Abstract
Comprehending how environmental variability shapes foraging behaviour across habitats is key to unlocking insights into consumer ecology. Seabirds breeding at high latitudes are exemplars of how marine consumers can adapt their behaviours to make use of predictable foraging opportunities, but prey tends to be less predictable in tropical oceanic ecosystems and may require alternative foraging behaviours. Here we used GPS and time-depth recorder loggers to investigate the foraging behaviour of central placed adult red-footed boobies (Sula sula rubripes), a tropical seabird that forages in oceanic waters via diving, or by capturing aerial prey such as flying fish in flight. Dive bout dynamics revealed that red-footed boobies appeared to exploit denser, but more sparsely distributed prey patches when diving further from the colony. Furthermore, although we found no evidence of environmentally driven habitat selection along their foraging routes, red-footed boobies preferentially dived in areas with higher sea surface temperatures and chlorophyll-a concentrations compared to conditions along their foraging tracks. This multi-scale variation implies that habitat selection differs between foraging routes compared to dive locations. Finally, red-footed booby dives were deepest during the middle of the day when light penetration was greatest. Ultimately, we highlight the importance of gaining insights into consumer foraging across different ecosystems, thereby broadening understanding of how animals might respond to changing environmental conditions.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Acquiring adequate trophic resources for growth, survival, and reproduction, carries strong ecological and evolutionary consequences in the animal kingdom (Tomlinson et al. 2014). Environmental variability shapes food availability and can have important repercussions for consumer foraging efficiency (Abrams 1991). For instance, in habitats where food is widely distributed but stable in time and space, evolutionary selection should favour foraging behaviours (and in turn life history tactics) which exploit this consistency (Riotte-Lambert and Matthiopoulos 2020). However, to fully understand how spatio-temporal food availability influences predator ecology, studies are required from a wide range of different habitats.
Many species of marine vertebrates adopt foraging tactics that are consistent in time and space (Rodríguez-Malagón et al. 2020) and these behaviours are exemplified by breeding seabirds (Wakefield et al. 2015; Votier et al. 2017). During the breeding season, this consistent foraging behaviour is the result of seabirds adopting central place foraging strategies as well as the occurrence of predictable foraging opportunities such as those aggregated around upwellings and fronts (Scales et al. 2014). However, these findings are biased towards neritic and/or high latitude areas. In tropical pelagic waters, prey tends to be more unpredictably and sparsely distributed which may in turn explain marine predator movement ecology (Ballance and Pitman 1999; Humphries et al. 2010). Indeed, tropical seabirds forage across large pelagic areas (Soanes et al. 2021) and demonstrate low forage site fidelity (Weimerskirch 2007), irregularly encountering prey patches (Weimerskirch et al. 2004). Dive bout dynamics, whereby individuals perform distinct sequences of multiple aggregated dives, can be reflective of the irregularity of prey patches, as more intense diving (longer bouts) should coincide with denser prey patches (Boyd 1996), and the distances between dive bouts should be indicative of how spatially clustered prey patches are (Sommerfeld et al. 2015). Whilst in more productive neritic ecosystems seabird dive bouts often occur either at distal points (Votier et al. 2017) or at environmentally favourable locations (e.g., amongst sea ice; Le Guen et al. 2018), it is possible that within tropical systems dive bouts might occur less predictably throughout their foraging trips.
There is evidence that some seabirds in tropical oceanic regions preferentially select to forage within high productivity habitats (Weimerskirch et al. 2004, 2005a) or those where physical features, such as ocean depth, influence the distribution of their prey (Jaquemet et al. 2005; Mendez et al. 2017). However, animals select habitats at different scales (Mayor et al. 2009). For example, though shearwater species select certain wind conditions when transiting (Paiva et al. 2010), once at their foraging grounds, wind-induced increases in turbidity may restrict their ability to successfully dive and capture prey (Darby et al. 2022). How the environmental drivers of multi-scale tropical seabird habitat selection vary throughout their foraging trips, as they decide firstly where to transit and then where to dive, warrants further study.
In addition to environmental conditions influencing the horizontal distributions of seabirds, for diving species, varying light conditions, sun glare, and subsequent prey visibility across the diurnal cycle can influence their vertical distributions (Lewis et al. 2002). Furthermore, many tropical seabirds rely on subsurface predators such as tuna and billfish to aggregate and disorientate prey, leading to almost obligate multi-species commensal associations (Veit and Harrison 2017). Together, these conditions may lead to divergent foraging behaviour among tropical seabirds compared to better studied populations from higher latitudes and coastal waters.
Here we equipped breeding red-footed boobies (Sula sula rubripes, hereafter ‘RFB’) with biologging devices to study their dive behaviour in the Chagos Archipelago, within the tropical Indian Ocean. RFBs are a sulid species that breed colonially throughout the tropics and adopt central place foraging strategies during breeding, commuting to and from their colonies to perform diurnal shallow plunge and surface dives for prey, such as flying fish (Exocoetidae; Weimerskirch et al. 2005a). We hypothesised that, due to ephemeral and patchy prey, RFB dives would be clustered in multiple bouts throughout their foraging journeys, rather than at distal locations. We also assessed multi-scale RFB habitat selection, hypothesising that environmental variables would influence where they a) transited and b) dived. Finally, we hypothesised that, due to a reliance on vision and commensal foraging, dive depth would be related to time of day.
Materials and methods
Study area and deployment of biologging devices
Fieldwork was conducted in the Chagos Archipelago Marine Protected Area, in the central Indian Ocean where there are over 21,000 breeding pairs of RFB. In February 2019 and 2022 we caught shrub-nesting adult RFBs on the nest by hand or with a landing net, selecting individuals that were either incubating or brooding at two locations < 2 km apart within the Diego Garcia atoll, Chagos Archipelago: 15 individuals at Barton Point (7.23°S 72.43°E; 9,269 breeding pairs) in 2019, and 17 individuals at East Island (7.23°S 72.42°E; 1,113 breeding pairs) in 2022. Using marine Tesa tape (4651), we attached combined GPS and time-depth recorder loggers (TechnoSmArt Axy-Trek Marine, 18 g, www.technosmart.eu/axy-trek-marine/, representing 1.7–2.4% of body mass across the 32 individuals: 740–1080 g) on the underside of each bird’s central two to four tail feathers, depending on moult condition. In 2019, GPS loggers recorded a location (latitude and longitude) every 30 s and in 2022 they recorded a location in 15 s of 1 Hz every 5 min. Time-depth recorder loggers recorded a depth reading every 1 s. Geolocation and immersion loggers (Intigeo C330, Migrate technology, 3.3 g, representing 0.3–0.45% of body mass) were also attached to the birds via a plastic leg ring, but the resultant data were not analysed in this study due to them being outside the scope of this manuscript. Handling time lasted approximately 10 min and nest attendance was monitored daily following deployment, revealing no unusual behaviour and that any disturbance caused during the deployment process was short-lived. Individuals were recaptured after at least 24-h of deployment, and after completing at least one foraging trip. The biologging devices were then removed (73% recovery at Barton Point and 52% at East Island where lower recovery was reflective of more limited access to the island colony).
Processing biologging data
We performed all data processing and analyses in R version 4.2.2 (R Core Team 2022). To obtain GPS fixes from RFB foraging tracks only, we removed those from outside the deployment period and defined foraging trips as those that extended > 1 km from the colony for > 30 min (Trevail et al. 2023a). We used the ‘adehabitatHR’ R package (Calenge 2006) to interpolate the GPS location data to the same frequency as the pressure data (every 1 s). We converted raw pressure readings to depth data using XManager software (TechnoSmArt) and cropped these files to only include readings recorded during foraging tracks. Environmental conditions can cause time-depth recorder loggers to experience changes in pressure and therefore, to remove noise from the depth data and ensure that the surface of the water was set to 0 m, we offset all depth values within a 30 s window by the median of that window (Elliott and Gaston 2009). We visually inspected time-series plots of depth to identify sharp fluctuations (i.e., true dive events, as opposed to residual background noise) and thereby classified dives as instances where at least one depth value exceeded a threshold of 0.1 m (Grémillet et al. 2004).
The foraging activity of marine predators can often be separated into distinct sequences or ‘bouts’ (Halsey et al. 2007). We grouped individual dives into dive bouts using a log survivorship analysis to identify the minimum surface interval between two dives that belonged to different bouts (Sibly et al. 1990). Briefly, we plotted the cumulative frequency of post-dive surface intervals on the logarithmic scale against surface interval durations, and identified two breakpoints within the resultant log survivorship curve using the ‘segmented’ R package (Muggeo 2017). The first of these breakpoints (41 s) is likely to have encompassed time for behaviours such as food digestion in-between dives, whereas the second breakpoint (221 s) is likely to indicate the end of a period of intense diving, i.e., an inter-bout duration (Le Guen et al. 2018). Isolated dives that were not within a diving bout were excluded from all analyses to avoid the inclusion of miscellaneous events, such as small dips below the surface and preening behaviour, as well as signals associated with sudden pressure or temperature changes (Duckworth et al. 2021). We extracted the depth of each individual dive, and the time at which it took place. For each dive bout, we calculated its duration, the number of dives that occurred, the distance between the bout and its proceeding bout (inter-bout distance), and the distance between the bout and the colony (using the ‘geosphere’ R package; Hijmans 2022).
Environmental variables
We wanted to evaluate the impact of environmental conditions on the two-step habitat selection decision making process that influenced firstly how RFBs preferentially selected foraging routes, and secondly what then influenced their dive locations. A dataset of the conditions available to foraging RFBs was therefore generated by extracting environmental metrics that may influence foraging behaviour, namely bathymetry (sea depth), chlorophyll-a concentration (a proxy for primary productivity), and sea surface temperature (see Table 1). Using the ‘rerddapXtracto’ R package (Mendelssohn 2022), we extracted these environmental metrics from the average location of each dive bout as well as a) 50 random points from within the population’s foraging range (‘available locations’; i.e., the population’s minimum convex polygon), and b) 50 random points throughout the associated foraging track where diving did not occur ('traversed locations’).
Statistical analyses
We ran all the following statistical analyses in the Stan programming language using the ‘brms’ R package (Bürkner 2017). We ran all models with 4 Markov Chain Monte Carlo (MCMC) chains and with 2000 iterations per chain, including warmup. When necessary, to eliminate divergent transitions that caused a bias in the posterior draws, we increased ‘adapt_delta’, a tuning parameter, to 0.85 (Bürkner 2017). We confirmed the convergence of all our mixed effects models via visual inspection of the chains, posterior predictive check plots, and by calculating a Gelman-Ruban convergence statistic (\(\widehat{R}\)) of 1 (McElreath 2020).
Dive distribution throughout the foraging journey
To evaluate differences in diving effort throughout foraging trips, we modelled dive bout metrics (namely bout duration and inter-bout distance) as a function of distance from the colony, including a random effect of ‘trip ID’ nested within ‘individual ID’.
Foraging habitat selection
To investigate the two-step habitat selection process, we assessed RFB habitat selection probabilities by firstly comparing the environmental conditions along the foraging track (‘traversed locations’) to those available within the population’s foraging range (‘available locations’), and then secondly by comparing the conditions at dive locations with traversed locations. We therefore initially investigated whether birds selected to traverse over different areas of differing environmental conditions from those within the population’s foraging range. This habitat selection was modelled in a mixed effects linear regression model assuming that foraging was determined by a Bernoulli distribution (0 = available locations and 1 = traversed locations) in response to each environmental metric (i.e., depth, chlorophyll-a concentration, and sea surface temperature; Table 1). We only retained remotely sensed data where they were available for all locations, as opposed to being obscured by cloud cover, and we centred and standardised the predictors to allow for comparisons between them. We included distance from the colony as a fixed effect to account for the central placed foraging strategy that seabirds use during their breeding seasons and a random effect of ‘trip ID per individual’ (Trevail et al. 2021). We determined the ratio of available points (the aforementioned random points from within the population’s maximum foraging range) per traversed location by running our model with an increasing number of available locations and weighting them accordingly (Supporting Information 1, Table S1), determining what number was best to include by calculating the area under the receiver operator characteristic curve (AUC; Zweig and Campbell 1993) and Bayesian versions of R2 values (Gelman et al. 2019). We used the same methods to perform backward stepwise selection and identify the most parsimonious model (Supporting Information 1, Tables S3). AUC measures the accuracy of the habitat selection model’s explanatory and predictive powers; values range from 0 to 1, with values above 0.5 indicating that the model discriminates better than chance, and higher values representing better model performance (Fielding and Bell 1997).
These methods were then repeated to investigate whether birds selected to dive within areas that had different environmental conditions to those along their foraging routes. Here, we also used a Bernoulli distribution (0 = dive locations and 1 = traversed locations) to model diving habitat selection in response to the environmental metrics (Table 1), including a fixed effect of distance from the colony and a random effect of ‘trip ID per individual’. As with the previous habitat selection models, we again determined what number of traversed locations (the aforementioned random points throughout the associated foraging track where diving did not occur) was best to include by calculating the AUC (Zweig and Campbell 1993) and Bayesian versions of R2 values (Gelman et al. 2019), and used the same methods to perform backward stepwise selection and identify the most parsimonious model (Supporting Information 1, Tables S2 and S4).
Diurnal diving
To assess differences in RFB dive depth over the course of the diel cycle, we modelled the log-transformed maximum depth of each individual dive as a function of the time of day at which it took place within a Bayesian regression model. A log-transformation was used on the depth data due to the raw data being skewed. Time of day was fitted as a smooth term due to expected non-linearity following visual inspection of the raw data. We included a random effect of ‘bout ID’ nested within ‘individual ID’ to account for multiple dives occurring within the same bout, and multiple bouts being performed by the same individual.
Results
Dive distribution throughout the foraging journey
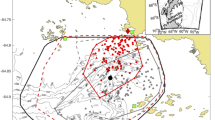
We recorded 1472 dives across 340 dive bouts from 43 foraging trips performed by 17 RFBs during their 2019 and 2022 breeding seasons. Dive bouts occurred between 31 and 411 km (mean = 140 km; sd = 91) from the colony and were composed of 2–30 individual dives (mean = 4; sd = 4; Fig. 1A). Distance from the colony had a small positive influence on dive bout duration (standardized effect size = 0.29, 95% highest posterior density (HPD) = − 0.03–0.63, with a 94% probability of the effect being above zero; Fig. 1B). Dive bouts varied in duration from 2 s (at 171 km from the colony) to 21 min (at 65 km from the colony), and 53% of bouts were less than 3 min in duration (mean = 4 min; sd = 13). Distance from the colony also had a small positive effect on distances between diving bouts (standardized effect size = 0.25, HPD = 0.18–0.36; Fig. 1C), 40% of which were less than 2 km apart (inter-bout distance mean = 14 km; sd = 29).
A Diving bout locations of red-footed boobies breeding on Diego Garcia, Chagos Archipelago, central Indian Ocean (yellow point). Foraging trips are indicated by a solid line and dive bouts are illustrated as points that vary in size in relation to the number of dives performed per bout. Data from 2019 and 2022 are shown in red and blue, respectively. The inset plot shows the locations of Barton Point (2019; yellow square) and East Island (2022; yellow triangle) within the Diego Garcia atoll. B and C The log-linear effects of distance from the colony (km) on bout duration (s) and inter-bout distance (km), respectively. The scatter plots illustrate raw data alongside the mean predicted model fits, with the shading representing 95% uncertainty intervals. The effect-size plots show the highest posterior density regions for the log-linear effects
Foraging habitat selection
Traversed vs available
There was no evidence that RFBs were more likely to traverse over waters of particular depths, (standardized effect size = 0.11, HPD = − 0.05–0.28), chlorophyll-a concentrations (standardized effect size = 0.19, HPD = − 0.02–0.42), or sea surface temperatures (standardized effect size = 0.11, HPD = − 0.12–0.36), in comparison to the conditions within the population’s foraging range (Fig. 2 A–C). Instead, birds generally selected to travel closer to the colony (standardized effect size of distance from the colony = − 0.72, HPD = − 0.91–− 053).
Probability of habitat selection whilst foraging by red-footed boobies breeding in the Chagos Archipelago during 2019 and 2022, given the sea floor depth (m) chlorophyll-a concentration (mg m−3) and sea surface temperature (oC). Solid lines indicate mean predicted model fit, the shaded areas represent 95% uncertainty intervals, and the points show the raw data. Area under the receiver operator characteristic curve (AUC) values are also shown. Panels A–C illustrate the probability of RFBs traversing a particular habitat, in comparison to other available locations. Panels D–F illustrate their probability of diving within a particular habitat as opposed to traversing it. Panels G–I illustrate the sea floor depth, chlorophyll-a concentration, and sea surface temperature in the central Indian Ocean in relation to the location of RFBs foraging tracks (lines) and dive bout locations (open circles)
Dive vs traversed
The areas where birds dived had relatively high chlorophyll-a concentrations (standardized effect size = 0.95, HPD = 0.57–1.35; Fig. 2E) and sea surface temperatures (standardized effect size = 1.04, HPD = 0.50–1.61; Fig. 2F) in comparison to those they traversed, but were not influenced by sea depth (the inclusion of depth within habitat selection models did not improve model fit; Supporting Information Table S4; standardised effect size when included = − 0.01, HPD = − 0.42–0.40; Fig. 2D). In comparison to where they traversed, birds selected to dive further from the colony (standardised effect size = 1.40. HPD = 0.93–1.89).
Diurnal diving
RFB diving was restricted to daylight hours, with few dives during dawn and dusk, and none at night (Fig. 3). Time of day had a non-linear effect on the maximum depth of individual dives (standardized effect size = 2.30, HPD = 0.60–4.72) which varied from 0.1 to 4.1 m (mean = 0.7 m; sd = 0.6), peaking at 10:56 (Fig. 3).
The maximum depth of individual dives (on the log scale; m) by red-footed boobies breeding in the Chagos Archipelago in 2019 and 2022 across the diel cycle. Night is shown in blue, dawn and dusk are in red, and daytime is in yellow. Dashed lines illustrate the mean predicted model fit of the non-linear relationship between time of day and log-transformed maximum dive depth (thickest dashed line), and the upper and lower 95% uncertainty intervals (thinner dashed lines)
Discussion
Dive distribution throughout the foraging journey
We show that RFB diving behaviour was influenced by their foraging trip ecology, the environmental conditions encountered throughout their foraging journeys, and the diel cycle. RFBs performed diving bouts throughout their foraging journeys (Fig. 1A) which were longest when birds were furthest from the colony (Fig. 1B), likely reflective of larger, higher density prey patches, far from land (Boyd 1996). High density prey patches may be less common close to the breeding colony because of prey depletion from the ca. 280,000 pairs of breeding seabirds in the Chagos Archipelago (Ashmole 1963; Carr et al. 2020). For example, at the Ascension Island seabird colony within the tropical Atlantic Ocean, flying fish densities were depleted close to the island and were highest ca. 300 km away, explained by an opposing trend in predatory seabird densities (Weber et al. 2021). Furthermore, a similar pattern of longer dive bouts in distant areas has been shown in other tropical sulids, including a Pacific Ocean population of Masked boobies (Sula dactylatra; Sommerfeld et al. 2015), evidencing a trade-off between diving when closer to the colony, and travelling further to forage on less depleted or less disrupted marine prey. Dive bout length may also be reflective of an individual’s current energy requirements and hunger levels (McHuron et al. 2018). Assuming that, due to physiological similarities, RFB flight costs are comparable to those of northern gannets (Morus bassanus; Birt-Friesen et al. 1989), it may be necessary for RFBs to engage in longer diving bouts when further from the colony to replenish the energetic reserves consumed via undertaking journeys of up to 1500 km (Trevail et al. 2023a).
RFB dive bouts were spatially distinct (Fig. 2C), in contrast to temperate, neritic foraging northern gannets whose dives tend to be clustered at the distal point (Hamer et al. 2009) and at stable oceanic features (Scales et al. 2014; Cox et al. 2016). Contrastingly, temperate breeding black-legged kittiwakes (Rissa tridactyla) feed throughout their foraging journeys when selecting ephemeral fronts (Trevail et al. 2021). Despite our sample size of only 17 individuals, the pattern observed in our study therefore highlights the ephemeral, patchy nature of prey in pelagic tropical ecosystems (Weimerskirch 2007).
Foraging habitat selection
When investigating multi-scale habitat selection, we found that habitat selection varied throughout the foraging process, differing between where birds decided to transit and where they decided to dive. There was no influence of the environment (namely sea depth, chlorophyll-a concentration and sea surface temperature) on foraging route choice, suggesting that alternative factors such as age (Votier et al. 2017), sex (Lewis et al. 2002), inter-colony competition (Trevail et al. 2023a), and knowledge gained outside the window when biologging devices were deployed, were more influential at this scale of habitat selection.
Although forage route choice was not influenced by environmental drivers, RFBs preferentially dived in warm, productive waters (Fig. 2E–F). Primary production is a key determinant of mid-trophic level prey and subsequent predator distributions (Green et al. 2020; de la Cruz et al. 2022), but we had not predicted that RFBs would demonstrate a preference for diving in warmer surface waters, hypothesising instead that cooler waters, indicative of productive upwellings, would be preferable (Soanes et al. 2021). However, warmer waters may impact fish behaviour with implications for catchability. For example, Indian Ocean billfish species prefer higher temperatures possibly due to the benefits of temperature-dependent swim speeds, which in turn may push prey towards the ocean’s surface (Thoya et al. 2022). Nevertheless, the influence of sea surface temperature on RFB prey capture is ambiguous, with different flying fish species (a main prey type) seeming to select for different temperatures (Randall et al. 2015; Churnside et al. 2017). We encourage further investigation into the factors driving the distribution and accessibility of fish and squid within the upper water column, critical drivers determining seabird foraging locations and dive probabilities (Boyd et al. 2015).
In addition to subsurface dives, RFBs likely also performed aerial captures of flying fish and squid that would not have been identified within our data processing, but which are evident from accelerometers (Weimerskirch et al. 2005b). To leap above the sea surface, flying fish require water temperatures of at least 20 °C to contract their muscles swiftly and take off (Davenport 1994), but whether their flight abilities have an upper temperature threshold remains nascent. Indeed, the occurrence of non-flying fishes in the diet of Abbott’s boobies (Papasula abbotti) increased with sea surface temperature around Christmas Island in the Indian Ocean, and the lengths of regurgitated flying fish were shorter (Hennicke and Weimerskirch 2014). We therefore pose the question of whether subsurface diving or aerial pursuit foraging is the most profitable strategy for RFBs and therefore whether areas of warmer sea surface temperatures really do represent the most favourable foraging conditions. Indeed, whilst we now know which variables are influencing when and where RFBs dive, knowledge gaps remain regarding both the mechanisms behind this, as well as what this might mean for the foraging ecology of tropical seabirds in a time of unprecedented global climate change and warming ocean temperatures. The aerial component of RFB prey gives rise to a complex system, and we encourage that future work seeks to quantify and compare the energetic costs of aerial pursuit foraging and foraging pelagically for subsurface prey.
Diurnal diving
RFBs dived during the daytime, this likely illustrative of factors including a predator avoidance strategy (Zavalaga et al. 2012), the diurnal availability of prey (Ménard et al. 2013), an affinity for foraging in association with subsurface predators (Weimerskirch et al. 2005b), and a reliance on daylight to aid visual prey identification and capture (Machovsky-Capuska et al. 2012). RFB dives were generally shallow, although maximum depth varied across the diel cycle (Fig. 3). Diving was most shallow around dawn and dusk, potentially due to tuna and billfish species feeding at high intensities at these times, forcing prey close to the ocean’s surface and within RFB capture range (Bestley et al. 2008; Williams et al. 2017). Contrastingly, RFBs dived deepest during the late morning when light penetration was high, resembling the diurnal dive depth patterns of northern gannets (Garthe et al. 2000).
The correlation between RFB dive depth and time of day is also possibly linked with prey detectability (Lewis et al. 2002; Darby et al. 2022), with deeper diving being reflective of prey availability. For example, birds may adjust their dive depths in response to variation in the location of prey species within the water column, or to target alternative deeper-dwelling prey (Chimienti et al. 2017). The diet of Indian Ocean RFBs is largely comprised of Exocoetidae flying fishes (e.g., Exocoetus volitans and Cheilopogon furcatus), Ommastrephinae flying squid (e.g., Sthenoteuthis oualaniensis), and Hemiramphidae halfbeak fishes (e.g., Euleptorhamphus viridis and Oxyporhamphus micropterus), all of which inhabit the top few meters of the water column during the day (Diamond 1974; Cherel et al. 2008). Despite their affinity for surface waters, it is likely that prey of different sizes, and different energetic values, are distributed across different depths, even within the upper epipelagic zone (Kappes et al. 2011). Indeed, although relationships between diurnal patterns in seabird dive depths and prey type have been explored at high latitudes (Wilson et al. 1993; Elliott and Gaston 2015), we know less about seabird diets and diel changes in prey availability within tropical waters. The consequences of diurnal changes in prey selection and dive depth may influence the energy budgets of RFBs, which are likely to experience heightened energetic costs whilst diving more deeply (Le Corre 1997).
Conclusion
Here, we reveal that RFB diving behaviour is influenced by environmental variability, with differences throughout foraging trips, in conjunction with environmental conditions, and across the diurnal cycle. We therefore shed light on the interplay of factors that influence how consumers find food in trophically unpredictable tropical ocean ecosystems. There is a pressing need to increase this understanding at a time when the world’s marine ecosystems are undergoing unprecedented change, altering environmental conditions, the ecology of lower trophic level species, and the energetics of the predators that consume them. Appropriate marine conservation and management is particularly critical at this time and should seek to focus on areas of importance for foraging tropical marine predators (Trevail et al. 2023b).
Data availability
All GPS data is available on the Seabird Tracking Database upon request (seabirdtracking.org, datasets #2099, Barton Point, and #2100, East Island). Associated R code is available on GitHub (github.com/RuthDunn/RFB_Diving).
References
Abrams PA (1991) Life history and the relationship between food availability and foraging effort. Ecology 72:1242–1252
Ashmole NP (1963) The regulation of numbers of tropical ocean birds. Ibis 103b:458–473
Ballance L, Pitman R (1999) S34.4: Foraging ecology of tropical seabirds. Proceedings of the 22nd International Ornithological Congress 2057–2071
Bestley S, Patterson TA, Hindell MA, Gunn JS (2008) Feeding ecology of wild migratory tunas revealed by archival tag records of visceral warming. J Anim Ecol 77:1223–1233
Birt-Friesen VL, Montevecchi WA, Cairns DK, Macko SA (1989) Activity-Specific Metabolic Rates of Free-Living Northern Gannets and Other Seabirds. Ecology 71:1257–1257
Boyd IL (1996) Temporal Scales of Foraging in a Marine Predator. Ecology 77:426–434
Boyd C, Castillo R, Hunt GL Jr, Punt AE, VanBlaricom GR, Weimerskirch H, Bertrand S (2015) Predictive modelling of habitat selection by marine predators with respect to the abundance and depth distribution of pelagic prey. J Anim Ecol 84:1575–1588
Bürkner P-C (2017) brms: An R Package for Bayesian Multilevel Models Using Stan. J Stat Softw 80:1–28
Calenge C (2006) The package adehabitat for the R software: a tool for the analysis of space and habitat use by animals. Ecol Modell 197:516–519
Carr P, Votier S, Koldewey H, Godley B, Wood H, Nicoll MAC (2020) Status and phenology of breeding seabirds and a review of Important Bird and Biodiversity Areas in the British Indian Ocean Territory. Bird Conserv Int 31:14–34
Cherel Y, Corre ML, Jaquemet S, Ménard F, Richard P, Weimerskirch H (2008) Resource partitioning within a tropical seabird community: New information from stable isotopes. Mar Ecol Prog Ser 366:281–291
Chimienti M, Cornulier T, Owen E, Bolton M, Davies IM, Travis JMJ, Scott BE (2017) Taking movement data to new depths: Inferring prey availability and patch profitability from seabird foraging behavior. Ecol Evol 7:10252–10265
Churnside J, Wells RJD, Boswell K, Quinlan J, Marchbanks R, McCarty B, Sutton T (2017) Surveying the distribution and abundance of flying fishes and other epipelagics in the northern Gulf of Mexico using airborne lidar. Bull Mar Sci 93:591–609
Cox SL, Miller PI, Embling CB, Scales KL, Bicknell AWJ, Hosegood PJ, Morgan G, Ingram SN, Votier SC (2016) Seabird diving behaviour reveals the functional significance of shelf-sea fronts as foraging hotspots. R Soc Open Sci 3:160317
Darby J, Clairbaux M, Bennison A, Quinn JL, Jessopp MJ (2022) Underwater visibility constrains the foraging behaviour of a diving pelagic seabird. Proc Biol Sci 289:20220862
Davenport J (1994) How and why do flying fish fly? Rev Fish Biol Fish 4:184–214
de la Cruz A, Ramos F, Tornero J, Rincón MM, Jiménez MP, Arroyo GM (2022) Seabird distribution is better predicted by abundance of prey than oceanography. A case study in the Gulf of Cadiz (SW, Iberian Peninsula). ICES J Mar Sci 79:204–217
Diamond AW (1974) The Red-footed booby on Aldabra Atoll, Indian Ocean. Ardea 62:196–218
Duckworth J, O’Brien S, Petersen IK, Petersen A, Benediktsson G, Johnson L, Lehikoinen P, Okill D, Väisänen R, Williams J, Williams S, Daunt F, Green JA (2021) Spatial and temporal variation in foraging of breeding red-throated divers. J Avian Biol 52:e02702
Elliott KH, Gaston AJ (2009) Accuracy of depth recorders. Waterbirds 32:183–191
Elliott KH, Gaston AJ (2015) Diel vertical migration of prey and light availability constrain foraging in an Arctic seabird. Mar Biol 162:1739–1748
Fielding AH, Bell JF (1997) A review of methods for the assessment of prediction errors in conservation presence/absence models. Environ Conserv 24:38–49
Garthe S, Benvenuti S, Montevecchi WA (2000) Pursuit plunging by northern gannets (Sula bassana) feeding on capelin (Mallotus villosus). Proc Biol Sci 267:1717–1722
Gelman A, Goodrich B, Gabry J, Vehtari A (2019) R-squared for Bayesian Regression Models. Am Stat 73:307–309
Genin A (2004) Bio-physical coupling in the formation of zooplankton and fish aggregations over abrupt topographies. J Mar Syst 50:3–20
Green DB, Bestley S, Trebilco R, Corney SP, Lehodey P, McMahon CR, Guinet C, Hindell MA (2020) Modelled mid-trophic pelagic prey fields improve understanding of marine predator foraging behaviour. Ecography 43:1014–1026
Grémillet D, Dell’Omo G, Ryan PG, Peters G, Ropert-Coudert Y, Weeks SJ (2004) Offshore diplomacy, or how seabirds mitigate intra-specific competition: a case study based on GPS tracking of Cape gannets from neighbouring colonies. Mar Ecol Prog Ser 268:265–279
Halsey LG, Bost C-A, Handrich Y (2007) A thorough and quantified method for classifying seabird diving behaviour. Polar Biol 30:991–1004
Hamer KC, Humphreys EM, Magalhães MC, Garthe S, Hennicke J, Peters G, Grémillet D, Skov H, Wanless S (2009) Fine-scale foraging behaviour of a medium-ranging marine predator. J Anim Ecol 78:880–889
Hennicke JC, Weimerskirch H (2014) Coping with variable and oligotrophic tropical waters: foraging behaviour and flexibility of the Abbott’s booby Papasula abbotti. Mar Ecol Prog Ser 499:259–273
Hijmans RJ (2022) geosphere: Spherical Trigonometry. R package version 1.5–18
Humphries NE, Queiroz N, Dyer JRM, Pade NG, Musyl MK, Schaefer KM, Fuller DW, Brunnschweiler JM, Doyle TK, Houghton JDR, Hays GC, Jones CS, Noble LR, Wearmouth VJ, Southall EJ, Sims DW (2010) Environmental context explains Lévy and Brownian movement patterns of marine predators. Nature 465:1066–1069
Jaquemet S, Le Corre M, Marsac F, Potier M, Weimerskirch H (2005) Foraging habitats of the seabird community of Europa Island (Mozambique Channel). Mar Biol 147:573–582
Kappes MA, Weimerskirch H, Pinaud D, Le Corre M (2011) Variability of resource partitioning in sympatric tropical boobies. Mar Ecol Prog Ser 441:281–294
Le Corre M (1997) Diving depths of two tropical pelecaniformes: The red-tailed tropicbird and the red-footed booby. Condor 99:1004
Le Guen C, Kato A, Raymond B, Barbraud C, Beaulieu M, Bost CA, Delord K, MacIntosh AJJ, Meyer X, Raclot T, Sumner M, Takahashi A, Thiebot JB, Ropert-Coudert Y (2018) Reproductive performance and diving behaviour share a common sea-ice concentration optimum in Adélie penguins (Pygoscelis adeliae). Glob Chang Biol 24:5304–5317
Lewis S, Benvenuti S, Dall Antonia L, Griffiths R, Money L, Sherratt TN, Wanless S, Hamer KC (2002) Sex-specific foraging behaviour in a monomorphic seabird. Proceed Royal Soc B Biol Sci 269:1687–1693
Machovsky-Capuska GE, Howland HC, Raubenheimer D, Vaughn-Hirshorn R, Würsig B, Hauber ME, Katzir G (2012) Visual accommodation and active pursuit of prey underwater in a plunge-diving bird: the Australasian gannet. Proc Biol Sci 279:4118–4125
Mayor SJ, Schneider DC, Schaefer JA, Mahoney SP (2009) Habitat Selection at Multiple Scales. Écoscience 16:238–247
McElreath R (2020) Statistical Rethinking: A Bayesian Course with Examples in R and Stan, 2nd edn. Chapman & Hall, London
McHuron EA, Hazen E, Costa DP (2018) Constrained by consistency? Repeatability of foraging behavior at multiple timescales for a generalist marine predator. Mar Biol 165:222
Ménard F, Potier M, Jaquemet S, Romanov E, Sabatié R, Cherel Y (2013) Pelagic cephalopods in the western Indian Ocean: New information from diets of top predators. Deep Sea Res Part 2 Top Stud Oceanogr 95:83–92
Mendelssohn R (2022) rerddapXtracto: Extracts Environmental Data from ‘ERDDAP’ Web Services. R Package Version 1(1):4
Mendez L, Borsa P, Cruz S, de Grissac S, Hennicke J, Lallemand J, Prudor A, Weimerskirch H (2017) Geographical variation in the foraging behaviour of the pantropical red-footed booby. Mar Ecol Prog Ser 568:217–230
Muggeo VMR (2017) Interval estimation for the breakpoint in segmented regression: a smoothed score-based approach. Aust N Z J Stat 59:311–322
Paiva VH, Guilford T, Meade J, Geraldes P, Ramos JA, Garthe S (2010) Flight dynamics of Cory’s shearwater foraging in a coastal environment. Zoology (jena) 113:47–56
R Core Team (2022) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria
Randall LL, Smith BL, Cowan JH, Rooker JR (2015) Habitat characteristics of bluntnose flyingfish Prognichthys occidentalis (Actinopterygii, Exocoetidae), across mesoscale features in the Gulf of Mexico. Hydrobiologia 749:97–111
Riotte-Lambert L, Matthiopoulos J (2020) Environmental predictability as a cause and consequence of animal movement. Trends Ecol Evol 35:163–174
Rodríguez-Malagón MA, Camprasse ECM, Angel LP, Arnould JPY (2020) Geographical, temporal and individual factors influencing foraging behaviour and consistency in Australasian gannets. R Soc Open Sci 7:181423
Scales KL, Miller PI, Embling CB, Ingram SN, Pirotta E, Votier SC (2014) Mesoscale fronts as foraging habitats: composite front mapping reveals oceanographic drivers of habitat use for a pelagic seabird. J R Soc Interface 11:20140679
Sibly RM, Nott HMR, Fletcher DJ (1990) Splitting behaviour into bouts. Anim Behav 39:63–69
Soanes LM, Green JA, Bolton M, Milligan G, Mukhida F, Halsey LG (2021) Linking foraging and breeding strategies in tropical seabirds. J Avian Biol 52:1–13
Sommerfeld J, Kato A, Ropert-Coudert Y, Garthe S, Wilcox C, Hindell MA (2015) Flexible foraging behaviour in a marine predator, the Masked booby (Sula dactylatra), according to foraging locations and environmental conditions. J Exp Mar Bio Ecol 463:79–86
Thoya P, Kadagi NI, Wambiji N, Williams SM, Pepperell J, Möllmann C, Schiele KS, Maina J (2022) Environmental controls of billfish species in the Indian Ocean and implications for their management and conservation. Divers Distrib 28:1554–1567
Tomlinson S, Arnall SG, Munn A, Bradshaw SD, Maloney SK, Dixon KW, Didham RK (2014) Applications and implications of ecological energetics. Trends Ecol Evol 29:280–290
Trevail AM, Green JA, Bolton M, Daunt F, Harris SM, Miller PI, Newton S, Owen E, Polton JA, Robertson G, Sharples J, Patrick SC (2021) Environmental heterogeneity promotes individual specialisation in habitat selection in a widely distributed seabird. J Anim Ecol 90:2875–2887
Trevail AM, Wood H, Carr P, Dunn R, Nicoll MA, Votier SC, Freeman R (2023a) Multi-colony tracking reveals segregation in foraging range, space use, and timing in a tropical seabird. Mar Ecol Prog Ser 724:155–165
Trevail AM, Nicoll MAC, Freeman R, Le Corre M, Schwarz J, Jaeger A, Bretagnolle V, Calabrese L, Feare C, Lebarbenchon C, Norris K, Orlowski S, Pinet P, Plot V, Rocamora G, Shah N, Votier SC (2023b) Tracking seabird migration in the tropical Indian Ocean reveals basin-scale conservation need. Curr Biol 33:5247-5256.e4
Veit RR, Harrison NM (2017) Positive interactions among foraging seabirds, marine mammals and fishes and implications for their conservation. Front Ecol Evol 5:1–8
Votier SC, Fayet AL, Bearhop S, Bodey TW, Clark BL, Grecian J, Guilford T, Hamer KC, Jeglinski JWE, Morgan G, Wakefield E, Patrick SC (2017) Effects of age and reproductive status on individual foraging site fidelity in a long-lived marine predator. Proc Biol Sci 284:20171068
Wakefield ED, Cleasby IR, Bearhop S, Bodey TW, Davies RD, Miller PI, Newton J, Votier SC, Hamer KC (2015) Long-term individual foraging site fidelity—why some gannets don’t change their spots. Ecology 96:3058–3074
Weber SB, Richardson AJ, Brown J, Bolton M, Clark BL, Godley BJ, Leat E, Oppel S, Shearer L, Soetaert KER, Weber N, Broderick AC (2021) Direct evidence of a prey depletion ‘halo’ surrounding a pelagic predator colony. Proc Natl Acad Sci U S A 118:e2101325118
Weimerskirch H (2007) Are seabirds foraging for unpredictable resources? Deep Sea Res Part 2 Top Stud Oceanogr 54:211–223
Weimerskirch H, Le Corre M, Jaquemet S, Potier M, Marsac F (2004) Foraging strategy of a top predator in tropical waters: great frigatebirds in the Mozambique Channel. Mar Ecol Prog Ser 275:297–308
Weimerskirch H, Le Corre M, Jaquemet S, Marsac F (2005a) Foraging strategy of a tropical seabird, the red-footed booby, in a dynamic marine environment. Mar Ecol Prog Ser 288:251–261
Weimerskirch H, Le Corre M, Ropert-Coudert Y, Kato A, Marsac F (2005b) The three-dimensional flight of red-footed boobies: adaptations to foraging in a tropical environment? Proceed Royal Soc B Biol Sci 272:53–61
Williams SM, Holmes BJ, Tracey SR, Pepperell JG, Domeier ML, Bennett MB (2017) Environmental influences and ontogenetic differences in vertical habitat use of black marlin (Istiompax indica) in the southwestern Pacific. R Soc Open Sci 4:170694
Wilson F, Puetz K, Bost CA, Culik BM, Bannasch R, Reins T, Adelung D (1993) Diel dive depth in penguins in relation to diel Vertical migration of prey: whose dinner by candlelight? Mar Ecol Prog Ser 94:101–104
Zavalaga CB, Emslie SD, Estela FA, Müller MS, Dellomo G, Anderson DJ (2012) Overnight foraging trips by chick-rearing Nazca Boobies Sula granti and the risk of attack by predatory fish. Ibis 154:61–73
Zweig MH, Campbell G (1993) Receiver-operating characteristic (ROC) plots: a fundamental evaluation tool in clinical medicine. Clin Chem 39:561–577
Acknowledgements
We thank the Bertarelli Foundation for supporting this work as well as two anonymous reviewers for helpful comments on earlier drafts of this manuscript.
Funding
This work was funded by the Bertarelli Foundation and contributed to the Bertarelli Programme in Marine Science. The authors have no relevant financial or non-financial interests to disclose.
Author information
Authors and Affiliations
Contributions
This manuscript originated from work done by Julia Ramsden during a Masters project. All authors contributed to the study conception and design. Robin Freeman, Alice Trevail, Hannah Wood, and Stephen Votier performed data collection. Ruth Dunn performed the analysis and wrote the first draft of the manuscript. All authors commented on previous versions of the manuscript and read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have not disclosed any competing interests.
Ethical approval
All applicable institutional guidelines for sampling, care, and experimental use of birds were followed and the necessary approvals were obtained. For birds that were captured and handled in 2019, methods were reviewed by the Zoological Society of London Ethics Committee and were approved by the British Trust for Ornithology special methods panel. In 2022, data we collected under ethics approval from the University of Exeter, and British Trust for Ornithology approved special methods. Avian research on Diego Garcia was undertaken under permit numbers 0000SE19 and 0011SE22. Documentary evidence is available on request.
Additional information
Responsible Editor: V. Paiva.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Dunn, R.E., Freeman, R., Nicoll, M.A. et al. From route to dive: multi-scale habitat selection in a foraging tropical seabird. Mar Biol 171, 124 (2024). https://doi.org/10.1007/s00227-024-04445-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00227-024-04445-y