinction: Native Fish Management in the American West (The University of Arizona Press, 1991).
Albrecht, B. A. et al. Use of inflow areas in two Colorado River basin reservoirs by the endangered Razorback Sucker (Xyrauchen texanus). West. N. Am. Nat. 77, 500–514. https://doi.org/10.3398/064.077.0410 (2018).
Pennock, C. A. et al. Reservoir fish assemblage structure across an aquatic ecotone: Can river-reservoir interfaces provide conservation and management opportunities?. Fish. Manag. Ecol. 28, 1–13. https://doi.org/10.1111/fme.12444 (2021).
Gido, K. B. & Propst, D. L. Habitat use and association of native and nonnative fishes in the San Juan River, New Mexico and Utah. Copeia 1999, 321–332. https://doi.org/10.2307/1447478 (1999).
Gido, K. B., Franssen, N. R. & Propst, D. L. Spatial variation in δ15N and δ13C isotopes in the San Juan River, New Mexico and Utah: implications for the conservation of native fishes. Environ. Biol. Fish. 75, 197–207. https://doi.org/10.1007/s10641-006-0009-1 (2006).
Ryden, D. W. & Ahlm, L. A. Observations on the distribution and movements of Colorado Squawfish, Ptychocheilus lucius, in the San Juan River, New Mexico, Colorado, and Utah. Southwest. Nat. 41, 161–168 (1996).
Cathcart, C. N. et al. Waterfall formation at a desert river-reservoir delta isolates endangered fishes. River Res. Appl. 34, 948–956. https://doi.org/10.1002/rra.3341 (2018).
Thomsen, M. S. et al. Impacts of marine invaders on biodiversity depend on trophic position and functional similarity. Mar. Ecol. Prog. Ser. 495, 39–47. https://doi.org/10.3354/meps10566 (2014).
McIntyre, P. B. & Flecker, A. S. Rapid turnover of tissue nitrogen of primary consumers in tropical freshwaters. Oecologia 148, 12–21. https://doi.org/10.1007/s00442-005-0354-3 (2006).
Franssen, N. R., Gilbert, E. I., James, A. P. & Davis, J. E. Isotopic tissue turnover and discrimination factors following a laboratory diet switch in Colorado Pikeminnow (Ptychocheilus lucius). Can. J. Fish. Aq. Sci. 74, 265–272. https://doi.org/10.1139/cjfas-2015-0531 (2017).
Busst, G. M. A. & Britton, J. R. Tissue-specific turnover rates of the nitrogen stable isotope as functions of time and growth in a cyprinid fish. Hydrobiologia 805, 49–60. https://doi.org/10.1007/s10750-017-3276-2 (2018).
Arrington, D. A. & Winemiller, K. O. Preservation effects on stable isotope analysis of fish muscle. Trans. Am. Fish. Soc. 131, 337–342. https://doi.org/10.1577/1548-8659(2002)131%3c0337:PEOSIA%3e2.0.CO;2 (2002).
Hubert, W. A., Pope, K. L. & Dettmers, J. M. Passive capture techniques. In Fisheries Techniques 3rd edn (eds Zale, A. V. et al.) 223–265 (American Fisheries Society, 2012).
Bates, D., Maechler, M., Bolker, B. & Walker, S. Fitting linear mixed effects models using lme4. J. Stat. Softw. 67, 1–48. https://doi.org/10.18637/jss.v067.i01 (2015).
Fox, J., & Weisberg, S. An {R} Companion to Applied Regression, 2nd edn. (Sage 2011). http://socserv.socci.mcmaster.ca/jfox/Books/Companion
Lefcheck, S. piecewiseSEM: Piecewise structural equation modeling in R for ecology, evolution, and systematics. Methods Ecol. Evo. 7, 573–579. https://doi.org/10.1111/2041-210X.12512 (2016).
Nakagawa, S., Johnson, P. C. D. & Schielzeth, H. The coefficient of determination R2 and intra-class correlation coefficient from generalized linear mixed-effects models revisited and expanded. J. R. Soc. Interface 14, 20170213. https://doi.org/10.1098/rsif.2017.0213 (2017).
Lysy, M., Stasko, A. D., Swanson, H. K. nicheROVER: (Niche) (R)egion and Niche (Over)lap metrics for multidimensional ecological niches. R package version 1.0 (2014). https://CRAN.R-project.org/package=nicheROVER
R Core Team. R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna (2019). Available: https://www.R-project.org/
Franssen, N. R., Davis, J. E., Ryden, D. W. & Gido, K. B. Fish community responses to mechanical removal of nonnative fishes in a large southwestern river. Fisheries 8, 352–363. https://doi.org/10.1080/03632415.2014.924409 (2014).
Kelly, D. J. & Jellyman, D. J. Changes in trophic linkages to shortfin eels (Anguilla australis) since the collapse of submerged macrophytes in Lake Ellesmere, New Zealand. Hydrobiologia 579, 161–173. https://doi.org/10.1007/s10750-006-0400-0 (2007).
Zambrano, L., Valiente, E. & Vander Zanden, M. J. food web overlap among native axolotl (Ambystoma mexicanum) and two exotic fishes: carp (Cyprinus carpio) and tilapia (Oreochromis niloticus) in Xochimilco, Mexico City. Biol. Invasions 12, 3061–3069. https://doi.org/10.1007/s10530-010-9697-8 (2010).
Córdova-Tapia, F., Contreras, M. & Zambrano, L. Trophic niche overlap between native and non-native fishes. Hydrobiologia 746, 291–301. https://doi.org/10.1007/s10750-014-1944-z (2015).
Portz, D. E. & Tyus, H. M. Fish humps in two Colorado River fishes: a morphological response to cyprinid predation?. Environ. Biol. Fishes 71, 233–245. https://doi.org/10.1007/s10641-004-0300-y (2004).
Pennock, C. A. et al. Predicted and observed responses of a nonnative Channel Catfish population following managed removal to aid the recovery of endangered fishes. N. Am. J. Fish. Mgmt. 38, 565–578. https://doi.org/10.1002/nafm.10056 (2018).
Hedden, S. C. et al. Quantifying consumption of native fishes by nonnative Channel Catfish in a desert river. N. Am. J. Fish. Manag. https://doi.org/10.1002/nafm.10514 (2020).
Nogueira, M. G., Oliveira, P. C. R. & Britto, Y. T. Zooplankton assemblages (Copepoda and Cladocera) in a cascade of reservoirs of a large tropical river (SE Brazil). Limnetica 27, 151–170 (2008).
Slaveska-Stamenković, V. et al. Factors affecting distribution pattern of dominant macroinvertebrates in Mantovo Reservoir (Republic of Macedonia). Biologia 67, 1129–1142. https://doi.org/10.2478/s11756-012-0102-1 (2012).
Behn, K. E. & Baxter, C. V. The trophic ecology of a desert river fish assemblage: influence of season and hydrologic variability. Ecosphere 10, e02583. https://doi.org/10.1002/ecs2.2583 (2019).
Glenn, E. P., Lee, C., Felger, R. & Zengel, S. Effects of water management on the wetlands of the Colorado River Delta, Mexico. Conserv. Biol. 10, 1175–1186. https://doi.org/10.1046/j.1523-1739.1996.10041175.x (1996).
Sykes, G. The Colorado River Delta. Publication no. 460. (Carnegie Institution of Washington, D.C. 1937).
Dalrymple, G. B. & Hamblin, W. K. K-Ar of Pleistocene lava dams in the Grand Canyon in Arizona. Proc. Natl. Acad. Sci. U.S.A. 95, 9744–9749. https://doi.org/10.1073/pnas.95.17.9744 (1998).
Minckley, W. L. Status of the razorback sucker, Xyrauchen texanus (Abbott), in the Lower Colorado River Basin. Southwest. Nat. 28, 165–187. https://doi.org/10.2307/3671385 (1983).
Doi, H. Spatial patterns of autochthonous and allochthonous resources in aquatic food webs. Popul. Ecol. 51, 57–64. https://doi.org/10.1007/s10144-008-0127-z (2009).
Thorp, J. H. & Delong, M. D. Dominance of autochthonous autotrophic carbon in food webs of heterotrophic rivers. Oikos 96, 543–550. https://doi.org/10.1034/j.1600-0706.2002.960315.x (2002).
Rennie, M. D., Sprules, W. G. & Johnson, T. B. Resource switching in fish following a major food web disruption. Oecologia 159, 789–802. https://doi.org/10.1007/s00442-008-1271-z (2009).
Cummings, B. M. & Schindler, D. E. Depth variation in isotopic composition of benthic resources and assessment of sculpin feeding patterns in an oligotrophic Alaskan lake. Aquat. Ecol. 47, 403–414. https://doi.org/10.1007/s10452-013-9453-0 (2013).
Fera, S. A., Rennie, M. D. & Dunlop, E. S. Broad shifts in the resource use of a commercially harvested fish following the invasion of dreissenid mussels. Ecology 98, 1681–1692. https://doi.org/10.1002/ecy.1836 (2017).
Pennock, C. A., McKinstry, M. C. & Gido, K. B. Razorback Sucker movement strategies across a river-reservoir habitat complex. Trans. Am. Fish. Soc. 149, 620–634. https://doi.org/10.1002/tafs.10262 (2020).
Vatland, S. & Budy, P. Predicting the invasion success of an introduced omnivore in a large heterogeneous reservoir. Can. J. Fish. Aquat. Sci. 64, 1329–1345. https://doi.org/10.1139/f07-100 (2007).
Romanuk, T. N., Hayward, A. & Hutchings, J. A. Trophic level scales positively with body size in fishes. Glob. Ecol. Biogeogr. 20, 231–240. https://doi.org/10.1111/j.1466-8238.2010.00579.x (2011).
Franssen, N. R., Gilbert, E. I., Gido, K. B. & Propst, D. L. Hatchery-reared endangered Colorado pikeminnow (Ptychocheilus lucius) undergo a gradual transition to piscivory after introduction to the wild. Aquat. Conserv. 29, 24–38. https://doi.org/10.1002/aqc.2995 (2019).
Hoeinghaus, D. J., Winemiller, K. O. & Agostinho, A. A. Hydrogeomorphology and river impoundment affect food-chain length of divers Neotropical food webs. Oikos 117, 984–995. https://doi.org/10.1111/j.2008.0030-1299.16458.x (2008).
Grill, G. et al. Mapping the world’s free-flowing rivers. Nature 569, 215–221. https://doi.org/10.1038/s41586-019-1111-9 (2019).
Pennock, C. A. & Gido, K. B. Spatial and temporal dynamics of fish assemblages in a desert reservoir over 38 years. Hyrdobiologia 848, 1231–1248. https://doi.org/10.1007/s10750-021-04514-z (2021).
Oliveira, E. F., Minte-Vera, C. V. & Goulart, E. Structure of fish assemblages along spatial gradients in a deep subtropical reservoir (Itaipu Reservoir, Brazil-Paraguay border). Environ. Biol. Fish. 72, 283–304. https://doi.org/10.1007/s10641-004-2582-5 (2005).
Buckmeier, D. L., Smith, N. G., Fleming, B. P. & Bodine, K. A. Intra-annual variation in river-reservoir interface fish assemblages: implications for fish conservation and management in regulated rivers. River Res. Appl. 30, 780–790. https://doi.org/10.1002/rra.2667 (2014).
Albrecht, B. A., Holden, P. B., Kegerries, R. B. & Golden, M. E. Razorback sucker recruitment in Lake Mead, Nevada-Arizona, why here?. Lake Reserv. Manage. 26, 336–344. https://doi.org/10.1080/07438141.2010.511966 (2010).
Acknowledgements
This work was funded by the U.S. Bureau of Reclamation through a variety of contracts, grants, and cooperative agreements to the Upper Colorado and San Juan River Basin recovery programs. We thank B. Hines, M. Bogaard, S. Brockdorf, R. McGrew, K. Burke, M. Akland, S. Wood, N. Zabloski, C. Michaud, K. Creighton (UDWR), C. Hedden, S. Hedden, L. Bruckerhoff, G. Hopper, L. Renner (KSU), P. MacKinnon, T. Arnold, and G. Thiede (USU) for assistance in the field and with logistics. We also thank J. Nippert (KSU) and T. Larkin (USU) for help with isotope analysis. Discussion with and comments from L. Bruckerhoff, N. Franssen, and three anonymous reviewers improved the manuscript. This work was conducted under the auspices of USU IACUC #10169 and permits provided by the National Park Service (Permit No. GLCA-2017-SCI-0003), The Navajo Nation (Special Permit, Scientific Collecting 1187), and U.S. Fish and Wildlife Service (TE067729-6). The data that support the findings of this study are available from the corresponding author upon reasonable request. Any use of trade, firm, or product names is for descriptive purposes only and does not imply endorsement by the U.S. Government. The study was performed in accordance with ARRIVE guidelines.
Author information
Authors and Affiliations
Contributions
C.A.P., Z.A., M.C.M., P.B., and K.B.G. designed the study and collected the data. C.A.P. and Z.A carried out laboratory work. C.A.P. performed the analyses and drafted the manuscript with input from Z.A., M.C.M., P.B., and K.B.G.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Pennock, C.A., Ahrens, Z.T., McKinstry, M.C. et al. Trophic niches of native and nonnative fishes along a river-reservoir continuum. Sci Rep 11, 12140 (2021). https://doi.org/10.1038/s41598-021-91730-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-021-91730-1
- Springer Nature Limited
This article is cited by
-
Trophic niche partitioning among non-native fish species coexisting in a Colombian high Andean reservoir
Biological Invasions (2024)
-
Isotopic diversity and niche patterns reveal contrasting resource use among co-occurring non-native fishes within a flow-altered African river system
Biological Invasions (2024)
-
Exploring fish assemblage structure, feeding guild, and water quality in a typical river-reservoir interface of tropical large reservoir environment, Central India
Environmental Science and Pollution Research (2023)
-
Determining resource intake of a nonnative fish highlights potential predatory and competitive interactions
Biological Invasions (2022)