Abstract
Purpose
The primary objective of this study was to evaluate the associations between centre/country-based factors and two important process and outcome indicators in patients with hospital-acquired bloodstream infections (HABSI).
Methods
We used data on HABSI from the prospective EUROBACT-2 study to evaluate the associations between centre/country factors on a process or an outcome indicator: adequacy of antimicrobial therapy within the first 24 h or 28-day mortality, respectively. Mixed logistical models with clustering by centre identified factors associated with both indicators.
Results
Two thousand two hundred nine patients from two hundred one intensive care units (ICUs) were included in forty-seven countries. Overall, 51% (n = 1128) of patients received an adequate antimicrobial therapy and the 28-day mortality was 38% (n = 839). The availability of therapeutic drug monitoring (TDM) for aminoglycosides everyday [odds ratio (OR) 1.48, 95% confidence interval (CI) 1.03–2.14] or within a few hours (OR 1.79, 95% CI 1.34–2.38), surveillance cultures for multidrug-resistant organism carriage performed weekly (OR 1.45, 95% CI 1.09–1.93), and increasing Human Development Index (HDI) values were associated with adequate antimicrobial therapy. The presence of intermediate care beds (OR 0.63, 95% CI 0.47–0.84), TDM for aminoglycoside available everyday (OR 0.66, 95% CI 0.44–1.00) or within a few hours (OR 0.51, 95% CI 0.37–0.70), 24/7 consultation of clinical pharmacists (OR 0.67, 95% CI 0.47–0.95), percentage of vancomycin-resistant enterococci (VRE) between 10% and 25% in the ICU (OR 1.67, 95% CI 1.00–2.80), and decreasing HDI values were associated with 28-day mortality.
Conclusion
Centre/country factors should be targeted for future interventions to improve management strategies and outcome of HABSI in ICU patients.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
The role of different structural indicators or centre-/country-based factors has been widely disregarded in the literature |
Therapeutic drug monitoring strategies, availability of clinical pharmacists, weekly screening for multidrug-resistant microorganism carriage, vancomycin-resistant enterococci prevalence in hospital-acquired bloodstream infections, and Human Development Index could be associated with adequacy of antimicrobial therapy and mortality |
Introduction
Hospital-acquired bloodstream infections (HABSI) are one of the hospital-acquired infections with the highest health burden measured in disability-adjusted life years [1, 2]. HABSI are frequently observed in the intensive care unit (ICU) setting and are associated with high morbidity, and increased hospital costs and length of stay. To investigate HABSI, initial adequate therapy and mortality represent one of the most important process and outcome indicators [3,4,5]. Interestingly, international cohorts investigating indicators focussed mostly on individual patient factors [6, 7]. Due to the difficulty to perform large multicentre cohorts, the role of centre-/country-based factors has been widely disregarded in the literature. Several surveys or international surveillance systems showed that the structure of the ICU and microbiological laboratory as well as epidemiological resistance data may substantially differ between different centres and countries [8,9,10,11]. However, associations between these structural indicators and the adequacy of antimicrobial therapy or mortality remain unknown due to the paucity of standardised data globally.
From 2019 to 2021, we conducted the EUROBACT-2 study, a prospective cohort that was designed to update the epidemiology and main factors associated with mortality in ICU patients with HABSI from ICUs worldwide [12]. We sought to use data from this high-quality cohort to evaluate the associations between centre-/country-based factors and two important process and outcome indicators, the adequacy of antimicrobial therapy within the first 24 h, and the 28-day mortality, respectively.
Material and methods
EUROBACT-2 study design
The EUROBACT-2 was a prospective multicontinental cohort study performed between September 2019 and June 2021 [12]. This clinical study was registered within ClinicalTrials.org (NCT03937245) and the results are reported in accordance with the STrengthening the Reporting of OBservational studies in Epidemiology guidelines [13].
Setting
Endorsement, logistic, and financial support was obtained from the European Society of Clinical Microbiology and Infectious Diseases (ESCMID) study Group for Infections in Critically Ill Patients (ESGCIP) and the European Society of Intensive Care Medicine (ESICM). An operational committee (AT, JFT, FB, SR, and NB) oversaw all study operations. National coordinators (NCs) recruited participating centres, applied for regulatory and ethical approvals, and facilitated communication within different countries.
Centres and patients
The included centres were ICUs, defined as a unit specifically designed to manage patients with organ failure within an hospital and able to provide invasive mechanical ventilation for at least 24 h. The EUROBACT-2 study recruited centres with patients with HABSI from 1st June 2019 to 30th January 2021. For this specific analysis, amongst all EUROBACT-2 participating centres, we selected those that included a minimum of ten consecutive HABSI patients or those that recruited patients for a period > 2 months. A flexible start of the inclusion period was allowed for each centre to facilitate participation in the cohort.
Adult patients (≥ 18 years old) with a first episode of HABSI treated in ICU were enrolled. A HABSI was defined as a positive blood culture sampled 48 h after hospital admission. Treatment in the ICU was defined as either the blood culture having been sampled in the ICU or the patient having been transferred to the ICU (i.e. in 48 h) for the treatment of the HABSI.
Data collection, definitions, and indicators
Hospital, centre (ICU), and country data.
Data on hospital and centre characteristics were stratified into the following subgroups: (1) structure of the ICU (i.e. variables that described type of ICU, personal, [infra]structure and organisation), (2) organisation of the microbiology laboratory (i.e. microbiological processing and reporting) and infectious diseases (i.e. variables that described availability of specialists, therapy drug monitoring [TDM] and institutional policy about treatments) and (3) aggregated ICU antimicrobial resistance (AMR)-related factors (i.e. variables that described or could influence AMR). We also collected data from countries of each of the included centres (e.g. geographical, life expectancy, education and income data). We extracted Human Development Index [HDI] data which is composite index of life expectancy, education and per capita income indicators [14, 15]. Further, country data from WHO Tripartite AMR Country Self-Assessment Survey [TrACSS] were also extracted [16, 17].
Individual patient data and outcomes.
We collected patient data from the hospital charts and no additional tests or interventions were performed. Our primary process indicator was adequate antimicrobial therapy within the first 24 h after HABSI. It was defined as a therapy with at least one antimicrobial with in vitro activity for the microorganism at the first day, with adequacy of antimicrobial selection, dosing and administration manually reviewed for all infections and sources of HABSI by three experts (NB, AT and FB). Antimicrobials administered at ineffective or very low dose and/or route of administration, relative to the source of infection, were considered as not adequate.
Our primary outcome indicator was 28-day mortality. Patients were followed for up to 28 days or until hospital discharge and vital status was assessed on day 28.
Statistical analyses
Continuous variables were expressed as medians (interquartile range [IQR]) and categorical variables as absolute frequencies and percentages.
The statistical plan consisted of two steps. First, we described the differences in adequacy of antimicrobial therapy within the first 24 h and 28-day mortality using Chi-square (or Fisher) and Student T (or Wilcoxon) tests for categorical and numeric variables, respectively, for the following subgroups: (1) structure of the ICU, (2) organisation of the microbiology laboratory and infectious diseases, (3) aggregated ICU AMR-related factors and (4) country factors. Second, to identify factors associated with adequate antimicrobial therapy in the first 24 h or day-28 mortality, we built a two-tiered hierarchical logistic mixed model using the GLIMMIX procedure of the SAS software for each subgroup. The effects of centre-based variables were included as random intercepts. Multilevel modelling considered the hierarchical structure of the data, which may manifest as intraclass correlations. We performed mixed univariable and multivariable logistic models for each subgroup. All non-colinear clinically relevant variables with p values < 0.10 by univariate analysis were introduced into the multivariable model. A backward process was then used for further variable selection. To mitigate bias introduced by severity of patients at the time of HABSI diagnosis, we forced in our multivariable models the variable “presence of septic shock” at the individual level. Moreover, we performed sensitivity analysis excluding centres that recruited patients affected by coronavirus disease 2029 (COVID-19).
Further details on skin contaminants, centre/country or individual patient variables, data quality, definitions, missing data and statistical analyses are described in the electronic supplementary material (ESM).
Results
Participating ICUs and patients
Amongst the 333 ICUs included in the EUROBACT-2 cohort, we excluded 132 ICUs that included less than 10 patients or for a period < 3 months (eFigure 1), leaving 201 ICUs from 47 countries eligible for the study (eFigure 2). Half of them were located in Europe (n = 105, 52%) and two thirds were in Organization for Economic Cooperation and Development (OECD) member countries (n = 137, 68%).
We included 2209 patients with a HABSI. Most patients were male (n = 1388, 63%) with a median age of 64 (IQR 52; 73) years. The most common admission diagnoses were respiratory diseases (n = 472, 21%) and sepsis or septic shock (n = 427, 19%). Most HABSI (n = 1766, 80%) were acquired in ICU.
Overall, 51% (n = 1128) of patients received an adequate antimicrobial therapy within the first 24 h and 38% (n = 839) died in-hospital within 28 days.
Structure of the ICU
In descriptive analyses, patients recruited from teaching hospitals received less frequently adequate antimicrobial therapy within the first 24 h, whereas patients recruited from burn units and in ICUs with higher number of senior doctors received more frequently adequate antimicrobial therapy within the first 24 h (Table 1). However, when univariable and multivariable mixed logistic models were used, these variables were not associated with adequate therapy within the first 24 h (eTable 1).
In descriptive analyses, type of ICU, number of beds per doctor, number of senior doctors, number of beds in the ICU, number of ventilator and non-ventilator beds in the ICU, and recruitment from general and paediatric wards were associated with increased 28-day mortality (Table 1). Using multivariable mixed logistic models with adjustment for the presence of septic shock, the availability of intermediate care beds in the ICU was associated with decreased 28-day mortality (odds ratio [OR] 0.63, 95% confidence interval [CI] 0.47–0.84, p = 0.0017, Fig. 1, eTable 2).
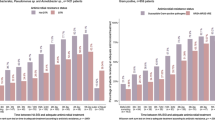
Multivariable mixed logistical models for adequate antimicrobial therapy within the first 24 h and 28-day mortality. The first panel relates to adequate antimicrobial therapy within the first 24 h, the second panel 28-day mortality. ICU intensive care unit, OR odds ratio, CI confidence interval, TDM therapeutic drug monitoring, AMR antimicrobial resistance
Organisation of the microbiology laboratory and infectious diseases
Having an infectious diseases (ID) specialist in the ICU, scheduled ID rounds or multidisciplinary meetings with ID specialists, the presence of a clinical pharmacist as part of the permanent ICU staff, empirical antibiotic treatment determined by local infection treatment guidelines, TDM for aminoglycosides, vancomycin and beta-lactam, automated blood culture incubation, monitoring for positive blood cultures every 24 h and 7 days a week, and performing molecular tests in case of multidrug-resistant bacteria were associated with an increased percentage of adequate therapy within the first 24 h (Table 2). Using multivariable mixed logistic models with adjustment for the presence of septic shock, only TDM for aminoglycosides was associated with an increased probability of adequate therapy within the first 24 h (TDM available at least once a week, OR 1.22 [95% CI 0.76–1.95]; TDM available every day, OR 1.48 [95% CI 1.03–2.14]; TDM available every day within a few hours, OR 1.79 [95% CI 1.34–2.38]; p < 0.01, Fig. 1, eTable 3).
Scheduled ID rounds or multidisciplinary meetings, frequent consultation with clinical pharmacists, the presence of a clinical pharmacist as part of the permanent ICU staff, empirical antibiotic treatment determined by national/international or local guidelines, collection of microbiological surgical site or procedural site specimens, TDM for aminoglycosides, vancomycin and beta-lactam, results of positive blood culture reported on personal contact and 24/7, antibiotic susceptibility test directly performed from the positive blood culture, and molecular test performed in case of multidrug-resistant bacteria were associated with decreased 28-day mortality. Using multivariable mixed logistic models with adjustment for the presence of septic shock, TDM for aminoglycosides was associated with decreased probability of 28-day mortality (TDM available at least once a week, OR 0.81 [95% CI 0.48–1.36]; TDM available every day, OR 0.66 [95% CI 0.44–1.00]; TDM available every day within a few hours, OR 0.51 [95% CI 0.37–0.70]; p < 0.01, Fig. 1, eTable 4). Moreover, consultation of clinical pharmacists was associated with decreased 28-day mortality (24/7 consultation, OR 0.67 [95% CI 0.47–0.95]; business hours consultation, OR 0.75 [95% CI 0.53–1.06]).
ICU AMR-related factors
Selective oropharyngeal and/or digestive tract decontamination, surveillance cultures and screening for multidrug-resistant organism carriage, different percentage of vancomycin-resistant enterococci (VRE) and extended-spectrum beta-lactamase (ESBL)-producing Enterobacterales were associated with adequate therapy within the first 24 h (Table 3). Using multivariable mixed logistic models with adjustment for the presence of septic shock, surveillance cultures and screening for multidrug-resistant organism carriage were associated with an increased probability of adequate therapy within the first 24 h (screening for all patients on admission, OR 1.07 [95% CI 0.75–1.53]; screening for all patients on admission and at least once weekly, OR 1.45 [95% CI 1.09–1.93]; Fig. 1, eTable 5).
Surveillance cultures and screening for multidrug-resistant organism carriage, different percentage of methicillin-resistant Staphylococcus aureus (MRSA), VRE, ESBL-producing Enterobacterales and carbapenemase-producing Enterobacterales were associated with 28-day mortality (Table 3). Using multivariable mixed logistic models with adjustment for the presence of septic shock, percentage of VRE between 10 and 25% was associated with increased 28-day mortality (OR 1.67 [95% CI 1.00–2.80], Fig. 1, eTable 6).
Country factors
Median values of HDI and current health expenditure were increased in patients who received an adequate therapy within the first 24 h. Moreover, patients recruited in countries with training and professional education on AMR, in countries with data on reports from national surveillance system for AMR, and in countries with policies for optimising antimicrobial use implemented in most healthcare facilities had higher proportions of adequate therapy within the first 24 h (Table 4). Using multivariable mixed logistic models with adjustment for the presence of septic shock, increasing HDI values were associated with increased OR for adequate therapy within the first 24 h (Fig. 1, eTable 7).
Median values of HDI and current health expenditure were decreased in patients who died within 28 days (Table 4). Moreover, patients recruited in countries with training and professional education on AMR, in countries with data reports from national surveillance system for AMR and with policies for optimising antimicrobial use implemented in most healthcare facilities were associated with decreased 28-day mortality. Using multivariable mixed logistic models with adjustment for the presence of septic shock, decreasing values of HDI were associated with increased 28-day mortality (Fig. 1, eTable 8).
Sensitivity analysis excluding the centres that recruited COVID-19 patients
A sensitivity analysis excluding centres (n = 59) that recruited COVID-19 patients (980 patients, 319 patients with COVID-19) during the study period showed similar results regarding the structure of the ICU factors, organisation of the microbiology laboratory and infectious diseases, ICU AMR-related and country factors for both indicators (eFigure 3).
Discussion
Using a large international prospective cohort, we provided a detailed description of the organisation of ICUs, microbiology laboratories and antimicrobial stewardship processes worldwide. We showed that several factors related to the centre and country were associated with the adequacy of antimicrobial therapy and mortality in critically ill patients with HABSI. To our knowledge, such an in-depth analysis on centre- and country-specific factors has never been performed. Compared to the initial EUROBACT-2 publication [12], this analysis differs in the study population by including only the largest centres, centre-related factors were investigated in detail that were not investigated in the initial publication, including those relevant to the organisation of the microbiology laboratory, infectious diseases and AMR, as were country-level factors. Individual HABSI data were used only as adjustment factors, and this analysis provides an in-depth analysis of a process indicator (i.e. adequacy of antimicrobial therapy).
Indeed, cohort studies analysing adequacy of antimicrobial therapy or mortality have mostly focussed on individual risk factors [18, 19].
Aminoglycoside TDM was associated with an increased probability of adequate antimicrobial therapy within the first 24 h and with decreased mortality. TDM is frequently used to optimise exposure whilst minimising toxicity in antibiotics with complex pharmacokinetics or those with a narrow therapeutic window [20]. Aminoglycosides could lead to acute kidney injury due to acute tubular necrosis; therefore, pharmacokinetically monitored aminoglycoside therapy in critically ill patients may reduce toxicity [20]. In addition, aminoglycoside TDM could optimise antibiotic dosing in an attempt to achieve pharmacokinetic/pharmacodynamic targets and outcomes of severe infections in critically ill patients [20, 21]. It is, therefore, possible that the frequency of TDM may be associated with reduced mortality. However, TDM for aminoglycoside may simply represent a proxy measure for access to a highly functional laboratory system in a mature healthcare setting with multiple other protective factors. Further analysis showed that 64% (n = 128) of centres administered aminoglycoside during the study period and, amongst them, 35% (n = 45) did not perform TDM for aminoglycoside. Interestingly, models conducted in this subpopulation showed similar results (eTables 9–10).
We observed that frequent consultation with clinical pharmacists was significantly associated with decreased 28-day mortality. Up to now, a positive impact of pharmacy consultation for ICU patients with severe infections has been reported only from retrospective database linkage [22], or by small localised studies [23, 24]. By ensuring optimal drug choice, avoiding interactions and improving delivery with pharmacodynamic/pharmacokinetic optimisation, clinical pharmacists could have a significant role in providing safe and effective care to ICU patients with severe infections.
In settings with a low prevalence of multidrug-resistant microorganisms, screening for multidrug-resistant organism carriage could prevent the spread of such microorganisms by allowing a prompt implementation of infection prevention and control measures, thus decreasing the risk of cross-transmission [25]. We also found that screening for multidrug-resistant organism carriage was associated with an increased probability of adequate therapy within the first 24 h. This association was significant when multidrug microorganisms were tested on admission and at least once weekly. Awareness of multidrug-resistant microorganism colonisation in critically ill patients could, therefore, be crucial for the implementation of the best therapeutic management strategies. In this context, a recent systematic review and meta-regression analysis showed that patients colonised with carbapenem-resistant microorganisms were at increased risk of subsequent infection [26]. Interestingly, one third of patients included in the EUROBACT-2 study were not screened for multidrug-resistant organism carriage, highlighting room for improvement in several centres. Further discussion on ICU AMR factors associated with mortality is described in the supplementary material.
Our study showed an association between the presence of intermediate care beds in the ICU and 28-day mortality in the EUROBACT-2 cohort. The role of intermediate care beds combined with ICU beds has been debated in the literature in the last 2 decades [27]. On one hand, intermediate care beds could provide more intensive monitoring and patient management than the general ward, thus impacting prognosis [27]. In this context, a large cohort study highlighted the benefits of intermediate care beds in term of prognosis for severely ill patients [28] and our international study underlined the importance of the presence of these beds, especially during COVID-19 pandemic. On the other hand, the presence of intermediate care beds may simply represent a less-severe patient population. However, after adjusting for severity on admission, we observed a significant association with mortality, thus refuting this hypothesis.
Country factors were also associated with our process and outcome indicators. We showed that decreasing values of HDI were associated with a low probability of adequate antimicrobial therapy and increased probability of mortality, respectively. To our knowledge, country factors have not been investigated as dependent variables on process and outcome indicators in critically ill patients with HABSI due to difficult to obtain worldwide data. HDI includes long and healthy life expectancy, education, and a decent standard of living measured by gross national income per capita [29]: our findings clearly highlighted the need for policy-mediated large-scale improvements even for critically ill patients.
Our study has several limitations. Centres were predominantly from high-income and upper-middle-income countries, which limits the generalisability of our results. Second, data collection continued during the first year of the COVID-19 pandemic, whereas the management of HABSI in the different centres may have been modified during this period. For this reason, we performed a sensitivity analysis excluding centres that recruited COVID-19 patients. Third, data collection was performed by individual investigators in several ICUs, without on-site monitoring. We mitigated the risk of inconsistencies with online checks through the electronic case report file, and by a close monitoring of the data quality and coherence within each case report by at least one expert. Fourth, for our multivariable models, we used backward selection which could be sensitive to the sample size, the order of variables, the correlation amongst variables, and the significance level. Fifth, aggregated AMR ICU data on Acinetobacter spp. and Pseudomonas spp. were not available in the EUROBACT-2 database. Sixth, adequacy within 24 h may be a debatable process outcome and could only represent a proxy for the true adequacy of antimicrobial treatment. Of note, a recent meta-analysis showed that 24 versus 48 h cut-off showed similar impact on mortality [30]. Seventh, the number of HABSI included in each centre was low, thus decreasing reliability of adjusted analyses.
Conclusion
Using a large high-quality international database, we showed that TDM strategies, availability of clinical pharmacists, weekly screening for multidrug-resistant microorganisms carriage, VRE prevalence in HABSI and Human Development Index could substantially be associated with process and outcome indicators. Centre- and country-specific factors should be included in further prospective international studies investigating severe infections in critically ill patients.
Availability of data and material
The datasets used and/or analysed during the current study are available from the OUTCOMEREA organisation on reasonable request.
Change history
08 April 2024
A Correction to this paper has been published: https://doi.org/10.1007/s00134-024-07415-6
References
Cassini A, Plachouras D, Eckmanns T, Abu Sin M, Blank HP, Ducomble T et al (2016) Burden of six healthcare-associated infections on european population health: estimating incidence-based disability-adjusted life years through a population prevalence-based modelling study. PLoS Med 13(10):e1002150
Cassini A, Colzani E, Pini A, Mangen MJ, Plass D, McDonald SA, et al. Impact of infectious diseases on population health using incidence-based disability-adjusted life years (DALYs): results from the Burden of Communicable Diseases in Europe study, European Union and European Economic Area countries, 2009 to 2013. Euro Surveill Bull Eur Malad Transm Eur Commun Dis Bull. 2018;23(16).
Niederman MS, Baron RM, Bouadma L, Calandra T, Daneman N, DeWaele J et al (2021) Initial antimicrobial management of sepsis. Crit Care 25(1):307
Ten Oever J, Jansen JL, van der Vaart TW, Schouten JA, Hulscher M, Verbon A (2019) Development of quality indicators for the management of Staphylococcus aureus bacteraemia. J Antimicrob Chemother 74(11):3344–3351
Klompas M, Rhee C, Singer M (2023) The importance of shifting sepsis quality measures from processes to outcomes. JAMA 329(7):535–536
Vincent JL, Sakr Y, Singer M, Martin-Loeches I, Machado FR, Marshall JC et al (2020) Prevalence and outcomes of infection among patients in intensive care units in 2017. JAMA 323(15):1478–1487
Tabah A, Koulenti D, Laupland K, Misset B, Valles J, de Carvalho FB et al (2012) Characteristics and determinants of outcome of hospital-acquired bloodstream infections in intensive care units: the EUROBACT International Cohort Study. Intensive Care Med 38(12):1930–1945
ECDC. European Centre for Disease Prevention and Control. Healthcare-associated infections in intensive care units - Annual Epidemiological Report for 2017 [Internet]. ECDC; 2019 Oct. https://www.ecdc.europa.eu/sites/default/files/documents/AER_for_2017-HAI.pdf. 2019.
Checkley W, Martin GS, Brown SM, Chang SY, Dabbagh O, Fremont RD et al (2014) Structure, process, and annual ICU mortality across 69 centers: United States critical illness and injury trials group critical illness outcomes study. Crit Care Med 42(2):344–356
Frankel SK, Moss M (2014) The effect of organizational structure and processes of care on ICU mortality as revealed by the United States critical illness and injury trials group critical illness outcomes study. Crit Care Med 42(2):463–464
Sakr Y, Moreira CL, Rhodes A, Ferguson ND, Kleinpell R, Pickkers P et al (2015) The impact of hospital and ICU organizational factors on outcome in critically ill patients: results from the Extended Prevalence of Infection in Intensive Care study. Crit Care Med 43(3):519–526
Tabah A, Buetti N, Staiquly Q, Ruckly S, Akova M, Aslan AT et al (2023) Epidemiology and outcomes of hospital-acquired bloodstream infections in intensive care unit patients: the EUROBACT-2 international cohort study. Intensive Care Med 49(2):178–190
von Elm E, Altman DG, Egger M, Pocock SJ, Gotzsche PC, Vandenbroucke JP et al (2007) Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: guidelines for reporting observational studies. BMJ 335(7624):806–808
UN Statistics Division. Standard country and area codes for statistical use. https://unstats.un.org/unsd/methodology/m49/. Accessed 7 June 2023.
World bank open database. Data on current health expenditure. Last accessed 8 Sept 2023. https://data.worldbank.org/. 2020.
WHO. Food and Agriculture Organization of the United Nations, World Organisation for Animal Health, WHO. Global Database for the Tripartite Antimicrobial Resistance (AMR) Country Self- assessment Survey (TrACSS). 2022. https://amrcountryprogress.org/#/response-overview. Accessed 6 June 2023.
Patel J, Harant A, Fernandes G, Mwamelo AJ, Hein W, Dekker D et al (2023) Measuring the global response to antimicrobial resistance, 2020–21: a systematic governance analysis of 114 countries. Lancet Infect Dis 23(6):706–718
Kallel H, Houcke S, Resiere D, Roy M, Mayence C, Mathien C et al (2020) Epidemiology and prognosis of intensive care unit-acquired bloodstream infection. Am J Trop Med Hyg 103(1):508–514
Timsit JF, Ruppe E, Barbier F, Tabah A, Bassetti M (2020) Bloodstream infections in critically ill patients: an expert statement. Intensive Care Med 46(2):266–284
Wong G, Sime FB, Lipman J, Roberts JA (2014) How do we use therapeutic drug monitoring to improve outcomes from severe infections in critically ill patients? BMC Infect Dis 14:288
van Lent-Evers NA, Mathot RA, Geus WP, van Hout BA, Vinks AA (1999) Impact of goal-oriented and model-based clinical pharmacokinetic dosing of aminoglycosides on clinical outcome: a cost-effectiveness analysis. Ther Drug Monit 21(1):63–73
MacLaren R, Bond CA, Martin SJ, Fike D (2008) Clinical and economic outcomes of involving pharmacists in the direct care of critically ill patients with infections. Crit Care Med 36(12):3184–3189
Lee H, Ryu K, Sohn Y, Kim J, Suh GY, Kim E (2019) Impact on patient outcomes of pharmacist participation in multidisciplinary critical care teams: a systematic review and meta-analysis. Crit Care Med 47(9):1243–1250
Berger NJ, Wright ME, Pouliot JD, Green MW, Armstrong DK (2021) The impact of a pharmacist-driven staphylococcus aureus bacteremia initiative in a community hospital: a retrospective cohort analysis. Pharmacy (Basel). 9(4):191
Birgand G, Schouten J, Ruppe E (2020) Less contact isolation is more in the ICU: con. Intensive Care Med 46(9):1732–1734
Willems RPJ, van Dijk K, Vehreschild M, Biehl LM, Ket JCF, Remmelzwaal S et al (2023) Incidence of infection with multidrug-resistant Gram-negative bacteria and vancomycin-resistant enterococci in carriers: a systematic review and meta-regression analysis. Lancet Infect Dis 23(6):719–731
Vincent JL, Rubenfeld GD (2015) Does intermediate care improve patient outcomes or reduce costs? Crit Care 19(1):89
Capuzzo M, Volta C, Tassinati T, Moreno R, Valentin A, Guidet B et al (2014) Hospital mortality of adults admitted to Intensive Care Units in hospitals with and without Intermediate Care Units: a multicentre European cohort study. Crit Care 18(5):551
WHO. Human development index definition. Available from: https://www.who.int/data/nutrition/nlis/info/human-development-index. Accessed 6 Sep 2023.
Hung YP, Lee CC, Ko WC (2022) Effects of inappropriate administration of empirical antibiotics on mortality in adults with bacteraemia: systematic review and meta-analysis. Front Med (Lausanne) 9:869822
Acknowledgements
The EUROBACT-2 study was endorsed by the European Society of Intensive Care Medicine (ESICM), the infection section of the ESCIM and the European Society of Clinical Microbiology and Infectious Diseases (ESCMID) study Group for Infections in Critically Ill Patients (ESGCIP), with scientific input of the OUTCOMEREA network. We thank the members of the EUROBACT-2 Study Group. The authors thank Céline Féger, MD (EMIBiotech) for her editorial support. The members of the Eurobact 2 Study Group are: Alexis Tabah, Hamish Pollock, Ben Margetts, Meredith Young, Neeraj Bhadange, Steven Tyler, Anne Ledtischke, Mackenzie Finnis, Anne Ledtischke, Mackenzie Finnis, Jyotsna Dwivedi, Manoj Saxena, Vishwanath Biradar, Natalie Soar, Vineet Sarode, David Brewster, Adrian Regli, Elizabeth Weeda, Samiul Ahmed, Cheryl Fourie, Kevin Laupland, Mahesh Ramanan, James Walsham, Jason Meyer, Edward Litton, Anna Maria Palermo, Timothy Yap, Ege Eroglu, Antony George Attokaran, C'havala Jaramillo, Khalid Mahmood Khan Nafees, Nurhikmahtul Aqilah Haji Abd Rashid, Haji Adi Muhamad Ibnu Walid, Tomas Mon, P. Dhakshina Moorthi, Shah Sudhirchandra, Dhadappa Damodar Sridharan, Qiu Haibo, Xie Jianfeng, Lu Wei-Hua, Wang Zhen, Chuanyun Qian, Jili Luo, Xiaomei Chen, Hao Wang, Peng Zhao, Juan Zhao, Qiu Wusi, Chen Mingmin, Lei Xu, Chengfen Yin, Ruilan Wang, Jinfeng Wang, Yongjie Yin, Min Zhang, Jilu Ye, Chungfang Hu, Suming Zhou, Min Huang, Jing Yan, Yan Wang, Bingyu Qin, Ling Ye, Xie Weifeng, Li Peije, Nan Geng, Yoshiro Hayashi, Toshiyuki Karumai, Masaki Yamasaki, Satoru Hashimoto, Koji Hosokawa, Jun Makino, Takeo Matsuyoshi, Akira Kuriyama, Hidenobu Shigemitsu, Yuka Mishima, Michio Nagashima, Hideki Yoshida, Shigeki Fujitani, Koichiro Omori, Hiroshi Rinka, Hiroki Saito, Kaori Atobe, Hideaki Kato, Shunsuke Takaki, M. Shahnaz Hasan, Muhamad Fadhil Hadi Jamaluddin, Lee See Pheng, Sheshendrasurian Visvalingam, Mun Thing Liew, Siong Ling Danny Wong, Kean Khang Fong, Hamizah Bt Abdul Rahman, Zuraini Md Noor, Lee Kok Tong, Abd. Hamid Azman, Mohd Zulfakar Mazlan, Saedah Ali, Kyeongman Jeon, Sang-Min Lee, Sunghoon Park, Seung Yong Park, Sung Yoon Lim, Qing Yuan Goh, Shin Yi Ng, Sui An Lie, Andrea Lay Hoon Kwa, Ken Junyang Goh, Andrew Yunkai Li, Caroline Yu Ming Ong, Jia Yan Lim, Jessica Lishan Quah, Kangqi Ng, Louis Xiang Long Ng, Yu Chang Yeh, Nai-Kuan Chou, Cong-Tat Cia, Ting-Yu Hu, Li-Kuo Kuo, Shih-Chi Ku, Phunsup Wongsurakiat, Yutthana Apichatbutr, Supattra Chiewroongroj, Rashid Nadeem, Ashraf El Houfi, Adel Alsisi, Amr Elhadidy, Mina Barsoum, Nermin Osman, Tarek Mostafa, Mohamed Elbahnasawy, Ahmed Saber, Amer Aldhalia, Omar Elmandouh, Ahmed Elsayed, Merihan A. Elbadawy, Ahmed K. Awad, Hanan M. Hemead, Farid Zand, Maryam Ouhadian, Seyed Hamid Borsi, Zahra Mehraban, Davood Kashipazha, Fatemeh Ahmadi, Mohsen Savaie, Farhad Soltani, Mahboobeh Rashidi, Reza Baghbanian, Fatemeh Javaherforoosh, Fereshteh Amiri, Arash Kiani, Mohammad Amin Zargar, Ata Mahmoodpoor, Fatemeh Aalinezhad, Gholamreza Dabiri, Golnar Sabetian, Hakimeh Sarshad, Mansoor Masjedi, Ramin Tajvidi, Seyed Mohammad Nasirodin Tabatabaei, Abdullah Khudhur Ahmed, Pierre Singer, Ilya Kagan, Merav Rigler, Daniel Belman, Phillip Levin, Belal Harara, Adei Diab, Fayez Abilama, Rebecca Ibrahim, Aya Fares, Ahmad Buimsaedah, Marwa Gamra, Ahmed Aqeelah, Almajdoub Ali Mohammed Ali, Ahmed Gaber Sadik Homaidan, Bushray Almiqlash, Hala Bilkhayr, Ahmad Bouhuwaish, Ahmed Sa Taher, Eman Abdulwahed, Fathi A. Abousnina, Aisha Khaled Hdada, Rania Jobran, Hayat Ben Hasan, Rabab Shaban Ben Hasan, Issam Serghini, Rachid Seddiki, Brahim Boukatta, Nabil Kanjaa, Doumiri Mouhssine, Maazouzi Ahmed Wajdi, Tarek Dendane, Amine Ali Zeggwagh, Brahim Housni, Oujidi Younes, Abdelhamid Hachimi, A. Ghannam, Z. Belkhadir, Sarah Amro, Mustafa Abu Jayyab, Ali Ait Hssain, Abdurahaman Elbuzidi, Edin Karic, Marcus Lance, Shaikh Nissar, Hend Sallam, Omar Elrabi, Ghaleb A. Almekhlafi, Maher Awad, Ahmed Aljabbary, Mohammad Karam Chaaban, Natalia Abu-Sayf, Mohammad Al-Jadaan, Lubna Bakr, Mounir Bouaziz, Olfa Turki, Walid Sellami, Pablo Centeno, Lic Natalia Morvillo, José Oscar Acevedo, Patricia Mabel Lopez, Rubén Fernández, Matías Segura, Dra Marta Aparicio, Microbiologa Irene Alonzo, Yanina Nuccetelli, Pablo Montefiore, Luis Felipe Reyes, Luis Felipe Reyes, Silvio A. Ñamendys-Silva, Juan P. Romero-Gonzalez, Mariana Hermosillo, Roberto Alejandro Castillo, Jesús Nicolás Pantoja Leal, Candy Garcia Aguilar, Mara Ocotlan Gonzalez Herrera, Missael Vladimir Espinoza Villafuerte, Manuel Lomeli-Teran, Jose G. Dominguez-Cherit, Adrian Davalos-Alvarez, Silvio A. Ñamendys-Silva, Luis Sánchez-Hurtado, Brigitte Tejeda-Huezo, Orlando R. Perez-Nieto, Ernesto Deloya Tomas, Liesbet De Bus, Jan De Waele, Isabelle Hollevoet, Wouter Denys, Marc Bourgeois, Sofie F. M. Vanderhaeghen, Jean-Baptiste Mesland, Pierre Henin, Lionel Haentjens, Patrick Biston, Cindérella Noel, Nathalie Layos, Benoît Misset, Nicolas De Schryver, Nicolas Serck, Xavier Wittebole, Elisabeth De Waele, Godelive Opdenacker, Pedja Kovacevic, Biljana Zlojutro, Aida Custovic, Ina Filipovic-Grcic, Radovan Radonic, Ana Vujaklija Brajkovic, Jasminka Persec, Sanja Sakan, Mario Nikolic, Hrvoje Lasic, Marc Leone, Charlotte Arbelot, Jean-François Timsit, Juliette Patrier, N. Zappela, P. Montravers, Thierry Dulac, Jérémy Castanera, Johann Auchabie, Anthony Le Meur, A. Marchalot, M. Beuzelin, Alexandre Massri, Charlotte Guesdon, Etienne Escudier, Philippe Mateu, Jérémy Rosman, Olivier Leroy, Serge Alfandari, Alexandru Nica, Bertrand Souweine, Elisabeth Coupez, Thibault Duburcq, Eric Kipnis, Perrine Bortolotti, Mathieu Le Souhaitier, Jean-Paul Mira, Pierre Garcon, Matthieu Duprey, Martial Thyrault, Rémi Paulet, François Philippart, Marc Tran, Cédric Bruel, Emmanuel Weiss, Sylvie Janny, Arnaud Foucrier, Pierre-François Perrigault, Flora Djanikian, François Barbier, Marc Gainnier, Jérémy Bourenne, Guillaume Louis, Roland Smonig, Laurent Argaud, Thomas Baudry, Armand Mekonted Dessap, Keyvan Razazi, Pierre Kalfon, Gaëtan Badre, Romaric Larcher, Jean-Yves Lefrant, Claire Roger, Benjamine Sarton, Stein Silva, Sophie Demeret, Loïc Le Guennec, Shidasp Siami, Christelle Aparicio, Guillaume Voiriot, Muriel Fartoukh, Claire Dahyot-Fizelier, Nadia Imzi, Kada Klouche, Hendrik Bracht, Sandra Hoheisen, Frank Bloos, Daniel Thomas-Rueddel, Sirak Petros, Bastian Pasieka, Simon Dubler, Karsten Schmidt, Antje Gottschalk, Carola Wempe, Philippe Lepper, Carlos Metz, Dmitriy Viderman, Yerlan Umbetzhanov, Miras Mugazov, Yelena Bazhykayeva, Zhannur Kaligozhin, Baurzhan Babashev, Yevgeniy Merenkov, Talgat Temirov, Kostoula Arvaniti, Dimitrios Smyrniotis, Vasiliki Psallida, Georgios Fildisis, Vasiliki Soulountsi, Evangelos Kaimakamis, Cristina Iasonidou, Sofia Papoti, Foteini Renta, Maria Vasileiou, Vasiliki Romanou, Vasiliki Koutsoukou, Mariana Kristina Matei, Leora Moldovan, Ilias Karaiskos, Harry Paskalis, Kyriaki Marmanidou, M. Papanikolaou, C. Kampolis, Marina Oikonomou, Evangelos Kogkopoulos, Charikleia Nikolaou, Anastasios Sakkalis, Marinos Chatzis, Maria Georgopoulou, Anna Efthymiou, Vasiliki Chantziara, Aikaterini Sakagianni, Zoi Athanasa, Eirini Papageorgiou, Fadi Ali, Georges Dimopoulos, Mariota Panagiota Almiroudi, Polychronis Malliotakis, Diamantina Marouli, Vasiliki Theodorou, Ioannis Retselas, Vasilios Kouroulas, Georgios Papathanakos, Giorgia Montrucchio, Gabriele Sales, Gennaro De Pascale, Luca Maria Montini, Simone Carelli, Joel Vargas, Valentina Di Gravio, Daniele Roberto Giacobbe, Angelo Gratarola, Elisa Porcile, Michele Mirabella, Ivan Daroui, Giovanni Lodi, Francesco Zuccaro, Maria Grazia Schlevenin, Paolo Pelosi, Denise Battaglini, Andrea Cortegiani, Mariachiara Ippolito, Davide Bellina, Andrea Di Guardo, Lorella Pelagalli, Marco Covotta, Monica Rocco, Silvia Fiorelli, Antonella Cotoia, Anna Chiara Rizzo, Adam Mikstacki, Barbara Tamowicz, Irmina Kaptur Komorowska, Anna Szczesniak, Jozef Bojko, Anna Kotkowska, Paulina Walczak-Wieteska, Dominika Wasowska, Tomasz Nowakowski, Hanna Broda, Mariusz Peichota, Iwona Pietraszek-Grzywaczewska, Ignacio Martin-Loeches, Alessandra Bisanti, Nuno Cartoze, Tiago Pereira, Nádia Guimarães, Madalena Alves, Ana Josefina Pinheiro Marques, Ana Rios Pinto, Andriy Krystopchuk, Ana Teresa, António Manuel Pereira de Figueiredo, Isabel Botelho, Tiago Duarte, Vasco Costa, Rui Pedro Cunha, Elena Molinos, Tito da Costa, Sara Ledo, Joana Queiró, Dulce Pascoalinho, Cristina Nunes, José Pedro Moura, Énio Pereira, António Carvalho Mendes, Liana Valeanu, Serban Bubenek-Turconi, Ioana Marina Grintescu, Cristian Cobilinschi, Daniela Carmen Filipescu, Cornelia Elena Predoi, Dana Tomescu, Mihai Popescu, Alexandra Marcu, Ioana Grigoras, Olguta Lungu, Alexey Gritsan, Anastasia Anderzhanova, Yulia Meleshkina, Marat Magomedov, Nadezhda Zubareva, Maksim Tribulev, Denis Gaigolnik, Aleksandr Eremenko, Natala Vistovskaya, Maria Chukina, Vladislav Belskiy, Mikhail Furman, Ricard Ferrer Rocca, Maria Martinez, Vanessa Casares, Paula Vera, Matias Flores, Joaquin Amador Amerigo, Maria Pilar Gracia Arnillas, Rosana Munoz Bermudez, Fernando Armestar, Beatriz Catalan, Regina Roig, Laura Raguer, María Dolores Quesada, Emilio Diaz Santos, Gemma Gomà, Alejandro Ubeda, Dra Maria Salgado, Lorena Forcelledo Espina, Emilio Garcia Prieto, Dra Mj Asensio, Dra M. Rodriguez, Emilio Maseda, Alejandro Suarez De La Rica, J. Ignacio Ayestaran, Mariana Novo, Miguel Angel Blasco-Navalpotro, Alberto Orejas Gallego, Fredrik Sjövall, Dzana Spahic, Carl Johan Svensson, Michael Haney, Alicia Edin, Joyce Åkerlund, Lina De Geer, Josef Prazak, Stephan Jakob, Jl Pagani, S. Abed-Maillard, Murat Akova, Abdullah Tarik Aslan, Arif Timuroglu, Sesin Kocagoz, Hulya Kusoglu, Selcuk Mehtap, Solakoğlu Ceyhun, Neriman Defne Altintas, Leyla Talan, Bircan Kayaaslan, Ayşe Kaya Kalem, Ibrahim Kurt, Murat Telli, Barcin Ozturk, Çiğdem Erol, Emine Kubra Dindar Demiray, Sait Çolak, Türkay Akbas, Kursat Gundogan, Ali Sari, Canan Agalar, Onur Çolak, Nurcan N. Baykam, Ozlem O. Akdogan, Mesut Yilmaz, Burcu Tunay, Rumeysa Cakmak, Nese Saltoglu, Ridvan Karaali, Iftihar Koksal, Firdevs Aksoy, Ahmet Eroglu, Kemal Tolga Saracoglu, Yeliz Bilir, Seda Guzeldag, Gulden Ersoz, Guliz Evik, Hulya Sungurtekin, Cansu Ozgen, Cem Erdoğan, Yunus Gürbüz, Nilgün Altin, Yasar Bayindir, Yasemin Ersoy, Senay Goksu, Ahmet Akyol, Ayse Batirel, Sabahat Cagan Aktas, Andrew Conway Morris, Matthew Routledge, Andrew Conway Morris, Ari Ercole, David Antcliffe, Roceld Rojo, Kate Tizard, Maria Faulkner, Amanda Cowton, Melanie Kent, Ashok Raj, Artemis Zormpa, George Tinaslanidis, Reena Khade, Tomasz Torlinski, Randeep Mulhi, Shraddha Goyal, Manan Bajaj, Marina Soltan, Aimee Yonan, Rachael Dolan, Aimee Johnson, Caroline Macfie, James Lennard, Maie Templeton, Sonia Sousa Arias, Uwe Franke, Keith Hugill, Hollie Angell, Benjamin J. Parcell, Katherine Cobb, Stephen Cole, Tim Smith, Clive Graham, Jaroslav Cerman, Allison Keegan, Jenny Ritzema, Amanda Sanderson, Ashraf Roshdy, Tamas Szakmany, Tom Baumer, Rebecca Longbottom, Daniel Hall, Kate Tatham, S. Loftus, A. Husain, E. Black, S. Jhanji, R. Rao Baikady, Peter Mcguigan, Rachel Mckee, Santhana Kannan, Supriya Antrolikar, Nicholas Marsden, Valentina Della Torre, Dorota Banach, Ahmed Zaki, Matthew Jackson, Moses Chikungwa, Ben Attwood, Jamie Patel, Rebecca E. Tilley, Miss Sally K. Humphreys, Paul Jean Renaud, Anton Sokhan, Yaroslava Burma, Wendy Sligl, Nadia Baig, Lorena McCoshen, Demetrios J. Kutsogiannis, Wendy Sligl, Patricia Thompson, Tayne Hewer, Raihan Rabbani, Shihan Mahmud Redwanul Huq, Rajib Hasan, Mohammad Motiul Islam, Mohan Gurjar, Arvind Baronia, Nikhil Kothari, Ankur Sharma, Saurabh Karmakar, Priya Sharma, Janardan Nimbolkar, Pratit Samdani, R. Vaidyanathan, Noor Ahmedi Rubina, Nikhilesh Jain, Madhumati Pahuja, Ritu Singh, Saurav Shekhar, Syed Nabeel Muzaffar, Ahmad Ozair, Suhail Sarwar Siddiqui, Payel Bose, Avijatri Datta, Darshana Rathod, Mayur Patel, M. K. Renuka, Sailaja K. Baby, Carol Dsilva, Jagadish Chandran, Pralay Ghosh, Sudipta Mukherjee, Kaladhar Sheshala, Krushna Chandra Misra, Saidu Yusuf Yakubu, Euphemia Mgbosoro Ugwu, John O. Olatosi, Ibironke Desalu, Gabriel Asiyanbi, Motunrayo Oladimeji, Olusola Idowu, Fowotade Adeola, Melanie Mc Cree, Ali Adil Ali Karar, Elfayadh Saidahmed, Hytham K. S. Hamid.
Funding
Open access funding provided by University of Geneva. Research grants were obtained from the European Society of Intensive Care Medicine (ESICM), the European Society of Clinical Microbiology and Infectious Diseases (ESCMID) study Group for Infections in Critically Ill Patients (ESGCIP), the Norva Dahlia foundation and the Redcliffe Hospital Private Practice Trust Fund.
Author information
Authors and Affiliations
Consortia
Contributions
NB, SR, AT and JFT designed and conceptualised the study. All co-authors acquired the data in their countries. SR, NB, NS and JFT did the statistical analysis. SR performed data curation. JFT and AT acquired funding. NB, SR, AT and JFT analysed and interpreted the data. NB, AT, NS and JFT drafted the manuscript. All authors critically reviewed the manuscript and approved the final report.
Corresponding author
Ethics declarations
Conflicts of interest
NB received a Mobility grant from the Swiss National Science Foundation (Grant number: P400PM_183865). JJDW is a senior clinical investigator funded by the Research Foundation Flanders (FWO, Ref. 1881020N). ACM is supported by a Medical Research Council Clinician Scientist Fellowship (MR/V006118/1) and received speaking fees from Fischer & Paykel, Biomerieux; he participated in scientific advisory board of Cambridge infection diagnostics; he received support for the present manuscript by the medical research council (Clinician Scientist Fellowship grant number: MR/V006118/1). ML received consulting fees from LFB, Shionogi, AOP Pharma and Viatris outside of this project. RF has received consulting fees from Inotrem, Pfizer and Cytosorbent and honoraria for lectures from Shionogi, MSD, Menarini, Thermofisher and Gilead. GM received payment for honoraria for lectures, presentations, speakers bureaus, manuscript writing or educational events from Thermofisher, Gilead, Pfizer and 3 M; and she participated on a data Safety Monitoring Board or Advisory Board by Gilead and 3 M. AC received fees for lectures and scientific consultancies by Gilead, MSD, Mundipharma, Pfizer. LDB participated on data safety monitoring or advisory board of MSD. MB received payment or honoraria for lectures, presentations, speakers bureaus, manuscript writing or educational events from Angelini, Biomerieux, Cidara, Gilead, Menarini, MSD, Pfizer, and Shionogi. MB participated on data safety monitoring board or advisory board of Angelini, Biomerieux, Cidara, Gilead, Menarini, MSD, Pfizer, and Shionogi. MA participated on a data safety board for GSK and is the president of the International Immunocompromised Host Society.
Ethics approval
The initial ethical approval as a low-risk research project with waiver of individual consent was granted by the Ethics Committee of the Royal Brisbane & Women’s Hospital, Queensland, Australia (number: LNR/2019/QRBW/48376). Each study centre obtained ethical, governance and any other relevant approvals according to regional and/or national regulations.
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Buetti, N., Tabah, A., Setti, N. et al. The role of centre and country factors on process and outcome indicators in critically ill patients with hospital-acquired bloodstream infections. Intensive Care Med 50, 873–889 (2024). https://doi.org/10.1007/s00134-024-07348-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00134-024-07348-0