Abstract
Purpose
There is an emerging role of the use of Prostate-Specific Membrane Antigen (PSMA) Positron Emission Tomography (PET) in renal cell carcinoma. Herein, we report our experience in use of PSMA PET in recurrent or metastatic renal cell carcinoma (RCC).
Methods
A retrospective analysis of all patients who underwent PSMA PET for suspected recurrent or de-novo metastatic RCC between 2015 and 2020 at three institutions was performed. The primary outcome was change in management (intensification or de-intensification) following PSMA PET scan. Secondary outcomes included histopathological correlation of PSMA avid sites, comparison of sites of disease on PSMA PET to diagnostic CT and time to systemic treatment.
Results
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
51 PSMA PET scans were performed in 48 patients for assessment for recurrence on conventional imaging (n=35, 73%) or de-novo metastatic disease (n=13, 27%). PSMA PET changed management in 11 patients (11/35, 31%) with recurrent disease, and 3 patients (3/13, 23%) with de-novo metastatic disease. For recurrent disease, better delineation of suspected recurrence led to treatment intensification in 23% (8/35; additional/altered resection, 17%; systemic therapy rather than resection, 6%), and treatment de-intensification in 9% (3/35; surveillance rather than systemic therapy, 3%; resection, 3%; and biopsy, 3%). For de-novo metastatic disease, PSMA PET lead to treatment intensification in 23% (3/13) of patients (systemic from local therapy, 15%; different systemic therapy, 8%). 30 patients underwent biopsy of PSMA avid sites, with clear cell RCC histology in 87%, as well as de-differentiated sarcomatoid RCC (7%), carcinoid (3%) and urothelial cancer (3%).
PSMA PET provided clinically useful information for patients with diagnostic CT staging, leading to a change in management in 29% of patients with advanced RCC. Prospective research is warranted to further investigate the oncological outcome of PSMA PET determined management in advanced RCC.
Introduction
Kidney cancer is the 7th most diagnosed malignancy in the developed world, with renal cell carcinoma (RCC) encompassing approximately 75% [1]. Unfortunately, 20-30% of patients are diagnosed with metastatic disease at diagnosis, and up to 40% develop recurrence despite adequate surgery for local disease [2, 3]. The management of advanced RCC is dictated by radiological imaging for staging. Oligo-metastatic RCC for instance, can be managed with local therapies such as metastasectomy or radiation, which can delay the requirement for systemic therapy and improve survival outcomes [4, 5]. More extensive recurrence, or de-novo metastatic disease is more typically managed with systemic therapy such as tyrosine kinase inhibitors, immunotherapy or their combination [6]. Computerised tomography (CT) scan is routinely used for delineation of extent of disease, or staging, and assessing response to treatment. However, CT is limited by standard criteria for accurate assessment of nodal disease, and in the detection of small volume local recurrence and metastasis [7].
Prostate-specific membrane antigen (PSMA) positron emission tomography (PET) uses a radioactive tracer, that binds to PSMA protein, which has better diagnostic accuracy than CT and bone scan for prostate cancer due to PSMA protein over-expression [8]. Interestingly, PSMA protein is also expressed in the neo-vasculature of other solid tumours including RCC, with tracers binding to the extra-cellular site [9, 10]. Preliminary reports have suggested improved performance for PSMA PET over conventional imaging in detecting RCC metastases [11, 12].
Therefore, this study aimed to assess the clinical impact of PSMA PET on the management of patients with recurrent or de-novo metastatic RCC when used across multiple institutions.
Methods
Study design and patient population
A multicentre retrospective study was performed across three tertiary hospitals (Royal Brisbane Women’s Hospital, Princess Alexandra Hospital and Wesley Hospital). Inclusion criteria were adult patients who underwent PSMA PET during the period of January 2015 to June 2020 for restaging of resected RCC with suspected recurrence on conventional imaging or initial staging of metastatic RCC. Patients were excluded if PSMA PET was performed for localised RCC or other cancers. This study received ethics and governance approval (Reference numbers HREC/2020/QRBWH/64912; UCH 2020.19.329)
Imaging
Siemens Biograph PET scanner (Siemens AG, Munich, Germany) were used at two sites (Royal Brisbane and Women’s Hospital and Princess Alexandra Hospital), while either Philips Ingenuity TF 128 slice PET/CT scanner (Phillips Healthcare, Amsterdam, Netherlands) or GE Discovery MI DR PET CT scanner (GE Healthcare, Chicago, USA) was used at one site (Wesley Hospital).
All sites used [68Ga]Ga-PSMA-11 initially, with two sites (Royal Brisbane and Women’s Hospital and Princess Alexandra Hospital) migrating to use of [18F]F-PSMA-1007 due to lower renal excretion and preference. Imaging were performed from skull vertex to the upper thighs. [68Ga]Ga-PSMA-11 PET images were obtained following intravenous delivery of average 145 MBq (Range: 100-162 MBq) and an average uptake time of 62 minutes (Range: 35-98 minutes). [18F]F-PSMA-1007 PET images were obtained following intravenous delivery of average 242 MBq (Range: 163-296 MBq) and an average uptake time of 120 min (Range: 57-169 minutes). Low dose CT was also performed for anatomical correlation at Royal Brisbane and Women’s Hospital and Princess Alexandra Hospital, while diagnostic CT was performed at Wesley Hospital. PET/CT Maximum intensity projection (MIP) images are shown in Supplementary Fig. 1, illustrating the differences in normal physiological distribution between the two tracers. Lesions were considered suspicious for metastasis when PSMA uptake was greater than background physiological uptake in a typical distribution for metastatic disease, with anatomical correlation on CT. More complex cases (e.g., solitary metastatic disease, or an unusual metastatic site) were discussed at multidisciplinary meetings for further evaluation, including additional imaging or biopsy. Images were interpreted by an experienced nuclear medicine specialist, while diagnostic CT was similarly interpreted by an experienced radiologist.
Data collection
PSMA PET databases at each institution were generated and queried for RCC patients with recurrent or de-novo metastatic disease. All available electronic medical records were accessed for data collection. Demographic data included age, sex, primary tumour histology (including variant morphology, such as sarcomatoid or rhabdoid differentiation) and line of systemic therapy prior to PSMA PET. Haemoglobin, neutrophil, platelet count, corrected calcium, Karnofsky performance status (≥80% or <80%) and time to systemic therapy were recorded for patients with metastatic disease. International metastatic renal cell carcinoma database consortium (IMDC) prognostic score at time of systemic therapy, or at time of PSMA PET (if not receiving systemic therapy) was calculated [13].
PSMA PET reports were reviewed and sites of disease and maximum standardized uptake (SUVMax) values were recorded. Diagnostic CT reports were reviewed, and sites of disease recorded if performed within 4 weeks prior to PSMA PET. Concordant findings were defined as the same sites of disease identified on PSMA PET and CT. Dis-concordant findings were defined as new sites of disease identified on PSMA PET, not identified on diagnostic CT, or PSMA PET not showing avidity in suspected metastatic sites on diagnostic CT. Histopathology of biopsied PSMA avid sites were also recorded if performed.
Disease classification
Patients were grouped into either 1) recurrent disease, where PSMA PET scan was performed to delineate diagnosis and management of suspected local or distant recurrence on surveillance conventional imaging post nephrectomy for RCC. 2) In the initial or subsequent management of de-novo metastatic disease.
Oligometastatic disease was defined as less than 5 metastatic sites that were amenable to definitive surgery or radiation.
Outcomes
The primary outcome of this study was to determine the change in clinical management following PSMA PET. Change in management was classified as intensification (change in modality of treatment, change in line of treatment or treatment to different site), or de-intensification (surveillance or biopsy rather than systemic therapy or resection). Each medical record was reviewed by two independent investigators for data confirmation, including consensus on management before and after PSMA PET, with a difference of opinion moderated by a third independent investigator.
The secondary outcomes were histological correlation of PSMA avid sites with PET parameters [SUVmax], comparison of PSMA PET to diagnostic CT, and time to systemic treatment in patients who underwent surveillance or local treatment [surgery or radiation] based on PSMA PET.
Statistical analysis
Data was managed with Microsoft Excel. Management plans before and after PSMA PET are illustrated via Sankey diagram which was generated via Microsoft Excel. Baseline characteristics were compared using Fisher exact test. P value <0.05 was considered significant. Kaplan Meier time to event analysis was used to calculate median time to treatment. STATA v17 software was used for the above analysis.
Results
120 patients were screened for eligibility, of which 51 PSMA PET scans in 48 patients were included for analysis. Patients were excluded because PSMA PET was performed for primary staging or similar or inadequate documentation (Fig. 1). 35 patients (35/48, 73%) underwent [68Ga]Ga-PSMA-11, while 13 patients (13/48, 27%) underwent [18F]F-PSMA-1007. 35 patients (35/48, 73%) had suspected recurrent locally or distant metastatic disease, while 13 patients (13/48, 27%) had de-novo metastatic disease. Median age was similar between recurrent and de-novo metastatic groups [64 v 63 years]. Primary tumour was predominantly clear cell RCC in both groups [100% v 92% p=0.27]. PSMA PET was positive in 86% (44/51) of scans. A higher proportion of favourable IMDC risk was observed in the recurrent group (60% v 23%; p=0.05), and lower proportion of intermediate IMDC risk [26% v 62% p=0.04), likely driven by short interval (less than 1 year) from diagnosis to treatment (11% vs 62%; p<0.01), and thrombocytosis (0% v 23% p=0.02). The de-novo metastatic group had a higher proportion of previous systemic therapy use (31% vs 3%, p=0.02), which was predominantly Tyrosine kinase inhibitor (77% vs 31%, p=<0.01). Demographics are outlined in Table 1.
Overall, there was a change in management in 14 patients (14/48, 29%), with intensification observed for 11 patients (11/48, 23%) and de-intensification observed for 3 patients (3/48, 6%) due to PSMA PET.
Management change - Recurrent disease
37 PSMA PET scans were performed in 35 patients for suspected recurrent disease on conventional imaging. Change in management was observed for 11 patients (11/35, 31%), grouped according to pre- and post-PET pathways (Fig. 2a) with details included in Table 2.
Treatment or investigation intensification after PSMA PET was observed for 8 patients (8/35, 23%). We observed conversion from initial surveillance to resection or biopsy (4/35, 11%) because of PSMA avidity at suspected sites that were equivocal on CT scan, or more extensive surgery for recurrence (2/35, 6%), when PSMA PET identified a greater burden of disease. Conversion from resection or radiation for oligometastatic disease based on CT to systemic therapy (2/35, 6%) occurred due to poly-metastatic disease being identified on PSMA PET.
De-intensification of treatment or investigation after PSMA PET was observed for 3 patients (3/35, 9%) due to a lack of avidity in a suspected site of recurrence on CT, from initial systemic therapy (1/35, 3%), resection (1/35, 3%) and biopsy (1/35, 3%).
Although management was not altered, PSMA PET influenced management by confirming oligometastatic disease in 7 patients (7/35, 20%), proceeding with surgery or radiation as planned. In 2 patients (2/35, 6%) a lack of PSMA avidity of suspected recurrence site confirmed management of surveillance.
Management change – De-novo metastatic disease
A total of 14 PSMA PET scans were performed in the de-novo metastatic setting in 13 patients. Change in management was observed for 3 patients (3/13, 23%), grouped according to pre- and post-PET pathways (Fig. 2b) with details included in Table 2.
All 3 patients had intensification of treatment, with two patients proceeding to systemic therapy instead of oligometastatic therapy, when unresectable or poly-metastatic disease was identified on PSMA PET. One patient had a change in line of systemic therapy when a new gastric metastasis was identified on PSMA PET which was not evident on CT.
PSMA PET provided confirmation of oligometastatic progression identified on CT (1/13, 8%), which proceeded with planned radiation to a right hilar node, while the same line of systemic therapy was continued in 2 patients based on PSMA PET findings.
Imaging comparison
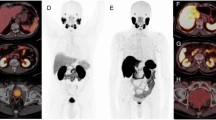
Corresponding diagnostic CT within one month prior to PSMA PET was available for 27 patients. The majority of CT scans were discordant with PSMA PET (17/27, 63%), mostly due to PSMA PET highlighting more sites of disease (n=14; Fig. 3a), including sub-diaphragmatic nodal disease (18.5%), bone metastasis (14.8%) and adrenal metastasis (14.8%; Supplementary Table 1). PSMA PET was discordant with CT scan findings (n=3; Fig. 3b), due to lack of avidity, in pulmonary metastasis (7.4%) and mediastinal nodal disease (3.7%).
[68Ga]Ga-PSMA-11 was performed for 17 patients (17/27, 63%), with PSMA PET highlighting more sites of disease in comparison to CT in 9 patients (9/17, 53%). [18F]F-PSMA-1007 was performed for 10 patients (10/27, 37%) and identified more sites of disease in 50% (5/10) of patients. All 3 patients who had discordant findings due to lack of avidity on PSMA PET received [18F]F-PSMA-1007.
For patients with recurrent disease, 25 patients (25/35, 71%) underwent [68Ga]Ga-PSMA-11, while 10 patients (10/35, 29%) underwent [18F]F-PSMA-1007 in this cohort. Percentage of patients requiring change in management was similar in both groups [28% vs 30% p=1.0]. For patients with de-novo metastatic disease, 10 patients (10/13, 77%) underwent [68Ga]Ga-PSMA-11, while 3 patients (3/13, 23%) underwent [18F]F-PSMA-1007 in this subgroup. All 3 patients who had a change in management received [68Ga]Ga-PSMA-11 [30% vs 0% p=0.53].
Histopathological correlation with imaging
Biopsy of PSMA avid sites occurred for 30 patients, of which most (28/30, 93%) were clear cell renal carcinoma (2 of these had sarcomatoid differentiation). Other morphologies included carcinoid (1/30, 3%, [18F]F-PSMA-1007) and urothelial carcinoma (1/30, 3%, [68Ga]Ga-PSMA-11). Two patients (2/30; 6%, [18F]F-PSMA-1007 and [68Ga]Ga-PSMA-11) proceeded to biopsy/resection of non-PSMA avid site due to suspected recurrence on CT, both returned benign histology. Median SUVmax of patients with clear cell renal carcinoma was 14 (IQR: 8.35-23.3). The median SUVmax did not differ for clear cell renal carcinoma between [68Ga]Ga-PSMA-11 and [18F]F-PSMA-1007 tracer [14 vs 14.4]. The median SUVmax and histology is located in Table 3.
Oncological outcomes – Recurrent disease
Patients with oligometastatic recurrent disease based on PSMA PET (14/35, 40%) were treated with local therapies, such as surgical resection (12/35, 34%) and radiation (2/35, 6%). During the follow-up (median 36 months, IQR 16-68), 4 patients (4/35, 11%) required systemic therapy after local therapy with a median time to commencement not reached [IQR: 52- NR months]. 2 patients (2/35, 6%) started systemic therapy within 12 months. 2 patients died from non-RCC causes before requiring systemic therapy.
During the follow-up (median 47 months, IQR: 34-55) of patients with suspected recurrence on CT who underwent surveillance based on PSMA PET results (10/35,29%), 5 patients (5/35, 14%) commenced systemic therapy. Median time to starting systemic therapy from PSMA PET was 36.1 months (IQR 21-49), with 1 patient requiring systemic therapy within 12 months. 1 patient died from non-RCC cause. 1 patient died from RCC cause who declined systemic therapy. Median time to starting systemic therapy was numerically longer for patients with negative PSMA PET in comparison to positive [36 v 21.1months p=0.28] but did not reach statistical significance.
Discussion
PSMA PET has the potential to improve precision in RCC staging and appropriateness of local or systemic therapy. Here, we observed that PSMA PET performed in patients with recurrent or de-novo metastatic RCC changed management in 29% (14/48), which was numerically higher for patients with recurrent than de-novo metastatic disease (31% vs 23%, p=0.73). Management plans were more likely to be intensified than de-intensified (23% vs 6%) due to additional or absent PSMA activity, respectively. Despite initial de-intensification, some patients still progressed to require systemic therapy (n=5) with variable interval treatment period. This is consistent with the natural history of RCC, with variable and late recurrences noted. The percentage of patients who required change in management was similar despite the tracer used.
Other centres have reported higher impacts, such as Udovicich and colleagues who reported management change for 49% [14]. Their cohort had a higher proportion of oligometastatic disease considered for local therapies based on conventional imaging compared to the current cohort (72% vs 33%, respectively), where management is more likely to change according to the current dataset (44%). The management impact is likely to be most significant for the oligometastatic cohort, with potential for change to either systemic therapy or surveillance based on either more or less extensive disease noted on PSMA PET, respectively. Correct characterisation of disease is important to avoid potentially unnecessary morbidity (e.g. surgery), which was outlined in a series of 14 patients with oligometastatic clear cell RCC based on CT, where 21.4% had poly-metastatic disease when evaluated with PSMA PET [15]. Conversely, accurate and earlier identification of oligometastatic disease with PSMA PET may prompt earlier local therapy (3/51, 6% in this study) and delay the initiation of systemic therapy post local therapy, shown here with 57% (8/14) of patients alive and not requiring systemic therapy to date, with a median time to systemic treatment not reached.
CT with intravenous contrast is the current standard of care for the staging of RCC. Limitations of CT include inability to distinguish between reactive or metastasis in enlarged lymph nodes and, difficulty in detecting small volume local recurrence and metastases. Several case series have highlighted the improved ability of PSMA PET to detect metastasis, including nodal disease too small to be characterised on CT and bone metastasis [16]. A summary of these studies is outlined in Supplementary Table 2. Conversely, a negative PSMA PET may be useful to rule out recurrence despite CT findings, indicated here by both management change patterns and negative histopathology of non-avid PSMA sites. While some patients with negative PSMA PET progressed to systemic therapy, 43% of patients in this subgroup are alive and have not required systemic therapy to date. Other molecular imaging techniques, such as FDG PET, may complement PSMA PET for improving precision of staging [17], while PSMA PET may assist with local staging (including tumour thrombus characterisation) [18].
In addition to precise staging, molecular imaging techniques may assist with assessing treatment response to systemic therapy in metastatic RCC. We found that, despite stable disease on CT, PSMA PET indicated progressive disease prompting change in systemic therapy for 1 patient, while partial responses were found in 2 patients, with one ceasing immunotherapy due to toxicity. These findings are supported by an observational study by Mittlemier and colleagues, where 9 out of 11 patients had stable disease after systemic therapy per CT assessment, however PSMA PET showed that most (6 out of 9 patients) had either complete or partial response and 1 patient had progressive disease [19].
The strengths of this study include the multi-centre, real-world data including multiple tracers and is one of the largest available series of PSMA PET. The larger number of biopsies or resections performed on PSMA avid sites for histopathological correlation was also a strength. Histopathological correlation of all PSMA avid sites is not appropriate in clinical practice, but was mitigated by assessment of atypical cases, such as solitary sites or equivocal diagnostic CT findings. The limitations include the retrospective nature and biased patient selection for PSMA PET imaging due to suspicion on CT, as well as different tracers used which may affect PSMA ligand avidity for non-biological reasons [20]. Retrospective determination of change in management is a limitation, but was mitigated through use of multiple, independent assessors.
In conclusion, PSMA PET provided clinically useful information in addition to conventional imaging, with high pathological concordance, to result in change in management for 29% of patients. These preliminary data support potential benefit of PSMA PET in renal cancer staging, both for accurate disease characterisation and potential response to treatment. Prospective trials are encouraged to further investigate the prognostic and predictive characteristics of PSMA PET for advanced RCC.
Data availability
The datasets generated during and/or analysed during the current study are not publicly available in order to protect patient privacy. The corresponding author may provide anonymised data on reasonable request.
References
Padala SA, Barsouk A, Thandra KC, Saginala K, Mohammed A, Vakiti A, et al. Epidemiology of Renal Cell Carcinoma. World J Oncol. 2020;11:79–87. https://doi.org/10.14740/wjon1279.
Ahn T, Roberts MJ, Abduljabar A, Joshi A, Perera M, Rhee H, et al. A Review of Prostate-Specific Membrane Antigen (PSMA) Positron Emission Tomography (PET) in Renal Cell Carcinoma (RCC). Mol Imaging Biol. 2019;21:799–807. https://doi.org/10.1007/s11307-018-01307-0.
Tyson MD, Chang SS. Optimal Surveillance Strategies After Surgery for Renal Cell Carcinoma. J Natl Compr Canc Netw. 2017;15:835–40. https://doi.org/10.6004/jnccn.2017.0102.
Kavolius JP, Mastorakos DP, Pavlovich C, Russo P, Burt ME, Brady MS. Resection of metastatic renal cell carcinoma. J Clin Oncol. 1998;16:2261–6. https://doi.org/10.1200/JCO.1998.16.6.2261.
Tang C, Msaouel P, Hara K, Choi H, Le V, Shah AY, et al. Definitive radiotherapy in lieu of systemic therapy for oligometastatic renal cell carcinoma: a single-arm, single-centre, feasibility, phase 2 trial. Lancet Oncol. 2021;22:1732–9. https://doi.org/10.1016/S1470-2045(21)00528-3.
Roberto M, Botticelli A, Panebianco M, Aschelter AM, Gelibter A, Ciccarese C, et al. Metastatic Renal Cell Carcinoma Management: From Molecular Mechanism to Clinical Practice. Front Oncol. 2021;11:657639. https://doi.org/10.3389/fonc.2021.657639.
Brufau BP, Cerqueda CS, Villalba LB, Izquierdo RS, González BM, Molina CN. Metastatic renal cell carcinoma: radiologic findings and assessment of response to targeted antiangiogenic therapy by using multidetector CT. Radiographics. 2013;33:1691–716. https://doi.org/10.1148/rg.336125110.
Roberts MJ, Maurer T, Perera M, Eiber M, Hope TA, Ost P, et al. Using PSMA imaging for prognostication in localized and advanced prostate cancer. Nat Rev Urol. 2023;20:23–47. https://doi.org/10.1038/s41585-022-00670-6.
Baccala A, Sercia L, Li J, Heston W, Zhou M. Expression of prostate-specific membrane antigen in tumor-associated neovasculature of renal neoplasms. Urology. 2007;70:385–90. https://doi.org/10.1016/j.urology.2007.03.025.
Tariq A, McCart Reed AE, Morton A, Porten S, Vela I, Williams ED, et al. Urothelial Carcinoma and Prostate-specific Membrane Antigen: Cellular, Imaging, and Prognostic Implications. Eur Urol Focus. 2022;8:1256–69. https://doi.org/10.1016/j.euf.2021.07.016.
Rhee H, Blazak J, Tham CM, Ng KL, Shepherd B, Lawson M, et al. Pilot study: use of gallium-68 PSMA PET for detection of metastatic lesions in patients with renal tumour. EJNMMI Res. 2016;6:76. https://doi.org/10.1186/s13550-016-0231-6.
Tariq A, Raman V, Alexander H, Roberts MJ, Thomas P. Pituitary Metastasis of Renal Cell Carcinoma Characterized by 18F-Prostate-Specific Membrane Antigen-1007 PET/CT. Clin Nucl Med. 2022;47:e346–e7. https://doi.org/10.1097/rlu.0000000000004055.
Heng DY, Xie W, Regan MM, Harshman LC, Bjarnason GA, Vaishampayan UN, et al. External validation and comparison with other models of the International Metastatic Renal-Cell Carcinoma Database Consortium prognostic model: a population-based study. Lancet Oncol. 2013;14:141–8. https://doi.org/10.1016/S1470-2045(12)70559-4.
Udovicich C, Callahan J, Bressel M, Ong WL, Perera M, Tran B, et al. Impact of Prostate-specific Membrane Antigen Positron Emission Tomography/Computed Tomography in Management of Oligometastatic Renal Cell Carcinoma. European Association of Urology. 2022;44:60–8.
Meyer AR, Carducci MA, Denmeade SR, Markowski MC, Pomper MG, Pierorazio PM, et al. Improved identification of patients with oligometastatic clear cell renal cell carcinoma with PSMA-targeted. Ann Nucl Med. 2019;33:617–23. https://doi.org/10.1007/s12149-019-01371-8.
Rowe SP, Gorin MA, Hammers HJ, Som Javadi M, Hawasli H, Szabo Z, et al. Imaging of metastatic clear cell renal cell carcinoma with PSMA-targeted 18F-DCFPyL PET/CT. Ann Nucl Med. 2015;29:877–82. https://doi.org/10.1007/s12149-015-1017-z.
Tariq A, Kwok M, Pearce A, Rhee H, Kyle S, Marsh P, et al. The role of dual tracer PSMA and FDG PET/CT in renal cell carcinoma (RCC) compared to conventional imaging: A multi-institutional case series with intra-individual comparison. Urol Oncol. 2022;40:66.e1–9. https://doi.org/10.1016/j.urolonc.2021.11.006.
Tariq A, McGeorge S, Pearce A, Rhee H, Wood S, Kyle S, et al. Characterization of tumor thrombus in renal cell carcinoma with prostate specific membrane antigen (PSMA) positron emission tomography (PET)/computed tomography (CT). Urol Oncol. 2022;40:276.e1–9. https://doi.org/10.1016/j.urolonc.2022.03.007.
Mittlmeier LM, Unterrainer M, Rodler S, Todica A, Albert NL, Burgard C, et al. F-PSMA-1007 PET/CT for response assessment in patients with metastatic renal cell carcinoma undergoing tyrosine kinase or checkpoint inhibitor therapy: preliminary results. Eur J Nucl Med Mol Imaging. 2021;48:2031–7. https://doi.org/10.1007/s00259-020-05165-3.
Pattison DA, Debowski M, Gulhane B, Arnfield EG, Pelecanos AM, Garcia PL, et al. Prospective intra-individual blinded comparison of [(18)F]PSMA-1007 and [(68) Ga]Ga-PSMA-11 PET/CT imaging in patients with confirmed prostate cancer. Eur J Nucl Med Mol Imaging. 2022;49:763–76. https://doi.org/10.1007/s00259-021-05520-y.
Funding
Open Access funding enabled and organized by CAUL and its Member Institutions The authors declare that no funds, grants, or other support were received during the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by Shivanshan Pathmanathan, Arsalan Tariq, Chun Gan and Matthew Roberts. The first draft of the manuscript was written by Shivanshan Pathmanathan, Arsalan Tariq, Chun Gan and Matthew Roberts and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors have no relevant financial or non-financial interests to disclose.
Ethics Approval
This study received ethics and governance approval (Reference numbers HREC/2020/QRBWH/64912; UCH 2020.19.329).
Consent to Participate
This was a retrospective study and granted ethics approval.
Consent to Publish
This was a retrospective study, with no patient identifying features, and granted ethics approval for publication. The authors affirm that there no identifying features for publications of images in Fig. 3 requiring consent to publish.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
ESM 1
(DOCX 156 kb)
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Pathmanathan, S., Tariq, A., Pearce, A. et al. Clinical impact of Prostate-Specific Membrane Antigen Positron Emission Tomography (PET) on intensification or deintensification of advanced renal cell carcinoma management. Eur J Nucl Med Mol Imaging 51, 295–303 (2023). https://doi.org/10.1007/s00259-023-06380-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-023-06380-4