Abstract
Background
Non-visual hallucinations in Parkinson’s disease (PD) can be prevalent and distressing. Most existing research has however, focused on visual hallucinations as well as related risk factors. The current study thus conducted a systematic review to collate existing evidence on non-visual hallucinations in PD, focusing on their prevalence, phenomenology, and clinical-cognitive correlates.
Methods
Ninety-one relevant studies were included from a systematic search across PsycINFO APA, PubMed, and Web of Science, for peer-reviewed publications in the English language, from 1970 to the present. These comprised a mix of case (30 studies; n = 56) and group design (62 studies; n = 7346) studies, divided into three somewhat overlapping collections to address our three research foci.
Results
Prevalence estimates for hallucinations were: auditory 1.5–72.0%, olfactory 1.6–21.0%, somatic-tactile 0.4–22.5%, gustatory 1.0–15.0%, and sensed presence 0.9–73.3%. Phenomenological inquiries revealed descriptions of vivid, consuming events replete with elaborate detail, adversely affecting PD patients in different ways. Overt experiences of multisensory hallucinations were also highly variable (0.4–80%) but exceedingly common, reported by almost half of the 45 included prevalence studies. There was some evidence for modality-specific hallucination predictors, but this was largely tentative, pending robust replication.
Conclusions
Marked prevalence figures coupled with phenomenological descriptions implicating distress denote that non-visual and multisensory hallucinations in PD are of clinical significance. More direct research and clinical attention need to be devoted to the study and management of such hallucinatory experiences.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Parkinson’s disease (PD) is a neurodegenerative disorder characterised by prominent motor and non-motor symptoms [1]. Classification as a movement disorder derives from its disabling motor symptoms, such as a resting tremor, bradykinesia, and rigidity, but non-motor symptoms also exert significant impact [2]. Hallucinations are one such symptom, typically regarded as belonging to the psychiatric realm, but can be prevalent and distressing [3, 4]. Another non-motor symptom relates to cognitive decline; 31% of PD patients were diagnosed with dementia [5], with 83% eventually transitioning over 20 years [6]. In fact, presence of visual hallucinations has been identified as a significant risk factor for developing dementia [7, 8]. Yet beyond the visual mode, scant attention has been given to hallucinatory experiences in other sensory modalities in PD as well as their potential clinical and cognitive correlates.
Hallucinations in PD: prevalence, phenomenological characteristics and correlates
With the publication of a consensus definition of psychosis in PD, clinical and research attention to the role of hallucinations in the disorder has intensified [9]. An early epidemiological review reported point prevalence of up to 38% visual hallucinations (lifetime 50%) and 22% auditory hallucinations in PD, observing that olfactory and somatic-tactile hallucination rates were not systematically noted [10]. A comparative review of visual hallucination incidence across Parkinsonism disorders instead cited rates up to 75% [11]. A more recent meta-analysis calculated pooled point prevalence of 28% and 9% for visual and auditory hallucinations respectively [12]; whereas a separate review noted 25% for sensed presence [13]. These authors concluded that hallucinations tended to increase and worsen over time [10], and highlighted that use of validated assessment tools yielded elevated estimates [12].
Detailed phenomenological descriptions of visual hallucinations in PD are plentiful. These include complex images of variable content, but often comprise familiar or unfamiliar people, or animals [14,15,16,17,18]. Frequency of visual hallucinations has varied from numerous times a day to weekly or less, most likely in the evening or at night, experienced as kinetic and stereotyped [15], typically of brief duration lasting seconds to minutes, with no specific localisation [14], and were somewhat blurred, with no known triggers [18]. Though non-threatening themes have been regularly endorsed [15], visual hallucinations could be perceived as emotionally neutral [14], or unpleasant and anxiety-provoking [16]. Conviction has similarly been mixed [14] or preserved, yet involving complex interactions, such as inviting ‘guests’ to dinner, or pushing an unwanted companion away [17]. Visual hallucinations have occurred in isolation, or in conjunction with other hallucinations or delusions [15]. For instance, comorbid visual and tactile perception of hairs on an arm, or lice and worms burrowing into the skin, have been described, with such multisensory events often perceived as distressing [17]. Conversely, phenomenological information regarding auditory hallucinations in PD has received limited attention. Most common were voices perceived as originating outside the head, often incomprehensible or indistinct [12], or familiar, and even persecutory on occasion [15]. Auditory hallucinations have sometimes been described as providing a “soundtrack” to visual hallucinations, for example, hearing people in a vision conversing [14]. Non-verbal sounds have also been reported, but musical hallucinations were deemed rare [12]. Inclusive descriptions of hallucinatory experiences across other sensory modalities were generally lacking, but tactile hallucinations have been cursorily described as the feeling of being touched by a person, or involving contact with small animals [14].
Cognitive decline is often seen in PD [5, 6], with cognitive impairment and visual hallucinations described as risk factors for mutual emergence [19]. Increased incidence of visual hallucinations during cognitive decline has been noted, with these experiences reported as qualitatively different, inciting combative behaviours, and exacerbated by lack of insight [17]. Several reviews have shown that PD patients with visual hallucinations exhibited significantly poorer cognition overall as well as within specific domains, involving attention, executive function, memory, and visual processing [20,21,22]. Those with visual hallucinations were also troubled by more severe clinical symptoms, such as greater duration and severity of PD illness, and depressed mood [18]. Alongside older age and sleep disturbances, all these factors have been noted as significant predictors of (visual) hallucinations in PD [10].
Other than these disease-related factors, there has also been scrutiny around possible neurophysiological underpinnings of visual hallucinations in PD. These have ranged from structural atrophy and/or aberrant activation across multiple brain regions, including frontal and visual cortices, genetic and familial origins or even the potential contribution of dopaminergic or anticholinergic therapies [10, 14, 18, 20, 23]. Some authors have attempted to offer up integrative theoretical models seeking to explain the rise and progression of visual hallucinations in PD [24, 25], though more work remains to be done. Likewise, visual hallucinations tend to be reported in other neurodegenerative (e.g. dementia with Lewy bodies), or ophthalmological (e.g. Charles Bonnet syndrome) disorders, though wide-ranging prevalence estimates make it difficult to draw any firm conclusions about relative frequencies [26,27,28]. In contrast, studies delving into non-visual or multisensory hallucinations in PD, including their neurophysiological causes, are notably lacking.
Aim and research questions
Based on a brief review of the literature, it is evident that hallucinations studies in PD have focused on the visual realm. Reviews have covered topics ranging from aetiological models [25] to neuroanatomical correlates [29] and management strategies [30] for visual hallucinations, but less so in other sensory modalities. Even studies broadly mentioning hallucinations were often simply referring to those in the visual domain. Yet hallucinations are not a unitary construct, encompassing a complex, multifaceted array of discrete yet overlapping perceptual experiences. Discrimination of modality-specific hallucinations in PD may aid prognostic and therapeutic outcomes. However, we first need to better understand these experiences.
The aim of the current study was to examine non-visual and multisensory hallucinations in PD, with the latter referring to experiences in two or more sensory modalities (often but not always including visual), concurrent or otherwise. The following research questions were put forward:
-
(i)
What was the prevalence of non-visual (i.e. auditory, olfactory, somatic-tactile, gustatory, and sensed presence) and multisensory hallucinations in PD?
-
(ii)
What were the phenomenological characteristics of these hallucinatory experiences, including physical (e.g. frequency, duration, intensity), cognitive (e.g. controllability, conviction, interaction) and emotional (e.g. valence, distress, functional impact) facets?
-
(iii)
What were the key clinical and cognitive correlates underlying the occurrence of these modality-specific hallucinations in PD? In line with existing research [10], clinical correlates of interest were participant age, age of PD diagnosis, length of illness, PD severity, and presence of depression. Cognitive correlates included presence of dementia or cognitive decline (general or domain-specific).
Methods
The review adhered to guidelines described in the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement [31]. The study protocol was pre-registered on the International Prospective Register of Systematic Reviews (PROSPERO; CRD42019124981). The authors confirm that the approval of an institutional review board and patient consent was not required for this work. We confirm that we have read the Journal’s position on issues involved in ethical publication and affirm that this work is consistent with those guidelines.
Search strategy
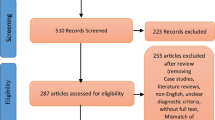
A flowchart illustrating the search strategy and study selection is presented in Fig. 1. A systematic search was conducted across PsycINFO APA, PubMed, and Web of Science, for peer-reviewed publications in the English language, from 1970 to the present, with a cut-off date of 15 February 2022. Search syntax was optimised for each database, centred on three dominant themes: PD, non-visual or multisensory hallucinations, as well as key clinical and cognitive correlates. These terms (and their permutations) were entered into a search matrix using Boolean operators to ensure pertinent combinations were incorporated (see Table A in Supplementary materials for the search syntax). Reference lists of eligible studies were cross-checked for further related works.
PRISMA flow diagram 1Search cut-off date: 15/02/2022 2Incorrect outcomes included: a Non-modality-specific hallucinations n = 91; b Limited phenomenological information n = 30; and c Main outcome not prevalence, phenomenology or correlates n = 8.3Pertaining to or combined with other neurological disorder(s)
Study selection
After duplicates were removed, the title, abstract and keywords of retrieved publications were screened by CY, where irrelevant studies were excluded. A fidelity check was conducted where WLT also independently reviewed the first 100 abstracts, yielding an excellent interrater reliability of k = 0.852 (i.e. complete agreement for 99/100 papers, with CY taking the more conservative stance of screening in one additional paper). This corroborated that the screening approach undertaken by CY for the remaining abstracts was targeted and robust. Full-text articles were then independently reviewed by WLT and CY for eligibility, and included if they comprised case report or group design studies investigating the prevalence, phenomenology and/or correlates of non-visual or multisensory hallucinations in PD. For intervention studies, only baseline data was used (if relevant); longitudinal studies were also permitted. Studies focusing solely on visual (or non-modality-specific) hallucinations were excluded (n = 91; nb. this inflated figure provides further support showing how non-visual hallucination studies have been neglected in lieu of those focusing on visual hallucinations), and unpublished data were not pursued. Discrepancies in study inclusion between CY and WLT were resolved through iterative discussions with senior author SLR, until consensus was achieved.
Data extraction and risk of bias assessment
Data extraction was jointly performed by WLT and CY, and comprised: Study identifiers (e.g. authors, year of publication and aims), participant characteristics (e.g. age, sex, subgroup sizes), prevalence data (e.g. percentages of non-visual or multisensory hallucinations), phenomenological descriptions (e.g. physical, cognitive and emotional facets of hallucinatory experiences), as well as key clinical and cognitive correlates (e.g. length and severity of PD illness, general and domain-specific cognition). Where information pertaining to visual hallucinations was presented alongside that for non-visual hallucinations, it was included in data extraction. This approach ensured completeness, especially critical for phenomenology, though extracted data for visual hallucinations should be interpreted with caution in light of this. To avert replication for multiple publications using the same cohort, the study with the most representative data aimed at answering our research questions was included; studies employing similar datasets were included only if distinct aspects of the empirical data were analysed.
The search strategy yielded a total of 8111 records, from which 91 case or group design studies met inclusion criteria. These studies were divided into three somewhat overlapping collections to address our three research foci, namely: (i) prevalence (n = 45), (ii) phenomenology (n = 56), and (iii) key clinical and cognitive correlates (n = 16). To fulfil a more stringent consideration of prevalence, smaller-scale studies where n ≤ 50 participants were removed from the first collection (n = 17 studies; see Table B in Supplementary materials). Likewise, studies focusing on overall hallucinations (i.e. not modality-specific) were omitted from the third collection (n = 15; see Tables C and D in Supplementary materials). Owing to the nature of our research questions, the first and third collections considered group design studies only, whereas the second collection encompassed case and group design studies. The varied nature of data extracted meant that a narrative synthesis of findings is provided.
A risk of bias assessment was conducted by CY using the JBI Critical Appraisals Checklist for Case Studies [JBI-CAC, supplemented by one additional item from the corresponding checklist for Case Series; [32] and Newcastle–Ottawa Quality Assessment Scale for Case Control Studies [NOQAS; [33] for case and group design studies respectively, adapted for our purpose (see Tables E and F in Supplementary materials). Within each of the six domains, 0–2 points may be awarded based on how well each response satisfied the criterion under assessment (nb. only 0–1 point may be awarded for representativeness and comparability in the NOQAS). Unweighted summed scores provided an overall measure of quality and bias. Categorical ratings were devised where: Excellent 10–12/12, good 7–9/12, fair 4–6/12 and poor ≤ 3/12 for JBI-CAC; and Excellent 9–10/10, good 7–8/10, fair 5–6/10 and poor ≤ 4/10 for NOQAS).
Results
Prevalence of multisensory hallucinations in PD
Prevalence data are presented in Table 1, where 6968 PD patients across 45 studies were included. Of these, a fraction was further classed as having dementia (n = 48) and/or psychosis (n = 319). Prevalence of overall hallucinations ranged from 12.8 to 100% (54.2% and 77.1–100% respectively for those with dementia or psychosis in PD), whereas that for multisensory hallucinations ranged from 0.4 to 80% (modalities not specified). Within each primary sensory domain, these figures were: auditory 1.5–72%, olfactory 1.6–21%, somatic-tactile 0.4–22.5%, gustatory 1.0–15.0%, and sensed presence 0.9–73.3% (visual 3.0–96.9%). Where available, we also extracted prevalence data for hallucinations in a single modality (see Table 1). Given the small proportions of PD patients classed as having dementia and/or psychosis, it was difficult to draw any firm conclusions as to whether prevalence estimates across single hallucination modalities appeared to differ in typical versus atypical PD. When interpreting these wide ranges, it is imperative to pay heed to the specific topic examined within each study, as this would have influenced the corresponding figures in some way (e.g. a study focused on sensed presence would likely have recruited a surfeit of participants experiencing this phenomenon, neglecting other hallucinatory experiences). On this note, focusing explicitly on studies that named prevalence as their predominant aim (n = 6), more precise point prevalence rates of 12.8–39.8% for overall (46.3% lifetime), and 2.1–10.3% for olfactory hallucinations as well as 23% for sensed presence were recorded (14.3% for visual). No existing studies specifically aimed to examine the prevalence of auditory, somatic-tactile or gustatory hallucinations in PD. It is noted that the sporadic and inconsistent nature of figures extracted across the sensory domains precluded a more systematic approach to data summation, including use of meta-analysis or forest plots, in a meaningful way.
Phenomenology of non-visual and multisensory hallucinations in PD
Descriptive phenomenology of non-visual and multisensory hallucinations in PD is shown in Table 2, where 1093 patients across 56 studies were included. To contextualise these findings, we opted to present participant characteristics, comprising age, sex (and number of participants for group design studies), length of illness as well as neurological and psychiatric comorbidities, where available. Hallucinatory experiences in single as well as multiple modalities were reported, spanning a range of non-visual (and visual) domains. For specific phenomenological parameters, there did not appear to be consistent patterns in how these diverse events were experienced by individual PD patients, whether in terms of their physical (e.g. frequency, duration, localisation and time of occurrence), cognitive (e.g. conviction, controllability and compliance) or emotional (e.g. content, valence, distress and functional impact) characteristics. Nor did these experiences seem to relate to participant characteristics (e.g. age) in a systematic manner. A major conclusion that may be drawn is that many of these experiences, regardless of hallucination modality, appeared to be vivid, consuming events replete with elaborate and possibly idiosyncratic detail, seemingly affecting a range of PD patients in different ways. Overt experiences of multisensory hallucinations were exceedingly common, described by almost half (48.2%) of included studies (though this could represent underreporting, given the question was mostly not explicitly asked).
Significant clinical and cognitive correlates of modality-specific hallucinations in PD
Select clinical and cognitive correlates of modality-specific hallucinations in PD are presented in Table 3, where 2008 patients across 16 studies were included. Subgroups for comparisons were those with auditory, olfactory (or with or without visual) hallucinations or sensed presence (assessed against control groups without hallucinations or psychosis at times). Broadly, it can be concluded that participants with any modality-specific hallucinations fared worse across all clinical and cognitive variables, when significant findings were established (though non-significant findings were also recorded). The picture was less clear when considering correlates from a modality-specific lens; PD patients with olfactory hallucinations had significantly more severe PD (1/2 studies) and poorer cognition (1/2 studies). Similarly, those with sensed presence had significantly increased length of illness (1/1 studies), more severe PD (2/2 studies), and poorer cognition overall or in select domains (2/2 studies). The two studies involving auditory hallucinations had too small subgroup numbers to form meaningful conclusions.
Significant associations or risk factors for modality-specific hallucinations in PD are shown in Table 4. Significant associations were identified between increased modalities of hallucinations and poorer overall and domain-specific cognition, as well as between olfactory hallucinations and female sex, presence of visual/auditory hallucinations and PD severity. Significant predictors of olfactory hallucinations were female sex and presence of visual or auditory hallucinations in one study [34], whereas significant predictors of sensed presence were PD severity, poorer cognition, presence of visual hallucinations or illusions, and increased levodopa equivalent (or other PD medication) dose across three studies [35,36,37]. Notably, Goetz et al. [38] demonstrated that length of illness at baseline, time elapsed, and first onset of visual and non-visual hallucinations predicted future increased occurrence of visual and non-visual hallucinations.
Risk of bias assessment
For the 30 case studies rated on the JBI-CAC, 90% were excellent, and 10% were good. For the 62 empirical studies rated on the NOQAS, 19.4% were excellent, 38.7% were good, 29% were fair, and 12.9% were poor (see Tables E and F in Supplementary materials). A significant fraction of adverse ratings for case and group design studies were due to lack of valid hallucination assessments, with a proportion of group design studies also marked down owing to lack of a control group for comparison (as prioritised in the tool employed).
Discussion
This systematic review aimed to examine the prevalence, phenomenological characteristics as well as major clinical and cognitive correlates of non-visual or multisensory hallucinations in PD, by collating findings from existing studies in the field. For our first research question regarding prevalence, rates for auditory (and visual) hallucinations as well as sensed presence were largely in line with figures cited in the literature [10,11,12,13]. There is no doubt that visual hallucinations (up to 96.9%), alongside sensed presence (up to 73.3%), remain the most typical sensory mode in PD. Most reviewed studies were of prospective, cross-sectional design, with those specifying prevalence as their predominant aim likely offering more accurate numbers. Though prevalence estimates for olfactory, somatic-tactile and gustatory hallucinations were of lesser magnitude (than visual hallucinations), sizeable figures (15–22.5%) were still recorded, signifying that these phenomena clearly affect a notable proportion of PD patients. Yet these wide-ranging numbers were also subject to methodological variations in terms of patient selection, evaluation methods, as well as possible divergence in symptoms assessed, thereby precluding conclusive prevalence rates being endorsed.
For our second research question on phenomenology, anecdotal descriptions of non-visual and multisensory hallucinations in PD demonstrated variability in individual experience, with little consistency across any of the physical, cognitive or emotional characteristics (see Table 2). In other words, frequency, duration, intensity, clarity, localisation, time of occurrence, personification, controllability, conviction, interaction, beliefs regarding origin, content, valence, distress and functional impact varied within and between individuals, depending on the specific hallucination episode and/or modality under examination. Similarly, whilst visual involvement in multisensory hallucinations was likely, non-visual symptoms, whether in single or multiple modalities, were also observed. These hallucinations were typically described with graphic detail, with PD patients fully immersed in their experiences. Coupled with the fact that these events were often not spontaneously divulged [40], this raises the speculation of whether non-visual hallucinations in PD may actually be more clinically significant than previously postulated. Involvement of greater number of sensory modalities in multisensory hallucinations also begets the question of whether there could be an incremental adverse impact on PD patients who have these experiences (see Sect. "Clinical implications and recommendations").
For our third research question, we focused on elucidating significant clinical and cognitive correlates of modality-specific hallucinations in PD. At the outset, we need to qualify that findings relating to the visual modality are incomplete. This is because our aim and search strategy focused on non-visual and multisensory hallucinations (with results for visual hallucinations presented alongside, only when they were concurrently analysed). Notably, our review demonstrated a clear lack of relevant studies in non-visual domains. Only a handful of studies were uncovered in auditory and olfactory hallucinations as well as sensed presence (see Tables 3 and 4), with the remaining focused on the visual domain (see Tables C and D). Nonetheless, we largely corroborated that PD patients with hallucinations (in any modality) tended to fare significantly worse in terms of increased length of illness, greater PD severity, presence of major depressive disorder and/or dementia as well as poorer cognition overall and in specific domains [18]. Other than depression, these variables have been verified as significant risk factors for developing hallucinations [10]. Furthermore, we identified added factors of interest to modality-specific hallucinations, where visual hallucinations were identified as a significant risk factor for sensed presence, and female sex and visual or auditory hallucinations were identified as significant risk factors for olfactory hallucinations. Yet caution must be exercised when focusing on findings relating to non-visual hallucinations owing to: (i) limited number of studies, (ii) small subgroup sizes [40, 41], and (iii) comorbid visual hallucinations in those subgroups [34, 41]. Further replicative efforts are thus essential.
Finally, our risk of bias assessment revealed that case studies were generally of high quality, whereas empirical studies were of mixed quality, meaning we need to be mindful of these limitations when drawing interpretations and subsequent conclusions from the latter body of work.
Clinical implications and recommendations
There are several clinical implications and recommendations we can draw from our findings. First, non-visual and multisensory hallucinations in PD are likely of clinical significance. Empirical evidence supporting this statement comes from prevalence figures noted, coupled with vivid phenomenological information, substantially contributing to patient distress in some cases. To increase focus in this area, clinicians and researchers first need to specifically ask about patient experiences of non-visual and multisensory symptoms during routine assessments. Some PD patients are unlikely to volunteer these symptoms because of embarrassment or fear of stigma in trying to avert a further psychiatric diagnosis [39, 42]. Being transparent and matter-of-fact about asking these questions should somewhat allay these concerns. Having a well-validated assessment tool will also be helpful in guiding conversations. Though the Movement Disorders Society Unified Parkinson Disease Rating Scale (UPDRS) remains the gold-standard measure, hallucinations (not modality-specific) and delusions are jointly addressed within a single item (nb. for this reason, PD studies that assessed hallucinations with this measure only were not able to be included within the current review). A brief accompanying hallucinations screen, such as the Psycho-Sensory Hallucinations Scale [43, 44], will circumvent this issue. More comprehensive investigations can alternatively employ the Questionnaire for Psychotic Experiences [45]. Related to this, sensed presence (included within this review), alongside passage hallucinations and visual illusions, has often been termed a ‘minor’ hallucination [46], implicitly deserving of lesser concern. This in turn suggests that hallucinatory experiences in other forms are ‘major’ in nature. Being cautious about such terminology may aid in managing patients’ views and expectations of these symptoms, noting that ‘minor’ hallucinations are often (though not always) comorbid with non-visual (and visual) hallucinations in PD.
Second, a factor that does motivate self-report of hallucinations in PD is when a patient is bothered by these experiences [47]. Many patients with symptoms established as characteristic in clinical presentations for PD, including visual hallucinations and sensed presence, exhibit good insight and minimal distress, despite the somewhat anomalous nature of these events [49, 50]. We can therefore speculate that a lack of patient awareness of non-visual hallucinatory symptoms in the disorder may contribute to feelings of fear and anxiety. Moreover, involvement of increasing number of modalities in multisensory hallucinations may further elevate patient distress (and possibly reduce insight), owing to enhanced veracity of the experience given multiple sensory inputs, especially if these coincide temporally. Supportive therapies that may be introduced to patients and/or their carers include psychoeducation, cognitive-behaviour techniques (if suitable), and where hearing loss may be a factor, the use of hearing aids [50]. It is hoped that simply asking about these experiences, and putting basic management strategies in place may be of benefit in the first instance.
Third, some authors have suggested that non-visual hallucinations tend to emerge in older age [14] or later-stage PD [13], but empirical support for this is still tentative. The most convincing evidence to date stems from a 10-year longitudinal study, where isolated visual hallucinations predominated early on, with visual plus non-visual hallucinations accounting for progressively higher proportion of experiences as time elapsed [38]. Conversely, some of our studies have demonstrated the presence of non-visual hallucinations in isolation [52,53,54], whereas multisensory hallucinations without visual involvement seemed rare [54]. Phenomenologically, certain sensory combinations appeared more prevalent, such as comorbidity of visual and tactile components as well as co-occurrence of sensed presence with hallucinations in one or more sensory modalities [55]. Should non-visual or multisensory hallucinations predominate later in PD, we may expect a discrete cognitive profile or more severe cognitive deficits to accompany this. Such a conjuncture was however overturned by Katzen et al. [56], who found no significant cognitive differences between those with visual or multisensory hallucinations, though small subgroup sizes was a major drawback. Yet patterns of clustering of hallucination modalities, along with their timelines in relation to PD illness progression, remain a clinically significant issue to bear in mind.
Study limitations
The current review was subject to several methodological limitations. Owing to its scope focused on non-visual and multisensory hallucinations, established PD studies centred on visual hallucinations only were excluded (though the number of existing studies likely necessitates a systematic review in its own right). By the same token, research examining hallucinations more broadly without specifying modality were also left out (including those that utilised the UPDRS as their main hallucination assessment tool). Consequently, any findings relating solely to visual hallucinations were incomplete and conclusions drawn from this review must take this into account. The other major limitation relates to lack of consideration of PD medications, especially in terms of its effects and associated timelines. This was a conscious decision, as the range of drugs and varying doses employed to treat a myriad of PD symptoms renders this a complex and somewhat contentious topic. For instance, some PD drugs are known or held to induce psychosis [13, 57, 58], whereas other neuropsychiatric medications are evidently prescribed to treat hallucinations [59, 60]. Until clear effects of these substances on hallucinatory events can be established, current findings will again need to be interpreted within this lens. Moreover, a lack of neuropathological confirmation of clinically suspected PD across the included studies has limited the generalisability of our conclusions, given some cases may be re-classified as dementia with Lewy bodies or some other Parkinsonism syndrome post-mortem. Finally, it is possible that we may have missed papers that presented non-hallucinations data in their full-text, but not the abstract. However, our comprehensive and extensive search strategy, would hopefully have mitigated this. On the other hand, the chief strength of our review is a much needed focus on non-visual and multisensory hallucinations in PD, a hitherto neglected area, despite repeated calls from established PD researchers in the field [38, 61].
Future research
It is thus clear that future research needs to holistically focus on all hallucination modalities in PD, but especially non-visual and multisensory modes given their relative neglect to date [38, 61], several aspects of which are especially deserving of attention. Evidently, prevalence studies, particularly in auditory, olfactory, somatic-tactile and multisensory (with specified modality combinations) are lacking. Scrutinising descriptive phenomenology of the latter, where thematic and temporal associations are explicitly clarified, will also facilitate a better understanding of what these experiences are like from a patient’s perspective. Given heavy involvement of the visual domain, its intersection with tactile symptoms in so-called ‘string’ hallucinations [62] is of interest, whereas little is known about comorbid olfactory and gustatory hallucinations in PD [34, 46]. Given known olfactory difficulties in the disorder [40] as well as interplay with the gustatory domain [63], this area of enquiry could prove fruitful.
In terms of clinical and cognitive correlates, more targeted studies with regards to non-visual hallucinations are needed, where it is essential to clearly delineate hallucinations by modality. From a practical perspective, achieving ‘pure’ modality subgroups may be impossible, but the next best logical step would be to ensure that these ‘mixed’ modality subgroups are well-documented. Existing studies (even in visual hallucinations) have mostly focused on detecting the presence of hallucinations, but it is equally important to consider what contributes to hallucination severity. Besides the variables considered in this review, there are other clinical correlates for which there is preliminary evidence for further study, including sex, anxiety and even delusions. There is also scope to undertake a more nuanced investigation of intersections between phenomenological facets and cognitive domains. For instance, PD patients with visual hallucinations involving unfamiliar content were found to have more profound deficits in executive inhibition relative to those whose hallucinations comprised recognised content [64]. Such efforts may be replicated considering hallucinations in non-visual modalities. Finally, the few longitudinal studies in the area have been especially enlightening [38, 65]. In this vein, a comprehensive longitudinal examination of hallucination modality in line with PD illness progression will help to address existing gaps in knowledge, and significantly advance the field.
Conclusions
Even with the limited evidence base analysed in the current review, it is apparent that non-visual and multisensory hallucinations in PD are of clinical significance, and impact a notable proportion of patients. More research attention needs to be devoted to their study, especially in terms of establishing more accurate prevalence rates, as well as elucidating the involvement and clustering of specific non-visual and visual modalities, and their primary clinical-cognitive correlates. Doing so will likely yield prognostic and therapeutic benefits in a bid towards holistic management of the disorder.
Data availability statement
N/A.
Change history
25 February 2023
Missing Open Access funding information has been added in the Funding Note
References
Sprenger F, Poewe W (2013) Management of motor and non-motor symptoms in Parkinson’s disease. CNS Drugs 27(4):259–272
Poewe W (2008) Non-motor symptoms in Parkinson’s disease. Eur J Neurol 15:14–20
Poewe W (2003) Psychosis in Parkinson’s disease. Mov Disord 18:S80–S87
Factor SA et al (2003) Longitudinal outcome of Parkinson’s disease patients with psychosis. Neurology 60(11):1756–1761
Aarsland D, Zaccai J, Brayne C (2005) A systematic review of prevalence studies of dementia in Parkinson’s disease. Mov Disord 20(10):1255–1263
Hely MA et al (2008) The Sydney multicenter study of Parkinson’s disease: the inevitability of dementia at 20 years. Mov Disord 23(6):837–844
Galvin JE, Pollack J, Morris JC (2006) Clinical phenotype of Parkinson disease dementia. Neurology 67(9):1605–1611
Ramirez-Ruiz B et al (2006) Neuropsychological deficits in Parkinson’s disease patients with visual hallucinations. Mov Disord 21(9):1483–1487
Ravina B et al (2007) Diagnostic criteria for psychosis in Parkinson’s disease: report of an NINDS, NIMH work group. Mov Disord 22(8):1061–1068
Fenelon G, Alves G (2010) Epidemiology of psychosis in Parkinson’s disease. J Neurol Sci 289(1–2):12–17
Bertram K, Williams DR (2012) Visual hallucinations in the differential diagnosis of Parkinsonism. J Neurol Neurosurg Psychiatry 83(4):448–452
Eversfield CL, Orton LD (2019) Auditory and visual hallucination prevalence in Parkinson’s disease and dementia with Lewy bodies: a systematic review and meta-analysis. Psychol Med 49(14):2342–2353
Ffytche DH et al (2017) The psychosis spectrum in Parkinson disease. Nat Rev Neurol 13(2):81–95
Diederich NJ et al (2009) Hallucinations in Parkinson disease. Nat Rev Neurol 5(6):331–342
Frei K, Truong DD (2017) Hallucinations and the spectrum of psychosis in Parkinson’s disease. J Neurol Sci 374:56–62
Mosimann UP et al (2006) Characteristics of visual hallucinations in Parkinson disease dementia and dementia with lewy bodies. Am J Geriatr Psychiatry 14(2):153–160
Onofrj M, Thomas A, Bonanni L (2007) New approaches to understanding hallucinations in Parkinson’s disease: phenomenology and possible origins. Expert Rev Neurother 7(12):1731–1750
Barnes J, David AS (2001) Visual hallucinations in Parkinson’s disease: a review and phenomenological survey. J Neurol Neurosurg Psychiatry 70(6):727–733
Lenka A et al (2016) Interactions of visual hallucinations, rapid eye movement sleep behavior disorder and cognitive impairment in Parkinson’s disease: a review. Parkinsonism Relat Disord 22:1–8
Alzahrani H, Venneri A (2015) Cognitive and neuroanatomical correlates of neuropsychiatric symptoms in Parkinson’s disease: a systematic review. J Neurol Sci 356(1–2):32–44
Montagnese M et al (2022) Cognitive and visual processing performance in Parkinson’s disease patients with vs without visual hallucinations: a meta-analysis. Cortex 146:161–172
Pezzoli S et al (2021) Neuroanatomical and cognitive correlates of visual hallucinations in Parkinson’s disease and dementia with Lewy bodies: voxel-based morphometry and neuropsychological meta-analysis. Neurosci Biobehav Rev 128:367–382
Lenka A et al (2015) Structural and functional neuroimaging in patients with Parkinson’s disease and visual hallucinations: a critical review. Parkinsonism Relat Disord 21(7):683–691
Diederich NJ, Goetz CG, Stebbins GT (2005) Repeated visual hallucinations in Parkinson’s disease as disturbed external/internal perceptions: focused review and a new integrative model. Mov Disord 20(2):130–140
Muller AJ et al (2014) Visual hallucinations in Parkinson’s disease: theoretical models. Mov Disord 29(13):1591–1598
Burghaus L et al (2012) Hallucinations in neurodegenerative diseases. CNS Neurosci Ther 18(2):149–159
O’Brien J et al (2020) Visual hallucinations in neurological and ophthalmological disease: pathophysiology and management. J Neurol Neurosurg Psychiatry 91(5):512–519
Waters F et al (2014) Visual hallucinations in the psychosis spectrum and comparative information from neurodegenerative disorders and eye disease. Schizophr Bull 40(Suppl 4):233–245
Marques A et al (2022) Structural and functional correlates of hallucinations and illusions in Parkinson’s disease. J Parkinsons Dis 12(1):397–409
Swann P, O’Brien JT (2019) Management of visual hallucinations in dementia and Parkinson’s disease. Int Psychogeriatr 31(6):815–836
Moher D et al (2009) Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. BMJ 339:b2535
Munn Z et al (2020) Methodological quality of case series studies: an introduction to the JBI critical appraisal tool. JBI Evid Synth 18(10):2127–2133
Stang A (2010) Critical evaluation of the Newcastle-Ottawa scale for the assessment of the quality of nonrandomized studies in meta-analyses. Eur J Epidemiol 25(9):603–605
Solla P et al (2021) Frequency and determinants of olfactory hallucinations in Parkinson’s disease patients. Brain Sci 11(7):841
Fenelon G et al (2011) Feeling of presence in Parkinson’s disease. J Neurol Neurosurg Psychiatry 82(11):1219–1224
Kataoka H, Ueno S (2015) Predictable risk factors for the feeling of presence in patients with Parkinson’s disease. Mov Disord Clin Pract 2(4):407–412
Wood RA et al (2015) Fifty percent prevalence of extracampine hallucinations in Parkinson’s disease patients. Front Neurol 6:263
Goetz CG, Stebbins GT, Ouyang B (2011) Visual plus nonvisual hallucinations in Parkinson’s disease: development and evolution over 10 years. Mov Disord 26(12):2196–2200
Inzelberg R, Kipervasser S, Korczyn AD (1998) Auditory hallucinations in Parkinson’s disease. J Neurol Neurosurg Psychiatry 64(4):533–535
Bannier S et al (2012) Prevalence and phenomenology of olfactory hallucinations in Parkinson’s disease. J Neurol Neurosurg Psychiatry 83(10):1019–1021
Matsui H et al (2007) Thalamic hyperperfusion in verbal hallucination of Parkinsonian patients. Intern Med 46(21):1765–1769
Fenelon G et al (2002) Tactile hallucinations in Parkinson’s disease. J Neurol 249(12):1699–1703
de Chazeron I et al (2015) Validation of a psycho-sensory hallucinations scale (PSAS) in schizophrenia and Parkinson’s disease. Schizophr Res 161(2–3):269–276
Llorca PM et al (2016) Hallucinations in schizophrenia and Parkinson’s disease: an analysis of sensory modalities involved and the repercussion on patients. Sci Rep 6:38152
Rossell SL et al (2019) The questionnaire for psychotic experiences: an examination of the validity and reliability. Schizophr Bull 45(S1):S78–S87
Kulick CV, Montgomery KM, Nirenberg MJ (2018) Comprehensive identification of delusions and olfactory, tactile, gustatory, and minor hallucinations in Parkinson’s disease psychosis. Parkinsonism Relat Disord 54:40–45
Rana AQ et al (2013) Predicting treatment-seeking for visual hallucinations among Parkinson’s disease patients. Psychiatry Clin Neurosci 67(7):509–516
Fenelon G et al (2000) Hallucinations in Parkinson’s disease: prevalence, phenomenology and risk factors. Brain 123(Pt 4):733–745
Reckner E, Cipolotti L, Foley JA (2020) Presence phenomena in parkinsonian disorders: phenomenology and neuropsychological correlates. Int J Geriatr Psychiatry 35(7):785–793
Powell A, Matar E, Lewis SJG (2022) Treating hallucinations in Parkinson’s disease. Expert Rev Neurother 22(6):455–468
Factor SA, Molho ES (2004) Threatening auditory hallucinations and Cotard syndrome in Parkinson disease. Clin Neuropharmacol 27(5):205–207
Landis BN, Burkhard PR (2008) Phantosmias and Parkinson disease. Arch Neurol 65(9):1237–1239
Jimenez-Jimenez FJ et al (1997) Cenesthetic hallucinations in a patient with Parkinson’s disease. J Neurol Neurosurg Psychiatry 63(1):120
Ting SKS et al (2019) Phantom bedside intruder in Parkinson’s disease. Neurol Clin Neurosci 7(2):91–93
Whitehead DL et al (2008) Circadian rest-activity rhythm is altered in Parkinson’s disease patients with hallucinations. Mov Disord 23(8):1137–1145
Katzen H et al (2010) Multi-modal hallucinations and cognitive function in Parkinson’s disease. Dement Geriatr Cogn Disord 30(1):51–56
Chou KL et al (2005) Drug-induced psychosis in Parkinson disease: phenomenology and correlations among psychosis rating instruments. Clin Neuropharmacol 28(5):215–219
Goetz CG et al (1998) Early dopaminergic drug-induced hallucinations in Parkinsonian patients. Neurology 51(3):811–814
J.D. Mellers, N.P. Quinn, M.A. Ron (1995) Psychotic and depressive symptoms in Parkinson’s disease A study of the growth hormone response to apomorphine. Br J Psychiatry 167(4): 522–526
Trosch RM et al (1998) Clozapine use in Parkinson’s disease: a retrospective analysis of a large multicentered clinical experience. Mov Disord 13(3):377–382
Fenelon G et al (2010) The changing face of Parkinson’s disease-associated psychosis: a cross-sectional study based on the new NINDS-NIMH criteria. Mov Disord 25(6):763–766
Perez-Perez J et al (2016) “String hallucinations”: Multimodal tactile and visual hallucinations in Parkinson’s disease. Mov Disord Clin Pract 3(2):180–183
Finger TE, Silver WL (1991) Neurobiology of taste and smell. Krieger Publishing, Malabar, Florida
Boubert L, Barnes J (2015) Phenomenology of visual hallucinations and their relationship to cognitive profile in Parkinson’s disease patients: preliminary observations. Sage Open. https://doi.org/10.1177/2158244015585827
Pagonabarraga J et al (2016) Minor hallucinations occur in drug-naive Parkinson’s disease patients, even from the premotor phase. Mov Disord 31(1):45–52
Aarsland D et al (2001) A comparative study of psychiatric symptoms in dementia with Lewy bodies and Parkinson’s disease with and without dementia. Int J Geriatr Psychiatry 16(5):528–536
Barrett MJ et al (2017) Characteristics, correlates, and assessment of psychosis in Parkinson disease without dementia. Parkinsonism Relat Disord 43:56–60
Chendo I et al (2021) High frequency of psychosis in late-stage Parkinsons disease. Clin Park Relat Disord 5:100119
de Maindreville AD, Fenelon G, Mahieux F (2005) Hallucinations in Parkinson’s disease: a follow-up study. Mov Disord 20(2):212–217
Factor SA et al (2014) Cognitive correlates of hallucinations and delusions in Parkinson’s disease. J Neurol Sci 347(1–2):316–321
Holroyd S, Currie L, Wooten GF (2001) Prospective study of hallucinations and delusions in Parkinson’s disease. J Neurol Neurosurg Psychiatry 70(6):734–738
Ikeda M, Kataoka H, Ueno S (2016) Fatigue is associated with the onset of hallucinations in patients with Parkinson’s disease: a 3-year prospective study. eNeurologicalSci 4:25–29
Lee AH, Weintraub D (2012) Psychosis in Parkinson’s disease without dementia: common and comorbid with other non-motor symptoms. Mov Disord 27(7):858–863
Lenka A et al (2017) Predictors of onset of psychosis in patients with Parkinson’s disease: who gets it early? Parkinsonism Relat Disord 44:91–94
Leu-Semenescu S et al (2011) Hallucinations in narcolepsy with and without cataplexy: contrasts with Parkinson’s disease. Sleep Med 12(5):497–504
Mack J et al (2012) Prevalence of psychotic symptoms in a community-based Parkinson disease sample. Am J Geriatr Psychiatry 20(2):123–132
Marsh L et al (2004) Psychiatric comorbidities in patients with Parkinson disease and psychosis. Neurology 63(2):293–300
McAuley JH, Gregory S (2012) Prevalence and clinical course of olfactory hallucinations in idiopathic Parkinson’s disease. J Parkinsons Dis 2(3):199–205
Muller AJ et al (2018) Informant- and self appraisals on the psychosis and hallucinations questionnaire (PsycH-Q) enhances detection of visual hallucinations in Parkinson’s disease. Mov Disord Clin Pract 5(6):607–613
Nishio Y et al (2017) Deconstructing psychosis and misperception symptoms in Parkinson’s disease. J Neurol Neurosurg Psychiatry 88(9):722–729
Omoto S et al (2021) Risk factors for minor hallucinations in Parkinson’s disease. Acta Neurol Scand 143(5):538–544
Pacchetti C et al (2005) Relationship between hallucinations, delusions, and rapid eye movement sleep behavior disorder in Parkinson’s disease. Mov Disord 20(11):1439–1448
Paleacu D, Schechtman E, Inzelberg R (2005) Association between family history of dementia and hallucinations in Parkinson disease. Neurology 64(10):1712–1715
Papapetropoulos S et al (2008) A questionnaire-based (UM-PDHQ) study of hallucinations in Parkinson’s disease. BMC Neurol 8:21
Rai NK et al (2015) Neuropsychiatric co-morbidities in non-demented Parkinson’s disease. Ann Indian Acad Neurol 18(1):33–38
Sawada H et al (2013) Trigger medications and patient-related risk factors for Parkinson disease psychosis requiring anti-psychotic drugs: a retrospective cohort study. BMC Neurol 13:145
Shine JM et al (2015) Validation of the psychosis and hallucinations questionnaire in non-demented patients with Parkinson’s disease. Mov Disord Clin Pract 2(2):175–181
Svetel M et al (2012) Hallucinations in Parkinson’s disease: cross-sectional study. Acta Neurol Belg 112(1):33–37
Zhang Y et al (2021) Clinical characteristics of minor hallucinations in Chinese Parkinson’s disease patients. Front Aging Neurosci 13:723405
Zhong M et al (2021) Prevalence and risk factors for minor hallucinations in patients with Parkinson’s disease. Behav Neurol 2021:3469706
Zhu J et al (2017) Prevalence and risk factors for visual hallucinations in Chinese patients with Parkinson’s disease. J Neurol Sci 372:471–476
Abe K et al (2016) A case of Parkinson disease with both visual hallucination and pain improved by gabapentin. Clin Neuropharmacol 39(1):55–56
Amar BR et al (2014) A clinical profile of patients with Parkinson’s disease and psychosis. Ann Indian Acad Neurol 17(2):187–192
Arnulf I et al (2000) Improvement of sleep architecture in PD with subthalamic nucleus stimulation. Neurology 55(11):1732–1734
Benbir G et al (2006) Features associated with the development of hallucinations in Parkinson’s disease. Acta Neurol Scand 114(4):239–243
Clark J (1998) Case history of a patient with musical hallucinations and Parkinson’s disease. Int J Geriatr Psychiatry 13(12):886–887
Dashtipour K et al (2021) Pimavanserin treatment for Parkinson’s disease psychosis in clinical practice. Parkinsons Dis 2021:2603641
Ergun U et al (2009) Musical hallucinations and Parkinson disease. Neurologist 15(3):150–152
Fernandez W, Stern G, Lees AJ (1992) Hallucinations and Parkinsonian motor fluctuations. Behav Neurol 5(2):83–86
Goetz CG, Tanner CM, Klawans HL (1982) Pharmacology of hallucinations induced by long-term drug therapy. Am J Psychiatry 139(4):494–497
Gondim Fde A et al (2010) Transient amantadine-induced musical hallucinations in a patient with Parkinson’s disease. Mov Disord 25(10):1505–1506
Gupta M et al (2004) Hallucinations in Parkinson’s disease: a study of forty three patients. J Assoc Physicians India 52:703–706
Jimenez-Jimenez FJ et al (1997) Acoustic voice analysis in untreated patients with Parkinson’s disease. Parkinsonism Relat Disord 3(2):111–116
Kataoka H et al (2014) Can dopamine agonists trigger tactile hallucinations in patients with Parkinson’s disease? J Neurol Sci 347(1–2):361–363
Kataoka H, Ueno S (2014) Auditory musical hallucinations associated with extended-release pramipexole in an elderly patient with Parkinson’s disease. Medicine (Baltimore) 93(27):e251
Kesserwani H (2021) Delusional infestation secondary to the dopamine agonist ropirinole in a patient with Parkinson’s disease: a case report with an outline of the biology of psychosis. Cureus 13(1):e12880
Lenka A, Reddy YCJ, Pal PK (2018) Isolated tactile hallucination in a patient with Parkinson’s disease: Quetiapine conquers the snakes and stones. Asian J Psychiatr 35:34–35
Meco G, Bernardi S (2007) Antidepressant use in treatment of psychosis with comorbid depression in Parkinson’s disease. Prog Neuropsychopharmacol Biol Psychiatry 31(1):311–313
Mittal M, Giron LT Jr (2010) Elvis is back: Musical hallucinations in a Parkinson disease patient. South Med J 103(8):837–841
Moskovitz C, Moses H 3rd, Klawans HL (1978) Levodopa-induced psychosis: a kindling phenomenon. Am J Psychiatry 135(6):669–675
Muralidharan K et al (2011) Bifrontal ECT for drug-induced psychosis in Parkinson’s disease. Indian J Psychiatry 53(2):156–158
Nagata T et al (2013) A case in which mirtazapine reduced auditory hallucinations in a patient with Parkinson disease. Int Psychogeriatr 25(7):1199–1201
Nishioka K et al (2014) Quantitative evaluation of electroconvulsive therapy for Parkinson’s disease with refractory psychiatric symptoms. J Neural Transm (Vienna) 121(11):1405–1410
Onofrj M et al (2000) Leucopenia induced by low dose clozapine in Parkinson’s disease recedes shortly after drug withdrawal. Clinical case descriptions with commentary on switch-over to olanzapine. Neurol Sci 21(4):209–215
Parsa MA, Bastani B (1998) Quetiapine (seroquel) in the treatment of psychosis in patients with Parkinson’s disease. J Neuropsychiatry Clin Neurosci 10(2):216–219
Potheegadoo J et al (2022) Presence hallucinations during locomotion in patients with Parkinson’s disease. Mov Disord Clin Pract 9(1):127–129
Roberts HE, Dean RC, Stoudemire A (1989) Clozapine treatment of psychosis in Parkinson’s disease. J Neuropsychiatry Clin Neurosci 1(2):190–192
Tousi B, Frankel M (2004) Olfactory and visual hallucinations in Parkinson’s disease. Parkinsonism Relat Disord 10(4):253–254
Voon V, Lang AE (2004) Antidepressants in the treatment of psychosis with comorbid depression in Parkinson disease. Clin Neuropharmacol 27(2):90–92
Wand AP (2012) Transient citalopram-induced auditory hallucinations in a patient with Parkinson’s disease and depression. Aust N Z J Psychiatry 46(2):178
Yoshida F et al (2009) Subthalamic nucleus stimulation does not cause deterioration of preexisting hallucinations in Parkinson’s disease patients. Stereotact Funct Neurosurg 87(1):45–49
Marques A et al (2021) Psycho-sensory modalities of visual hallucinations and illusions in Parkinson’s disease. Rev Neurol (Paris) 177(10):1228–1236
Acknowledgements
The authors would like to thank the Barbara Dicker Brain Sciences Foundation for their support and for funding this study.
Funding
Open Access funding enabled and organized by CAUL and its Member Institutions.
Author information
Authors and Affiliations
Contributions
Study conception and design were led by WLT, SLR and CY. The literature search was performed by CY. Data screening, extraction and analysis were performed by WLT and CY. The first draft of the manuscript was written by WLT, and critically reviewed by CY and SLR. All authors read, commented and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no financial or non-financial interests relevant to the manuscript to disclose.
Ethical standards
This is a systematic review for which no ethical approval is required. All analysed data were derived from existing peer reviewed publications.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Toh, W.L., Yolland, C., Gurvich, C. et al. Non-visual hallucinations in Parkinson’s disease: a systematic review. J Neurol 270, 2857–2889 (2023). https://doi.org/10.1007/s00415-022-11545-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-022-11545-6