Abstract
Objective
Cranial computed tomography (CT) scan is the most widely used tool to rule out intracranial lesions after pediatric traumatic brain injury (TBI). However, in pediatric population, the radiation exposure can lead to an increased risk of hematological and brain neoplasm. Defined in 2019 National Institute for Health and Care Excellence (NICE) guidelines as “troponins for the brain”, serum biomarkers measurements, particularly S100B, have progressively emerged as a supplementary tool in the management of TBI thanks to their capacity to predict intracranial post-traumatic lesions.
Methods
This systematic review was conducted following the PRISMA protocol (preferred reporting items for systematic reviews and meta-analyses). No chronological limits of study publications were included. Studies reporting data from children with TBI undergoing serum S100B measurement and computed tomography (CT) scans were included.
Results
Of 380 articles screened, 10 studies met the inclusion criteria. Patients admitted with mild-TBI in the Emergency Department (ED) were 1325 (80.25%). The overall pooled sensitivity and specificity were 98% (95% CI, 92–99%) and 45% (95% CI, 29–63%), respectively. The meta-analysis revealed a high negative predictive value (NVP) (99%; 95% CI, 94–100%) and a low positive predictive value (PPV) (41%; 95% CI, 16–79%). Area under the curve (AUC) was 76% (95% CI, 65–85%). The overall pooled negative predictive value (NPV) was 99% (95% CI, 99–100%).
Conclusions
The measurement of serum S100B in the diagnostic workflow of mTBI could help informed decision-making in the ED setting, potentially safely reducing the use of CT scan in the pediatric population. The high sensitivity and excellent negative predictive values look promising and seem to be close to the values found in adults. Despite this, it must be pointed out the high heterogeneity (> 90%) found among studies. In order for S100B to be regularly introduced in the pediatric workflow for TBI, it is important to conduct further studies to obtain cut-off levels based on pediatric reference intervals.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
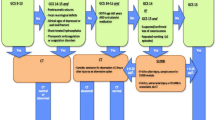
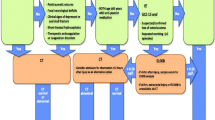
Traumatic brain injury (TBI) is one of the most common encountered pathologies in the emergency department (ED), with an estimated annual incidence of 475.000 cases in children younger than 14 years in the USA [1]. Glasgow Coma Scale (GCS) and Pediatric Glasgow Coma Scale represent reliable clinical tools to rapidly assess the impairment of consciousness level and therefore TBI severity in an emergency setting [2]. In recent years, the incidence of mild TBI (mTBI) has progressively increased [3].
Nonetheless, imaging exams such as cranial computed tomography (CT) are essential to safely rule out potential intracranial complication after TBI [4]. In this context, radiation exposure after cranial CT scans in pediatric population represent a relevant concern due to the increased risk of hematological and brain neoplasm [5, 6]. Consequently, the use of cranial CT scan in children with mTBI must be carefully rationed [7].
Firstly defined in 2019 National Institute for Health and Care Excellence (NICE) guidelines as “troponins for the brain” [8], specific serum biomarkers measurements have progressively emerged as a supplementary tool in the management of TBI due to their capacity to predict intracranial post-traumatic lesions [9]. Among the multitude of biomarkers proposed, S100b is one of the most assessed and it is potentially one of the bio-markers to be evaluated in pediatric TBI cases [10].
In a recent meta-analysis, Oris et al. [11] reported a potential 30% reduction of cranial CT scans for the diagnosis of mTBI when supplemented by serum S100B measurements [11]. Nonetheless, serum S100B concentration is highly variable and strictly depends on the age of the patient, making it difficult to clearly identify a specific serum cut-off [12].
To this purpose, this meta-analysis aims to evaluate the efficacy of serum S100B levels in the identification of intracranial lesion after TBI in pediatric population, defining a potential cut-off to reduce unnecessary cranial CT scan in an emergency setting.
Materials and methods
Literature search
This systematic review was conducted following the PRISMA protocol (preferred reporting items for systematic reviews and meta-analyses) [13]. Potentially relevant literature was retrieved from PubMed/MEDLINE, Embase, and the Cochrane Library. The final search was completed on the 21st of December 2023. A detailed search strategy is reported in Supplementary Material 1. Word variations and expanded medical subject headings were searched for whenever feasible.
Inclusion and exclusion criteria
Articles written in English and involving human subjects were eligible for inclusion. No chronological limits of study publications were included. Prospective and retrospective clinical studies, reports of case series with at least five patients per group and studies enrolling pediatric patients (age < 18 years) presenting to the ED with a history of possible brain injury and undergoing CT scan or inpatient stay, with at least one quantitative blood measurement of S100B on admission and studies that included the possibility of extracting of biomarker sensitivity and specificity were eligible for inclusion. We included studies containing mixed populations; that is, participants with mild, moderate, and severe TBI (according to GCS). We excluded studies using non-quantitative methods to assess biomarker concentrations and studies that analyzed quantitative measurements other than blood (e.g., saliva or urine). Meta-analyses, case reports, or studies with less than five patients per group, cadaver studies, laboratory, and animal studies were excluded.
Screening and full-text review
Title and abstract screening, full-text review, and data extraction were undertaken in parallel by two reviewers (A.G. and A.M.). Any disagreements at any stage were resolved by discussion and consensus. Persistent disagreements were resolved with the involvement of a third reviewer (E.L.B.). The process was carried out using Rayyan, a tool for undertaking literature and systematic reviews [14].
Data extraction
The names of the first authors, type of study, publication date, sample size, patient characteristics (age, sex ratio, GCS score), laboratory aspects of S100B (type of assay, concentrations, reference ranges, sampling information, time between TBI and blood sampling), comparison of CT scan versus S100B blood values (negative predictive value (NPV), positive predictive value (PPV), sensitivity, specificity, area under the curve [AUC], and cutoffs), and eventual clinical evolution (CE) were extracted from the studies and collected into a table format.
Quality assessment
The Cochrane risk-of-bias tool for nonrandomized studies of interventions (ROBINS-I tool) was used for risk-of-bias assessment of the included studies [15]. This was performed by two authors (A.G., and A.M.).
Statistical analysis
Sensitivity, specificity, NPV, PPV, and AUC were meta-analyzed in R using the Meta and Metafor packages (Version 4.6–0). Missing confidence intervals (CI) were imputed as follows: the lower limit was set as the minimum between sensitivity and 1, while the upper limit was set as the maximum between sensitivity and 0, multiplied by 100. Baseline characteristics were summarized for each study sample and reported as mean (SD) and number (percent) for continuous and categorical variables, respectively, or median (minimum, maximum). The meta-analysis results were visualized using a forest plot with a random effect model. Statistical heterogeneity among studies was assessed by examining forest plots, 95% CIs, and I2. A likelihood ratio scattergram was plotted to visualize likelihood ratios across studies, facilitating comparison and interpretation of test performance in terms of sensitivity and specificity. Finally, a funnel plot was utilized to investigate potential publication bias. Type I error was fixed at α = 0.05.
Results
Literature search
Our search strategy identified a total of 380 citations. An initial screening to remove duplicate studies produced 207 unique articles. After excluding review articles, experimental studies, meta-analyses, animal studies, and studies with adults, 105 articles were found to be relevant. Finally, 11 studies meeting the inclusion and exclusion criteria, reporting on 1675 patients were identified and included in the qualitative synthesis [16,17,18,19,20,21,22,23,24,25], with 10 studies analyzing 1651 patients available for inclusion in the quantitative synthesis (Fig. 1, PRISMA flowchart). The study of Yeung was excluded for quantitative synthesis due to the measurement of a salivary biomarker rather than serum [26].
Study characteristics and quality
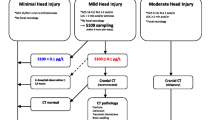
Table 1 provides an overview of the included studies and their quality ratings according to risk-of-bias assessment (ROBINS-I tool). All 10 (100%) studies included in the quantitative analysis were prospective. The majority (80%) of the studies were single-center studies. Patients with mTBI (GCS 15–14-13) at admission to the ED were 1451 (88.47%). In the clinical evaluation at admission, Hallen et al. did not specify the GCS. In most of the cases, S100B serum values were measured within a maximum time-frame of 6 h [16, 17, 21,22,23,24]; two authors [19, 20] and just one author [25] recommended a maximum time of 3 and 24 h, respectively. Cutoff values for S100B differed between the studies (concentration threshold 0.006–0.869 µg/L) and researchers interpreted S100B concentrations on the basis of reference ranges adapted for age in only two articles [20, 24]. Cranial CT was the reference examination for all studies included in the quantitative analysis.
Specificity was relatively heterogeneous, in some cases weak (25.56 to 100%). Optimal sensitivity was measured in most cases, but in one study researchers obtained a sensitivity of 48% [25]. Areas under ROC curves ranged between 0.51 and 0.985. PPV and NPV showed relatively heterogeneous results, ranging from 0.11 to 1.0 for PPV and from 0.91 to 1.0 for NPV.
Quantitative results
After removing Yeung et al. study [26], the meta-analysis revealed a sensitivity of 98% (95% CI, 92–99%) and specificity of 45% (95% CI, 29–63%), resulting in a high NVP (99%; 95% CI, 94–100%) and a low PPV (41%; 95% CI, 16–79%). AUC was 76% (95% CI, 65–85%) (Figs. 2 and 3). There was a significant heterogeneity (> 90%) among the studies for all metrics except the NVP (73%). The likelihood ratio scattergram confirmed the heterogeneity of the studies for both sensitivity and specificity (Fig. 4A).
Publication bias
Funnel plots of the investigated outcomes can be found in the Fig. 4B, C.
Discussion
Management of TBI after head trauma in pediatric population represents a challenging situation for the ED clinician. The decision to acquire a cranial CT balances the incidence of detecting an intracranial injury with the risks of unnecessary radiation exposure [7, 27]. Approximately 90% of head CT scan performed for mTBI in pediatric patients did not demonstrate neurological injury [28]. Between 1996 and 2005, the overall use of CT imaging in the ED setting has doubled in children under 5 years and tripled in children between 5 and 14 years, with an estimated 50% increase of head CT [29]. In this context, in the last years, the Pediatric Emergency Care Applied Research Network (PECARN) has produced several studies focused on this specific topic [7]. In 2009, Kuppermann et al. conducted a multicenter prospective cohort study with the largest pediatric population with mTBI, with the primary endpoint to identify patients with very low risk of clinically relevant TBI in which the CT exam could possibly be unnecessary [7]. In 2011, the group of Pandor et al. proposed a methodological algorithm for the management of mTBI in pediatric population [30].
Specifically, Miglioretti et al. in 2017 analyzed the correlation between the lifetime risk of leukemia and a history of previous cranioencephalic CT scans, showing an estimate 1 case per 10,000 CT scans in patients younger than 5 years, and 0.5 cases per 10,000 CT scans in the population between 10 and 14 years [29].
While cranial CT scan remains a standard diagnostic tool, rapid sequence MRI without sedation has progressively been used in the ED, taking advantage of its ability to rule out a wider spectrum of intracranial pathologies compared to conventional CT scan [31, 32]. Nevertheless, the limited availability of MRI compared to CT still represents a major restriction regarding its application in the diagnostic algorithm of TBI in the ED setting.
In this context, the challenges associated with accurate diagnostic and therapeutic workflow for TBI have progressively shed light on the search for blood and fluid biomarkers as potential complementary diagnostic tools, aiming to ultimately optimize neuroimaging management in the ED and improve cost savings for healthcare systems [33]. Despite the promising outlook and multiple research studies, systematic adoption of biomarkers into clinical practice remains limited. Standardization of biomarker assays, validation across heterogeneous patient populations, and systematic integration of biomarkers into existing clinical workflows still represent relevant concerns. Nevertheless, there has been a considerable contribution to the evidence supporting the relevance of biomarkers in TBI diagnostic workflow, with multiple attempts to identify accurate and reliable biomarker suitable for an ED setting. To date, the most evidence for the use of biomarkers as complementary tools for diagnosis and prognosis in pediatric patients with TBI exists for S100 calcium-binding protein B (S100B), glial fibrillary acidic protein (GFAP), ubiquitin C-terminal hydrolase (UCH-L1), and osteopontin (OPN) [25]. Overall, serum and saliva S100B levels have emerged as a sensible and reliable biomarker for brain injury after head trauma in children [23, 34].
S100B is a calcium-binding protein highly expressed by astroglial and neuronal cells. Structurally, is a small dimeric protein with a molecular weight of approximately 21 kDa [20]. It is released from brain cells and enters the systemic circulation probably due to increased permeability of the blood–brain barrier, subsequently being excreted within approximately 6 h from injury [35, 36]. S100B is also expressed in adipocytes, chondrocytes, melanocytes, and epidermal cells to a lesser extent, potentially leading to increased systemic levels even in the absence of brain injury [17], although minor peripheral injuries typically do not cause significant rises in S100B serum levels [37]. Several studies have shown an inverse correlation between S100B serum levels and age, with levels decreasing from infancy to adolescence, particularly during the first 2–3 years of life. This trend can be explained by multiple factors, such as increased permeability of the blood–brain barrier, accelerated protein turnover in neuronal cells, and diminished renal excretion of S100B. All these factors must be considered when analyzing potential reference intervals of serum S100B in a pediatric population, especially during the first years of life [20]. In addition, considering its relatively short half-life ranging from 20 to 120 min, S100B can be used as a biomarker for traumatic brain injury when measured within a timeframe that aligns with its clearance kinetics [16, 20]. Typically, it is most useful for assessing acute injury within 3 h of the traumatic event.
The clinical use of S100B as a biomarker for mTBI was firstly reported in 1995 [38]. Since then, several studies demonstrated its capacity to reduce the number of CT scan conducted in patients with mTBI, especially in adults [39]. Integration of S100B concentrations and clinical decision in adult patients with mTBI can reduce the number of negative CT scan by up to 30% [30, 40]. On this basis, in 2013 the clinical use of S100B in the management of mTBI in adult patients has been firstly introduced in the Scandinavian Guidelines [41].
Nonetheless, the role of S100B in clinical practice for pediatric TBI is still debated. In 2015, another major work was carried out on the Scandinavian Guidelines for the Management of Mild and Moderate Head Trauma in Pediatric Children by Astrand et al. [42]. In the Scandinavian Guidelines for initial management of minor and moderate head trauma in children, Astrand et al. did not include serum S100B evaluation considering the limited number of available studies in the literature, and the high heterogeneity of reference levels related to the age of the patients. In this context, Bouvier et al. demonstrated that serum concentration of S100B in healthy children is higher in the first 3 years of life [43]. As emerges by our analysis, only two studies interpreted serum S100B concentrations references considering also the age of the patients [20, 24].
In this meta-analysis, the high sensitivity and excellent negative predictive values look promising and seem to be close to the values found in adults [36]. Despite this, we must point out the high heterogeneity (> 90%) among the studies for all metrics except the NVP (73%) (Fig. 3). Although it has high sensitivity, using only serum S100B concentration for initial mTBI screening is not ideal due to the risk of false-negative results. We revealed an important deviation with one study [25]. Finally, the variability of protein concentration by sampling time and particularly by age is an important limitation [44].
In this context, a randomized, multicenter, open-label, prospective, interventional study (nine centers) was recently conducted in France in which researchers used a stepped wedge cluster design with two arms (“S100B management” intervention group and “conventional management” control group) [45]. In this randomized clinical trial including a cohort of 2078 children, S100B biomonitoring produced a reduction in the number of CT scans and in-hospital observation. However, the difference in CT scans performed between the control group and the S100B biomonitoring group was not statistically significant (P = 0.44). Another interesting result of this study is that Bouvier et al. found a relative risk of 0.49 (95% CI, 0.30–0.77) in the post hoc analysis for CT scans and 0.46 (95% CI, 0.39–0.51) in the modified intention-to-treat analysis for in-hospital observations.
From the available data, there are no major studies in the literature in which S100B identified specific types of intracranial lesions. In some cases, subdural hematomas have been classified slightly more frequently as false negatives [46, 47]. We speculate that this may be due to the location and/or extent of the brain lesion and the pathoanotomic and neurovascular characteristics of the different lesions that cause altered or delayed leakage of S100B into the circulation. On the other hand, Bouvier et al. found that the S100B identified patients with poor clinical evolution (CE) with a sensitivity of 100% (95% CI, 84–100) and specificity of 36% (95% CI, 31–41) [20]. Specifically, poor CE was defined by the following clinical symptoms: vomiting, facial paralysis, movement disorders, vertigo, photomotor reflex disorders, seizures, progressive headache, or behavioral changes. They showed a significant (P = 0.0001) capacity of S100B to differentiate between poor CE and good CE in patients after mTBI and the best threshold conserving a sensitivity of 100% was 0.19 µg/L.
Conclusion
Despite the undoubtable role of CT imaging, clinical assessment represents a fundamental complementary diagnostic element in the management of pediatric mTBI. The primary objective of this meta-analysis was exploratory and aimed to highlight the possibility of using serum S100B levels in the diagnostic workflow of pediatric TBI. The promising potential of S100B integration in the management of pediatric head injury is evident. Based on this meta-analysis, the measurement of serum S100B could help informed decision-making in the ED setting, potentially safely reducing the use of CT scan in the pediatric population. S100B protein serum levels, in combination with the PECARN algorithm, could ultimately reduce the need for CT scans. The primary goal of this analysis has been to highlight the sufficiency of the evidence in this area, rather than offering specific treatment recommendations. The number of standardized studies is still insufficient, and the variability of protein concentration by age and sampling time should be studied in more detail. In order for S100B to be regularly introduced in the pediatric workflow for TBI, it is important to conduct further studies to obtain cut-off levels based on pediatric reference intervals.
Data availability
Data is provided within the manuscript or supplementary information files.
Abbreviations
- TBI:
-
Traumatic brain injury
- GCS:
-
Glasgow Coma Scale
- PECARN:
-
Pediatric Emergency Care Applied Research Network
- AUC:
-
Area under the curve
- CI:
-
Confidence interval
- NPV:
-
Negative predictive value
- CT:
-
Computed tomography
References
Araki T, Yokota H, Morita A (2017) Pediatric traumatic brain injury: characteristic features, diagnosis, and management. Neurol Med Chir (Tokyo) 57(2):82–93
Teasdale G, Maas A, Lecky F, Manley G, Stocchetti N, Murray G (2014) The Glasgow Coma Scale at 40 years: standing the test of time. Lancet Neurol 13(8):844–854
Chen C, Peng J, Sribnick EA, Zhu M, Xiang H (2018) Trend of age-adjusted rates of pediatric traumatic brain injury in U.S. Emergency Departments from 2006 to 2013. Int J Environ Res Public Health 15(6):1171
Sakkas A, Weiß C, Wilde F, Ebeling M, Scheurer M, Thiele OC et al (2023) Justification of indication for cranial CT imaging after mild traumatic brain injury according to the current national guidelines. Diagnostics (Basel, Switzerland) 13(11):1826
Lee S, Kim HY, Lee KH, Cho J, Lee C, Kim KP et al (2024) Risk of hematologic malignant neoplasms from head CT radiation in children and adolescents presenting with minor head trauma: a nationwide population-based cohort study. Eur Radiol 13(11):1826. https://doi.org/10.1007/s00330-024-10646-2
Pearce MS, Salotti JA, Little MP, McHugh K, Lee C, Kim KP et al (2012) Radiation exposure from CT scans in childhood and subsequent risk of leukaemia and brain tumours: a retrospective cohort study. Lancet (London, England) 380(9840):499–505
Kuppermann N, Holmes JF, Dayan PS, Hoyle JDJ, Atabaki SM, Holubkov R et al (2009) Identification of children at very low risk of clinically-important brain injuries after head trauma: a prospective cohort study. Lancet (London, England) 374(9696):1160–1170
(NICE) LNI for H and CE, editor. 2019 surveillance of head injury: assessment and early management (NICE guideline CG176) [Internet]. London; Available from: https://www.ncbi.nlm.nih.gov/books/NBK551814/
Bouvier D, Oris C, Brailova M, Durif J, Sapin V (2020) Interest of blood biomarkers to predict lesions in medical imaging in the context of mild traumatic brain injury. Clin Biochem [Internet] 85:5–11
Marzano LAS, Batista JPT, de Abreu Arruda M, de Freitas Cardoso MG, de Barros JLVM, Moreira JM et al (2022) Traumatic brain injury biomarkers in pediatric patients: a systematic review. Neurosurg Rev [Internet] 45(1):167–97
Oris C, Pereira B, Durif J, Simon-Pimmel J, Castellani C, Manzano S et al (2018) The biomarker s100b and mild traumatic brain injury: a meta-analysis. Pediatrics [Internet] 141(6)
Bouvier D, Castellani C, Fournier M, Dauphin J-B, Ughetto S, Breton M et al (2011) Reference ranges for serum S100B protein during the first three years of life. Clin Biochem 44(10–11):927–929
Moher D, Shamseer L, Clarke M, Ghersi D, Liberati A, Petticrew M et al (2015) Preferred reporting items for systematic review and meta-analysis protocols (PRISMA-P) 2015 statement. Syst Rev 4(1):1
Ouzzani M, Hammady H, Fedorowicz Z, Elmagarmid A (2016) Rayyan-a web and mobile app for systematic reviews. Syst Rev 5(1):210
Sterne JA, Hernán MA, Reeves BC, Savović J, Berkman ND, Viswanathan M et al (2016) ROBINS-I: a tool for assessing risk of bias in non-randomised studies of interventions. BMJ 355:i4919
Castellani C, Bimbashi P, Ruttenstock E, Sacherer P, Stojakovic T, Weinberg A-M (2009) Neuroprotein s-100B - a useful parameter in paediatric patients with mild traumatic brain injury? Acta Paediatr Int J Paediatr [Internet] 98(10):1607–12
Manzano S, Holzinger IB, Kellenberger CJ, Lacroix L, Klima-Lange D, Hersberger M et al (2016) Diagnostic performance of S100B protein serum measurement in detecting intracranial injury in children with mild head trauma. Emerg Med J [Internet] 33(1):42–6
Mozafari J, Fahimi MA, Mohammadi K, Barzegari H, Hanafi MG, Saki-Malehi A (2019) The diagnostic accuracy of serum and urinary S100B protein in children and adolescents with mild traumatic brain injury. New Zeal J Med Lab Sci [Internet] 73(3):88–91
Kelmendi FM, Morina AA, Mekaj AY, Blyta A, Alimehmeti R, Dragusha S et al (2018) Serum S100B levels can predict computed tomography findings in paediatric patients with mild head injury. BioMed Res Int Vol [Internet]. Available from: https://doi.org/10.1155/2018/6954045
Bouvier D, Fournier M, Dauphin J-B, Amat F, Ughetto S, Labbé A et al (2012) Serum S100B determination in the management of pediatric mild traumatic brain injury. Clin Chem 58(7):1116–1122
Papa L, Mittal MK, Ramirez J, Ramia M, Kirby S, Silvestri S et al (2016) In children and youth with mild and moderate traumatic brain injury, glial fibrillary acidic protein out-performs s100β in detecting traumatic intracranial lesions on computed tomography. J Neurotrauma [Internet] 33(1):58–64
Babcock L, Byczkowski T, Mookerjee S, Bazarian JJ (2012) Ability of S100B to predict severity and cranial CT results in children with TBI. Brain Inj [Internet] 26(11):1372–80
Hallén M, Karlsson M, Carlhed R, Hallgren T, Bergenheim M (2010) S-100B in serum and urine after traumatic head injury in children. J Trauma - Inj Infect Crit Care [Internet] 69(2):284–9
Simon-Pimmel J, Lorton F, Guiziou N, Levieux K, Vrignaud B, Masson D et al (2015) Serum S100β neuroprotein reduces use of cranial computed tomography in children after minor head trauma. Shock 44(5):410–416
Blackwell LS, Wali B, Xiang Y, Alawieh A, Sayeed I, Reisner A (2023) Prognostic value of plasma biomarkers S100B and osteopontin in pediatric TBI: a prospective analysis evaluating acute and 6-month outcomes after mild to severe TBI. Biomedicines. 11(8):2167
Yeung C, Bhatia R, Bhattarai B, Ayutyanont N, Sinha M (2018) Role of salivary biomarkers in predicting significant traumatic brain injury. Pediatrics [Internet] 141(1)
Berger RP, Adelson PD, Pierce MC, Dulani T, Cassidy LD, Kochanek PM (2005) Serum neuron-specific enolase, S100B, and myelin basic protein concentrations after inflicted and noninflicted traumatic brain injury in children. J Neurosurg [Internet] 103 Pediatrics (Suppl. 1):61–8
Dewan MC, Mummareddy N, Wellons JC 3rd, Bonfield CM (2016) Epidemiology of global pediatric traumatic brain injury: qualitative review. World Neurosurg 91:497-509.e1
Miglioretti DL, Johnson E, Williams A, Robert T, Weinmann S, Solberg LI et al (2017) Pediatric computed tomography and associated radiation exposure and estimated cancer risk. JAMA Pediatr 167(8):6–14
Pandor A, Goodacre S, Harnan S, Holmes M, Pickering A, Fitzgerald P et al (2011) Diagnostic management strategies for adults and children with minor head injury: a systematic review and an economic evaluation. Health Technol Assess 15(27):1–202
Ferrazzano PA, Rosario BL, Wisniewski SR, Shafi NI, Siefkes HM, Miles DK et al (2019) Use of magnetic resonance imaging in severe pediatric traumatic brain injury: assessment of current practice. J Neurosurg Pediatr 23(4):471–479
Morello A, Scala M, Schiavetti I, Diana MC, Severino M, Tortora D et al (2024) Surgical revascularization as a procedure to prevent neurological complications in children with moyamoya syndrome associated with neurofibromatosis I: a single institution case series. Child’s Nerv Syst ChNS Off J Int Soc Pediatr Neurosurg 13(8):1165
Calcagnile O, Anell A, Undén J (2016) The addition of S100B to guidelines for management of mild head injury is potentially cost saving. BMC Neurol [Internet] 16:200. https://doi.org/10.1186/s12883-016-0723-z
Yeung C, Bhatia R, Bhattarai B, Sinha M (2021) Role of salivary biomarkers in predicting significant traumatic brain injury. Pediatr Emerg Care [Internet] 37(12):E1373-6
Rothoerl RD, Woertgen C, Holzschuh M, Metz C, Brawanski A (1998) S-100 serum levels after minor and major head injury. J Trauma 45(4):765–767
Mondello S, Sorinola A, Czeiter E, Vámos Z, Amrein K, Synnot A et al (2021) Blood-based protein biomarkers for the management of traumatic brain injuries in adults presenting to emergency departments with mild brain injury: a living systematic review and meta-analysis. J Neurotrauma 38(8):1086–1106
Kelmendi FM, Morina AA, Mekaj AY, Blyta A, Alimehmeti R, Dragusha S et al (2018) Serum S100B levels can predict computed tomography findings in paediatric patients with mild head injury. Biomed Res Int [Internet] 2018:1–7
Ingebrigtsen T, Romner B, Kongstad P, Langbakk B (1995) Increased serum concentrations of protein S-100 after minor head injury: a biochemical serum marker with prognostic value? J Neurol Neurosurg Psychiatry [Internet] 59(1):103–4. Available from: https://pubmed.ncbi.nlm.nih.gov/7608699
Biberthaler P, Linsenmeier U, Pfeifer K-J, Kroetz M, Mussack T, Kanz K-G et al (2006) Serum S-100B concentration provides additional information fot the indication of computed tomography in patients after minor head injury: a prospective multicenter study. Shock 25(5):446–453
Undén J, Romner B (2010) Can low serum levels of S100B predict normal CT findings after minor head injury in adults?: an evidence-based review and meta-analysis. J Head Trauma Rehabil [Internet] 25(4):228–240. https://doi.org/10.1097/HTR.0b013e3181e57e22
Undén L, Calcagnile O, Undén J, Reinstrup P, Bazarian J (2015) Validation of the Scandinavian guidelines for initial management of minimal, mild and moderate traumatic brain injury in adults. BMC Med 13:292
Astrand R, Rosenlund C, Undén J, Neurotrauma S, Snc C (2016) Scandinavian guidelines for initial management of minor and moderate head trauma in children Scandinavian guidelines for initial management of minor and moderate head trauma in children. BMC Med [Internet]. https://doi.org/10.1186/s12916-016-0574-x
Bouvier D, Duret T, Rouzaire P, Jabaudon M, Rouzaire M, Nourrisson C et al (2016) Preanalytical, analytical, gestational and pediatric aspects of the S100B immuno-assays. Clin Chem Lab Med [Internet] 54(5):833–42
Roumpf SK, Welch JL (2019) Can S100B serum biomarker testing reduce head computed tomography scanning in children with mild traumatic brain injury? Ann Emerg Med [Internet] 73(5):456–8
Bouvier D, Cantais A, Laspougeas A, Lorton F, Plenier Y, Cottier M et al (2024) Serum S100B level in the management of pediatric minor head trauma: a randomized clinical trial. JAMA Netw open 7(3):e242366
Müller B, Evangelopoulos DS, Bias K, Wildisen A, Zimmermann H, Exadaktylos AK (2011) Can S-100B serum protein help to save cranial CT resources in a peripheral trauma centre? A study and consensus paper. Emerg Med J [Internet] 28(11):938–40
Injury MH, Zongo D, Ribéreau-gayon R, Masson F, Laborey M (2011) S100-B protein as a screening tool for the early assessment of. YMEM [Internet] 59(3):209–18. Available from: https://doi.org/10.1016/j.annemergmed.2011.07.027
Funding
Open access funding provided by Università degli Studi di Torino within the CRUI-CARE Agreement.
Author information
Authors and Affiliations
Contributions
Author’s contribution Study concept: A.M.—Study design: A.M.—Data acquisition: A.G.—Quality control of data and algorithms: A.M.; I.S.—Data analysis and interpretation: A.M.; I. S.; F.C.—Statistical analysis: I.S.; A.M.—Manuscript preparation: E.L.B.; I.P.; S.C.—Manuscript editing: A.M.; F.C.—Manuscript review: M.P.; M.M.L.; D.G. All authors agreed to the publication of this work.
Corresponding author
Ethics declarations
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Morello, A., Schiavetti, I., Lo Bue, E. et al. Update on the role of S100B in traumatic brain injury in pediatric population: a meta-analysis. Childs Nerv Syst (2024). https://doi.org/10.1007/s00381-024-06565-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00381-024-06565-8