Abstract
Purpose
To investigate the associations of overall diet quality and dietary factors with serum biomarkers for lipid and amino acid metabolism in a general population of children.
Methods
We studied 194 girls and 209 boys aged 6–8 years participating in the Physical Activity and Nutrition in Children study. Food consumption was assessed by 4-day food records and diet quality was quantified by the Finnish Children Healthy Eating Index (FCHEI). Fasting serum fatty acids, amino acids, apolipoproteins, as well as lipoprotein particle sizes were analyzed with high-throughput nuclear magnetic resonance spectroscopy. Data were analyzed using linear regression adjusted for age, sex, and body fat percentage.
Results
FCHEI was directly associated with the ratio of polyunsaturated (PUFA) to saturated fatty acids (SFA) (PUFA/SFA), the ratio of PUFA to monounsaturated fatty acids (MUFA) (PUFA/MUFA), the ratio of PUFA to total fatty acids (FA) (PUFA%), the ratio of omega-3-fatty acids to total FA (omega-3 FA%), and inversely associated with the ratio of MUFA to total FA (MUFA%), alanine, glycine, histidine and very-low density lipoprotein (VLDL) particle size. Consumption of vegetable oils and vegetable-oil-based margarine (≥ 60% fat) was directly associated with PUFA/SFA, PUFA/MUFA, PUFA%, the ratio of omega-6 FA to total FA (omega-6 FA%), and inversely associated with SFA, MUFA, SFA to total FA (SFA%), MUFA%, alanine and VLDL particle size. Consumption of high-fiber grain products directly associated with PUFA/SFA, PUFA/MUFA, omega-3 FA%, omega-6 FA%, PUFA% and inversely associated with SFA and SFA%. Fish consumption directly related to omega-3 FA and omega-3 FA%. Consumption of sugary products was directly associated with histidine and VLDL particle size. Vegetable, fruit, and berry consumption had direct associations with VLDL particle size and the ratio of apolipoprotein B to apolipoprotein A1. Consumption of low fat (< 1%) milk was directly associated with phenylalanine. A higher consumption of high-fat (≥ 1%) milk was associated with lower serum MUFA/SFA and higher SFA%. Sausage consumption was directly related to SFA% and histidine. Red meat consumption was inversely associated with glycine.
Conclusions
Better diet quality, higher in intake of dietary sources of unsaturated fat and fiber, and lower in sugary product intake were associated with more favorable levels of serum biomarkers for lipid and amino acid metabolism independent of adiposity.
Trial Registration
ClinicalTrials.gov: NCT01803776, registered March 3, 2013.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Poor diet quality is associated with metabolic disturbances, including increased body fat content, dyslipidemia, higher blood pressure and metabolic syndrome in adults [1] and children [2, 3]. Moreover, a diet abundant with unhealthy choices, such as a high consumption of sugar-sweetened beverages and a low consumption of fruits and vegetables, has been linked to an increased risk of cardiometabolic diseases in adults [4]. Metabolomics, the study of small molecules in biofluids, provides an opportunity to explore the possible mediating factors in the complex pathophysiological pathways between diet and health [5].
In cross-sectional and cohort studies among adults, increased serum, or plasma concentrations of saturated fatty acids (SFA), aromatic amino acids (AAA), branched-chain amino acids (BCAA) and apolipoprotein (apo) B, a small low-density lipoprotein (LDL) particle size, and a large very-low-density (VLDL) particle size have been linked to metabolic disturbances and cardiometabolic diseases [6,7,8,9,10]. Furthermore, increased serum or plasma concentrations of SFA, AAA, and BCAA as well as a small LDL particle size have been associated with insulin resistance, an atherogenic lipid profile, and cardiometabolic risk score in cross-sectional studies among children [11,12,13].
Better diet quality has been linked to a more cardioprotective metabolic profile characterized as higher concentrations of serum or plasma polyunsaturated fatty acids (PUFA), lower serum concentrations of BCAA, AAA [10] and apoB [14], a smaller VLDL particle size, and a larger LDL particle size in adults [15]. However, the evidence in children on the associations between diet and metabolites, which might be the mediating factors between diet and health, is scarce. Since diet quality is relatively low among children globally [16, 17], and dietary habits track from childhood to adulthood [18], research focusing on children’s nutrition and health is extremely important in the prevention of multiple diseases and in improving overall public health. Therefore, we wanted to perform a study examining multiple dietary factors and metabolites in a large population sample of children.
We examined the associations of overall diet quality, assessed by the Finnish Children Healthy Eating Index (FCHEI), and single dietary factors with serum concentrations of fatty acids, amino acids, apoB and apoA1, as well as the sizes of VLDL, LDL, and high-density lipoprotein (HDL) particles, measured by a high-throughput nuclear magnetic resonance (NMR) spectroscopy metabolomics analysis, in a population sample of 6–8-year-old Finnish children.
Materials and methods
Study population
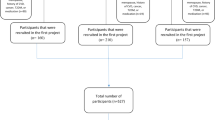
These cross-sectional analyses are based on the baseline data of the Physical Activity and Nutrition in Children (PANIC) study, which is an 8-year physical activity and diet intervention study with an ongoing follow-up in a population sample of children from the city of Kuopio, Finland. The study has been described in more detail elsewhere [19]. In short, 736 children were invited to participate in the baseline examinations of the study between 2007 and 2009. Of all invited, 512 (70%) attended. The PANIC study protocol was approved by the Research Ethics Committee of the Hospital District of Northern Savo in 2006 (Statement 69/2006). The parents or caregivers gave their written informed consent, and the children provided their assent to participation. The present study sample consisted of 403 children (194 girls, 209 boys) with complete data on variables used in the analyses, except linoleic acid for which data from 382 children were available.
Assessment of diet
Food consumption and energy intake were assessed by food records filled out by the parents or caregivers on four predefined consecutive days including either two weekdays and two weekend days (99.5% of participants) or three weekdays and one weekend day (0.5% of participants), as described earlier [19]. At the first study visit, a clinical nutritionist instructed the parents to record all food and drinks consumed by their child at home and outside home. At the second study visit, the clinical nutritionist reviewed and completed, if necessary, the food records together with the parents. Food records were analyzed using the Micro Nutrica® dietary analysis software, version 2.5 (The Social Insurance Institution of Finland), that uses Finnish and international data on the nutrient compositions of foods [20] and that was regularly updated by a clinical nutritionist.
We computed FCHEI [21] to assess overall diet quality. FCHEI consists of five categories: (1) vegetables, fruit, and berries, (2) vegetable oils and vegetable-oil-based margarine (≥ 60% fat), (3) low-fat (< 1%) milk, (4) fish, and (5) foods with high sugar content. The scoring of the index has been described elsewhere [22]. Briefly, the consumption of these foods was divided by energy intake and categorized to deciles. Deciles were scored, a higher decile getting a higher score apart from sugary products that were inversely scored. The sum of scores from the five categories was calculated, with a minimum of 0 indicating lower diet quality and a maximum of 50 indicating better diet quality.
Biochemical analyses
Venous blood samples were collected from the children after a 12-h fast. Blood samples were centrifuged and stored at a temperature of − 75 °C until analyses. Serum concentrations of total fatty acids (FA), SFA, monounsaturated fatty acids (MUFA), PUFA, omega-3-fatty acids, omega-6-fatty acids, docosahexaenoic acid (DHA), linoleic acid (LA), alanine, glutamine, glycine, histidine, isoleucine, leucine, valine, total BCAA (including isoleucine, leucine and valine), phenylalanine, tyrosine, apoB and apoA1 as well as the sizes of VLDL, LDL, and HDL particles were measured using high-throughput NMR spectroscopy metabolomics analysis (Nightingale Health Ltd, Kuopio Finland) as described in detail earlier [23]. Finally, the degree of serum fatty acid unsaturation (i.e., the presence and amount of double bonds in the carbon chains of the fatty acids), the ratios of MUFA to SFA (MUFA/SFA), PUFA to SFA (PUFA/SFA), PUFA to MUFA (PUFA/MUFA), SFA to total FA (expressed as a percentage, SFA%), MUFA to total FA (expressed as a percentage, MUFA%,), PUFA to total FA (expressed as a percentage, PUFA%), omega-3-FA to total FA (expressed as a percentage, omega-3-FA%), omega-6-FA to total FA (expressed as a percentage, omega-6-FA%), DHA to total FA (expressed as a percentage, DHA%), LA to total FA (expressed as a percentage, LA%), and the ratio of apoB to apoA1 (apoB/apoA1) were assessed.
Other assessments
Body height and weight were measured, and body mass index (BMI) and body mass index standard deviation score (BMI-SDS) were calculated [19]. Body fat percentage was assessed by a dual energy X-ray absorptiometry (DXA) method with a Lunar® DXA device (Lunar Prodigy Advance; GE Medical Systems, Madison, WI, USA).
Statistical analyses
The sample size of this study is based on the power calculations for our study on the intervention effects on fasting insulin and homeostatic model assessment of insulin resistance in children [24]. Briefly, we determined the number of children required to detect at least 0.30 standard deviation difference between the intervention group (60% of children) and the control group (40% of children) with a power of 80% and a two-sided p value for the difference between the groups of 0.05 allowing for a 20% loss to follow-up or missing data. These power calculations provided a required sample size of at least 275 children in the intervention group and 183 children in the control group. Data were analyzed using the SPSS Statistics software, version 27.0 (IBM Corporation, IBM SPSS Statistics for Windows, Armonk, NY, USA). Differences and associations with p values of < 0.05 were considered statistically significant. All continuous variables were checked for normality by observing histograms and using the Kolmogorov–Smirnov test. If not normally distributed, variables were transformed with logarithmic or square root transformation. Not all variables were normally distributed after these transformations, and non-parametric tests were used. To compare basic characteristics, dietary factors and metabolites between boys and girls, we used the Student’s t test for normally distributed variables or the Mann–Whitney’s U test for variables with skewed distributions.
Linear regression analyses adjusted for age and sex were used to examine the associations of FCHEI and food consumption with serum biomarkers of lipid and amino acid metabolism. For statistically significant associations, we further adjusted the data for body fat percentage that may confound or mediate the observed associations [25, 26]. We used the non-transformed dietary variables as the residuals of the linear regression analyses were normally distributed. Of the outcome variables, only serum MUFA was used as logarithmically transformed. Benjamini–Hochberg False Discovery Rate (FDR) using the FDR value of 0.2 was used to adjust results for multiple comparison [27].
Results
Basic characteristics and serum metabolites in children
Boys were taller, had lower body fat percentage and higher energy intake, consumed more red meat and sausages and had higher serum PUFA/SFA, PUFA/MUFA, PUFA%, omega-6 FA% and LA% than girls (Table 1). Girls had higher serum total FA, SFA, MUFA, MUFA/SFA, MUFA%, glutamine, apoB and apoB/apoA1 and lower serum apoA1 than boys (Table 2).
Associations of FCHEI and single dietary factors with serum fatty acids
A higher FCHEI score was associated with higher serum PUFA/SFA, PUFA/MUFA, PUFA% and omega-3 FA% as well as lower MUFA% after adjustment for age and sex (Table 4). A higher consumption of low-fat (< 1%) milk was associated with a higher serum DHA (Table 3). A higher consumption of vegetable oils and vegetable-oil-based margarine (≥ 60% fat) was associated with higher PUFA/SFA, PUFA/MUFA, PUFA%, omega-6 FA%, and lower serum SFA, MUFA, and lower ratios of SFA and MUFA to total FA. In addition, a higher consumption of high-fiber grain products was associated with higher serum PUFA/SFA, PUFA/MUFA, PUFA%, omega-3 FA%, omega-6 FA%, and lower serum SFA, MUFA, and SFA%. A higher consumption of low-fiber grain products was associated with lower serum omega-3 fatty acids and a lower degree of serum fatty acid unsaturation. A higher consumption of high-fat (≥ 1%) milk was associated with lower serum MUFA/SFA and higher SFA%. A higher fish consumption was associated with higher serum omega-3 fatty acids and the ratio of omega-3 FA%. A higher consumption of sugary products was associated with higher SFA%. Last, higher consumption of sausages was associated with lower PUFA/SFA and higher SFA%. The associations of FCHEI with higher serum, PUFA/SFA, PUFA/MUFA, PUFA%, omega-3 FA%, and lower MUFA%, the associations of vegetable oils and vegetable-oil-based margarine (≥ 60% fat) and high-fiber grain products with higher serum PUFA/SFA, PUFA/MUFA, PUFA%, omega-6 FA%, and with lower serum SFA and SFA% remained statistically significant after further adjustment for body fat percentage (Tables 3 and 4). In addition, the association of vegetable oils and vegetable-oil-based margarine (≥ 60% fat) with lower MUFA%, the association of high-fiber grain products with higher serum omega-3 FA%, the association of high-fat (≥ 1%) milk with MUFA/SFA and SFA%, the association of fish consumption with serum omega-3 fatty acids and omega-3 FA%, and the association of sausage consumption with SFA% remained statistically significant after further adjustment for body fat percentage. The associations of FCHEI with PUFA/SFA, PUFA/MUFA, MUFA%, PUFA%, omega-3 FA%, the associations of vegetable oils and vegetable-oil-based margarine (≥ 60% fat) with SFA, MUFA, PUFA/SFA, PUFA/MUFA, SFA%, MUFA%, PUFA%, omega-6 FA%, and the associations of high-fiber grain products with SFA, MUFA, PUFA/SFA, PUFA/MUFA, SFA%, PUFA%, omega-3 FA% and with omega 6-FA% remained statistically significant after FDR correction.
Associations of FCHEI and single dietary factors with serum amino acids
FCHEI was inversely associated with serum alanine, glycine, and histidine after adjustment for age and sex (Table 5). The consumption of vegetable oils and vegetable-oil-based margarine (≥ 60% fat) was associated with lower serum alanine. A higher consumption of low-fat (< 1%) milk was associated with higher serum phenylalanine and lower glutamine. A higher consumption of sugary products and sausages was associated with higher serum histidine. A higher consumption of red meat was associated with lower serum glycine. All these associations, except that between low-fat (< 1%) milk and serum glutamine, remained statistically significant after additional adjustment for body fat percentage. The associations of FCHEI with alanine, glycine, and histidine, the association of vegetable oils and vegetable-oil-based margarine (≥ 60% fat) with lower serum alanine, the association of low-fat (< 1%) milk with phenylalanine, and the association of sugary products with histidine remained statistically significant even after FDR correction.
Associations of FCHEI and single dietary factors with lipoprotein particle size
A higher FCHEI was associated with a smaller VLDL particle size after adjustment for age and sex (Table 6). A higher consumption of sugary products and a higher consumption of vegetables, fruit, and berries were associated with a larger VLDL particle size whereas a higher consumption of vegetable oils and vegetable-oil-based margarine (≥ 60% fat) was associated with a smaller VLDL particle size. Additional adjustment for body fat percentage did not affect these associations. All the associations except for the association of a higher consumption of vegetables, fruit, and berries with a larger VLDL particle size remained after FDR correction.
Associations of FCHEI and single dietary factors with apolipoproteins
FCHEI was not associated with serum apolipoproteins after adjustment for age and sex (Table 7). However, a higher consumption of vegetables, fruit, and berries was associated with higher serum apoB/apoA1. This association remained statistically significant after further adjustment for body fat percentage but not after FDR correction.
Discussion
We found several associations of overall diet quality and single dietary factors with serum biomarkers of lipid and amino acid metabolism, particularly fatty acids, in a population sample of primary school-aged children. For instance, better diet quality was associated with higher ratios of PUFA and omega-3 FA, lower serum alanine, glycine, and histidine and a smaller VLDL particle size. Higher consumption of vegetable oils and vegetable-oil-based margarine (≥ 60% fat) and high-fiber grain products were associated with higher serum PUFAs, omega-6 FA, and lower serum SFA and MUFA. In addition, consumption of high-fiber grain products was associated with higher serum omega-3 FA%. A higher fish consumption was associated with higher serum omega-3 fatty acids, whereas a higher consumption of sugary products, high-fat dairy and meat products was associated mainly with fatty acids, serum amino acids, such as higher SFA%, lower PUFA/SFA, higher serum phenylalanine and histidine, and lower glycine. Also, a higher consumption of vegetable oils and vegetable-oil-based margarine (≥ 60% fat) was associated with a smaller VLDL particle size and a higher consumption of sugary products was associated with a larger VLDL particle size. Six of the observed associations, mainly the association of dietary factors with fatty acids such as a higher consumption of low-fiber grain products with lower serum omega-3 fatty acids and a lower degree of serum fatty acid unsaturation, were partly explained by body fat percentage, suggesting that adiposity mediates these associations. Several associations remained after the correction for multiple testing. However, concerning serum fatty acids, mainly the associations of FCHEI, vegetable oils, and vegetable-oil-based margarine (≥ 60% fat) and high-fiber grain products with serum fatty acid ratios remained statistically significant after the FDR correction.
Fatty acids
Better diet quality was mostly associated with ratios of fatty acids, such as higher PUFA/SFA, PUFA/MUFA, MUFA%, PUFA%, omega-3 FA% but not with concentrations of any separate serum fatty acids. A previous study in Brazilian children did not find associations of diet quality and serum fatty acids [28]. However, many studies have found associations particularly between better diet quality and higher circulating n-3-fatty acids in children [29, 30]. Our results indicate that better diet quality may enhance the ratios of fatty acids in the serum to a more favorable direction, that meaning higher PUFA concentrations in relation to MUFA, SFA or total FA. Our findings concerning the associations of a higher consumption of vegetable oils and vegetable-oil-based margarine (≥ 60% fat) with lower serum levels of SFA, and a higher fish consumption with higher serum omega-3 fatty acids are consistent with the findings of previous studies in children [31, 32]. Furthermore, the results of this study suggest that a higher consumption of vegetable oils and vegetable-oil-based margarine (≥ 60% fat) may influence the ratios of serum fatty acids to a more favorable direction, as in the case of overall diet quality. We also observed direct associations of the consumption of high-fiber grain products with serum PUFA/SFA, PUFA/MUFA, PUFA%, omega-3 FA%, omega-6 FA%, and inverse associations with serum SFA, MUFA, and SFA%. We have previously reported that high-fiber grain product consumption is directly associated with the dietary intake of PUFAs but not MUFAs or SFAs in children [33]. Thus, children who consume high-fiber grain products may have a higher PUFA intake and a lower MUFA and SFA intake which is expected to improve serum fatty acid profile. Higher blood PUFAs have been viewed as cardioprotective in adults [6] and children [34]. Our results suggest that vegetable oils and vegetable-oil-based margarine (≥ 60% fat) and fish, foods that are rich in PUFAs might be cardioprotective by improving fatty acid metabolism since childhood. It has been reported that dietary counselling from childhood to adulthood, focusing on the quality of dietary fat, improves cardiovascular health [35]. Some of the observed associations of dietary factors with serum fatty acids were explained by body fat percentage, suggesting that adiposity may mediate these associations.
Amino acids
Studies on the relationships between dietary factors and serum or plasma amino acids in children are scarce. We observed that better overall diet quality was associated with lower serum alanine, glycine, and histidine. An intervention study showed that plasma alanine was lower in participants following a healthy Nordic diet compared with participants following a typical Danish diet in adults [36], consistently with our findings. One explanation for these findings could be that better diet quality enhances insulin sensitivity [37] and thereby increases muscle protein synthesis [38] and decreases serum alanine. Importantly, higher circulating alanine has been directly associated with cardiovascular events in adults [7]. The results of our study suggest that better diet quality could improve cardiometabolic health by influencing amino acid metabolism since childhood. However, alanine is a non-essential amino acid that can be endogenously synthetized [39]. Hence, there might be factors affecting alanine concentrations that we were not able to account for and that would explain the observed associations.
A Korean intervention study found that serum glycine decreased after following a healthy diet with less red and processed meats compared to controls following a Western diet [40]. We found inverse associations of overall diet quality and red meat consumption with serum glycine, the latter being inconsistent with the results of the Korean intervention study. Glycine is a conditionally essential amino acid and can be synthetized, like alanine, endogenously from other sources [41] and thus it is possible that serum glycine does not directly reflect dietary intake.
The observed association of better diet quality and lower serum histidine may be partly explained by the association between a higher consumption of sugary products and higher serum histidine since higher consumption of sugary products can lower the FCHEI score. Histidine is an essential amino acid derived from animal-based products, such as meat and dairy [42]. Typical sources of sugar in our study population include dairy products, such as sweetened sour milk products and ice cream [33]. Thus, it is possible that a high consumption of these products increases serum histidine in children. However, not all milk products, such as sour milk products, were associated with serum histidine. Histidine was also directly associated with sausage consumption, which is logical considering that meat is a predominant source of histidine [42]. However, the consumption of red meat was not related to serum histidine.
Phenylalanine is an aromatic amino acid whose serum concentrations have been directly associated with cardiovascular outcomes in adults [7] and with insulin resistance in children [12]. We observed a direct association between the consumption of low-fat (< 1%) milk and serum phenylalanine. Consistent results have been reported in a study examining the effects of dairy- and meat-based complementary diets among infants [43]. The consumption of milk with higher fat content was not associated with phenylalanine in our data. Finnish children have, however, been reported to consume notably more low-fat milk than milk with higher fat content [33]. Thus, it is possible that an excessive consumption of milk leads to higher serum phenylalanine although the association is logical considering that milk and dairy products are a source of phenylalanine in diets [42]. Also, a slightly higher protein content of low-fat milk may partly explain this association.
Surprisingly, our results do not indicate that there is an association between diet and BCAA concentration, which is contrary to the results of cross-sectional studies among adults [10, 44] and an intervention study among children [45]. However, in one of the cross-sectional studies [10], the association between diet and BCAA levels was not detected when analyses were repeated in a younger population (ages between 3 and 18 years). In addition, our findings are in line with the results of a dietary counselling intervention aiming to maintain a healthier diet, which had no effect on BCAA levels in children and adolescents [46]. It is possible that adults consume bigger portions and thus larger amounts of the dietary sources of BCAA’s compared to youth or children, which is then reflected in the BCAA levels in the bloodstream. This is supported by the fact that in the intervention study among children where diet was reflected in the BCAA levels [45], the children consumed notably high portions of dairy or meat. In the present study, the consumption of dietary sources of BCAA’s might have been subtle, thus not having an impact on the BCAA levels. It has also been observed that amino acid concentrations in the plasma and skeletal muscle change with age [47]. Therefore, differences in skeletal muscle and amino acid metabolism between different age groups might also explain the contrary results.
Lipoprotein particle size
Better overall diet quality was associated with a smaller VLDL particle size, which is in line with findings among adults [15]. We have previously found that a higher FCHEI score is associated with lower triglyceride levels in Finnish children [48]. The concentration of triglycerides, especially in the liver, might influence the formation of different VLDL subfractions; for example, the larger and less dense VLDL particles contain more triglycerides [49]. It is thus possible that triglyceride levels might explain the association between better overall quality and a smaller VLDL particle size in the present study. We also found an association between a higher consumption of sugary products and a larger VLDL particle size that could be explained by a higher fructose intake related to a high sugar product consumption. We have previously observed that sugary products are the main source of sucrose in Finnish children [33]. Since fructose is a structural unit of sucrose, it is possible that the intake of fructose is higher in children consuming higher amounts of sugary products. Fructose intake has been directly associated with liver fat content in adults [50]. VLDL particles are mostly produced in and secreted from the liver [51] and elevated fatty acid content of the liver has been observed to induce the formation of larger VLDL particles [52]. Therefore, a higher intake of fructose might lead to higher secretion of larger VLDL particles from the liver through increased liver adiposity. We also found an association between a higher consumption of vegetables, fruit, and berries and a larger VLDL size. It is possible that fructose intake from this food group increases VLDL particle size. However, there might be other lifestyle factors that explain the observed association that we could not account for. The inverse association between the consumption of vegetable oils and vegetable-oil-based margarine (≥ 60% fat), rich in PUFAs and low in SFAs, and VLDL particle size in the present study might also be explained by the accumulation of liver fat since the intake of SFAs has been directly linked to liver fat content in adults and children [53, 54]. We did not observe associations of dietary factors with LDL particle size, inconsistent with previous intervention studies in children [55, 56]. In accordance with the results of some previous studies [56], we observed no associations of dietary factors with HDL particle size.
Apolipoproteins
Lifestyle interventions have been shown to decrease circulating apoB and increase circulating apoA1 in children with obesity and hypercholesterolemia [57, 58]. We found no associations of overall diet quality or single dietary factors with serum apolipoproteins, except the direct association of vegetables, fruit, and berries consumption with apoB/apoA1, in a general population of children. Apolipoproteins are structural components of lipoprotein particles [51], mainly LDL cholesterol particles [59]. We did not find an association between overall diet quality and serum LDL cholesterol in our previous study in children [48]. Together with the results of previous studies [57, 58], our observations suggest that overall diet quality does not influence serum apolipoproteins in metabolically healthy children, but it may be seen in children with hypercholesterolemia and with elevated circulating LDL cholesterol levels. The direct association between the consumption of vegetables, fruit, and berries and apoB/apoA1 in our study might be related to fructose intake from this food group, since it has been shown that a short-term fructose intake restriction reduces serum apoB in children [60]. However, other lifestyle factors that we could not account for mediate this association.
Study strengths and limitations
We examined a general population sample of prepubertal children who have not yet been exposed to possible confounding factors for the associations of dietary factors with serum biomarkers for lipid and amino acid metabolism, such as alcohol consumption, smoking, diseases, and medications. Moreover, food consumption was assessed comprehensively by 4-day dietary records, reviewed by clinical nutritionists. Overall diet quality was assessed using FCHEI, which has been validated in Finnish children [21] and represents accurately the dietary challenges in our study population. Also, we used high-throughput NMR spectroscopy analysis to assess the serum biomarkers of lipid and amino acid metabolism. This method is well-suited to identify blood metabolites in large study populations [5]. This study also has limitations. First, the sample size of this study was not originally based on the current research question but is based on the power calculations for our previous study on the intervention effects on fasting insulin and homeostatic model assessment of insulin resistance in children [24]. However, we estimated that 395 participants were required to detect an association with a small effect size (f2 ≥ 0.02) in a multivariate linear regression analysis at the power of 80. Second, the assessment of diet using any measure administrated by the parents or caregivers of children, including dietary records, is prone to inaccurate reporting. Also, due to the complexity of eating behavior and human metabolism, we could not consider all possible confounding factors in the statistical analyses. Thus, the results must be interpreted carefully, simultaneously reviewing other studies on the topic. We could not conclude all possible mechanisms explaining the observed associations due to the lack of research on this topic in children. Last, we cannot draw conclusions about the causality between diet and serum metabolites due to the cross-sectional nature of this study.
Conclusion
We observed many associations of overall diet quality and single dietary factors with serum biomarkers of lipid and amino acid metabolism in children, and these associations were mainly independent of adiposity. Better overall diet quality, a higher consumption of dietary sources of unsaturated fatty acids and high-fiber grain products, and a lower consumption of sugary products were associated with higher serum PUFAs, lower serum MUFAs and SFAs, and smaller serum VLDL particles. The results indicate that good diet quality is important in improving lipid and amino acid metabolism from an early age. Further evidence from long-term diet intervention studies is warranted to confirm whether diet modification has beneficial effects on lipid and amino acid metabolism, and whether these have meaningful effects on cardiometabolic health since childhood.
Data availability
The PANIC study is ongoing, and therefore the data are not fully anonymized and, thus, not openly available. Part of the data can be shared by request.
References
Nicklas TA, O’Neil CE, Fulgoni VL (2012) Diet quality is inversely related to cardiovascular risk factors in adults. J Nutr 142:2112–2118. https://doi.org/10.3945/jn.112.164889
Krijger JA, Nicolaou M, Nguyen AN, Voortman T, Hutten BA, Vrijkotte TG (2021) Diet quality at age 5–6 and cardiovascular outcomes in preadolescents. Clin Nutr ESPEN 43:506–513. https://doi.org/10.1016/j.clnesp.2021.02.011
Kerr JA, Liu RS, Gasser CE, Mensah FK, Burgner D, Lycett K, Gillespie AN, Juonala M, Clifford SA, Olds T, Saffery R, Gold L, Liu M, Azzopardi P, Edwards B, Dwyer T, Wake M (2021) Diet quality trajectories and cardiovascular phenotypes/metabolic syndrome risk by 11–12 years. Int J Obes (Lond) 45:1392–1403. https://doi.org/10.1038/s41366-021-00800-x
Miller V, Micha R, Choi E, Karageorgou D, Webb P, Mozaffarian D (2022) Evaluation of the quality of evidence of the association of foods and nutrients with cardiovascular disease and diabetes: a systematic review. JAMA Netw Open 5:e2146705. https://doi.org/10.1001/jamanetworkopen.2021.46705
Brennan L (2014) NMR-based metabolomics: from sample preparation to applications in nutrition research. Prog Nucl Magn Reson Spectrosc 83:42–29. https://doi.org/10.1016/j.pnmrs.2014.09.001
Kaikkonen JE, Jula A, Viikari JS, Juonala M, Hutri-Kähönen N, Kähönen M, Lehtimäki T, Raitakari OT (2021) Associations of serum fatty acid proportions with obesity, insulin resistance, blood pressure, and fatty liver: the cardiovascular risk in young finns study. J Nutr 151:970–978. https://doi.org/10.1093/jn/nxaa409
Jauhiainen R, Vangipurapu J, Laakso A, Kuulasmaa T, Kuusisto J, Laakso M (2021) The association of 9 amino acids with cardiovascular events in finnish men in a 12-year follow-up study. J Clin Endocrinol Metab 106:3448–3454. https://doi.org/10.1210/clinem/dgab562
Zuber V, Gill D, Ala-Korpela M, Langenberg C, Butterworth A, Bottolo L, Burgess S (2021) High-throughput multivariable Mendelian randomization analysis prioritizes apolipoprotein B as key lipid risk factor for coronary artery disease. Int J Epidemiol 50:893–901. https://doi.org/10.1093/ije/dyaa216
Hartz J, Krauss RM, Göttsater M, Melander O, Nilsson P, Mietus-Snyder M (2020) Lipoprotein particle predictors of arterial stiffness after 17 years of follow up: the Malmö Diet and Cancer study. Int J Vasc Med 2020:4219180. https://doi.org/10.1155/2020/4219180
Akbaraly T, Würtz P, Singh-Manoux A, Shipley MJ, Haapakoski R, Lehto M, Desrumaux C, Kähönen M, Lehtimäki T, Mikkilä V (2018) Association of circulating metabolites with healthy diet and risk of cardiovascular disease: analysis of two cohort studies. Sci Rep 8:8620. https://doi.org/10.1038/s41598-018-26441-1
Venäläinen T, Ågren J, Schwab U (2016) Cross-sectional associations of plasma fatty acid composition and estimated desaturase and elongase activities with cardiometabolic risk in Finnish children—The PANIC study. J Clin Lipidol 10:82–91. https://doi.org/10.1016/j.jacl.2015.09.004
Zhang X, Ojanen X, Zhuang H, Wu N, Cheng S, Wiklund P (2019) Branched-chain and aromatic amino acids are associated with insulin resistance during pubertal development in girls. J Adolesc Health 65:337–343. https://doi.org/10.1016/j.jadohealth.2019.01.030
Shimabukuro T, Sunagawa M, Ohta T (2004) Low-density lipoprotein particle size and its regulatory factors in school children. J Clin Endocrinol Metabol 89:2923–2927. https://doi.org/10.1210/jc.2003-031818
Poggio R, Elorriaga N, Gutierrez L, Irazola V, Rubinstein A, Danaei G (2017) Associations between dietary patterns and serum lipids, apo and C-reactive protein in an adult population: evidence from a multi-city cohort in South America. Br J Nutr 117(4):548–555. https://doi.org/10.1017/S0007114517000514
Millar SR, Navarro P, Harrington JM, Shivappa N, Hébert JR, Perry IJ, Phillips CM (2021) Comparing dietary score associations with lipoprotein particle subclass profiles: a cross-sectional analysis of a middle-to older-aged population. Clin Nutr 40:4720–4729. https://doi.org/10.1016/j.clnu.2021.06.005
Liu J, Rehm CD, Onopa J, Mozaffarian D (2020) Trends in diet quality among youth in the United States, 1999–2016. JAMA 323:1161–1174. https://doi.org/10.1001/jama.2020.0878
Diethelm K, Jankovic N, Moreno LA, Huybrechts I, De Henauw S, De Vriendt T, Gonzalez-Gross M, Leclercq C, Gottrand F, Gilbert CC (2012) Food intake of European adolescents in the light of different food-based dietary guidelines: results of the HELENA (Healthy Lifestyle in Europe by Nutrition in Adolescence) Study. Public Health Nutr 15:386–398. https://doi.org/10.1017/S1368980011001935
Kaikkonen JE, Mikkilä V, Magnussen CG, Juonala M, Viikari JS, Raitakari OT (2013) Does childhood nutrition influence adult cardiovascular disease risk? Insights from the Young Finns Study. Ann Med 45:120–128. https://doi.org/10.3109/07853890.2012.671537
Eloranta AM, Lindi V, Schwab U, Tompuri T, Kiiskinen S, Lakka HM, Laitinen T, Lakka TA (2012) Dietary factors associated with overweight and body adiposity in Finnish children aged 6–8 years: the PANIC Study. Int J Obes (Lond) 36:950–955. https://doi.org/10.1038/ijo.2012.89
Rastas M, Seppänen R, Knuts L, Hakala P, Karttila V (1997) Nutrient composition of foods. The Social Insurance Institution, Helsinki
Kyttälä P, Erkkola M, Lehtinen-Jacks S, Ovaskainen M, Uusitalo L, Veijola R, Simell O, Knip M, Virtanen SM (2014) Finnish Children Healthy Eating Index (FCHEI) and its associations with family and child characteristics in pre-school children. Public Health Nutr 17:2519–2527. https://doi.org/10.1017/S1368980013002772
Eloranta A, Jääskeläinen J, Venäläinen T, Jalkanen H, Kiiskinen S, Mäntyselkä A, Schwab U, Lindi V, Lakka TA (2018) Birth weight is associated with dietary factors at the age of 6–8 years: the Physical Activity and Nutrition in Children (PANIC) study. Public Health Nutr 21:1278–1285. https://doi.org/10.1017/S1368980017004013
Soininen P, Kangas AJ, Würtz P, Suna T, Ala-Korpela M (2015) Quantitative serum nuclear magnetic resonance metabolomics in cardiovascular epidemiology and genetics. Circ Cardiovasc Genet 8:192–206. https://doi.org/10.1161/CIRCGENETICS.114.000216
Lakka TA, Lintu N, Väistö J, Viitasalo A, Sallinen T, Haapala EA, Tompuri TT, Soininen S, Karjalainen P, Schnurr TM, Mikkonen S, Atalay M, Kilpeläinen TO, Laitinen T, Laaksonen DE, Savonen K, Brage S, Schwab U, Jääskeläinen J, Lindi V, Eloranta AM (2020) A 2 year physical activity and dietary intervention attenuates the increase in insulin resistance in a general population of children: the PANIC study. Diabetologia 63(11):2270–2281. https://doi.org/10.1007/s00125-020-05250-0
Rainwater DL, Mitchell BD, Comuzzie AG, Haffner SM (1999) Relationship of low-density lipoprotein particle size and measures of adiposity. Int J Obes Relat Metab Disord 23:180–189. https://doi.org/10.1038/sj.ijo.0800813
Gibney MJ, Walsh M, Brennan L, Roche HM, German B, Van Ommen B (2005) Metabolomics in human nutrition: opportunities and challenges. Am J Clin Nutr 82:497–503. https://doi.org/10.1093/ajcn.82.3.497
Benjamini Y, Hochberg Y (1995) Controlling the false discovery rate: a practical and powerful approach to multiple testing. J Roy Stat Soc Ser B (Methodol) 57:289–300. https://doi.org/10.1111/j.2517-6161.1995.tb02031.x
Toffano RB, Hillesheim E, Mathias MG, Coelho-Landell CA, Salomão RG, Almada MO, Camarneiro JM, Barros TT, Camelo-Junior JS, Rezzi S (2018) Validation of the Brazilian healthy eating index-revised using biomarkers in children and adolescents. Nutrients 10:154. https://doi.org/10.3390/nu10020154
Aparicio-Ugarriza R, Cuenca-García M, Gonzalez-Gross M, Julián C, Bel-Serrat S, Moreno LA, Breidenassel C, Kersting M, Arouca AB, Michels N (2019) Relative validation of the adapted Mediterranean Diet Score for Adolescents by comparison with nutritional biomarkers and nutrient and food intakes: The Healthy Lifestyle in Europe by Nutrition in Adolescence (HELENA) study. Public Health Nutr 22:2381–2397. https://doi.org/10.1017/S1368980019001022
Jauregibeitia I, Portune K, Rica I, Tueros I, Velasco O, Grau G, Trebolazabala N, Castaño L, Larocca AV, Ferreri C (2020) Fatty acid profile of mature red blood cell membranes and dietary intake as a new approach to characterize children with overweight and obesity. Nutrients 12:3446. https://doi.org/10.3390/nu12113446
Venäläinen T, Schwab U, Ågren J, de Mello V, Lindi V, Eloranta A, Kiiskinen S, Laaksonen D, Lakka TA (2014) Cross-sectional associations of food consumption with plasma fatty acid composition and estimated desaturase activities in Finnish children. Lipids 49:467–479. https://doi.org/10.1007/s11745-014-3894-7
Lauritzen L, Harsløf LB, Hellgren LI, Pedersen MH, Mølgaard C, Michaelsen KF (2012) Fish intake, erythrocyte n-3 fatty acid status and metabolic health in Danish adolescent girls and boys. Br J Nutr 107:697–704. https://doi.org/10.1017/S0007114511002418
Eloranta A, Venäläinen T, Soininen S, Jalkanen H, Kiiskinen S, Schwab U, Lakka TA, Lindi V (2016) Food sources of energy and nutrients in Finnish girls and boys 6–8 years of age–the PANIC study. Food Nutr Res 60:32444. https://doi.org/10.3402/fnr.v60.32444
Wolters M, Pala V, Russo P, Risé P, Moreno LA, De Henauw S, Mehlig K, Veidebaum T, Molnár D, Tornaritis M (2016) Associations of whole blood n-3 and n-6 polyunsaturated fatty acids with blood pressure in children and adolescents–results from the IDEFICS/I. Family cohort PLoS One 11:e0165981. https://doi.org/10.1371/journal.pone.0165981
Laitinen TT, Nuotio J, Rovio SP, Niinikoski H, Juonala M, Magnussen CG, Jokinen E, Lagström H, Jula A, Viikari JS (2020) Dietary fats and atherosclerosis from childhood to adulthood. Pediatrics 145:e20192786. https://doi.org/10.1542/peds.2019-2786
Khakimov B, Poulsen SK, Savorani F, Acar E, Gürdeniz G, Larsen TM, Astrup A, Dragsted LO, Engelsen SB (2016) New Nordic diet versus average Danish diet: a randomized controlled trial revealed healthy long-term effects of the new Nordic diet by GC–MS blood plasma metabolomics. J Proteome Res 15:1939–1954. https://doi.org/10.1021/acs.jproteome.6b00109
Aljahdali AA, Peterson KE, Cantoral A, Ruiz-Narvaez E, Tellez-Rojo MM, Kim HM, Hébert JR, Wirth MD, Torres-Olascoaga LA, Shivappa N (2022) Diet quality scores and cardiometabolic risk factors in Mexican children and adolescents: a longitudinal analysis. Nutrients 14:896. https://doi.org/10.3390/nu14040896
Abdulla H, Smith K, Atherton PJ, Idris I (2016) Role of insulin in the regulation of human skeletal muscle protein synthesis and breakdown: a systematic review and meta-analysis. Diabetologia 59:44–55. https://doi.org/10.1007/s00125-015-3751-0
Biolo G, Williams BD, Fleming RY, Wolfe RR (1999) Insulin action on muscle protein kinetics and amino acid transport during recovery after resistance exercise. Diabetes 48:949–957. https://doi.org/10.2337/diabetes.48.5.949
Kim MJ, Park S, Yang HJ, Shin P, Hur HJ, Park S, Lee K, Hong M, Kim JH, Choi S (2022) Alleviation of dyslipidemia via a traditional balanced Korean diet represented by a low glycemic and low cholesterol diet in obese women in a randomized controlled trial. Nutrients 14:235. https://doi.org/10.3390/nu14020235
Wang W, Wu Z, Dai Z, Yang Y, Wang J, Wu G (2013) Glycine metabolism in animals and humans: implications for nutrition and health. Amino Acids 45:463–477. https://doi.org/10.1007/s00726-013-1493-1
Górska-Warsewicz H, Laskowski W, Kulykovets O, Kudlińska-Chylak A, Czeczotko M, Rejman K (2018) Food products as sources of protein and amino acids—the case of Poland. Nutrients 10:1977. https://doi.org/10.3390/nu10121977
Tang M, Weaver NE, Berman LM, Brown LD, Hendricks AE, Krebs NF (2021) Different blood metabolomics profiles in infants consuming a meat-or dairy-based complementary diet. Nutrients 13:388. https://doi.org/10.3390/nu13020388
Hamaya R, Mora S, Lawler PR, Cook NR, Buring JE, Lee IM, Manson JE, Tobias DK (2022) Association of modifiable lifestyle factors with plasma branched-chain amino acid metabolites in women. J Nutr 152(6):1515–1524. https://doi.org/10.1093/jn/nxac056
Hoppe C, Mølgaard C, Vaag A, Barkholt V, Michaelsen KF (2005) High intakes of milk, but not meat, increase s-insulin and insulin resistance in 8-year-old boys. Eur J Clin Nutr 59:393–398. https://doi.org/10.1038/sj.ejcn.1602086
Lehtovirta M, Pahkala K, Niinikoski H, Kangas AJ, Soininen P, Lagström H, Viikari JSA, Rönnemaa T, Jula A, Ala-Korpela M, Würtz P, Raitakari OT (2018) Effect of dietary counseling on a comprehensive metabolic profile from childhood to adulthood. J Pediatr 195(190–198):e3. https://doi.org/10.1016/j.jpeds.2017.11.057
Hammarqvist F, Angsten G, Meurling S, Andersson K, Wernerman J (2010) Age-related changes of muscle and plasma amino acids in healthy children. Amino Acids 39(2):359–366. https://doi.org/10.1007/s00726-009-0446-1
Eloranta AM, Schwab U, Venäläinen T, Kiiskinen S, Lakka HM, Laaksonen DE, Lakka TA, Lindi V (2016) Dietary quality indices in relation to cardiometabolic risk among Finnish children aged 6–8 years–the PANIC study. Nutr Metabol Cardiovasc Dis 26:833–841. https://doi.org/10.1016/j.numecd.2016.05.005
Berneis KK, Krauss RM (2002) Metabolic origins and clinical significance of LDL heterogeneity. J Lipid Res 43(9):1363–1379. https://doi.org/10.1194/jlr.r200004-jlr200
Ouyang X, Cirillo P, Sautin Y, McCall S, Bruchette JL, Diehl AM, Johnson RJ, Abdelmalek MF (2008) Fructose consumption as a risk factor for non-alcoholic fatty liver disease. J Hepatol 48:993–999. https://doi.org/10.1016/j.jhep.2008.02.011
Fisher EA, Ginsberg HN (2002) Complexity in the secretory pathway: the assembly and secretion of apolipoprotein B-containing lipoproteins. J Biol Chem 277:17377–17380. https://doi.org/10.1074/jbc.R100068200
Adiels M, Taskinen M, Packard C, Caslake MJ, Soro-Paavonen A, Westerbacka J, Vehkavaara S, Häkkinen A, Olofsson S, Yki-Järvinen H (2006) Overproduction of large VLDL particles is driven by increased liver fat content in man. Diabetologia 49:755–765. https://doi.org/10.1007/s00125-005-0125-z
Rosqvist F, Iggman D, Kullberg J, Cedernaes J, Johansson H, Larsson A, Johansson L, Ahlström H, Arner P, Dahlman I (2014) Overfeeding polyunsaturated and saturated fat causes distinct effects on liver and visceral fat accumulation in humans. Diabetes 63:2356–2368. https://doi.org/10.2337/db13-1622
Papandreou D, Rousso I, Malindretos P, Makedou A, Moudiou T, Pidonia I, Pantoleon A, Economou I, Mavromichalis I (2008) Are saturated fatty acids and insulin resistance associated with fatty liver in obese children? Clin Nutr 27:233–240. https://doi.org/10.1016/j.clnu.2007.11.003
Kaitosaari T, Rönnemaa T, Raitakari O, Talvia S, Kallio K, Volanen I, Leino A, Jokinen E, Välimäki I, Viikari J (2003) Effect of 7-year infancy-onset dietary intervention on serum lipoproteins and lipoprotein subclasses in healthy children in the prospective, randomized Special Turku Coronary Risk Factor Intervention Project for Children (STRIP) study. Circulation 108:672–677. https://doi.org/10.1161/01.CIR.0000083723.75065.D4
Lehtovirta M, Matthews LA, Laitinen TT, Nuotio J, Niinikoski H, Rovio SP, Lagström H, Viikari JS, Rönnemaa T, Jula A (2021) Achievement of the targets of the 20-year infancy-onset dietary intervention—association with metabolic profile from childhood to adulthood. Nutrients 13:533. https://doi.org/10.3390/nu13020533
Pedrosa C, Oliveira B, Albuquerque I, Simões-Pereira C, Vaz-de-Almeida MD, Correia F (2011) Markers of metabolic syndrome in obese children before and after 1-year lifestyle intervention program. Eur J Nutr 50:391–400. https://doi.org/10.1007/s00394-010-0148-1
Giannini C, Diesse L, D’adamo E, Chiavaroli V, De Giorgis T, Di Iorio C, Chiarelli F, Mohn A (2014) Influence of the Mediterranean diet on carotid intima–media thickness in hypercholesterolaemic children: a 12-month intervention study. Nutr Metab Cardiovasc Dis 24:75–82. https://doi.org/10.1016/j.numecd.2013.04.005
Marcovina S, Packard CJ (2006) Measurement and meaning of apolipoprotein AI and apolipoprotein B plasma levels. J Intern Med 259:437–446. https://doi.org/10.1111/j.1365-2796.2006.01648.x
Gugliucci A, Lustig RH, Caccavello R, Erkin-Cakmak A, Noworolski SM, Tai VW, Wen MJ, Mulligan K, Schwarz J (2016) Short-term isocaloric fructose restriction lowers apoC-III sas and yields less atherogenic lipoprotein profiles in children with obesity and metabolic syndrome. Atherosclerosis 253:171–177. https://doi.org/10.1016/j.atherosclerosis.2016.06.048
Acknowledgements
We are grateful to all children and their parents and caregivers who have participated in the PANIC study. We are also indebted to all members of the PANIC research team for their invaluable contribution in the acquisition of the data throughout the study.
Funding
Open access funding provided by University of Eastern Finland (including Kuopio University Hospital). The PANIC study has been supported financially by grants from Ministry of Education and Culture of Finland, Ministry of Social Affairs and Health of Finland, Academy of Finland, Research Committee of the Kuopio University Hospital Catchment Area (State Research Funding), Finnish Innovation Fund Sitra, Social Insurance Institution of Finland, Finnish Cultural Foundation, Foundation for Paediatric Research, Diabetes Research Foundation in Finland, Finnish Foundation for Cardiovascular Research, Juho Vainio Foundation, Paavo Nurmi Foundation, Yrjö Jahnsson Foundation, and the city of Kuopio. The funding sources have not been involved in designing the study, collecting the data, analyzing the data, interpreting the results, writing the manuscript, or deciding to submit the paper for publication.
Author information
Authors and Affiliations
Contributions
SL wrote the draft of the manuscript and conducted the statistical analyses. SL, AME, and EAL formulated the research question. TAL designed the study and received funding. SL, AME, EAL, US, TS, and TAL interpreted the findings, contributed to the critical review of the manuscript, and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no competing interests to declare that are relevant to the content of this article.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Laamanen, S.E., Eloranta, AM., Haapala, E.A. et al. Associations of diet quality and food consumption with serum biomarkers for lipid and amino acid metabolism in Finnish children: the PANIC study. Eur J Nutr 63, 623–637 (2024). https://doi.org/10.1007/s00394-023-03293-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00394-023-03293-8