Abstract
Purpose
Bronchiectasis is predominantly marked by neutrophilic inflammation. The relevance of type 2 biomarkers in disease severity and exacerbation risk is poorly understood. This study explores the clinical significance of these biomarkers in bronchiectasis patients.
Methods
In a cross-sectional cohort study, bronchiectasis patients, excluding those with asthma or allergic bronchopulmonary aspergillosis, underwent clinical and radiological evaluations. Bronchoalveolar lavage samples were analyzed for cytokines and microbiology. Blood eosinophil count (BEC), serum total immunoglobulin E (IgE), and fractional exhaled nitric oxide (FeNO) were measured during stable disease states. Positive type 2 biomarkers were defined by established thresholds for BEC, total IgE, and FeNO.
Results
Among 130 patients, 15.3% demonstrated BEC ≥ 300 cells/μL, 26.1% showed elevated FeNO ≥ 25 ppb, and 36.9% had high serum total IgE ≥ 75 kU/L. Approximately 60% had at least one positive type 2 biomarker. The impact on clinical characteristics and disease severity was variable, highlighting BEC and FeNO as reflective of different facets of disease severity and exacerbation risk. The combination of low BEC with high FeNO appeared to indicate a lower risk of exacerbation. However, Pseudomonas aeruginosa colonization and a high neutrophil-to-lymphocyte ratio (NLR ≥ 3.0) were identified as more significant predictors of exacerbation frequency, independent of type 2 biomarker presence.
Conclusions
Our study underscores the distinct roles of type 2 biomarkers, highlighting BEC and FeNO, in bronchiectasis for assessing disease severity and predicting exacerbation risk. It advocates for a multi-biomarker strategy, incorporating these with microbiological and clinical assessments, for comprehensive patient management.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Bronchiectasis, a chronic respiratory disease, presents with cough, sputum production, dyspnea, and recurrent exacerbations often due to infections [1]. Historically, bronchiectasis was primarily linked to neutrophilic airway inflammation [2,3,4]. However, there has been a growing interest in understanding the role of eosinophilic inflammation in bronchiectasis [5, 6]. European cohort studies found 20% of patients have peripheral blood eosinophilia (≥ 300 eosinophils/μL) without comorbidities, termed “eosinophilic bronchiectasis” [6]. An Italian pilot study revealed a T2 high endotype—characterized by blood eosinophil counts (BEC) ≥ 300 cells/μL or fractional exhaled nitric oxide (FeNO) ≥ 25 ppb—in 31% of patients, associated with severe disease and significant quality of life impact [7]. Additionally, a Chinese study identified high serum total immunoglobulin E (IgE) (> 60 kU/L) or BEC (≥ 150 cells/µL) in 68.8% of patients, correlating with disease severity and radiological extent [8].
Recent strategies emphasize airway inflammation heterogeneity and biologics in asthma therapy [9]. This shift has led to a greater focus on evaluating composite Type 2 biomarkers, which include elevated serum IgE levels (≥ 75 kU/L), BEC (≥ 300 cells/μL), and FeNO values (≥ 25 ppb) [10]. In chronic obstructive pulmonary disease (COPD), despite less conclusive evidence, these biomarkers are instrumental for clinical outcome assessments [11,12,13,14] and in predicting responses to inhaled corticosteroids (ICS) [15] and biologics [16]. A multicenter European study in bronchiectasis linked inflammatory and microbial profiles, enhancing exacerbation prediction and biomarker development for targeted treatments [17].
Currently, the relationship between Type 2 biomarkers, clinical characteristics, disease severity, and related clinical outcomes in bronchiectasis remains poorly defined. Our study aims to explore the prevalence of positive Type 2 biomarkers and their clinical implications in East Asian individuals with bronchiectasis. To this end, we assessed three commonly used clinical markers of Type 2 inflammation: blood eosinophils, FeNO, and serum total IgE in bronchiectasis patients. Additionally, we analyzed airway microbiology and inflammation by examining bronchoalveolar lavage (BAL) samples to determine the clinical relevance of these factors in relation to disease severity and clinical outcomes.
Material and Methods
Study Design and Participants
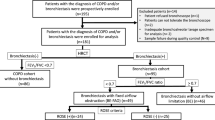
Patients with bronchiectasis were prospectively enrolled from November 2018 to February 2023 at the National Taiwan University Hospital (NTUH), Yunlin Branch, Taiwan. Inclusion criteria were clinical stability and bronchiectasis diagnosis confirmed by high-resolution computed tomography (HRCT), showing a bronchoarterial ratio > 1, absence of tapering, airway visibility within 1 cm of the pleural surface [18], and symptoms like cough, dyspnea, chronic sputum production, and recurrent respiratory infections [1]. Exclusion criteria included a diagnosis of asthma or allergic bronchopulmonary aspergillosis (ABPA) [19], recent exacerbations requiring antibiotics or hospitalization within the last 3 months, or fever or acute infections in the previous 4 weeks.
At enrollment, information regarding comorbidities, past exacerbations, current inhalation medications, and exacerbation history was collected. Exacerbation severity was classified based on treatment: moderate exacerbations required outpatient antibiotics or additional therapies, while severe exacerbations necessitated hospitalization or emergency department visits for airway diseases [20]. Each patient was followed up at intervals of 3–6 months. All exacerbation outcomes, laboratory data, and imaging studies are systematically documented by the attending physicians. The thresholds for positive Type 2 biomarkers were defined as follows: BEC ≥ 300 cells/μL, serum total IgE ≥ 75 kU/L with features of atopy, and FeNO ≥ 25 ppb, in accordance with previous definitions [9, 10, 13]. The NTUH Research Ethics Committee approved this study (NTUH-REC No. 201712075RINA, 201910082RINA, and 202110093RIND).
BAL Sample Collection
BAL sample collection and analysis were standardized. Participants fasted for 4 h before the procedure, and performed oral gargling with 20 mL 0.9% saline, followed by 0.12% chlorhexidine mouthwash. Under topical anesthesia and sedation, a bronchoscope was inserted to the selected lung region, and BAL was conducted using 200 mL of 0.9% saline [21]. The samples were processed within 2 h for analysis.
BAL Sample for Analysis of Immune Cells and Inflammatory Cytokines
For immune and cytokine profiling, the retrieved BAL fluid was strained and cells were isolated for monoclonal antibody staining (including CD14, CD15, CD16, CD45, CD49d, CD80, CD206, CD294, CD163, CD193). Post-staining, cells underwent flow cytometry for surface antigen analysis [22]. BAL supernatants were assessed for inflammatory markers (TNF-α, IL-1β, IL-6, IL-8) with multiplex immunoassays and Luminex technology [23].
Assessment of Severity of Bronchiectasis
Chest high-resolution computed tomography (HRCT) scans were reviewed by a trained chest specialist and a thoracic radiologist, both of whom were blinded to the clinical data. Radiological severity of bronchiectasis was quantified using a modified Reiff score, with a range from 1 (minimum) to 18 (maximum) for lobar involvement assessment [24]. The multidimensional bronchiectasis severity index (BSI) [25] and E-FACED score, encompassing exacerbation, forced expiratory volume in 1 s (FEV1), age, chronic colonization, disease extension, and dyspnea [26], were also utilized to comprehensively define bronchiectasis severity.
Statistical Analysis
Continuous variables were represented by medians and interquartile range (IQR), with t-tests and Mann–Whitney tests comparing parametric and nonparametric groups, respectively. Categorical variables underwent chi-square or Fisher’s exact tests, as appropriate. For multiple group analyses, the Kruskal–Wallis test was used for continuous variables, while chi-squared or Fisher’s exact tests were used for categorical data. Spearman’s test assessed correlations between quantitative variables. Receiver operating characteristic (ROC) curves evaluated the diagnostic accuracy of clinical variables for predicting moderate to severe exacerbations. Kaplan–Meier curves depicted exacerbation-free survival, with the log-rank test for comparison. Cox’s proportional hazards model, adjusted for significant variables, was applied in the multivariable analysis of exacerbations. A two-sided p-value < 0.05 indicated statistical significance. Analyses were performed using SPSS (version 18.0, IBM).
Results
Prevalence and Correlation of Type 2 Biomarkers in Patients with Bronchiectasis
A total of 145 patients with stable bronchiectasis were enrolled. Five patients could not cooperate with the bronchoscope examination, four patients had co-existing asthma, five patients lacked FeNO data, and one patient suspected of ABPA were excluded. In this analysis of 130 bronchiectasis patients, 15.3% (n = 20) had BEC ≥ 300 cells/µL, 26.1% (n = 34) exhibited FeNO levels ≥ 25 ppb, and 36.9% (n = 48) had serum total IgE levels ≥ 75 kU/L. Patients were classified into eight subgroups based on each biomarker’s positivity or negativity, as shown in Fig. 1A. Notably, 60.9% (n = 79) tested positive for at least one biomarker. A significant positive correlation was found between blood eosinophil and serum total IgE levels (r = 0.324, p < 0.001), which did not extend to their correlation with FeNO levels (r = 0.065, p = 0.459). The relationship between serum total IgE and FeNO levels was marginally negative (r = − 0.033, p = 0.708), as depicted in Fig. 1B–D.
A overlap of baseline positivity of three clinical used biomarkers, BEC ≥ 300 cells/uL, FeNO ≥ 25 ppb, total IgE ≥ 75 kU/L in the bronchiectasis cohort (n = 130). B–D the correlation between BEC, FeNO and serum total IgE in the bronchiectasis cohort. Spearman’s test assessed correlations between quantitative variables. BEC blood eosinophil counts, FeNO fractional exhaled nitric oxide, IgE immunoglobulin E
Clinical Profiles of Bronchiectasis Patients by Type 2 Biomarkers
Table 1 summarizes the demographics and clinical characteristics of the bronchiectasis cohort, sorted by BEC, FeNO, and IgE levels. The cohort’s median age was 67.6, predominantly male (56.9%) and nonsmoking (60.7%). Elevated BEC (≥ 300 cells/µL) was linked to a higher proportion of males (85.0%, p = 0.006), increased body mass index (BMI) (25.2, p < 0.001), and a higher percentage of smokers (65.0%, p = 0.01). The high FeNO group showed an elevated BMI (23.2, p = 0.005) but no correlation with smoking. Increased IgE levels were associated with more males (70.8%, p = 0.014) and smokers (52.1%, p = 0.022). Symptomatic and etiological profiles were consistent across biomarker stratifications. Table 2 indicates that higher IgE levels correlated with lower FEV1/FVC ratios (p = 0.003). BEC ≥ 300 cells/µL showed a trend towards reduced FEV1/FVC (p = 0.054), while high FeNO was associated with normal lung function (p = 0.033) and less radiological severity (p = 0.012). Cardiovascular disease was more common in patients with high FeNO (p = 0.024), and allergic rhinitis was notably prevalent in those with increased BEC (p = 0.001). Treatment patterns varied, with patients having high FeNO less often on dual bronchodilator therapy (p = 0.017); however, no significant differences in ICS or prophylactic macrolide use were observed across the biomarker stratifications.
Laboratory, Microbiological, and Clinical Outcomes by Type 2 Biomarkers in Bronchiectasis
Table 3 delineates the laboratory and microbiological findings in bronchiectasis patients, categorized by Type 2 biomarker status. Patients with BEC ≥ 300 cells/μL exhibited elevated IgE levels (p < 0.001, p = 0.035). Elevated BEC were also observed in those with IgE ≥ 75 kU/L (p < 0.001, p = 0.004). In contrast, FeNO ≥ 25 ppb correlated with lower neutrophil counts (p = 0.035), with no significant correlation to BEC or IgE, aligning with the findings depicted in Fig. 1B–D. Furthermore, a higher percentage of BAL lymphocytes was observed in patients with FeNO ≥ 25 ppb (p = 0.015), alongside significantly diminished cytokine concentrations (IL-1β, IL-6, IL-8, TNF-α). However, no notable differences were found in BAL macrophage, neutrophil, or eosinophil counts across biomarker statuses. Correlational analysis of BAL eosinophils with FeNO, BEC, and serum IgE revealed weak and statistically insignificant relationships. FeNO was marginally inversely related to BAL eosinophils (r = − 0.138), while BEC and IgE had slight positive correlations (r = 0.113 and r = 0.094, respectively), all lacking statistical significance (p > 0.05) as illustrated in Fig. S1. Predominant pathogens included Klebsiella pneumoniae (33.0%), Pseudomonas aeruginosa (26.9%), and others, with their prevalence consistent irrespective of biomarker levels. Clinically, the BEC ≥ 300 cells/μL group showed a trend towards more frequent exacerbations, nearing significance (p = 0.050), whereas patients with FeNO ≥ 25 ppb experienced significantly fewer exacerbations (p = 0.048).
Correlations of Type 2 Biomarkers with Inflammatory Markers and Disease Severity in Bronchiectasis
Figure 2 illustrates the relationships between Type 2 biomarkers, inflammatory markers, immunological profiles, and bronchiectasis severity. BEC showed a positive correlation with severity, notably with E-FACED (r = 0.190, p = 0.03) and modified Reiff scores (r = 0.222, p = 0.011). In contrast, FeNO was negatively correlated with severity, significant only for the modified Reiff score (r = − 0.181, p = 0.04). Serum total IgE levels did not significantly correlate with severity scores. Additionally, local (BAL IL-1β, IL-6, IL-8, TNF-α) and systemic markers [C-reactive protein (CRP), neutrophil-to-lymphocyte ratio (NLR), and neutrophils] demonstrated stronger positive correlations with severity than Type 2 biomarkers (Table S1).
The correlations between Type 2 biomarkers, inflammatory markers, and immunological profiles with the severity scores in bronchiectasis. Spearman’s test assessed correlations between quantitative variables. The presence of asterisks indicates the level of statistical significance, with (*) denoting p < 0.05, (**) indicating p < 0.01, and (***) representing p < 0.001. The direction of the correlations is represented by the position of the markers: above the zero line for positive correlations and below for negative correlations. BAL bronchoalveolar lavage, BEC blood eosinophil counts, BSI bronchiectasis severity index, CRP C reactive protein, E-FACED exacerbation, forced expiratory volume in 1 s (FEV1), age, chronic colonization by Pseudomonas aeruginosa, radiological extension and dyspnea, FeNO fractional exhaled nitric oxide, IgE immunoglobulin E, IL-1β interleukin [IL]-1beta, IL-6 interleukin[IL]-6, IL-8 interleukin [IL]-8, MCP-1 monocyte chemoattractant protein-1, NLR neutrophil–lymphocyte ratio, TNF-α tumor necrosis factor-alpha
Exacerbation Risk Prediction in Bronchiectasis Using Composite Type 2 Biomarkers
Exploring the differential impacts of FeNO and BEC on exacerbation risk, we analyzed clinical variables alongside bronchiectasis severity and exacerbation rates, stratified by BEC and FeNO thresholds (Table 4). Patients with BEC ≥ 300 cells/μL and FeNO < 25 ppb showed elevated modified Reiff scores compared to those with opposite biomarker levels (p = 0.025), while BSI and E-FACED scores remained consistent. BAL markers IL-1β, IL-6, and IL-8, linked to severity in Fig. 2, were elevated in patients with high BEC (≥ 300 cells/μL) and low FeNO (< 25 ppb) (Table 4). Longitudinal analysis with a median follow-up time of 2.76 years for exacerbation outcomes indicated that high FeNO (≥ 25 ppb) was associated with reduced exacerbation risks (p = 0.048) (Fig. 3B), whereas high BEC (≥ 300 cells/μL) tended toward an increased risk, albeit not significantly (p = 0.118) (Fig. 3A). Serum total IgE levels did not significantly correlate with exacerbation risk (Fig. 3C). Subgroup analysis highlighted that low BEC and high FeNO were predictive of fewer exacerbations, especially when compared to high BEC/low FeNO (p = 0.026) and low BEC/low FeNO groups (p = 0.03) (Fig. 3D).
Kaplan–Meier curves illustrate the risk of moderate or severe exacerbations in bronchiectasis patients, stratified by: A BEC levels ≥ 300 cells/μL or < 300 cells/μL, B FeNO levels ≥ 25 ppb or < 25 ppb, C serum total IgE levels ≥ 75 kU/L or < 75 kU/L, and D combined BEC and FeNO into four subgroup levels. BEC blood eosinophil counts, IgE immunoglobulin E, FeNO fractional exhaled nitric oxide
Type 2 Biomarkers and Clinical Indicators as Exacerbation Predictors in Bronchiectasis
Figure 4 stratifies bronchiectasis patients by exacerbation status, showing that those with higher FeNO levels experience fewer exacerbations (p = 0.006) and those with exacerbations present with elevated BAL cytokines (IL-6, IL-8, IL-1β, TNF-α). ROC analysis indicates BAL IL-1β as the strongest predictor of exacerbations (AUC: 0.792), alongside significant BSI scores and NLR (Fig. 5). Figs. S2 and S3 highlight clinical factors that increase exacerbation risk, including Pseudomonas aeruginosa colonization, FEV1/FVC < 0.7, high NLR [NLR ≥ 3.0, cutoff value based on the mean NLR (2.9 ± 2.2, p < 0.001)], elevated BSI scores (≥ 9), mMRC dyspnea scale (≥ 2), prior macrolide therapy, and current inhalation therapies, but not ICS use. Multivariable analysis in Table 5 designates Pseudomonas aeruginosa colonization and high NLR (≥ 3.0) as independent predictors of exacerbations with HRs of 2.065 (p = 0.035) and 1.877 (p = 0.048), respectively. The combined low BEC and high FeNO indicated a decreased risk in univariable analysis but did not reach statistical significance in the multivariable analysis (HR: 0.459, p = 0.098).
Receiver operating characteristic (ROC) curve and area under the curve (AUC) with 95% confidence intervals (CIs) for clinical variables and biomarkers predictive of future exacerbations. BAL bronchoalveolar lavage, BSI bronchiectasis severity index, IL-6 interleukin[IL]-6, IL-8 interleukin [IL]-8, IL-1β interleukin [IL]-1beta, mMRC modified Medical Research Council, NLR neutrophil–lymphocyte ratio, TNF-α tumor necrosis factor-alpha, E-FACED exacerbation, forced expiratory volume in 1 s (FEV1), age, chronic colonization by Pseudomonas aeruginosa, radiological extension and dyspnea
Discussion
Our study is the first to apply three Type 2 biomarkers (BEC, total IgE, and FeNO) in an East Asian bronchiectasis cohort, revealing that 60% of patients had at least one positive marker. The impacts of these biomarkers on disease severity and outcomes varied, indicating that BEC and FeNO may represent distinct aspects of disease severity. Notably, a combination of low BEC with high FeNO suggested lower disease severity and a trend towards lower exacerbation risk. However, Pseudomonas aeruginosa colonization and high NLR (≥ 3.0) were identified as stronger exacerbation predictors. These findings imply that the clinical and pathophysiological complexity of bronchiectasis may not be sufficiently explained by the binary classification of ‘Type 2 high’ or ‘Type 2 low’ that is based solely on traditional type 2 biomarkers, as commonly applied in asthma management. This study underscores the benefits of employing multi-biomarker strategies instead of relying on single markers for a more effective risk stratification and precise treatment of bronchiectasis.
Our study on Type 2 biomarkers within an East Asian bronchiectasis cohort reveals distinct inflammation patterns when compared to asthma [10] and COPD [27]. Defining Type 2 inflammation by the presence of any positive marker, we found that 60% of our patients exhibited such inflammation, a prevalence situated between asthma (88%) [10] and COPD (42.5%) [27]. Specifically, eosinophilic bronchiectasis (BEC ≥ 300 cells/µL) was present in 15.3% of patients, lower than the 22% seen in European cohorts [6], hinting at potential regional differences due to genetic or environmental factors [6, 28, 29]. Elevated FeNO levels (≥ 25 ppb) occurred in 26.1% of patients, less than the prevalence in COPD (42.8%) [30] and asthma (51%) [10], with weakened correlation with BEC possibly resulting from the exclusion of patients with asthma [31], significant bacterial colonization [32], or the inherent heterogeneity of bronchiectasis [33]. Additionally, 36.9% had high serum IgE levels (≥ 75 kU/L), slightly below the 43.2% identified with > 60 kU/L in other studies [8]. Contrary to the findings of Ren et al. [8], our data indicate a positive correlation between IgE and BEC rather than FeNO, and a non-significant negative correlation with FeNO, highlighting the complex interplay of Type 2 biomarkers in bronchiectasis. Moreover, our analysis showed only weak and non-significant correlations between BAL eosinophils and FeNO, BEC, and serum IgE (p > 0.05), suggesting that BAL eosinophil percentages do not correlate strongly with systemic biomarkers commonly associated with inflammation and allergic responses in bronchiectasis patients. Further research is warranted to elucidate these findings.
In our study, male bronchiectasis patients who smoked with fixed airflow obstruction demonstrated elevated BEC and IgE levels, paralleling observations in COPD [34,35,36] and bronchiectasis [8, 37]. This pattern suggests a connection between these biomarkers and reduced pulmonary function. Yet, radiological severity of bronchiectasis was associated only with BEC, not total IgE, diverging from prior findings [8, 37], despite the positive BEC-IgE correlation. In contrast, higher FeNO levels (≥ 25 ppb) correlated with less radiological severity and fewer obstructive patterns, a divergence from earlier reports [7, 8, 32], but consistent with a recent Chinese bronchiectasis cohort [38], illustrating variable FeNO impacts. Furthermore, patients with high FeNO had lower blood neutrophil counts and reduced levels of airway pro-inflammatory cytokines (IL-1β, IL-6, IL-8, and TNF-α), which may explain why elevated FeNO is associated with less severe disease and a lower risk of exacerbation. Further research is warranted to clarify the relationship between elevated FeNO and bronchiectasis severity, particularly in patients without concurrent asthma.
The findings elucidate the nuanced relationship between type 2 biomarkers and exacerbation risk in bronchiectasis. Elevated BEC (≥ 300 cells/µL) correlates with higher E-FACED scores and a tendency for more frequent exacerbations, whereas FeNO levels ≥ 25 ppb are linked to a lower risk. Notably, a combination of low BEC and high FeNO significantly reduced modified Reiff scores and exacerbation rates, suggesting that BEC and FeNO reflect distinct aspects of disease severity, as seen in a retrospective Chinese cohort [38], akin to findings in a prior COPD study [30]. Building on these observations, we infer that in eosinophilic COPD, particularly in cases of severe COPD and emphysema, reduced FeNO levels often result from severe peripheral airway narrowing or obliteration, or the development of emphysema [30, 39, 40]. Similar phenomena may be observed in bronchiectasis, where severe ciliary damage and excess mucus can sequester liberated FeNO in the lung peripheries, rendering it inaccessible for proximal airway measurement [41, 42]. This pattern suggests a shared pathophysiological mechanism in these conditions, where structural changes in the lungs can significantly impact FeNO levels.
However, the relationship between exacerbation risk and other biomarkers such as serum IgE levels is less straightforward. Notably, serum IgE levels did not show a strong correlation with exacerbation risk, challenging the predictability based on Type 2 biomarkers alone. Our research highlights BEC and FeNO as composite biomarkers for enhancing risk stratification in eosinophilic bronchiectasis, a treatable trait that predisposes patients to exacerbation risks [43, 44]. The potential benefits of therapies such as inhaled corticosteroids (ICS) [44,45,46] and anti-IL-5/anti-IL-5 receptor biologics [47] in mitigating exacerbations emphasize their importance for personalized treatment, pending further evaluation.
Despite 86.9% of our cohort showing pathogen colonization detected by BAL samples, no significant differences in microbiome profiles, including Klebsiella pneumoniae and Pseudomonas aeruginosa, were observed across Type 2 biomarker levels, consistent with prior studies [7, 8]. However, contrasts with European cohorts [6, 17] might stem from our use of conventional culture methods instead of next-generation sequencing (NGS) analysis [6, 17] and different inflammatory cluster classifications [17]. Our findings align with existing literature that identifies Pseudomonas aeruginosa colonization [2, 3], airflow limitation [2, 48, 49], increased airway neutrophilic inflammation, NLR [2, 3, 38, 50], and high BSI scores [2, 25] as significant predictors of exacerbations. Multivariable analysis identified Pseudomonas aeruginosa colonization and high NLR (≥ 3.0) as independent predictors of exacerbations. The interplay of low BEC and high FeNO hinted at reduced exacerbation risks, albeit without statistical significance. Our data indicate that while traditional risk factors like Pseudomonas colonization and high NLR are confirmed predictors of exacerbations, Type 2 biomarker combinations could enhance patient stratification, supporting a multi-biomarker approach in developing personalized bronchiectasis treatment plans [6,7,8, 17, 33].
The strength of this study lies in its comprehensive integration of three Type 2 biomarkers, systemic and local inflammatory markers, and microbiological data within a well-defined bronchiectasis cohort, with exclusion criteria applied to patients with clinical asthma and ABPA. However, it encounters several limitations. Firstly, the lack of research on T2-high endotypes and the establishment of thresholds for type 2 biomarkers in bronchiectasis underscores the need for consensus on diagnostic benchmarks. Secondly, its cross-sectional design precludes the observation of longitudinal changes in Type 2 biomarkers. Thirdly, the relatively small sample size in specific subgroups, especially those with low BEC and high FeNO, as well as in the eosinophilic bronchiectasis subgroup, limits our ability to fully evaluate their impact on exacerbation risks. This limitation underscores the need for further studies to confirm these preliminary findings and to enhance our understanding of their clinical implications. Fourthly, the absence of control groups constrains comparative analyses. Fifthly, reliance on conventional culture methods rather than NGS restricts the scope of our microbiome analysis. Sixthly, sample collection during stable disease states necessitates follow-up studies to monitor biomarker changes during exacerbations. Lastly, conducting this study within a Taiwanese East Asian population highlights the necessity for further validation across diverse geographical and ethnic contexts.
In conclusion, our investigation offers groundbreaking perspectives on Type 2 biomarkers within an East Asian bronchiectasis cohort, demonstrating the differential roles of BEC, FeNO, and serum IgE in influencing exacerbation risk. These findings highlight the multifaceted nature of bronchiectasis, advocating for holistic patient assessments that combine biomarker insights with airway microbiology and clinical indices. Our study reinforces the utility of multi-biomarker strategies, as opposed to single-marker methods, for nuanced risk stratification and individualized bronchiectasis treatment.
Data Availability
No datasets were generated or analysed during the current study.
Abbreviations
- AUC:
-
Area under the curve
- BAL:
-
Bronchoalveolar lavage
- BEC:
-
Blood eosinophil counts
- BMI:
-
Body mass index
- BSI:
-
Bronchiectasis severity index
- CAT:
-
COPD assessment test
- CIs:
-
Confidence intervals
- COPD:
-
Chronic obstructive pulmonary disease
- CRP:
-
C reactive protein
- E-FACED:
-
Exacerbation, Forced expiratory volume in 1 s (FEV1), Age, Chronic colonization by Pseudomonas aeruginosa, Radiological extension and Dyspnea
- FeNO:
-
Fractional exhaled nitric oxide
- FEV1:
-
Forced expiratory volume in 1 s
- FVC:
-
Forced vital capacity
- ICS:
-
Inhaled corticosteroid
- IL-1β:
-
Interleukin [IL]-1β
- IL-6:
-
Interleukin [IL]-6
- IL-8:
-
Interleukin [IL]-8
- IgE:
-
Immunoglobulin E
- LABA:
-
Long-acting β2 sympathomimetic agonists
- LAMA:
-
Long-acting muscarinic antagonist
- MCP-1:
-
Monocyte chemoattractant protein-1
- mMRC:
-
Modified Medical Research Council
- NLR:
-
Neutrophil–lymphocyte ratio
- NTM:
-
Non-tuberculosis mycobacteria
- ROC:
-
Receiver operating characteristic
- TB:
-
Tuberculosis
- TNF-α:
-
Tumor necrosis factor [TNF]-α
References
Polverino E, Goeminne PC, McDonnell MJ et al (2017) European Respiratory Society guidelines for the management of adult bronchiectasis. Eur Respir J 50:1700629
Chalmers JD, Moffitt KL, Suarez-Cuartin G et al (2017) Neutrophil elastase activity is associated with exacerbations and lung function decline in bronchiectasis. Am J Respir Crit Care Med 195:1384
Tiew PY, Jaggi TK, Chan LLY et al (2021) The airway microbiome in COPD, bronchiectasis and bronchiectasis-COPD overlap. Clin Respir J 15:123
Flume PA, Chalmers JD, Olivier KNJ (2018) Advances in bronchiectasis: endotyping, genetics, microbiome, and disease heterogeneity. Lancet 392:880–890
Guan WJ, Oscullo G, He MZ et al (2023) Significance and potential role of eosinophils in non-cystic fibrosis bronchiectasis. J Allergy Clin Immunol Pract 11:1089–1099
Shoemark A, Shteinberg M, De Soyza A et al (2022) Characterization of eosinophilic bronchiectasis: a European multicohort study. Am J Respir Crit Care Med 205:894–902
Oriano M, Gramegna A, Amati F et al (2021) T2-high endotype and response to biological treatments in patients with bronchiectasis. Biomedicines 9:772
Ren J, Chen A, Wang J et al (2023) Association of blood total immunoglobulin E and eosinophils with radiological features of bronchiectasis. BMC Pulm Med 23:316
Brusselle GG, Koppelman GH (2022) Biologic therapies for severe asthma. N Engl J Med 386:157–171
Denton E, Price DB, Tran TN et al (2021) Cluster analysis of inflammatory biomarker expression in the international severe asthma registry. J Allergy Clin Immunol Pract 9:2680–2688
Yun JH, Lamb A, Chase R et al (2018) Blood eosinophil count thresholds and exacerbations in patients with chronic obstructive pulmonary disease. J Allergy Clin Immunol 141:2037–2047
Casanova C, Celli BR, de Torres JP et al (2017) Prevalence of persistent blood eosinophilia: relation to outcomes in patients with COPD. Eur Respir J 50:1701162
Lu Z, Huang W, Wang L et al (2018) Exhaled nitric oxide in patients with chronic obstructive pulmonary disease: a systematic review and meta-analysis. Int J Chronic Obstr Pulm Dis 13:2695–2705
Jamieson DB, Matsui EC, Belli A et al (2013) Effects of allergic phenotype on respiratory symptoms and exacerbations in patients with chronic obstructive pulmonary disease. Am J Respir Crit Care Med 188:187–192
Bafadhe M, Peterson S, De Blas MA et al (2018) Predictors of exacerbation risk and response to budesonide in patients with chronic obstructive pulmonary disease: a post-hoc analysis of three randomised trials. Lancet Respir Med 6:117–126
Bhatt SP, Rabe KF, Hanania NA et al (2023) BOREAS investigators. dupilumab for COPD with type 2 inflammation indicated by eosinophil counts. N Engl J Med 389:205–214
Choi H, Ryu S, Keir HR et al (2023) Inflammatory molecular endotypes in bronchiectasis: a European multicenter cohort study. Am J Respir Crit Care Med 208:1166–1176
Naidich DP, McCauley DI, Khouri NF et al (1982) Computed tomography of bronchiectasis. J Comput Assist Tomogr 6:437–444
Agarwal R, Sehgal IS, Dhooria S et al (2016) Developments in the diagnosis and treatment of allergic bronchopulmonary aspergillosis. Expert Rev Respir Med 10:1317–1334
Amati F, Simonetta E, Gramegna A et al (2019) The biology of pulmonary exacerbations in bronchiectasis. Eur Respir Rev 28:190055
Morris A, Beck JM, Schloss PD et al (2013) Comparison of the respiratory microbiome in healthy nonsmokers and smokers. Am J Respir Crit Care Med 187:1067–1075
Gauthier TW, Grunwell JR, Ping XD et al (2017) Impaired defenses of neonatal mouse alveolar macrophage with cftr deletion are modulated by glutathione and TGFβ1. Physiol Rep 5:e13086
Amatullah H, Shan Y, Beauchamp BL et al (2017) DJ-1/PARK7 impairs bacterial clearance in sepsis. Am J Respir Crit Care Med 195:889–905
Reiff DB, Wells AU, Carr DH et al (1995) CT findings in bronchiectasis: limited value in distinguishing between idiopathic and specific types. AJR Am J Roentgenol 165:261–267
Chalmers JD, Goeminne P, Aliberti S et al (2014) The bronchiectasis severity index. An international derivation and validation study. Am J Respir Crit Care Med 189:576–585
Rosales-Mayor E, Polverino E, Raguer L et al (2017) Comparison of two prognostic scores (BSI and FACED) in a Spanish cohort of adult patients with bronchiectasis and improvement of the FACED predictive capacity for exacerbations. PLoS ONE 12:e0175171
Oishi K, Matsunaga K, Shirai T et al (2020) Role of type2 inflammatory biomarkers in chronic obstructive pulmonary disease. J Clin Med 9:2670
Chandrasekaran R, Mac Aogáin M, Chalmers JD et al (2018) Geographic variation in the aetiology, epidemiology and microbiology of bronchiectasis. BMC Pulm Med 18:83
Dhar R, Singh S, Talwar D et al (2023) Clinical outcomes of bronchiectasis in India: data from the EMBARC/respiratory research network of India registry. Eur Respir J 61:00611
Liu X, Zhang H, Wang Y et al (2020) Fractional exhaled nitric oxide is associated with the severity of stable COPD. COPD 17:121–127
Chen FJ, Liao H, Huang XY et al (2016) Importance of fractional exhaled nitric oxide in diagnosis of bronchiectasis accompanied with bronchial asthma. J Thorac Dis 8:992–999
Tsang KW, Leung R, Fung PC et al (2002) Exhaled and sputum nitric oxide in bronchiectasis: correlation with clinical parameters. Chest 121:88–94
Saleh AD, Chalmers JD, De Soyza A et al (2017) The heterogeneity of systemic inflammation in bronchiectasis. Respir Med 127:33–39
Hartl S, Breyer MK, Burghuber OC et al (2020) Blood eosinophil count in the general population: typical values and potential confounders. Eur Respir J 55:1901874
Park HY, Chang Y, Kang D et al (2021) Blood eosinophil counts and the development of obstructive lung disease: the Kangbuk Samsung health study. Eur Respir J 58:2003823
Lommatzsch M, Speer T, Herr C et al (2022) IgE is associated with exacerbations and lung function decline in COPD. Respir Res 23:1
Hassan WA, Shalan I, Khalifa M (2014) Impact of immunoglobulin E and airway obstruction on bronchiectasis. Open J Respir Dis 4:34–40
Chen F, Zeng Z, Huang X, Liu Y (2023) Simultaneous evaluation of the fractional exhaled nitric oxide and blood eosinophil count of T2-high endotype in patients with non-cystic fibrosis bronchiectasis. Chron Respir Dis 20:1479973123121055
Malerba M, Radaeli A, Olivini A et al (2014) Exhaled nitric oxide as a biomarker in COPD and related comorbidities. Biomed Res Int 2014:271918
Machado RF, Stoller JK, Laskowski D et al (2002) Low levels of nitric oxide and carbon monoxide in α1-antitrypsin deficiency. J Appl Physiol 93:2038–2043
Boon M, Meyts I, Proesmans M et al (2014) Diagnostic accuracy of nitric oxide measurements to detect primary ciliary dyskinesia. Eur J Clin Invest 44:477–485
Paternò S, Pisani L, Zanconato S et al (2023) Role of nasal nitric oxide in primary ciliary dyskinesia and other respiratory conditions in children. Int J Mol Sci 24:16159
Pollock J, Goeminne PC (2023) Eosinophils in bronchiectasis: a U-turn for bronchiectasis management. Chest 64:561–563
Oscullo G, Gomez-Olivas JD, Martínez-García MÁ (2023) Eosinophilic bronchiectasis and therapeutic opportunities. Ann Allergy Asthma Immunol 131:689–690
Aliberti S, Sotgiu G, Blasi F et al (2020) Blood eosinophils predict inhaled fluticasone response in bronchiectasis. Eur Respir J 56:2000453
Martínez-García MÁ, Méndez R, Olveira C et al (2023) The U-shaped relationship between eosinophil count and bronchiectasis severity: the effect of inhaled corticosteroids. Chest 164(3):606–613
Rademacher J, Konwert S, Fuge J et al (2020) Anti-IL5 and anti-IL5Ra therapy for clinically significant bronchiectasis with eosinophilic endotype: a case series. Eur Respir J 55(1):1901333
Aliberti S, Lonni S, Dore S et al (2016) Clinical phenotypes in adult patients with bronchiectasis. Eur Respir J 47:1113–1122
Martinez-García MA, Oscullo G, Posadas T et al (2001) Spanish registry of bronchiectasis group of SEPAR (RIBRON). Pseudomonas aeruginosa and lung function decline in patients with bronchiectasis. Clin Microbiol Infect 27:428–434
Martinez-García MÁ, Olveira C, Girón R et al (2022) Peripheral neutrophil-to-lymphocyte ratio in bronchiectasis: a marker of disease severity. Biomolecules 12:1399
Acknowledgements
The authors would like to acknowledge the service provided by the First Common Laboratory of Department of Education and Medical Research at National Taiwan University Hospital Yunlin Branch.
Funding
This research was supported by the Ministry of Science and Technology, Taiwan (MOST 107-2314-B-002-235-MY3) and National Taiwan University Hospital, Yun-Lin Branch, Taiwan (NTUHYL109. X011, NTUHYL 111. X013).
Author information
Authors and Affiliations
Contributions
YFC and JYC collected the clinical and microbiology data and performed all analyses. HHH, YYC, KZL and ZCH contributed to specimen analysis and statistical analyses. NC and YFC provided radiological and clinical insights to data interpretation. CJY, HCW and JYC supervised the study. YFC and CJY drafted the manuscript. All authors provided critical revisions and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical Approval
This study was approved by the Research Ethics Committee of National Taiwan University Hospital (NTUH-REC No. 201712075RINA and NTUH-REC No.202110093RIND), in accordance with the Declaration of Helsinki.
Consent to Participate
Written informed consent was obtained from all patients/participants involved in the study.
Consent to Publish
The authors affirm that human research participants provided informed consent for publication.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Chen, YF., Hou, HH., Chien, N. et al. Type 2 Biomarkers and Their Clinical Implications in Bronchiectasis: A Prospective Cohort Study. Lung (2024). https://doi.org/10.1007/s00408-024-00707-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00408-024-00707-0