Abstract
Purpose
Enhanced responsiveness to external triggers is thought to reflect hypersensitivity of the cough reflex. It may involve an enhanced sensitivity of the afferent nerves in the airways and/or an abnormal processing of the afferent information by the central nervous system (CNS). The CNS processing of cough has been shown to involve the same regions as those in symptom amplification, a phenomenon that often manifests as multiple symptoms. The main purpose of the present study was to define whether the presence of several cough triggers is associated with multiple symptoms.
Methods
2131 subjects with current cough responding to two email surveys filled in a comprehensive questionnaire about social background, lifestyle, general health, doctors’ diagnoses and visits, symptoms, and medication. Multiple symptoms was defined as three or more non-respiratory, non-mental symptoms.
Results
A carefully controlled multiple regression analysis revealed that the number of cough triggers was the only cough characteristic associating with multiple non-respiratory, non-mental symptoms [aOR 1.15 (1.12–1.19) per one trigger, p < 0.001]. Among the 268 subjects with current cough both in the first survey and in the follow-up survey 12 months later, the repeatability of the trigger sum was good with an intraclass correlation coefficient of 0.80 (0.75–0.84).
Conclusion
The association between the number of the cough triggers and multiple symptoms suggests that the CNS component of cough hypersensitivity may be a manifestation of non-specific alteration in the CNS interpretation of various body sensations. The number of cough triggers is a repeatable measure of cough sensitivity.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Coughing in response to low levels of thermal, mechanical, or chemical triggers is nowadays regarded as the key clinical element in chronic cough. It is in the center of the concept ‘cough hypersensitive syndrome’ which has been suggested as an overarching diagnosis irrespective of the possible cough background disorders [1]. Furthermore, two recent data-driven analyses have revealed two cough phenotypes, the phenotype TBQ (several Triggers of cough, many cough Background disorders, and poor cough-related Quality of life), and the common phenotype. The presence of several triggers of cough was the most important cough characteristic to separate the two cough phenotypes. This phenotype can be recognized also among subjects with acute and subacute cough, in addition to chronic cough [2, 3].
Enhanced responsiveness to external triggers is thought to reflect hypersensitivity of the cough reflex arc [4, 5]. It may involve an enhanced sensitivity of the afferent nerves in the airways and/or abnormal processing of the afferent information by the central nervous system (CNS) [6]. The involvement of CNS is supported by functional brain imaging studies utilizing airway irritation by capsaicin, a potent cough provocation agent, which have found activity in right inferior frontal gyrus, insula, prefrontal cortex, and anterior and mid-cingulate cortex of the CNS [7,8,9]. Of note, patients with chronic cough manifest alterations in the function of these brain regions when compared to healthy subjects [7].
In our previous community-based studies, subjects with cough often reported multiple non-respiratory symptoms [10, 11]. This feature led to biologically unplausible, odd associations. For example, chronic cough was statistically significantly associated with toothache, headache, constipation etc. Presence of multiple symptoms is a key characteristic of a phenomenon called symptom amplification. It is a process whereby patients’ thoughts, emotions, and concerns heighten uncomfortable bodily sensations and symptoms, making them more salient, intense, unpleasant, and distressing. The mechanism behind this phenomenon is probably an altered processing of interoceptive stimuli in brain regions that impart cognitive and affective dimensions to physical sensations [12]. Of particular interest, these regions strongly overlap with those participating in the processing of cough-evoking stimulation by capsaicin. These findings suggest a mechanistic link between symptom amplification and cough hypersensitivity.
The main purpose of the present study was to define whether the presence of several cough triggers is associated with multiple non-respiratory symptoms. The second purpose was to investigate the long-term repeatability of the cough triggers.
Materials and Methods
Population
The population of the present study consists of the responders to two large population-based e-mail surveys originally planned to investigate the risk factors, prevalence, characteristics, and consequences of cough (Fig. 1) [10, 11]. The first survey was performed in spring 2017. The target population was the public service employees of two middle-sized towns in central Finland, Jyväskylä and Kuopio. The response rate was 26% [10]. The second one was performed in spring 2021. The target population was the members of the Finnish Pensioner’s Federation. The response rate was 24% [11]. In both studies, the sex and age distribution of the respondents closely resembled those of the target populations. Altogether, there were 9865 well-characterized subjects with an age range of 18–94 years (Table 1).
The subjects with current cough (cough within the past 2 weeks) in the spring 2017 survey were sent a shorter follow-up questionnaire 12 months later. The response rate was now 75.3%. The triggers were asked, again, and the repeatability of the trigger sum and individual triggers were thereby analyzed. Of note, the cough was not necessarily continuous throughout the 12 months’ period but was interrupted in many subjects.
The decision to respond was considered as an informed consent. The study was approved by the Ethics Committee of Kuopio University Hospital (289/2015). Permissions to conduct the surveys were obtained from the officials of Jyväskylä and Kuopio and from the Finnish Pensioners’ Federation.
The Questionnaires
The questionnaires of the surveys in 2017 and 2021 were almost identical but slightly modified due to the differences in the two populations’ age and employment status [10, 11]. Both questionnaires included questions about social background, lifestyle, general health, doctors’ diagnoses and visits, symptoms and medication. Validated questions for depressive symptoms were included [13]. Respondents with current cough answered additional cough-related questions including the Leicester Cough Questionnaire [14]. All surveys included a list of ten external triggers (Table 2) and the year 2021 survey also four allotussia-type triggers (supplementary Information, Table 1).
Definitions
Multiple non-respiratory, non-mental symptoms (MNNS) was defined as presence of at least three symptoms from a list consisting of 10 common non-respiratory, non-mental symptoms: chest pain on exertion, aching joints, back pain, sciatica (back pain that radiates to the leg), toothache, swollen feet, headache, constipation, other gut problems (flatulence, diarrhea), and urinary problems. The cut-off value three was chosen to indicate the highest tertile in the total population of 9865 subjects. Disorder sum was calculated by summing up the reported disorders diagnosed by a doctor from a list of 15 non-respiratory, non-mental disorders (supplementary Information, Table 2). Trigger sum was calculated by summing up the reported cough triggers from a list of the ten external triggers, which were asked in all surveys (Table 2). Sputum production was assessed utilizing the LCQ question about it (In the last 2 weeks, have you been bothered by sputum (phlegm) production when you cough? There were 7 alternative answers from ‘every time’ to ‘never’). Presence of depressive symptoms was defined as a Patient Health Questionnaire-2 score of three or more [13].
Statistical Analysis
Mann–Whitney U test or chi-square test were used to compare the groups. The multivariate analyses were conducted using binary logistic regression with a backward directed stepwise process. The dependent variable was the presence of MNNS. The independent variables were chosen on the basis of statistically significant association with MNNS in the bivariate analysis. The repeatability of the cough triggers and the trigger sum were analyzed utilizing the intraclass correlation coefficient, Bland–Altman plot and the Kappa value [15, 16]. SPSS software version 26.0 (IBM SPSS Statistics for Windows) was utilized and a p-value < 0.05 was considered statistically significant. The data is expressed as means and 95% confidence intervals (CI) unless stated otherwise.
Results
In the total population, the 7162 subjects without MNNS and the 2703 subjects with MNNS differed in many respects (Table 1). Also, among the 2131 subjects with current cough, those with and without MNNS differed significantly in terms of many background characteristics in the bivariate analyses (Table 3). Of the various cough characteristics, sputum production, presence of any cough trigger, trigger sum, and the LCQ scores differed significantly between the groups. On the contrary, cough bout frequency and the length of the current cough episode did not associate with MNNS. The subjects with MNNS visited a doctor due to cough more often than those without.
The multivariate analysis among the subjects with current cough included the following independent variables: body mass index, gender, presence of depressive symptoms, disorder sum, sputum production, trigger sum, and LCQ total score. Trigger sum was the only cough characteristic that showed an independent, statistically significant association with MNNS (Table 4, Fig. 2). When trigger sum was replaced by the presence of any trigger in this model, its adjusted OR was 1.58 (1.26–1.99, p < 0.001). All ten external triggers were also statistically significantly associated with MNNS (Table 2). The four allotussia-type triggers, which were used only in the year 2021 survey, were less strongly associated with MNNS (Supplementary Information, Table 1).
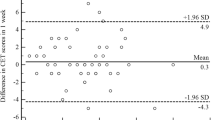
Among the 268 subjects with current cough both in the spring 2017 survey and in the survey 12 months later, the repeatability of the trigger sum was good with an intraclass correlation coefficient of 0.80 (0.75–0.84). The repeatability was not affected by the number of reported triggers (Fig. 3). The Kappa value for any trigger was 0.30. The kappa values for all triggers are expressed in Table 2.
A Bland–Altman plot to demonstrate the repeatability of the trigger sum in 12 months’ interval among 268 subjects with current cough at both time-points. The difference of the first trigger sum minus the second trigger sum is plotted against the mean of the two trigger sums. The two horizontal lines indicate mean ± 1.96 SD of the difference between the trigger sums
Discussion
In this large, community-based population with a wide age range, the number of cough triggers showed a statistically highly significant, independent association with MNNS. This finding supports the idea that cough responsiveness to several triggers may share similar pathophysiological mechanisms with symptom amplification. Furthermore, the present study for the first time reports the good long-term repeatability of the trigger sum.
As mentioned, airway irritation by capsaicin evokes activity in right inferior frontal gyrus, insula, prefrontal cortex, and anterior and mid-cingulate cortex of the CNS and patients with chronic cough manifest alterations in the function of these brain regions when compared to healthy subjects [7,8,9]. These CNS regions are also activated by a wide range of other body sensations like desire to void in response to bladder dilatation [17], pain, discomfort and urge-to-defecate by rectal distension [18], pain due to esophageal distension [19], breathlessness sensation due to resistive inspiratory loading [20], and palpitation sensation induced by isoproterenol [21]. In addition, these regions show abnormal activation among subjects with medically unexplained symptoms, i.e., somatoform disorders [12]. They impart cognitive and affective dimensions to body sensations and an altered function of them is thought to be the key pathophysiological basis for symptom amplification [12]. Clinically, this may manifest, for example, as a disproportionate symptom stress, non-specific medication side effects, and symptoms that persist despite adequate medical therapy. One of the cardinal features of symptom amplification is multiplicity of symptoms [12, 22]. We propose that the presented strong association between multiple cough triggers and MNNS indicate overlapping interpretative CNS mechanisms in cough reflex hypersensitivity and symptom amplification.
Of note, the list of symptoms to define MNNS did not include respiratory or mental symptoms, to exclude any plausible biological mechanisms that could link the symptoms with cough. For example, wheezing is typical for asthma, which also causes prolonged cough [10]. Depressive symptoms, in turn, can develop in long-standing cough as a secondary phenomenon [23]. Thus, it is unlikely that the symptoms that defined MNNS can relate to multiple cough triggers by other mechanisms than an abnormal CNS interpretation of body sensations, i.e., symptom amplification.
To the best of our knowledge, there are no previous studies investigating the repeatability of the reported cough triggers. In the present study, the long-term repeatability of trigger sum was good when measured twice 12 months apart. Furthermore, the repeatability was unaffected by the number of triggers. On the contrary, the repeatability of individual triggers was unsatisfactory, suggesting that the sum of triggers is the preferable way to clinically evaluate cough sensitivity. It seems that the subjective importance of individual cough triggers may vary with time but the tendency to experience them, as expressed in the trigger sum, is more constant.
An enhanced responsiveness to cough triggers is widely considered a clinical correlate for cough hypersensitivity [4, 5] and in the present study, the number of subject-reported cough triggers was assumed to reflect the degree of cough reflex arc hypersensitivity. However, the objective evidence about this association is scarce. Strong scents [24,25,26,27,28,29] and cold air [30] as cough triggers are associated with an enhanced cough response to capsaicin. In addition, objectively documented coughing in response to deep inspiration is associated with cough hypersensitivity to citric acid [31]. However, we are unaware about studies correlating the number of cough triggers with the degree of objectively measured cough sensitivity. Thus, the number of cough triggers needs more validating to be used as a clinical correlate for cough reflex arc hypersensitivity.
Another shortcoming in the present study is that the allotussia-type triggers were only asked in the year 2021 survey and were therefore not included in the trigger sum. However, these triggers were found to be less associated with MNNS than the external triggers and therefore, this shortcoming may not invalid the findings. Low response rate may also be regarded as a weakness. It is possible that subjects with more troublesome cough are over-represented. However, the age and the gender distributions of the responders resembled closely those of the original populations, as reported before [10, 11]. Finally, our study was a survey and objective tests to measure cough reflex sensitivity were not included.
The strengths of the present study include the large, well characterized population. Most of the characteristics that have shown to associate with multiple symptoms could be considered in the multivariate models and therefore, the independent association between the trigger sum and MNNS could be demonstrated with a great certainty. In particular, many other studies have not controlled adequately for general medical disorders. They form an important predictor of multiple symptoms: the more general medical disorders a person has, the more symptoms he/she probably reports [32]. This was considered in the present study by utilizing the variable ‘Disorder sum’.
In conclusion, the strong association between the number of the reported cough triggers and MNNS suggests that the CNS component of cough hypersensitivity may not be a specific phenomenon for cough, but a manifestation of a wider, non-specific alteration in the CNS interpretation of various body sensations, i.e., symptom amplification. This hypothesis is supported by functional brain imaging studies that have revealed overlapping functional CNS regions involved in the phenomena [7,8,9, 12]. This finding may provide a novel treatable trait for chronic cough, since subjects with symptom amplification have benefited from chronic disease management programs, cognitive behavior therapy, and psychotherapy [12]. The association of the cough hypersensitivity with symptom amplification may also clarify the mechanisms of the extraordinary high placebo effect of cough medications and the relatively modest effect of the new P2X3 antagonists, which act on peripheral afferent nerves only [33].
References
Morice AH, Millqvist E, Belvisi MG et al (2014) Expert opinion on the cough hypersensitivity syndrome in respiratory medicine. Eur Respir J 44:1132–1148
Koskela HO, Selander TA, Lätti AM (2020) Cluster analysis in 975 patients with current cough identifies a phenotype with several cough triggers, many background disorders, and low quality of life. Respir Res 21:219
Koskela HO, Kaulamo JT, Selander TA et al (2022) Validation of the cough phenotype TBQ among elderly Finnish subjects. ERJ Open Res 8:00284–02022
Chung KF, McGarvey L, Song W-J et al (2022) Cough hypersensitivity and chronic cough. Nat Rev Dis Primers 8:45
Sundar KM, Stark AC, Hu N et al (2021) Is laryngeal hypersensitivity the basis of unexplained or refractory chronic cough? ERJ Open Res 7:00793–02020
Turner RD, Birring SS (2019) Chronic cough: ATP, afferent pathways and hypersensitivity. Eur Respir J 54:1900889
Ando A, Smallwood D, McMahon M et al (2016) Neural correlates of cough hypersensitivity in humans: evidence for central sensitisation and dysfunctional inhibitory control. Thorax 71:323–329
Farrell MJ, Cole LJ, Chiapoco D et al (2012) Neural correlates coding stimulus level and perception of capsaicin-evoked urge-to-cough in humans. Neuroimage 61:1324–1335
Mazzone SB, Cole LJ, Ando A et al (2011) Investigation of the neural control of cough and cough suppression in humans using functional brain imaging. J Neurosci 31:2948–2958
Lätti AM, Pekkanen J, Koskela HO (2018) Defining the risk factors for acute, subacute and chronic cough: a cross-sectional study in a Finnish adult employee population. BMJ Open 8:e022950
Kaulamo JT, Lätti AM, Koskela HO (2022) Cough in the elderly during the COVID-19 pandemic. Lung 200:161–168
Barsky AJ, Silbersweig DA (2023) The amplification of symptoms in the medically ill. J Gen Intern Med 38:195–202
Kroenke K, Spitzer RL, Williams JB (2003) The Patient Health Questionnaire-2: validity of a two-item depression screener. MedCare 41:1284–1292
Birring SS, Prudon B, Carr AJ et al (2003) Development of a symptom specific health status measure for patients with chronic cough: Leicester Cough Questionnaire (LCQ). Thorax 58:339–343
Bland JM, Altman DG (1990) A note on the use of the intraclass correlation coefficient in the evaluation of agreement between two methods of measurement. Comput Biol Med 20:337–340
McHugh ML (2012) Interrater reliability: the kappa statistic. Biochem Med (Zagreb) 22:276–282
Jarrahi B, Mantini D, Balsters JH et al (2015) Differential functional brain network connectivity during visceral interoception as revealed by independent component analysis of fMRI TIME-series. Hum Brain Mapp 36:4438–4468
Rubio A, Van Oudenhove L, Pellissier S et al (2015) Uncertainty in anticipation of uncomfortable rectal distension is modulated by the autonomic nervous system—a fMRI study in healthy volunteers. Neuroimage 107:10–22
Hobson AR, Aziz Q (2004) Brain processing of esophageal sensation in health and disease. Gastroenterol Clin N Am 33:69–91
Faull OK, Hayen A, Pattinson KTS (2017) Breathlessness and the body: neuroimaging clues for the inferential leap. Cortex 95:211–221
Khalsa SS, Rudrauf D, Feinstein JS et al (2009) The pathways of interoceptive awareness. Nat Neurosci 12:1494–1496
Kroenke K (2014) A practical and evidence-based approach to common symptoms: a narrative review. Ann Intern Med 161:579–586
Sohn K-H, Song W-J, Kim S-H et al (2019) Chronic cough, not asthma, is associated with depression in the elderly: a community-based population analysis in South Korea. Korean J Intern Med 34:1363–1371
Johansson A, Löwhagen O, Millqvist E et al (2002) Capsaicin inhalation test for identification of sensory hyperreactivity. Respir Med 96:731–735
Millqvist E, Johansson A, Bende M (2004) Relationship of airway symptoms from chemicals to capsaicin cough sensitivity in atopic subjects. Clin Exp Allergy 34:619–623
Millqvist E, Ternesten-Hasséus E, Ståhl A et al (2005) Changes in levels of nerve growth factor in nasal secretions after capsaicin inhalation in patients with airway symptoms from scents and chemicals. Environ Health Perspect 113:849–852
Elberling J, Dirksen A, Johansen JD et al (2006) The capsaicin cough reflex in eczema patients with respiratory symptoms elicited by perfume. Contact Derm 54:158–164
Holst H, Arendt-Nielsen L, Mosbech H et al (2010) The capsaicin cough reflex in patients with symptoms elicited by odorous chemicals. Int J Hyg Environ Health 213:66–71
Pullerits T, Ternesten-Hasseus E, Johansson EL et al (2014) Capsaicin cough threshold test in diagnostics. Respir Med 108:1371–1376
Song W-J, Kim J-Y, Jo E-J et al (2014) Capsaicin cough sensitivity is related to the older female predominant feature in chronic cough patients. Allergy Asthma Immunol Res 6:401–408
Koskela HO, Nurmi HM, Song W-J (2021) Deep inspiration-provoked cough: a sign of cough reflex arc hypersensitivity. Lung 199:501–505
Creed F (2022) The predictors of somatic symptoms in a population sample: the lifelines cohort study. Psychosom Med 84:1056–1066
McGarvey LP, Birring SS, Morice AH et al (2022) Efficacy and safety of gefapixant, a P2X3 receptor antagonist, in refractory chronic cough and unexplained chronic cough (COUGH-1 and COUGH-2): results from two double-blind, randomised, parallel-group, placebo-controlled, phase 3 trials. Lancet 399:909–923
Acknowledgements
We thank Seppo Hartikainen from Istekki for the assistance in modifying the electronic questionnaire.
Funding
Open access funding provided by University of Eastern Finland (UEF) including Kuopio University Hospital. This study was supported by Kuopion Seudun Hengityssäätiö and Hengityssairauksien Tutkimussäätiö foundations.
Author information
Authors and Affiliations
Contributions
Substantial contribution to the conception and design of the work: all authors. Acquisition, analysis, or interpretation of data for the work: all authors. Drafting the article or revising it critically for important intellectual content: all authors. Agreement to be accountable for all aspects of the work, in ensuring that questions related to the accuracy and integrity of any part of the work are appropriately investigated and resolved: all authors.
Corresponding author
Ethics declarations
Competing interests
Heikki O Koskela has received funding for the present study from Kuopion Seudun Hengityssäätiö and Hengityssairauksien Tutkimussäätiö Foundations, payments for lectures from Boehringer Ingelheim and MSD, and owns shares of a medical company Orion. Johanna T Kaulamo has received funding for the present study from Kuopion Seudun Hengityssäätiö, Hengityssairauksien Tutkimussäätiö, Suomen Tuberkuloosin vastustamisyhdistyksen Säätiö, Väinö ja Laina Kiven Säätiö, and Suomen Kulttuurirahasto foundations, and travel support from Boehringer-Ingelheim for attending a scientific meeting. Anne M Lätti has received funding for the present study from Kuopion Seudun Hengityssäätiö, Hengityssairauksien Tutkimussäätiö, KYS:n Tutkimussäätiö, Suomen Tuberkuloosin Vastustamisyhdistyksen Säätiö, and Väinö ja Laina Kiven Säätiö Foundations, travel support from Orion, Boehringer Ingelheim and Roche for attending a scientific meeting, and payment for lectures and Advisory Board participations from Farmasian oppimiskeskus, MSD and GlaxoSmithKline. The authors have no other financial or non-financial competing interests.
Ethics Approval
This study was performed in line with the principles of the Declaration of Helsinki. Approval was granted by the Ethics Committee of Kuopio University Hospital (289/2015).
Consent to Participate
The decision to respond to the questionnaire was considered as an informed consent.
Consent to Publish
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Koskela, H.O., Kaulamo, J.T. & Lätti, A.M. Cough Sensitivity to Several External Triggers is Associated with Multiple Non-respiratory Symptoms. Lung 201, 267–274 (2023). https://doi.org/10.1007/s00408-023-00622-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00408-023-00622-w