Abstract
Strategies aiming to improve the longevity of resin–dentin adhesive interface developed so far have only been able to retard the problem. Different approaches are thus needed. The objective of this review was to determine whether the use of collagen-depletion strategies after acid-etching procedures may improve the bond strength of resin-based materials to dentin. A systematic review was planned following 2021 PRISMA statement guidelines, with a search strategy performed in five electronic databases: PubMed/Medline, Scopus, EMBASE, SciELO and IADR Abstract Archive (last search: 17/01/2022). Inclusion criteria encompassed studies which evaluated a collagen-depletion strategy in acid-etched human dentin and tensile/shear bond strength tests. Risk of bias assessment was carried out by two reviewers, working independently on an adapted five-domain risk of bias (RoB) checklist for laboratory studies. Results were synthesized qualitatively, as a meta-analysis was not possible due to limited number of studies and their RoB. A total of eight studies were eligible for inclusion in the systematic review after inclusion/exclusion criteria application. Out of these, two evaluated the effect of using NaOCl followed by an antioxidant, and the remaining six evaluated different enzymatic treatments (bromelain, chondroitinase ABC, papain, and trypsin). None of the studies reported a decrease of bond strength when a collagen-depletion strategy was used, in comparison to traditional hybrid layers (control). All enzymatic treatment studies which respected the inclusion criteria improved the bond strength to dentin. Some specific collagen-depletion strategies seem to play a favorable role in improving immediate bond strengths to dentin. Further research with sound methodology is required to consolidate these findings, since limitations in RoB and a low number of studies were found. The assessment of further proteolytic agents and long-term outcomes is also required.
Similar content being viewed by others
Introduction
Compositional differences between enamel and dentin, such as the absence of a highly sensitive and degradable organic matrix, are responsible for distinct predictability in restorative success1,2. In the case of dentin, adhesive procedures rely on the formation of a hybrid layer (HL), a dogma in adhesive dentistry first coined by Nakabayashi in 1982, known today to most important mechanism to secure micromechanical retention in such a substrate2,3,4. While in enamel a simplified and resistant bonding mechanism is tangible, considering that resin monomers are easily drawn into deep mineralized pits and pores, formed through acid etching5,6, in dentin, the HL formed within an intricate and complex collagen has an inevitable expiry-date stamp. The inconvenient truth is that virtually all HLs are bound to fail with time.
Indeed, it has been already argued that the foundation stone on which adhesion to dentin was set, is flawed7. On the nanoscale, complete envelopment of collagen fibrils by monomers, closing and filling in all spaces, is practically unattainable8. Furthermore, to even have a chance in penetrating the interfibrillar spaces of the collagen network, methacrylic acid-based monomers of relative hydrophilicity and low-viscosity must be used; this condemns them to future hydrolysis9,10. Thus, to summarize, the existence of exposed collagen, unfilled spaces and water inherently present within dentinal collagen, all either or simultaneously lead to hydrolysis and endogenous enzymatic activity, ultimately responsible for interface degradation2,7,11. These reasons led Bertassoni et al. to point out that the resin-dentin interface, from a molecular perspective, is the antithesis of successful bonding7.
While most recent research focuses on dealing with this problem by modifying the collagen12,13,14, developing anti-enzymatic strategies, to be used as pre-treatments or as functional components included in dental adhesives15,16,17, or even formulate degradation-resistant polymers18,19, it is key to understand that these strategies offer a one-sided solution to a multifactorial problem. While these strategies may improve the outcomes short-term, long-term, either the enzymatic or hydrolytic degradation pathway will inevitably occur, if the organic content remains5. Hence, a different approach may be needed.
Several researchers in the late 1990s and early 2000s contested the need for having collagen within the HL at all20,21. This would possibly solve the problems mentioned above. In fact, a considerable number of studies found that collagen-depletion by virtue of deproteinizing agents such as sodium hypochlorite (NaOCl) could increase the bond strength or improve interfacial properties of adhesives to dentin20,21,22,23. NaOCl, as reported by these authors, was able to dissolve most of the organic portion, leaving a mineral-rich layer easily infiltrated by resin monomers. However, a number of studies also found that these results were adhesive-dependent, while arguing that the collagen is indispensable in the HL24,25. Consequently, collagen-depletion strategies remained unpopular. At the time, attention was not given to the fact that NaOCl is an oxidizing agent, directly interfering with free-radical addition polymerization, via since the oxygen species it produces can inhibit free radical activity, so inducing an important reduction of bond strengths26,27. Furthermore, retention of NaOCl within demineralized dentin affects the resin-dentin interface25. This means that the negative results attributed with the removal of demineralized collagen fibrils could have been related to the detrimental effects of the agent used for that effect, rather than to not having collagen in the HL. Such results could have contributed to the unpopularity of the technique.
Interestingly, recent research has focused again on collagen-depletion strategies28,29,30. Indeed, other deproteinizing agents have been explored for the same end, namely enzymatic pre-treatments (i.e., trypsin, bromelain) to breakdown the collagen matrix or non-collagenous proteins in the extracellular matrix (ECM), such as proteoglycans (PGs) glycosaminoglycans (GAGs)30,31,32. These novel approaches have found positive and promising results, which may lead us to the verge of a new bonding approach to dentin. However, a qualitative and quantitative synthesis of preliminary studies whose objective is removing the collagen to alter hybridization, is still lacking. Considering the research that has been published in the matter, the objective of this systematic review was thus to investigate whether a correct collagen-depletion strategy, after acid-etching, could improve the bond strength of resin-based materials to dentin. Considering a systematic review typology, the null hypothesis would be that, based on the body of evidence available, there are no differences in the bond strength of adhesive materials to dentin, when collagen-depletion was carried out after acid-etching, compared to a conventional hybrid layer.
Materials and methods
Systematic search
The present systematic review (SR) was carried out following the latest PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analysis) statement guidelines and flowcharts. The protocol associated to this systematic review was registered using the international prospective register of systematic reviews (PROSPERO CRD42022303858). The databases used for the electronic search were PubMed/Medline, Scopus, EMBASE and SciELO and the search was performed by two reviewers, working independently. The IADR Abstract Archive was additionally searched for potentially relevant studies using a simple keyword search strategy. For systematic keyword search using the databases above, both MeSH terms and free keywords were used. The combinations used to search the different databases are shown in Table 1.
Following initial database search, and after paper screening, each primary study included was manually searched for additional relevant papers in the reference list. An online literature visualization tool (https://www.connectedpapers.com/) was also used to identify additional relevant studies not retrieved in the database search. The search period of this SR was between December 2021 and January 2022, with the last search being conducted on the 17 January 2022. No language or publication date restrictions were applied to the systematic search.
Review question, PICO and inclusion/exclusion criteria
This SR aims to answer the following general question: “Do collagen depletion strategies improve the bond strength of resin-dentin interfaces?”, which subdivides into specific questions, such as: (1) Is the use of sodium hypochlorite succeeded by an antioxidant, after acid-etching, effective in improving the bond strength of resin-dentin interfaces?; (2) are the use of enzymatic pre-treatments, after acid-etching, effective in improving the bond strength of resin-dentin interfaces?
A PICO strategy format was taken in consideration to organize the inclusion and exclusion criteria and to define the research question. PICO was defined as follows: P—permanent human posterior teeth; I—adhesive restorations, performed in dentin, which used a deproteinizing pre-treatment strategy, after acid etching, to remove surface collagen; C—traditional adhesive restorations relying on conventional hybrid layer formation; O—immediate bond strength, usually at 24 h (tensile or shear). The criteria used for inclusion and exclusion of studies was pre-determined and approved by all review members and is summarized in Table 2.
Studies were considered ineligible if they did not perform deproteinization after acid-etching, since the scope of this review comprises the removal of the collagen network exposed after acid-etching. Pre-etching strategies generally relate to smear layer deproteinization which is not the present aim. Moreover, regarding the studies which used sodium hypochlorite (NaOCl), it was decided to include only studies that used an antioxidant after application of NaOCl, to counter the oxidizing effect of hypochlorite. Antioxidants such as sodium ascorbate or grape seed extract are able to neutralize reactive species formed through the oxidizing effect of NaOCl, when in contact with dental hard tissues33,34. Without the use of an antioxidant, the results of the studies cannot be accurately trusted, as poor bond strength results may be attributed to the oxidizing effect, which affects the polymerization of the resin materials placed after, rather than to the strategy itself. Thus, to have reliable pooled results in the present study, references that did not use an antioxidant following NaOCl application were thus excluded.
Data collection and data items extraction
In the beginning, after the systematic search, and following the PRISMA flowchart recommendations, references were exported to a reference organization software (Mendeley Desktop for Mac v. 1.19.8), in which title and abstracts were subject to comprehensive screening by two review members, working independently (A.D. and M.B.C.). Duplicate removal was also performed and registered before screening. To resolve disagreements, the opinion of a third reviewer (A.M.A.) was sought and consensus was reached. Further screening and eligibility followed the pre-determined inclusion and exclusion criteria stringently. Reasons for study exclusion were also documented. When full texts could not be accessed, researchers were contacted via a digital platform (www.researchgate.net) or e-mail.
After final selection of references and their inclusion for the systematic review, quantitative and qualitative data were extracted from each record to a Microsoft Excel (v. 16.16; Microsoft, Redmond, WA, USA) spreadsheet containing a form previously made by the two assessors (A.D. and M.B.C.) and approved by all the review team members. Qualitative data extracted for the systematic review included authors, publication date, country, intervention, experimental groups, sample size, materials used, shear or microtensile bond strength outcome time-points, type of aging conducted and main conclusions. As for quantitative data, used in the meta-analysis, all microtensile bond strength results were gathered in the form of means, standard deviation at all time-points and also corresponding statistical inference tests for multi-comparisons. Data was collected independently, by both reviewers (A.D. and M.B.C.), and cross-checked by an additional reviewer (S.S.).
Quality assessment (risk of bias)
To assess the risk of bias (RoB) of the primary studies included in this review, and since no pre-specified tool exists for in vitro studies, let alone for dental studies, an in vitro evaluation scale was adapted as similarly done by previous authors35,36, but organized by domains and including relevant sources of bias for dental materials studies. Sources of bias were grouped in 4 different domains: Domain 1—bias in planning and allocation; Domain 2—bias in specimen preparation; Domain 3—bias in outcome assessment and Domain 4—bias in data treatment and reporting. The risk of bias was individually measured by two reviewers (A.D. and M.B.C.) and confirmed by a third member of the team (A.M.A.). Once again, disagreements were resolved by reaching consensus among the three assessors. A table summarizing the RoB results was made and the RobVis web visualization tool (www.riskofbias.info/welcome/robvis-visualization-tool) was used to build output figures. Grading of cumulative evidence was not performed as a meta-analysis was not possible considering the studies that were included.
Data analysis
Meta-analytical analyses were not carried out due to the reduced number of studies found for each deproteinization strategy retrieved in the systematic search (NaOCl application or enzymatic agents) and their methodological weaknesses. Due to this, qualitative synthesis was solely adopted in this systematic review.
Results
Study selection and summary of studies—SR
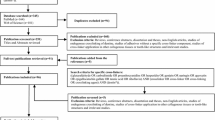
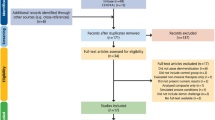
Subsequent to a careful screening of all retrieved references from the systematic search, 46 papers were short-listed for full-text reading. Out of these, 8 studies respected the inclusion criteria and were included in the systematic review30,37,38,39,40,41,42. The PRISMA workflow considered in this SR is depicted in Fig. 1. One additional paper was found through manual search, which was also included in the SR. The remaining excluded papers either applied no antioxidant after using NaOCl or used the deproteinizing strategy before acid-etching (smear layer deproteinization). In one study no bond strength test was performed32. The characteristics of these studies are summarized in Table 3.
Six studies dealt with enzymatic treatments, such as the application of trypsin and/or chondroitinase ABC, to remove PGs and GAGs, or bromelain as a collagenase30,38,39,40,41,42. Two studies involved the application of varying concentrations of NaOCl (4.25, 5.25 or 10%) followed by the application of an antioxidant. It is important to highlight that not a single study reported a decrease in bond strength, when a deproteinizing technique was used. Deproteinizing strategy application times varied from 1 min (NaOCl) up to 48 h (trypsin).
RoB analysis of the studies
A RoB analysis of the 8 studies shown above is presented in Table 4. An illustrative image of the RoB overall outcome, considering all sources of bias for all studies, can be seen in Fig. 2. Overall, studies included in this SR were classified as having a moderate to high risk of bias. Sources of bias such as sample size calculations (D1) and blinding of the testing operator (D3) were not reported in any study. Other important sources of bias, such as identical experimental conditions and standardization of samples and materials (D2) were only sufficiently reported in 50% and 25% of the studies, respectively.
Excluded studies using NaOCl without an antioxidant
To map the retrieved studies (26) where NaOCl was used as a deproteinizing agent after acid etching, but without a subsequent application of an antioxidant, a table summary with their highlights was constructed and shown (Table 5). Such a table can confirm the conflicting results reported in the literature during the last 20 years, since it is possible to observe that an improvement in bond strengths following the use of NaOCl varied upon concentration, application time, the adhesive type, and its chemistry.
Discussion
Numerous attempts have been made to develop resistant hybrid layers, able to withstand degradation processes in the oral cavity by limiting hydrolysis mechanisms and inhibiting enzymes which breakdown the unsupported organic content. Most of them have only been able to retard the problem rather than completely resolve it43,44. Thus, innovative, radical treatments and approaches are still needed7. Considering this, the objective of this systematic review was to investigate the evidence on the benefit of removing the acid-etched exposed collagen, by using deproteinizing agents, on the bond strength of adhesive materials to dentin.
Dentin is a highly mineralized substrate with a particular difference—it contains, in addition to a mineral apatite-based phase (mainly hydroxyapatite), a high volume of organic content that range from 30 to 50% of its total vol%; it is mainly composed of type-I collagen, and as much as 20 vol% of water45,46. Water molecules are substantially retained by water-binding proteins such as PGs30,31. To etch dental substrates, phosphoric acid (PA) has been preferentially used for more than 40 years47. In dentin, the recommended etching time using PA has been limited to 15 s46. This exposes a 5–8 μm dense collagen network devoid of hydroxyapatite minerals, which serves as a target for the priming agent present in modern adhesive systems48. However, deproteinization carried out after acid etching would remove this exposed collagen network, leaving a slightly roughened hydroxyapatite surface that was hidden underneath, with shallow pits thought to be receptable to resin-based materials20. This layer would therefore resemble the bonding mechanism achieved in enamel, which is virtually free from organic content due to its nature. Some authors have named this a “reverse hybrid layer”, since the collagen is no longer infiltrated by the resin-based materials, but rather the resin monomers instead occupy the space where the collagen was originally20,49. NaOCl is also able to solubilize underlying fibers, further contributing to the creation of submicron porosities that foster the retention of polymerized resin monomers49.
The term deproteinization encompasses not only collagen-depletion strategies but also smear layer deproteinization techniques, which should be distinguished from the former. With smear-layer deproteinization, agents such as NaOCl or hypochlorous acid (HOCl) are used as a surface pre-treatment to remove the organic portion of the smear layer, that generally contains not only organic residues but also residual hydroxyapatite, rotatory instrument debris and bacteria25. This was not the aim of the present review, as such technique is used to improve and strengthen subsequent hybridization of self-etch adhesives to dentin50,51.
Since a meta-analysis was not possible given the scarce number of studies found and their associated risk of bias, it cannot be possible to answer the review question with quantifiable confidence, although some important conclusions can be drawn, however. Low quality evidence suggests that there may be a beneficial effect of using deproteinizing agents to increase the immediate bond strength to dentin. None of the studies found a detrimental effect of deproteinizing agents in bonding outcome, when compared to a traditional hybrid layer mechanism, considered to be the control comparison in all studies that were included, as can be seen from the evidence gathered in Table 3. This had already been reported in a clinical study with a 5-year follow-up, although the authors did not counter the oxidizing effect of NaOCl52. Yet, the impact of these results cannot be ignored. Adhesive restorations performed in cavity preparations that involve dentin surfaces have relied on the formation of a hybrid layer, since it was first seen by John McLean in 195253, and later named and described by Nakabayashi in the early 1980’s3,54. Proving that collagen may not be as indispensable as once thought is daunting. A shift in this philosophy is cumbersome but may be required.
Excluding collagen from the bonding layer can be from some points of view advantageous for three concrete reasons: (1) it greatly reduces the technique sensitivity in bonding to dentin, related to its moisture state and the primer used (is it too dry? is it too wet? have I evaporated the solvent enough?)55,56; (2) it increases the longevity of the restorative procedure, since there is less water, less incomplete spaces that were not filled by the adhesive and there will be no enzyme-mediated collagen degradation7,57; finally (3) it provides a favorable substrate, increasing the wettability for hydrophobic resin mixtures to bond to27,30, which were otherwise too viscous to infiltrate the collagen network. In turn, this hydrophobicity also reduces chances of hydrolytic degradation happening at the resin-dentin restorative interface over time.
NaOCl is chemical compound widely used as an endodontic irrigant, known for its non-specific proteolytic and antibacterial activity against the organic layer present in radicular dentin58. Deproteinization accomplished with NaOCl at clinically relevant times, in percentages between 2.5 and 10%, results in an only partial removal of the collagen fibrils59,60. If a collagen-depletion technique is to be considered, leaving this residual organic content bound to the bondable layer may not be entirely desirable. In fact, such partial collagen depletion may also be responsible for some of the material-dependent results seen over the years. What can be clearly seen from Table 5, are the vast differences in the percentages and application times of NaOCl used. These aspects most likely influence not only the extent of the proteolytic activity—how much collagen is dissolved, but also the impact of the redox effect61, detrimental to resin-based materials dependent upon the generation of free-radicals for polymerization mechanisms. Furthermore, the rinsing protocol after the use of NaOCl will most likely also impact the detrimental effect of this agent since ineffective rinsing protocols will not get rid of the residual NaOCl. From the studies in Table 5, many disparities were also found regarding the rinsing protocols62,63,64,65. Prolonged use of NaOCl may also lead to alterations of the elastic modulus of dentin, making it more brittle and prone to cohesive fracture66.
Besides from NaOCl, other alternative irrigants able to dissolve organic content have been proposed and studied in the endodontic field over the years67,68,69. Irrigants such as calcium hypochlorite or chlorine dioxide have been for their organic dissolution capacity70,71. In fact, 5% chlorine dioxide has been reported to show interesting and comparable results, to NaOCl, in bond strength outcomes69. But limited evidence concerning these alternative oxidizing solutions is available.
Regarding the enzymatic strategies, interestingly, a recent SR and meta-analysis evaluated the role of deproteinizing agents on the bond strength to enamel, used before and after acid etching. Indeed, Fernández-Barrera et al. found that deproteinizing after acid etching enamel did not increase bond strengths, while the opposite did72. Papain-based agents were effective in improving the bond strength of resin-enamel interfaces. In the present SR, several different enzymatic agents were used in the eligible studies: bromelain, chondroitinase ABC, papain, or trypsin, with each having their own different targets. Bromelain is a plant protease obtained from the pineapple stem, broadly used in the food industry and able to catalyze the hydrolysis of proteins into amino acids73. Its capacity to promote collagen hydrolysis has been verified experimentally74,75. Similarly, papain is obtained from papaya and is a cysteine protease known for its specificity, commonly used in atraumatic dental procedures as a chemo-mechanical method to remove caries-affected substrate73. Bromelain was applied for 1 min in the study of Chauhan et al. and Khatib et al., with experimental groups achieving better results than the non-deproteinized control39,42. Another study which undertook SEM analysis of the surface-treated dentin, found that bromelain treatment for 1 min resulted in a surface free of collagen remnants when compared to 10% NaOCl32. The concentration of bromelain was not reported in three studies32,39. Papain is already used in caries-affected substrates, although its collagen degradation ability in acid-etched dentin, during a short application period, might be questionable. Still, future studies are needed to consolidate these findings and to determine optimal concentrations and application times of enzymatic solutions.
Removal of PGs and GAGs by virtue of trypsin or chondroitinase ABC digestions was also assessed in three studies included in this SR30,38,40. PGs are proteins that are heavily glycosylated, formed by core protein that have one or more GAG chains. They are water-binding proteins that regulate the hydraulic mechanical support system to the collagen network of the ECM in dentin76. Removing this excess intrinsic water, by cleaving these proteins, can thus aid wettability and interaction of materials with the surface. To cleave the proteins, chondroitinase ABC or trypsin have been used. While chondroitinase cleaves GAGs, trypsin, in contrast, can digest the protein core releasing the GAG chains. Trypsin may also remove other non-collagenous components of the ECM, that are present in dentin30.
It has been shown that trypsin digestion leads to an increased susceptibility of organic tissue degradation, promoting further proteolysis76. Gao et al. pioneered the study that first removed GAGs and PGs in dentin, to assess resulting bond strengths. These researchers found that upon removal of PGs, bond strengths were improved, while the opposite happened when GAGs were removed77. Farina et al., further to this, showed that a 24 h protocol of trypsin digestion, contrarily to the 48 h attempted by Bedran-Russo et al.31 in bovine teeth (excluded from this SR), led to a significant increase in the bond strength of a hydrophobic adhesive blend. This is a result of significant importance30. Viscous and hydrophobic resin mixtures used in adhesive dentistry (i.e., flowable or self-adhesive composites), bonded on their own, without a dental adhesive, have always struggled in securing acceptable bonding to dentin up to this day78,79,80.
One of the major gaps found in the evidence was that long-term bond strength data is scarce and largely unavailable. Studies featuring aged experimental groups are not only necessary, but fundamental in the future, to confirm that a collagen-depletion strategy may in fact stabilize bond degradation, eliminating the unpredictability associated to collagen. In fact, an optimal deproteinization strategy is still missing and more studies are required to reach a consensus. Specifically, it is pertinent for further studies to determine which is the best agent, its concentration, and a clinically relevant time frame at which it can be applied to dentin, to secure successful immediate bonding properties and their longevity. All of which are possible in coming studies.
To conclude, the present findings suggest that an effective collagen-depletion protocol will most likely increase immediate bond strengths. Even more importantly, a correct protocol can improve wettability to dentin surfaces depleted of collagen81, while enabling a greater chance in increasing bonds of hydrophobic blends to dentin.
Conclusion
Although a meta-analysis was not possible due to a reduced number of studies found and concerns regarding their methodological quality, collagen-depletion strategies in dentin, especially when enzymes are used, may improve immediate bond strengths. Only two studies that used NaOCl followed by an antioxidant agent were retrieved. Additional research is required to find the best agent, its concentration and application time. Further studies are needed to consolidate these findings and to determine the role of collagen-depletion strategies on the long-term bond strength. Such strategies would be particularly important to enable bonding of hydrophobic materials and mixtures.
References
Goldberg, M., Kulkarni, A. B., Young, M. & Boskey, A. Dentin structure composition and mineralization. Front. Biosci. E3, 281 (2011).
Van Meerbeek, B., Yoshihara, K., Van Landuyt, K., Yoshida, Y. & Peumans, M. From buonocore’s pioneering acid-etch technique to self-adhering restoratives. A status perspective of rapidly advancing dental adhesive technology. J. Adhes. Dent. 22, 7–34 (2020).
Nakabayashi, N., Kojima, K. & Masuhara, E. The promotion of adhesion by the infiltration of monomers into tooth substrates. J. Biomed. Mater. Res. 16, 265–273 (1982).
Nakabayashi, N., Nakamura, M. & Yasuda, N. Hybrid layer as a dentin-bonding mechanism. J. Esthet. Restor. Dent. 3, 133–138 (1991).
Van Meerbeek, B., Yoshihara, K., van Landuyt, K., Yoshida, Y. & Peumans, M. From buonocore’s pioneering acid-etch technique to self-adhering restoratlves. A status perspective of rapidly advancing dentai adheslve technology. J. Adhes. Dent. 22, 7–34 (2020).
Van Meerbeek, B. et al. Buonocore memorial lecture. Adhesion to enamel and dentin: Current status and future challenges. Oper. Dent. 28, 215–235 (2003).
Bertassoni, L. E., Orgel, J. P. R., Antipova, O. & Swain, M. V. The dentin organic matrix—Limitations of restorative dentistry hidden on the nanometer scale. Acta Biomater. 8, 2419–2433 (2012).
Vaidyanathan, T. K. & Vaidyanathan, J. Recent advances in the theory and mechanism of adhesive resin bonding to dentin: A critical review. J. Biomed. Mater. Res. B Appl. Biomater. 88, 558–578 (2009).
Salz, U., Zimmermann, J., Zeuner, F. & Moszner, N. Hydrolytic stability of self-etching adhesive systems. J. Adhes. Dent. 7, 107–116 (2005).
Iliev, G. et al. Shelf life and storage conditions of universal adhesives: A literature review. Polymer 13, 2708 (2021).
Spencer, P. et al. Adhesive/dentin interface: The weak link in the composite restoration. Ann. Biomed. Eng. 38, 1989–2003 (2010).
Singh, P., Nagpal, R. & Singh, U. P. Effect of dentin biomodifiers on the immediate and long-term bond strengths of a simplified etch and rinse adhesive to dentin. Restor. Dent. Endod. 42, 188–199 (2017).
Mehtala, P. et al. Effect of dimethyl sulfoxide on dentin collagen. Dent. Mater. 33, 915–922 (2017).
Stape, T. H. S. et al. Optimization of the etch-and-rinse technique: New perspectives to improve resin–dentin bonding and hybrid layer integrity by reducing residual water using dimethyl sulfoxide pretreatments. Dent. Mater. 34, 967–977 (2018).
Li, F., Majd, H., Weir, M. D., Arola, D. D. & Xu, H. H. K. Inhibition of matrix metalloproteinase activity in human dentin via novel antibacterial monomer. Dent. Mater. 31, 284–292 (2015).
Laronha, H. et al. Polymerizable matrix metalloproteinases’ inhibitors with potential application for dental restorations. Biomedicine 9, 366 (2021).
Münchow, E. A. et al. Development of an antibacterial and anti-metalloproteinase dental adhesive for long-lasting resin composite restorations. J. Mater. Chem. B 8, 10797–10811 (2020).
Yamauchi, S., Wang, X., Egusa, H. & Sun, J. High-performance dental adhesives containing an ether-based monomer. J. Dent. Res. 99, 189–195 (2020).
Decha, N. et al. Synthesis and characterization of new hydrolytic-resistant dental resin adhesive monomer HMTAF. Des. Monomers Polym. 22, 106–113 (2019).
Prati, C., Chersoni, S. & Pashley, D. H. Effect of removal of surface collagen fibrils on resin-dentin bonding. Dent. Mater. 15, 323–331 (1999).
Vargas, M. A., Cobb, D. S. & Armstrong, S. R. Resin-dentin shear bond strength and interfacial ultrastructure with and without a hybrid layer. Oper. Dent. 22, 159–166 (1997).
de Castro, A. K., Hara, A. T. & Pimenta, L. A. Influence of collagen removal on shear bond strength of one-bottle adhesive systems in dentin. J. Adhes. Dent. 2, 271–277 (2000).
Gomes Torres, C. R., De Araújo, M. A. M. & De Mello Torres, A. C. Effects of dentin collagen removal on microleakage of bonded restorations. J. Adhes. Dent. 6, 33–42 (2004).
Perdigão, J., Lopes, M., Geraldeli, S., Lopes, G. C. & García-Godoy, F. Effect of a sodium hypochlorite gel on dentin bonding. Dent. Mater. 16, 311–323 (2000).
Alshaikh, K. H., Hamama, H. H. H. & Mahmoud, S. H. Effect of smear layer deproteinization on bonding of self-etch adhesives to dentin: A systematic review and meta-analysis. Restor. Dent. Endod. 43, e14 (2018).
Gauthier, M. A., Stangel, I., Ellis, T. H. & Zhu, X. X. Oxygen inhibition in dental resins. J. Dent. Res. 84, 725–729 (2005).
de Almeida, L. et al. Effect of collagen matrix saturation on the surface free energy of dentin using different agents. J. Contemp. Dent. Pract. 16, 531–536 (2015).
Torres, C. R. G. et al. Five-year clinical performance of the dentine deproteinization technique in non-carious cervical lesions. J. Dent. 42, 816–823 (2014).
Siqueira, F. S. F. et al. Three-year effects of deproteinization on the in vitro durability of resin/dentin-eroded interfaces. Oper. Dent. 43, 60–70 (2018).
Farina, A. P., Cecchin, D., Vidal, C. M. P., Leme-Kraus, A. A. & Bedran-Russo, A. K. Removal of water binding proteins from dentin increases the adhesion strength of low-hydrophilicity dental resins. Dent. Mater. 36, e302–e308 (2020).
Bedran-Russo, A. K. B., Pereira, P. N. R., Duarte, W. R., Okuyama, K. & Yamauchi, M. Removal of dentin matrix proteoglycans by trypsin digestion and its effect on dentin bonding. J. Biomed. Mater. Res. B Appl. Biomater. 85, 261–266 (2008).
Dayem, R. & Tameesh, M. A new concept in hybridization: Bromelain enzyme for deproteinizing dentin before application of adhesive system. Contemp. Clin. Dent. 4, 421–426 (2013).
Lobo, S. et al. Antioxidant pre-treatments are able to reduce waiting time for restorative treatment after dental bleaching: A microtensile bond strength exploratory study. Appl. Adhes. Sci. 9, 1–12 (2021).
Prasansuttiporn, T. et al. Effect of antioxidant/reducing agents on the initial and long-term bonding performance of a self-etch adhesive to caries-affected dentin with and without smear layer-deproteinizing. Int. J. Adhes. Adhes. 102, 102648 (2020).
Belmar da Costa, M., Delgado, A. H. S., Pinheiro de Melo, T., Amorim, T. & Mano Azul, A. Analysis of laboratory adhesion studies in eroded enamel and dentin: A scoping review. Biomater. Investig. Dent. 8, 24–38 (2021).
Montagner, A. F., Sarkis-Onofre, R., Pereira-Cenci, T. & Cenci, M. S. MMP inhibitors on dentin stability: A systematic review and meta-analysis. J. Dent. Res. 93, 733–743 (2014).
Lai, S. C. N. et al. Reversal of compromised bonding to oxidized etched dentin. J. Dent. Res. 80, 1919–1924 (2001).
Gao, Y. et al. Effect of dentin proteoglycans on the bonding properties of dentin. Chin. J. Stomatol. 49, 753–757 (2014).
Chauhan, K., Basavanna, R. S. & Shivanna, V. Effect of bromelain enzyme for dentin deproteinization on bond strength of adhesive system. J. Conserv. Dent. 18, 360–363 (2015).
Gao, Y., Zhao, S. J., Wang, P. H., Lu, S. & Chen, J. H. Effect of dentin proteoglycans on the stability of resin-dentin bonds against artificial saliva storage. Zhonghua Kou Qiang Yi Xue Za Zhi 52, 486–491 (2017).
Khan, R. et al. Comparison of different dentin deproteinizing agents on the shear bond strength of resin-bonded dentin. Int. J. Clin. Pediatr. Dent. 13, S69–S77 (2020).
Khatib, M. S., Devarasanahalli, S. V., Aswathanarayana, R. M., Venkateswara, A. H. & Nadig, R. R. Microtensile bond strength of composite resin following the use of bromelain and papain as deproteinizing agents on etched dentin: An in vitro study. Int. J. Clin. Pediatr. Dent. 13, 43–47 (2020).
Göstemeyer, G. & Schwendicke, F. Inhibition of hybrid layer degradation by cavity pretreatment: Meta- and trial sequential analysis. J. Dent. 49, 14–21 (2016).
Montagner, A. F., Sarkis-Onofre, R., Pereira-Cenci, T. & Cenci, M. S. MMP inhibitors on dentin stability. J. Dent. Res. 93, 733–743 (2014).
Tjäderhane, L., Carrilho, M. R., Breschi, L., Tay, F. R. & Pashley, D. H. Dentin basic structure and composition—An overview. Endod. Top. 20, 3–29 (2009).
Breschi, L. et al. Dentin bonding systems: From dentin collagen structure to bond preservation and clinical applications. Dent. Mater. 34, 78–96 (2018).
Buonocore, M. G. A simple method of increasing the adhesion of acrylic filling materials to enamel surfaces. J. Dent. Res. 34, 849–853 (1955).
Sauro, S. et al. Resin-dentin bonds to EDTA-treated vs. acid-etched dentin using ethanol wet-bonding. Part II: Effects of mechanical cycling load on microtensile bond strengths. Dent. Mater. 27, 563–572 (2011).
Abuhaimed, T. S. & Neel, E. A. A. Sodium hypochlorite irrigation and its effect on bond strength to dentin. BioMed Res. Int. 2017, 1–8 (2017).
Kusumasari, C. et al. Effect of smear layer deproteinization with chemo-mechanical caries removal agents on sealing performances of self-etch adhesives. J. Dent. 94, 103300 (2020).
Thanatvarakorn, O. et al. Smear layer-deproteinizing improves bonding of one-step self-etch adhesives to dentin. Dent. Mater. 34, 434–441 (2018).
Rocha Gomes Torres, C. et al. Five-year clinical performance of the dentine deproteinization technique in non-carious cervical lesions. J. Dent. 42, 816–823 (2014).
Staehle, H. J. & Sekundo, C. The origins of acrylates and adhesive technologies in dentistry. J. Adhes. Dent. 23, 397–406 (2021).
Fusayama, T., Nakamura, M., Kurosaki, N. & Iwaku, M. Non-pressure adhesion of a new adhesive restorative resin. J. Dent. Res. 58, 1364–1370 (1979).
Ferrari, M. & Tay, F. R. Technique sensitivity in bonding to vital, acid-etched dentin. Oper. Dent. 28, 3–8 (2003).
Tay, F. R. & Pashley, D. H. Have dentin adhesives become too hydrophilic? J. (Can. Dent. Assoc.) 69, 726–731 (2003).
Tjäderhane, L. et al. Strategies to prevent hydrolytic degradation of the hybrid layer—A review. Dent. Mater. 29, 999–1011 (2013).
Gonçalves, L. S., Rodrigues, R. C. V., Andrade Junior, C. V., Soares, R. G. & Vettore, M. V. The effect of sodium hypochlorite and chlorhexidine as irrigant solutions for root canal disinfection: A systematic review of clinical trials. J. Endod. 42, 527–532 (2016).
Sauro, S. et al. Deproteinization effects of NaOCl on acid-etched dentin in clinically-relevant vs prolonged periods of application. A confocal and environmental scanning electron microscopy study. Oper. Dent. 34, 166–173 (2009).
Mountouris, G., Silikas, N. & Eliades, G. Effect of sodium hypochlorite treatment on the molecular composition and morphology of human coronal dentin. J. Adhes. Dent. 6, 175–182 (2004).
Celestine, M. J., Joseph, L. S. & Holder, A. A. Kinetics and mechanism of the oxidation of a cobaloxime by sodium hypochlorite in aqueous solution: Is it an outer-sphere mechanism? Inorg. Chim. Acta 454, 254–265 (2017).
Rodrigues, N. S. et al. Effect of different conditioning/deproteinization protocols on the bond strength and degree of conversion of self-adhesive resin cements applied to dentin. Int. J. Adhes. Adhes. 81, 98–104 (2018).
Saboia, V. P., Rodrigues, A. L. & Pimenta, L. A. Effect of collagen removal on shear bond strength of two single-bottle adhesive systems. Oper. Dent. 25, 395–400 (2000).
Sacramento, P. A. et al. Influence of NaOCl irrigation and water storage on the degradation and microstructure of the resin/primary dentin interface. J. Adhes. Dent. 13, 213–220 (2011).
Nima, G., Cavalli, V., Bacelar-Sá, R., Ambrosano, G. M. B. & Giannini, M. Effects of sodium hypochlorite as dentin deproteinizing agent and aging media on bond strength of two conventional adhesives. Microsc. Res. Tech. 83, 186–195 (2020).
Grigoratos, D., Knowles, J., Ng, Y. L. & Gulabivala, K. Effect of exposing dentine to sodium hypochlorite and calcium hydroxide on its flexural strength and elastic modulus. Int. Endod. J. 34, 113–119 (2001).
de Conto Ferreira, M. B., Carlini Júnior, B., Galafassi, D. & Gobbi, D. L. Calcium hypochlorite as a dentin deproteinization agent: Microleakage, scanning electron microscopy and elemental analysis. Microsc. Res. Tech. 78, 676–681 (2015).
Seballos, V. G. et al. Effect of post-space irrigation with NaOCl and CaOCl at different concentrations on the bond strength of posts cemented with a self-adhesive resin cement. Braz. Dent. J. 29, 446–451 (2018).
Kamalasanan, R. R. et al. Effect of 5% chlorine dioxide irrigant on micro push out bond strength of resin sealer to radicular dentin: An in vitro study. J. Clin. Diagn. Res. 11, 49–53 (2017).
Rossi-Fedele, G., Guastalli, A. R., Doǧramaci, E. J., Steier, L. & de Figueiredo, J. A. P. Influence of pH changes on chlorine-containing endodontic irrigating solutions. Int. Endod. J. 44, 792–799 (2011).
Khoroushi, M., Najafabadi, M. A. & Feiz, A. Effects of calcium hypochlorite and sodium hypochlorite, as root canal irrigants, on the bond strength of glass fiber posts cemented with self-adhesive resin cement. Front. Dent. 16, 214–223 (2019).
Fernández-Barrera, M. Á. et al. The effect of deproteinizing agents on bond strength of resin-based materials to enamel: A systematic review and meta-analysis of in vitro studies. J. Adhes. Dent. 23, 287–296 (2021).
Alshahli, R. Z. et al. Effect of smear layer deproteinization with enzyme solutions on bonding efficacy of one-step self-etch adhesives. Int. J. Adhes. Adhes. 102, 102672 (2020).
Ketnawa, S., Rawdkuen, S. & Chaiwut, P. Two phase partitioning and collagen hydrolysis of bromelain from pineapple peel Nang Lae cultivar. Biochem. Eng. J. 52, 205–211 (2010).
Faiza, W., Firouzi, A., Islam, M. R., Sumdani, G. & Taher, A. Degradation analysis of epoxy resin composites reinforced with bioprotein: Effects of hydrolysis using papain and bromelain. Polym. Compos. 42, 2717–2727 (2021).
de Mattos Pimenta Vidal, C., Leme-Kraus, A. A., Rahman, M., Farina, A. P. & Bedran-Russo, A. K. Role of proteoglycans on the biochemical and biomechanical properties of dentin organic matrix. Arch. Oral Biol. 82, 203–208 (2017).
Wang, W. et al. Structure-property relationships in hybrid dental nanocomposite resins containing monofunctional and multifunctional polyhedral oligomeric silsesquioxanes. Int. J. Nanomed. 9, 841–852 (2014).
Frankenberger, R., Lopes, M., Perdigão, J., Ambrose, W. W. & Rosa, B. T. The use of flowable composites as filled adhesives. Dent. Mater. 18, 227–238 (2002).
Monticelli, F., Osorio, R., Mazzitelli, C., Ferrari, M. & Toledano, M. Limited decalcification/diffusion of self-adhesive cements into dentin. J. Dent. Res. 87, 974–979 (2008).
Poitevin, A. et al. Bonding effectiveness of self-adhesive composites to dentin and enamel. Dent. Mater. 29, 221–230 (2013).
Toledano, M. et al. Effect of acid etching and collagen removal on dentin wettability and roughness. J. Biomed. Mater. Res. 47, 198–203 (1999).
Ribeiro, A. I. A. M. et al. Ação dos agentes desproteinizantes e antioxidantes sobre a resistência de união à microtração de sistemas adesivos convencionais. Rev. Gaucha Odontol. 59, 221–227 (2011).
Toledano, M., Perdigão, J., Osorio, E. & Osorio, R. Influence of NaOCl deproteinization on shear bond strength in function of dentin depth. Am. J. Dent. 15, 252–255 (2002).
Osorio, R., Ceballos, L., Tay, F., Cabrerizo-Vilchez, M. A. & Toledano, M. Effect of sodium hypochlorite on dentin bonding with a polyalkenoic acid-containing adhesive system. J. Biomed. Mater. Res. 60, 316–324 (2002).
Munksgaard, E. C. Wet or dry, normal or deproteinized dentin surfaces as substrate for dentin adhesives. Acta Odontol. Scand. 60, 60–64 (2002).
Uceda-Gómez, N. et al. Effect of sodium hypochlorite on the bond strength of an adhesive system to superficial and deep dentin. J. Appl. Oral Sci. 11, 223–228 (2003).
De Souza, F. B., Vicente Silva, C. H., Palma Dibb, R. G., Delfino, C. S. & De Souza Beatrice, L. C. Bonding performance of different adhesive systems to deproteinized dentin: Microtensile bond strength and scanning electron microscopy. J. Biomed. Mater. Res. Appl. Biomater. 75, 158–167 (2005).
Da Silva, E. M., Duarte, P. B. P. G., Poskus, L. T., Barcellos, A. A. L. & Guimarães, J. G. A. Nanoleakage and microshear bond strength in deproteinized human dentin. J. Biomed. Mater. Res. B Appl. Biomater. 81, 336–342 (2007).
Uceda-Gómez, N. et al. Long-term bond strength of adhesive systems applied to etched and deproteinized dentin. J. Appl. Oral Sci. 15, 475–479 (2007).
Erhardt, M. C. G. et al. Influence of dentin acid-etching and NaOCl-treatment on bond strengths of self-etch adhesives. Am. J. Dent. 21, 44–48 (2008).
Saboia, V. P. A. et al. Adhesion of a two-step etch-and-rinse adhesive on collagen-depleted dentin. J. Adhes. Dent. 10, 419–422 (2008).
Silva, G. O., Barcellos, D. C., Pucci, C. R., Borges, A. B. & Torres, C. R. G. Longitudinal bond strength evaluation using the deproteinized dentin technique. Gen. Dent. 57, 328–333 (2009).
Braz, R. et al. Influência da remoção de colágeno sobre a resistência de união de adesivos autocondicionantes. Pesqui. Bras. Odontopediatr. Clin. Integr. 9, 49–54 (2009).
Baseggio, W. et al. Effect of deproteinization and tubular occlusion on microtensile bond strength and marginal microleakage of resin composite restorations. J. Appl. Oral Sci. 17, 462–466 (2009).
Chaharom, M. E. E., Kahnamoii, M. A., Kimyai, S. & Moghaddam, M. H. Effect of sodium hypochlorite on the shear bond strength of fifth- and seventh-generation adhesives to coronal dentin. Afr. J. Biotechnol. 10, 12697–12701 (2011).
Barbosa De Souza, F., Sinclér Delfino, C., Lacalle Turbino, M. & Braz, R. Deproteinized dentin: A favorable substrate to self-bonding resin cements? J. Biomed. Mater. Res. B Appl. Biomater. 98, 387–394 (2011).
Aguilera, F. S., Osorio, R., Osorio, E., Moura, P. & Toledano, M. Bonding efficacy of an acetone/based etch-and-rinse adhesive after dentin deproteinization. Med. Oral Patol. Oral Cir. Bucal 17, 649–654 (2012).
Lisboa, D. S., dos Santos, S. V., Griza, S., Rodrigues, J. L. & Faria-e-Silva, A. L. Dentin deproteinization effect on bond strength of self-adhesive resin cements. Braz. Oral Res. 27, 73–75 (2013).
Faria-E-Silva, A. L., Araújo, J. E., Rocha, G. P., De Oliveira, A. D. S. & De Moraes, R. R. Solvent content and dentin bond strengths using water-wet, ethanol-wet and deproteinization bonding techniques. Acta Odontol. Scand. 71, 710–715 (2013).
Di Francescantonio, M. et al. Sodium hypochlorite effects on dentin bond strength and acid-base resistant zone formation by adhesive systems. Braz. J. Oral Sci. 14, 334–340 (2015).
Montagner, A. F. et al. Effect of sodium hypochlorite as dentinal pretreatment on bonding strength of adhesive systems. Indian J. Dent. Res. 26, 416–420 (2015).
De Souza, L. C. et al. Deproteinization stabilises dentin bonding of self-adhesive resin cements after thermocycling. Int. J. Adhes. Adhes. 66, 53–58 (2016).
Pucci, C. R. et al. Influence of dentin deproteinization on bonding degradation: 1-year results. J. Contemp. Dent. Pract. 17, 985–989 (2016).
Dikmen, B. & Tarim, B. The effect of endodontic irrigants on the microtensile bond strength of different dentin adhesives. Niger. J. Clin. Pract. 21, 280–286 (2018).
Author information
Authors and Affiliations
Contributions
Conceptualization of the paper and investigation was carried out by A.H.S.D. and M.B.C. The investigation was conducted by A.D. and M.B.C. First draft preparation was carried out by A.D., M.B.C and S.S. Writing and reviewing of the paper was carried out by A.H.S.D., M.B.C., A.M.A., M.P. and S.S. Supervision of the research was carried out by A.M.A, M.P. and S.S. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
H. S. Delgado, A., Belmar Da Costa, M., Polido, M.C. et al. Collagen-depletion strategies in dentin as alternatives to the hybrid layer concept and their effect on bond strength: a systematic review. Sci Rep 12, 13028 (2022). https://doi.org/10.1038/s41598-022-17371-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-022-17371-0
- Springer Nature Limited