Abstract
In the highly automated processing in broiler abattoirs, some process steps reduce the bacterial counts and inactivate or remove pathogens, while others can lead to an increase. The present study compared the reduction of Enterobacteriaceae counts (EBCs) on breast skin samples in 3 broiler abattoirs using different scalding techniques: (A) conventional immersion scalding (360 s), (B) conventional immersion scalding with thermal treatment of the water (204 s), and (C) the AeroScalder® using hot, humid, saturated air as the scalding medium (360 s in air). In 3 commercial broiler abattoirs in Germany and The Netherlands, a total of 320 breast skin samples per abattoir (before and after scalding, after plucking, before and after chilling) and water samples from the scalders were taken and examined for EBC. After scalding, a significant EBC reduction by 0.7 log was determined only for the conventional immersion scalder (Abbatoir A); the reductions of the other 2 scalders were 0.1 log (Abattoir B) and 0.2 log (Abattoir C) and not statistically significant. The EBCs after scalding differed by up to 0.5 log cfu/g when the 3 scalders were compared, and these counts can be seen as similar. For all 3 abattoirs, the largest EBC reductions (p < 0.001) of 2.8 to 3.6 logs were found after plucking. Compared to the immersion scalders, EBCs in water samples were lowest in those taken from the AeroScalder®. Hence, we conclude that the conventional immersion scalders and the AeroScalder® reduced EBCs in a comparable manner. However, the greatest reductions in EBCs were seen after the plucking steps in the studied abattoirs, not after the scalding as such.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Broilers when entering slaughter facilities carry on their feathers bacteria that originate from fecal contamination (Göksoy et al. 2004; Kotula and Pandya 1995; Seliwiorstow et al. 2015). A reduction of bacterial load can be seen after several mainly automated processing steps, so the risk of cross-contamination is lowered throughout the process, but cannot be excluded in total (Althaus et al. 2017; EFSA 2012; Göksoy et al. 2004; Kotula and Pandya 1995; Lillard 1990; Mead 2004). After entering the abattoir, the processing line for broilers starts with stunning and bleeding followed by scalding. This processing step is necessary to facilitate the plucking by loosening the feathers in the feather follicles (Götze 1979; Singh and Rama 2017). Today, scalding temperatures between 50 and 65 °C, depending on the chilling system in place, are used (Löhren 2012; Singh and Rama 2017). This encompasses scalding regimes that operate with water temperatures between 51 and 55 °C.
The traditional scalding principle is based on convection, which means that the carcasses are in direct contact with the scalding water, and the temperature on the carcass’ surfaces is the same as that of the scalding water. Conventional scalding tanks are constructed with several chambers, and the carcasses are pulled upside down through the hot water scalding tank in countercurrent principle. This means that the carcasses are pulled in one direction and the scalding water in the other direction while fresh water is added continuously in the last scalding tank (Löhren 2012). One reason for this is water replacement after its continuous losses via animals’ feathers (Fries et al. 2001). In addition, the introduction of fresh water has the advantage that the microbial load of the scalding water is minimized by a dilution effect. Therefore, the water is usually introduced in the last scalding tank and transported to the beginning in countercurrent to the process line. Thus, the microbial load is lowest in the last scalding tank (Cason et al. 2000; Heemskerk 2005), resulting in lower microbial loads of water remaining in the feathers of the scalded broilers (Löhren 2012).
A new scalding technique based on condensation is the so-called AeroScalder® (Marel, The Netherlands), which utilizes hot, humid, saturated air. The surface temperature is controlled by the dew point that protects the product from epidermal damage and cooking. The AeroScalder® consists of 2 chambers, the air conditioning chamber and the scalding chamber. In the first chamber, the air is moisturized and warmed up to 57 °C. The hot moisturized air is led into the scalding chamber and blown at high velocity onto specific parts of the product between the feathers to reach the feather follicles. The water in vapor phase contained in the saturated air condenses at the colder carcass surface. This results in a local drop of the dew point and similar temperatures on the surface as in low scalding regimes operating with temperatures ranging from 51 to 55 °C. In contrast to traditional scalding, the AeroScalder® is designed to actively direct the air onto the parts of the carcasses where the feathers are more difficult to pluck. The air is re-circulated and re-conditioned in the air conditioning chamber and used again (Anonymous 2021; personal communication Ricardo Hernández Arrondo 2023).
However, the microbial load of the feathers and the surface of the broiler carcasses must be considered. On the one hand, plucking leads to a reduction of the microbial load by removing the feathers (Buess et al. 2019; Stephan 2014; Zweifel et al. 2015). On the other hand, it can lead to recontamination from the plucking fingers, from the water-air-mixture used (Svobodová et al. 2012), and from release of feces during this processing step (Berrang et al. 2001; Heemskerk 2005). To counteract this, an outside washer can be installed after plucking (Buess et al. 2019; Puolanne and Ertbjerg 2014) to reduce the microbial load of the carcasses before they enter the evisceration line (Buess et al. 2019). After plucking, the next steps of the processing line normally include opening of the carcasses, evisceration, trimming, and inside-outside washers before the carcasses enter the chilling area (Puolanne and Ertbjerg 2014). In general, only slight changes in the microbial loads were achieved by these processing steps (Buess et al. 2019; Zweifel et al. 2015).
Several studies have investigated changes in microbial loads at different steps, which can represent the process hygiene in broiler abattoirs. Some of them used EBCs as an indicator of fecal contamination and showed reductions after scalding and after plucking (Althaus et al. 2017; Berrang and Dickens 2000; Göksoy et al. 2004; Pacholewicz et al. 2015a). However, plucking can also result in an EBC increase (Abu-Ruwaida et al. 1994). For Campylobacter, it can result in an increase, a stable situation, or a reduction, depending on the abattoir and the incoming flock (Althaus et al. 2017; Pacholewicz et al. 2015a, b, 2016; Seliwiorstow et al. 2015). All these studies described immersion scalding being in use in the participating broiler abattoirs, but not all used counterflow scalders, and the scalding temperature was not always mentioned. Additionally, not all studies started sampling for process control before scalding, so the incoming microbial loads were not determined. The scalding technique as such was not the focus of these studies. However, it can be assumed that different scalding techniques could produce a range of impacts on the microbial loads, and so could result in different loads in the post-scalding processing steps; this would result in different process hygiene at the end of the slaughter line.
Since the scalding water can act as vector for cross-contamination, we analyzed the impact of conventional immersion scalders and the AeroScalder® used in commercial broiler abattoirs on the EBC on broiler carcasses from the beginning of the slaughter lines to their end after chilling.
2 Material and methods
2.1 Slaughterhouse practices and scalding techniques in the abattoirs
The study was conducted at 3 commercial abattoirs located in Northwestern and Eastern Germany and in the Netherlands. All 3 abattoirs were large-scale abattoirs with line speeds of 10,000 birds/h (Abattoir C) or 13,500 birds/h (Abattoirs A & B). Stunning of live birds was conducted with CO2 in all 3 abattoirs. The scalders each operated in a low scalding regime, so the scalding temperature ranged between 51 and 55 °C for all scalders but the scalding techniques differed. In Abattoir A, a conventional immersion scalder with 2 scalding tanks using the counterflow-principle and a scalding time of approx. 360 s was used. Abattoir B operated a conventional immersion scalder with 3 tanks and thermal treatment of the re-used scalding water, operating in counterflow-principle with an approximate scalding time of 204 s. In Abattoir B, the scalding water was taken from the second scalding tank, thermally-treated at 71–74 °C, then reintroduced into the third scalding tank. The scalding water in this tank was kept at a stable temperature of 52–54 °C. Abattoir C used an AeroScalder® with a scalding time of around 360 s (Table 1). The following processing steps including plucking, opening of the carcasses, and evisceration, as well as the washing-steps before chilling were similar in all 3 abattoirs (Fig. 1).
2.2 Sampling
Samples were taken between May 2016 and February 2018, resulting in 2 summer and 2 winter sampling periods with 24 sampling dates in total. On each sampling date, 2 flocks were randomly chosen at the abattoir for sampling. At the end, 48 flocks were included. The birds sampled for the study were 39–41 days old and had an average slaughter weight of 2.57 kg. Sampling took place at 5 different sampling points at the slaughter line: before scalding, after scalding, after plucking, before chilling, and after chilling. At each sampling point, breast skin samples without feathers from 4 randomly chosen carcasses from each flock were taken. For this, an abattoir employee took a carcass out of the processing line and held it head down without contacting the carcass’ surface. For sampling, the breast skin was grasped in the center of the breast with sterile forceps and was gently pulled off a little from the carcass. Using a sterile scalpel, incisions were then made carefully along imaginary lines surrounding the area to be sampled (Fig. 2) and behind the skin to excise only the skin. This sampling aseptically removed all the breast skin on the carcass. Each sample was placed in an individual sterile blender bag with lateral filter for volumes up to 400 mL (VWR International GmbH, Darmstadt, Germany) and cooled immediately to 4 °C. After sampling of the second flock, all samples were transported at 4 °C to a laboratory in Berlin and were stored in a refrigerator that maintained a temperature of around 4 °C until the next morning when the laboratory work started. In total, 320 breast skin samples per abattoir were taken, meaning that 64 samples per sampling point were included in the laboratory analysis.
Additionally, 114 scalding water samples (n = 36 in Abattoir A, n = 46 in Abattoir B, and n = 32 in Abattoir C) were taken with sterile 15 mL tubes (Carl Roth GmbH & Co. KG, Karlsruhe, Germany) after sampling the respective flock, but within the time birds from the sampled flock were passing through the scalder. At Abattoir A, superficial scalding water from the beginning and end areas of the scalding tank was taken. The scalding water at Abattoir B was sampled from the beginning, middle, and end areas. At Abattoir C, the condensation water that accumulates with the moisturized air during scalding and is recycled during processing was sampled. For this, a water tap was installed in the middle area of the scalding tunnel. After draining the water standing in the water pipe, 2 sampling tubes were aseptically filled successively.
2.3 Microbiological examinations
Breast skin samples and scalding water samples were analyzed for EBC using the drop plating technique. For this, each breast skin sample was weighed and blended in a 1:10 ratio with Luria Bertani broth (Carl Roth GmbH & Co. KG, Karlsruhe, Germany) in the stomacher bag that had been used for sampling. From each scalding water sample, 10 mL were taken und transferred to a sterile stomacher bag and mixed in a 1:10 ratio with Luria Bertani broth. After homogenization for 120 s at 500–540 strokes per minute in a stomacher (Smasher™ High-Performance Blender/Homogenizer, bioMerieux, Marcy-l’Étoile, France), dilution series in sodium chloride peptone agar test tubes were created. From each dilution series, 0.05 mL were dropped on MacConkey Agar No 3 plates (Thermo Fisher Diagnostics GmbH [former Oxoid Deutschland GmbH], Wesel, Germany) in duplicate and streaked out on each agar surface by using the pipette tip. After incubating the agar plates aerobically for 24 h at 37 °C, colony forming units (cfu) were counted. To determine the EBC, all samples with a minimum of 3 typical colonies (after the manufacturer’s manual) on the duplicate agar plates were included in the count. EBC was expressed as cfu/g (breast skin samples) or cfu/mL (water samples).
2.4 Statistical analyses
Statistical analyses were carried out using IBM SPSS for Windows version 24 (SPSS for Windows, IBM® - Armonk, New York, USA). For practical purposes and following Hübner et al. (2002), mean values differing by less than 0.5 log units were defined as typical laboratory variance. EBC values were transformed to the logarithm of base 10 to achieve normal distribution.
The 3 processing steps scalding, plucking, and chilling were investigated in detail using mixed linear regression models with abattoir and processing position as fixed (including interaction), and flock as random variables. Log EBC was the dependent variable. In total, 4 models were applied, comparing EBCs from:
-
before and after scalding,
-
after scalding and after plucking,
-
before scalding and after chilling,
-
after plucking and after chilling.
Model diagnostics included visual inspection of residuals for normality and homogeneity. Multiple testing of specific positions was corrected using Bonferroni method. Thus, p-values < 0.025 were regarded as significant in terms of a 5% level of significance.
3 Results
3.1 Breast skin samples
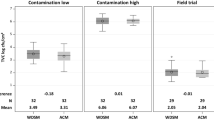
Mean EBCs before scalding for the 3 abattoirs ranged from 6.0 logcfu/g to 6.4 log cfu/g breast skin. Following the sampling points over the whole processing line for each abattoir, decreases of mean EBC were identified, so mean EBCs were reduced to between 2.6 and 2.7 log cfu/g after chilling. The scalding technique itself reduced the EBC by 0.7, 0.2, and 0.1 log for Abattoirs A, B, and C, respectively (Fig. 3).
Distribution of Enterobacteriaceae counts on breast skin samples in 3 abattoirs examined. * indicates significant reductions of p < 0.05; *** indicates significant reduction of p < 0.001. A – abattoir A with immersion scalding; B – Abattoir B with immersion scalding and thermal treatment of the scalding water, C – Abattoir C with AeroScalder® (Marel); EBC – Enterobacteriaceae count; x – arithmetic mean
The main reduction of EBC was seen after plucking in all 3 abattoirs. The reduction due to plucking was greatest in Abattoir A, with a mean reduction of 3.6 logs after plucking compared to the initial EBC on the breast skin before scalding. Reductions of mean EBC after plucking were 2.8 and 3.5 logs for Abattoirs C and B, respectively (Fig. 3).
The result of the mixed linear regression model for EBC before and after scalding showed an intra-flock variance of 28%. Comparing the 3 scalding techniques, no overall significant differences were seen (p = 0.103). However, significant interactions between scalding technique and the sampling points of before and after scalding were identified (p < 0.001, Fig. 4). Among the abattoirs, scalding at Abattoir A produced the greatest EBC reduction. Between Abattoir A and Abattoirs B and C, there were significant differences between EBCs at the sampling points of before and after scalding (p = 0.003, n = 64 samples per abattoir) (Fig. 4; EBCs at Abattoir A were lower than in the other abattoirs). Independent of the abattoir, the EBCs were significantly lower after scalding than before scalding (p < 0.001). The conventional immersion scalder in Abattoir A produced the greatest EBC reduction [0.7 log; from 6.3 log cfu/g (95%-CI 6.2–6.4) to 5.6 log cfu/g (95% CI 6.4–5.8), p = 0.003], while the conventional immersion scalder with thermally-treated water in Abattoir B and the AeroScalder® in Abattoir C produced similar reductions of 0.1 log [reduction from 6.0 log cfu/g (95%-CI 5.8–6.2) to 5.9 log cfu/g (95%-CI 5.7–6.1), p = 0.471) and 0.2 log (reduction from 6.4 log cfu/g (95%-CI 6.3–6.5) to 6.2 log cfu/g (95%-CI 6.0–6.4), p = 0.049], respectively (Fig. 3).
Interaction between abattoir and the sampling point before and after scalding of mean predicted Enterobacteriaceae counts on breast skin samples. A – Abattoir A with immersion scalding; B – Abattoir B with immersion scalding and thermal treatment of the scalding water; C – Abattoir C with AeroScalder® (Marel); EBC – Enterobacteriaceae count
Comparing the EBCs between after scalding and after plucking, the values decreased significantly in all abattoirs (p < 0.001). This reduction was greatest in Abattoir C (p = 0.017, Fig. 5). The inter-cluster coefficient was 8.1%. Abattoir B had the highest and Abattoir A the lowest EBCs (p = 0.003).
Interaction between abattoir and the sampling point after scalding and after plucking of mean predicted Enterobacteriaceae counts on breast skin samples. A – Abattoir A with immersion scalding; B – Abattoir B with immersion scalding and thermal treatment of the scalding water; C – Abattoir C with AeroScalder® (Marel); EBC – Enterobacteriaceae count
Since no great impact on mean EBC after the processing steps following plucking were measured, a mixed linear regression model was applied to compare EBCs at the beginning and the end of the slaughter line. The inter-cluster coefficient was 10%. In all 3 abattoirs, significant reductions of mean EBC were found at the end of the slaughter line compared to the beginning (p < 0.001) (Fig. 3). Abattoir C had significantly higher EBCs at the end of the line than the other 2 abattoirs (p < 0.001). There were no significant interactions between abattoir and sampling point (p = 0.246).
In all abattoirs, EBCs were significantly lower after chilling compared to after plucking (p < 0.001). Abattoir B had the greatest reduction, from 3.8 log cfu/g to 2.8 log cfu/g EBC (interaction p = 0.001) (Fig. 6). The inter-cluster coefficient was 8.8%.
Interaction between abattoir and the sampling point after plucking and after chilling of mean predicted Enterobacteriaceae counts on breast skin samples. A – Abattoir A with immersion scalding; B – Abattoir B with immersion scalding and thermal treatment of the scalding water; C – Abattoir C with AeroScalder® (Marel); EBC – Enterobacteriaceae count
3.2 Scalding water samples
EBCs reduced in scalding water samples for Abattoirs A and B from the beginning to the end of the counterflow scalding tanks. For Abattoir C with the AeroScalder®, only a mean EBC of all water samples (2.48 log cfu/mL) could be calculated, since the water samples were taken successively at the same place in the middle of the scalding tunnel on each sampling day. Compared to the 2 other abattoirs, the lowest mean EBCs were found in the water samples from Abattoir C (Table 2).
4 Discussion
Each of the 3 scalding techniques produced a reduction of the EBC, either after scalding (Abattoir A and C) or after plucking (all abattoirs). The assumption that a particularly good microbial reduction effect is achieved by the washing effect of the scalding water was not confirmed, as the AeroScalder® (using hot, humid, saturated air) led to similar reductions of the EBC on breast skin overall. Some studies investigating the impact of scalding water on the microbial load showed a reduction of microbial loads from the beginning to the end in counterflow immersion scalding tanks (Althaus et al. 2017; Cason et al. 2000; Gardner and Golan 1976; Heemskerk 2005; Lillard 1990). Gardner (1976) concluded that the water usage and the scalding process are the main causes of EBC reduction, whereas plucking of the feathers can result in an increase of EBC.
In our study, the microbial loads of the water samples taken from the AeroScalder® at Abattoir C were the lowest among the 3 abattoirs. This could be explained by the lack of a washing effect. Additionally, at Abattoir C, we could only sample the condensed water that is recycled during the process to produce the hot, humid, saturated air that is the scalding medium and in contact with the carcasses. Therefore, it can be considered that the condensed water itself is only indirectly in contact with the carcasses, although this water can also act as a vector. The greater EBC after scalding for Abattoir C (using the AeroScalder®) (Fig. 1) can be explained by the lack of a washing effect, as only hot, humid, saturated air is blown between the feather follicles. Comparing the water samples from the other scalders, we confirmed that the counterflow-principle led to reductions in the microbial loads through the different scalding tanks, as shown before by Althaus et al. (2017), Cason et al. (2000), Heemskerk (2005), and Lillard (1990). Nonetheless, in conclusion, the AeroScalder® in Abattoir C, with its new technology, led to comparable EBCs as were determined for the 2 conventional immersion scalders used in Abattoirs A and B.
The conventional immersion scalder in Abattoir A utilized a longer scalding time than the scalder in Abattoir B and produced a significantly better reduction of EBC after scalding. However, it must be concluded that all 3 scalders achieved good but not highly relevant reductions of EBC after scalding, as the best mean log reduction (Abattoir A) was only 0.7 log, and a log reduction of 0.5 log is within the expected laboratory variance (Hübner et al. 2002). Therefore, we stress that the found significance is only from a statistical point of view. Nonetheless, we determined that the scalding in combination with the following plucking step resulted in small but significant EBC reductions in all 3 abattoirs.
Plucking produced the most extensive reductions of EBCs in our study. This is consistent with other studies investigating microbial loads (total viable count, EBC, extended-spectrum β-lactamase producing Escherichia coli counts) on poultry carcasses (Buess et al. 2019; Chen et al. 2020; Göksoy et al. 2004; Lillard 1990; Mead et al. 1993; Pacholewicz et al. 2015a, b; Zweifel et al. 2015). In contrast to this, other studies showed that scalding resulted in extensive reductions of the microbial loads of the carcasses, while the plucking afterwards led to a subsequent increase (Gardner and Golan 1976; Hutchison et al. 2022; Izat et al. 1988). For Campylobacter, different studies also showed contradictory results regarding the microbial loads after plucking, i.e., an increase, reduction, or stable loads depending on the abattoir and flock investigated (Althaus et al. 2017; Pacholewicz et al. 2015a, b, 2016; Seliwiorstow et al. 2015; Stephan 2014). As reasons for these reported contradictory results, it could be assumed that different scalding temperatures other than that used in our investigation, or cross-contamination caused by the plucking fingers, were influencing factors. Not all studies explained the scalding temperature and process or the defeathering process in detail, so we can only assume possible influences.
Compared to other studies using skin samples (Althaus et al. 2017; Göksoy et al. 2004; Zweifel et al. 2015), the influence of scalding in our study was lower. Overall, when comparing different studies, the sampling material and the sampling method have to be taken into account. We decided to use breast skin samples for sampling immediately before scalding and defeathering. If we had sampled other carcass parts or had used the carcass rinse method, we would inevitably also have sampled the feathers still in the skin, which are often heavily contaminated with bacteria (Kotula and Pandya 1995; Seliwiorstow et al. 2015), and which would have had a great impact on the results. However, it is possible to derive trends and, thus, compare studies conducted at different abattoirs using different sampling materials or methods. In conclusion, the measured reductions of EBC on breast skin samples in the current study, especially at the processing step after plucking, were in line with studies that showed general EBC reductions along the processing line at broiler abattoirs; the studies used neck skin samples (Althaus et al. 2017; Buess et al. 2019; Göksoy et al. 2004; Zweifel et al. 2015), the carcass rinse method (Lillard 1990; Pacholewicz et al. 2015a, b; Svobodová et al. 2012), or the swabbing technique (Gardner and Golan 1976; Matias et al. 2010).
Depending on the sampling points compared, the results of the performed mixed linear models showed intra-flock variances for EBC of between 28% and 8.1%, meaning the differences in EBCs were also due to the flocks. This is in concordance with other authors who also concluded that abattoir- and flock-specific influences can be seen and can result in differences when comparing abattoirs (Althaus et al. 2017; Pacholewicz et al. 2015a, b, 2016; Seliwiorstow et al. 2015). From our study, we can conclude that plucking had the main influence on EBC, as all feathers with adhered feces were removed during this processing step.
5 Conclusions
The EBC reductions caused by the scalding were much smaller than the reductions caused by plucking in all 3 abattoirs. The greatest reductions of EBC were seen after the plucking step in all 3 abattoirs included in the study. EBC reductions at the abattoir using the AeroScalder® were comparable to the reductions achieved at abattoirs with conventional immersion scalders. Intra-flock differences in EBC reductions were measured, and these can have an impact on the process hygiene additionally to abattoir-specific influences, such as scalding regime, plucking, or others.
Data availability
The data presented in this study are available on request from the corresponding author. The data are not publicly available due to privacy.
References
Abu-Ruwaida A, Sawaya W, Dashti B, Murad M, Al-Othman H (1994) Microbiological quality of broilers during processing in a modern commercial slaughterhouse in Kuwait. J Food Prot 57(10):887–892. https://doi.org/10.4315/0362-028X-57.10.887
Althaus D, Zweifel C, Stephan R (2017) Analysis of a poultry slaughter process: influence of process stages on the microbiological contamination of broiler carcasses. Ital J Food Saf 6(4):7097. https://doi.org/10.4081/ijfs.2017.7097
Anonymous (2021) Product: Aeroscalder. https://marel.com/en/products/aeroscalder. Accessed 31 March 2023
Berrang M, Dickens J (2000) Presence and level of Campylobacter spp. on broiler carcasses throughout the processing plant. J Appl Poult Res 9(1):43–47. https://doi.org/10.1093/japr/9.1.43
Berrang M, Buhr R, Cason J, Dickens J (2001) Broiler carcass contamination with Campylobacter from feces during defeathering. J Food Prot 64(12):2063–2066. https://doi.org/10.4315/0362-028X-64.12.2063
Buess S, Zurfluh K, Stephan R, Guldimann C (2019) Quantitative microbiological slaughter process analysis in a large-scale Swiss poultry abattoir. Food Control 105:86–93. https://doi.org/10.1016/j.foodcont.2019.05.012
Cason J, Hinton A Jr, Ingram K (2000) Coliform, Escherichia coli, and salmonellae concentrations in a multiple-tank, counterflow poultry scalder. J Food Prot 63(9):1184–1188. https://doi.org/10.4315/0362-028X-63.9.1184
Chen S, Fegan N, Kocharunchitt C, Bowman J, Duffy L (2020) Impact of poultry processing operating parameters on bacterial transmission and persistence on chicken carcasses and their shelf life. Appl Environ Microbiol 86(12):e00594–e00520. https://doi.org/10.1128/AEM.00594-20
EFSA (2012) Scientific opinion on the public health hazards to be covered by inspection of meat (poultry). EFSA J 10(6):2741. https://doi.org/10.2903/j.efsa.2012.2741
European Commission (2005) Commission Regulation (EC) No 2073/2005 of 15 November 2005 on microbiological criteria for foodstuffs; current consolidated version: 08/03/2020. http://data.europa.eu/eli/reg/2005/2073/oj
Fries R (2001) Chap. 5. Geflügelfleischgewinnung, 5.1.4 Brühen. In: Fries R, Bergmann V, Fehlhaber K (eds) Praxis Der Geflügelfleischuntersuchung. Schlütersche GmbH & Co. KG, Verlag und Druckerei, Hannover, pp 39–41
Gardner F, Golan F (1976) Water usage in poultry processing-an effective mechanism for bacterial reduction. In: Proceedings of the 7th National Symposium on Food Processing Wastes. 7–9 April 1976, Atlanta, Georgia, USA; Environmental Protection Technology Series: EPA-600/2-76-304, pp 338–355
Göksoy EO, Kirkan S, Kok F (2004) Microbiological quality of broiler carcasses during processing in two slaughterhouses in Turkey. Poult Sci 83(8):1427–1432. https://doi.org/10.1093/ps/83.8.1427
Götze U (1979) Chap. 5. Technologie und Hygiene Der Geflügelschlachtung, 5.3.4 Brühen. In: Großklaus D (ed) Geflügelfleischhygiene: Tierhaltung, Schlachtung, Lebendtier- und Fleischuntersuchung, Erzeugnisse, Rechtsgrundlagen. Verlag Paul Parey, Hamburg und Berlin, pp 116–118
Heemskerk W (2005) Preventive strategies during slaughter of poultry, to improve food safety. In Proceedings of the XVII European Symposium on the Quality of Poultry Meat and XI European Symposium on the Quality of Eggs and Egg Products. https://www.cabi.org/Uploads/animal-science/worlds-poultry-science-association/WPSA-the-netherlands-2005/119.pdf. Accessed 31 March 2023
Hübner P, Gautsch S, Jemmi T (2002) In house validation (single laboratory validation) of microbiological methods. Mitt Lebensmittelunters Hyg 93(2):118–139
Hutchison ML, Harrison D, Tchorzewska MA, Gonzalez-Bodi S, Madden RH, Corry JEL, Allen VM (2022) Quantitative determination of Campylobacter on broilers along 22 United Kingdom processing lines to identify potential process control points and cross-contamination from colonized to uncolonized flocks. J Food Prot 85(12):1696–1707. https://doi.org/10.4315/jfp-22-204
Izat A, Gardner F, Denton J, Golan F (1988) Incidence and level of Campylobacter jejuni in broiler processing. Poult Sci 67(11):1568–1572. https://doi.org/10.3382/ps.0671568
Kotula KL, Pandya Y (1995) Bacterial contamination of broiler chickens before scalding. J Food Prot 58(12):1326–1329. https://doi.org/10.4315/0362-028X-58.12.1326
Lillard H (1990) The impact of commercial processing procedures on the bacterial contamination and cross-contamination of broiler carcasses. J Food Prot 53(3):202–204. https://doi.org/10.4315/0362-028X-53.3.202
Löhren U (2012) Overview on current practices of poultry slaughtering and poultry meat inspection. EFSA Supporting Publications 9(6):298E [58pp.]. https://doi.org/10.2903/sp.efsa.2012.EN-298
Matias BG, de Arruda Pinto PS, Cossi MVC, Nero LA (2010) Salmonella spp. and hygiene indicator microorganisms in chicken carcasses obtained at different processing stages in two slaughterhouses. Foodborne Pathog Dis 7(3):313–318. https://doi.org/10.1089/fpd.2009.0392
Mead G (2004) Microbial hazards in production and processing. In: Mead CG (ed) Poultry meat processing and quality. Woodhead Publishing Limited, Cambridge, England, pp 232–257
Mead G, Hudson W, Hinton M (1993) Microbiological survey of five poultry processing plants in the UK. Br Poult Sci 34(3):497–503. https://doi.org/10.1080/00071669308417605
Pacholewicz E, Liakopoulos A, Swart A, Gortemaker B, Dierikx C, Havelaar AH, Schmitt H (2015a) Reduction of extended-spectrum-β-lactamase-and AmpC-β-lactamase-producing Escherichia coli through processing in two broiler chicken slaughterhouses. Int J Food Microbiol 215:57–63. https://doi.org/10.1016/j.ijfoodmicro.2015.08.010
Pacholewicz E, Swart A, Schipper M, Gortemaker BG, Wagenaar JA, Havelaar AH, Lipman LJ (2015b) A comparison of fluctuations of Campylobacter and Escherichia coli concentrations on broiler chicken carcasses during processing in two slaughterhouses. Int J Food Microbiol 205:119–127. https://doi.org/10.1016/j.ijfoodmicro.2015.04.006
Pacholewicz E, Swart A, Wagenaar JA, Lipman LJ, Havelaar AH (2016) Explanatory variables associated with Campylobacter and Escherichia coli concentrations on broiler chicken carcasses during processing in two slaughterhouses. J Food Prot 79(12):2038–2047. https://doi.org/10.4315/0362-028x.jfp-16-064
Puolanne E, Ertbjerg P (2014) Chap. 4: the slaughter process, 4.5 poultry. In: Ninios T, Lundén J, Korkeala H, Fredriksson-Ahomaa M (eds) Meat inspection and control in the slaughterhouse. John Wiley & Sons Ltd., pp 41–43. https://doi.org/10.1002/9781118525821.ch4
Seliwiorstow T, Baré J, Van Damme I, Uyttendaele M, De Zutter L (2015) Campylobacter carcass contamination throughout the slaughter process of Campylobacter-positive broiler batches. Int J Food Microbiol 194:25–31. https://doi.org/10.1016/j.ijfoodmicro.2014.11.004
Singh M, Rama EN (2017) Chap. 6: safety management and pathogen monitoring in poultry slaughterhouse operations: the case of the United States. 3 effects of processing operations on carcass contamination: from live transportation to carcass washing. In: Rick SC (ed) Achieving sustainable production of poultry meat, vol 1. Burleigh Dodds Science Publishing, pp 113–117
Stephan R (2014) Mikrobiologische Prozessanalyse Der Geflügelschlachtung in Drei Schlachtbetrieben Der Schweiz: Wie Entwickeln Sich die Keimzahlen Im Schlachtprozess? Schweizer Geflügelzeitung 12:12–13
Svobodová I, Bořilová G, Hulánková R, Steinhauserová I (2012) Microbiological quality of broiler carcasses during slaughter processing. Acta Vet Brno 81(1):37–42. https://doi.org/10.2754/avb201281010037
Zweifel C, Althaus D, Stephan R (2015) Effects of slaughter operations on the microbiological contamination of broiler carcasses in 3 abattoirs. Food Control 51:37–42. https://doi.org/10.1016/j.foodcont.2014.11.002
Acknowledgements
The authors like to thank Prof. Dr. Uwe Rösler, Institute for Animal Hygiene and Environmental Health, Freie Universität Berlin for coordinating the joint research project “EsRAM”.
We thank Prof. Dr. Thomas Alter, Institute of Food Safety and Food Hygiene, Freie Universität Berlin for his help with laboratory work and administrative issues.
The authors thank Ricardo Hernández Arrondo from Marel for his help describing the technical parts of the AeroScalder®.
We acknowledge the support of the included abattoirs and their assistance in sampling.
Funding
Open Access funding enabled and organized by Projekt DEAL. The study was conducted in the framework of the joint research project “EsRAM”. This project was supported by funds of the Federal Ministry of Food and Agriculture (BMEL) based on a decision of the Parliament of the Federal Republic of Germany via the Federal Office for Agriculture and Food (BLE) under the innovation support program; grand ID: 2817701014.
Open Access funding enabled and organized by Projekt DEAL.
Author information
Authors and Affiliations
Contributions
Conceptualization and methodology: N.L., L.E., R.F., and D.M.; formal analysis: R.M. and N.L.; investigation: S.F. and N.L.; data curation: S.F. and N.L.; writing—original draft preparation: N.L. and R.M.; writing—review and editing: R.F., D.M., L.E., R.M., and N.L.; supervision: D.M., L.E., and N.L.; project administration: N.L.; funding acquisition: R.F.
All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors do not have any conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Langkabel, N., Freter, S., Merle, R. et al. Enterobacteriaceae counts influenced by different scalding techniques in broiler processing. J Consum Prot Food Saf 19, 49–58 (2024). https://doi.org/10.1007/s00003-023-01470-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00003-023-01470-9