Abstract
The aim of this study was to systematically review and meta-analyze published data on the diagnostic performance of combined 18F-fluoro-2-deoxyglucose positron emission tomography/computed tomography (FDG-PET/CT) in the detection of primary tumors in patients with cancer of unknown primary (CUP). A systematic search for relevant studies was performed of the PubMed/MEDLINE and Embase databases. Methodological quality of the included studies was assessed. Reported detection rates, sensitivities and specificities were meta-analyzed. Subgroup analyses were performed if results of individual studies were heterogeneous. The 11 included studies, comprising a total sample size of 433 patients with CUP, had moderate methodological quality. Overall primary tumor detection rate, pooled sensitivity and specificity of FDG-PET/CT were 37%, 84% (95% CI 78–88%) and 84% (95% CI 78–89%), respectively. Sensitivity was heterogeneous across studies (P = 0.0001), whereas specificity was homogeneous across studies (P = 0.2114). Completeness of diagnostic workup before FDG-PET/CT, location of metastases of unknown primary, administration of CT contrast agents, type of FDG-PET/CT images evaluated and way of FDG-PET/CT review did not significantly influence diagnostic performance. In conclusion, FDG-PET/CT can be a useful method for unknown primary tumor detection. Future studies are required to prove the assumed advantage of FDG-PET/CT over FDG-PET alone and to further explore causes of heterogeneity.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Cancer of unknown primary (CUP), defined as the presence of histologically proven metastatic disease for which the site of origin cannot be identified at the time of diagnosis (despite comprehensive diagnostic workup), is one of the ten most frequent cancers (accounting for 3–5% of all malignancies) and is the fourth most common cause of cancer-related death [1, 2]. Failure to detect the primary tumor impedes optimization of treatment planning, which, in turn, may negatively influence patient prognosis. 18F-fluoro-2-deoxyglucose positron emission tomography (FDG-PET) allows whole-body tumor detection [3] and has proven to be useful in patients with CUP for the detection of the primary tumor [4–6]. A disadvantage of FDG-PET, however, is its lack of anatomic information, which may impede precise localization of FDG accumulation. Furthermore, tumors with low or even no FDG uptake may be missed by FDG-PET. Complimentary anatomic information, provided by computed tomography (CT) or magnetic resonance (MR) imaging, may improve the diagnostic performance of FDG-PET alone. The relatively recently introduced combined FDG-PET/CT scanner allows obtaining both functional and anatomic images in a single examination [7, 8] and may be of great value for the detection of primary tumors in patients with CUP. The purpose of this study was therefore to systematically review and meta-analyze published data on the diagnostic performance of FDG-PET/CT in unknown primary tumor detection.
Methods
Search strategy
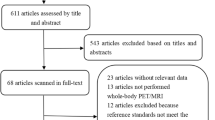
A computer-aided search of the PubMed/MEDLINE and Embase databases was conducted to find relevant published articles on the diagnostic performance of combined FDG-PET/CT in primary tumor detection in patients with CUP. The search strategy is presented in Table 1. No beginning date limit was used. The search was updated until 13 March 2008. Only English-, German-, French-, Italian- or Spanish-language studies were considered because the investigators were familiar with these languages. To expand our search, bibliographies of articles that finally remained after the selection process were screened for potentially suitable references.
Study selection
Studies or subsets in studies investigating the diagnostic performance of FDG-PET/CT in primary tumor detection in patients with CUP were eligible for inclusion. Review articles, meta-analyses, abstracts, editorials or letters, case reports, guidelines for management and studies examining ten or fewer patients with CUP were excluded. Studies or subsets in studies were excluded if metastases were not histologically confirmed. Studies that provided insufficient data to construct a 2 × 2 contingency table to calculate sensitivity and specificity for primary tumor detection in patients with CUP were also excluded. When data were presented in more than one article, the article with the largest number of patients or the article with the most details was chosen.
Two researchers (T.C.K., R.M.K.) independently reviewed the titles and abstracts of the retrieved articles, applying the inclusion and exclusion criteria mentioned above. Articles were rejected if they were clearly ineligible. The same two researchers then independently reviewed the full-text version of the remaining articles to determine their eligibility for inclusion. Disagreements were resolved in a consensus meeting.
Data abstraction
For each included study, information was collected concerning basic study and patient characteristics (author names, year of publication, country of origin, study design, number, age and sex ratio of investigated patients, location of metastases of unknown primary, histology of metastases of unknown primary and diagnostic workup before FDG-PET/CT), FDG-PET/CT parameters (time of fasting before FDG administration, FDG dose, time interval between FDG administration and data acquisition, number of CT detector rows, reconstructed CT slice width, administration of intravenous and/or oral CT contrast agents and area of body examined) and FDG-PET/CT evaluation (evaluation of attenuation-corrected and/or non-attenuation corrected images, interpreters(s) of FDG-PET/CT, criteria for positivity and applied reference standard).
To calculate estimates of diagnostic performance (i.e., primary tumor detection rate, sensitivity and specificity), a true-positive result was considered when FDG-PET/CT suggested the location of the primary tumor and was subsequently confirmed. A false-positive result was considered when this location was not confirmed. The sites suggested by FDG-PET/CT were confirmed by histopathological analysis of tissue obtained by biopsy or surgery, considered as the reference standard. However, imaging procedures or clinical follow-up was accepted if no histopathological proof could be obtained. A true-negative result was considered when neither FDG-PET/CT nor the reference standard could detect the primary tumor. A false-negative result was considered if the primary tumor was detected in a particular location that was negative on FDG-PET/CT [4, 6].
Study quality
For each included study, the methodological quality was assessed by using the Quality Assessment of Studies of Diagnostic Accuracy Included in Systematic Reviews (QUADAS) criteria, which is a 14-item instrument [9, 10]. The item “Is the time period between reference standard and index test short enough to be reasonably sure that the target condition did not change between the two tests?” was removed from the standard QUADAS list, since follow-up is required to verify negative FDG-PET/CT findings. The item “Did patients receive the same reference standard regardless of the index test result?” was removed from the standard QUADAS list, since positive FDG-PET/CT findings can be confirmed by means of histology, but negative FDG-PET/CT findings require follow-up. The item “Were the reference standard results interpreted without knowledge of the results of the index test?” was also removed from the standard QUADAS list, since positive FDG-PET/CT findings can be verified by means of histopathological analysis of tissue obtained by biopsy or surgery. One item was added to the standard QUADAS list: “Was comparator review bias avoided?” The complete list of quality items is displayed in Table 2. For each item, the two researchers (TCK, RMK) independently assessed whether it was fulfilled (yes or no). If it was unclear from the information provided in an article as to whether an item was fulfilled, the item was rated as “unclear.” Both “no” and “unclear” responses were interpreted as indicating that the quality criterion was not met. Disagreements were discussed and resolved by consensus. The total quality score was expressed as a percentage of the maximum score of 12.
Statistical analysis
Primary tumor detection rates of individual studies were calculated and totaled. Locations of primary tumors detected by FDG-PET/CT, locations of false-positives FDG-PET/CT findings and locations of false-negative FDG-PET/CT findings were recorded and summarized. Sensitivities and specificities of FDG-PET/CT in primary tumor detection (with corresponding 95% CIs) were calculated from the original numbers given in the included studies and meta-analyzed using a random effects model. Where sensitivity or specificity estimates for an individual study were zero, a continuity correction of 0.5 was added to every value for that study in order to make the calculation of sensitivity and specificity defined.
A chi-squared test was performed to test for heterogeneity between studies. Heterogeneity was defined as P < 0.10. Differences in sensitivities and specificities due to different cut-offs (thresholds) used in different studies to define a positive (or negative) FDG-PET/CT results were assessed by computing the Spearman correlation coefficient between the logit of sensitivity and logit of 1-specificity. A strong positive correlation would suggest the presence of a threshold effect. Other potential sources for heterogeneity were explored by assessing whether certain predefined covariates significantly influenced (i.e., P < 0.05) the relative diagnostic odds ratio (RDOR) [11]. Although the findings of such analyses should be regarded mainly as hypothesis generating, statistical significance may suggest substantial changes in the diagnostic performance of the test under study as the covariate changes. Specifically, analyses were performed according to completeness of diagnostic workup before FDG-PET/CT (studies that fulfilled quality item 1 vs. studies that did not fulfill quality item 1 [Table 2]), location of metastases of unknown primary (cervical vs. extracervical), administration of CT contrast agents (both intravenous and oral contrast vs. no intravenous or oral contrast agent, or not reported), type of FDG-PET/CT images evaluated (both attenuation-corrected and non-attenuation-corrected images vs. attenuation-corrected images only, or not reported) and way of FDG-PET/CT review (reported blinding to reference test vs. no or unreported blinding to reference test.
Statistical analyses were executed using the Statistical Package for the Social Sciences version 14.0 software (SPSS Inc., Chicago, IL) and Meta-DiSc statistical software version 1.4 (Unit of Clinical Biostatistics, Ramón y Cajal Hospital, Madrid, Spain) [11].
Results
Literature search
The computer-aided search revealed 230 articles from PubMed/MEDLINE and 185 articles from Embase (Table 1). Reviewing titles and abstracts from PubMed/MEDLINE revealed 17 articles potentially eligible for inclusion. Reviewing titles and abstracts from Embase revealed 17 articles potentially eligible for inclusion, of which 15 were all already identified by the PubMed/MEDLINE search. Thus, 19 studies [12–30] remained for possible inclusion and were retrieved in full text version. Screening of the references of these 19 articles did not bring up new articles. After reviewing the full article, two articles [17, 19] were excluded because a part of the study population underwent FDG-PET alone and was not separately analyzed from patients undergoing FDG-PET/CT, two articles [25, 30] were excluded because sensitivity and specificity for primary tumor detection could not be calculated, one article [28] was excluded because it did not separately analyze patients with CUP, one article [21] was excluded because fewer than ten patients with CUP were included, one article [23] was excluded because it was an editorial, and one article [24] was excluded because the same data were used in a later study. Eventually, 11 studies [12–16, 18, 20, 22, 26, 27, 29], comprising a total sample size of 433 patients with CUP, met all inclusion and exclusion criteria, and they were included in this systematic review. The characteristics of the included studies are presented in Tables 3, 4, 5 and 6.
Methodological quality assessment
Twelve methodological quality items were assessed for each of the 11 included studies (Table 7). The total methodological quality score, expressed as a fraction of the maximum score, ranged from 42% to 75% (median, 50%).
Diagnostic performance
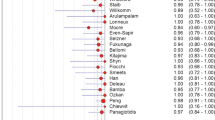
The results of the 11 included studies are presented in Tables 8 and 9, and Figs. 1, 2 and 3. Primary tumor detection rates ranged from 22 to 73%, with an overall detection rate of 37% (162/433) (Table 8). The most commonly detected location of the primary tumor by FDG-PET/CT was the lung, 33% (Fig. 1). The most common locations of false-positive FDG-PET/CT findings were the lung and the oropharynx, both 15% (Fig. 2). The most common cause of false-negative FDG-PET/CT findings was breast cancer, 27% (Fig. 3). Sensitivity and specificity of FDG-PET/CT in primary tumor detection ranged from 55% to 100% and from 73% to 100%, with pooled estimates of 84% (95% CI 78–88%) and 84% (95% CI 78–89%), respectively (Table 8). The included studies were statistically heterogeneous in their estimates of sensitivity (P = 0.0001), but homogeneous in their estimates of specificity (P = 0.2114).
A Spearman correlation coefficient of -0.201 (P = 0.554) between the logit of sensitivity and logit of 1-specificity did not suggest the presence of a threshold effect. No significantly increased RDORs were observed in any of the subgroup analyses according to completeness of diagnostic workup before FDG-PET/CT, location of metastases of unknown primary, administration of CT contrast agents, type of FDG-PET/CT images evaluated and way of FDG-PET/CT review (Table 9).
Discussion
CUP represents a heterogeneous group of metastatic tumors for which no primary site can be detected following a thorough medical history, careful clinical examination and extensive diagnostic workup [1, 2]. Attempts to identify the primary tumor in patients with CUP are are often time consuming, expensive and ultimately unsuccessful [1, 2]. The results of this study indicate that, overall, FDG-PET/CT is able to detect 37% of primary tumors in patients with CUP, and sensitivity and specificity are reasonably high (both 84%). Sensitivity, however, was heterogeneous across studies. Subgroup analysis could not clarify the observed heterogeneity. It should be noted, however, that results from our subgroup analysis may not be conclusive because of the relatively small number of included studies. Furthermore, it was not possible to perform subgroup analysis according to the number of CT detector rows and CT slice width due to incomplete reporting of included studies. Future studies are required to further investigate potential sources of heterogeneity.
The results of this systematic review should be interpreted carefully, since the methodological quality of the included studies was moderate. Several methodological shortcomings were identified, of which spectrum bias, bias due to the use of an inadequate reference standard and verification bias may seriously have affected the results. According to Pavlidis et al. [1, 2], the precise clinical definition of CUP should refer to patients who present with histologically confirmed metastatic cancer in whom medical history, physical examination, full blood count, basic biochemistry battery, urinalysis, stool occult blood testing, immunohistochemistry with specific markers as well as imaging technology with chest X-ray, CT of the chest abdomen and pelvis or mammography and MR imaging in certain cases have failed to detect the primary tumor. However, only two studies [22, 26] (18%) included patients who fulfilled these criteria. In the other nine studies [12–16, 18, 20, 27, 29], diagnostic performance of FDG-PET/CT might have been overestimated because of (possible) incomplete diagnostic workup. Furthermore, only five studies [12, 15, 20, 22, 27] applied an adequate reference standard of histology and follow-up in all patients. Consequently, in the other six studies [13, 14, 16, 18, 26, 29], diagnostic performance of FDG-PET/CT might have been overestimated. Other methodological flaws include an inadequate description of selection criteria in seven studies [13, 16, 18, 22, 26, 27, 29] (64%), the possibility that FDG-PET/CT formed part of the reference standard (incorporation bias) in five studies [13, 15, 18, 20, 22] (46%), inadequate description of the execution of FDG-PET/CT in six studies [14, 15, 18, 20, 22, 26] (55%), inadequate description of the reference standard in six studies [12–15, 20, 22] (55%), possible interpretation of FDG-PET/CT while knowing the results of the reference standard (test review bias) in eight studies [12–15, 18, 20, 22, 27] (73%) and possible interpretation of FDG-PET/CT without knowledge of the histological nature of the metastases of unknown primary in nine studies [12–15, 16, 20, 22, 27, 29] (82%).
Lung, oropharyngeal and pancreatic carcinoma were the most frequently detected primary tumors by FDG-PET/CT in this meta-analysis (33%, 16% and 5%, respectively). This is partly in line with previous autopsy studies in patients with CUP [31–34], which have shown that the most common locations of the primary tumor are the lung and the pancreas. The high detection rate of oropharyngeal carcinoma by FDG-PET/CT in this meta-analysis, however, is discrepant with the results of autopsy studies [31–34]. This finding can by explained by the fact that 4 of 11 included studies [13, 16, 20, 27] exclusively investigated patients with cervical metastases of unknown primary, whose primary tumors are most frequently located in the oropharynx [5, 35]. Indeed, these four studies [13, 16, 20, 27] comprised 69% of all oropharyngeal carcinomas detected by FDG-PET/CT in this meta-analysis. The most commonly reported locations of false-positive FDG-PET/CT results were the lung and the oropharynx (both 15%). Causes of false-positive results may be FDG uptake in benign conditions with increased glycolysis (e.g., one false-positive FDG-PET/CT finding in the lung proved to be a pulmonary infarction [29]), high physiological FDG uptake (e.g., muscle FDG uptake) and failure to evaluate both attenuation-corrected and non-attentuation-corrected images to minimize the chance of misinterpreting (FDG-PET/CT) artifacts as pathologic (only three of the included studies [12, 16, 29] explicitly stated that both attenuation-corrected and non-attentuation-corrected images were evaluated) [36–38]. Breast cancer was the most common cause of false-negative FDG-PET/CT results (27%). This may be explained by the fact that small (<1 cm) and slow-growing, low-grade (breast) cancers with low or no FDG uptake (e.g., tubular carcinoma and noninvasive cancers such as ductal or lobular carcinoma in situ) may be overlooked on FDG-PET/CT [39].
FDG-PET alone has been thoroughly investigated for primary tumor detection in patients with CUP; meta-analyses on FDG-PET reported primary tumor detection rates ranging between 24.5% and 43%, sensitivities ranging between 87% and 91.9%, and specificities ranging between 71% and 81.9% [4–6]. An advantage of FDG-PET/CT over FDG-PET alone is more accurate localization of foci with increased FDG uptake, and this may reduce the problems of physiological FDG uptake being misinterpreted as pathological and false localization of disease. In addition, tumors with low or no FDG uptake, or tumors of a size below the spatial resolution of FDG-PET, may be depicted by the CT component of FDG-PET/CT. Recently introduced combined FDG-PET/CT scanners with a 64-section multidetector CT component and less than 2.5-mm collimation may detect small primary cancers in the lungs or oropharynx earlier [40] and are clearly superior to FDG-PET/CT alone. Furthermore, the additional anatomic data obtained using FDG-PET/CT may increase the accuracy of FDG-PET-directed biopsies. Another advantage of FDG-PET/CT is the use of the CT images for attenuation correction of the PET emission data, which reduces whole-body scanning times from 45 min to 30 min or less. This approach also provides low-noise attenuation correction factors, compared with those from standard PET transmission measurements using an external radiation source, and eliminates bias from emission contamination of postinjection transmission scans [7, 8]. On the other hand, a disadvantage of CT-based attenuation correction may be misclassification of regions containing high concentrations of CT contrast medium with high-density bone (CT contrast agents have high atomic numbers relative to the atomic number of bone, and as the concentration of a contrast agent increases, its corresponding CT number will fall within the CT number range for bone), which results in overcorrection for photon attenuation, consequently leading to an overestimation of FDG uptake in the contrast-enhanced region [41, 42]. Three of the included studies [13, 27, 29] directly compared FDG-PET/CT and (CT-based attenuation-corrected) FDG-PET alone. In all three studies [13, 27, 29], FDG-PET/CT was able to detect a few more primary tumors than FDG-PET alone, but these differences were not statistically significant. Therefore, FDG-PET/CT has not yet been proven to be diagnostically superior to FDG-PET alone. More studies directly comparing FDG-PET/CT and FDG-PET alone are required to prove the assumed advantage of FDG-PET/CT over FDG-PET alone.
Whole-body MR imaging may be an alternative to FDG-PET/CT [43]; it does not require the operator to work with a potentially harmful radiotracer, the safety profile of MR contrast agents is favorable compared with that of iodinated contrast with CT [44], and the costs for a whole-body MR imaging examination (about 575 euros) are approximately two times less than that of a whole-body FDG-PET/CT examination (about 1,123 euros) [45]. In 1998, Eustace et al. [46] showed the potential of whole-body MR imaging in primary tumor detection in four patients with CUP, using a short-tau-inversion-recovery sequence. However, to our knowledge, no other reports on the diagnostic performance of whole-body MR imaging in unknown primary tumor detection have been published since then, while (both anatomical and functional whole-body) MR technology has continued to evolve [47, 48].
Identification of the primary tumor in patients with CUP enables accurate tumor staging, which allows optimizing treatment planning; this, in turn, may improve patient prognosis. On the other hand, it should be realized that FDG-PET/CT is an expensive examination, and false-positive FDG-PET/CT findings may result in unnecessary additional invasive diagnostic procedures, which have associated morbidities and costs [49]. In general, it appears that patients with CUP have a limited life expectancy, with a median survival of approximately 6–9 months [1, 2], but a median survival of 23 months has been reported for patients with CUP and an identified primary tumor subsequently treated with specific therapy [50]. Similarly, one study [51] reported that the 3-year survival rate for patients with cervical metastases and occult oropharyngeal primary tumors was 100% after treatment, while the patients with cervical metastases in which a primary tumor was not detected showed a survival rate of 58%. Only four studies included in this systematic review reported the therapeutic impact of FDG-PET/CT; in these four studies, FDG-PET/CT modified therapy in 18.2–60% of patients [15, 16, 20, 22]. Although one study [12] reported that the survival rate of CUP patients with at least one hypermetabolic lesion was significantly lower (P < 0.0279) than that of the remaining CUP patients, none of the included studies reported FDG-PET/CT-modified patient outcomes. Therefore, the additional value of FDG-PET/CT to patients with CUP and its cost-effectiveness should be further investigated; the currently presented data can be used to perform such an analysis.
In conclusion, although included studies were of moderate methodological quality and their results were heterogeneous, the results of this systematic review and meta-analysis indicate that FDG-PET/CT can be a useful method for unknown primary tumor detection. Future studies are required to prove the assumed advantage of FDG-PET/CT over FDG-PET alone and to further explore causes of heterogeneity.
References
Pavlidis N (2007) Forty years experience of treating cancer of unknown primary. Acta Oncol 46:592–601
Pavlidis N, Briasoulis E, Hainsworth J, Greco FA (2003) Diagnostic and therapeutic management of cancer of an unknown primary. Eur J Cancer 39:1990–2005
Rohren EM, Turkington TG, Coleman RE (2004) Clinical applications of PET in oncology. Radiology 231:305–332
Sève P, Billotey C, Broussolle C, Dumontet C, Mackey JR (2007) The role of 2-deoxy-2-[F-18]fluoro-D-glucose positron emission tomography in disseminated carcinoma of unknown primary site. Cancer 109:292–299
Rusthoven KE, Koshy M, Paulino AC (2004) The role of fluorodeoxyglucose positron emission tomography in cervical lymph node metastases from an unknown primary tumor. Cancer 101:2641–2649
Delgado-Bolton RC, Fernández-Pérez C, González-Maté A, Carreras JL (2003) Meta-analysis of the performance of 18F-FDG PET in primary tumor detection in unknown primary tumors. J Nucl Med 44:1301–1314
Blodgett TM, Meltzer CC, Townsend DW (2007) PET/CT: form and function. Radiology 242:360–385
Von Schulthess GK, Steinert HC, Hany TF (2006) Integrated PET/CT: current applications and future directions. Radiology 238:405–422
Whiting P, Rutjes AW, Reitsma JB, Bossuyt PM, Kleijnen J (2003) The development of QUADAS: a tool for the quality assessment of studies of diagnostic accuracy included in systematic reviews. BMC Med Res Methodol 3:25
Whiting PF, Weswood ME, Rutjes AW, Reitsma JB, Bossuyt PN, Kleijnen J (2006) Evaluation of QUADAS, a tool for the quality assessment of diagnostic accuracy studies. BMC Med Res Methodol 6:9
Zamora J, Abraira V, Muriel A, Khan K, Coomarasamy A (2006) Meta-DiSc: a software for meta-analysis of test accuracy data. BMC Med Res Methodol 6:31
Fencl P, Belohlavek O, Skopalova M, Jaruskova M, Kantorova I, Simonova K (2007) Prognostic and diagnostic accuracy of [18F]FDG-PET/CT in 190 patients with carcinoma of unknown primary. Eur J Nucl Med Mol Imaging 34:1783–1792
Nassenstein K, Veit-Haibach P, Stergar H et al (2007) Cervical lymph node metastases of unknown origin: primary tumor detection with whole-body positron emission tomography/computed tomography. Acta Radiol 23:1–8
Fleming AJ Jr, Smith SP Jr, Paul CM et al (2007) Impact of [18F]-2-fluorodeoxyglucose-positron emission tomography/computed tomography on previously untreated head and neck cancer patients. Laryngoscope 117:1173–1179
Bruna C, Journo A, Netter F et al (2007) On the interest of PET with 18F-FDG in the management of cancer of unknown primary (CUP). Med Nucl 31:242–249
Wartski M, Le Stanc E, Gontier E et al (2007) In search of an unknown primary tumor presenting with cervical metastases: performance of hybrid FDG-PET-CT. Nucl Med Commun 28:365–371
Paul SA, Stoeckli SJ, von Schulthess GK, Goerres GW (2007) FDG PET and PET/CT for the detection of the primary tumor in patients with cervical non-squamous cell carcinoma metastasis of an unknown primary. Eur Arch Otorhinolaryngol 264:189–195
Ambrosini V, Nanni C, Rubello D et al (2006) 18F-FDG PET/CT in the assessment of carcinoma of unknown primary origin. Radiol Med (Torino) 111:1146–1155
Schneider K, Hrasky A, Aschoff P, Bihl H, Hagen R (2006) Significance of PET and integrated PET/CT in the diagnostics of occult primary tumors. Laryngorhinootologie 85:819–823
Fakhry N, Barberet M, Lussato D et al (2006) Role of [18F]-FDG PET-CT in the management of the head and neck cancers. Bull Cancer 93:1017–1025
Schneider K, Aschoff P, Bihl H, Hagen R (2006) The integrated PET/CT: technological advance in diagnostics of head and neck recurrencies and CUP? Laryngorhinootologie 85:179–183
Pelosi E, Pennone M, Deandreis D, Douroukas A, Mancini M, Bisi G (2006) Role of whole body positron emission tomography/computed tomography scan with 18F-fluorodeoxyglucose in patients with biopsy proven tumor metastases from unknown primary site. Q J Nucl Med Mol Imaging 50:15–22
Maldonado A, Gonzalez Alenda J (2006) The anatomicometabolic PET-CT in carcinoma of unknown primary site. Oncologia (Spain) 29:93–94
Fakhry N, Jacob T, Paris J et al (2006) Contribution of 18-F-FDG PET for detection of head and neck carcinomas with an unknown primary tumor. Ann Otolaryngol Chir Cervicofac 123:17–25
Zanation AM, Sutton DK, Couch ME, Weissler MC, Shockley WW, Shores CG (2005) Use, accuracy, and implications for patient management of [18F]-2-fluorodeoxyglucose-positron emission/computerized tomography for head and neck tumors. Laryngoscope 115:1186–1190
Nanni C, Rubello D, Castellucci P et al (2005) Role of 18F-FDG PET-CT imaging for the detection of an unknown primary tumor: preliminary results in 21 patients. Eur J Nucl Med Mol Imaging 32:589–592
Freudenberg LS, Fischer M, Antoch G et al (2005) Dual modality of 18F-fluorodeoxyglucose-positron emission tomography/computed tomography in patients with cervical carcinoma of unknown primary. Med Princ Pract 14:155–160
Syed R, Bomanji JB, Nagabhushan N et al (2005) Impact of combined (18)F-FDG PET/CT in head and neck tumors. Br J Cancer 92:1046–1050
Gutzeit A, Antoch G, Kühl H et al (2005) Unknown primary tumors: detection with dual-modality PET/CT – initial experience. Radiology 234:227–234
Rödel R, Straehler-Pohl HJ, Palmedo H et al (2004) PET/CT imaging in head and neck tumors. Radiologe 44:1055–1059
Blaszyk H, Hartmann A, Bjornsson J (2007) Cancer of unknown primary origin: a decade of experience in a community-based hospital. Am J Surg 194:833–837
Al-Brahim N, Ross C, Carter B, Chorneyko K (2005) The value of postmortem examination in cases of metastasis of unknown origin-20-year retrospective data from a tertiary care center. Ann Diagn Pathol 9:77–80
Mayordomo JI, Guerra JM, Guijarro C et al (1993) Neoplasms of unknown primary site: a clinicopathological study of autopsied patients. tumori 79:321–324
Le Chevalier T, Cvitkovic E, Caille P et al (1988) Early metastatic cancer of unknown primary origin at presentation. A clinical study of 302 consecutive autopsied patients. Arch Intern Med 148:2035–2039
Werner JA, Dünne AA, Myers JN (2003) Functional anatomy of the lymphatic drainage system of the upper aerodigestive tract and its role in metastasis of squamous cell carcinoma. Head Neck 25:322–332
Mawlawi O, Pan T, Macapinlac HA (2006) PET/CT imaging techniques, considerations, and artifacts. J Thorac Imaging 21:99–110
Truong MT, Pan T, Erasmus JJ (2006) Pitfalls in integrated CT-PET of the thorax: implications in oncologic imaging. J Thorac Imaging 21:111–122
Blodgett TM, Fukui MB, Snyderman CH et al (2005) Combined PET-CT in the head and neck: part 1. Physiologic, altered physiologic, and artifactual FDG uptake. Radiographics 25:897–912
Lim HS, Yoon W, Chung TW et al (2007) FDG PET/CT for the detection and evaluation of breast diseases: usefulness and limitations. Radiographics 27(suppl 1):S197–S213
Fischbach F, Knollmann F, Griesshaber V, Freund T, Akkol E, Felix R (2003) Detection of pulmonary nodules by multislice computed tomography: improved detection rate with reduced slice thickness. Eur Radiol 13:2378–2383
Ay MR, Zaidi H (2006) Assessment of errors caused by X-ray scatter and use of contrast medium when using CT-based attenuation correction in PET. Eur J Nucl Med Mol Imaging 33:1301–1313
Antoch G, Freudenberg LS, Egelhof T et al (2002) Focal tracer uptake: a potential artifact in contrast-enhanced dual-modality PET/CT scans. J Nucl Med 43:1339–1342
Lauenstein TC, Semelka RC (2006) Emerging techniques: whole-body screening and staging with MRI. J Magn Reson Imaging 24:489–498
Runge VM (2001) Safety of magnetic resonance contrast media. Top Magn Reson Imaging 12:309–314
Plathow C, Walz M, Lichy MP et al (2008) Cost considerations for whole-body MRI and PET/CT as part of oncologic staging. Radiologe 48:384–396
Eustace S, Tello R, DeCarvalho V, Carey J, Melhem E, Yucel EK (1998) Whole body turbo STIR MRI in unknown primary tumor detection. J Magn Reson Imaging 8:751–753
Brauck K, Zenge MO, Vogt FM et al (2008) Feasibility of whole-body MR with T2- and T1-weighted real-time steady-state free precession sequences during continuous table movement to depict metastases. Radiology 246:910–916
Kwee TC, Takahara T, Ochiai R, Nievelstein RAJ, Luijten PR (2008) Diffusion-weighted whole-body imaging with background body signal suppression (DWIBS): features and potential applications in oncology. Eur Radiol 18:1937–1952
Schapira DV, Jarrett AR (1995) The need to consider survival, outcome, and expense when evaluating and treating patients with unknown primary carcinoma. Arch Intern Med 155:2050–2054
Raber MN, Faintuch J, Abbruzzese JL, Sumrall C, Frost P (1991) Continuous infusion 5-fluorouracil, etoposide and cis-diamminedichloroplatinum in patients with metastatic carcinoma of unknown primary origin. Ann Oncol 2:519–520
Haas I, Hoffmann TK, Engers R, Ganzer U (2002) Diagnostic strategies in cervical carcinoma of an unknown primary (CUP). Eur Arch Otorhinolaryngol 259:325–333
Open Access
This article is distributed under the terms of the Creative Commons Attribution Noncommercial License which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This is an open access article distributed under the terms of the Creative Commons Attribution Noncommercial License (https://creativecommons.org/licenses/by-nc/2.0), which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
About this article
Cite this article
Kwee, T.C., Kwee, R.M. Combined FDG-PET/CT for the detection of unknown primary tumors: systematic review and meta-analysis. Eur Radiol 19, 731–744 (2009). https://doi.org/10.1007/s00330-008-1194-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-008-1194-4