Abstract
Synthesis and investigation of antimicrobial activity of fifteen novel thiazoles containing cyclohexene moiety are presented. Among the derivatives, compounds 3a–3d, 3f, 3n, and 3o showed very strong activity against the reference Candida spp. strains with MIC = 0.015–3.91 µg/ml. The activity of these compounds is similar and even higher than the activity of nystatin used as positive control. Compounds 3d, 3f, 3n, 3o showed the highest activity with very strong effect towards most of yeasts isolated from clinical materials with MIC = 0.015–7.81 µg/ml. The cytotoxicity studies for the most active compounds showed that Candida spp. growth was inhibited at noncytotoxic concentrations for the mammalian L929 fibroblast. In addition, a good correlation was obtained between lipophilicity of compounds determined using reversed phase thin-layer chromatography and their antifungal activity.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Nowadays, the incidence of Invasive Fungal Infection is increasing worldwide. They are mostly caused by yeasts belonging to Candida spp., especially Candida albicans, but most recently also by other species like Candida glabrata, Candida tropicalis, Candida parapsilosis, or Candida krusei called as non-albicans Candida species (Ishida et al. 2011; Silva et al. 2017; Pappas et al. 2016). These fungi are a part of the microbiota colonizing mucocutaneus areas, with a reported prevalence 15–75%, mainly within the oral cavity, upper airways, gastrointestinal tract, and vagina of healthy individuals (Ghannoum et al. 2010). They are one of the most important opportunistic fungal pathogens causing mucosal and systemic infections (Hacioglu et al. 2018). Invasive candidiasis are considered the life-threatening infections associated with high morbidity and mortality (Pappas et al. 2016; Hacioglu et al. 2018). The mortality rate of bloodstream infections caused by these yeasts was reported to be as high as 40–60% among immunosuppressed and hospitalized patients (Sun et al. 2015; Li et al. 2018; Turecka et al. 2018).
The incidence of fungal infection is increasing, especially in populations of immunosuppressed individuals (persons with HIV/AIDS or primary immune deficiency) and due to the continuous development of modern medicines, such as organ transplantation, hemodialysis, parenteral nutrition, extensive applicability of catheters or medical implants, cancer chemotherapy, and the widespread use of broad-spectrum antibiotics, glucocorticoids, and immunosuppressants (Turecka et al. 2018; Roemer and Krysan 2014; Gomes da Silva Dantas et al. 2018). In addition to the predisposing factors of the host, the pathogenicity of Candida species is affected by their virulence factors, such as the polymorphism (growth in two forms, unicellular and filamentous), production of extracellular enzymes (proteases, phospholipases, and hemolysins), as well as the expression of adhesins, which is related to the biofilm formation (on host tissues or on medical device surfaces) (Hacioglu et al. 2018; Sun et al. 2015; Li et al. 2018; Gomes da Silva Dantas et al. 2018).
At present, the available antifungal drugs are limited to three major classes: polyenes (amphotericin B or nystatin), which bind fungal cell membrane ergosterol leading to cell lysis; azoles (fluconazole or posaconazole) that inhibit ergosterol biosynthesis; and echinocandins (caspofungin or micafungin) inhibiting fungal (1,3)-β-D-glucan cell wall synthesis (Ishida et al. 2011; Pappas et al. 2016; Li et al. 2018; Turecka et al. 2018; Roemer and Krysan 2014; Ahmad et al. 2017). Some characteristics of these drugs restrict their use in prophylaxis and clinical practice, such as fungistatic activity (azoles), which extends the required time of treatment, high toxicity (polyenes) and nonspecific interaction with other drugs, which increases their side effects (Gomes da Silva Dantas et al. 2018; Ahmad et al. 2017). Moreover, as a consequence of the wide use of these antifungals, drug resistance of Candida spp. is increasing, which poses a serious threat to antifungal therapy (Li et al. 2018). Thus, discovering new antifungal agents or overcoming drug resistance become a hot topic in the antifungal field (Li et al. 2018; Ahmad et al. 2017).
The last years of the research have confirmed that thiazole scaffold has diverse biological properties that can be used to treat many diseases, such as microbial infections (Bikobo et al. 2017; Secci et al. 2012; Łączkowski et al. 2018a, 2018b; Zaki et al. 2018; Ansari et al. 2018), Parkinson’s disease (Carradori et al. 2018), cancer (de Santana et al. 2018; Łączkowski et al. 2014, 2016a, 2016b; Piechowska et al. 2019; Gomha et al. 2018), epilepsy (Siddiqui and Ahsan 2010; Ahangar et al. 2011; Łączkowski et al. 2016a, 2016b, 2017), toxoplasmosis (Schultz et al. 2014; Łączkowski et al. 2018a, 2018b), and antioxidants (Geronikaki et al. 2013; Djukic et al. 2018). Considering the above-mentioned findings, we decided to design and synthesize the structure of hybrids incorporating both the thiazole ring and the cyclohexenyl moiety. Our earlier studies as well as literature data suggested that such hybrid structures will show some synergistic effects responsible for achieving high antimicrobial activity (Secci et al. 2012). The investigated compounds have been rationalized, so that they contain such substituents that in earlier studies were responsible for the highest antimicrobial activity (Fig. 1). Thanks to the use of electron-donating as well as electron-withdrawing character of substituents we were able to modify the hydrophilic and hydrophobic properties of the designed molecules.
Next, the synthesized compounds were evaluated for their antimicrobial activity against a panel of 32 different strains of microorganisms, including Gram-positive bacteria, Gram-negative bacteria, and fungi belonging to yeasts. These microorganisms came from American Type Culture Collection (ATCC), routinely used for the evaluation of antimicrobials. Moreover, the isolates of Candida spp. from clinical materials were included.
Materials and methods
Chemistry
All experiments were carried out under air atmosphere unless stated otherwise. Reagents were generally the best quality commercial-grade products and were used without further purification. 1H NMR (400 MHz) and 13C NMR (176 MHz) spectra were recorded on a Bruker Avance III multinuclear instrument. Melting points were determined in open glass capillaries and are uncorrected. Analytical TLC was performed using Macherey-Nagel Polygram Sil G/UV254 0.2 mm plates. 3-Cyclohexene-1-carboxaldehyde, thiosemicarbazide, and appropriate bromoketones were commercial materials (Aldrich).
2-(Cyclohex-3-enylmethylene)hydrazinecarbothioamide (2)
Thiosemicarbazide (1.82 g, 20.0 mmol) was added to a stirred solution of 3-cyclohexene-1-carboxaldehyde (1) (2.20 g, 20.0 mmol) in absolute ethyl alcohol (30 ml) and then (1.0 ml) of acetic acid was added. The reaction mixture was stirred under reflux for 20 h under nitrogen atmosphere. Next, the reaction mixture was added to water (100 ml) and neutralized with NaHCO3 solution. The product was filtered off and subsequently washed with water to afford the desired product to yield 2.93 g (80%); mp 122–123 °C; eluent: dichloromethane/methanol (95:5), Rf = 0.74. 1H NMR (DMSO-d6, 700 MHz) δ (ppm): 1.41–1.46 (m, 1H, CH); 1.78–1.84 (m, 1H, CH); 2.00–2.06 (m, 3H, CH, CH2); 2.08–2.14 (m, 1H, CH); 2.41–2.44 (m, 1H, CH); 5.66 (s, 2H, 2CH); 7.39 (d, 1H, CH, J = 5.0 Hz); 7.44 (bs, 1H, NH); 7.98 (bs, 1H, NH); 11.05 (bs, 1H, NH). 13C NMR (176 MHz, DMSO-d6) δ (ppm): 24.29 (CH2); 26.06 (CH2); 28.28 (CH2); 36.32 (CH); 125.95 (CH); 127.18 (CH); 150.54 (CH=N); 178.18 (C=S). Anal. Calcd. for C8H13N3S: C, 52.43; H, 7.15; N, 22.93. Found: C, 52.45; H, 7.20; N, 22.98.
2-(2-(Cyclohex-3-enylmethylene)hydrazinyl)-4-(4-fluorophenyl)thiazole (3a). Typical procedure
Carbothioamide 2 (0.183 g, 1.0 mmol) was added to a stirred solution of 2-bromo-1-(4-fluorophenyl)ethanone (0.217 g, 1.0 mmol) in absolute ethyl alcohol (15 ml). The reaction mixture was stirred at room temperature for 20 h. Next, the reaction mixture was added to water (50 ml) and neutralized with NaHCO3 solution. The product was extracted with dichloromethane (2 × 100 ml), the solvent was evaporated in vacuo, and the product was purified on silica gel column chromatography (230–400 mesh) using (dichloromethane, Rf = 0.51) to afford the desired product: 0.27 g, 90%; mp 153–155 °C. 1H NMR (DMSO-d6, 400 MHz) δ (ppm): 1.41–1.46 (m, 1H, CH); 1.78–1.84 (m, 1H, CH); 2.00–2.06 (m, 3H, CH, CH2); 2.08–2.14 (m, 1H, CH); 2.46–2.49 (m, 1H, CH); 5.69 (s, 2H, 2CH); 7.15–7.24 (m, 3H, 3CH); 7.34 (d, 1H, CH, J = 5.0 Hz); 7.84–7.87 (m, 2H, 2CH); 11.59 (bs, 1H, NH). 13C NMR (176 MHz, DMSO-d6) δ (ppm): 23.90 (CH2); 25.84 (CH2); 28.17 (CH2); 36.01 (CH); 102.78 (CH); 115.45 (d, 2CH; JC–F = 19.7 Hz); 125.53 (CH); 126.87 (CH); 127.50 (d, 2CH, JC–F = 6.2 Hz); 131.51 (C); 148.77 (C); 160.90 (CH=N); 162.27 (C–F); 168.78 (C–NH). Anal. Calcd. for C16H16FN3S: C, 63.76; H, 5.35; N, 13.94. Found: C, 63.74; H, 5.40; N, 13.99.
4-(4-Bromophenyl)-2-(2-(cyclohex-3-enylmethylene)hydrazinyl)thiazole (3b)
2-Bromo-1-(4-bromophenyl)ethanone was reacted with 2. Yield: 0.29 g, 80%, (dichloromethane, Rf = 0.72); mp 165–167 °C. 1H NMR (DMSO-d6, 400 MHz) δ (ppm): 1.41–1.46 (m, 1H, CH); 1.78–1.84 (m, 1H, CH); 2.00–2.06 (m, 3H, CH, CH2); 2.08–2.14 (m, 1H, CH); 2.46–2.49 (m, 1H, CH); 5.69 (s, 2H, 2CH); 7.31 (s, 1H, CH); 7.35–7.38 (d, 1H, CH, J = 5.0 Hz); 7.59 (d, 2H, 2CH, J = 8.0 Hz); 7.77 (d, 2H, 2CH, J = 8.0 Hz); 11.64 (bs, 1H, NH). 13C NMR (176 MHz, DMSO-d6) δ (ppm): 23.85 (CH2); 25.76 (CH2); 28.04 (CH2); 36.05 (CH); 104.31 (CH); 120.88 (C); 125.46 (CH); 126.87 (CH); 127.74 (2CH); 131.61 (2CH); 132.97 (C); 147.50 (C); 150.54 (CH=N); 168.91 (C–NH). Anal. Calcd. for C16H16BrN3S: C, 53.04; H, 4.45; N, 11.60. Found: C, 53.02; H, 4.50; N, 11.66.
4-(4-Chlorophenyl)-2-(2-(cyclohex-3-enylmethylene)hydrazinyl)thiazole (3c)
2-Bromo-1-(4-chlorophenyl)ethanone was reacted with 2. Yield: 0.28 g, 88%, (dichloromethane, Rf = 0.36); mp 155–158 °C. 1H NMR (DMSO-d6, 400 MHz) δ (ppm): 1.41–1.46 (m, 1H, CH); 1.78–1.84 (m, 1H, CH); 2.00–2.06 (m, 3H, CH, CH2); 2.08–2.14 (m, 1H, CH); 2.46–2.49 (m, 1H, CH); 5.69 (s, 2H, 2CH); 7.29 (s, 1H, CH); 7.36 (d, 1H, CH, J = 5.0 Hz); 7.45 (d, 2H, 2CH, J = 8.0 Hz); 7.84 (d, 2H, 2CH, J = 8.0 Hz); 11.61 (bs, 1H, NH). 13C NMR (176 MHz, DMSO-d6) δ (ppm): 23.90 (CH2); 25.88 (CH2); 28.17 (CH2); 36.01 (CH); 103.86 (CH); 125.53 (CH); 126.87 (CH); 127.25 (2CH); 128.63 (2CH); 131.86 (C); 133.73 (C); 148.87 (CH=N); 149.21 (C); 168.82 (C–NH). Anal. Calcd. for C16H16ClN3S: C, 60.46; H, 5.07; N, 13.22. Found: C, 60.50; H, 5.01; N, 13.18.
2-(2-(Cyclohex-3-enylmethylene)hydrazinyl)-4-p-tolylthiazole (3d)
2-Bromo-1-(4-methylphenyl)ethanone was reacted with 2. Yield: 0.29 g, 97%, (dichloromethane, Rf = 0.41); mp 156–158 °C. 1H NMR (DMSO-d6, 400 MHz) δ (ppm): 1.41–1.46 (m, 1H, CH); 1.78–1.84 (m, 1H, CH); 2.00–2.06 (m, 3H, CH, CH2); 2.08–2.14 (m, 1H, CH); 2.31 (s, 3H, CH3); 2.41–2.44 (m, 1H, CH); 5.70 (s, 2H, 2CH); 7.13 (s, 1H, CH); 7.20 (d, 2H, 2CH, J = 8.0 Hz); 7.34 (d, 1H, CH, J = 5.0 Hz); 7.70 (d, 2H, 2CH, J = 8.0 Hz); 11.56 (bs, 1H, NH). 13C NMR (176 MHz, DMSO-d6) δ (ppm): 20.89 (CH3); 23.90 (CH2); 25.91 (CH2); 28.22 (CH2); 36.00 (CH); 102.11 (CH); 125.49 (CH); 125.55 (2CH); 126.87 (CH); 129.19 (2CH); 132.25 (C); 136.71 (C); 148.58 (C); 150.51 (CH=N); 168.57 (C–NH). Anal. Calcd. for C17H19N3S: C, 68.65; H, 6.44; N, 14.13. Found: C, 68.70; H, 6.45; N, 14.18.
2-(2-(Cyclohex-3-enylmethylene)hydrazinyl)-4-(4-(trifluoromethyl)phenyl)thiazole (3e)
2-Bromo-1-(4-trifluoromethylphenyl)ethanone was reacted with 2. Yield: 0.31 g, 89%, (dichloromethane, Rf = 0.54); mp 132–134 °C. 1H NMR (DMSO-d6, 400 MHz) δ (ppm): 1.41–1.46 (m, 1H, CH); 1.78–1.84 (m, 1H, CH); 2.00–2.06 (m, 3H, CH, CH2), 2.08–2.14 (m, 1H, CH); 2.41–2.44 (m, 1H, CH); 5.70 (s, 2H, 2CH); 7.37 (d, 1H, CH, J = 5.0 Hz); 7.47 (s, 1H, CH); 7.75 (d, 2H, 2CH, J = 8.0 Hz); 8.03 (d, 2H, 2CH, J = 8.0 Hz); 11.68 (bs, 1H, NH). 13C NMR (176 MHz, DMSO-d6) δ (ppm): 23.87 (CH2); 25.86 (CH2); 28.15 (CH2); 36.00 (CH); 105.89 (CH); 125.51 (CH); 125.61 (2CH); 126.07 (2CH); 126.87 (CH); 127.52 (q, C, JC–F = 29.5 Hz); 138.47 (C); 148.92 (C); 149.05 (CH=N); 168.93 (C–NH). Anal. Calcd. for C17H16F3N3S: C, 58.11; H, 4.59; N, 11.96. Found: C, 58.08; H, 4.63; N, 12.01.
2-(2-(Cyclohex-3-enylmethylene)hydrazinyl)-4-(4-methoxyphenyl)thiazole (3f)
2-Bromo-1-(4-methoxyphenyl)ethanone was reacted with 2. Yield: 0.28 g, 88%, (dichloromethane, Rf = 0.23); mp 121–123 °C. 1H NMR (DMSO-d6, 400 MHz) δ (ppm): 1.41–1.53 (m, 1H, CH); 1.81–1.89 (m, 1H, CH); 1.98–2.23 (m, 4H, 2CH, CH2), 2.46–2.49 (m, 1H, CH); 3.77 (s, 3H, OCH3); 5.69 (s, 2H, 2CH); 6.96 (d, 2H, 2CH, J = 8.0 Hz); 7.04 (s, 1H, CH); 7.34 (d, 1H, CH, J = 5.0 Hz); 7.75 (d, 2H, 2CH, J = 8.0 Hz); 11.55 (bs, 1H, NH). 13C NMR (176 MHz, DMSO-d6) δ (ppm): 24.28 (CH2); 26.31 (CH2); 28.59 (CH2); 36.38 (CH); 55.57 (CH3); 101.28 (CH); 114.38 (2CH); 125.94 (CH); 127.26 (2CH); 128.18 (CH); 133.37 (C); 148.95 (C); 150.63 (CH=N); 159.16 (C–OCH3); 168.93 (C–NH). Anal. Calcd. for C17H19N3OS: C, 65.15; H, 6.11; N, 13.41. Found: C, 65.20; H, 6.10; N, 13.46.
2-(2-(Cyclohex-3-enylmethylene)hydrazinyl)-4-(4-nitrophenyl)thiazole (3g)
2-Bromo-1-(4-nitrophenyl)ethanone was reacted with 2. Yield: 0.31 g, 93%, (dichloromethane, Rf = 0.33); mp 145–147 °C. 1H NMR (DMSO-d6, 400 MHz) δ (ppm): 1.41–1.46 (m, 1H, CH); 1.78–1.84 (m, 1H, CH); 2.00–2.06 (m, 3H, CH, CH2), 2.08–2.14 (m, 1H, CH); 2.41–2.44 (m, 1H, CH); 5.70 (s, 2H, 2CH); 7.39 (d, 1H, CH, J = 5.0 Hz); 7.63 (s, 1H, CH); 8.09 (d, 2H, 2CH, J = 9.0 Hz); 8.26 (d, 2H, 2CH, J = 9.0 Hz); 11.74 (bs, 1H, NH). 13C NMR (176 MHz, DMSO-d6) δ (ppm): 24.28 (CH2); 26.22 (CH2); 28.52 (CH2); 36.41 (CH); 108.39 (CH); 124.52 (2CH); 125.90 (CH); 126.73 (2CH); 127.26 (CH); 141.25 (C); 146.60 (C); 148.83 (C); 149.69 (CH=N); 169.49 (C-NH). Anal. Calcd. for C16H16N4O2S: C, 58.52; H, 4.91; N, 17.06. Found: C, 58.50; H, 4.93; N, 17.08.
4-(2-(2-(Cyclohex-3-enylmethylene)hydrazinyl)thiazol-4-yl)benzonitrile (3h)
4-(Bromoacetyl)benzonitrile was reacted with 2. Yield: 0.30 g, 98%, (dichloromethane, Rf = 0.26); mp 180–182 °C. 1H NMR (DMSO-d6, 400 MHz) δ (ppm): 1.41–1.46 (m, 1H, CH); 1.78–1.84 (m, 1H, CH); 2.00–2.06 (m, 3H, CH, CH2); 2.08–2.14 (m, 1H, CH); 2.41–2.44 (m, 1H, CH); 5.70 (s, 2H, 2CH); 7.37 (d, 1H, CH, J = 5.0 Hz); 7.54 (s, 1H, CH); 7.85 (d, 2H, 2CH, J = 8.5 Hz); 7.99 (d, 2H, 2CH, J = 8.5 Hz); 11.69 (bs, 1H, NH). 13C NMR (176 MHz, DMSO-d6) δ (ppm): 23.88 (CH2); 25.83 (CH2); 28.13 (CH2); 36.01 (CH); 106.98 (CH); 109.54 (C); 119.08 (C); 125.50 (CH); 126.14 (2CH); 126.86 (CH); 132.72 (2CH); 138.93 (C); 148.74 (C); 149.15 (CH=N); 168.98 (C–NH). Anal. Calcd. for C17H16N4S: C, 66.21; H, 5.23; N, 18.17. Found: C, 66.28; H, 5.25; N, 18.22.
2-(2-(Cyclohex-3-enylmethylene)hydrazinyl)-4-(2,4-difluorophenyl)thiazole (3i)
2-Bromo-1-(2′4′-difluorophenyl)ethanone was reacted with 2. Yield: 0.30 g, 94%, (dichloromethane, Rf = 0.52); mp 130–132 °C. 1H NMR (DMSO-d6, 400 MHz) δ (ppm): 1.41–1.46 (m, 1H, CH); 1.78–1.84 (m, 1H, CH); 2.00–2.06 (m, 3H, CH, CH2); 2.08–2.14 (m, 1H, CH); 2.41–2.44 (m, 1H, CH); 5.70 (s, 2H, 2CH); 7.11–7.19 (m, 2H, 2CH); 7.30–7.38 (m, 2H, 2CH); 7.95–8.04 (m, 1H, CH); 11.63 (bs, 1H, NH). 13C NMR (176 MHz, DMSO-d6) δ (ppm): 24.27 (CH2); 26.25 (CH2); 28.55 (CH2); 36.33 (CH); 104.92 (t, C, JC–F = 47.0 Hz); 107.56 (d, C, JC–F = 27.0 Hz); 112.19 (dd, C, JC–F = 5.9 Hz, JC–F = 37.0 Hz); 112.19 (dd, C, JC–F = 6.8 Hz, JC–F = 19.2 Hz); 125.91 (CH); 127.26 (CH); 130.81 (dd, C, JC–F = 8.5 Hz, JC–F = 16.4 Hz); 143.69 (C); 149.36 (CH=N); 159.53 (dd, C, JC–F = 23.0 Hz, JC–F = 289.0 Hz); 162.02 (dd, C, JC–F = 23.0 Hz, JC–F = 281.0 Hz); 168.54 (C–NH). Anal. Calcd. for C16H15F2N3S: C, 60.17; H, 4.73; N, 13.16. Found: C, 60.21; H, 4.69; N, 13.19.
4-(4-Azidophenyl)-2-(2-(cyclohex-3-enylmethylene)hydrazinyl)thiazole (3j)
2-Bromo-1-(4-azidophenyl)ethanone was reacted with 2. Yield: 0.33 g, 99%, (dichloromethan, Rf = 0.34); mp 124–126 °C. 1H NMR (DMSO-d6, 400 MHz) δ (ppm): 1.41–1.46 (m, 1H, CH); 1.78–1.84 (m, 1H, CH); 2.00–2.06 (m, 3H, CH, CH2); 2.08–2.14 (m, 1H, CH); 2.41–2.44 (m, 1H, CH); 5.70 (s, 2H, 2CH); 7.15 (d, 2H, 2CH, J = 8.5 Hz); 7.23 (s, 1H, CH); 7.35 (d, 1H, CH, J = 5.0 Hz); 7.87 (d, 2H, 2CH, J = 8.5 Hz); 11.61 (bs, 1H, NH). 13C NMR (176 MHz, DMSO-d6) δ (ppm): 23.89 (CH2); 25.88 (CH2); 28.16 (CH2); 36.02 (CH); 102.10 (CH); 119.38 (2CH); 125.53 (CH); 126.87 (CH); 127.17 (2CH); 131.77 (C); 138.28 (C); 149.07 (CH=N); 149.25 (C); 168.73 (C–NH). Anal. Calcd. for C16H16N6S: C, 59.24; H, 4.97; N, 25.91. Found: C, 59.25; H, 5.01; N, 25.95.
2-(2-(Cyclohex-3-enylmethylene)hydrazinyl)-4-(3,4-dichlorophenyl)thiazole (3k)
2-Bromo-1-(3′4′-dichlorophenyl)ethanone was reacted with 2. Yield: 0.22 g, 63%, (dichloromethane, Rf = 0.50); mp 117–120 °C. 1H NMR (DMSO-d6, 400 MHz) δ (ppm): 1.41–1.46 (m, 1H, CH); 1.81–1.89 (m, 1H, CH); 1.98–2.10 (m, 3H, CH, CH2); 2.11–2.20 (m, 1H, CH); 2.42–2.50 (m, 1H, CH); 5.70 (s, 2H, 2CH); 7.36 (d, 1H, CH, J = 5.0 Hz); 7.44 (s, 1H, CH); 7.64 (d, 1H, CH, J = 8.5 Hz); 7.80 (dd, 1H, CH, J = 3.0 Hz, J = 9.0 Hz); 8.04 (d, 1H, CH, J = 2.0 Hz); 11.65 (bs, 1H, NH). 13C NMR (176 MHz, DMSO-d6) δ (ppm): 24.27 (CH2); 26.24 (CH2); 28.54 (CH2); 36.39 (CH); 105.73 (CH); 125.90 (C); 125.97 (C); 127.25 (C); 127.58 (C); 130.06 (C); 131.25 (C); 131.85(C); 137.76 (C); 148.15 (CH=N); 149.59 (C); 169.29 (C–NH). Anal. Calcd. for C16H15Cl2N3S: C, 54.55; H, 4.29; N, 11.93. Found: C, 54.57; H, 4.32; N, 11.96.
4-(Chloromethyl)-2-(2-(cyclohex-3-enylmethylene)hydrazinyl)thiazole (3l)
1,3-Dichloroacetone was reacted with 2. Yield: 0.14 g, 55%, (dichloromethane/methanol (95:5), Rf = 0.73); mp 127–129 °C. 1H NMR (DMSO-d6, 400 MHz) δ (ppm): 1.41–1.46 (m, 1H, CH); 1.78–1.84 (m, 1H, CH), 2.00–2.06 (m, 3H, CH, CH2); 2.08–2.14 (m, 1H, CH); 2.41–2.44 (m, 1H, CH); 4.57 (s, 2H, 2CH); 5.68 (s, 2H, CH2); 6.84 (s, 1H, CH); 7.30 (d, 1H, CH, J = 6.0 Hz); 11.50 (bs, 1H, NH). 13C NMR (176 MHz, DMSO-d6) δ (ppm): 23.85 (CH2); 25.84 (CH2); 28.15 (CH2); 35.96 (CH); 42.01 (CH2-Cl); 107.58 (CH); 125.51 (CH); 126.87 (CH); 147.87 (C–CH2Cl); 148.84 (CH=N); 169.10 (C–NH). Anal. Calcd. for C11H14ClN3S: C, 51.66; H, 5.52; N, 16.43. Found: C, 51.67; H, 5.55; N, 16.46.
2-(2-(Cyclohex-3-enylmethylene)hydrazinyl)-4-(adamant-1-yl)thiazole (3m)
1-Adamantyl bromomethyl ketone was reacted with 2. Yield: 0.31 g, 92%, (dichloromethane, Rf = 0.51); mp 155–157 °C. 1H NMR (DMSO-d6, 400 MHz) δ (ppm): 1.41–1.53 (m, 1H, CH); 1.65–1.76 (m, 7H, 7CH); 1.80–1.88 (m, 7H, 7CH); 1.98–2.09 (m, 6H, 6CH); 2.11–2.20 (m, 1H, CH); 5.69 (s, 2H, 2CH); 6.40 (s, 1H, CH); 7.49 (d, 1H, CH, J = 5.0 Hz); 11.69 (bs, 1H, NH). 13C NMR (176 MHz, DMSO-d6) δ (ppm): 24.12 (CH2); 26.01 (CH2); 28.18 (6CH2); 28.30 (CH2); 36.00 (CH); 36.47 (CH); 36.54 (2CH); 41.14 (C); 100.83 (CH); 125.72 (CH); 127.28 (CH); 153.77 (CH=N); 169.25 (C–NH). Anal. Calcd. for C20H27N3S: C, 70.34; H, 7.97; N, 12.30. Found: C, 70.34; H, 7.99; N, 12.35.
Ethyl 2-(2-(2-(cyclohex-3-enylmethylene)hydrazinyl)thiazol-4-yl)acetate (3n)
Ethyl 4-chloro-3-oxobutanoate was reacted with 2. Yield: 0.20 g, 68%, (dichloromethane, Rf = 0.51); mp 125–127 °C. 1H NMR (DMSO-d6, 400 MHz) δ (ppm): 1.17 (t, 3H, CH3, J = 7.0 Hz); 1.41–1.46 (m, 1H, CH); 1.78–1.84 (m, 1H, CH); 2.00–2.06 (m, 3H, CH, CH2); 2.08–2.14 (m, 1H, CH); 2.41–2.44 (m, 1H, CH); 3.53 (s, 2H, CH2); 4.05 (q, 2H, CH2, J = 7.0 Hz); 5.68 (s, 2H, 2CH); 6.53 (s, 1H, CH); 7.28 (d, 1H, CH, J = 6.0 Hz); 11.39 (bs, 1H, NH). 13C NMR (176 MHz, DMSO-d6) δ (ppm): 14.20 (CH3); 23.86 (CH2); 25.87 (CH2); 28.19 (CH2); 35.95 (CH); 37.15 (CH2); 60.26 (CH2–O); 104.89 (CH); 125.53 (CH); 126.86 (CH); 145.00 (C); 148.44 (CH=N); 168.52 (C=O); 170.13 (C–NH). Anal. Calcd. for C14H19N3O2S: C, 57.31; H, 6.53; N, 14.32. Found: C, 57.28; H, 6.56; N, 14.36.
2-(2-(Cyclohex-3-enylmethylene)hydrazinyl)-4-methylthiazole (3o)
Chloroacetone was reacted with 2. Yield: 0.12 g, 52%, (dichloromethane, Rf = 0.29); mp 128–129 °C. 1H NMR (DMSO-d6, 400 MHz) δ (ppm): 1.39–1.52 (m, 1H, CH); 1.78–1.88 (m, 1H, CH); 1.96–2.01 (m, 3H, CH, CH2); 2.15–2.20 (m, 1H, CH); 2.33 (s, 3H, CH3); 2.39–2.49 (m, 1H, CH); 5.68 (s, 2H, 2CH); 6.28 (s, 1H, CH); 7.29 (d, 1H, CH, J = 5.5 Hz); 11.24 (bs, 1H, NH). 13C NMR (176 MHz, DMSO-d6) δ (ppm): 17.65 (CH3); 24.28 (CH2); 26.34 (CH2); 28.64 (CH2); 36.34 (CH); 102.27 (CH); 125.95 (CH); 127.25 (CH); 148.02 (C–CH3); 148.78 (CH=N); 168.70 (C–NH). Anal. Calcd. for C11H15N3S: C, 59.69; H, 6.83; N, 18.99. Found: C, 59.71; H, 6.83; N, 19.04.
Microbiology
Microbial material
The examined 1,3-tiazole derivatives 3a–3o were screened in vitro for antibacterial and antifungal activities using the broth microdilution method according to European Committee on Antimicrobial Susceptibility Testing (EUCAST) (EUCAST 2003) and Clinical and Laboratory Standards Institute (CLSI) guidelines (Wayne Clinical and Laboratory Standards Institute 2012) against reference strains of microorganisms from American Type Culture Collection (ATCC), including Gram-positive bacteria (Staphylococcus epidermidis ATCC 12228, Staphylococcus aureus ATCC 6538, Staphylococcus aureus ATCC 25923, Staphylococcus aureus ATCC 43300, Micrococcus luteus ATCC 10240, Bacillus cereus ATCC 10876, and Bacillus subtilis ATCC 6633), Gram-negative bacteria (from the Enterobacteriaceae family: Escherichia coli ATCC 25922, Klebsiella pneumoniae ATCC 13883, Proteus mirabilis ATCC 12453, Salmonella typhimurium ATCC 14028 and other: Pseudomonas aeruginosa ATCC 9027 and Bordetella bronchiseptica ATCC 4617) and fungi belonging to yeasts (Candida albicans ATCC 2091, Candida albicans ATCC 10231, Candida glabrata ATCC 90030, Candida parapsilosis ATCC 22019, and Candida krusei ATCC 14243).
In the study of antifungal activity of the compounds 3a–3o 14 clinical strains of different species of yeasts from Candida species were also used, namely C. albicans, C. dubliniensis C. famata, C. glabrata, C. guilliermondii, C. inconspicua, C. kefyr, C. krusei, C. lambica, C. lusitaniae, C. parapsilosis, C. pulcherrima, C. sake, and C. tropicalis. These fungi were isolated by the authors (from Department of Pharmaceutical Microbiology of Medical University in Lublin, Poland) from various clinical materials, e.g., from upper respiratory tract of hospitalized patients including cancer persons (i.e., with lung cancer or hematological malignancies), patients with chronic hepatitis C, patients with diabetes and elderly people, aged of 65 years old or older. The Ethical Committee of the Medical University of Lublin approved the study protocol (No. KE-0254/75/2011). These isolates were identified by standard diagnostic methods–biochemical microtests, e.g., API Candida, API 20 C AUX and ID 32 C (bioMérieux) on the basis of assimilation of different substrates.
Antimicrobial activity
All the used microbial cultures were first subcultured on appropriate nutrient media. Microbial suspensions were prepared in 0.85% NaCl with an optical density of 0.5 McFarland standard scale. The surface of Mueller–Hinton agar and RPMI 1640 with MOPS were inoculated with the suspensions of bacterial or fungal species, respectively. The examined compounds 3a–3o were first dissolved in dimethyl sulfoxide (DMSO). Next, suspensions were plated on solid media containing 2 mg/ml of the tested compounds followed incubation under appropriate conditions. The inhibition of microbial growth was assessed by comparison with a control culture prepared without any sample tested. Ciprofloxacin or nystatin (Sigma) were used as a reference antibacterial or antifungal compounds, respectively.
Subsequently minimal inhibitory concentration (MIC) of these compounds was examined by the two-fold microdilution broth method in Mueller-Hinton broth (for bacteria) and RPMI 1640 broth with MOPS (for fungi) prepared in 96-well polystyrene plates. Final concentrations of the compounds in the liquid media ranged from 1000 to 0.0038 µg/ml. Microbial suspensions were added per each well containing broth and various concentrations of the examined compounds. After incubation, the MIC was determined spectrophotometric as the lowest concentration of the samples showing complete bacterial or fungal growth inhibition. Appropriate DMSO, sterile, and growth controls were carried out. The media with no tested substances were used also as controls.
The minimal bactericidal concentration (MBC) or minimal fungicidal concentration (MFC) are defined as the lowest concentration of the compound that is required to kill a particular bacterial or fungal species. MBC or MFC of substances 3a–3o were assessed by removing the culture using for MIC determinations from each well and spotting onto appropriate agar medium. The plates were incubated. The lowest compounds concentrations with no visible growth observed were assessed as a bactericidal or fungicidal concentrations. All the experiments were repeated three times and representative data are presented (Wiegand et al. 2008).
In this study was defined: no bioactivity (MIC > 1000 µg/ml), mild (MIC = 501–1000 µg/ml), moderate (MIC = 126–500 µg/ml), good (MIC = 26–125 µg/ml), strong (MIC = 10–25 µg/ml), and very strong bioactivity (MIC < 10 µg/ml). The MBC/MIC or MFC/MIC ratios were calculated in order to determine bactericidal/fungicidal (MBC/MIC ≤ 4, MFC/MIC ≤ 4) or bacteriostatic/fungistatic (MBC/MIC > 4, MFC/MIC > 4) effect of the tested compounds (O'Donnell et al. 2010).
Cytotoxic assay
Preparation of compounds and drugs
Selected compounds 3a, 3d, 3f, 3n, and 3o were dissolved in dimethyl sulfoxide (DMSO, Sigma-Aldrich, St. Louis, MO, USA) to 50 mg/ml. Dilution of the all compounds were freshly prepared before the cells were exposed. The final concentration of DMSO in the compounds dilutions was not higher than 1.00%.
Cell viability assay
Cell viability assay were performed according to international standards (ISO 10993-5:2009(E)), using the tetrazolium salt [3-(4,5-dimethylthiazol-2-yl)- 2,5-diphenyltetrazolium bromide] (MTT, Sigma-Aldrich) and mouse adherent fibroblasts L929 cells (ATTC® CCL-1™). Briefly, 1 × 104/100 μL/well of L929 cells placed into 96-well plates were incubated 24 h in 37 °C and 10% CO2 to achieve a confluent monolayer. Afterwards, the growth medium (GM) was replaced by 100 μL of the compounds dilutions in culture medium (CM) and the cells were treated next 24 h. In addition, cells were treated with a 4.0–0.03% concentration of DMSO and 1 M NaOH as the compound solvent (data not shown). Then, 50 μL of 1 mg/ml of MTT solution in CM was added to each well for more 2 h (37 °C, 10% CO2) incubation. Following, CM was carefully aspirated and 150 μL of DMSO and 25 μL 0.1M glycine buffer (pH = 10.5) (Sigma-Aldrich) was added. The optical density at 570 nm on the ELISA reader (Multiskan EX, Labsystems, Vienna, VA, USA), was read. The results were expressed as a percentage of viability compared with untreated cells. All experiments were performed in triplicate.
Growth Medium (GM)—RPMI 1640 media R8758 (Sigma-Aldrich), supplemented with 5% fetal bovine serum (FBS, ATCC® 30-2020™), 100 U/ml penicillin and 100 μg/ml streptomycin (Penicillin–Streptomycin Solution ATCC® 30-2300™).
Culture Medium (CM)—RPMI 1640, without phenol red (Biowest, Nuaille, France), supplemented with 5% fetal bovine serum (FBS, ATCC® 30-2020™), 2 mM l-glutamine (Sigma-Aldrich), 100 U/ml penicillin and 100 μg/ml streptomycin (Penicillin–Streptomycin Solution ATCC® 30-2300™).
Lipophilicity
Experimental lipophilicity of the synthesized thiazole derivatives 3a–3o was determined using reversed phase thin-layer chromatography (RP-TLC–reversed phase thin-layer chromatography) on 10 × 10 cm HPTLC plates coated with C18 silica F254 (Merck, Darmstadt, Germany). Binary solvents were prepared by mixing appropriate volumes of water and one polar modifier (60–80% acetone, 60–90% acetonitrile, 60–85% 1,4-dioxane, 70–95% methanol, in volumes increasing by 5%). All organic modifiers were of analytical grade and they were supplied by POCH (Gliwice, Poland) and Merck (Darmstadt, Germany). The examined compounds 3a–3o and reference substances with known lipophilicity: 2-aminophenol (S1), 8-hydroxyquinoline (S2), 2-naphtol (S3), diphenylamine (S4), 3,4-benzopyrene (S5) were dissolved in methanol to obtain the concentrations 2.0 mg/ml. Volumes of 0.2 μl were spotted to the plates. The chromatograms were developed to a distance of 9 cm from the origin, in a horizontal Teflon chamber with an eluent distributor (Chromdes, Lublin, Poland) at 23 ± 1 °C. After developing, the spots were located under ultraviolet (UV) illumination at 254 nm.
Results and discussion
Chemistry
Synthesis of the title compounds was accomplished following the well known Hantzsch cyclization reaction. In the first step 2-(cyclohex-3-enylmethylene)hydrazinecarbothioamide (2) was synthesized with 80% yield through condensation of 3-cyclohexene-1-carboxaldehyde with thiosemicarbazide in absolute ethyl alcohol containing catalytic amount of glacial acetic acid. In the next step cyclization of hydrazinecarbothioamide 2 with variety of para-substituted bromoacetophenones in ethanolic solution and under room temperature produced (2-(cyclohex-3-enylmethylene)-hydrazinyl)thiazole 3a–3o with high yield (52–99%) and with high chemical purity. The reaction pathway has been summarized in Scheme 1. All obtained products were purified on silica gel column chromatography, and fully characterized spectroscopically using 1H and 13C NMR, and elemental analyses.
In the 1H NMR spectrum of compound 2, three characteristic signals derived from the NH2 and NH groups at 7.44, 7.98 and 11.05 ppm can be observed. These three signals characteristic for hydrazinecarbothioamides are the result of the exchange of a hydrogen atom between the NH2 group and the sulfur atom. The 13C NMR of carbon atoms present in C=N and C–SH groups resonate around 150 and 178 ppm, respectively. Also 1H NMR spectra of (2-(cyclohex-3-enylmethylene)hydrazinyl)thiazole (3a–3o) showed characteristic singlet at δ (6.28–7.63) ppm due to the presence of the H-5 atom in the thiazole ring and broadened hydrazine NH singlet at δ (11.24–11.74) ppm. The 13C NMR of carbon atoms present in C=N and C–NH groups resonate around 150 and 170 ppm, respectively, which proves that the conversion of substrates to the expected products was successful. Purity of the products was confirmed by the elemental analyses, whose results were in good agreement with the calculated values. All reactions were repeated at least two times and are fully reproducible.
Biological evaluation
Antifungal activity
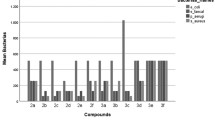
According to the results presented in Table 1, on the basis of minimal inhibitory concentration values obtained by the broth microdilution method, it was shown that compounds 3a–3d, 3f, 3n, and 3o exhibited very strong activity against reference Candida spp. with MIC = 0.015–3.91 µg/ml. The activity of these compounds is similar and even higher than the activity of nystatin used as positive control. The compounds 3n and 3o showed the highest activity with very strong fungicidal effect towards yeasts with MIC = 0.015–0.03 µg/ml and similar MFC = 0.015–0.06 µg/ml (MFC/MIC = 1–4) (Tables 1 and 2). Moreover, the compounds 3h–3j indicated very strong anticandidal effect (MIC = 0.24–7.81 µg/ml, MFC = 1.95 to ≥1000 µg/ml) towards reference C. albicans and C. krusei strains. Also substance 3l with MIC = 1.95–125 µg/ml and MFC = 3.91 to >1000 µg/ml had the high activity (Tables 1 and 2). The compounds 3e, 3k, 3m did not show any antifungal effect against reference Candida spp. strains. All tested compounds were inactive towards C. glabrata ATTC 90030, which is naturally insensitive to some antimycotics, especially to the azole drugs (Tumbarello et al. 2008). In addition, compounds 3g–3j were inactive towards C. parapsilosis ATTC 22019.
In view of the fact that these substances were highly active against the reference yeasts, we decided to examine their activity against the clinical isolates of Candida spp. The compounds 3n and 3o showed the highest activity with very strong antifungal effect towards most of yeasts (C. albicans, C. dubliniensis C. famata, C. inconspicua, C. krusei, C. tropicalis, C. lusitaniae, C. parapsilosis, C. guilliermondii, and C. sake) with MIC = 0.015–3.91 µg/ml and MFC = 0.015–125 µg/ml (Tables 1 and 2). The anticandidal effect was both fungicidal (MFC = 1–4) and fungistatic (MFC = 8–64). The activity of these substances was similar to nystatin. The compounds 3d and 3f showed also very strong activity towards C. albicans, C. dubliniensis C. inconspicua, C. krusei, C. tropicalis, C. lusitaniae, and C. sake (MIC = 3.91–7.81 µg/ml and MFC = 7.81 to ≥1000 µg/ml). The substances 3a–3c had slightly lower effect in relation to the same clinical isolates (MIC = 1.95–125 µg/ml and MFC = 7.81 to ≥1000 µg/ml). Substances 3a–3d and 3f indicated a weaker activity against other strains of Candida such as C. kefyr, C. famata, C. guilliermondii, and C. parapsilosis with MIC = 31.25–1000 µg/ml and MFC = 500 to ≥1000 µg/ml. Also compounds 3g–3j and 3l showed some anticandidal activity. These studies showed that the most susceptible to substances 3a–3o was C. lusitaniae, while C. glabrata, C. pulcherrima, and C. lambica were the least sensitive to them. The antifungal effect was different - both fungicidal (MFC/MIC = 1–4) and fungistatic (MFC/MIC = 8 to >256) (Table 2). The compounds 3e, 3k, and 3m did not show any activity towards clinical isolates.
Antibacterial activity
The studies of activity of the compounds 3a–3o against bacteria demonstrated that compounds 3b, 3c, 3g, 3k–3m, and 3o exhibited some activity against reference Gram-positive bacteria and Bordetella bronchiseptica ATTC 4617 belonging to Gram-negative microorganisms. Among them, the compound 3k exhibited moderate or good effect (MIC = 62.5–500 µg/ml and MBC = 500 to ≥1000 µg/ml) against these bacteria as well as compound 3o had very strong effect towards B. bronchiseptica ATTC 4617 (MIC = 0.06 µg/ml and MBC = 0.48 µg/ml), moderate activity against Bacillus cereus ATCC 10876 (MIC = 500 µg/ml and MBC > 1000 µg/ml) and mild effect towards the rest of these bacteria (MIC = 1000 µg/ml, MBC ≥ 1000 µg/ml). The compounds 3l and 3n showed strong (MIC = 15.62 µg/ml and MBC > 62.5 µg/ml) and very strong activity (MIC = 0.06 µg/ml and MBC = 0.24 µg/ml) towards B. bronchiseptica ATTC 4617, respectively.
The structure–activity relationship analysis showed that the type of substituent has a decisive influence on the activity. As can be seen, compounds 3n and 3o containing CH2COOEt and CH3 groups directly bound to the thiazole ring show the highest activity against Candida spp. Also, compounds 3d and 3f containing CH3 and OCH3 substituents in the phenyl ring show high activity against yeasts. These results show that compounds containing electron-donating groups promote high antifungal activity of compounds. The only exception is the adamantyl group in compound 3m, which did not show any activity towards Candida spp., and the only explanation of this fact is that this group is sterically hindered. Also compounds 3e and 3k containing the CF3 and 3,4-diCl substituents did not show any activity against Candida spp. It can be noticed that the presence of electron-withdrawing groups reduces the activity of compounds. An important result that we have noticed earlier in our research is the lack of activity of derivatives containing the CF3 group.
Cytotoxicity against L929 cells
Selected compounds 3a, 3d, 3f, 3n, and 3o were tested for their cytotoxicity against mouse adherent fibroblast cells line (L929 - ATTC® CCL-1™) according to the international standards: ISO 10993-5:2009(E) using MTT assay. The percent of viable cells ± standard derivation in the concentrations range between 0 to 50.00 µg/ml together with the no cytotoxic concentration values are presented in Table 3. The results of the cytotoxicity evaluation showed that Candida spp. growth was inhibited at noncytotoxic concentrations for the mammalian L929 fibroblast (NCC 10–50 µg/ml).
Lipophilicity determination
On the basis of retardation coefficients (RF) for reference substances and tested compounds, RM values were calculated using the equation: RM = log (1–RF/RF). Then, RM0 values (equivalent to the retention of a solute extrapolated to pure water as mobile phase) were calculated using the equation: RM = RM0–Sφ, where φ is the volume fraction of the organic modifier in the mobile phase and S is the slope of the regression curve (Tables 4 and 5).
The calculated RM0 values for the reference substances were correlated with their log P values found in the literature (Komsta et al. 2010) and appropriate calibration curves were obtained with sufficient linearity:
The lipophilicity of the synthesized 1,3-tiazole derivatives 3a–3o was calculated on the basis of the above calibration equations and their RM0 values. The calculated lipophilicity values were presented in Table 6.
It is well known that RP-TLC method allows fast and reproducible determination of lipophilicity values, especially for a wide range of new synthesized compounds. In the present study, determination coefficients (r2) obtained for calibration equations were sufficiently high (>0.75) providing accuracy of further lipophilicity determination. At the same time, lipophilicity of the reference substances was highly correlated with their RM0 values (Table 5). As far as the tested compounds were concerned, the determination coefficients were also sufficiently high (>0.95) for all eluents. Lipophilicity values obtained for the tested compounds were similar to each other using eluents containing methanol, acetonitrile, and acetone while higher values were calculated for 1,4-dioxane systems. At the same time, in 1,4-dioxane systems, the calculated r2 were the highest values (almost all r2 > 0.99). In the literature, methanol and 1,4-dioxane were recommended as the most suitable organic modifiers for such estimations (Rutkowska et al. 2013).
The lowest lipophilicity values were obtained for the compounds 3n and 3o containing the CH2COOEt and CH3 group, respectively. These two compounds also had the highest antifungal activity. Similarly, the compound 3l with the CH2Cl substituent showed low lipophilicity value. However, probably due to the presence of electron-withdrawing chlorine atom, its antifungal activity is only moderate. On the contrary, the highest values of lipophilicity were obtained for the compounds 3e, 3m, and 3k, independently of the organic modifier used in the binary solvent. This substances, as it turned out, showed no antimicrobial activity. As can be seen, while the additional phenyl ring was present in the structure, the lipophilicity increased (3d, 3e, 3f). When fluoride atoms were introduced instead the chlorine ones, the lipophilicity decreased (3i versus 3k). It was also confirmed that removal of chlorine atom decreased the lipophilicity (3c versus 3k). When CF3 group was introduced to the structure instead of chlorine atom, a small increase of lipophilicity occurred (3e versus 3c).
Conclusions
As a result of our research, we have identified a promising scaffold, (2-(cyclohex-3-enylmethylene)hydrazinyl)thiazole which significantly increases antifungal activity in all tested Candida spp. strains. Among the derivatives, compounds 3a–3d, 3f, 3n, and 3o showed very strong activity against reference Candida spp., and also towards most of yeasts isolated from clinical materials with MIC = 0.015–7.81 µg/ml. The activity of these compounds is similar and even higher than the activity of nystatin used as positive control. The cytotoxicity studies for the most active compounds showed that Candida spp. growth was inhibited at noncytotoxic concentrations for the mammalian L929 fibroblast. In addition, a good correlation was obtained between lipophilicity of compounds determined using reversed phase thin-layer chromatography (RP-TLC) and their antifungal activity. The highest antifungal activity was demonstrated by the compounds 3n and 3o containing the CH2COOEt and CH3 group, which are characterized by the lowest lipophilicity values. All these results provide a new understanding of the relationship between the structure of thiazole derivatives, their lipophilicity and antifungal activity.
References
Ahangar N, Ayati A, Alipour E, Pashapour A, Foroumadi A, Emami S (2011) 1-((2-Arylthiazol-4-yl)methyl)azoles as a new class of anticonvulsants: design, synthesis, in vivo screening, and in silico drug-like properties. Chem Biol Drug Des 78:844–852
Ahmad A, Wani MY, Patel M, Sobrals AJFN, Duse AG, Aqlan FM, Al-Bogami AS (2017) Synergistic antifungal effect of cyclized chalcone derivatives and fluconazole against Candida albicans. Med Chem Comm 8:2195–2207
Ansari A, Ali A, Asif M, Rauf MA, Shamsuzzaman OM (2018) Facile one-pot multicomponent synthesis and molecular docking studies of steroidal oxazole/thiazole derivatives with effective antimicrobial, antibiofilm and hemolytic properties. Steroids 134:22–36
Bikobo DSN, Vodnar DC, Stana A, Tiperciuc B, Nastasă C, Douchet M, Oniga O (2017) Synthesis of 2-phenylamino-thiazole derivatives as antimicrobial agents. J Saudi Chem Soc 21:861–868
Carradori S, Ortuso F, Petzer A, Bagetta D, De Monte C, Secci D, De Vita D, Guglielmi P, Zengin G, Aktumsek A, Alcaro S, Petzer JP (2018) Design, synthesis and biochemical evaluation of novel multi-target inhibitors as potential anti-Parkinson agents. Eur J Med Chem 143:1543–1552
Clinical and Laboratory Standards Institute (2012) Reference method for broth dilution antifungal susceptibility testing of yeasts. M27-S4. Clinical and Laboratory Standards Institute, Wayne, PA, USA
Djukic M, Fesatidou M, Xenikakis I, Geronikaki A, Angelova VT, Savic V, Pasic M, Krilovic B, Djukic D, Gobeljic B, Pavlica M, Djuric A, Stanojevic I, Vojvodic D, Saso L (2018) In vitro antioxidant activity of thiazolidinone derivatives of 1,3-thiazole and 1,3,4-thiadiazole. Chem Biol Interact 286:119–131
European Committee for Antimicrobial Susceptibility Testing (EUCAST) determination of minimum inhibitory concentrations (MICs) of antibacterial agents by broth dilution (2003) EUCAST discussion document E. Dis 5.1. Clin Microbiol Infect 9:1–7
Ghannoum MA, Jurevic RJ, Mukherjee PK, Cui F, Sikaroodi M, Naqvi A, Gillevet PM (2010) Characterization of the oral fungal microbiome (mycobiome) in healthy individuals. PLoS Pathog 6:e1000713
Gomha SM, Abdelhamid AO, Abdelrehem NA, Kandeel SM (2018) Efficient synthesis of new benzofuran‐based thiazoles and investigation of their cytotoxic activity against human breast carcinoma cell lines. J Heterocycl Chem 55:995–1001
Geronikaki AA, Pitta EP, Liaras KS (2013) Thiazoles and thiazolidinones as antioxidants. Curr Med Chem 20:4460–4480
Gomes da Silva Dantas F, Araújo de Almeida-Apolonio A, Pires de Araújo R, Regiane Vizolli Favarin L, Fukuda de Castilho P, de Oliveira Galvão F, Estivalet Svidzinski TI, Casagrande GA, Pires de Oliveira KM (2018) A promising copper (II) complex as antifungal and antibiofilm drug against yeast infection. Molecules 23:1856–1869
Hacioglu M, Tan ASB, Dosler S, Inan N, Otuk G (2018) In vitro activities of antifungals alone and in combination with tigecycline against Candida albicans biofilms. PeerJ 6:e5263
Ishida K, Fernandes Rodrigues JC, Cammerer S, Urbina JA, Gilbert I, de Souza W, Rozental S (2011) Synthetic arylquinuclidine derivatives exhibit antifungal activity against Candida albicans, Candida tropicalis and Candida parapsilopsis. Ann Clin Microbiol Antimicrob 10:3
Komsta Ł, Skibiński R, Berecka A, Gumieniczek A, Radkiewicz B, Radoń M (2010) Revisiting thin-layer chromatography as a lipophilicity determination tool—a comparative study on several techniques with a model solute set. J Pharm Biomed Anal 53:911–918
Li Y, Sun L, Lu Ch, Gong Y, Li M, Sun S (2018) Promising antifungal targets against Candida albicans based on ion homeostasis. Front Cell Infect Microbiol 8:286–298
Łączkowski KZ, Konklewska N, Biernasiuk A, Malm A, Sałat K, Furgała A, Dzitko K, Bekier A, Baranowska-Łączkowska A, Paneth A (2018a) Thiazoles with cyclopropyl fragment as antifungal, anticonvulsant, and anti-Toxoplasma gondii agents: synthesis, toxicity evaluation, and molecular docking study. Med Chem Res 27:2125–2140
Łączkowski KZ, Misiura K, Świtalska M, Wietrzyk J, Baranowska-Łączkowska A, Fernández B, Paneth A, Plech T (2014) Synthesis and in vitro antiproliferative activity of thiazole-based nitrogen mustards. The hydrogen bonding interaction between model systems and nucleobases. Anticancer Agents Med Chem 14:1271–1281
Łączkowski KZ, Świtalska M, Baranowska-Łączkowska A, Plech T, Paneth A, Misiura K, Wietrzyk J, Czaplińska B, Mrozek-Wilczkiewicz A, Malarz K, Musioł R, Grela I (2016) Thiazole-based nitrogen mustards: design, synthesis, spectroscopic studies, DFT calculation, molecular docking, and antiproliferative activity against selected human cancer cell lines. J Mol Struct 1119:139–150
Łączkowski KZ, Sałat K, Misiura K, Podkowa A, Malikowska N (2016) Synthesis and anticonvulsant activities of novel 2-(cyclopentylmethylene)hydrazinyl-1,3-thiazoles in mouse models of seizures. J Enzym Inhib Med Chem 31:1576–1582
Łączkowski KZ, Landowska K, Biernasiuk A, Sałat A, Furgała A, Plech T, Malm A (2017) Synthesis, biological evaluation and molecular docking studies of novel quinuclidinone derivatives as potential antimicrobial and anticonvulsant agents. Med Chem Res 26:2088–2104
Łączkowski KZ, Anusiak J, Świtalska M, Dzitko K, Cytarska J, Baranowska-Łączkowska A, Plech T, Paneth A, Wietrzyk J, Białczyk J (2018b) Synthesis, molecular docking, ctDNA interaction, DFT calculation and evaluation of antiproliferative and anti-Toxoplasma gondii activities of 2,4-diaminotriazine-thiazole derivatives. Med Chem Res 27:1131–1148
O'Donnell F, Smyth TJ, Ramachandran VT, Smyth WF (2010) A study of the antimicrobial activity of selected synthetic and naturally occurring quinolones. Int J Antimicrob Agents 35:30–38
Pappas PG, Kauffman CA, Andes DR, Clancy CJ, Marr KA, Ostrosky-Zeichner L, Reboli AC, Schuster MG, Vazquez JA, Walsh TJ, Zaoutis TE, Sobel JD (2016) Executive summary: clinical practice guideline for the management of candidiasis: 2016 update by the Infectious Diseases Society of America. Clin Infect Dis 15:62 e1–50
Piechowska K, Świtalska M, Cytarska J, Jaroch K, Łuczykowski K, Chałupka J, Wietrzyk J, Misiura K, Bojko B, Kruszewski S, Łączkowski KZ (2019) Discovery of tropinone-thiazole derivatives as potent caspase 3/7 activators, and noncompetitive tyrosinase inhibitors with high antiproliferative activity : rational design, one-pot tricomponent synthesis, and lipophilicity determination. Eur J Med Chem 175:162–171
Roemer T, Krysan DJ (2014) Antifungal drug development: challenges, unmet clinical needs, and new approaches. Cold Spring Harb Perspect Med 4:pii: a019703
Rutkowska E, Pająk K, Jóźwiak K (2013) Lipophilicity—methods of determination and its role in medicinal chemistry. Acta Pol Pharm—Drug Res 70:3–18
Silva S, Rodrigues CF, Araújo D, Rodrigues ME, Henriques M (2017) Candida species biofilms’ antifungal resistance. J Fungi 3:8–24
Sun L, Liao K, Wang D (2015) Effects of magnolol and honokiol on adhesion, yeast-hyphal transition, and formation of biofilm by Candida albicans. PLoS ONE 10:e0117695
Secci D, Bizzarri B, Bolasco A, Carradori S, D'Ascenzio M, Rivanera D, Mari E, Polletta L, Zicari A (2012) Synthesis, anti-Candida activity, and cytotoxicity of new (4-(4-iodophenyl)thiazol-2-yl)hydrazine derivatives. Eur J Med Chem 53:246–253
de Santana TI, Barbosa MO, Gomes PATM, da Cruz ACN, da Silva TG, Leite ACL (2018) Synthesis, anticancer activity and mechanism of action of new thiazole derivatives. Eur J Med Chem 144:874–886
Siddiqui N, Ahsan W (2010) Triazole incorporated thiazoles as a new class of anticonvulsants: design, synthesis and in vivo screening. Eur J Med Chem 45:1536–1543
Schultz TL, Hencken CP, Woodard LE, Posner GH, Yolken RH, Jones-Brando L, Carruthers VB (2014) A thiazole derivative of artemisinin moderately reduces Toxoplasma gondii cyst burden in infected mice. J Parasitol 100:516–521
Tumbarello M, Sanguinetti M, Trecarichi EM, La Sorda M, Rossi M, De Carolis E, De Gaetano Donati K, Fadda G, Cauda R, Posteraro B (2008) Fungaemia caused by Candida glabrata with reduced susceptibility to fluconazole due to altered gene expression: risk factors, antifungal treatment and outcome. J Antimicrob Chemother 62:1379–1385
Turecka K, Chylewska A, Kawiak A, Waleron KF (2018) Antifungal activity and mechanism of action of the Co(III) coordination complexes with diamine chelate ligands against reference and clinical strains of Candida spp. Front Microbiol 9:1954–1607
Wiegand I, Hilpert K, Hancock REW (2008) Agar and broth dilution methods to determine the minimal inhibitory concentration (MIC) of antimicrobial substances. Nat Protoc 3:163–175
Zaki YH, Al-Gendey MS, Abdelhamid AO (2018) A facile synthesis, and antimicrobial and anticancer activities of some pyridines, thioamides, thiazole, urea, quinazoline, β-naphthyl carbamate, and pyrano[2,3-d]thiazole derivatives. Chem Cent J 12:70
Acknowledgements
This study was supported by the Nicolaus Copernicus University (project No. 786/2014).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Biernasiuk, A., Kawczyńska, M., Berecka-Rycerz, A. et al. Synthesis, antimicrobial activity, and determination of the lipophilicity of ((cyclohex-3-enylmethylene)hydrazinyl)thiazole derivatives. Med Chem Res 28, 2023–2036 (2019). https://doi.org/10.1007/s00044-019-02433-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00044-019-02433-2