Abstract
Metastasis is the leading cause of death in colorectal cancer (CRC) patients, and the liver is the most common site of metastasis. Tumor cell metastasis can be thought of as an invasion-metastasis cascade and metastatic organotropism is thought to be a process that relies on the intrinsic properties of tumor cells and their interactions with molecules and cells in the microenvironment. Many studies have provided new insights into the molecular mechanism and contributing factors involved in CRC liver metastasis for a better understanding of the organ-specific metastasis process. The purpose of this review is to summarize the theories that explain CRC liver metastasis at multiple molecular dimensions (including genetic and non-genetic factors), as well as the main factors that cause CRC liver metastasis. Many findings suggest that metastasis may occur earlier than expected and with specific organ-anchoring property. The emergence of potential metastatic clones, the timing of dissemination, and the distinct routes of metastasis have been explained by genomic studies. The main force of CRC liver metastasis is also thought to be epigenetic alterations and dynamic phenotypic traits. Furthermore, we review key extrinsic factors that influence CRC cell metastasis and liver tropisms, such as pre-niches, tumor stromal cells, adhesion molecules, and immune/inflammatory responses in the tumor microenvironment. In addition, biomarkers associated with early diagnosis, prognosis, and recurrence of liver metastasis from CRC are summarized to enlighten potential clinical practice, including some markers that can be used as therapeutic targets to provide new perspectives for the treatment strategies of CRC liver metastasis.
Similar content being viewed by others
Background
Colorectal cancer (CRC) is the world’s second leading cause of cancer deaths, and liver metastasis from CRC accounts for the majority of fatalities in CRC patients [1, 2]. CRC’s most common target metastatic sites are the liver, lung, bone, and brain, known as organ tropism, and the liver is the most common site of CRC metastasis [3]. Up to 50% of CRC patients have liver metastasis, and approximately 15–23% of patients have metastasis at the time of diagnosis. Hepatic resection combined with modern adjuvant systemic regimens is only effective in 20% of colorectal cancer liver metastasis (CRLM) patients. Even after curative hepatic resection, the 5-year overall survival rate is around 48% [4]. In practice, however, approximately 80% of CRLM patients have unresectable metastatic lesions [5, 6].They are typically downstaged by systemic and local therapy (including stereotactic radiotherapy, transarterial radioembolization, and transarterial chemoembolization) to achieve metastatic liver lesion resection, improving patients’ long-term survival and prognosis. Although the development of adjuvant systemic therapy has significantly improved the clinical outcome of patients with stage IV CRC liver metastases [7], early detection of CRLM remains critical to good results. Despite the high resolution of computed tomography (CT) as the most commonly used detection modality, up to 30% of liver metastasis cannot be detected in their early stages. MRI and PET-CT may have higher sensitivity and specificity, but the costs are prohibitive [2].
Many studies have found that cancer metastasis is a complex selective process influenced by anatomical, biological, and microenvironmental factors [8]. Theories such as “seed-soil,” pre-niche, and the crosstalk between tumor cells and immune cells provide direct evidence for metastatic propensity and organ-specific tropism of metastatic cancer cells, implying that metastasis is a process that is dependent on organ-targeted anchoring characteristics [9,10,11]. Thus, investigating how metastatic CRC appears, when it appears, and the underlying mechanism provides clues for the treatment of CRC and its liver metastasis.
Theories underlying organ-specific metastasis process
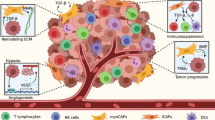
According to a traditional model of metastatic spread, cancer cells undergo the following general steps known as metastatic cascade, which can be divided into two major phases: (1) dissemination from the primary lesion to distant organs by entering systemic circulation and adapting to a new microenvironment (intravasation, extravasation), and (2) colonization followed by expanding growth [12]. The multi-step process typically involves an invasion-metastasis cascade. According to Hanahan and Weinberg, the hallmark of “activating invasion and metastasis” is one of the six important features of cancer. Invading cancer cells pass through or collaborate with stroma to avoid elimination by immune system cells such as neutrophils, monocytes/macrophages, and endothelial cells. Epithelial-mesenchymal transition (EMT) program can be activated by carcinoma cells and orchestrate most steps of invasion and metastasis, except for colonization. Disseminated tumor cells act in dormancy in circulation and new environment tissue to avoid immune surveillance, and then they interact with the tissue microenvironment to be awakened from dormancy. Moreover, the development of metastatic colonization needs multiple biological programs, and these adaptations require intrinsic capabilities of cancer cells and a permissive tumor microenvironment with stromal support cells. The invasion-metastasis process, which appears to be a linear progression from the primary tumor to metastatic colonies, operates under the guidance of a specific paradigm [13,14,15]. However, several studies have suggested that metastasis cannot be defined solely by chronological order, as many interdigitated and mutually exclusive metastasis events do not appear to follow a linear progression model [16]. In our understanding of CRC metastasis to the liver, the general “seed and soil” theory and the “mechanistic theory” are highly complementary [17]. Anatomically, CRC metastasis is thought to occur in a stepwise fashion, with the majority of venous drainage from the intestine entering the portal system to flow into the liver, and then the disseminated cells in the bloodstream are arrested by the first available liver capillary beds with endothelial cells and basement membrane [18]. Although the physical characteristics influence organ tropism and affect the non-random organ-specificity, the fact is that organs receiving similar blood volumes have distinct metastatic-formation efficacy. The special ability of circulating tumor cells to form secondary growth cannot be explained as purely mechanistic [19, 20]. Cancer cells entering the circulation disperse in various directions, but their anchorage to specific metastatic sites is determined by various factors [21]. Stephen Paget proposed a hypothesis in 1889 that described cancer cells as “seeds” and receptive microenvironments as “soils,” both of which are required for rate-limiting steps in the formation of micrometastasis [9]. Recently, growing evidence for the process of pre-metastatic niche formation adds new insights to the “seed-soil” theory. For example, metastasis-initiating cells co-opt the metastatic microenvironment to facilitate colonization. Before cancer cells disseminate from the primary tumor, a subpopulation of primary tumor cells has the “prime” potential and reprograms the distant microenvironments [22]. Pre-metastatic niche formation is important in CRLM. By recruiting various cellular components (Kupffer cells, macrophages, and fibroblasts), producing CRC-derived factors (chemokines and cytokines) and exosomes, primary CRC prepares a favorable microenvironment in the liver. It mediates liver-target metastasis [23, 24]. As a result, the concept of pre-metastasis niches may trump the chronological metastasis pattern that is dependent on circulation. Recently, many discoveries have clarified the key molecules and cells involved in liver-specific metastasis of CRC. For example, LINC00485 is a newly discovered class of Long non-coding RNAs (lncRNAs), and low expression of LINC00485 predicts a poor prognosis for CRC patients. LINC00485 attenuated CRC cell invasion and liver metastasis by directly modulating the miR-581/EDEM1 axis. Overexpression of LINC00485 enhanced the expression of epithelium markers E-cadherin and significantly down-regulated the expression of mesenchymal markers N-cadherin, indicating a loss of malignant phenotype in cancer cells [25]. Higher levels of tumor suppressor microRNAs (miR-25-3p, miR-130b-3p, miR-425-5p, miR-934) in the exosomes were secreted by CRC cells in more advanced disease, and these exosomal miRNAs induced macrophages M2 polarization to promote liver metastasis of CRC. CXCL13 secreted by M2-polarized macrophages promoted the transcription of exosomal miR-934 in CRC cells, forming a positive feedback loop to foster CRLM [26, 27]. Recent studies have revealed the complex interactions between immune cells (myeloid-derived suppressor cells, macrophages, Kupffer cells) and cancer cells in the tumor microenvironment. For example, primary CRC tumor-secreted VEGF-A stimulated tumor-associated macrophages to produce CXCL1, which recruited CXCR2-positive myeloid-derived suppressor cells (MDSCs) accumulated in the pre-metastatic site and facilitated liver metastasis [28]. Furthermore, a high level of IL-6 secreted by tumor cells recruited MDSCs to accumulate in the pre-metastatic niche in the liver, which was stimulated by the S1PR1–STAT3 signaling pathway and positively correlated with the number of metastatic liver nodes. Increased CD14+HLA-DR−/low MDSCs in CRLM patients were shown to inhibit T-cell proliferation and predict poor prognosis [10]. ANGPTL1 was significantly down-regulated in CRC-derived exosomes. It attenuated CRLM by reprogramming Kupffer cells (KCs) to reduce their MMP9 expression, which helped prevent vascular leakage [29]. Under hypoxic conditions, exosomal miR-135a-5p was released from primary tumor cells and phagocytosed by KCs, which selectively initiated a favorable pre-metastatic formation in the liver by establishing an immunosuppressive microenvironment [30]. In addition, some cell adhesion molecules have been demonstrated to play an important role in CRC liver metastasis. It has been reported that α5β1 Integrin was highly expressed in metastatic CRC cells [31]. E-cadherin ensures the epithelial integrity, and membrane E-cadherin in metastatic cancer cells is lost during tissue dissociation or tumor invasion. Disruption of E-cadherin relieved REST-mediated repression of L1CAM and upregulated L1CAM expression [32] (Fig. 1).
Key molecules and cells for liver-specific metastasis of CRC. Invasive cancer cells from primary CRC tumors invade the adjacent vasculature from the primary site. The invading tumor cells circulate within the blood vessels protected by platelets, extravasate, and finally colonize the liver. The key molecules and cells involved in liver-specific metastasis of CRC include immune cells (MDSCs, TAMs, Kupffer cells), cytokines (IL-6, VEGF-A), chemokines (CXCL13, CXCL1, CXCL12), exosomes (miR-25-3p, miR-130b-3p, miR-425-5p, miR-934, miR-135a-5p, miR581, ANGPTL1) and cell adhesion molecules (L1CAM, E-cadherin, α5β1 Integrin and N-cadherin). Several molecular and cellular interactions have been identified to play critical roles in CRC liver metastasis, and these factors may influence the organ tropism of CRC metastasis
Metastasis is recognized as an evolutionary process, with the genomes carrying the archeological record of each metastatic cancer cell. Indeed, many genomic studies have been conducted to explain metastatic subclones’ emergence and and investigate the “timing, route, and direction” of metastatic spread [33]. It appears that the metastasis process is not constrained to a specific model of evolution but rather follows complex evolution patterns, which means that single or multiple clone(s) with metastatic traits leave the primary tumor at distinct timing points and form metastases in distant organs via various trajectories, including monophyletic seeding and polyphyletic seeding [34, 35]. The relative timing of metastatic spread is determined by comparing the genetic divergence between the primary tumors and metastasis. In the classic “linear evolution model,” the metastasis-initiating clone(s) emerge late in the primary tumor and seed at the metastatic sites as a byproduct of tumor development. Instead, in the “parallel evolution model,” the metastatic subclone(s) spread from the primary tumor to distant sites early, and both the primary and metastatic subclones evolve concurrently. As a result, compared with the “linear evolution model,” a greater degree of Primary-Metastasis genetic divergence is expected in the “parallel evolution model” [36]. In addition to the time of metastasis, studies of the clonal relationship between primary tumors and metastases explained metastatic seeding patterns based on genomic data analysis: identification of monoclonal/polyclonal metastasis and monophyletic/polyphyletic metastasis may provide information for treatment improvement. In metastatic CRC, both monoclonal/polyclonal metastasis and monophyletic/polyphyletic seeding patterns were observed. Polyclonal metastasis appeared to be the most common type of CRC metastasis. A polyphyletic seeding pattern was observed in the case of CRC with liver metastasis followed by lung metastasis [37, 38]. For the distinct routes of metastasis, Hai-ning et al. investigated the genomic evolution for the clonal origin and revealed three metastatic models (sequential, branch-off, and diaspora) by phylogenetic reconstruction using Treeomics. The results of the genomic analysis showed that liver and lung metastasis might originate from primary tumors independently rather than subsequently, providing genomic evidence for the organotropisms of metastatic CRC cells. However, the relationship between the characteristics of primary site subclones and their potential for liver metastasis has not been thoroughly investigated [38].
The underlying molecular mechanisms and contributing factors involved in CRLM
Genetic and epigenetic changes associated with liver metastasis of CRC
Cancer cells are thought to acquire metastatic capacity due to genetic and epigenetic changes. Genetic mutations can potentially disrupt epigenetic patterns, and the interaction between these two mechanisms can promote metastasis. [33, 39,40,41,42].
Although numerous genetic alterations have been detected between the primary tumor and metastatic sites in CRC [43,44,45], much remains unknown about the interaction between tumor genomic features and metastatic potential and organ-specific metastatic patterns [34, 46]. The clonal relationship observed between paired primary tumors, and metastasis explains at least part of the dissemination of metastasis-competent clones in different temporal patterns and trajectories [36, 47, 48]. This section will will sort out the genetic/epigenetic alterations associated with CRC liver-specific metastasis and summarize recent studies on the metastatic evolution patterns (temporal patterns and routes) observed in the metastatic CRC cohort.
Genetic alterations associated with liver metastasis of CRC
Several whole-genome sequencing analyses on metastatic tumors have been performed in recent years to gain insight into the critical genetic events involved in CRLM. Several studies have been conducted to investigate single nucleotide variations (SNVs), mutated genes, and chromosome copy numbers of CRLM. An analysis of metastatic solid tumor genomes revealed that consistent genetic changes indicate cancer metastasis remains to be further identified [44]. A pan-cancer cohort study of 25,000 patients’ tumor genomic profiling identified the associations between genetic alterations and metastatic patterns in 50 tumor types. The result showed that copy number alterations were not significantly associated with the metastatic burden for CRC. Chromosomal instability may be established early in tumor development and was already high in patients with low metastatic burden [45]. Oga et al. discovered 6855 mutations in primary CRC tumors without liver metastasis, primary metastatic CRC, and paired liver metastasis (LM) lesions using whole-exome sequencing (WES) analysis. The result showed that the somatic genomic profiles of primary CRC tumors and LM lesions were not significantly different; however, LM regions showed an enriched A-to-C nucleotide conversion in the context of “AAG,” an event that may be specific to liver metastases [49]. Li et al. used WES to look for somatic SNVs (sSNVs) in primary tumors and matched liver metastasis samples from 16 CRC patients with liver metastasis. The SNVs data were analyzed using ABSOLUTE software to calculate the proportion of mutational genes in each sample. An average of 34% (8–63%) mutations were shared by both primary tumors and liver metastasis, indicating a common ancestral trunk among them. Furthermore, an average of 34% (12%–88%) mutations were metastasis-private, which may be a result obtained or lost during the tumor metastasis process. The probable timing order of mutation events has been investigated by analyzing the distribution of cancer cell fractions (CCF). A higher median CCF value indicates that the mutation occurred earlier. Data on the median CCF value of TP53 and KRAS showed that TP53 mutations occurred earlier than KRAS in primary tumors but later than KRAS in liver metastasis [50].
Several studies have been conducted to identify frequently mutated genes involved in metastasis. For example, 707 genes have been identified as LM-associated genes, which specifically mutated in the LM regions but not in CRC tumors without liver metastasis, including ADAMTS10, NELL1, and RXFP3, implying their roles in liver metastasis. Furthermore, ADAP1 fusions were discovered in the RNA-seq dataset, indicating that ADAP1 was fused to GET4, SUN1, or NOC4L in an out-of-frame manner in the LM region. Two in-frame fusions of the ADAP1’s ArfGAP domain with proteins from GEMIN4 and TMEM8A have been discovered, which may facilitate metastasis by activating GTPase [49]. A study used targeted sequencing of primary tumors and matched liver metastasis samples to describe the genome landscape of Chinese CRLM patients. The most frequently mutated genes were found to be TP53 (324/396, 82%), PC (302/396, 76%), KRAS (166/396, 42%), SMAD4 (54/396, 14%), FLG (52/396, 13%), and FBXW7 (43/396, 11%). Furthermore, the distribution of genomic changes was related to the time of metastasis (synchronous/metachronous liver metastasis). Alterations in genes of FBXW7, FLT3, XIRP2, TSC2, LATS1, and CREBBP were significantly enriched in metachronous lesions, and alterations in CDK12 were significantly enriched in synchronous LM [51].
The differences in chromosome copy number between primary and secondary tumors revealed that genetic aberrations in liver metastasis are a dynamic process, such as the presence of a focal amplification of chromosome 7p in primary tumors but not in the LM region. The loss or gain of copy number variations (CNVs) most likely allows clones to be more fit in a new environment [49]. Anand and colleagues investigated the link between aneuploidy and CRC metastasis. Aneuploidy is not just a byproduct of chromosomal instability; it has a direct influence on cancer cells’ metastatic capability, either promoting or inhibiting metastasis behavior. HCT116 colon cells with an extra copy of chromosome 5 exhibit increased invasive behavior by activating an EMT program and upregulated matrix metalloproteinases (MMPs) [52]. In addition, CNV alterations, as a common biological event during tumor progression and therapy, usually involve multiple genes. There are potentially complex interactions between co-amplified or co-deleted genes affected by CNV events, acting as a whole. It has been reported that CDK12 and HER2 were frequently co-amplified in CRC, and inhibition of CDK12 can enhance the sensitivity of CRC cells to lapatinib, an anti-HER2 tyrosine kinase inhibitor (TKI) [53].
Genome events related to metastatic evolution pattern
According to phylogenetic analysis of non-synonymous SNVs from the primary tumor and metastatic liver lesions, there were three main clonal evolution patterns from primary to liver metastases: clonal-clonal pattern (C–C) (early events), subclonal-clonal pattern (S–C) (middle-stage events), none-clonal pattern (0-C) (later events). In terms of CNV events, Chr 20q amp, 17p del, 18q del, and 8p del in clonal- clonal evolution were considered as early events, 8q amp in liver metastasis-specific evolution was considered as later events, and 8q amp, 13q amp, and 8p del in subclonal-clonal evolution were considered as middle-stage events. SYNE1 was a mutant gene with S–C clonal evolution characteristics. Its mRNA expression level in normal, CRC primary, and liver metastasis gradually decreased; however, its functional mechanism in CRLM remains unknown [50]. Tumor mutation burden, an immunotherapy biomarker, in conjunction with HLALOH (HLA, Human leukocytes antigens, LOH, Loss of heterozygosity), is used as an indicator to assess the efficacy of immunotherapy [54]. Subclonal mutation loads were higher in primary tumors than in clonal mutation loads. In contrast, the proportion of clonal mutation was increased in metastatic lesions, which is consistent with the S–C evolutionary pattern, indicating the role of selection in metastasis. HLA LOH occurred in samples with recurrent mutations of S–C changing pattern, including KRAS, SYNE1, FBXL2, DNAH11, and CACNA1H, indicating that this mutational clonal pattern promotes CRC cells evading the immune system during liver metastasis [50].
Epigenetic modifications associated with liver metastasis of CRC
No genetic changes have been identified as consensus metastasis-specific drivers in the process of CRC metastasis. However, epigenetic changes may provide an alternative mechanism to induce tumor cells for metastatic phenotypes [55]. The core content of epigenetic modification is the covalent modification of histones and nucleic acids (including methylation, acetylation, ubiquitination, etc.). In addition, epigenetic regulation also includes chromatin remodeling and transcriptional mediators (mainly non-coding RNAs, such as microRNAs and long ncRNAs) of the RNA splicing machinery. They affect gene expression without sequence changes in DNA [56, 57] (Fig. 2). Epigenetic changes play an important role in CRLM (Table 1), but whether there are metastasis-specific epigenetic drivers and their mechanism need to be investigated further [40].
The role of epigenetic modifications in CRC liver metastasis. Epigenetic modification plays a vital role in gene regulation, mainly for various covalent modifications of histones and nucleic acids. The change of nucleic acid is in DNA and RNA. In addition, epigenetic modification also includes chromatin remodeling, non-coding RNA regulation, and other mechanisms. DNA methylation mainly occurs at the C of 5′-CpG-3′ to generate 5-methylcytosine (5mC). Under the action of DNA methyltransferase (DNMT), methyl groups are covalently bonded to the 5' carbon of cytosines of CpG dinucleotide residues. Hypermethylated gene expression is suppressed. Chromatin remodeling can regulate gene expression by regulating chromatin changes in chromatin structure and location, such as PU.1 opening chromatin regions of downstream effector genes and recruiting additional epigenetic modifiers to regulate gene expression. N6-Adenylate methylation (m6A), which inserts a methyl substituent on the N atom at the 6-position of adenosine. During transcription, m6A deposited on RNA transcripts affects gene expression post-transcriptionally by altering the structure of RNA or the specific recognition of m6-binding proteins. Non-coding RNAs are endogenous RNA molecules that cannot be translated into proteins but have particular gene expression regulatory functions, regulating post-transcriptional gene expression by complementary binding to RNA transcripts of the target gene
Dysregulation in DNA methylation is the mainly studied DNA modification in tumor and metastasis [64]. The methylation changes of primary CRC, metastatic CRC, and liver metastases differ between individuals. CRC primary tumors exhibited global hypomethylation and CpG island (CGI) hypermethylation compared to healthy tissues, whereas metastatic colorectal lesions exhibit high-level global methylation but lower CGI methylation [65]. The study by Udali et al. came to the same conclusion. Primary CRC and synchronous liver metastases had similar epigenetic DNA hypomethylation status when compared with homologous cancer-free colon tissues, indicating that these epigenetic mechanisms occurred in the early stages of CRC development and were maintained till the stage of liver metastasis progression [66]. However, the mechanism by which methylation inhibits tumor suppressor gene expression may be compromised during metastasis. Mahdi et al. discovered that the regulatory mechanism of methylation on gene expression might be compromised during the process of CRC tumor cell metastasis and colonization in the liver. The expression levels of three endothelin system genes changed significantly during the liver colonization of CC531 cells. When metastatic cell lines were exposed to Decitabine (DAC, which inhibits DNA methyltransferases), the expression of endothelin system genes did not increase, indicating that these gene expression changes were not caused by DNA methylation. This suggests that the regulatory function of epigenetic alterations may be gradually lost in the late stage of metastasis [67]. The microenvironment-induced epigenetic mutation is an essential mechanism for metastatic tumor cells to grow in their new niche. The hepatic growth factor (HGF) is abundant in the microenvironment of liver metastases. HGF from the metastatic liver microenvironment was shown to activate the c-Met/PI3K/AKT/mTOR axis in CRC cells, activating the SREBP2-dependent cholesterol biosynthesis pathway to promote CRC liver metastasis [68]. PU.1 is a pioneer factor that remodels chromosomes by opening the enclosed chromatin and enlisting the help of additional epigenetic modifiers. According to one study, HGF caused PU.1 phosphorylation in metastatic cells. The phosphorylated PU.1 regulated downstream regulatory elements to activate the effector gene DPP4. The HGF/PU.1/DPP4 axis was activated, which promoted the growth of CRC tumor cells at the site of the liver. Targeting the chromatin remodeling pathway in the future may provide additional treatment options for metastatic cancer [58].
RNA N6-methyladenosine (m6A) is the most prevalent internal modification in messenger (m)RNA in eukaryotes, and recent studies have shown that m6A modification also exists in lncRNAs, Circular RNAs (circRNAs), and pre- Micro RNAs (pre-miRNAs), and plays essential roles for their biogenesis and functions [69]. In CRC, m6A modification accelerates CRC progression and metastasis by promoting glycolysis in cancer cells, inducing immunosuppression in the tumor microenvironment, maintaining tumor cell stemness, and promoting chemoresistance [64]. One study found that YTHDF1, a translation-promoting cytoplasmic m6A reader, was the most significantly upregulated m6A regulator in CRC cells. YTHDF1 activated RhoA signaling by binding to the m6A site of ARHGEF2 messenger RNA, leading to enhanced translation of ARHGEF2, and the YTHDF1-m6A-ARHGEF2 axis promoted the growth of CRC cell lines in primary organs as well as in lung and liver metastases [59]. circNSUN2 was found to be frequently upregulated in tumor tissue and serum samples from CRC patients with liver metastases, suggesting poor overall survival. The experimental results showed that the N6-methyladenosine modification of circNSUN2 regulated its cytoplasmic export and enhanced the stability of HMGA2 mRNA by forming a circNSUN2/IGF2BP2/HMGA2 RNA–protein ternary complex, which led to the LM of CRC. And circNSUN2 may serve as a novel therapeutic target of CRLM, providing treatment options for patients with CRLM [60].
The role of miRNA expression and miRNA-gene regulation in the progression and metastasis of CRC is relatively well understood. Anne-Marie et al. demonstrated that c-met was a direct target of miR-146a. Overexpression of miR-146a inhibited the expression of the proto-oncogene c-met, which reduced the development of liver metastasis and may be used as a therapeutic target for treating CRLM [61]. Lee et al. compared the miRNA and gene expression profiles of two groups of primary tumor samples with and without metastasis to identify the miRNA-target regulators to initiate metastasis [70]. The miR-424 has previously been shown to be upregulated and specifically target FGFR1 to inhibit its expression in placental trophoblasts [71], which is consistent with the miRNA-target network results. While PDGFRB inhibited miR-30b expression, which may be linked to the initiation of CRLM, miR-30b has been shown to promote cancer cell apoptosis in human gastric cancer tissues [72]. Circ RNAs have been shown to contain many miRNA binding sites and function as miRNA sponges, allowing them to indirectly regulate miRNA-target genes' expression. It has also been demonstrated that it encodes peptides with regulatory procedures. Zheng et al. discovered that the expression of circPPP1R12A, which encoded a conserved 73-aa small peptide, circPPP1R12A-73aa, was significantly increased in primary colon cancer tissues. Not circRNA circPPP1R12A itself but circPPP1R12A-73aa can promote CRC metastasis by inhibiting MST1/2 and LATS1/2 to activate the Hippo-YAP signaling pathway, which may be used as a therapeutic target for CRLM [62]. LncRNAs modulate the biological process of tumor metastasis via mechanisms such as transcriptional regulation and chromosome modification. Ben Yue and colleagues discovered a new positive feed-forward loop mechanism between LncRNA cytoskeleton regulator RNA (CYTOR) and Wnt/β-catenin signaling that promotes EMT phenotype and CRC metastasis. Targeting the CYTOR/β-catenin axis may be a promising treatment strategy for CRLM [63].
Dynamic stem cell hierarchies and stem cell states in CRLM
Several cellular properties, including cancer stem cell (CSC)-like traits, EMT, and autophagy, among others, are linked to the acquisition of metastatic ability [73, 74]. According to the cancer stem cells hypothesis, CSC-like properties (self-renewal ability and cellular plasticity) of primary tumor cells are an obvious prerequisite for tumor initiation and metastatic clone formation [75, 76].
Dynamic hierarchical and phenotypic changes related to cell plasticity in liver metastasis of CRC
The dominant view of hierarchical organization is a critical concept for understanding the role of cellular plasticity within cancer stem cells, differentiated cells, and cells in the intermediate state, which reflects cell–cell cooperation between different clones [77]. Many studies have shown that both primary tumors and metastatic lesions of CRC have hierarchically organized structures with heterogeneous cell populations (CSCs, niche cells, transient amplifying cells, and differentiated cells) in dedicated niches (Fig. 3A). According to the “hardwired stem cell hierarchy” model, it believes that CSCs are located at the bottom of the structure (intestinal soil crypts), split asymmetrically into a CSC and a transient amplification (TA) cell, with the latter dedicated to differentiation (Fig. 3B). In contrast, increasing evidence supports the “CSC-driven tumor hierarchy” model, which allows daughter cells (TA cells) and fully differentiated cells to dedifferentiate into stem cells to replenish the damaged CSC pool [78,79,80] (Fig. 3C). Many studies have shown that CSCs are the root of CRC metastasis. For example, according to Dieter et al. research, three types of tumor-initiating cells (TICs) with stem-like properties were identified, and only the long-term TICs (LT-TICs) characterized as extensively self-renewing potential was assumed to drive CRC metastasis [76].
Dynamic hierarchy and phenotypic plasticity of CSCs are prerequisites for CRLM. The intestinal crypt-villus structure is a dynamic stem cell hierarchical organization essential in colorectal cancer progression and metastasis. The compartment of hierarchical organization of colorectal cancer contains tumor cells in different states: SC cells, TA cells, and TD cells. Stem cells (SC) at the apex of hierarchies generate transient amplifying (TA) progenitor cells, and TA cells differentiate into terminally differentiated (TD) cells. B and C represent the hardwired stem cell hierarchy and the novel dynamic stem cell hierarchy, respectively. The latter model has a more plastic stem cell hierarchy, meaning that TA and TD cells can dedifferentiate to replace lost SCs through reprogramming. D. The role of cellular plasticity in colorectal cancer metastasis. CRC models demonstrate that Lgr5 + CSCs initiate and maintain metastasis, but tumor cells that escape the primary tumor and disseminated cells found in the blood circulation are overwhelmingly Lgr5- cells. Lgr5- cells were seeded in the metastatic site, and Lgr5 + cells appeared in the metastatic site after development period. When Lgr5 + cells are lost, Lgr5- cells replenish the eliminated Lgr5 + cells by dedifferentiation
Dynamic phenotypic properties of stem cells
Many recent studies have looked into the relationships between CSCs and non-CSCs, as well as the role of cellular plasticity in CRC with metastasis. CSCs, as the tumor’s clonogenic core, have specific markers such as leucine-rich repeat-containing G-protein-coupled receptor 5 (LGR5). Because selective depletion of colon Lgr5 + CSCs prevented liver metastasis, the experimental evidence has proven that colon cancer-derived liver metastasis depends on a specific subpopulation of Lgr5 + cells. However, once the Lgr5 + cell consumption was stopped, the tumor regenerated rapidly, indicating that the tumor cannot be eliminated by ablating Lgr5 + cells. A study further discovered that colorectal tumors were maintained by proliferative Lgr5 − cells. Lgr5 − cells replenished the Lgr5 + cell pools to promote tumor growth. Furthermore, other experiments revealed a dynamic phenotype of Lgr5 cells that showed that Lgr5 re-expressed after arriving the support microenvironmrnt of the stem cell niche, but the most disseminated and seeding cells are Lgr5 − rather than Lgr5 + . Finally, both Lgr5 − and Lgr5 + cells were observed enriched in liver metastasis, and Lgr5 + cells were required for metastatic tumor growth, indicating that cellular plasticity is a deterministic step in the metastasis process [81] (Fig. 3D).
The phenotypic properties of stem cells (such as CD44) [82] and their interaction with the microenvironment remain unknown. Fixed CSCs and migrating CSCs (MCSCs) are the two supposed types of CSCs. Gao et al. identified organ-specific metastatic MCSCs in human CRC. CD110 + cells have stem cell characteristics that promote liver metastasis, which may be used as a surface marker of liver-specific metastasis [83]. Intestinal stem cells (ISC) located at the bottom of the mucosal invagination maintain intestinal tissue regeneration. EphB2 receptors were highly expressed in ISC, and EphB2 + ISC-like tumor cells promoted CRC recurrence after primary tumor excision in immunocompromised mice [84]. Compared with normal tissues, HIST2H2BF protein was overexpressed in CRC tissues and was associated with a poor prognosis in the patients. HIST2H2BF may promote the development of stem cell-like properties by activating the Notch pathway, indicating its therapeutic potential [85]. Similarly, Wang et al. discovered BMI-1 expressed on the surface of cancer cells in primary colorectal tumors and played an essential role in the self-renewal of CRC stem cells. And because inhibiting BMI-1 with the small molecule compound QW24 can reduce CRC metastasis to the liver, BMI-1 may be used as a new therapeutic target for CRLM [86].
Microenvironment factors associated with CRC liver metastasis: pre-niche, CAFs, and inflammation/immune response
The tumor microenvironment includes innate and adaptive immune cells, stromal cells, endothelial cells, and cancer-associated fibroblasts. Since crosstalk between tumor cells and immune cells ultimately results in an environment that promotes tumor growth and metastasis, the tumor microenvironment is closely related to tumor formation and metastasis [87]. CRLM is frequently regarded as a late-stage random event. And it is believed that metastatic clones emerge late and are formed by the long-term evolution of primary tumor cells. Then the disseminated cancer cells circulate through the portal vein into the liver [88]. However, new evidence suggested that tumor metastasis may occur earlier than expected, which is influenced by both the intrinsic factors of tumor cells and the tumor microenvironment, such as pre-niche formation [10], whose related molecular and cellular events also help to explain the organotropism of CRC metastasis [24, 89]. The appearance of invisible micrometastases (1–2 mm) is caused by disseminated tumor cells choosing to enter a dormant period in the new microenvironment, influenced by molecules and cells. FBX8 is a member of the F-box protein family and directly binds to proteins such as HIF-1, CDK4, and C-myc via its Sec7 domain, promoting the dormancy of liver metastatic CRC cells [90]. Targeting dormant cells to inhibit colonization provides a new theory for treating CRLM.
Pre-niche formation in liver metastases
Seed-soil theory and pre-niche theory both support the idea that cancer metastasis is not random but rather organ-specific [91]. Some researchers have found that primary CRC tumors can promote the formation of hepatic pre-niches by releasing miRNA-containing exosomes [92]. Furthermore, the primary tumor’s pro-inflammatory cytokines, chemoattractants, and angiogenic factors are thought to initiate the formation of the pre-niche [93], recruiting and mobilizing bone marrow-derived cells, and marrow-derived granulocytic MDSCs to the target metastasis site. MDSCs have been demonstrated to contribute to inflammation-associated tumorigenesis and metastasis through multiple mechanisms [94]; Wang et al. discovered that primary CRC tumors secrete VEGF-A, which stimulated macrophages to secrete CXCL1. CXCR2 is the CXCL1 receptor found on the surface of MDSC. CXCL1 overexpression attracted MDSC to the metastatic liver site. Targeting CXCR2 on MDSCs in the pre-niche could be a new therapeutic or preventive strategy for CRC metastasis [28]. Lin et al. discovered that an S1PR1-STAT3-IL6-MDSCs axis may be involved in promoting CRC liver metastasis and that MDSCs can form a “pre-metastasis niche” for CRLM. In tumor cells and the tumor microenvironment, sphingosine 1-phosphate receptor 1 (S1PR1) was involved in the continuous activation of signal transducer and activator transcription 3 (STAT3). Increased S1PR1 protein expression activated STAT3 and increased IL-6 expression; IL-6, in turn, activated STAT3 and recruited MDSCs to the liver’s pre-niche. STAT3 was a key transcriptional effector of the signaling pathway dependent on S1PR1, promoting CRC cell growth and liver metastasis. As a result, inhibiting STAT3/S1PR1 signaling in CRC cell can reduce IL-6 expression and myeloid cells infiltration, thereby preventing metastasis [10]. Exosomes play a critical role in the formation of the pre-metastasis niche. Zhao et al. discovered that miR-934 was overexpressed in CRC patients with liver metastasis and was linked to a poor prognosis. miR-934 in exosomes of primary CRC cells was transferred to macrophages, where it induced polarization of M2 macrophages by down-regulating the expression of PTEN and activating the PI3K/AKT signaling pathway. Notably, CXCL13 secretion by polarized M2 macrophages can trigger the CXCL13/CXCR5/NFB/p65/miR-934 positive feedback loop in metastatic CRC tumor cells. Crosstalk between CRC cells and tumor-associated macrophages (TAM) promotes CRLM and induces pre-metastasis niche formation, suggesting a potential treatment strategy for CRLM [27]. Kupffer cells (KCs) were also shown to be involved in the pre-niche formation process of CRLM, and the hypoxic microenvironment induced the release of exosomal miR-135a-5p from primary CRC lesions. KCs phagocytosed exosomes of miR-135a-5p in the blood circulation and transferred to the liver, promoting CRC liver metastasis [30]. The exosomal protein ANGPTL1 released by CRC tumors was mainly taken up by KCs. ANGPTL1 down-regulated the expression of MMP9 and impeded vascular leakiness in the pre-niche site to alleviate CRC liver metastasis. [29]
CAFs and molecular mechanisms related to liver metastasis of CRC
The tumor microenvironment is made up of tumor stroma and tumor cells, and their interaction promotes cancer growth and metastasis [95]. Cancer-associated fibroblasts (CAFs) have been shown to interact with cancer cells to promote tumor progression. Endoglin is highly expressed on endothelial cells and is required for vascular development, and it is also specifically expressed on the surface of other stromal cells, such as CAFs. The bone morphogenetic protein (BMP)-9 binded to endoglin on CAFs, causing Smad1 phosphorylation and promoting CAFs invasion. TRC105, a neutralizing antibody, can inhibit Smad1 phosphorylation by preventing BMP-9 binding to endoglin, resulting in inhibition of CAFs invasion in vitro and reducing the metastatic spread of CRC cells in the liver, indicating CAFs are also potential therapeutic targets [96]. Furthermore, CAFs also contribute to the EMT, stemness, and metastasis processes by secreting exosomes [97]. J. L. Hu and colleagues discovered that miR-92a-3p-containing exosomes secreted by CAFs in the tumor microenvironment were important mediators of communication between CRC tumor cells and stromal cells. CAFs-exosomal miR-92a-3p was delivered to tumor cells to increase its amount in the cytoplasm. miR-92a-3p bound directly to the target genes of FBXW7 and MOAP1, suppressing their expression via the Wnt/- βcatenin signaling pathway and inhibiting CRC cell EMT and metastasis. Using miR-92a-3p or blocking the function of CAFs to secrete exosomes may be an alternative treatment for CRC metastasis [98]. Although CAFs in the tumor microenvironment has a significant impact on CRLM, the origin of CAFs is unknown. Hao-Xiang Tan et al. discovered that in the environment of CRLM, CAFs were induced by SDF-1. SDF-1 was released by liver-specific pericytes and hepatic stellate cells (HSCs). HSCs were activated by tumor-derived CXCR4 and differentiated into CAFs via the SDF-1-CXCR4-TGF-β pathway to promote liver metastasis. By inhibiting CAFs differentiation via the CXCR4/TGF-β1 pathway, a clinical strategy for anti-metastatic therapy may be developed [99].
Immune/inflammation response in the tumor microenvironment
The two cores of the tumor microenvironment are immunity and inflammation, but the relationship between the two is unclear. Inflammation promotes tumor development; persistent inflammatory cells and factors can transform the tumor-related inflammatory microenvironment into an immunosuppressive microenvironment, promoting tumor progression and metastasis [100].
Inflammation promotes metastasis
Influential microenvironmental factors may act as metastasis initiators in cancer cells, allowing them to spread to distant organs and establish metastasis at a secondary site. The cytoplasmic membrane-bound pattern recognition receptor nucleotide-binding oligomerization domain 1 (NOD1) receptor can recognize microorganisms and mediate inflammation. Under inflammatory stimulation, NOD1 promoted the metastatic phenotype of CRC cells via the p38 MAPK pathway. It enhanced the adhesion and metastasis of CRC cells to the liver sinusoids, as Henry Y. et al. It is demonstrated that NOD1 can be a novel target of inflammation-mediated cancer metastasis [101]. Wenjing et al. investigated a transcription program mediated by the inflammatory environment. GFI1, a six-zinc finger transcription repressor, was significantly down-regulated after CRC cells were treated with LSMCM (LPS-stimulated Monocyte Conditioned Medium) for 24 h, and the TGFβ signaling pathway was responsible for the down-regulation of GFI1. LSMCM activated the GFI1-STAT3-EP2 signaling pathway, promoting EMT, migratory, and invasive behavior in CRC cells, indicating that inflammation promotes metastatic spread [102].
Inflammation results in immunosuppression and promotes metastasis
As the most common immune cells in the tumor microenvironment, macrophages play a critical regulatory role in the progression of inflammation and tumor metastasis. Circulating monocyte precursors are recruited to tumor sites, infiltrating tumors, termed TAMs. Macrophages have diverse phenotypes and functions, and at least two phenotypes of TAMs are involved in tumor promotion. Classically-activated (M1) macrophages secrete pro-inflammatory factors and pronounced anti-tumor activity, but the alternatively-activated (M2) macrophages can suppress the immune system by forming a barrier around invasive cells and establishing a “microenvironment” in which tumor cells can escape immune surveillance and metastasize. The intrinsic plasticity of TAMs controlled the balance between tumor-suppressing and tumor-promoting activities. [54]. Li et al. discovered that Ndrg2 deficiency increases the activation of the NF-κB pathway in TAMs, which regulated TAMs polarization toward the tumor suppressor phenotype (M1). Besides hepatic macrophages, Kupffer cells, a subtype of bone marrow-derived macrophages, are also responsible for inhibiting CRLM [103]. As previously stated, certain secreted factors (such as exosomes) influence distant tumor metastasis by regulating TAM polarization, forming a regulatory loop between tumor cells, TAMs, and the microenvironment [103]. Dong Wang et al. discovered that CXCR4 was overexpressed in CRC tissues and patients with high CXCR4 expression have a higher risk of metastasis. miR-25-3p, miR-130b-3p, and miR-425-5p were highly expressed in CRC cells activated by the CXCL12/CXCR4 axis. These miRNAs secreted by CRC cells were encapsulated in exosomes and delivered to TAMs. TAMs with increased miR-25-3p, miR-130b-3p, and miR-425-5p, promoted their M2 polarization, and PTEN acted as a downstream target to activate the PI3K/Akt signaling pathway. M2 polarization of macrophages enhanced the CRC cells’ metastatic capacity, promoting CRC liver metastasis [104]. The ability of monocytes to be recruited into the tumor microenvironment and differentiate into TAMs has intrigued researchers. According to research, the heterogeneity of myeloid cell phenotype appeared to be selective for tumors and organs, implying that bone marrow cells with different markers drive metastasis to other sites. A study, for example, confirmed that CRC expresses CCL2 to mobilize inflammatory monocytes (IM) from the bone marrow via the CCL2/CCR2 chemokine axis. CCL2 were highly expressed in metastatic CRC liver tumors compared with normal liver tissue, and CCL2 mediated bone marrow-derived circulating CCR2 + IM (CD11b + , CD14 + , CCR2 +) migration to the site of liver metastasis in CRC patients, where they differentiated into immunosuppressive TAMs (CD115 + , CD14 + , CD68 +) and supported metastatic tumor growth. As a result, the ability of CCR2 + IM to decrease CD8 + T cells and increase regulatory T-cells (CD4 + , FoxP3 + , CD25 +) may be a potential mechanism for its promotion of tumor growth at the metastatic site. In addition, it has recently been discovered that blocking CCR2 produced an anti-tumor immune response at the metastatic site, providing a new target for clinical treatment [105]. According to a recent single-cell analysis of autologous samples of liver metastasized CRC, suppressor cells such as dendritic cells (DC3s) and SPP1 + macrophages were predominant in liver metastasis, forming a more immunosuppressive environment, resulting in the pro-metastasis effect [106]. In addition, one study found that the metastatic liver microenvironment had a marked spatial reprogramming effect on immunosuppressive cells such as MRC1 + CCL18 + M2-like macrophages. By defining the immune landscape of CRLM using single-cell RNA-seq and spatial transcriptomics, MRC1 + CCL18 + macrophages exhibited the highest metabolic activity at metastatic sites. This immune reprogramming may be induced by chemotherapy (Fig. 4) [107].
Immune/inflammation response in the tumor Microenvironment. M2 macrophages, inflammatory monocytes, bone marrow-derived macrophages, and dendritic cells cause an immunosuppressive microenvironment. CRC cells with high expression of CXCR4 were activated through the CXCL12/CXCR4 axis and secreted exosomal miRNAs (miR-25-3p, miR-130b-3p and miR-425-5p) to promote M2 polarization of M1 macrophages; CRC tissues with high Expressing CCL2 recruited inflammatory monocytes to metastatic sites through the CCL2/CCR2 chemokine axis promoting immunosuppression; suppressor cells such as myeloid-derived macrophages and dendritic cells were predominant during CRLM process
Biomarkers related to early detection, therapeutic efficacy prediction, and recurrence risk of liver metastasis from CRC
The unpredictability of distant metastasis limits treatment options as well. Thus, identifying biomarkers for liver metastasis and treating CRC will benefit the patients [108]. In Table 2, all therapeutic targets mentioned are summarized. Table 3 summarizes the potential diagnostic, predictive, or prognostic biomarkers for CRLM and their strengths and limitations are summarized.
Mutated genes as biomarkers
In recent years, mutated and differentially expressed genes have been identified as potential predictive and prognostic biomarkers for CRLM. The incidence of BRAF mutation in metastatic CRC is less than 10%, but BRAF V600E mutation is thought to be a relevant therapeutic target for metastatic CRC management. Although the data showed that CRC patients with KRAS mutations had a higher risk of liver metastasis, the difference was not statistically significant, and the risk of liver metastasis in patients with BRAF mutations was lower than that in patients with Wt BRAF. As a result, there was no clear link between KRAS and BRAF mutations and liver metastasis in patients with CRC [109]. A multivariate Cox regression model has been used in studies to predict genes with potential prognostic significance after CRLM resection. Mutations in the TP53, KRAS, PIK3CA, BRAF, and SMAD families have been identified as prognostic markers for overall survival after CRLM liver surgery. The SMAD family of genes, which included SMAD2, SMAD3, and SMAD4, had the best predictive value for poor tumor outcomes [110].
Differentiated expressed molecules as biomarkers
ERCC1 may serve as a predictive biomarker for the effectiveness of chemotherapy. The expression of ERCC1 was primarily examined in the relationship between CRC and liver metastasis. There was no clear evidence of a link between ERCC1 expression and concurrent/metachronous liver metastasis, and no evidence of a link between the primary tumor and liver metastasis was found. A study examines ERCC1 protein in primary colorectal cancer and corresponding liver metastases. Good reproducibility of IHC expression benefited ERCC1 as a predictive biomarker for platinum-based chemotherapy. However, no consistency has been observed when comparing IHC expression in primary tumors and matched liver metastases. It means ERCC1 expression of liver metastases can not be assessed in archived material (primary tumor biopsies), implying the need to use tissue from actual tumor burden to evaluate ERCC1 expression [111, 112]. The EMX2 gene belongs to the family of homeobox genes. The Cox proportional hazard model was used to examine the distant metastasis rate of EMX2 overexpression tumor patients and EMX2 low expression tumor patients. The results showed that there is a significant correlation between tumor EMX2 expression down-regulation and CRLM [113].
chromosome 4 deletion
A study published in 2013 used array-based comparative genomic hybridization (aCGH) to examine genetic changes in metastatic site samples from two groups of CRC patients with liver metastases who had survival rates of less than five years and more than five years. Through survival analysis, metastatic tissue samples with whole chromosome 4 loss indicated a higher survival rate in the patients, implying that the absence of chromosome 4 may reduce the risk of recurrence of CRLM [114].
Non-coding RNAs as biomarkers
Non-coding RNAs regulate the development and metastasis of CRC and have a significant effect on cancer biology. Moreover, the primary tumors release a variety of exosomes containing various LncRNAs to promote the formation of the anterior liver niche, molecules released by exosomes can be detected by liquid biopsy, and LncRNAs can be used as an effective biomarker to predict early metastasis, especially micrometastases that cannot be detected by pathological methods [115]. Li Sun et al. discovered, for example, that miR-122, packaged into exosomes, was significantly overexpressed in CRC patients with liver metastasis compared with CRC patients without liver metastasis [116]. Compared with primary CRC tissues, circRNA_0001178 and circRNA_0000826 were significantly upregulated in CRC liver metastasis tissues. And it was found that circ102049 was highly expressed in primary CRC tumors with liver metastasis and closely correlated with the prognosis of CRC patients. These non-coding RNAs may serve as potential diagnostic biomarkers for CRC liver metastasis [117, 118].
miRNAs
miRNA is a type of short single-stranded endogenous non-coding RNA that can bind to the 3'-untranslated region (3'-UTR) of mRNA to regulate transcription, resulting in translational inhibition. When miRNA expression in the plasma of CRC patients with and without liver metastases was compared, miR-21, miR-203, and miR-210 were significantly dysregulated in patients with liver metastatic CRC, indicating their roles as early detection biomarkers. Significantly downregulated miR-19a, miR-203, and miR-21 have been shown to have the ability to predict patients at high risk of recurrence [119].
LncRNAs
LncRNAs, typically > 200 bp in length, are natural prognostic biomarkers. Yiya and GAS5 expression were upregulated at liver metastasis compared with the primary CRC, and CRC patients with high expression of Yiya and GAS5 had a higher risk of liver metastasis. Yiya’s expression was found to be an independent risk factor for liver metastasis in early-stage patients using Cox proportional-risk regression analysis, suggesting that it could be the most potential predictive biomarker for liver metastasis [120]. The most crucial catalytic enzyme for mRNA methylation modification has been identified as METTL4, a major m6A methyltransferase. METTL14’s downstream target was lncRNA XIST, according to Xiao Yang et al. Down-regulation of oncogenic lncRNA XIST via METTL14 inhibited CRC cell metastasis and growth; thus, METTL14 may be a promising therapeutic target [121].
CircRNA
Hanchen Xu et al. discovered a difference in circRNA expression between tissue samples from CRC patients with and without liver metastasis. It was discovered that circRNA_0001178 and circRNA_0000826 might be potential diagnostic biomarkers for CRLM by constructing a circRNA-miRNA-mRNA network [117].
Other prognostic biomarkers of colorectal cancer liver metastasis
Some small-scale cohort studies have discovered that various types of circulating molecules can be used as potential biomarkers for prognostic prediction of CRLM. However, their actual clinical applications were still minimal [122]. There was evidence that apoptotic circulating tumor cell (CTC) was a possible prognostic marker for CRC metastasis to the liver or other metastases. Among the 17 patients with metastatic CRC included in the study, 6/11 had apoptotic CTCs in the liver, and 1/9 had apoptotic CTCs in other metastatic sites [123]. The expression of leptin and the chitinase activity can predict the ability of CRC to metastasize. Leptin expression was significantly higher in metastatic CRC tissue than in primary tumors without liver metastasis, indicating that increased leptin expression was associated with a higher risk of liver metastasis. Chitinase is an inflammatory biomarker that causes inflammatory factors to be produced. According to the findings of multi-factor regression analysis, serum chitinase activity was an independent risk factor for predicting liver metastasis, and the power was superior to traditional CEA. This conclusion was consistent with the view that inflammation promotes cancer metastasis [124].
Challenges and perspectives
CRC liver spread is difficult to detect very early, with limited treatment options and a poor prognosis, resulting in median survival of about six months for CRLM patients [125]. There was no clear stratification for patients with resectable metastatic tumors. Fong clinical risk scores included multiple variables that stratify patients with distant metastasis into high-risk (5-year OS: 47%) and low-risk (5-year OS: 24%) groups, but they lack validation from a large cohort of neoadjuvant chemotherapy patients, limiting their accuracy [126, 127]. The development of adjuvant systemic therapy has significantly improved the clinical outcome of patients with stage IV CRLM [7, 128]. CRC heterogeneity presents significant challenges in disease diagnosis and treatment; genetic/epigenetic alterations may be a driving force at the genome level, giving rise to subclones with potential metastatic spread. Furthermore, the non-genetic diversity of phenotypic and behavioral states of cancer cell clones has been proposed as a source of intra-tumor heterogeneity, which is influenced by changing microenvironments [129]. Although many studies have identified one or more genes, molecules, and pathways related to metastasis, their influence on phenotypes of metastatic advantages has yet to be investigated.
CRC metastasis may occur at the stage of tumorigenesis. In recent years, genomic analysis in the difference between primary tumors and metastatic sites has challenged the notion that CRC metastasis is an early event. In addition to the evidence-based genomic analysis, the “seed-soil” theory and the formation of pre-niches also suggest that metastasis occurs at an early stage and that specific molecules secreted by the primary tumor predispose to the occurrence of CRC in the liver. “Parallel evolution” discovered that the genetic divergence between the primary tumor and the metastatic site is greater than that of the “linear evolution model”, implying that in CRC, some metastatic clones leave the primary site at an early stage and evolve further within the metastases [33]. The central question is the mechanisms underlying the formation of metastatic clones, like whether metastatic capacity is an advantage that has been selected during tumor evolution in the primary tumor or just an unselected, incidental consequence of tumor progression [43]. Because only a few “fit” subclones among all clones can survive and give rise to metastasis in a distant organ, several cancer initiation evolution models have been proposed to explain metastatic progression, and the feasibility of these models in explaining the development of metastasis is still being explored [122]. Tumor initiation is thought to be driven by the sequential stepwise acquisition of genetic alternations involving driver gene mutations in the classical “linear progression model” (Note: this should be distinguished from the “linear progression model” in metastatic evolution). A series of selective sweeps can fixed new driver mutations leading to clones expanding and providing strong selective advantages. The extent to which the selection effect promotes clonal diversity and generates dominant clones, on the other hand, is still debatable. The “Neutral evolution model,” which contradicts classic Darwinian evolution, hypothesizes that “random drift, rather than non-random selection based on reproductive fitness, results in the generation of new clones.” Rather than the sequential clonal evolution (“linear progression model”), the “Big Bang” model proposes that heterogeneous cell populations with potential metastasis advantages are simply “incidental byproducts” of the primary tumor formation process. Clonal selection has no effect on tumor progression, and these intermixed subclones have similar fitness and grow at the same rate [47, 130]. It has been discovered that the “Big Bang” model and its subsequent “Neutral evolution model” best explain the emergence of metastatic clones in CRC.
The role of molecular genetic changes in metastatic evolution is still largely unknown. Here, we found that epigenetic changes were more likely than genetic changes to be the direct driver of metastasis, and certain gene mutations were associated with specific metastasis patterns. However, studies at the genome level have demonstrated that metastatic CRC is of monoclonal and polyclonal origin and follows both monophyletic and polyphyletic seeding patterns. By identifying critical genes in metastatic evolution, it may be possible to improve the ability to determine whether CRC has metastasized. Recent research on the genome landscape of patients with metastatic CRC has revealed a link between mutant genes and metastasis patterns. S-C evolutionary pattern was associated with KRAS, SYNE1, CACNA1H, PCLO, FBXL2, and DNAH11 [50]. However, no universal metastasis-specific driver genes have been identified in CRC comparative genome studies. The epigenetic alterations associated with CRLM observed in the studies primarily included DNA methylation, non-coding gene regulation, and chromosome remodeling [40, 131].
Clinical evidence indicates that reasonable combination strategies are essential in the clinical treatment and efficacy of CRC liver metastases [132]. The tumor immune microenvironment influences the progression of malignant tumors. For example, immune checkpoint proteins (PD-L1, CTLA-4, TIM3), the main targets of immunotherapy, are present in exosomes and can promote tumor progression and metastasis [133, 134]. Some immune infiltrations can suppress adaptive immunity. The liver- metastasized lesion contains a complex tumor microenvironment that is easier to produce an immunosuppressive microenvironment than other organs to promote tumor metastasis. As a result, the immune microenvironment is regarded as a promising therapeutic target for CRLM [135]. For example, combining immune checkpoint blocking therapy with conventional therapy opens up new treatment options. According to one study, increased circulating fms-related tyrosine kinase 3 ligand (FLT3LG) caused by neoadjuvant cytotoxic treatment in rectal cancer patients activated the anti-tumor immune response. Functionally active adaptive immune cells may eliminate microscopic tumor cells, lowering the risk of tumor metastasis [136]. The combination of IL-12 gene therapy and chemotherapy has shown the ability to transform the metastatic microenvironment into a more immunogenic phenotype by reducing Treg, MDSC, and M2 macrophages [132].
Conclusions
This review summarized the main perspectives related to metastasis, especially focusing on the factors influencing organ-specific metastasis. Beginning with general metastasis principles to understand the process of metastasis, we summarize and integrate the hypothesis of the metastatic evolution patterns (including origin of metastatic cloning and the pattern of metastatic seeding), genetic/epigenetic changes related to CRLM, molecular and cellular mechanisms, and biomarkers providing ideas for challenging metastatic CRC. The phenotypic change of CSCs is also one of the essential intrinsic factors for CRLM, which is a prerequisite for metastasis formation. For extrinsic factors of CRLM, cell/molecular interactions between tumor cells and the tumor microenvironment have been shown to mediate metastasis. Among them, CAFs are critical cellular components that induce CRLM, and the establishment of an immunosuppressive microenvironment has been established in the metastatic site. The relationship between inflammation and immune response needs to be further explored. Several central questions remain to be clarified: (1) Whether and what kind of the evidence based on the genome level can predict the potential of CRC to develop liver metastases in the early stage and predict the efficacy of immunotherapy, to provide a reference for clinical treatment strategies? (2) The dynamic evolution of cancer stem cell phenotypes is a prerequisite for CRLM. How can it be an effective therapeutic target or a biomarker for early detection? (3) Since macrophages and CAFs play an essential role in CRLM, whether they affect the organ tropism of metastasis? What is possible to detect CRLM early by detecting cell phenotypes in the tumor microenvironment? And whether can we predict a state of suppressive immune microenvironment for a tumor? Exploration of these issues will provide helpful information for early detection and appropriate treatment strategies for CRLM. Finally, we summarize the current therapeutic targets for CRLM, as well as diagnostic/prognostic biomarkers, and analyze the strengths and limitations of these methods, hoping to provide valuable clues for the diagnosis and treatment of CRLM.
Availability of data and materials
All data included in this study are available upon request by contact with the corresponding author.
Abbreviations
- CRC:
-
Colorectal cancer
- CRLM:
-
Colorectal cancer liver metastasis
- SNVs:
-
Single nucleotide variations
- sSNVs:
-
Somatic single nucleotide variations
- LM:
-
Liver metastasis
- CCF:
-
Cancer cell fractions
- EMT:
-
Epithelial-mesenchymal transition
- CNVs:
-
Copy number variations
- MMPs:
-
Matrix metalloproteinases
- TKI:
-
Tyrosine kinase inhibitor
- C–C:
-
Clonal- Clonal
- S-C:
-
Subclonal- Clonal
- 0-C:
-
None- Clonal
- HLA:
-
Human leukocytes antigens
- LOH:
-
Loss of heterozygosity
- CGI:
-
CpG island
- HGF:
-
Hepatic growth factor
- m6A:
-
N6-methyladenosine
- Circ RNAs:
-
Circular RNAs
- LncRNAs:
-
Long non-coding RNAs
- CYTOR:
-
Cytoskeleton regulator RNA
- CSC:
-
Cancer stem cell
- MCSCs:
-
Migrating CSCs
- TA:
-
Transient amplification
- TICs:
-
Tumor- initiating cells
- LGR5:
-
LT-TICs: long term tumor- initiating cells G-protein-coupled receptor 5
- ISC:
-
Intestinal stem cell
- MDSCs:
-
Myeloid- derived suppressor cells
- S1PR1:
-
Sphingosine 1-phosphate receptor 1
- STAT3:
-
Signal transducer and activator of transcription 3
- CAFs:
-
Cancer-related fibroblasts
- BMP:
-
Bone morphogenetic protein
- NOD:
-
Nucleotide-binding oligomerization domain 1
- LSMCM:
-
LPS-stimulated Monocyte Conditioned Medium
- IM:
-
Inflammatory monocytes
- DC:
-
Dendritic cell
- aCGH:
-
Array-based comparative genomic hybridization
- 3'-UTR:
-
3'-Untranslated region
- lncRNAs:
-
Long non-coding RNAs
- CTC:
-
Circulating tumor cell
- circRNAs:
-
Circular RNAs
- pre-Micro RNA:
-
Pre-miRNAs
- FLT3LG:
-
Fms-related tyrosine kinase 3 ligand
References
Riihimaki M, Hemminki A, Sundquist J, Hemminki K. Patterns of metastasis in colon and rectal cancer. Sci Rep. 2016;6:29765.
Sawatzki M, Guller U, Gusewell S, Husarik DB, Semela D, Brand S. Contrast-enhanced ultrasound can guide the therapeutic strategy by improving the detection of colorectal liver metastases. J Hepatol. 2021;74(2):419–27.
Tamas K, Walenkamp AM, de Vries EG, van Vugt MA, Beets-Tan RG, van Etten B, de Groot DJ, Hospers GA. Rectal and colon cancer: not just a different anatomic site. Cancer Treat Rev. 2015;41(8):671–9.
Zarour LR, Anand S, Billingsley KG, Bisson WH, Cercek A, Clarke MF, Coussens LM, Gast CE, Geltzeiler CB, Hansen L, et al. Colorectal cancer liver metastasis: evolving paradigms and future directions. Cell Mol Gastroenterol Hepatol. 2017;3(2):163–73.
Baba K. Successful treatment of conversion chemotherapy for initially unresectable synchronous colorectal liver metastasis. World J Gastroenterol. 2015. https://doi.org/10.3748/wjg.v21.i6.1982.
Cherradi S, Ayrolles-Torro A, Vezzo-Vie N, Gueguinou N, Denis V, Combes E, Boissiere F, Busson M, Canterel-Thouennon L, Mollevi C, et al. Antibody targeting of claudin-1 as a potential colorectal cancer therapy. J Exp Clin Cancer Res. 2017;36(1):89.
Adam R, Kitano Y. Multidisciplinary approach of liver metastases from colorectal cancer. Ann Gastroenterol Surg. 2019;3(1):50–6.
Urosevic J, Garcia-Albeniz X, Planet E, Real S, Cespedes MV, Guiu M, Fernandez E, Bellmunt A, Gawrzak S, Pavlovic M, et al. Colon cancer cells colonize the lung from established liver metastases through p38 MAPK signalling and PTHLH. Nat Cell Biol. 2014;16(7):685–94.
Langley RR, Fidler IJ. The seed and soil hypothesis revisited–the role of tumor-stroma interactions in metastasis to different organs. Int J Cancer. 2011;128(11):2527–35.
Lin Q, Ren L, Jian M, Xu P, Li J, Zheng P, Feng Q, Yang L, Ji M, Wei Y, et al. The mechanism of the premetastatic niche facilitating colorectal cancer liver metastasis generated from myeloid-derived suppressor cells induced by the S1PR1-STAT3 signaling pathway. Cell Death Dis. 2019;10(10):693.
Wang M, Su Z, Amoah Barnie P. Crosstalk among colon cancer-derived exosomes, fibroblast-derived exosomes, and macrophage phenotypes in colon cancer metastasis. Int Immunopharmacol. 2020;81: 106298.
Chaffer CL, Weinberg RA. A perspective on cancer cell metastasis. Science. 2011;331(6024):1559–64.
Suhail Y, Cain MP, Vanaja K, Kurywchak PA, Levchenko A, Kalluri R. Kshitiz: systems biology of cancer metastasis. Cell Syst. 2019;9(2):109–27.
Ganesh K, Massague J. Targeting metastatic cancer. Nat Med. 2021;27(1):34–44.
Hanahan D, Weinberg RA. Hallmarks of cancer: the next generation. Cell. 2011;144(5):646–74.
Bhattacharya R, Panda CK, Nandi S, Mukhopadhyay A. An insight into metastasis: random or evolving paradigms? Pathol Res Pract. 2018;214(8):1064–73.
Zheng W, Wu F, Fu K, Sun G, Sun G, Li X, Jiang W, Cao H, Wang H, Tang W. Emerging mechanisms and treatment progress on liver metastasis of colorectal cancer. Onco Targets Ther. 2021;14:3013–36.
Weidle UH, Birzele F, Kruger A. Molecular targets and pathways involved in liver metastasis of colorectal cancer. Clin Exp Metastasis. 2015;32(6):623–35.
Obenauf AC, Massague J. Surviving at a Distance: Organ-Specific Metastasis. Trends Cancer. 2015;1(1):76–91.
Budczies J, von Winterfeld M, Klauschen F, Bockmayr M, Lennerz JK, Denkert C, Wolf T, Warth A, Dietel M, Anagnostopoulos I, et al. The landscape of metastatic progression patterns across major human cancers. Oncotarget. 2015;6(1):570–83.
Rubin MA. Insights into the mechanism of organ-specific cancer metastasis. Cancer Discov. 2014;4(11):1262–4.
Klein CA. Selection and adaptation during metastatic cancer progression. Nature. 2013;501(7467):365–72.
Hoshino A, Costa-Silva B, Shen TL, Rodrigues G, Hashimoto A, Tesic Mark M, Molina H, Kohsaka S, Di Giannatale A, Ceder S, et al. Tumour exosome integrins determine organotropic metastasis. Nature. 2015;527(7578):329–35.
Yang L, Li T, Shi H, Zhou Z, Huang Z, Lei X. The cellular and molecular components involved in pre-metastatic niche formation in colorectal cancer liver metastasis. Expert Rev Gastroenterol Hepatol. 2021;15(4):389–99.
Li C, Pan B, Liu X, Qin J, Wang X, He B, Pan Y, Sun H, Xu T, Xu X, et al. Long intergenic non-coding RNA LINC00485 exerts tumor-suppressive activity by regulating miR-581/EDEM1 axis in colorectal cancer. Aging. 2021;13(3):3866–85.
Zhou H, Liu Z, Wang Y, Wen X, Amador EH, Yuan L, Ran X, Xiong L, Ran Y, Chen W, et al. Colorectal liver metastasis: molecular mechanism and interventional therapy. Signal Transduct Target Ther. 2022;7(1):70.
Zhao S, Mi Y, Guan B, Zheng B, Wei P, Gu Y, Zhang Z, Cai S, Xu Y, Li X, et al. Tumor-derived exosomal miR-934 induces macrophage M2 polarization to promote liver metastasis of colorectal cancer. J Hematol Oncol. 2020;13(1):156.
Wang D, Sun H, Wei J, Cen B, DuBois RN. CXCL1 Is critical for premetastatic niche formation and metastasis in colorectal cancer. Cancer Res. 2017;77(13):3655–65.
Jiang K, Chen H, Fang Y, Chen L, Zhong C, Bu T, Dai S, Pan X, Fu D, Qian Y, et al. Exosomal ANGPTL1 attenuates colorectal cancer liver metastasis by regulating Kupffer cell secretion pattern and impeding MMP9 induced vascular leakiness. J Exp Clin Cancer Res. 2021;40(1):21.
Sun H, Meng Q, Shi C, Yang H, Li X, Wu S, Familiari G, Relucenti M, Aschner M, Wang X, et al. Hypoxia-inducible exosomes facilitate liver-tropic premetastatic niche in colorectal cancer. Hepatology. 2021;74(5):2633–51.
Shao S, Zhu Y, Meng T, Liu Y, Hong Y, Yuan M, Yuan H, Hu F. Targeting high expressed alpha5beta1 integrin in liver metastatic lesions to resist metastasis of colorectal cancer by rpm peptide-modified chitosan-stearic micelles. Mol Pharm. 2018;15(4):1653–63.
Ganesh K, Basnet H, Kaygusuz Y, Laughney AM, He L, Sharma R, O’Rourke KP, Reuter VP, Huang YH, Turkekul M, et al. L1CAM defines the regenerative origin of metastasis-initiating cells in colorectal cancer. Nat Cancer. 2020;1(1):28–45.
Patel SA, Rodrigues P, Wesolowski L, Vanharanta S. Genomic control of metastasis. Br J Cancer. 2021;124(1):3–12.
Kim TM, Jung SH, An CH, Lee SH, Baek IP, Kim MS, Park SW, Rhee JK, Lee SH, Chung YJ. Subclonal genomic architectures of primary and metastatic colorectal cancer based on intratumoral genetic heterogeneity. Clin Cancer Res. 2015;21(19):4461–72.
Saito T, Niida A, Uchi R, Hirata H, Komatsu H, Sakimura S, Hayashi S, Nambara S, Kuroda Y, Ito S, et al. A temporal shift of the evolutionary principle shaping intratumor heterogeneity in colorectal cancer. Nat Commun. 2018;9(1):2884.
Turajlic S, Swanton C. Metastasis as an evolutionary process. Science. 2016;352(6282):169–75.
Leung ML, Davis A, Gao R, Casasent A, Wang Y, Sei E, Vilar E, Maru D, Kopetz S, Navin NE. Single-cell DNA sequencing reveals a late-dissemination model in metastatic colorectal cancer. Genome Res. 2017;27(8):1287–99.
Chen HN, Shu Y, Liao F, Liao X, Zhang H, Qin Y, Wang Z, Luo M, Liu Q, Xue Z, et al. Genomic evolution and diverse models of systemic metastases in colorectal cancer. Gut. 2022;71(2):322–32.
Chiang AC, Massague J. Molecular basis of metastasis. N Engl J Med. 2008;359(26):2814–23.
Grady WM, Carethers JM. Genomic and epigenetic instability in colorectal cancer pathogenesis. Gastroenterology. 2008;135(4):1079–99.
Testa U, Castelli G, Pelosi E. Genetic alterations of metastatic colorectal cancer. Biomedicines. 2020. https://doi.org/10.3390/biomedicines8100414.
Alderton GK. Tumour evolution: epigenetic and genetic heterogeneity in metastasis. Nat Rev Cancer. 2017;17(3):141.
Vignot S, Lefebvre C, Frampton GM, Meurice G, Yelensky R, Palmer G, Capron F, Lazar V, Hannoun L, Miller VA, et al. Comparative analysis of primary tumour and matched metastases in colorectal cancer patients: evaluation of concordance between genomic and transcriptional profiles. Eur J Cancer. 2015;51(7):791–9.
Priestley P, Baber J, Lolkema MP, Steeghs N, de Bruijn E, Shale C, Duyvesteyn K, Haidari S, van Hoeck A, Onstenk W, et al. Pan-cancer whole-genome analyses of metastatic solid tumours. Nature. 2019;575(7781):210–6.
Nguyen B, Fong C, Luthra A, Smith SA, DiNatale RG, Nandakumar S, Walch H, Chatila WK, Madupuri R, Kundra R, et al. Genomic characterization of metastatic patterns from prospective clinical sequencing of 25,000 patients. Cell. 2022;185(3):563–75.
Joung JG, Oh BY, Hong HK, Al-Khalidi H, Al-Alem F, Lee HO, Bae JS, Kim J, Cha HU, Alotaibi M, et al. Tumor heterogeneity predicts metastatic potential in colorectal cancer. Clin Cancer Res. 2017;23(23):7209–16.
Sottoriva A, Barnes CP, Graham TA. Catch my drift? Making sense of genomic intra-tumour heterogeneity. Biochim Biophys Acta Rev Cancer. 2017;1867(2):95–100.
Loeb LA, Kohrn BF, Loubet-Senear KJ, Dunn YJ, Ahn EH, O’Sullivan JN, Salk JJ, Bronner MP, Beckman RA. Extensive subclonal mutational diversity in human colorectal cancer and its significance. Proc Natl Acad Sci USA. 2019;116(52):26863–72.
Oga T, Yamashita Y, Soda M, Kojima S, Ueno T, Kawazu M, Suzuki N, Nagano H, Hazama S, Izumiya M, et al. Genomic profiles of colorectal carcinoma with liver metastases and newly identified fusion genes. Cancer Sci. 2019;110(9):2973–81.
Li C, Xu J, Wang X, Zhang C, Yu Z, Liu J, Tai Z, Luo Z, Yi X, Zhong Z. Whole exome and transcriptome sequencing reveal clonal evolution and exhibit immune-related features in metastatic colorectal tumors. Cell Death Discov. 2021;7(1):222.
Wang HW, Yan XL, Wang LJ, Zhang MH, Yang CH, Wei L, Jin KM, Bao Q, Li J, Wang K, et al. Characterization of genomic alterations in Chinese colorectal cancer patients with liver metastases. J Transl Med. 2021;19(1):313.
Vasudevan A, Baruah PS, Smith JC, Wang Z, Sayles NM, Andrews P, Kendall J, Leu J, Chunduri NK, Levy D, et al. Single-Chromosomal gains can function as metastasis suppressors and promoters in colon cancer. Dev Cell. 2020;52(4):413–28.
Li H, Wang J, Yi Z, Li C, Wang H, Zhang J, Wang T, Nan P, Lin F, Xu D, et al. CDK12 inhibition enhances sensitivity of HER2+ breast cancers to HER2-tyrosine kinase inhibitor via suppressing PI3K/AKT. Eur J Cancer. 2021;145:92–108.
Pancione M, Giordano G, Remo A, Febbraro A, Sabatino L, Manfrin E, Ceccarelli M, Colantuoni V. Immune escape mechanisms in colorectal cancer pathogenesis and liver metastasis. J Immunol Res. 2014;2014: 686879.
Banerjee R, Smith J, Eccles MR, Weeks RJ, Chatterjee A. Epigenetic basis and targeting of cancer metastasis. Trends Cancer. 2022;8(3):226–41.
Hogg SJ, Beavis PA, Dawson MA, Johnstone RW. Targeting the epigenetic regulation of antitumour immunity. Nat Rev Drug Discov. 2020;19(11):776–800.
Jung G, Hernandez-Illan E, Moreira L, Balaguer F, Goel A. Epigenetics of colorectal cancer: biomarker and therapeutic potential. Nat Rev Gastroenterol Hepatol. 2020;17(2):111–30.
Wang L, Wang E, Prado Balcazar J, Wu Z, Xiang K, Wang Y, Huang Q, Negrete M, Chen KY, Li W, et al. Chromatin remodeling of colorectal cancer liver metastasis is mediated by an HGF-PU 1-DPP4 Axis. Adv Sci. 2021;8(19):e2004673.
Wang S, Gao S, Zeng Y, Zhu L, Mo Y, Wong CC, Bao Y, Su P, Zhai J, Wang L, et al. N6-Methyladenosine reader YTHDF1 Promotes ARHGEF2 translation and RhoA signaling in colorectal cancer. Gastroenterology. 2022;162(4):1183–96.
Chen RX, Chen X, Xia LP, Zhang JX, Pan ZZ, Ma XD, Han K, Chen JW, Judde JG, Deas O, et al. N(6)-methyladenosine modification of circNSUN2 facilitates cytoplasmic export and stabilizes HMGA2 to promote colorectal liver metastasis. Nat Commun. 2019;10(1):4695.
Bleau AM, Redrado M, Nistal-Villan E, Villalba M, Exposito F, Redin E, de Aberasturi AL, Larzabal L, Freire J, Gomez-Roman J, et al. miR-146a targets c-met and abolishes colorectal cancer liver metastasis. Cancer Lett. 2018;414:257–67.
Zheng X, Chen L, Zhou Y, Wang Q, Zheng Z, Xu B, Wu C, Zhou Q, Hu W, Wu C, et al. A novel protein encoded by a circular RNA circPPP1R12A promotes tumor pathogenesis and metastasis of colon cancer via Hippo-YAP signaling. Mol Cancer. 2019;18(1):47.
Yue B, Liu C, Sun H, Liu M, Song C, Cui R, Qiu S, Zhong M. A positive feed-forward loop between LncRNA-CYTOR and Wnt/beta-catenin signaling promotes metastasis of colon cancer. Mol Ther. 2018;26(5):1287–98.
Su R, Wu X, Tao L, Wang C. The role of epigenetic modifications in colorectal cancer metastasis. Clin Exp Metastasis. 2022;39(4):521–39.
Ili C, Buchegger K, Demond H, Castillo-Fernandez J, Kelsey G, Zanella L, Abanto M, Riquelme I, Lopez J, Viscarra T, et al. Landscape of genome-wide DNA methylation of colorectal cancer metastasis. Cancers. 2020. https://doi.org/10.3390/cancers12092710.
Udali S, De Santis D, Ruzzenente A, Moruzzi S, Mazzi F, Beschin G, Tammen SA, Campagnaro T, Pattini P, Olivieri O, et al. DNA methylation and hydroxymethylation in primary colon cancer and synchronous hepatic metastasis. Front Genet. 2017;8:229.
Mahdi MR, Georges RB, Ali DM, Bedeer RF, Eltahry HM, Gabr AHZ, Berger MR. Modulation of the endothelin system in colorectal cancer liver metastasis: influence of epigenetic mechanisms? Front Pharmacol. 2020;11:180.
Zhang KL, Zhu WW, Wang SH, Gao C, Pan JJ, Du ZG, Lu L, Jia HL, Dong QZ, Chen JH, et al. Organ-specific cholesterol metabolic aberration fuels liver metastasis of colorectal cancer. Theranostics. 2021;11(13):6560–72.
Chen Y, Lin Y, Shu Y, He J, Gao W. Interaction between N(6)-methyladenosine (m(6)A) modification and noncoding RNAs in cancer. Mol Cancer. 2020;19(1):94.
Lee J, Hong HK, Peng SB, Kim TW, Lee WY, Yun SH, Kim HC, Liu J, Ebert PJ, Aggarwal A, et al. Identifying metastasis-initiating miRNA-target regulations of colorectal cancer from expressional changes in primary tumors. Sci Rep. 2020;10(1):14919.
Mouillet JF, Donker RB, Mishima T, Cronqvist T, Chu T, Sadovsky Y. The unique expression and function of miR-424 in human placental trophoblasts. Biol Reprod. 2013;89(2):25.
Zhu ED, Li N, Li BS, Li W, Zhang WJ, Mao XH, Guo G, Zou QM, Xiao B. miR-30b, down-regulated in gastric cancer, promotes apoptosis and suppresses tumor growth by targeting plasminogen activator inhibitor-1. PLoS ONE. 2014;9(8): e106049.
Chen T, You Y, Jiang H, Wang ZZ. Epithelial-mesenchymal transition (EMT): A biological process in the development, stem cell differentiation, and tumorigenesis. J Cell Physiol. 2017;232(12):3261–72.
Smith AG, Macleod KF. Autophagy, cancer stem cells and drug resistance. J Pathol. 2019;247(5):708–18.
Miyazaki T, Chung S, Sakai H, Ohata H, Obata Y, Shiokawa D, Mizoguchi Y, Kubo T, Ichikawa H, Taniguchi H, et al. Stemness and immune evasion conferred by the TDO2-AHR pathway are associated with liver metastasis of colon cancer. Cancer Sci. 2022;113(1):170–81.
Dieter SM, Ball CR, Hoffmann CM, Nowrouzi A, Herbst F, Zavidij O, Abel U, Arens A, Weichert W, Brand K, et al. Distinct types of tumor-initiating cells form human colon cancer tumors and metastases. Cell Stem Cell. 2011;9(4):357–65.
Malanchi I, Santamaria-Martinez A, Susanto E, Peng H, Lehr HA, Delaloye JF, Huelsken J. Interactions between cancer stem cells and their niche govern metastatic colonization. Nature. 2011;481(7379):85–9.
Walter RJ, Sonnentag SJ, Orian-Rousseau V, Munoz-Sagredo L. Plasticity in colorectal cancer: why cancer cells differentiate. Cancers. 2021. https://doi.org/10.3390/cancers13040918.
Batlle E, Clevers H. Cancer stem cells revisited. Nat Med. 2017;23(10):1124–34.
Walcher L, Kistenmacher AK, Suo H, Kitte R, Dluczek S, Strauss A, Blaudszun AR, Yevsa T, Fricke S, Kossatz-Boehlert U. Cancer Stem Cells-Origins and Biomarkers: Perspectives for Targeted Personalized Therapies. Front Immunol. 2020;11:1280.
de SousaSousa e F, Kurtova AV, Harnoss JM, Kljavin N, Hoeck JD, Hung J, Anderson JE, Storm EE, Modrusan Z, Koeppen H, et al. A distinct role for Lgr5(+) stem cells in primary and metastatic colon cancer. Nature. 2017;543(7647):676–80.
Todaro M, Gaggianesi M, Catalano V, Benfante A, Iovino F, Biffoni M, Apuzzo T, Sperduti I, Volpe S, Cocorullo G, et al. CD44v6 is a marker of constitutive and reprogrammed cancer stem cells driving colon cancer metastasis. Cell Stem Cell. 2014;14(3):342–56.
Gao W, Chen L, Ma Z, Du Z, Zhao Z, Hu Z, Li Q. Isolation and phenotypic characterization of colorectal cancer stem cells with organ-specific metastatic potential. Gastroenterology. 2013;145(3):636–46.
Merlos-Suarez A, Barriga FM, Jung P, Iglesias M, Cespedes MV, Rossell D, Sevillano M, Hernando-Momblona X, da Silva-Diz V, Munoz P, et al. The intestinal stem cell signature identifies colorectal cancer stem cells and predicts disease relapse. Cell Stem Cell. 2011;8(5):511–24.
Qiu L, Yang X, Wu J, Huang C, Miao Y, Fu Z. HIST2H2BF potentiates the propagation of cancer stem cells via notch signaling to promote malignancy and liver metastasis in colorectal carcinoma. Front Oncol. 2021;11: 677646.
Wang J, Xing Y, Wang Y, He Y, Wang L, Peng S, Yang L, Xie J, Li X, Qiu W, et al. A novel BMI-1 inhibitor QW24 for the treatment of stem-like colorectal cancer. J Exp Clin Cancer Res. 2019;38(1):422.
Oura K, Morishita A, Tani J, Masaki T. Tumor immune microenvironment and immunosuppressive therapy in hepatocellular carcinoma: a review. Int J Mol Sci. 2021. https://doi.org/10.3390/ijms22115801.
Milette S, Sicklick JK, Lowy AM, Brodt P. Molecular pathways: targeting the microenvironment of liver metastases. Clin Cancer Res. 2017;23(21):6390–9.
Pretzsch E, Bosch F, Neumann J, Ganschow P, Bazhin A, Guba M, Werner J, Angele M. Mechanisms of metastasis in colorectal cancer and metastatic organotropism: hematogenous versus peritoneal spread. J Oncol. 2019;2019:7407190.
Zhu X, Wang F, Wu X, Li Z, Wang Z, Ren X, Zhou Y, Song F, Liang Y, Zeng Z, et al. FBX8 promotes metastatic dormancy of colorectal cancer in liver. Cell Death Dis. 2020;11(8):622.
Brodt P. Role of the microenvironment in liver metastasis: from pre- to prometastatic niches. Clin Cancer Res. 2016;22(24):5971–82.
Wen XQ, Qian XL, Sun HK, Zheng LL, Zhu WQ, Li TY, Hu JP. MicroRNAs: multifaceted regulators of colorectal cancer metastasis and clinical applications. Onco Targets Ther. 2020;13:10851–66.
Vidal-Vanaclocha F, Crende O, de DurangoGarcia C, Herreros-Pomares A, Lopez-Domenech S, Gonzalez A, Ruiz-Casares E, Vilboux T, Caruso R, Duran H, et al. Liver prometastatic reaction: stimulating factors and responsive cancer phenotypes. Semin Cancer Biol. 2021;71:122–33.
Zhang Y, Zhao Y, Li Q, Wang Y. Macrophages, as a promising strategy to targeted treatment for colorectal cancer metastasis in tumor immune microenvironment. Front Immunol. 2021;12: 685978.
Liu C, Pei H, Tan F. Matrix stiffness and colorectal cancer. Onco Targets Ther. 2020;13:2747–55.
Paauwe M, Schoonderwoerd MJA, Helderman R, Harryvan TJ, Groenewoud A, van Pelt GW, Bor R, Hemmer DM, Versteeg HH, Snaar-Jagalska BE, et al. Endoglin expression on cancer-associated fibroblasts regulates invasion and stimulates colorectal cancer metastasis. Clin Cancer Res. 2018;24(24):6331–44.
Ma R, Feng Y, Lin S, Chen J, Lin H, Liang X, Zheng H, Cai X. Mechanisms involved in breast cancer liver metastasis. J Transl Med. 2015;13:64.
Hu JL, Wang W, Lan XL, Zeng ZC, Liang YS, Yan YR, Song FY, Wang FF, Zhu XH, Liao WJ, et al. CAFs secreted exosomes promote metastasis and chemotherapy resistance by enhancing cell stemness and epithelial-mesenchymal transition in colorectal cancer. Mol Cancer. 2019;18(1):91.
Tan HX, Gong WZ, Zhou K, Xiao ZG, Hou FT, Huang T, Zhang L, Dong HY, Zhang WL, Liu Y, et al. CXCR4/TGF-beta1 mediated hepatic stellate cells differentiation into carcinoma-associated fibroblasts and promoted liver metastasis of colon cancer. Cancer Biol Ther. 2020;21(3):258–68.
Garner H, de Visser KE. Immune crosstalk in cancer progression and metastatic spread: a complex conversation. Nat Rev Immunol. 2020;20(8):483–97.
Jiang HY, Najmeh S, Martel G, MacFadden-Murphy E, Farias R, Savage P, Leone A, Roussel L, Cools-Lartigue J, Gowing S, et al. Activation of the pattern recognition receptor NOD1 augments colon cancer metastasis. Protein Cell. 2020;11(3):187–201.
Xing W, Xiao Y, Lu X, Zhu H, He X, Huang W, Lopez ES, Wong J, Ju H, Tian L, et al. GFI1 downregulation promotes inflammation-linked metastasis of colorectal cancer. Cell Death Differ. 2017;24(5):929–43.
Li M, Lai X, Zhao Y, Zhang Y, Li M, Li D, Kong J, Zhang Y, Jing P, Li H, et al. Loss of NDRG2 in liver microenvironment inhibits cancer liver metastasis by regulating tumor associate macrophages polarization. Cell Death Dis. 2018;9(2):248.
Wang D, Wang X, Si M, Yang J, Sun S, Wu H, Cui S, Qu X, Yu X. Exosome-encapsulated miRNAs contribute to CXCL12/CXCR4-induced liver metastasis of colorectal cancer by enhancing M2 polarization of macrophages. Cancer Lett. 2020;474:36–52.
Grossman JG, Nywening TM, Belt BA, Panni RZ, Krasnick BA, DeNardo DG, Hawkins WG, Goedegebuure SP, Linehan DC, Fields RC. Recruitment of CCR2(+) tumor associated macrophage to sites of liver metastasis confers a poor prognosis in human colorectal cancer. Oncoimmunology. 2018;7(9): e1470729.
Liu Y, Zhang Q, Xing B, Luo N, Gao R, Yu K, Hu X, Bu Z, Peng J, Ren X, et al. Immune phenotypic linkage between colorectal cancer and liver metastasis. Cancer Cell. 2022;40(4):424-437.e425.
Wu Y, Yang S, Ma J, Chen Z, Song G, Rao D, Cheng Y, Huang S, Liu Y, Jiang S, et al. Spatiotemporal immune landscape of colorectal cancer liver metastasis at single-cell level. Cancer Discov. 2022;12(1):134–53.
Thomassen I, van Gestel YR, Lemmens VE, de Hingh IH. Incidence, prognosis, and treatment options for patients with synchronous peritoneal carcinomatosis and liver metastases from colorectal origin. Dis Colon Rectum. 2013;56(12):1373–80.
Garcia-Carbonero N, Martinez-Useros J, Li W, Orta A, Perez N, Carames C, Hernandez T, Moreno I, Serrano G, Garcia-Foncillas J. KRAS and BRAF mutations as prognostic and predictive biomarkers for standard chemotherapy response in metastatic colorectal cancer: a single institutional study. Cells. 2020. https://doi.org/10.3390/cells9010219.
Lang H, Baumgart J, Heinrich S, Tripke V, Passalaqua M, Maderer A, Galle PR, Roth W, Kloth M, Moehler M. Extended molecular profiling improves stratification and prediction of survival after resection of colorectal liver metastases. Ann Surg. 2019;270(5):799–805.
Tóth C, Sükösd F, Valicsek E, Herpel E, Schirmacher P, Renner M, Mader C, Tiszlavicz L, Kriegsmann J. Expression of ERCC1, RRM1, TUBB3 in correlation with apoptosis repressor ARC, DNA mismatch repair proteins and p53 in liver metastasis of colorectal cancer. Int J Mol Med. 2017;40(5):1457–65.
Olsen LM, Fiehn AK, Hasselby JP. ERCC1 expression in advanced colorectal cancer and matched liver metastases. Pathol Res Pract. 2020;216(3): 152826.
Aykut B, Ochs M, Radhakrishnan P, Brill A, Hocker H, Schwarz S, Weissinger D, Kehm R, Kulu Y, Ulrich A, et al. EMX2 gene expression predicts liver metastasis and survival in colorectal cancer. BMC Cancer. 2017;17(1):555.
Aust N, Schule S, Altendorf-Hofmann AK, Chen Y, Knosel T, Dirsch O, Settmacher U, Weise A, Mrasek K, Liehr T. Loss of chromosome 4 correlates with better long-term survival and lower relapse rate after R0-resection of colorectal liver metastases. J Cancer Res Clin Oncol. 2013;139(11):1861–7.
Rehman AH, Jones RP, Poston G. Prognostic and predictive markers in liver limited stage IV colorectal cancer. Eur J Surg Oncol. 2019;45(12):2251–6.
Sun L, Liu X, Pan B, Hu X, Zhu Y, Su Y, Guo Z, Zhang G, Xu M, Xu X, et al. Serum exosomal miR-122 as a potential diagnostic and prognostic biomarker of colorectal cancer with liver metastasis. J Cancer. 2020;11(3):630–7.
Xu H, Wang C, Song H, Xu Y, Ji G. RNA-Seq profiling of circular RNAs in human colorectal cancer liver metastasis and the potential biomarkers. Mol Cancer. 2019;18(1):8.
Zhi Q, Wan D, Ren R, Xu Z, Guo X, Han Y, Liu F, Xu Y, Qin L, Wang Y. Circular RNA profiling identifies circ102049 as a key regulator of colorectal liver metastasis. Mol Oncol. 2021;15(2):623–41.
Nassar FJ, Msheik ZS, Itani MM, Helou RE, Hadla R, Kreidieh F, Bejjany R, Mukherji D, Shamseddine A, Nasr RR, et al. Circulating miRNA as biomarkers for colorectal cancer diagnosis and liver metastasis. Diagnostics. 2021. https://doi.org/10.3390/diagnostics11020341.
Kong H, Wu Y, Zhu M, Zhai C, Qian J, Gao X, Wang S, Hou Y, Lu S, Zhu H. Long non-coding RNAs: novel prognostic biomarkers for liver metastases in patients with early stage colorectal cancer. Oncotarget. 2016;7(31):50428–36.
Yang X, Zhang S, He C, Xue P, Zhang L, He Z, Zang L, Feng B, Sun J, Zheng M. METTL14 suppresses proliferation and metastasis of colorectal cancer by down-regulating oncogenic long non-coding RNA XIST. Mol Cancer. 2020;19(1):46.
Gao W, Chen Y, Yang J, Zhuo C, Huang S, Zhang H, Shi Y. Clinical perspectives on liquid biopsy in metastatic colorectal cancer. Front Genet. 2021;12: 634642.
Allen JE, Saroya BS, Kunkel M, Dicker DT, Das A, Peters KL, Joudeh J, Zhu J, El-Deiry WS. Apoptotic circulating tumor cells (CTCs) in the peripheral blood of metastatic colorectal cancer patients are associated with liver metastasis but not CTCs. Oncotarget. 2014;5(7):1753–60.
Song Z, Chen E, Qian J, Xu J, Cao G, Zhou W, Wang F, Chen M, Xu D, Wang X, et al. Serum chitinase activity prognosticates metastasis of colorectal cancer. BMC Cancer. 2019;19(1):629.
Garcia-Alfonso P, Ferrer A, Gil S, Duenas R, Perez MT, Molina R, Capdevila J, Safont MJ, Castanon C, Cano JM, et al. Neoadjuvant and conversion treatment of patients with colorectal liver metastasis: the potential role of bevacizumab and other antiangiogenic agents. Target Oncol. 2015;10(4):453–65.
Filip S, Vymetalkova V, Petera J, Vodickova L, Kubecek O, John S, Cecka F, Krupova M, Manethova M, Cervena K, et al. Distant metastasis in colorectal cancer patients-do we have new predicting clinicopathological and molecular biomarkers? a comprehensive review. Int J Mol Sci. 2020. https://doi.org/10.3390/ijms21155255.
Alhumaid A, AlYousef Z, Bakhsh HA, AlGhamdi S, Aziz MA. Emerging paradigms in the treatment of liver metastases in colorectal cancer. Crit Rev Oncol Hematol. 2018;132:39–50.
Saad AM, Abdel-Rahman O. Initial systemic chemotherapeutic and targeted therapy strategies for the treatment of colorectal cancer patients with liver metastases. Expert Opin Pharmacother. 2019;20(14):1767–75.
Punt CJ, Koopman M, Vermeulen L. From tumour heterogeneity to advances in precision treatment of colorectal cancer. Nat Rev Clin Oncol. 2017;14(4):235–46.
Sottoriva A, Kang H, Ma Z, Graham TA, Salomon MP, Zhao J, Marjoram P, Siegmund K, Press MF, Shibata D, et al. A Big Bang model of human colorectal tumor growth. Nat Genet. 2015;47(3):209–16.
Singovski G, Bernal C, Kuciak M, Siegl-Cachedenier I, Conod A. Ruiz i Altaba A: in vivo epigenetic reprogramming of primary human colon cancer cells enhances metastases. J Mol Cell Biol. 2016;8(2):157–73.
Hu M, Zhou X, Wang Y, Guan K, Huang L. Relaxin-FOLFOX-IL-12 triple combination therapy engages memory response and achieves long-term survival in colorectal cancer liver metastasis. J Control Release. 2020;319:213–21.
Poureau P-G, Metges J-P. Fundamentals of digestive cancers immunology, especially gastric and hepatocellular carcinomasfondamentaux de l’immunologie des cancers digestifs (Gastriques et Hépatocellulaires). Oncologie. 2021;23(1):47–59.
Xing C, Li H, Li RJ, Yin L, Zhang HF, Huang ZN, Cheng Z, Li J, Wang ZH, Peng HL. The roles of exosomal immune checkpoint proteins in tumors. Mil Med Res. 2021;8(1):56.
Zhang Y, Song J, Zhao Z, Yang M, Chen M, Liu C, Ji J, Zhu D. Single-cell transcriptome analysis reveals tumor immune microenvironment heterogenicity and granulocytes enrichment in colorectal cancer liver metastases. Cancer Lett. 2020;470:84–94.
Kalanxhi E, Meltzer S, Ree AH. Immune-modulating effects of conventional therapies in colorectal cancer. Cancers. 2020. https://doi.org/10.3390/cancers12082193.
Chen DL, Lu YX, Zhang JX, Wei XL, Wang F, Zeng ZL, Pan ZZ, Yuan YF, Wang FH, Pelicano H, et al. Long non-coding RNA UICLM promotes colorectal cancer liver metastasis by acting as a ceRNA for microRNA-215 to regulate ZEB2 expression. Theranostics. 2017;7(19):4836–49.
Acknowledgements
Not applicable.
Funding
This work was financially supported by grants from the National Key Research and Development Program of China (2021YFF1201300, 2022YFE0103600), the National Natural Science Foundation of China (No. 81872280, 82073094), the CAMS Innovation Fund for Medical Sciences (CIFMS)(2021-I2M-1-014), the Open Issue of State Key Laboratory of Molecular Oncology (No. SKL-KF-2021–16), and the Independent Issue of State Key Laboratory of Molecular Oncology (No. SKL-2021–16).
Author information
Authors and Affiliations
Contributions
YN have drafted the work and substantively revised it. WY contributed to the literature review. HQ and YS contributed to the supervision and supported final approval of the article. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare that there are no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Niu, Y., Yang, W., Qian, H. et al. Intracellular and extracellular factors of colorectal cancer liver metastasis: a pivotal perplex to be fully elucidated. Cancer Cell Int 22, 341 (2022). https://doi.org/10.1186/s12935-022-02766-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12935-022-02766-w