Abstract
Osteoarthritis (OA) is one of the most common causes of musculoskeletal disability in the world. Traditionally, it has been thought that obesity contributes to the development and progression of OA by increased mechanical load of the joint structures. Nevertheless, studies have shown that adipose tissue-derived cytokines (adipocytokines) are a possible link between obesity and OA. Furthermore, according to recent findings, not only articular cartilage may be the main target of these cytokines but also the synovial membrane, subchondral bone and infrapatellar fat pad may be encompassed in the process of degradation. This review presents the most recent reports on the contribution of adipocytokines to the knee joint cartilage degradation, osteophyte formation, infrapatellar fat pad alterations and synovitis.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Osteoarthritis (OA) is a progressive and disabling disease, one of the most common musculoskeletal disorders. OA originates as a result of the action of risk factors such as genetics, old age, female gender, obesity and injury [1, 2]. The disease is characterized by the progressive loss of articular cartilage with concomitant joint space narrowing, osteophyte formation, subchondral sclerosis and synovitis. An important feature of OA is the extension of the spectrum of damage from the articular cartilage to the entire joint structures [3]. The pathological processes taking place in cartilage, bone and synovial membrane result in pain, loss of function and in consequence disability. It has been estimated that in the USA symptomatic knee OA occurs in 10 % of men and 13 % of women aged 60 years or older and it is likely to increase because of the aging of the population and the increasing numbers of obese persons [4].
Although numerous risk factors contribute to the process of joint degradation, over the past ten years the association of obesity and OA has been well documented [5, 6]. One of the most important problems addressed by this intensive research until now is whether the impact of obesity on the development of OA is solely caused by an increased mechanical load in obese individuals, or rather, whether this influence is due to specific biochemical processes activated by, or directly related to the endocrine function of adipose tissue [7, 8]. The white adipose tissue (WAT) has the potential to produce various substances that have some functional similarity to classic endocrine hormones, although they differ from them in many aspects. Those substances were named adipocytokines, and in line with the latter hypothesis, recent investigations reveal a role played by adipocytokines in the degradation of cartilage, synovium and bone [9, 10].
This review summarizes the most recent reports on the role of adipocytokines in the pathogenesis of knee joint OA.
Adipocytokines
Adipocytokines are bioactive proteins secreted mainly by WAT, first identified in the early 1990s. So far, the role of adipocytokines in the metabolism of bone tissue and its contribution to the differentiation of bone marrow cells to osteoblasts [11, 12], as well as an important role in the pathogenesis of metabolic syndrome have been demonstrated [13, 14]. Some adipocytokines might also play a direct role in maintaining an inflammatory state in the joints of obese individuals (the so-called low-grade inflammatory state) [15]. These proteins have been detected in synovial fluid (SF) and the plasma of patients with OA [16–18]. A putative source of adipocytokine secretion in the region of the knee joint is the infrapatellar fat pad (IPFP) [19, 20]. In addition, recent findings indicate that deteriorated articular cartilage is another source of adipocytokines within the joint [21]. Furthermore, adipocytokines originating from the adipose tissue, which acts as an endocrine organ, can directly initiate the abnormalities in structure and function of articular cartilage in obese individuals [14]. This “external” action is then added to the action of adipocytokines produced inside the joint, which further deteriorates the condition of the joint. Of numerous adipocytokines, the best characterized are leptin, resistin, adiponectin, vaspin, omentin, visfatin and adipsin [22].
Leptin
From all adipocytokines, leptin was discovered first and is best characterized [23]. It is a polypeptide of 146 amino acids, and M r 16 kDa, encoded by the obese gene (ob) localized on chromosome 7q31.3. Leptin is a multi-functional protein. It has s metabolic, neuroendocrine, and immunomodulatory effects. The activity of leptin is mediated by the Ob receptor (ObR) encoded by the diabetes gene (db). Leptin is produced mainly in the adipocytes of subcutaneous WAT and in the glandular cells of the stomach, in the liver and in the endothelial cells of the mammary gland [24–26]. The concentration of leptin fluctuates the highest being during the night and the lowest during the day. In healthy individuals, the plasma concentration of leptin ranges from 5 to 15 μg/L [27]. The concentration of leptin is higher in women, which is connected with larger stores of subcutaneous adipose tissue [28]. There are significantly higher ratios of plasma leptin concentrations to fat mass in girls than boys at the late stage of puberty. However, there are no significant differences in leptin concentrations in prepubertal and early pubertal children [29].
Leptin is secreted in a free form or is bound to its receptor. The bound form regulates mainly the energy balance. Leptin plays the role of a “guardian” of adipose tissue stores in the body. As a result of direct action on the central nervous system, it restrains hunger evoking the feeling of satiety and regulating thermogenesis as well as increasing energy expenditure [30]. Acting similarly to insulin, leptin lowers blood glucose level [24, 30]. It was also suggested that leptin acts as an acute phase protein having pro-inflammatory action through T-lymphocyte stimulation resulting in the liberation of cytokines such as interleukins (ILs) and tumor necrosis factor-α (TNF-α) and increasing the activation level of NK cells, macrophages and neutrophils [31].
Resistin
Resistin is a polypeptide of 105 amino acids and M r 12 kDa encoded by the RSTN gene localized on chromosome 19p13.3 [32]. It is produced mainly in WAT and also in bone marrow, the spleen, lungs, placentas and the pancreas. Resistin takes part in adipogenesis, insulin resistance and inflammatory processes and exhibits a positive correlation with the occurrence of obesity, insulin resistance, and chronic inflammation [33]. It induces the secretion of pro-inflammatory cytokines such as IL-1, IL-6, IL-12 and TNF-α in a NF-κB-mediated fashion [34, 35]. Resistin elevates the concentrations of TNFα, IL-1β and IL-6 in plasma and increases catabolism and proteoglycan levels after experimental joint injury [36]. It has also been demonstrated that resistin up-regulates the intercellular adhesion molecule-1 (ICAM1), vascular cell-adhesion molecule-1 (VCAM1) and chemokine ligand 2 (CCL2) [37].
Adiponectin
Adiponectin is a polypeptide of 244 amino acids, but it spontaneously polymerizes to form trimers, hexamers and dodecamers. Adiponectin is encoded by the ADPOQ gene, which is localized on chromosome 3q27. It is produced by the majority of WAT cells and to a lesser extent by visceral adipose tissue (VAT). It takes part in the metabolism of carbohydrates and lipids, and plays an important role in thermoregulation by combusting ectopic adipose tissue. It also increases the insulin sensitivity of muscle and liver tissues. The plasma concentration of adiponectin reveals sexual dimorphism with females having higher levels than males. It ranges from 500 to 30,000 μg/L [24] and increases in degenerative joint disease and rheumatoid arthritis [15]. Adiponectin binds to a number of G protein-coupled receptors and the expression level of these receptors is elevated in articular cartilage, which is affected by degeneration [38]. The role of adiponectin played in the inflammatory process is not completely elucidated. It was suggested that it affects IL-1β, IL-6, MMP-13 [39], MMP-1, MMP-3, NO [38], and MMP-9 secretion [40].
Visfatin
Visfatin is a protein of M r 52 kDa, a nicotinamide phosphoribosyltransferase enzyme, which acts as a dimer. Visfatin, first detected in skeletal muscles, liver and bone marrow, is encoded by the PBEF1 gene [41]. It is secreted by visceral fat tissue [42] and is implicated in a number of conditions, such as aging, atherosclerosis, pathogenesis of type 2 diabetes and rheumatoid arthritis [43]. Its role in metabolic processes is still controversial, but it has been reported that the properties of visfatin are similar to insulin. It has been suggested that the levels of visfatin are higher in obese [44, 45] and in OA patients [46]. In an experimental study, it has been found that visfatin level could be down-regulated by the overexpression of miR34a, which binds directly to the 3′UTR of visfatin mRNA. In obese mice, adipocytokine level correlated inversely with the elevated levels of miR-34a [47].
Other adipocytokines
Omentin is also known as an intestinal lactoferrin receptor. It is a 313-amino-acid protein encoded by the ITLN1 gene, which is secreted by the visceral stromal vascular cells of the abdominal adipose tissue (but not by adipocytes), and is also synthesized in small intestine, lungs and heart muscles [48, 49]. Omentin causes vasodilation and attenuates C-reactive protein-induced angiogenesis via the nuclear factor kappa B signaling pathway. Although its function is not completely elucidated, it is known that it potentiates the effect of insulin on glucose metabolism [48] and its gene expression is altered in inflammation and in obese patients [50].
Vaspin (visceral adipose tissue-derived serine protease inhibitor) is a protein of 395 amino acids, with a M r of 45.2 kDa, encoded by the Serpina12 gene. Its expression in adipose tissue is regulated in a fat depot-specific manner and is associated with obesity, insulin resistance, and abnormal glucose metabolism. It is produced by visceral tissue adipocytes, shares about 40 % sequence homology with α1-antitrypsin, and its secretion alters glucose tolerance and insulin sensitivity [51]. These findings indicate that vaspin exerts an insulin-sensitizing effect targeted toward WAT in states of obesity. Vaspin structure is similar to adiponectin and it acts as an anti-inflammatory agent, suppressing TNFα, leptin and resistin secretion [52, 53].
Adipsin (complement factor D) is a protein encoded by the CFD gene. It affects both the lipid and glucose metabolism. It is a serine protease that stimulates glucose transport for triglyceride accumulation in fats cells and inhibits lipolysis. It has been initially indicated that adipsin level correlates with obesity, dyslipidemia, insulin resistance and cardiovascular diseases [54].
Adipocytokines in cartilage degradation
A crucial feature of OA is the progressive loss of articular cartilage. Cartilage degradation is a result of the increased expression of genes encoding proteolytic enzymes such as metalloproteinases (MMPs) and aggrecanases (AGGs), as well as inflammatory cytokines, nitric oxide (NO) and prostaglandins (PGEs). The secretion of enzymes involved in the degradation of cartilage accompanied by insufficient cartilage repair result in the breakdown of homeostasis and a shift toward catabolic processes. Adipocytokines acting in an autocrine or paracrine manner stimulate chondrocytes in a dual way. One of the first studies on the impact of leptin on cartilage cells revealed that this peptide increases the anabolic activity of chondrocytes by inducing IGF-1 and TGF-β expression at both mRNA and protein levels [55]. However, further studies have shown contradictory results. Leptin was reported to increase MMP-2, MMP-9, cathepsin D and type II collagen expression in vivo, at both mRNA and protein levels. This peptide also up-regulated ADAMTS-4 and ADAMTS-5 expression, causing proteoglycan depletion from the articular cartilage of rats [46]. The latest data suggest that leptin induces ADAMTS-4, ADAMTS-5, and ADAMTS-9 gene expression by mitogen-activated protein kinases and NF-ĸB signaling pathways in human chondrocytes, which results in the subsequent increase of inflammatory processes [56]. Vuolteenaho et al. [57] showed that leptin enhanced NO synthesis and PGE2, IL-6 and IL-8 secretion in OA cartilage. Berry et al. [58] found that the presence of the soluble leptin receptor (sOB-Rb) is associated with reduced cartilage synthesis and increased levels of the tissue degradation marker (N-terminal type IIA procollagen propeptide, PIIANP).
Similarly, adiponectin has both a pro- and anti-inflammatory effect on tissues. Acting in vitro, this peptide induced iNOS activity and IL-6, MMP-3, MMP-9 and MCP-1 (monocyte chemattractant protein-1) expression [40]. In the study of Francin et al. [59] the adiponectin transcript level was significantly correlated with those encoding prostaglandin E2 and MMP-13. By contrast, Chen et al. [60] showed the protective effect of adiponectin on articular cartilage by up-regulating the tissue metallo-proteinase inhibitor (TIMP-2) and decreasing IL-1β-mediated MMP-13 expression. In patients with severe OA, higher plasma adiponectin levels were found and their chondrocytes expressed both adiponectin receptors (AdipoR1 and AdipoR2) and AdipoR1 was mainly expressed in the superficial layers of OA cartilage [40]. Visfatin had a catabolic effect on articular cartilage. Produced by OA chondrocytes it increased MMP-3, MMP-13 as well as ADAMTS-4 and ADAMTS-5 activity in a mouse model [61]. Yammani and Loeser [62] demonstrated that, in human cartilage, visfatin inhibited IGF-1 and led to IGF-1-mediated proteoglycan synthesis. Moreover, Hong et al. [63] suggested that visfatin could alter the expression, of chondrogenic factors such as the sex-determining region Y-box 9 (SOX-9) and type II collagen. In OA patients visfatin SF level positively correlated with the degradation markers of collagen, (CTX-II) and aggrecans (AGG1, AGG2) [64]. Recent studies show that most articular visfatin derives from synovium, and the activity of visfatin is involved in chondrocyte and osteoblast activation, so targeting this enzymatic activity to disrupt joint tissue interactions may be novel in OA therapy [65].
Adipocytokines in osteophyte formation
Osteophyte formation is one of the hallmarks of OA. It has been suggested that osteophytes result from the abnormal healing response of subchondral trabeculae, or from the blood vessels penetration into the degrading cartilage [66]. It has been also shown that in OA the patient’s osteophytes express TGF-β, which induces osteophyte formation in an experimental model [67, 68]. Thomas et al. [11] suggested that the action of leptin on bone tissue may be a result of bone marrow stromal cell stimulation and enhanced cell differentiation into osteoblasts with limited differentiation into adipocytes. In the study of Berry et al. [58], high leptin levels were associated with increased bone formation markers, such as osteocalcin and PINP (N-terminal type I procollagen propeptide). However, no associations were found between adipocytokine level and bone resorption markers (CTX-I, NTX-I and ICTP). Another study revealed that in subchondral bone osteoblasts of OA patients leptin expression levels were about fivefold than in normal osteoblasts and protein level was approximately twofold in OA cells compared to normal [69]. In the magnetic resonance imaging (MRI)-assessed knee joints, leptin levels were also strongly associated with osteophyte formation. Higher adipocytokine levels were found among women with larger osteophytes (>10 mm) than in those with no or smaller osteophytes (<10 mm) [70]. Visfatin has been also found to influence osteoblasts proliferation and type II collagen production [71].
Adipocytokines secretion by infrapatellar fat pad
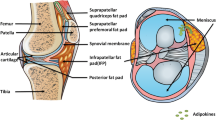
The infrapatellar fat pad (IPFP), situated within the knee joint capsule, contains in addition to adipocytes, macrophages, lymphocytes and granulocytes, and may serve as a local source of adipose tissue-derived cytokines [72]. Several studies demonstrated that IPFP-derived adipocytokines could have a direct impact on articular cartilage. When the subcutaneous fat tissue (SCFT) and fat pad tissue (FPT)-derived cells were compared, IPFP adipocytes showed a twofold increase in IL-6 gene expression and in IL-6 release. Interestingly, leptin secretion was 40 % lower and adiponectin was increased by 70 % in IPFP cells compared with the subcutaneous adipose tissue [73]. Moreover, it was demonstrated that the culture media from OA patients’ IPFP adipocytes induced MMP13 and MMP1 expression in articular chondrocytes and that the leptin level positively correlated with the expression of both MMPs and cartilage collagen destruction [20]. The end-stage OA patients’ fat pad cells also showed an increased expression of inflammatory cytokines and down-regulation of anabolic peptides, such as VCAM1 (vascular cell adhesion molecule-1), CTGF (connective tissue growth factor) and CD44 (cluster of differentiation) [74].
Adipocytokines and synovitis
Changes of the synovial membrane in the course of OA include synovial hyperthrophy and hyperplasia, sometimes accompanied by tissue thickening. It is believed that a low-grade inflammatory state induced by synovial membrane may enhance IL-1β and TNF-α expression, which contributes to cartilage degradation [75]. Adipocytokines can be detected locally both in the synovium and synovial fluid (SF) of OA individuals. Nevertheless, the exact role of these peptides in inducing synovitis is unknown. Several studies provide information regarding this issue. De Boer et al. [9] have found that serum leptin, resistin and adiponectin levels were significantly correlated with the inflammation of local synovial tissue, but no association was found between adipocytokine level and cartilage damage markers as well as GAGs content. However, Hao et al. [76] demonstrated that the SF adiponectin level correlated with aggrecan depletion markers (AGG1 and AGG2), but not with the type II collagen degradation marker (CTX-II). By contrast, Honsawek et al. [77] found that SF adiponectin level was lower in severe OA and negatively correlated with OA grade and BMI. Correspondingly, the SF/plasma leptin ratio has been found to be significantly lower in the end-stage OA compared to early stages of the disease [78]. More recently, a study by Bas et al. [79] revealed that low SF adiponectin level and high SF leptin level were associated with worse pre-operative pain scores in the knees of OA patients.
Conclusions
Adipocytokines participate in numerous metabolic and inflammatory processes in the body. The currently published data suggest their pivotal role in the course of OA. It has been shown that in the knee joint the source of cytokines is not only systemic but also local, since cartilage, synovium or infrapatellar fat could be the source itself. Therefore, the role of obesity as a risk factor and cause of OA may not be limited to biomechanical loading, but also associated with adipose tissue secretory activity. Given the epidemic of obesity and OA, it is clear that these two conditions cannot be ignored and a modifiable risk factor, such as being overweight, should be targeted in order to reduce knee OA incidence.
References
Garner M, Alshameeri Z, Khanduja V (2013) Osteoarthritis: genes, nature-nurture interaction and the role of leptin. Int Orthop 37:2499–2505. doi:10.1007/s00264-013-2088-x
Blagojevic M, Jinks C, Jeffery A, Jordan KP (2010) Risk factors for onset of osteoarthritis in older adults: a systematic review and meta-analysis. Osteoarthr Cartil 18:24–33. doi:10.1016/j.joca.2009.08.010
Houard X, Goldring MB, Berenbaum F (2013) Homeostatic mechanisms in articular cartilage and role of inflammation in osteoarthritis. Curr Rhematol Rep 15:375. doi:10.1007/s11926-013-0375-6
Zhang Y, Jordan JM (2010) Epidemiology of osteoarthritis. Clin Geriatr Med 26:355–369. doi:10.1016/j.cger.2010.03.001
Pottie P, Presle N, Terlain B, Netter P, Mainard D, Berenbaum F (2006) Obesity and osteoarthritis: more complex than predicted. Ann Rheum Dis 65:1403e5. doi:10.1136/ard.2006.061994
Grotle M, Hagen KB, Natvig B, Dahl FA, Kvien TK (2008) Obesity and osteoarthritis in knee, hip and/or hand: an epidemiological study in the general population with 10 years follow-up. BMC Musculoskelet Disord 9:132. doi:10.1186/1471-2474-9-132
Berenbaum F (2013) Osteoarthritis as an inflammatory disease (osteoarthritis is not osteoarthrosis!). Osteoarthr Cartil 21:16–21. doi:10.1016/j.joca.2012.11.012
Felson DT (2013) Osteoarthritis as a disease of mechanics. Osteoarthritis Cartilage 21:10–15. doi:10.1016/j.joca.2012.09.012
De Boer TN, van Spil WE, Huisman AM, Polak AA, Bijlsma JWJ, Lafeber FPJG, Mastbergen SC (2012) Serum adipokines in osteoarthritis; comparison with controls and relationship with local parameters of synovial inflammation and cartilage damage. Osteoarthr Cartil 20:846–853. doi:10.1016/j.joca.2012.05.002
Migliaccio S, Greco EA, Wannenes F, Donini LM, Lenzi A (2014) Adipose, bone and muscle tissues as new endocrine organs: role of reciprocal regulation for osteoporosis and obesity development. Horm Mol Biol Clin Invest 17:39–51. doi:10.1515/hmbci-2013-0070
Thomas T, Gori F, Khosla S, Jensen MD, Burguera B, Riggs BL (1999) Leptin acts on human marrow stromal cells to enhance differentiation to osteoblasts and to inhibit differentiation to adipocytes. Endocrinology 140:1630–1638. doi:10.1210/endo.140.4.6637
Cornish J, Callon KE, Bava U, Lin C, Naot D, Hill BL, Grey AB, Broom N, Myers DE, Nicholson GC, Reid IR (2002) Leptin directly regulates bone cell function in vitro and reduces bone fragility in vivo. J Endocrinol 175:405–415
Matsuzawa Y (2006) Therapy insight: adipocytokines in metabolic syndrome and related cardiovascular disease. Nat Clin Pract Cardiovasc Med 3(1):35–42. doi:10.1038/ncpcardio0380
Abella V, Scotece M, Conde J, Lopez V, Lazzaro V, Pino J, Gomez-Reino JJ, Gualillo O (2014) Adipokines, metabolic syndrome and rheumatic diseases. J Immunol Res ID 343746. doi:10.1155/2014/343746
Scotece M, Conde J, Gómez R, López V, Lago F, Gómez-Reino JJ (2011) Beyond fat mass: exploring the role of adipokines in rheumatic disease. Sci World J 11:1932–1947. doi:10.1100/2011/290142
Hu PF, Tang JL, Chen WP, Bao JP, Wu LD (2011) Increased apelin serum levels and expression in human chondrocytes in osteoarthritis patients. Int Orthop 35:1421–1426. doi:10.1007/s00264-010-1100-y
Lübbeke A, Finckh A, Puskas GJ, Suva D, Lädermann A, Bas S, Fritschy D, Gabay C, Hoffmeyer P (2013) Do synovial leptin levels correlate with pain in end stage arthritis? Int Orthop 37:2071–2079. doi:10.1007/s00264-013-1982-6
Vuolteenaho K, Koskinen A, Moilanen E (2014) Leptin- a link between obesity and osteoarthritis. Applications for prevention and treatment. Basic Clin Pharmacol Toxicol:103–108. doi:10.1111/bcpt.12160
Clockaerts S, Bastiaansen-Jenniskens YM, Runhaar J, Van Osch GJVM, Van Offel JF, Verhaar JAN, De Clerck LS, Somville J (2010) The infrapatellar fat pad should be considered as an active osteoarthritic joint tissue: a narrative review. Osteoatrhr Cartil 18:876–882. doi:10.1016/j.joca.2010.03.014
Hui W, Litherland J, Elias MS, Kitson GI, Cawston TE, Rowan AD, Young DA (2012) Leptin produced by joint white adipose tissue induces cartilage degradation via upregulation and activation of matrix metalloproteinases. Ann Rheum Dis 71:455–462. doi:10.1136/annrheumdis-2011-200372
Koskinen A, Juslin S, Nieminen R, Moilanen T, Vuolteenaho K, Moilanen E (2011) Adiponectin associates with markers of cartilage degradation in osteoarthritis and induces production of proinflammatory and catabolic factors through mitogen-activated protein kinase pathways. Arthr Res Ther 13:R184. doi:10.1186/ar3512
Presle N, Pottie P, Dumond H, Guillame C, Lapicque F, Pallu S, Mainard D, Netter P, Terlain B (2006) Differential distribution of adipokines between serum and synovial fluid in patients with osteoarthritis. Contribution of joint tisues to their articular production. Osteoarthr Cartil 14:690–695. doi:10.1016/j.joca.2006.01.009
Zhang Y, Proenca R, Maffei M, Barone M, Loepold L, Friedman JM (1994) Positional cloning of the mouse obese gene and its human homologue. Nature 372:425–432. doi:10.1038/372425a0
Meier U, Gressner AM (2004) Endocrine regulation of energy metabolism: review of pathobiochemical and clinical chemical aspects of leptin, Ghrelin, adiponectin and resistin. Clin Chem 50:1511–1525. doi:10.1373/clinchem.2004.032482
Koerner A, Kratzsch J, Kiess W (2005) Adipocytokines: leptin- the classical, resistin- the controversical, adiponectin- the promising, and more to come. Best Prac Res Clin Endocrinol Metab 19(4):525–546. doi:10.1016/j.beem.2005.07.008
Rasouli N, Kern FA (2008) Adipocytokines and the metabolic complications of obesity. J Clin Endocrinol Metab 93:64–73. doi:10.1210/jc.2008-1613
Jasińska A, Pietruczuk M (2010) Adipocytokiny białka o wielokierunkowym działaniu. J Lab Diagn 46(3):331–338
Saad MF, Damani S, Gingerich RL, Riad-Gabriel MG, Khan A, Boyadjian R, Jinagouda SD, el-Tawil K, Rude RK, Kamdar V (1997) Sexual dimorphism in plasma leptin concentration. J Clin Endocrinol Metab 82(2):579–584
Horlick MB, Rosenbaum M, Nicolson M, Levine LS, Fedun B, Wang J, Pierson RN Jr, Leibel RL (2000) Effect of puberty on the relationship between circulating leptin and body composition. J Clin Endocrinol Metab 85(7):2509–2518
Ashima RS, Lazar MA (2008) Adipocytokines and the peripheral and neutral control of energy balance. Mol Endocrinol 22(5):1023–1031. doi:10.1210/me.2007-0529
Fernández-Riejos P, Najib S, Santos-Alvarez J, Martín-Romero C, Pérez-Pérez A, González-Yanes C, Sanchez-Margalet V (2010) Role of leptin in the activation of immune cells. Mediators Inflamm 568343. doi:10.1155/2010/568343
Yang RZ, Huang Q, Xu A, McLenithan JC, Eisen JA, Shuldiner AR, Alkan S, Gong DW (2003) Comparative studies of resistin expression and phylogenomics in human and mouse. Biochem Biophys Res Commun 310(3):927–935. doi:10.1016/j.bbrc.2003.09.093
Wulster-Radcliffe MC, Ajuwon KM, Wang J, Christian JA, Spurlock ME (2004) Adiponectin differentially regulates cytokines in porcine macrophages. Biochem Biophys Res Commun 316(3):924–929. doi:10.1016/j.bbrc.2004.02.130
Milan G, Granzotto M, Scarda A, Calcagno A, Pagano C, Federspil G, Vettor R (2002) Resistin and adiponectin expression in visceral fat of obese rats: effect of weight loss. Obes Res 10(11):1095–1103
Silswal N, Singh AK, Aruna B, Mukhopadhyay S, Ghosh S, Ehtesham NZ (2005) Human resistin stimulates the pro-inflammatory cytokines TNF-alpha and IL-12 in macrophages by NF-kappaB-dependent pathway. Biochem Biophys Res Commun 334(4):1092–1101. doi:10.1016/j.bbrc.2005.06.202
Bokarewa M, Nagaey I, Dahlberg L, Smith U, Tarkowski A (2005) Resistin, an adipokine with potent proinflammatory properties. J Immunol 174(9):5789–5795. doi:10.4049/ jimmunol.174.9.5789
Verma S, Li SH, Wang CH, Fedak PW, Li RK, Weisel RD, Mickle DA (2003) Resistin promotes endothelial cell activation: further evidence of adipokine-endothelial interaction. Circulation 108(6):736–740. doi:10.1161/01.CIR.0000084503.91330.49
Kang EH, Lee YL, Kim TK, Chang CB, Chung J-H, Shin K, Lee EY, Lee EB, Song YW (2010) Adiponectin is a potential catabolic mediator in osteoarthritis cartilage. Arthr Res Ther 12(6):R231. doi:10.1186/ar3218
Hu P, Bao J, Wu L (2011) The emerging role of adipokines in osteoarthritis: a narrative review. Mol Biol Rep 38(2):873–878. doi:10.1007/s11033-010-0179-y
Lago R, Gomez R, Otero M, Lago F, Gallego R, Dieguez C, Gomez-Reino JJ, Gualillo O (2008) A new player in cartilage homeostasis: adiponectin induces nitric oxide synthase type II and pro-inflammatory cytokines in chondrocytes. Osteoarthr Cartil 16(9):1101–1109. doi:10.1016/j.joca.2007.12.008
Skoczylas A (2009) The role of visfatin in the pathophysiology of human. Wiad Lek 62(3):190–196
Samal B, Sun Y, Stearns G, Xie C, Suggs S, McNiece I (1994) Cloning and characterization of the cDNA encoding a novel human pre-B-cell colony-enhancing factor. Mol Cell Biol 14(2):1431–1437. doi:10.1128/MCB.14.2.1431
Versini M, Jeandel PY, Rosenthal E, Shoenfeld Y (2014) Obesity in autoimmune diseases: not a passive bystander. Autoimmun Rev 13(9):981–1000. doi:10.1016/j.autrev.2014.07.001
Friebe D, Neef M, Kratzsch J, Erbs S, Dittrich K, Garten A, Petzold-Quinque S, Bluhler S, Reinehr T, Stumvoll M, Bluher M, Kiess W, Korner A (2011) Leucocytes are a major source of circulating nicotinamide phosphoribosyltransferase (NAMPT)/pre-B cell colony (PBEF)/visfatin linking obesity and inflammation in humans. Diabetologia 54(5):1200–1211. doi:10.1007/s00125-010-2042-z
Catalan V, Gomez-Ambrosi J, Rodriguez A, Ramirez B, Silva C, Rotellar F, Cienfuegos JA, Fruhbeck G (2011) Association of increased Visfatin/PBEF/NAMPT circulating concentrations and gene expression levels in peripheral blood cells with lipid metabolism and fatty liver in human morbid obesity. Nutr Metab Cardiovasc Dis 21(4):245–253. doi:10.1016/j.numecd.2009.09.008
Bao JP, Chen WP, Feng J, Hu PF, Shi ZL, Wu LD (2010) Leptin plays a catabolic role on articular cartilage. Mol Biol Rep 37(7):3265–3272. doi:10.1007/s11033-009-9911-x
Choi S-E, Fu T, Seok S, Kim D-H, Yu E, Lee K-W, Kang Y, Li X, Kemper JK (2013) Elevated microRNA-34a in obesity reduces NAD+ levels and SIRT1 activity by directly targeting NAMPT. Aging Cell 12(6):1062–1072. doi:10.1111/acel.12135
Schäffler A, Neumeier M, Herfarth H, Fürst A, Schölmerich J, Büchler C (2005) Genomic structure of human omentin, a new adipocytokine expressed in omental adipose tissue. Biochim Biophys Acta 1732(1–3):96–102. doi:10.1016/j.bbaexp.2005.11.005
Yang RZ, Lee MJ, Hu H, Pray J, Wu HB, Hansen BC, Shuldiner AR, Sk F, McLenithan JC, Gong DW (2006) Identification of omentin as a novel depot- specific adipokine in human adipose tissue: possible role in modulating insulin action. Am J Physiol Endocrinol Metab 290(6):E1253–E1261. doi:10.1152/ajpendo.00572.2004
De Souza Batista CM, Yang RZ, Lee MJ, Glynn NM, Yu DZ, Pray J, Ndubuizu K, Patil S, Schwartz A, Kligman M, Fried SK, Gong DW, Shuldiner AR, Pollin TI, McLenithan JC (2007) Omentin plasma levels and gene expression are decreased in obesity. Diabetes 56(6):1655–1661. doi:10.2337/db06-1506
Hida K, Wada J, Eguchi J, Zhang H, Baba M, Seida A et al (2005) Visceral adipose tissue-derived serine protease inhibitor: a unique insulin-sensitizing adipocytokine in obesity. PNAS 102(30):10610–10615. doi:10.1073/pnas.0504703102
Fantuzzi G (2005) Adipose tissue, adipokines, and inflammation. J Allergy Clin Immunol 115(5):911–919. doi:10.1016/j.jaci.2005.02.023
Tilg H, Moschen AR (2006) Adipocytokines: mediators linking adipose tissue, inflammation and immunity. Nat Rev Immunol 6(10):772–783. doi:10.1038/nri1937
Cianflone K, Xia Z, Chen LY (2003) Critical review of acylation-stimulating protein physiology in humans and rodents. Biochim Biophys Acta 1609(2):127–143. doi:10.1016/S0005-2736(02)00686-7
Dumond H, Presle N, Terlain B, Mainard D, Loeuille D, Netter P, Pottie P (2003) Evidence for a key role of leptin in osteoarthritis. Arthritis Rheum 48(11):3118–3129. doi:10.1002/art.11303
Yaykasli KO, Hatipoglu OF, Yaykasli E, Yildirim K, Kaya E, Ozsahin M, Uslu M, Gunduz E (2014) Leptin induces ADAMTS-4, ADAMTS-5, and ADAMTS-9 genes expression by mitogen-activated protein kinases and NF-ĸB signaling pathways in human chondrocytes. Cell Biol Int. doi:10.1002/cbin.10336
Vuolteenaho K, Koskinen A, Kukkonen M, Nieminen R, Päivärinta U, Moilanen T, Moilanen E (2009) Leptin enhances synthesis of proinflammatory mediators in human osteoarthritic cartilage—mediator role of NO in leptin-induced PGE2, IL-6, and IL-8 production. Mediators Inflamm 2009:345838. doi:10.1155/2009/345838
Berry PA, Jones SW, Cicuttini FM, Wluka AE, Maciewicz RA (2011) Temporal relationship between serum adipokines, biomarkers of bone and cartilage turnover, and cartilage volume loss in a population with clinical knee osteoarthritis. Arthritis Rheum 63:700–707. doi:10.1002/art.30182
Francin PJ, Abot A, Guillaume C, Moulin D, Bianchi A, Gegout-Pottie P, Jouzeau JY, Mainard D, Presle N (2014) Association between adiponectin and cartilage degradation in human osteoarthritis. Osteoarthr Cartil 22:519–526. doi:10.1016/j.joca.2014.01.002
Chen TH, Chen L, Hsieh MS, Chang CP, Chou DT, Tsai SH (2006) Evidence for a protective role for adiponectin in osteoarthritis. Biochim Biophys Acta 1762:711–718. doi:10.1016/j.bbadis.2006.06.008
Gosset M, Berenbaum F, Salvat C, Sautet A, Pigenet A, Tahiri K, Jacques C (2008) Crucial role of visfatin/pre-B cell colonyenhancing factor in matrix degradation and prostaglandin E2 synthesis in chondrocytes: possible influence on osteoarthritis. Arthritis Rheum 58:1399–1409. doi:10.1002/art.23431
Yammani RR, Loeser RF (2012) Extracellular nicotinamide phosphoribosyltransferase (NAMPT/visfatin) inhibits insulin- like growth factor-1 signaling and proteoglycan synthesis in human articular chondrocytes. Arthritis Res Ther 14:R23. doi:10.1186/ar3705
Hong EH, Yun HS, Kim J, Um HD, Lee KH, Kang CM, Lee SJ, Chun JS, Hwang SG (2011) Nicotinamide phosphoribosyltransferase is essential for interleukin-1beta-mediated dedifferentiation of articular chondrocytes via SIRT1 and extracellular signal-regulated kinase (ERK) complex signaling. J Biol Chem 286:28619–28631. doi:10.1074/jbc.M111.219832
Duan Y, Hao D, Li M, Wu Z, Li D, Yang X, Qiu G (2012) Increased synovial fluid visfatin is positively linked to cartilage degradation biomarkers in osteoarthritis. Rheumatol Int 32:985–990. doi:10.1007/s00296-010-1731-8
Laiguillon MC, Houard X, Bougault C, Gosset M, Nourissat G, Sautet A, Jacques C, Berenbaum F, Sellam J (2014) Expression and function of visfatin (Nampt), an adipokine-enzyme involved in inflammatory pathways of osteoarthritis. Arthritis Res Ther 16:R38. doi:10.1186/ar4467
Gilbertson EM (1975) Development of periarticular osteophytes in experimentally induced osteoarthritis in the dog. A study using microradiographic, microangiographic, and fluorescent bone-labelling techniques. Ann Rheum Dis 34:12–25
Bakker AC, van de Loo FA, van Beuningen HM, Sime P, van Lent PL, van der Kraan PM, Richards CD, van den Berg WB (2001) Overexpression of active TGF-beta-1 in the murine knee joint: evidence for synovial-layer dependent chondro-osteophyte formation. Osteoarthr Cartil 9:128–136. doi:10.1053/joca.2000.0368
Uchino M, Izumi T, Tominaga T, Wakita R, Minehara H, Sekiguchi M, Itoman M (2000) Growth factor expression in the osteophytes of the human femoral head in osteoarthritis. Clin Orthop Relat Res 377:119–125
Mutabaruka MS, Aoulad Aissa M, Delalandre A, Lavigne M, Lajeunesse D (2010) Local leptin production in osteoarthritis subchondral osteoblasts may be responsible for their abnormal phenotypic expression. Arthritis Res Ther 12(1):R20. doi:10.1186/ar2925
Karvonen-Gutierrez CA, Harlow SD, Jacobson J, Mancuso P, Jiang Y (2014) The relationship between longitudinal serum leptin measures and measures of magnetic resonance imaging-assessed knee joint damage in a population of mid-life women. Ann Rheum Dis 73(5):883–889. doi:10.1136/annrheumdis-2012-202685
Xie H, Tang SY, Luo XH, Huang J, Cui RR, Yuan LQ, Zhou HD, Wu XP, Liao EY (2007) Insulin-like effects of visfatin on human osteoblasts. Calcif Tissue Int 80:201–210. doi:10.1007/s00223-006-0155-7
Ioan-Facsinay A, Kloppenburg M (2013) An emerging player in knee osteoarthritis: the infrapatellar fat pad. Arthritis Res Ther 15:225
Distel E, Cadoudal T, Durant S, Poignard A, Chevalier X, Benelli C (2009) The infrapatellar fat pad in knee osteoarthritis: an important source of interleukin-6 and its soluble receptor. Arthritis Rheum 60:3374–3377. doi:10.1002/art.24881
Gandhi R, Takahashi M, Virtanen C, Syed K, Davey JR, Mahomed NN (2011) Microarray analysis of the infrapatellar fat pad in knee osteoarthritis: relationship with joint inflammation. J Rheumatol 38:1966–1972. doi:10.3899/jrheum.101302
Ayral X, Pickering EH, Woodworth TG, Mackillop N, Dougados M (2005) Synovitis: a potential predictive factor of structural progression of medial tibiofemoral knee osteoarthritis—results of a 1 year longitudinal arthroscopic study in 422 patients. Osteoarthr Cartil 13:361–367. doi:10.1016/j.joca.2005.01.005
Hao D, Li M, Wu Z, Duan Y, Li D, Qiu G (2011) Synovial fluid level of adiponectin correlated with levels of aggrecan degradation markers in osteoarthritis. Rheumatol Int 31:1433–1437. doi:10.1007/s00296-010-1516-0
Honsawek S, Chayanupatkul M (2010) Correlation of plasma and synovial fluid adiponectin with knee osteoarthritis severity. Arch Med Res 41:593–598. doi:10.1016/j.arcmed.2010.11.007
Staikos C, Ververidis A, Drosos G, Manolopoulos VG, Verettas DA, Tavridou A (2013) The association of adipokine levels in plasma and synovial fluid with the severity of knee osteoarthritis. Rheumatology 52(6):1077–1083. doi:10.1093/rheumatology/kes422
Bas S, Finckh A, Puskas GJ, Suva D, Hoffmeyer P, Gabay C, Lübbeke A (2014) Adipokines correlate with pain in lower limb osteoarthritis: different associations in hip and knee. Int Orthop 38:2577–2583. doi:10.1007/s00264-014-2416-9
Founding source
This work was supported by grant from the Dean of Medical Faculty II for Young Scientists, Poznan University of Medical Sciences (grant number 502-14-02217342).
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution License which permits any use, distribution, and reproduction in any medium, provided the original author(s) and the source are credited.
About this article
Cite this article
Richter, M., Trzeciak, T., Owecki, M. et al. The role of adipocytokines in the pathogenesis of knee joint osteoarthritis. International Orthopaedics (SICOT) 39, 1211–1217 (2015). https://doi.org/10.1007/s00264-015-2707-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00264-015-2707-9