Abstract
Increased cardiovascular (CV) morbidity and mortality have been found in rheumatoid arthritis (RA). Tumour necrosis factor α (TNF-α) inhibitors may improve vascular function. In the first part of this study, we determined microcirculation during postoocclusive reactive hyperemia (PORH) representing endothelial function. In a nonselected population (n = 46) we measured flow-mediated vasodilation (FMD) of the brachial artery and laser Doppler flow (LDF) by ultrasound. Among LDF parameters, we determined TH1 (time to half before hyperemia), TH2 (time to half after hyperemia), Tmax (time to maximum) and total hyperemic area (AH). We measured von Willebrand antigen (vWF:Ag) by ELISA. In the second part of the study, we assessed the effects of adalimumab treatment on microcirculatory parameters in 8 early RA patients at 0, 2, 4, 8 and 12 weeks. We found significant positive correlations between FMD and LDF Tmax (R = 0.456, p = 0.002), FMD and TH2 (R = 0.435, p = 0.004), and negative correlation between vWF:Ag and Tmax (R = − 0.4, p = 0.009) and between vWF:Ag and TH2 (R = − 0.446, p = 0.003). Upon adalimumab therapy in early RA, TH2 times improved in comparison to baseline (TH2baseline = 26.9 s vs. TH24weeks = 34.7 s, p = 0,032), and this effect prolonged until the end of treatment (TH28weeks = 40.5, p = 0.026; TH212weeks = 32.1, p = 0.013). After 8 weeks of treatment, significant improvement was found in AHa (AHbaseline = 1599 Perfusion Units [PU] vs. AH8weeks = 2724 PU, p = 0.045). The PORH test carried out with LDF is a sensitive option to measure endothelial dysfunction. TH1 and TH2 may be acceptable and reproducible markers. In our pilot study, treatment with adalimumab exerted favorable effects on disease activity, endothelial dysfunction and microcirculation in early RA patients.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Increased cardiovascular morbidity and mortality have been associated with rheumatoid arthritis (RA) and other inflammatory arthritides [1,2,3,4,5,6,7,8]. Both systemic inflammation and traditional cardiovascular risk factors are involved in this process [4, 8]. Endothelial dysfunction (ED) is considered to be the earliest sign of vascular pathophysiology underlying atherosclerosis. Thus, it may be a promising tool to detect high cardiovascular risk early [9, 10]. Despite the 25 years of experience in the detection of ED, its clinical utility has not been clearly defined due mainly to technical pitfalls and high variability [10, 11].
The most attractive approach to the assessment of ED seems to be postocclusive reactive hyperemia (PORH), where dynamic changes of capillary flow and resistance, conduit vessel flow and/or diameter may be detected. The brachial artery flow mediated, endothelium-dependent dilation (FMD) is the prototype of noninvasive endothelial function tests [10,11,12,13,14]. Under standard conditions, FMD may be the method of choice for the noninvasive determination of macrovascular ED [2, 10, 14]. In contrast, the laser Doppler flowmetry (LDF) allows to record blood flow during the PORH test on the microvascular level [10, 14]. In the case of LDF, most investigators applied acetylcholine (Ach) or sodium nitroprusside (SNP) iontophoresis during the assessment to detect endothelium-dependent and -independent microvascular function, respectively [10, 14,15,16]. To date, there have been few studies using LDF in RA. Most studies included Ach and/or SNP iontophoresis and were carried out in established but not early RA [15,16,17,18,19,20,21]. Only Ruiz-Limon et al. [22] applied PORH and LDF. Most studies did not correlate micro- and macrovascular pathophysiology [15,16,17, 21, 23,24,25,26]. In general, impaired endothelium-dependent microcirculation was found in RA [15, 17, 18, 21, 22, 24,25,26,27]. Two studies involved early RA patients, but LDF results were not correlated with FMD [26, 28].
In this study, we wished to determine the relationship of brachial artery FMD to the measured and calculated LDF PORH parameters to validate the probably best microcirculatory measure of ED. Then, as there have been very few studies carried out in early, untreated RA, we conducted a longitudinal pilot study to verify the clinical utility of microcirculatory PORH in inflammatory arthritis.
Subjects and methods
Healthy subjects and patients
In the first cross-sectional part of the study, 46 consecutive, unselected subjects were recruited from hospital practitioners and inpatient relatives representing the general population. There were 28 females and 18 males, with a mean age of 38 years (range 21–70 years). Exclusion criteria included known cardio- and cerebrovascular diseases, hypertension (blood pressure > 140/90 mmHg), diabetes mellitus, cigarette smoking, obesity (body mass index, BMI ≥ 30 kg/m2), current inflammatory, infectious disease or renal failure (serum creatinine ≥ 117 mmol/l). None of the patients received aspirin, clopidogrel, heparin or warfarin or vasoactive drugs.
In the second, pilot part of the study we longitudinally assessed the effects of the tumour necrosis factor α (TNF-α) inhibitor adalimumab on microcirculatory (LDF), macrovascular (brachial artery FMD) function, as well as vWF:Ag levels (biomarker of endothelial activation) in patients with early, untreated RA. Eight early RA patients (6 females and 2 males) with a mean age of 37.8 years (range 24–66 years) were included in the study. All patients had a disease duration ≤ 1 year. The mean disease duration was 5.6 months (range 3–11 months). All early RA patients satisfied the 2010 EULAR/ACR classification criteria for RA [29]. We recruited these RA patients from a pool of > 700 patients continuously undergoing follow-ups in our institution. None of the patients received corticosteroids, traditional or biologic disease-modifying antirheumatic drugs (DMARD) prior to the study. All patients received 40 mg adalimumab sc biweekly and vascular examinations were performed at baseline and 2, 4, 8 and 12 weeks after the initiation of treatment.
The study was approved by the Hungarian Scientific Research Council Ethical Committee (approval No. 14,804–2/2011/EKU). Written informed consent was obtained from each patient and assessments were carried out according to the Declaration of Helsinki.
Assessment of microvascular and macrovascular function
All assessments were carried out under standard conditions in an air-conditioned room at 21 °C after 20-min acclimatization, always in the morning. All subjects were fasting and had not used alcohol, tobacco, antioxidants and vasoactive drugs during the past 24 h according to current guidelines [30, 31].
For LDF assessments, individuals were placed in supine position and the right arm was fixed. A pneumatic cuff was firmly placed around the lower arm just below the antecubital fossa and two, one-point LDF probes (Periflux 4001 instrument, PF408, Perimed AB, Järfälla, Sweden) were applied to the forearm skin just below the cuff very close to each other proximally and distally aiming to record a primary and a control perfusion curves. Laser signal (780 nm wavelength) enters the upper layer of the skin and gets reflected from red blood cells in the circulation. A frequency shift is detected and flux is expressed in perfusion units (PU) which may be continuously recorded. Among LDF PORH parameters, the most relevant ones were time to maximum (TM), time to half before (TH1) and after hyperemia (TH2), acceleration phase of laser Doppler curve (AUCaccel), total hyperemic area (AH), its decelerating (AUCdecel) and one-minute long segment (AUC1min), peak flux (PF) (Table 1).
To detect FMD, a B-mode longitudinal section was obtained of the brachial artery above the antecubital fossa using a 10 MHz linear array transducer (ultrasound system: HP Sonos 5500), and the probe was fixed. After 2 min of simultaneous recording of LDF signals and the measurement of brachial artery resting diameter, the pneumatic cuff around the forearm was inflated to suprasystolic pressure for 5 min. To evoke PORH, the cuff was suddenly released and both the arterial diameter and the laser Doppler signals were 90 min long continuously recorded. The baseline and postocclusive diameters of brachial artery were ECG gated and detected offline. FMD values were expressed as % change from baseline value measured at 60 s after cuff deflation. The primary and control laser Doppler PORH curves were saved and later thoroughly evaluated (Perisoft for Windows, version 2.5.5.). Beyond the incorporated parameters a single observer (G.K.) dissolved the LDF PORH curves into resting, occlusion, acceleration, deceleration and at 60 s areas, and measured and calculated the absolute and relative time, flux, slope and area parameters (Fig. 1, Table 1).
A representative laser Doppler flow postocclusive reactive hyperemia curve. See Table 1 for abbreviation explanations
Every ultrasound examination was performed by a single trained sonographer (G.K.). In our hands, the intraobserver variability (coefficient of variation) of FMD test was 5% and the intraclass correlation coefficient (ICC) required for agreement studies was 0.935.
Determination of circulating von Willebrand factor levels
Circulating vWF:Ag is a marker of endothelial cell activation. Plasma levels of vWF:Ag were determined by STA Liatest vWF:Ag immunoturbidimetric assay using microlatex particles coated with polyclonal rabbit anti-human vWF:Ag antibodies (Diagnostica Stago, Asnieres, France). After mixing the reagent with plasma, the degree of agglutination was proportional to the amount of vWF:Ag present in the plasma sample. The reference range was 50–160%. The blood sampling was carried out early in the morning after overnight fasting before the acclimatisation from the left arm.
Data analysis
Descriptive data of normal variables are expressed as the mean ± SD. Statistical analysis for comparison was carried out by the SPSS 22.0 software. Distributions were assessed by Kolmogorow–Smirnov test. Correlations between variables were determined using Pearson correlation analysis for normally distributed values and Spearman correlation analysis as non-parametric test. R values of theses correlations were determined and corresponding p values < 0.05 were considered significant. As we had only eight early RA patients in the follow-up pilot study, only limited statistical analysis could be performed in the second part of the study. In case of the normal distribution, differences were assessed by Student’s paired t test using the SPSS 22.0 software.
To define agreement of PORH parameters, the intrasession variability were calculated using the identical measured and calculated parameters derived from primary and control laser Doppler probe. Intraclass colleration coefficients (ICC), coefficients of variations (CV) and Bland–Altman plots were applied (MedCalc 11.5.0).
Results
Simultaneous assessment of brachial artery FMD and laser Doppler PORH curves
After PORH test carried out in healthy individuals representing the general population (n = 46), FMD as a measure of macrovascular endothelial function were assessed on the forearm and the microcirculatory LDF PORH parameters detected by the primary probe were correlated with the reference FMD methods. LDF parameters are listed and explained in Table 1.
In this cohort, LDF time parameters significantly correlated with FMD. Both TM (p = 0.002) TH2 (p = 0.004) exerted significant, positive correlations with FMD. Only weak but statistically significant association was found between the FMD and the area of AUCaccel (p = 0.048). Neither the absolute and relative flux data, nor the most commonly investigated area of hyperemia and its derivatives showed any correlation with FMD (Table 2).
Association of laser Doppler parameters with vWF:Ag levels
As described above, circulating vWF:Ag levels are indicators of endothelial activation. Plasma vWF:Ag concentrations exerted significant negative correlation with TM (p = 0.009), TH1 (p = 0.014) and TH2 (p = 0.003) (Table 2). The absolute and relative flux and area parameters could not be associated with vWF:Ag concentrations.
Intrasession reproducibility of laser Doppler PORH parameters
Due to the significant variability of local skin circulation, in the healthy subject cohort, another control probe was placed in the near proximity of the primary detector. The agreement of corresponding PORH parameters derived from these one point probes were determined to assess their true clinical utility. Reproducibility values are seen in Table 2. Regarding LDF time parameters, TH1 seems to be the most reproducible. The intrasession reproducibility of TH2 is also acceptable, while the intrasession agreement of one point TM is considered to be poor. The reproducibility of all flux parameters was relatively poor. However, AH, AUCdecel and AUC1min exerted moderate reproducibility (Table 2). Thus, according to the ICC, CV results and the Bland–Altman plots the TH1 and TH2 parameters are the most stable methods in the estimation of microcirculatory endothelial function, and determination of TH2 may be the optimal method for a follow-up study (Fig. 2; Table 2).
Effects of adalimumab treatment on endothelial function in early RA
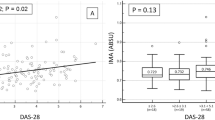
After the validation of the LDF PORH technique on healthy subjects, in the second part of the study, LDF was tested in a prospective pilot study. In 8 patients with early, untreated RA, adalimumab therapy successfully improved arthritis as it decreased CRP levels (p = 0.04) and disease activity (DAS28, p < 0.0001) (data not shown). Effects of adalimumab on endothelial function were assessed at baseline and after 2, 4, 8 and 12 weeks of therapy. Changes in the most relevant LDF parameters are included in Table 3. Microvascular endothelial function characterized by TH2 times, as the most reliable indicator, significantly improved in comparison to baseline as early as by week 4 (p = 0.032), and this effect was sustained until the end of treatment (p = 0.013) (Table 3, Fig. 3). TH1, the most reproducible parameter, which otherwise poorly represents endothelial function, also improved by week 12 in comparison to baseline (p = 0.001). TM, a good marker of endothelial function with poor reproducibility did not show any significant changes. In this small pilot study, PF increased markedly, however, due to its clinical unstability, the elevation was not significant (Table 3).
Some of the time- and flow-dependent area parameters also changed. After 8 weeks of treatment, significant improvement could be observed in AH (p = 0.045) and in AUC1min (p = 0.019) (Table 3). However, the latter paramaters may not reflect endothelial function as shown in our previous study. AUCaccel, which is a moderate marker of endothelial function, did not show any significant improvement as a consequence of poor reproducibility (Table 3).
As a positive control, brachial artery FMD and vWF:Ag levels were also assessed during the 12-week treatment follow-up period. FMD significantly improved from week 2 compared to baseline. This improvement was sustained until week 12. In contrast, vWF:Ag levels did not show significant decreases upon anti-TNF therapy (Fig. 4).
Discussion
The assessment of ED is the earliest and most sensitive marker for vascular damage, also in inflammatory diseases, such as RA [10, 14, 30, 31]. In the first part of our study, we tested PORH-provoked microcirculatory changes by LDF in healthy volunteers. Surprisingly, only the TM and TH2 parameters correlated moderately, and AUCaccel weakly with FMD. The validity of this observation was supported by the negative correlation between serum levels of the endothelial activation marker vWF:Ag and the time parameters. In summary, the prolongation of PORH curves due to decreased microcirculatory flow indicate better endothelial function. Our results also suggest, that only the time parameters, ideally TH2 may be used as a marker of endothelial function. Our results are in agreement with the early results of Carlsson et al. [32] indicating that PORH area is mainly prostaglandin- and adenosine-dependent. The NO antagonist L-NMMA pretreatment affected only the deceleration segment of PORH curve decreasing the TH2 [33], which result indirectly supports our conclusion. Other authors observed the same effect of L-NMMA on the deceleration phase of PORH curve on the level of macrocirculation [34, 35]. Thus, the detailed analysis of time parameters, particularly the deceleration phase of PORH curves allows to detect endothelial function [36, 37]. Furthermore, the measurement of total AH, which partly depends on the prolongation of PORH curve and TH2, may be used as a complex vascular function test [38]. Stiefel et al. [39] previously tested the excellent clinical utility of PORH studies: the complex vascular parameter AH showed good sensitivity and excellent specificity to predict coronary artery disease. The TH2 time prolongation seemed partly the cause of increased AH, and the TH2 parameter prolongation itself exerted good sensitivity and specificity for coronary artery disease. The last parameter has been above validated by our group as a true endothelial function marker. We also observed good intrasession reproducibility of time, moderate of area and poor of flux parameters.
The detailed analysis and validation of the PORH-mediated LDF technique were carried out in healthy volunteers. To assess its utility in a small group of rheumatic patients, in the second, pilot part of our study, we carried out an LDF and FMD pilot study in 8 patients with early untreated RA. To our knowledge, ours is the first pilot study to assess LDF parameters after PORH and not iontophoresis in early RA patients undergoing anti-TNF therapy. We found significant improvement in some LDF PORH curve parameters including TH1, TH2, AH and AUC1min. This was associated with significant improvement in FMD. Some LDF studies have previously been performed in chronic RA [15,16,17,18,19,20,21,22,23,24,25, 27, 39, 40]. Except for one recent study that utilized PORH [22], all other groups used Ach and/or SNP iontophoresis to detect endothelium-dependent and –independent microvascular endothelial function, respectively [15,16,17,18,19,20,21, 23,24,25, 27, 28]. Most studies reported impaired skin microcirculation in RA as determined by LDF [15,16,17,18,19, 21,22,23,24, 27, 40]. Van Eijk et al. [28] reported preserved microcirculatory capacity in a cohort of early RA patients with low disease activity. Impaired skin microcirculation was associated with inflammatory activity [18] and increased CRP levels [17, 19] but not with low RA disease activity [28]. Others did not find associations between micro- or macrocirculation and inflammatory activity [24]. In one study, impaired Ach-induced, microcirculatory endothelial function could be associated with global cardiovascular risk [24]. Very few longitudinal studies were conducted to determine the effects of antirheumatic therapy including biologics on LDF parameters. Treatment of RA resulted in improved microcirculation in some studies [22, 24, 27] but not in others [17, 18]. In the active, established RA cohort of Galarraga et al. [18], no improvement of Ach or SNP iontophoresis-induced microcirculation could be observed upon anti-TNF therapy in the whole population. However, LDF parameters improved in responders [18]. In the only available, recent LDF PORH study, Ruiz-Limon et al. [22] reported improvement of skin microcirculation upon tocilizumab treatment of chronic RA patients. Only very few studies included early RA patients [26, 28]. Foster et al. [26] assessed iontophoresis-induced LDF and FMD in 18 patients with new-onset RA. Interestingly, only endothelium-independent micro- (SNP-induced) and macrocirculation (GTN-induced) were found to be impaired [26]. Similarly, as described above, Van Eijk et al. [28] could not demonstrate impairment of microcirculation in early RA patients with low CRP. Finally, only two studies compared micro- and macrovascular endothelial function in RA patients [15, 41]. Neither Arosio et al. [15] nor Sandoo et al. [41] could not demonstrate correlation between LDF parameters and FMD in patients with established RA. Thus, most LDF studies using iontophoresis demonstrated impaired skin microcirculation, probably associated with higher CRP and disease activity.
Our study may have important advantages and limitations. Among the advantages… Limitations include the small number of patients in the second, pilot part of our study.
In conclusion, in our two-stage study, first we demonstrated that the TH2 parameter of LDF PORH test may be a reliable and relatively stable marker of endothelial function in the normal population. The most commonly used area parameters did not correlate with macrovascular endothelial function indicating they would rather complex vascular function parameters. We verified in a pilot study conducted in early RA patients that in a pathological condition, where reversible ED may be an important phenomenon, early, effective treatment may improve microcirculation and this can be monitored by the operator-independent laser Doppler technique. It remains to be determined whether LDF could have an additional value in cardiovascular risk stratification. One possible limitation of our study is that we used single-point laser, which was sufficient to detect the common denominators of different endothelial function parameters. However, we do not know, whether these result could be extrapolated to modern laser techniques, such as LSCI. LSCI may, at least partly, eliminate the disadvantages of the one-point laser method probably by introducing more reproducible parameters to our experimental and clinical armamentarium.
Change history
06 January 2020
In the Original article, the legend of Figures 3 and 4 are interchanged and line number 387 is incomplete.
Abbreviations
- Ach:
-
Acetylcholine
- BMI:
-
Body mass index
- CRP:
-
C-reactive protein
- DAS28:
-
28-Joint disease activity scale
- DMARD:
-
Disease-modifying antirheumatic drug
- ED:
-
Endothelial dysfunction
- FMD:
-
Flow-mediated vasodilation
- GTN:
-
Glyceryl trinitrate
- LDF:
-
Laser Doppler flowmetry
- LSCI:
-
Laser speckle contrast imaging
- NO:
-
Nitric oxide
- PORH:
-
Post-occlusive reactive hyperemia
- RA:
-
Rheumatoid arthritis
- SNP:
-
Sodium nitroprusside
- TNF-α:
-
Tumour necrosis factor alpha,
- vWF:Ag:
-
Von Willebrand factor antigen
References
Szekanecz Z, Kerekes G, Der H, Sandor Z, Szabo Z, Vegvari A et al (2007) Accelerated atherosclerosis in rheumatoid arthritis. Ann N Y Acad Sci 1108:349–358. https://doi.org/10.1196/annals.1422.036
Kerekes G, Szekanecz Z, Der H, Sandor Z, Lakos G, Muszbek L et al (2008) Endothelial dysfunction and atherosclerosis in rheumatoid arthritis: a multiparametric analysis using imaging techniques and laboratory markers of inflammation and autoimmunity. J Rheumatol 35:398–406
Shoenfeld Y, Gerli R, Doria A, Matsuura E, Cerinic MM, Ronda N et al (2005) Accelerated atherosclerosis in autoimmune rheumatic diseases. Circulation 112:3337–3347. https://doi.org/10.1161/CIRCULATIONAHA.104.507996
Agca R, Heslinga SC, Rollefstad S, Heslinga M, McInnes IB, Peters MJ et al (2017) EULAR recommendations for cardiovascular disease risk management in patients with rheumatoid arthritis and other forms of inflammatory joint disorders: 2015/2016 update. Ann Rheum Dis 76:17–28. https://doi.org/10.1136/annrheumdis-2016-209775
Wallberg-Jonsson S, Ohman M, Rantapaa-Dahlqvist S (2004) Which factors are related to the presence of atherosclerosis in rheumatoid arthritis? Scand J Rheumatol 33:373–379. https://doi.org/10.1080/03009740410010308
Nurmohamed MT, Kitas G (2011) Cardiovascular risk in rheumatoid arthritis and diabetes: how does it compare and when does it start? Ann Rheum Dis 70:881–883. https://doi.org/10.1136/ard.2010.145839
Gonzalez-Gay MA, Szekanecz Z, Popa CD, Dessein P (2012) Atherosclerosis in rheumatoid arthritis. Mediators Inflamm 2012:489608. https://doi.org/10.1155/2012/489608
Szekanecz Z, Kerekes G, Vegh E, Kardos Z, Barath Z, Tamasi L et al (2016) Autoimmune atherosclerosis in 3D: how it develops, how to diagnose and what to do. Autoimmun Rev 15:756–769. https://doi.org/10.1016/j.autrev.2016.03.014
Giannotti G, Landmesser U (2007) Endothelial dysfunction as an early sign of atherosclerosis. Herz 32:568–572. https://doi.org/10.1007/s00059-007-3073-1
Kerekes G, Soltesz P, Nurmohamed MT, Gonzalez-Gay MA, Turiel M, Vegh E et al (2012) Validated methods for assessment of subclinical atherosclerosis in rheumatology. Nat Rev Rheumatol 8:224–234. https://doi.org/10.1038/nrrheum.2012.16
Anderson TJ, Uehata A, Gerhard MD, Meredith IT, Knab S, Delagrange D et al (1995) Close relation of endothelial function in the human coronary and peripheral circulations. J Am Coll Cardiol 26:1235–1241. https://doi.org/10.1016/0735-1097(95)00327-4
Lefroy DC, Crake T, Uren NG, Davies GJ, Maseri A (1993) Effect of inhibition of nitric oxide synthesis on epicardial coronary artery caliber and coronary blood flow in humans. Circulation 88:43–54. https://doi.org/10.1161/01.cir.88.1.43
Joannides R, Haefeli WE, Linder L, Richard V, Bakkali EH, Thuillez C et al (1995) Nitric oxide is responsible for flow-dependent dilatation of human peripheral conduit arteries in vivo. Circulation 91:1314–1319. https://doi.org/10.1161/01.cir.91.5.1314
Khan F, Galarraga B, Belch JJ (2010) The role of endothelial function and its assessment in rheumatoid arthritis. Nat Rev Rheumatol 6:253–261. https://doi.org/10.1038/nrrheum.2010.44
Arosio E, De Marchi S, Rigoni A, Prior M, Delva P, Lechi A (2007) Forearm haemodynamics, arterial stiffness and microcirculatory reactivity in rheumatoid arthritis. J Hypertens 25:1273–1278. https://doi.org/10.1097/HJH.0b013e3280b0157e
Fenton SAM, Sandoo A, Metsios GS, Duda JL, Kitas GD, Veldhuijzen van Zanten JJ (2018) Sitting time is negatively related to microvascular endothelium-dependent function in rheumatoid arthritis. Microvasc Res 117:57–60. https://doi.org/10.1016/j.mvr.2018.01.005
Foster W, Carruthers D, Lip GY, Blann AD (2010) Inflammation and microvascular and macrovascular endothelial dysfunction in rheumatoid arthritis: effect of treatment. J Rheumatol 37:711–716. https://doi.org/10.3899/jrheum.090699
Galarraga B, Belch JJ, Pullar T, Ogston S, Khan F (2010) Clinical improvement in rheumatoid arthritis is associated with healthier microvascular function in patients who respond to antirheumatic therapy. J Rheumatol 37:521–528. https://doi.org/10.3899/jrheum.090417
Galarraga B, Khan F, Kumar P, Pullar T, Belch JJ (2008) C-reactive protein: the underlying cause of microvascular dysfunction in rheumatoid arthritis. Rheumatology (Oxford) 47:1780–1784. https://doi.org/10.1093/rheumatology/ken386
Sandoo A, Carroll D, Metsios GS, Kitas GD, Veldhuijzen van Zanten JJ (2011) The association between microvascular and macrovascular endothelial function in patients with rheumatoid arthritis: a cross-sectional study. Arthritis Res Ther 13:R99. https://doi.org/10.1186/ar3374
Klimek E, Sulicka J, Gryglewska B, Skalska A, Kwasny-Krochin B, Korkosz M et al (2017) Alterations in skin microvascular function in patients with rheumatoid arthritis and ankylosing spondylitis. Clin Hemorheol Microcirc 65:77–91. https://doi.org/10.3233/CH-15112
Ruiz-Limon P, Ortega R, Arias de la Rosa I, Abalos-Aguilera MDC, Perez-Sanchez C, Jimenez-Gomez Y et al (2017) Tocilizumab improves the proatherothrombotic profile of rheumatoid arthritis patients modulating endothelial dysfunction, NETosis, and inflammation. Transl Res 183:87–103. https://doi.org/10.1016/j.trsl.2016.12.003
Sandoo A, Dimitroulas T, Veldhuijzen van Zanten JJ, Smith JP, Metsios GS, Nightingale P et al (2012) Lack of association between asymmetric dimethylarginine and in vivo microvascular and macrovascular endothelial function in patients with rheumatoid arthritis. Clin Exp Rheumatol 30:388–396
Sandoo A, Kitas GD, Carroll D, Veldhuijzen van Zanten JJ (2012) The role of inflammation and cardiovascular disease risk on microvascular and macrovascular endothelial function in patients with rheumatoid arthritis: a cross-sectional and longitudinal study. Arthritis Res Ther 14:R117. https://doi.org/10.1186/ar3847
Dimitroulas T, Hodson J, Sandoo A, Smith J, Kitas GD (2017) Endothelial injury in rheumatoid arthritis: a crosstalk between dimethylarginines and systemic inflammation. Arthritis Res Ther 19:32. https://doi.org/10.1186/s13075-017-1232-1
Foster W, Lip GY, Raza K, Carruthers D, Blann AD (2011) An observational study of endothelial function in early arthritis. Eur J Clin Invest 42:510–516. https://doi.org/10.1111/j.1365-2362.2011.02607.x
Datta D, Ferrell WR, Sturrock RD, Jadhav ST, Sattar N (2007) Inflammatory suppression rapidly attenuates microvascular dysfunction in rheumatoid arthritis. Atherosclerosis 192:391–395. https://doi.org/10.1016/j.atherosclerosis.2006.05.034
van Eijk IC, Serne EH, Dijkmans BA, Smulders Y, Nurmohamed M (2011) Microvascular function is preserved in newly diagnosed rheumatoid arthritis and low systemic inflammatory activity. Clin Rheumatol 30:1113–1118. https://doi.org/10.1007/s10067-011-1750-1
Aletaha D, Neogi T, Silman AJ, Funovits J, Felson DT, Bingham CO 3rd et al (2010) 2010 rheumatoid arthritis classification criteria: an American College of Rheumatology/European League Against Rheumatism collaborative initiative. Ann Rheum Dis 69:1580–1588. https://doi.org/10.1002/art.27584
Corretti MC, Anderson TJ, Benjamin EJ, Celermajer D, Charbonneau F, Creager MA et al (2002) Guidelines for the ultrasound assessment of endothelial-dependent flow-mediated vasodilation of the brachial artery: a report of the International Brachial Artery Reactivity Task Force. J Am Coll Cardiol 39:257–265. https://doi.org/10.1016/s0735-1097(01)01746-6
Thijssen DH, Black MA, Pyke KE, Padilla J, Atkinson G, Harris RA et al (2010) Assessment of flow-mediated dilation in humans: a methodological and physiological guideline. Am J Physiol Heart Circ Physiol 300:H2–H12. https://doi.org/10.1152/ajpheart.00471.2010
Carlsson I, Sollevi A, Wennmalm A (1987) The role of myogenic relaxation, adenosine and prostaglandins in human forearm reactive hyperaemia. J Physiol 389:147–161. https://doi.org/10.1113/jphysiol.1987.sp016651
Cankar K, Finderle Z, Strucl M (2009) The effect of alpha-adrenoceptor agonists and L-NMMA on cutaneous postocclusive reactive hyperemia. Microvasc Res 77:198–203. https://doi.org/10.1016/j.mvr.2008.10.004
Tagawa T, Imaizumi T, Endo T, Shiramoto M, Harasawa Y, Takeshita A (1994) Role of nitric oxide in reactive hyperemia in human forearm vessels. Circulation 90:2285–2290. https://doi.org/10.1161/01.cir.90.5.2285
Kooijman M, Thijssen DH, de Groot PC, Bleeker MW, van Kuppevelt HJ, Green DJ et al (2008) Flow-mediated dilatation in the superficial femoral artery is nitric oxide mediated in humans. J Physiol 586:1137–1145. https://doi.org/10.1113/jphysiol.2007.145722
Hansell J, Henareh L, Agewall S, Norman M (2004) Non-invasive assessment of endothelial function—relation between vasodilatory responses in skin microcirculation and brachial artery. Clin Physiol Funct Imaging 24:317–322. https://doi.org/10.1111/j.1475-097X.2004.00575.x
Debbabi H, Bonnin P, Ducluzeau PH, Leftheriotis G, Levy BI (2010) Noninvasive assessment of endothelial function in the skin microcirculation. Am J Hypertens 23:541–546. https://doi.org/10.1038/ajh.2010.10
Anderson TJ, Phillips SA (2015) Assessment and prognosis of peripheral artery measures of vascular function. Prog Cardiovasc Dis 57:497–509. https://doi.org/10.1016/j.pcad.2014.11.005
Stiefel P, Moreno-Luna R, Vallejo-Vaz AJ, Beltran LM, Costa A, Gomez L et al (2012) Which parameter is better to define endothelial dysfunction in a test of postocclusive hyperemia measured by laser-Doppler flowmetry? Coron Artery Dis 23:57–61. https://doi.org/10.1097/MCA.0b013e32834e4f34
Dimitroulas T, Sandoo A, Hodson J, Smith J, Panoulas VF, Kitas GD (2014) Relationship between dimethylarginine dimethylaminohydrolase gene variants and asymmetric dimethylarginine in patients with rheumatoid arthritis. Atherosclerosis 237:38–44. https://doi.org/10.1016/j.atherosclerosis.2014.07.033
Sandoo A, Hodson J, Douglas KM, Smith JP, Kitas GD (2013) The association between functional and morphological assessments of endothelial function in patients with rheumatoid arthritis: a cross-sectional study. Arthritis Res Ther 15:R107. https://doi.org/10.1186/ar4287
Acknowledgements
Open access funding provided by University of Debrecen (DE).
Funding
This work was supported by the European Union and the State of Hungary co-financed by the European Social Fund in the framework of TAMOP-4.2.4.A/2-11/1-2012-0001 ‘National Excellence Program’(Z.S.); also by the European Union grants GINOP-2.3.2-15-2016-00015 and GINOP-2.3.2-15-2016-00050 (Z.S.).
Author information
Authors and Affiliations
Contributions
GK: study design, patient examination, LDF and FMD measurements, manuscript draft. LD: patient examination, data analysis, manuscript draft corrections. VP: patient examination, data analysis, LDF and FMD measurements. EAM: data analysis, manuscript draft corrections. SS: patient and control subject supply, patient exam, manuscript correction. GS: patient and control subject supply, patient exam, manuscript correction. AV: patient and control subject supply, patient exam, manuscript correction. OT: LDF and FMD measurement, manuscript correction. ZC: study design, data analysis, manuscript correction. EV: LDF and FMD measurement, manuscript correction. PS: study design, manuscript review. ZS, project manager, expert advices, study design, manuscript draft.
Corresponding author
Ethics declarations
Conflict of interest
None of the authors have any potential conflicts of interest.
Informed consent
Informed consent was obtained from each healthy subject or RA patient.
Ethical approval
For this study, we also obtained institutional review board approval from the University of Debrecen.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Dávida, L., Pongrácz, V., Mohamed, E.A. et al. A prospective, longitudinal monocentric study on laser Doppler imaging of microcirculation: comparison with macrovascular pathophysiology and effect of adalimumab treatment in early rheumatoid arthritis. Rheumatol Int 40, 415–424 (2020). https://doi.org/10.1007/s00296-019-04503-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00296-019-04503-5