Abstract
Objectives
To evaluate the incidence of adverse drug reactions (ADRs), including hypersensitivity reactions (HSRs) and post-contrast acute kidney injury (PC-AKI), after intravenous (IV) administration of ioversol.
Materials and methods
A systematic literature search (1980–2021) of studies documenting IV use of ioversol and presence or absence of ADRs, HSRs, or PC-AKI was performed. Key information including patients’ characteristics, indication and dose of ioversol, safety outcome incidence, intensity and seriousness were extracted.
Results
Thirty-one studies (> 57,000 patients) were selected, including 4 pediatric studies. The incidence of ADRs in adults was reported in 12 studies from ioversol clinical development with a median (range) of 1.65% (0–33.3%), and 3 other studies with an incidence between 0.13 and 0.28%. The incidence of HSRs (reported in 2 studies) ranged from 0.20 to 0.66%, and acute events (4 studies) from 0.23 to 1.80%. Severe reactions were rare with a median (range) of 0 (0–4%), and none were reported among pediatric patients. The incidence of ADRs and HSRs with ioversol, especially those of severe intensity, was among the lowest in studies comparing different iodinated contrast media (ICM) of the same class. PC-AKI incidence was variable (1–42% in 5 studies); however, ioversol exposure per se did not increase the incidence.
Conclusions
When administered by the IV route, ioversol has a good safety profile comparable to that of other ICM within the same class, with a low incidence of severe/serious ADRs overall, and particularly HSRs. PC-AKI incidence does not seem to be increased compared to patients who did not receive ioversol. Further well-designed studies are warranted to confirm these results.
Key Points
• Ioversol has a good safety profile in adult and pediatric patients when IV administered.
• ADR and HSR incidence with ioversol, especially those of severe intensity, was among the lowest compared to other ICM.
• IV administration of ioversol per se did not increase PC-AKI incidence.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Iodine-based contrast media (ICM) are widely used in clinical practice for various X-ray-based modalities, and can be classified, according to their osmolality, into hyperosmolar CM (HOCM), low-osmolar CM (LOCM), and iso-osmolar CM (IOCM) [1]. They can be further subdivided into ionic and non-ionic CM, which do not dissociate into ions in water and are therefore lower in osmolality [2].
Ioversol (Optiray®, Guerbet) is a non-ionic, monomeric LOCM, with an osmolality between 502 and 792 mOsm/kg, depending on iodine concentration (240, 300, 320, or 350 mg I/mL).
Despite the generally good safety profile of ICM, adverse drug reactions (ADRs) may occur and can be life threatening. Among these reactions, there are hypersensitivity reactions (HSRs) [3]. Immediate (acute) HSRs occur within 1 h after ICM administration and may include urticaria, angioedema, bronchospasm, laryngeal edema, and anaphylactic shock. Non-immediate (delayed) HSRs, with symptoms occurring between 1 h and several days after ICM administration, commonly manifest as delayed urticaria and maculopapular exanthema, and rarely as severe cutaneous adverse reactions (SCARs) [3].
Post-contrast acute kidney injury (PC-AKI) is a complication that might occur after intravascular exposure to ICM. PC-AKI has been associated with excess morbidity and mortality [4,5,6], and chronic kidney disease (CKD) is the most well-known risk factor [7]. The risk of PC-AKI could increase from 5% at an estimated glomerular filtration rate (eGFR) ≥ 60 to 30% at an eGFR < 30 mL/min/1.73 m2 [8]. Several definitions of PC-AKI, based on serum creatinine (SCr) concentration, have been proposed by different initiatives, the European Society of Urogenital Radiology (ESUR) [9], the Acute Kidney Injury Network (AKIN) [10], and the Kidney Disease Improving Global Outcomes (KDIGO) being the most recent [11].
As the causal relationship between ICM exposure and the occurrence of AKI is often confounded by several patient- and procedure-related factors, the term PC-AKI is preferred for AKI associated with CM administration for studies lacking a control population [9]. Only when the ICM is demonstrated as the causative factor is the term contrast-induced acute kidney injury (CI-AKI) or contrast-induced nephropathy (CIN) appropriate.
To support radiologists in their clinical practice, we sought to perform this systematic analysis of literature on the incidence of ADRs, HSRs, and PC-AKI after intravenous (IV) administration of ioversol and to position the safety profile of ioversol among the different ICM. Complications after intra-arterial administration will be discussed in a future review.
Materials and methods
This systematic literature review was performed according to the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) guidelines [12].
Data sources and searches
A search of MEDLINE (PubMed) and EMBASE (Elsevier) references from January 1980 to May 2021 was performed using keywords related to adverse events usually associated with the use of ICM such as “allergic reaction,” “hypersensitivity,” “anaphylactic,” “nephrotoxicity,” and “kidney injury” (Appendix 1).
Study selection
Clinical studies documenting exposure to IV ioversol and the presence or absence of ADRs, and/or HSRs, and/or PC-AKI were included. Systematic or descriptive reviews, commentaries, letters, or case reports were excluded. Studies with less than 5 patients exposed to ioversol were excluded.
Study selection was conducted and reconciled between two independent authors. After a first screening step of all identified references, based on titles and abstracts, a full-text screening of potentially relevant publications was performed. Additional relevant publications were identified by cross-referencing.
Data extraction and study quality assessment
Key data extracted from selected articles were as follows: study design, patient characteristics, indication for which ioversol was used, number of patients exposed to ioversol and other ICM (if any) or number of administered doses, ICM dose, type of safety outcome and incidence, intensity [13] and seriousness if reported, and definition of PC-AKI (when applicable).
The methodological quality of the non-randomized studies was assessed using a modified Newcastle-Ottawa Scale (NOS) [14]. The score ranged from 0 to 8, based on 8 questions (one question excluded as not appropriate for safety outcomes) related to patient selection, comparability of cohorts, and outcomes assessment. Scores of 7–8 and 5–6 indicated high-quality and moderate-quality studies, respectively. The revised Cochrane Risk of Bias assessment tool for randomized trials (ROB 2) algorithm was used for randomized controlled trials (RCT) [15].
Results
Study selection
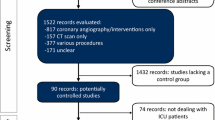
Among the 556 articles identified, 132 underwent a full-text screening and 4 articles were identified through citation tracking [16,17,18,19]. Finally, 31 articles were included: 16 related to the ioversol clinical development program [20,21,22,23,24,25,26,27,28,29,30,31,32,33,34,35] and 15 from other studies (Fig. 1). Twenty-five studies had a prospective design and 11 were RCT [20,21,22,23,24,25, 28, 29, 31, 36, 37]. Four studies were on pediatric patients [27, 30, 38, 39].
The NOS was applied to all non-RCT and one RCT (randomized for patient hydration and not for ICM allocation) [36], indicating high quality for 4 studies and medium quality for 18 studies. All RCTs had a low risk of bias, except one [37] where some concerns linked to a potential performance bias were raised as the study was not double blinded.
Twenty-nine studies indicated the number of patients exposed to ioversol (total of 57,837 patients, including 13,484 pediatric patients) while two studies indicated the number of administered doses of ioversol, with more than 1.5 million in An et al [17] and 20,958 doses in Morales et al [40] (Table 1).
In adult studies conducted during the clinical development of ioversol, the mean administered dose ranged between 50 and 176 mL, while sparse information was retrieved from the other adult studies. In pediatric patients, the injected dose was 1–3 mL/kg [27, 30, 38, 39].
Among the selected studies, 26 [17, 18, 20,21,22,23,24,25,26,27,28,29,30,31,32,33,34,35,36,37,38, 40, 42,43,44,45] documented the incidence of all ADRs or specifically HSRs (56,502 patients and 1,613,481 doses) and 5 studies [16, 19, 39, 46, 47] reported the incidence of PC-AKI (1335 patients). Contrast-enhanced CT was the main indication for which ioversol was used, followed by venography and urography. The mean age was 28–78 years old in adult studies and 5–10 years old in pediatric studies.
Twelve publications reported information on intensity of reactions (Table 2), with detailed information on the methodology of classification in 4 of them (Table 3). In addition, 4 publications reported information on seriousness of reactions (Table 2).
Adverse drug reactions and hypersensitivity reactions
The overall incidence of ADRs in adults was reported in 15 studies [17, 20,21,22,23,24,25,26, 28, 29, 32, 33, 35, 42, 43] with a median of 0.23%. In two studies where heat sensation was assessed in a specific questionnaire, a higher incidence of ADRs was reported (42–63%) [31, 34].
In 12 studies of ioversol clinical development (658 patients), the median incidence of ADRs was 1.65% (range: 0–33.3%), with 6 studies reporting no ADRs (Table 2). The highest incidence was reported in a pharmacokinetic study [21], where 6 of 18 patients reported ADRs, none of which was severe. Overall, most of the reported ADRs were minor and consisted of nausea, vomiting, and headache.
Three other studies reported incidences between 0.13 and 0.23% [17, 42, 43]. Vogl et al [42] reported ADRs in 0.28% of 10,836 patients, mainly urticaria (0.12%), nausea (0.10%), and erythema (0.06%). Four serious ADRs (0.037%) were reported, including 3 anaphylactoid reactions requiring hospitalization (0.028%).
An et al [17] reported an incidence of ADRs with ioversol of 0.23%, with urticaria (47.3%) and itching (43.9%) being the most frequent acute ADRs, and maculopapular rash (88%) the most frequent delayed ADR. The incidence of serious ADRs with ioversol was 0.01% (no deaths reported) (Table 2).
Chen et al [43] showed that ADRs were mainly evocative of HSRs, with an incidence of 0.13% for ioversol. Only one anaphylactic shock reaction (0.019%) and no case of laryngeal edema was reported with ioversol for 5261 patients exposed. The incidence of moderate and severe ADRs with ioversol was 0.02%, no deaths induced by ICM were reported, and all ADRs resolved.
The incidence of HSRs with ioversol was explicitly reported in two studies (0.2–0.66%) [40, 44] (Table 2). Morales et al [40] included patients with a previous history of HSRs to ICM. The incidence of HSRs was 0.2% with ioversol (mostly cutaneous symptoms [88.7%]), and severe HSRs represented 6.4% of all cases (no specific data with ioversol). In the study by Cha et al [44], HSR incidence was 0.66% and no severe HSRs were reported among 24,220 patients who received ioversol.
The incidence of acute ADRs was explicitly reported in two studies [18, 37], and in a third study, acute ADRs represented the majority of the reported ADRs (88.6%) [17]. The incidence was 0.23–1.8% [17, 18, 37]. In the study by Gomi et al [37], the acute ADR incidence was significantly lower with ioversol (1.8%) compared to iomeprol (3.9%) and iopromide (3.5%). Overall, 0.7% of the reported reactions required treatment and resolved, with no association with the type of ICM. No patient experienced life-threatening severe complications requiring immediate transfer to the emergency department.
In the study by Juchem et al [18], acute ADRs corresponding to two cases of vomiting (1%) were reported with ioversol, while the incidence of acute ADRs with meglumine diatrizoate was 12.5% (85% were anaphylactoid reactions). All acute ADRs were mild and patients recovered spontaneously.
Furthermore, in the study by Motosugi et al [36], acute allergic-like reaction incidence with ioversol was 1.8% and that of acute physiologic reactions was 1.1%, and none were severe.
Anaphylactoid reaction incidence in patients exposed to ioversol was reported in two studies ranging from 0.18% [42] to 2.5% [45]. Federle et al [45] reported more than a threefold higher incidence of anaphylactoid reactions with iothalamate compared to ioversol at both slow (8.3% vs. 2.0%, respectively) and fast (9.1% vs. 2.5%, respectively) injection rates.
The incidence of ADRs in pediatric patients exposed to ioversol for CT or urography was reported by Callahan et al [38], with a total of 12,494 pediatric patients and a mean (SD) age of 9.5 (5.9) years. Mild symptoms such as nausea, warm sensation, altered taste, and anxiety were not recorded as ADRs in this study. No ADRs were reported among 941 patients who underwent excretory urography. Only mild (0.38%) and moderate ADRs (0.08%) were reported. In patients aged ≤ 6 years old, only ADRs of mild intensity were reported. Two other pediatric studies from ioversol clinical development (mean age ≈ 5 years) reported ADRs in 3 of 65 patients (4.6%): metallic taste, nausea, and vomiting in two patients and not defined in the third patient [27, 30].
Studies with a comparison with other ICM
Ioversol was compared to a non-ionic, monomeric LOCM in 5 studies [20, 24, 25, 29, 31] during its clinical development, and no difference was shown regarding ADR incidence (Table 2). In 6 other studies [17, 36, 37, 40, 43, 44], the incidence of all ADRs and HSRs and severe/serious events (when reported) with ioversol was among the lowest (Table 2). In 3 studies [17, 43, 44], also including data with the IOCM iodixanol, the incidences of ADRs and HSRs with ioversol were 0.13–0.66% vs. 0.27–0.99% with iodixanol, and severe/serious events were 0.00–0.02% vs. 0.03–0.48%, respectively.
Five studies reported that the incidence of ADRs or HSRs was significantly different between ICM, with the highest incidences reported with iomeprol and/or iopromide [17, 37, 40, 43, 44]. Two studies compared the nature of ADRs between ICM. In Chen et al, rash was the predominant ADR reported with all ICM, but was more frequent with iodixanol. Facial swelling was more often reported with iodixanol compared with iopamidol and iopromide and was not reported with ioversol [43]. An et al analyzed the prevalence of ADRs by system organ class (SOC) and reported that “skin and appendages disorders” were more frequent with iodixanol, and “gastrointestinal system disorders” and “respiratory system disorders” more frequent with iomeprol [17].
Post-contrast acute kidney injury
PC-AKI prophylactic measures were described in two studies, and consisted of oral or IV hydration [16, 19]. A large heterogeneity in PC-AKI incidence was observed among the 5 studies (1–42%), due to heterogenous patient populations and differences in used PC-AKI definitions (Table 4).
In Louvel et al [46], one patient (1.1%) aged 82 years had a 25% increase in sCr (87 to 109 mmol/L) which rapidly improved. An increase > 10% in sCr was observed in 8 patients aged > 69 years and 4 patients aged < 60 years, with no significant difference between the two age groups. In Gomez et al [19] (98 diabetic patients using metformin), PC-AKI was observed for only one patient (1%) with an eGFR < 60 mL/min/1.73 m2 (incidence of 4.7% in this subpopulation), without clinical repercussion. During a 1-month follow-up period, no patient had alteration of renal function requiring medical care.
Ng et al [47] included two matched groups of patients who underwent CT with or without ioversol, and showed no difference in PC-AKI incidence (17%), sCr increase (0.25 and 0.11 mg/dL, respectively), need for hemodialysis (2% and 1%, respectively), and in-hospital mortality (17% and 21%, respectively). Moura et al [16] included a high-risk population of patients admitted to intensive care unit (ICU) with a length of stay > 3 days. The broader PC-AKI definition used in this study resulted in an incidence of 42%. Hemodialysis was needed for seven patients (12%) and deaths reported for 9 patients (6.5%).
Gilligan et al [39] included two matched groups of pediatric patients exposed to ioversol (aged 8 [6] years), and those who underwent abdominal US, and showed no difference in PC-AKI incidence (2.4% and 2.6%, respectively). In patients with an eGFR < 60 mL/min/1.73 m2, PC-AKI incidence was lower with ioversol (5.6% vs. 11.1%, respectively), although not statistically significant.
Discussion
This systematic literature review showed a large heterogeneity between studies regarding the way ADRs were collected and the type of ADRs reported. The median (range) incidence of ADRs with IV ioversol was 0.23% (0–33.3%). This variability is mainly emanating from ioversol clinical development studies, which included a low number of patients, and where heat and pain were specifically assessed in some studies. In the other studies, the incidence of ADRs in adults was low, independent of the type of ADR reported: 0.13–0.28% for all ADRs [17, 42, 43], 0.23–1.8% for acute ADRs [17, 18, 36, 37], and 0.2–0.66% for HSRs [40, 44]. In two studies, the relatively high incidence of events could be due to the systematic interview of patients [36] and a higher incidence of mild events (> 90% [36], 83% [44]). These incidences are comparable to those reported with other ICM. Indeed, two large retrospective studies with more than 246,000 patients who received IV non-ionic LOCM, reported an ADR incidence of 0.3% [49, 50].
The incidence of severe reactions to IV ioversol was low (0–0.02%) [18, 36, 38, 43, 44] and similar (if not lower) to what has been reported with other ICM (0.01–0.08%) [49,50,51,52]. Anaphylactic shock was reported in only one study, with a low incidence (0.019%) [43], consistent with a previous study using other non-ionic ICM (0.016%) [53]. Thus, the occurrence of severe events can be considered as rare with non-ionic ICM.
The risk of ADRs after using ICM in pediatric patients, and particularly life-threatening reactions, is low [54, 55]. Callahan et al reported a low incidence of ADRs (0.46%) and absence of severe events [38]. In one study, where non-ionic ICM were administered in 13,461 pediatric patients, the overall incidence of ADRs was 3.4%, and that of severe ADRs was 0.07% [55]. Another study reported an incidence of allergic-like reactions of 0.18% overall and 0.027% for severe reactions on 11,306 IV administrations [56]. This variability could be due to the different reporting (all ADRs or specific types, some mild symptoms not recorded as ADRs) [38]. ADR incidence was previously associated with the age of the patients with lower incidences observed in patients aged ≤ 10 years (0.22%) [50]. This could be linked to weak immune responses in pediatric patients compared to adults. Overall, it can be concluded that ioversol has a similar safety profile as other non-ionic ICM when IV administered to pediatric patients.
Several large retrospective studies investigated the safety profile of different ICM. Two studies using different non-ionic ICM reported that cutaneous and gastrointestinal disorders were the most frequent for mild events (51–69% and 12–14%, respectively) [49, 50]. In contrast, in a comparison of the safety profile of seven ICM, it was reported that skin (69.4%) and respiratory system disorders (8.9%) were the most frequent, followed by gastrointestinal disorders (5.7%). For ioversol, the proportion of gastrointestinal disorders and cardiovascular disorders was significantly higher than the general profile of LOCM (8% vs. 6% and 2% vs. 1%, respectively) and skin disorders significantly lower (65% vs. 70%) [57]. Despite some differences between LOCM, cutaneous and gastrointestinal manifestations are the most frequent and it could be concluded that ioversol has a similar safety profile to other LOCM.
PC-AKI incidence was highly variable, with the highest incidence reported in a critical care population with strong competing risk factors for AKI [16]. It is advised to use the lowest dose of ICM as possible in patients with diabetes and other co-morbidities and/or in patients with impaired renal function [7, 58, 59]. Consistent with what has been reported by Gomez et al [19], others reported a PC-AKI incidence of 1% in patients with normal renal function, which increased to 14% in those with severe renal impairment [60].
In the two studies comparing CT with ioversol to unenhanced CT or abdominal US, IV administration of ioversol per se did not increase the incidence of PC-AKI in adult and pediatric patients [39, 47]. Others reported that IV ICM administration for CT was not associated with an increased risk of PC-AKI [60], and large retrospective studies using propensity score matching suggested a lower incidence of PC-AKI than previously estimated [61]. In studies comparing the safety profile of iodixanol to that of other non-ionic LOCM, urinary system disorders were more frequently reported than with non-ionic LOCM [57]. However, this could be due to iodixanol being used more frequently in high-risk patients with underlying renal diseases [17]. The proportion of urinary system disorders with ioversol was comparable to the general profile of LOCM, suggesting a similar safety profile with regard to PC-AKI [17, 57]. In procedures involving IV administration of ICM, several meta-analyses showed that iodixanol was not associated with a reduction in PC-AKI compared to non-ionic LOCM [62,63,64].
In conclusion, the safety profile of ioversol, by IV route, is good and comparable to that of other non-ionic LOCM, with a low incidence of ADRs overall and particularly severe/serious ADRs, in adult and pediatric patients. PC-AKI incidence following IV administration of ioversol was not higher than in patients unexposed to ICM. Further well-designed studies are warranted in order to confirm these results.
Abbreviations
- ADR:
-
Adverse drug reaction
- AKIN:
-
Acute kidney injury network
- CIN:
-
Contrast-induced nephropathy
- CKD:
-
Chronic kidney disease
- eGFR:
-
Estimated glomerular filtration rate
- ESUR:
-
European Society of Urogenital Radiology
- HOCM:
-
Hyperosmolar contrast medium
- HSR:
-
Hypersensitivity reaction
- IA:
-
Intra-arterial
- ICM:
-
Iodinated contrast media
- ICU:
-
Intensive care unit
- IOCM:
-
Iso-osmolar contrast medium
- IV:
-
Intravenous
- KDIGO:
-
Kidney Disease Improving Global Outcomes
- LOCM:
-
Low-osmolar contrast medium
- NOS:
-
Newcastle-Ottawa Scale
- PC-AKI:
-
Post-contrast acute kidney injury
- PRISMA:
-
Preferred Reporting Items for Systematic Reviews and Meta-analyses
- RCT:
-
Randomized controlled trial
- ROB 2:
-
The revised Cochrane risk of bias assessment tool
- sCr:
-
Serum creatinine
- SOC:
-
System organ class
References
Chartrand-Lefebvre C, White CS, Bhalla S et al (2011) Comparison of the effect of low- and iso-osmolar contrast agents on heart rate during chest CT angiography: results of a prospective randomized multicenter study. Radiology 258:930–937
Aspelin P, Bellin MF, Jakobsen J Å., Webb JAW (2009) Classification and terminology. In: Thomsen HS, Webb JAW (eds) Contrast media: safety issues and ESUR guidelines. Springer, Berlin, Heidelberg, pp 3–9
Demoly P, Adkinson NF, Brockow K et al (2014) International consensus on drug allergy. Allergy 69:420–437
Rao QA, Newhouse JH (2006) Risk of nephropathy after intravenous administration of contrast material: a critical literature analysis. Radiology 239:392–397
Kooiman J, Pasha SM, Zondag W et al (2012) Meta-analysis: serum creatinine changes following contrast enhanced CT imaging. Eur J Radiol 81:2554–2561
From AM, Bartholmai BJ, Williams AW et al (2008) Mortality associated with nephropathy after radiographic contrast exposure. Mayo Clin Proc 83:1095–1100
Azzalini L, Kalra S (2020) Contrast-induced acute kidney injury-definitions, epidemiology, and implications. Interv Cardiol Clin 9:299–309
Davenport MS, Perazella MA, Yee J et al (2020) Use of intravenous iodinated contrast media in patients with kidney disease: consensus statements from the American College of Radiology and the National Kidney Foundation. Kidney Med 2:85–93
van der Molen AJ, Reimer P, Dekkers IA et al (2018) Post-contrast acute kidney injury - part 1: definition, clinical features, incidence, role of contrast medium and risk factors : recommendations for updated ESUR Contrast Medium Safety Committee guidelines. Eur Radiol 28:2845–2855
Mehta RL, Kellum JA, Shah SV et al (2007) Acute Kidney Injury Network: report of an initiative to improve outcomes in acute kidney injury. Crit Care 11:R31
Khwaja A (2012) KDIGO clinical practice guidelines for acute kidney injury. Nephron Clin Pract 120:c179–c184
PRISMA-P Group, Moher D, Shamseer L, et al (2015) Preferred reporting items for systematic review and meta-analysis protocols (PRISMA-P) 2015 statement. Syst Rev 4:1
American College of Radiology (2021) ACR manual on contrast media. American College of Radiology, Reston, Va
GA Wells, B Shea, D O’Connell, et al The Newcastle-Ottawa Scale (NOS) for assessing the quality if nonrandomized studies in meta-analyses. http://www.ohri.ca/programs/clinical_epidemiology/oxford.asp. Accessed 19 Oct 2020
Sterne JAC, Savović J, Page MJ et al (2019) RoB 2: a revised tool for assessing risk of bias in randomised trials. BMJ 366:l4898
Moura ELB de, Amorim FF, Huang W, Maia M de O (2017) Contrast-induced acute kidney injury: the importance of diagnostic criteria for establishing prevalence and prognosis in the intensive care unit. Rev Bras Ter Intensiva 29:303–309
An J, Jung H, Kwon OY et al (2019) Differences in adverse reactions among iodinated contrast media: analysis of the KAERS database. J Allergy Clin Immunol 7:2205–2211
Juchem BC, Dall’Agnol CM (2007) Immediate adverse reactions to intravenous iodinated contrast media in computed tomography. Rev Lat Am Enfermagem 15:78–83
Gómez Herrero H, De Arriba VC, Buldain Parra M, Arraiza Sarasa M (2013) Nephrotoxicity due to iodine contrasts in computerized tomography studies of diabetic outpatients on metformin. An Sist Sanit Navar 36:197–201
Wilkins RA, Spinks BC (1990) A double blind clinical study comparing the safety, tolerance and efficacy of ioversol 240 and iohexol 240 (omnipaque 240) in ascending venography. Clin Radiol 41:268–271
Wilkins RA, Whittington JR, Brigden GS, Lahiri A, Heber ME, Hughes LO (1989) Safety and pharmacokinetics of ioversol in healthy volunteers. Invest Radiol 24:781–788
Wilson AJ, Murphy WA, Destouet JM et al (1989) Ascending lower limb phlebography: comparison of ioversol and iothalamate meglumine. Can Assoc Radiol J 40:142–144
Scott H, Palmer FJ (1990) Ioversol in ascending phlebography--a clinical trial. Australas Radiol 34:44–46
Colthurst JR, Chan O, Creagh M et al (1990) A double-blind clinical study comparing the safety, tolerance and efficacy of ioversol and iohexol in intravenous urography. Clin Radiol 42:174–176
Voegeli E, Woessmer B (1992) Evaluation of clinical tolerability and diagnostic efficacy of ioversol 350 in UIV. A randomized double-blind study of ioversol 350 versus iohexol 350. Ann Radiol 35:293–296
Lemaître L (1992) Evaluation of efficacy and tolerability of ioversol in intravenous urography. Ann Radiol 35:303–306
Montagne J, Adamsbaum C (1992) Ioversol 300: clinical study in pediatric intravenous urography. Ann Radiol 35:307–310
McClennan BL, Heiken JP, Lee JK, James MA (1989) Computed body tomography with a new nonionic contrast agent. Comparison of ioversol with sodium/meglumine diatrizoate. Invest Radiol 24(Suppl 1):S35–S38
Rieser R, Beinborn W, Ney N (1992) A double-blind comparative study on the contrast quality, tolerance and safety of ioversol 300 versus iohexol 300 in central venous angiography (C.V. DSA). Ann Radiol 35:311–314
Panuel M, Devred P, Faure F, Bourlière-Najean B, Ternier F, Le Bail C (1992) Evaluation of diagnostic efficacy and clinical tolerability of ioversol in “whole body” computed tomography in children. A non comparative phase III trial. Ann Radiol 35:280–283
Chagnaud C, Moulin G, Delannoy L et al (1992) Ioversol 300 and iopamidol 300 in “whole body” computed tomography. A double-blind clinical trial. Ann Radiol 35:276–279
Kopecky KK, Becker GJ, Conces DJ (1989) Ioversol 320: a new nonionic, water-soluble contrast medium for body computed tomography clinical trial. Invest Radiol 24(Suppl 1):S33–S34
Sartor K, Gado MH, Hodges FJ (1989) Clinical experience with ioversol 320 in cranial computed tomographic scanning. Invest Radiol 24(Suppl 1):S29–S32
Gillard C, Tatu L, Menegazzo D, Bonneville JF (1992) Clinical tolerability of ioversol 300 in brain computed tomography. Ann Radiol 35:284–287
Théron J, Paugam JP, Courthéoux P (1991) Ioversol 350: clinical experience in skull x-ray computed tomography. Ann Radiol 34:413–417
Motosugi U, Ichikawa T, Sano K, Onishi H (2016) Acute adverse reactions to nonionic iodinated contrast media for CT: prospective randomized evaluation of the effects of dehydration, oral rehydration, and patient risk factors. Am J Roentgenol 207:931–938
Gomi T, Nagamoto M, Hasegawa M et al (2010) Are there any differences in acute adverse reactions among five low-osmolar non-ionic iodinated contrast media? Eur Radiol 20:1631–1635
Callahan MJ, Poznauskis L, Zurakowski D, Taylor GA (2009) Nonionic iodinated intravenous contrast material-related reactions: incidence in large urban children’s hospital--retrospective analysis of data in 12,494 patients. Radiology 250:674–681
Gilligan LA, Davenport MS, Trout AT et al (2020) Risk of acute kidney injury following contrast-enhanced CT in hospitalized pediatric patients: a propensity score analysis. Radiology 294:548–556
Morales-Cabeza C, Roa-Medellín D, Torrado I et al (2017) Immediate reactions to iodinated contrast media. Ann Allergy Asthma Immunol 119:553–557
McClennan BL (1989) Clinical summary of initial intravenous administration of ioversol. Invest Radiol 24 (Suppl 1):S43–S46
Vogl TJ, Wessling J, Buerke B (2012) An observational study to evaluate the efficiency and safety of ioversol pre-filled syringes compared with ioversol bottles in contrast-enhanced examinations. Acta Radiol 53:914–920
Chen Q, Zhao X, Wang X et al (2017) Retrospective analysis of non-laboratory-based adverse drug reactions induced by intravenous radiocontrast agents in a Joint Commission International-accredited academic medical center hospital in China. Ther Clin Risk Manag 13:565–573
Cha MJ, Kang DY, Lee W et al (2019) Hypersensitivity reactions to iodinated contrast media: a multicenter study of 196 081 patients. Radiology 293:117–124
Federle MP, Willis LL, Swanson DP (1998) Ionic versus nonionic contrast media: a prospective study of the effect of rapid bolus injection on nausea and anaphylactoid reactions. J Comput Assist Tomogr 22:341–345
Louvel JP, Primard E, Henry J et al (1996) Effects of the low-osmolality contrast medium ioversol (Optiray) on renal function in a geriatric population. Acta Radiol 37:950–953
Ng CS, Shaw AD, Bell CS, Samuels JA (2010) Effect of IV contrast medium on renal function in oncologic patients undergoing CT in ICU. Am J Roentgenol 195:414–422
Brown SGA (2004) Clinical features and severity grading of anaphylaxis. J Allergy Clin Immunol 114:371–376
Zhang B, Dong Y, Liang L et al (2016) The incidence, classification, and management of acute adverse reactions to the low-osmolar iodinated contrast media isovue and ultravist in contrast-enhanced computed tomography scanning. Medicine 95:e3170
Li X, Chen J, Zhang L et al (2015) Clinical observation of the adverse drug reactions caused by non-ionic iodinated contrast media: results from 109,255 cases who underwent enhanced CT examination in Chongqing, China. Br J Radiol 88:20140491
Palkowitsch PK, Bostelmann S, Lengsfeld P (2014) Safety and tolerability of iopromide intravascular use: a pooled analysis of three non-interventional studies in 132,012 patients. Acta Radiol 55:707–714
Honda T, Kuriyama K, Kiso K et al (2020) Incidence rate of severe adverse drug reactions to nonionic contrast media at the National Hospital Organization Osaka National Hospital. Allergo J Int 29:240–244
Kim MH, Lee SY, Lee SE, et al (2014) Anaphylaxis to iodinated contrast media: clinical characteristics related with development of anaphylactic shock. PLoS One 16;9(6):e100154
Gaca AM, Frush DP, Hohenhaus SM et al (2007) Enhancing pediatric safety: using simulation to assess radiology resident preparedness for anaphylaxis from intravenous contrast media. Radiology 245:236–244
Katayama H, Yamaguchi K, Kozuka T et al (1990) Adverse reactions to ionic and nonionic contrast media. A report from the Japanese Committee on the Safety of Contrast Media. Radiology 175:621–628
Dillman JR, Strouse PJ, Ellis JH et al (2007) Incidence and severity of acute allergic-like reactions to i.v. nonionic iodinated contrast material in children. Am J Roentgenol 188:1643–1647
Seong JM, Choi NK, Lee J et al (2013) Comparison of the safety of seven iodinated contrast media. J Korean Med Sci 28:1703–1710
Mehran R, Dangas GD, Weisbord SD (2019) Contrast-associated acute kidney injury. N Engl J Med 380:2146–2155
Wang Y, Liu K, Xie X, Song B (2021) Contrast-associated acute kidney injury: an update of risk factors, risk factor scores, and preventive measures. Clin Imaging 69:354–362
McDonald JS, McDonald RJ, Carter RE et al (2014) Risk of intravenous contrast material–mediated acute kidney injury: a propensity score–matched study stratified by baseline-estimated glomerular filtration rate. Radiology 271:65–73
Dekkers IA, van der Molen AJ (2018) Propensity score matching as a substitute for randomized controlled trials on acute kidney injury after contrast media administration: a systematic review. Am J Roentgenol 211:822–826
Eng J, Wilson RF, Subramaniam RM et al (2016) Comparative effect of contrast media type on the incidence of contrast-induced nephropathy: a systematic review and meta-analysis. Ann Intern Med 164:417–424
Dong M, Jiao Z, Liu T et al (2012) Effect of administration route on the renal safety of contrast agents: a meta-analysis of randomized controlled trials. J Nephrol 25:290–301
McCullough PA, Brown JR (2011) Effects of intra-arterial and intravenous iso-osmolar contrast medium (iodixanol) on the risk of contrast-induced acute kidney injury: A meta-analysis. Cardiorenal Med 1:220–234
Funding
The authors state that this work has not received any funding.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Guarantor
The scientific guarantor of this publication is Aart J. van der Molen.
Conflict of Interest
Ibrahim Bedioune and Elisabeth Darmon-Kern are medical writers employed by Guerbet.
Statistics and Biometry
No complex statistical methods were necessary for this paper.
Informed Consent
Written informed consent was not required for this study because only published data were used.
Ethical Approval
Institutional Review Board approval was not required because only published data were used.
Study subjects or cohorts overlap
Studies with duplicate data were excluded from this systematic review.
Methodology
• Multicenter study
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Part 2 of this article can be found under https://doi.org/10.1007/s00330-022-08637-2.
Supplementary Information
ESM 1
(DOCX 20 kb)
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
van der Molen, A.J., Dekkers, I.A., Bedioune, I. et al. A systematic review of the incidence of hypersensitivity reactions and post-contrast acute kidney injury after ioversol in more than 57,000 patients: part 1—intravenous administration. Eur Radiol 32, 5532–5545 (2022). https://doi.org/10.1007/s00330-022-08636-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-022-08636-3