Abstract
Objective
To determine longitudinal predictors of health-related quality of life (HR-QoL) in an international multicenter cohort of patients with isolated dystonia.
Methods
Out of 603 dystonia patients prospectively enrolled in the Natural History Dystonia Coalition study, 155 were assessed three times within 2 years for HR-QoL, symptoms of depression, generalized anxiety disorder (GAD), and social anxiety disorder (SAD), as well as dystonia severity and dystonic tremor. In addition, the impact of botulinum neurotoxin (BoNT) injections on HR-QoL was evaluated after 1 year.
Results
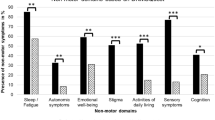
Depressive symptoms at baseline predicted lower HR-QoL on all subscales after 2 years (all p ≤ 0.001). Higher GAD scores at baseline predicted lower HR-QoL related to general health, pain and emotional well-being, whereas higher SAD scores predicted higher pain-related QoL after 2 years (all p ≤ 0.006). Dystonia severity at baseline predicted social functioning (p = 0.002). Neither dystonic tremor, age, or sex predicted HR-QoL at 2 years. Two latent categories were revealed across the three-time points: Category 1 with higher total HR-QoL scores (mean HR-QoL = 74.4% ± 16.1), susceptible to symptoms of depression and SAD, and Category 2 with lower total HR-QoL scores (mean HR-QoL = 45.5% ± 17.6), susceptible to symptoms of GAD. HR-QoL improved over the course of 1 year irrespective of the use of BoNT.
Conclusion
The longitudinal impact of psychiatric symptoms on HR-QoL emphasizes the importance of incorporating mental health treatment, in particular also the therapy of anxiety disorders, into treatment regimens for dystonia.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Nonmotor symptoms (NMS), i.e. depression, generalized anxiety disorder (GAD), social anxiety disorder (SAD), self-esteem and acceptance of illness often concomitate the motor manifestations in isolated dystonia, occur more frequently than in healthy controls [1,2,3,4,5,6,7] and impact on the health-related quality of life (HR-QoL) [8,9,10,11,12]. The previous studies on HR-QoL in dystonia have been cross-sectional [8,9,10,11,12,13,14] and do not allow conclusions on predictors of future QoL. Longitudinal examinations in cranial and cervical dystonia several weeks after botulinum neurotoxin (BoNT) injection revealed an improvement of dystonia severity and improvement of certain HR-QoL domains [3, 15,16,17,18,19,20]. The prevalence of NMS such as depression and anxiety and the reported QoL in a small group of patients with cervical dystonia did, however, not change within 2 years [21].
This is the first study analyzing HR-QoL data in a large, international, prospective cohort of patients with adult-onset isolated dystonia to identify predictors of HR-QoL at 2 years after baseline examination. As BoNT is an established therapy to alleviate the severity of MS in dystonia, we additionally evaluated the longitudinal effects of BoNT on HR-QoL. Identifying predictors of HR-QoL in dystonia can help clinicians screen for patients at high risk of low HR-QoL and develop long-term therapy regimens.
Methods
Participants
Data from the Natural History Project of the Dystonia Coalition enrolled across 36 clinical sites (USA, Canada, Australia, Germany, France, Italy) were analyzed. Additional Dystonia Coalition investigators that contributed subjects to the study are listed in Online Resource 1. The multicenter Dystonia Coalition study includes dystonia patients aged 18 years and older, and the Natural History Project focuses on patients with dystonia onset no more than 5 years before study enrollment (https://www.rarediseasesnetwork.org/cms/dystonia).
Participants included in this study were examined three times within 2 years (baseline, 1-year follow-up, 2-year follow-up). At each study visit, the subjects answered standardized questionnaires and were clinically examined using a standardized protocol. Dystonia patients receiving BoNT treatment were enrolled when symptoms returned. This usually meant they were enrolled 3 months after treatment, but never sooner than two months after treatment. Exclusion criteria were secondary or combined dystonia and medical/neurological conditions confounding diagnoses or precluding a complete assessment.
Questionnaire and rating scales
Information regarding the questionnaire and rating scales (Dystonia Coalition Questionnaire, RAND 36-Item Health Survey, Hospital Anxiety and Depression Scale (HADS), Liebowitz Social Anxiety Scale (LSAS)), as well as the examination protocol and rating (Burke − Fahn − Marsden Dystonia Rating Scale (BFMDRS), tremor) have been previously published [8].
HR-QoL was evaluated by the eight subscales (general health, physical functioning, role limitations due to physical health problems, pain, energy/fatigue, emotional well-being, role limitations due to emotional problems and social functioning) and the total score of the RAND 36-Item Health Survey [22]. Total HR-QoL is represented by the mean of the eight subscales of the RAND 36-Item Health Survey. The severity of generalized anxiety and depressive symptoms was assessed by the self-reported HADS version 4 [23] and symptoms of social anxiety by the LSAS [24]. The clinical examination included the evaluation of dystonia severity, using the validated BFMDRS [25], as well as the existence of tremors (yes/no).
Statistical analysis
Predictors of 2-year HR-QoL subdomains were obtained from eight cross-lagged path models (CLPM, one model per domain; Bonferroni corrected alpha ≤ 0.006). Further, an independent analysis was done to categorize patients into clusters of high and low total HR-QoL within the 2 years period using a latent class growth analysis (LCGA). Data at baseline included 603 patients. Only patients with complete data sets at time point three (2-year follow-up) were included in the CLPM (n = 155) and only patients with complete data sets across the three time points (baseline, 1-year follow-up, and 2-year follow-up) were included in the LCGA (n = 145). Both the CLPM and the LCGA were carried out in MPlus version 7.4 [26].
Within the CLPM, the statistical effects of baseline dystonia severity, tremor (yes/no), age, sex, and symptom severity of depression (HADS-D), GAD (HADS-A) and SAD (LSAS), as well as the eight subscales of the RAND 36-Item Health Survey, were assessed as direct paths from baseline on the same variables at 1-year follow-up and as indirect paths via (intermediate) direct effects on the same variables at 2-year follow up (Fig. 1 and Online Resource 2_Fig. 3). The eight cross-lagged path models assessed the strength of directional associations between constructs over time while controlling for all other constructs included in the model and the correlations between the concepts at each time point [27]. Model fit was evaluated based on the proportions of variation (r2) that were explained in each statistically dependent variable [28].
Direct effects of the cross-lagged path model for the general health subscale (stylized illustration). Direct paths of the cross-lagged path model for the general health subscale of HR-QoL are presented. Dashed lines illustrate the effect of age and sex on the different variables. Bonferroni corrected alpha is ≤ 0.006. Significant paths are marked in red, grey paths did not reach significance level. The other seven HR-QoL cross-lagged path models are presented in Online Resource 2_Fig. 3
To investigate the impact of dystonia severity, sex, age, the severity of depression, GAD and SAD on the development of total quality of life over time, linear growth in HR-QoL was estimated over the 2 years and combined with a statistical examination for distinct patterns of growth (‘latent classes’; hereafter, ‘categories’ to ease interpretation by clinicians). Robust maximum likelihood estimation was used to minimize biased results caused by non-normally distributed variables. An increasing number of categories were investigated, and the accuracy of the resulting categories was subjected to two formal statistical tests (Lo-Mendell − Rubin adjusted likelihood ratio test (LMR), Vuong − Lo-Mendell − Rubin likelihood ratio test (VLMR)) as to whether a statistical model estimating n categories offered a more accurate representation of the data than the n-1 category alternative. A two-category solution returned a significantly more accurate representation of the data than did a single category alternative (LMR: p = 0.042, VLMR: p = 0.041). However, there was also no significant improvement in the model fit by extending the estimate to three categories (LMR: p = 0.217, VLMR: p = 0.214). Furthermore, the two-category model was better able to represent HR-QoL over the three measurement points than was the less parsimonious three-category alternative (entropy value of 0.92 as compared to 0.87).
Annual changes of HR-QoL in dystonia were evaluated using a nonparametric ANalysis Of VAriance (ANOVA). Only patients with complete longitudinal data sets across the two time points were included in the ANOVA (n = 72). Data from two subgroups of patients were analyzed: patients not treated with BoNT (neither at baseline nor at follow-up, n = 37) and patients who received BoNT within 1-year of follow-up but not before baseline (n = 35). The analysis was corrected for oral medication intake such that only patients with unchanged or no oral medication at both time points were considered. Categorization of dystonia medication was performed according to Richardson et al.[29]. For this ANOVA, patients with local surgical treatment for dystonia (myectomy, denervation) were excluded (n = 3). The analysis was performed in R version 1.1.463. Mann − Whitney and Chi-square tests were run in SPSS Statistics 22 (IBM Corp., Armonk, NY) to evaluate group differences. All tests of significance were two-sided. P values ≤ 0.05 were considered significant unless Bonferroni-corrected.
Data availability
Anonymized data (study protocol, statistical analysis) will be shared by request from any qualified investigator. Data will be available for 10 years.
Results
Out of 603 participants, complete longitudinal data sets for the CLPM and the LCGA were available from 155 patients (representing 25.7% from our previous study [8]). The majority of patients were female (63.9%, 99/155), the mean age at baseline examination was 57.6 ± 11.1 years, and the mean age at dystonia onset was 54.8 ± 10.9 years. Table 1 displays mean BFMDRS scores, the ratio of patients with tremor, mean of RAND 36-Item Health Survey scores, HADS-D, HADS-A, and LSAS, as well as the ratio of patients with symptoms of depression (HADS-D > 7), GAD (HADS-A > 7), and SAD (LSAS > 30) at baseline.
Regarding therapy, 67.1% (104/155) were treated with BoNT, 41.3% (64/155) with oral antidystonic drugs (anticholinergics, benzodiazepines, nonbenzo-hypnotics, dopaminergics, antidopaminergics, muscle relaxants) and 27.7% (43/155) with antidepressants at baseline. None of the patients had a deep brain stimulator (DBS), whereas three patients had previously received a myectomy for blepharospasm or spasmodic dysphonia.
Predictors of the eight HR-QoL domains within 2 years
Higher depression scores at baseline predicted lower HR-QoL on all eight subscales at 2 years (Total indirect effect: General Health: β = – 1.82, Physical Functioning: β = – 2.22, Physical Role Functioning: β = – 3.41, Pain: β = – 1.87, Energy/Fatigue: β = – 2.23, Emotional Well-being: β = – 1.17, Emotional Role Functioning: β = – 3.46, Social Functioning: β = – 2.71, all p < 0.001). Higher GAD scores at baseline predicted lower HR-QoL related to general health (Total indirect effect: β = – 0.87, p = 0.005), pain (Total indirect effect: β = – 1.50, p = 0.002) and emotional well-being (Total indirect effect: β = – 1.26, p < 0.001) after 2 years, whereas higher SAD scores at baseline predicted higher pain-related QoL (Total indirect effect: β = 0.17, p = 0.002). Higher dystonia severity at baseline predicted lower HR-QoL in the context of social functioning (Total indirect effect: β = – 0.69, p = 0.002). The total indirect effects could not be calculated for binary variables such as tremor. Tremor had no direct effects on any of the eight HR-QoL subscales (all p > 0.006). The total direct effects of the eight cross-lagged path models are presented in Fig. 1 and Online Resource 2_Fig. 3.
Predictors of total HR-QoL within 2 years
LCGA revealed two categories distinguished by the total HR-QoL that was reported across the three time points by 145 longitudinally examined dystonia patients (Fig. 2).
Latent class growth analysis. The LCGA revealed two latent classes (here “categories”), distinguished by the total HR-QoL (in %) that was reported across the three time points (baseline, 1-year follow-up, 2-year follow-up). Category 1 (66% of patients) reported a consistently higher level of HR-QoL that was susceptible to symptoms of depression and SAD, whereas category 2 (34% of patients) reported a consistently lower level of HR-QoL that was susceptible to symptoms of GAD. y-axis represents HR-QoL (total) in percent with standard error
Category 1
66% of patients reported a higher level of quality of life over the three points (mean HR-QoL = 74.36% ± 16.07). In this group, HR-QoL was susceptible to symptoms of depression and SAD. Higher depression scores reported at baseline predicted lower HR-QoL after 2 years (β = – 0.40, p = 0.047) as did higher levels of SAD (β = – 0.42, p = 0.017). In contrast, patients with lower depression scores at baseline had a more stable HR-QoL score over the 2-year period (flatter statistical slope, β = 0.88, p < 0.001). Overall, the patients had no significant increase of HR-QoL trajectories over the 2 years (beta = 1.99, p = 0.09). No relationship was present between HR-QoL and severity of GAD (intercept β = – 0.37, p = 0.095; slope β = – 0.29, p = 0.32), age (intercept β = – 0.33, p = 0.093; slope β = – 0.29, p = 0.203), sex (intercept β = – 0.11, p = 0.622; slope β = – 0.47, p = 0.057), or dystonia severity (intercept β = 0.01, p = 0.972; slope β = 0.15, p = 0.510).
Category 2
34% of patients reported a consistently lower level of HR-QoL over the 2-year period (mean HR-QoL = 45.54% ± 17.64). In this group of patients, HR-QoL was susceptible to symptoms of GAD, i.e., higher GAD baseline levels were associated with lower levels of HR-QoL after 2 years (β = – 0.71, p < 0.001). There was no relationship between HR-QoL and severity of depression (intercept β = – 0.26, p = 0.187; slope β = 0.79, p = 0.190), severity of SAD (intercept β = – 0.12, p = 0.488; slope β = 0.27, p = 0.581), age (intercept β = – 0.08, p = 0.732; slope beta = 0.18, p = 0.789), sex (intercept β = 0.36, p = 0.052, slope β = 0.53, p = 0.314), or dystonia severity (intercept β = 0.13, p = 0.520; slope β = 0.31, p = 0.505). Overall, patients in this class had no significant decrease of HR-QoL trajectories over the 2 years (beta = – 3.78, p = 0.331).
Influence of BoNT therapy on HR-QoL within 1 year
A nonparametric ANOVA with HR-QoL total score as dependent variable, time (1 year) as a within-subject factor and treatment group (BoNT initiated/no BoNT) as a between-subject factor revealed a main effect for time [F(1,67.9) = 9.6, p = 0.002] to improve HR-QoL after 1 year, and no effect for group [F(1,67.9) = 1.0, p = 0.314] or the interaction between time and group [F(1,67.9) = 0.2, p = 0.697]. Another nonparametric ANOVA with BFMDRS as dependent variable, time as a within-subject factor and group as a between-subject factor revealed a main effect for time [F(1,64.3) = 5.5, p = 0.019] in the BFMDRS score decline, and no effect for group [F(1,64.3) = 0.1, p = 0.738] or the interaction between time and group [F(1,64.3) = 0.04, p = 0.846]. Bonferroni corrected alpha was ≤ 0.025.
The results of the nonparametric ANOVAs with the eight HR-QoL subscales (RAND 36-Item Health Survey) as a dependent variable, time as a within-subject factor and group as a between-subject factor are displayed in Table 2. Bonferroni corrected p was ≤ 0.006. Group differences were found for emotional well-being only.
The BoNT-treated group had higher depression scores (HADS-D) at baseline than the untreated group (U = 360.0, p = 0.002), whereas anxiety (HADS-A; U = 470.0, p = 0.066) and social anxiety scores (LSAS; U = 446.5, p = 0.036) did not differ between groups (Bonferroni corrected alpha was ≤ 0.017, Table 3). Age (U = 583.0, p = 0.467) and sex (χ2 = 0.498; p = 0.633) were not different between the two groups.
Discussion
Our international, multicenter study aimed to identify predictors of HR-QoL in isolated dystonia involving 155 patients over a time period of 2 years. Symptoms of depression predicted all eight subdomains of HR-QoL, whereas higher generalized anxiety scores predicted lower HR-QoL of three subdomains (general health, pain, emotional well-being) and higher social anxiety symptoms higher pain-related QoL. In contrast, dystonia severity was associated with limitations in social functioning only. We additionally separated two categories of patients with either higher or lower HR-QoL and identified a distinct profile of predictors in each. HR-QoL was more dependent on symptoms of depression and social anxiety in patients with a higher QoL whereas generalized anxiety predicted HR-QoL in the group of patients with a lower HR-QoL.
Symptoms of depression are a strong determinator of HR-QoL in dystonia which is in keeping with results of several previous cross-sectional studies in cervical and craniofacial dystonia [3, 9, 12] and other forms of isolated dystonia [8, 30, 31]. In our previous cross-sectional study involving 603 patients from the same cohort including the patients investigated here, and in a study in cervical dystonia, also symptoms of GAD and SAD were associated with all eight subdomains of HR-QoL [8, 9]. Interestingly, this effect was not as strong when the patients in the present study were investigated longitudinally, as fewer domains appeared to be influenced by symptoms of GAD (n = 3) and SAD (n = 1). This could be in part explained by the smaller sample size leading to less statistical power. However, a recent cross-sectional study in patients with craniofacial dystonia showed similar results using multiple linear regression analysis of depression, anxiety, and SF-36 scores. Anxiety correlated with all eight subdomains of SF36, but after adjusting for disease duration, education level, and depression correlated only with the pain subdomain [12]. Although depression predicted all subdomains of future HR-QoL in our study, GAD predicted less but relevant subdomains such as general health, emotional well-being and pain and GAD was associated with a significantly lower total QoL as compared to depression (mean HR-QoL: 45.5% ± 17.6 vs. 74.4% ± 16.1). This may in part be due to behavioral and pharmacologic therapy for GAD being less frequently employed as compared to the established guidelines of depression treatment. This highlights the importance of screening for symptoms of both psychiatric disorders. Of note, higher SAD scores predicted higher pain-related QoL at 2 years, which was unexpected. A possible interpretation of this association is, however, that patients with SAD may be less active, resulting in reduced bodily activity, which may consequently be associated with less occurrence of pain. Furthermore, there might be a pathway convergence of the anxiety and medial nociceptive system, which is connected to the cingulate gyrus, amygdala and hypothalamus, sub-serving the affective-motivational aspects of pain [32, 33].
Dystonia severity was a determinant of HR-QoL in cervical dystonia and was associated with four out of eight subdomains of HR-QoL in our cross-sectional study analysis [8, 9]. The effect vanished, however, in the longitudinal design of the present study with dystonia severity only being a predictor in the context of social functioning. The high burden on HR-QoL of NMS rather than solely MS of dystonia, as also proven e.g. in patients with writer’s cramp [34], thus emphasizes the impact of coping strategies, acceptance of illness, as well as management of mood and anxiety on the maintenance of an acceptable HR-QoL [10]. Nevertheless, methodological differences, such as dystonia severity being rated by patients [9] or movement disorder specialists [8], as well as the use of different rating scales, should be taken into account. The BFMDRS, which is a validated [25] and commonly used rating scale, can be limited when assessing different focal dystonia phenotypes. In our study, 93 patients had focal dystonia and only 40% segmental, multifocal or generalized dystonia. Furthermore, 67% of our patients were treated with BoNT about three months prior to the examination. Thus, dystonia severity may have been potentially higher in some patients without long-term BoNT therapy [35] and may have resulted in stronger associations with lower HR-QoL. This question could only be answered with a longitudinal study design in which a large number of untreated and significantly affected dystonia patients were included and BoNT therapy was initiated during the course of the study to evaluate the effect at the time of peak BoNT efficacy.
In contrast to our previous cross-sectional results on HR-QoL in dystonia showing tremor impacting HR-QoL in the context of physical functioning and pain [8], here tremor was not found to predict future HR-QoL. As in our cross-sectional study, only the existence of tremor, but not its severity or distribution, was evaluated which is a limitation [8]. This still highlights that NMS have a greater impact on the quality of life of dystonia patients than MS. Nevertheless, a long-term improvement in patients with long-term BoNT therapy cannot be excluded, although patients were evaluated about three months after BoNT treatment.
Interestingly, there was a similar 1-year improvement in dystonia severity and total HR-QoL in a subgroup of patients with and without BoNT therapy. There were no group differences regarding age, sex and HR-QoL subscales except for emotional well-being, which was most likely a result of higher baseline depression scores in the BoNT-treated group. These results are in contrast to recently published data describing unchanged total QoL in patients with cervical dystonia at 2 years. However, these results are not comparable because only patients with cervical dystonia were included and the assessment was performed approximately 12 weeks after the previous BoNT injection which is different from our design [21]. Furthermore, the CDIP‐58, used by Moriarty et al. [21], is a cervical dystonia-specific patient self-report of QoL [36] with a stronger focus on motor symptoms as compared to the RAND 36, a commonly used, generic quality of life questionnaire [37]. We can only speculate as to the significance of the finding of a similar 1-year improvement in dystonia severity and total HR-QoL independent of the BoNT effect. Several possibilities are proposed. First, there may be a nonspecific quality-enhancing effect created by the close clinical monitoring required in a research protocol, expanding into those not receiving BoNT. Furthermore, the factors unrelated to those targeted by chemodenervation and unaccounted for in this study may have yielded the magnitude of effects quantified by the HR-QoL [10]. Physical and cognitive − behavioral therapy has been shown to improve dystonia severity and HR-QoL in cervical dystonia [38,39,40,41,42]. These aspects, however, were not systematically assessed here. Alternatively, the beneficial effects of BoNT may not be readily captured by the evaluation of HR-QoL at the end of the BoNT treatment cycle resulting in the absence of a significant interaction between time and group. Examinations in cranial and cervical dystonia four, six or eight weeks after BoNT injection likewise revealed an improvement of dystonia severity and improvement of some HR-QoL domains [3, 15,16,17,18,19]. In patients with cervical dystonia, nonmotor symptoms as depression and anxiety improved one month after BoNT treatment [3, 15, 43] with a slighter but still significant improvement after three months [43]. Thus, the decreased effect of BoNT at the time point of the follow-up visit of the present study is likely to have an impact on the results of measurement in the patient group with BoNT treatment.
This longitudinal study has some limitations: To guarantee statistical power some potential confounding factors such as educational level, marital and socio-economic status were not assessed but may significantly influence HR-QoL. The BFMDRS is a validated [25] and commonly used rating scale for dystonia. Nevertheless, it may not be sufficiently representative of focal dystonias. Furthermore, only the existence and not the severity, distribution, frequency, amplitude and regularity of tremor were evaluated. Examination of patients at the end of the BoNT treatment cycle only allowed the evaluation of long-term effects. In addition, samples sizes of this subanalysis were not large enough to detect significant small effects (at α = 0.05). However, the groups with and without BoNT treatment were well defined and well corrected for other therapeutic influences.
In summary, the most comprehensive predictors of HR-QoL in isolated dystonia are symptoms of depression, followed by GAD, whereas severity of dystonia only predicts social functioning. Dystonia patients with higher levels of anxiety have lower HR-QoL than patients with symptoms of depression and social anxiety, possibly as a consequence of less utilization of medication and behavioral therapy for GAD. Although standard therapy regimens as BoNT mainly focus on the physical symptoms, depression and anxiety should be specifically identified and targeted to improve long-term HR-QoL in dystonia.
Standard Protocol Approvals and Patient Consents
All participants gave written informed consent for study participation prior to their enrollment. The study was approved by the local ethics committees of all clinical sites and performed in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki and its later amendments.
References
Bailey GA, Martin E, Peall KJ (2022) Cognitive and Neuropsychiatric Impairment in Dystonia. Curr Neurol Neurosci Rep 22:699–708. https://doi.org/10.1007/s11910-022-01233-3
Berman BD, Junker J, Shelton E, Sillau SH, Jinnah HA, Perlmutter JS, Espay AJ, Jankovic J, Vidailhet M, Bonnet C, Ondo W, Malaty IA, Rodríguez R, McDonald WM, Marsh L, Zurowski M, Bäumer T, Brüggemann N (2017) Psychiatric associations of adult-onset focal dystonia phenotypes. J Neurol Neurosurg Psychiatry 88:595–602. https://doi.org/10.1136/jnnp-2016-315461
Slawek J, Friedman A, Potulska A, Krystkowiak P, Gervais C, Banach M, Ochudlo S, Budrewicz S, Reclawowicz D, Rynkowski M, Opala G (2007) Factors affecting the health-related quality of life of patients with cervical dystonia and the impact of botulinum toxin type A injections. Funct Neurol 22:95–100
Lewis L, Butler A, Jahanshahi M (2008) Depression in focal, segmental and generalized dystonia. J Neurol 255:1750–1755. https://doi.org/10.1007/s00415-008-0020-x
Gundel H, Wolf A, Xidara V, Busch R, Ceballos-Baumann AO (2001) Social phobia in spasmodic torticollis. J Neurol Neurosurg Psychiatry 71:499–504
Kuyper DJ, Parra V, Aerts S, Okun MS, Kluger BM (2011) Nonmotor manifestations of dystonia: a systematic review. Mov Disord 26:1206–1217. https://doi.org/10.1002/mds.23709
Martino D, Brander G, Svenningsson P, Larsson H, de la Cruz LF (2020) Association and Familial Coaggregation of Idiopathic Dystonia with Psychiatric Outcomes. Mov Disord. https://doi.org/10.1002/mds.28257
Junker J, Berman BD, Hall J, Wahba DW, Brandt V, Perlmutter JS, Jankovic J, Malaty IA, Wagle Shukla A, Reich SG, Espay AJ, Duque KR, Patel N, Roze E, Vidailhet M, Jinnah HA, Brüggemann N (2021) Quality of life in isolated dystonia: non-motor manifestations matter. J Neurol Neurosurg Psychiatry 92:622–628. https://doi.org/10.1136/jnnp-2020-325193
Ben-Shlomo Y, Camfield L, Warner T, group Ec, (2002) What are the determinants of quality of life in people with cervical dystonia? J Neurol Neurosurg Psychiatry 72:608–614
O’Connor S, Hevey D, O’Keeffe F (2023) Illness Perceptions, Coping, Health-Related Quality of Life and Psychological Outcomes in Cervical Dystonia. J Clin Psychol Med Settings 30:129–142. https://doi.org/10.1007/s10880-022-09851-2
Liang Y, Lin J, Hou Y, Zhang L, Ou R, Li C, Wei Q, Cao B, Liu K, Jiang Z, Yang T, Yang J, Zhang M, Kang S, Xiao Y, Jiang Q, Yang J, Song W, Chen X, Zhao B, Wu Y, Shang H (2022) Health-Related Quality of Life in Cervical Dystonia Using EQ-5D-5L: A Large Cross-Sectional Study in China. Front Neurol 13:895272. https://doi.org/10.3389/fneur.2022.895272
Yi M, Li J, Liu G, Ou Z, Liu Y, Li J, Chen Y, Guo Y, Wang Y, Zhang W, Zeng J, Dang C (2022) Mental health and quality of life in patients with craniofacial movement disorders: A cross-sectional study. Front Neurol 13:938632. https://doi.org/10.3389/fneur.2022.938632
Girach A, Vinagre Aragon A, Zis P (2018) Quality of life in idiopathic dystonia: a systematic review. J Neurol. https://doi.org/10.1007/s00415-018-9119-x
Rafee S, Al-Hinai M, Douglas G, Ndukwe I, Hutchinson M (2023) Mood symptoms in cervical dystonia: Relationship with motor symptoms and quality of life. Clin Park Relat Disord 8:100186. https://doi.org/10.1016/j.prdoa.2023.100186
Weiss D, Hieber L, Sturm J, Bortlein A, Mayr I, Appy M, Kuhnler B, Buchthal J, Dippon C, Arnold G, Wachter T (2017) Botulinumtoxin Improves both Generic and Disease-Specific Quality of Life in Cervical Dystonia. Front Neurol 8:561. https://doi.org/10.3389/fneur.2017.00561
Muller J, Kemmler G, Wissel J, Schneider A, Voller B, Grossmann J, Diez J, Homann N, Wenning GK, Schnider P, Poewe W, Austrian Botulinum T, Dystonia Study G (2002) The impact of blepharospasm and cervical dystonia on health-related quality of life and depression. J Neurol 249:842–846. https://doi.org/10.1007/s00415-002-0733-1
Mordin M, Masaquel C, Abbott C, Copley-Merriman C (2014) Factors affecting the health-related quality of life of patients with cervical dystonia and impact of treatment with abobotulinumtoxinA (Dysport): results from a randomised, double-blind, placebo-controlled study. BMJ Open 4:e005150. https://doi.org/10.1136/bmjopen-2014-005150
Hilker R, Schischniaschvili M, Ghaemi M, Jacobs A, Rudolf J (2001) Health related quality of life is improved by botulinum neurotoxin type A in long term treated patients with focal dystonia. J Neurol Neurosurg Psychiatry 71:193–199
Biuk D, Karin AA, Matic S, Barac J, Benasic T, Stiglmayer N (2013) Quality of life in patients with blepharospasm. Coll Antropol 37:29–33
Yoshida K (2022) Effects of Botulinum Toxin Therapy on Health-Related Quality of Life Evaluated by the Oromandibular Dystonia Rating Scale. Toxins 14 https://doi.org/10.3390/toxins14100656
Moriarty A, Rafee S, Ndukwe I, O’Riordan S, Hutchinson M (2022) Longitudinal Follow-Up of Mood in Cervical Dystonia and Influence on Age at Onset. Movement disorders clinical practice 9:614–618. https://doi.org/10.1002/mdc3.13457
McHorney CA, Ware JE Jr, Raczek AE (1993) The MOS 36-Item Short-Form Health Survey (SF-36): II. Psychometric and clinical tests of validity in measuring physical and mental health constructs. Med Care 31:247–263
Bjelland I, Dahl AA, Haug TT, Neckelmann D (2002) The validity of the Hospital Anxiety and Depression Scale. An updated literature review. J Psychosom Res 52:69–77
Rytwinski NK, Fresco DM, Heimberg RG, Coles ME, Liebowitz MR, Cissell S, Stein MB, Hofmann SG (2009) Screening for social anxiety disorder with the self-report version of the Liebowitz Social Anxiety Scale. Depress Anxiety 26:34–38. https://doi.org/10.1002/da.20503
Burke RE, Fahn S, Marsden CD, Bressman SB, Moskowitz C, Friedman J (1985) Validity and reliability of a rating scale for the primary torsion dystonias. Neurology 35:73–77
Muthén L, Muthén B (1998–2012; Seventh Edition) Mplus User’s Guide. Los Angeles, CA
Huck SWCW, Bounds WG (1974) 6th edition) Reading statistics and research. Harper and Row
Tomarken AJ, Waller NG (2003) Potential problems with “well fitting” models. J Abnorm Psychol 112:578–598. https://doi.org/10.1037/0021-843X.112.4.578
Pirio Richardson S, Wegele AR, Skipper B, Deligtisch A, Jinnah HA, Dystonia Coalition I (2017) Dystonia treatment: Patterns of medication use in an international cohort. Neurology 88:543–550. https://doi.org/10.1212/WNL.0000000000003596
Page D, Butler A, Jahanshahi M (2007) Quality of life in focal, segmental, and generalized dystonia. Mov Disord 22:341–347. https://doi.org/10.1002/mds.21234
Basurovic N, Svetel M, Pekmezovic T, Kostic VS (2012) Evaluation of the quality of life in patients with segmental dystonia. Vojnosanitetski pregled Military-medical and pharmaceutical review 69:759–764
Hutchinson M, McGovern EM, Narasimham S, Beck R, Reilly RB, Walsh CD, Malone KM, Tijssen MAJ, O’Riordan S (2018) The premotor syndrome of cervical dystonia: Disordered processing of salient environmental stimuli. Mov Disord 33:232–237. https://doi.org/10.1002/mds.27229
Treede RD (2002) Spinothalamic and thalamocortical nociceptive pathways. J Pain 3:109–112;discussion 113–104. https://doi.org/10.1054/jpai.2002.122951
Zhang L, Hou Y, Lin J, Yang J, Cao B, Wei Q, Ou R, Shang H (2022) Comprehensive analysis of non-motor symptoms and their association with quality of life in Writer’s cramp. Parkinsonism Relat Disord 100:37–40. https://doi.org/10.1016/j.parkreldis.2022.05.025
Quagliato EM, Carelli EF, Viana MA (2010) A prospective, randomized, double-blind study comparing the efficacy and safety of type a botulinum toxins botox and prosigne in the treatment of cervical dystonia. Clin Neuropharmacol 33:22–26. https://doi.org/10.1097/WNF.0b013e3181c46f48
Cano SJ, Warner TT, Linacre JM, Bhatia KP, Thompson AJ, Fitzpatrick R, Hobart JC (2004) Capturing the true burden of dystonia on patients: the Cervical Dystonia Impact Profile (CDIP-58). Neurology 63:1629–1633. https://doi.org/10.1212/01.wnl.0000142962.11881.26
Hays RD, Sherbourne CD, Mazel RM (1993) The RAND 36-Item Health Survey 1.0. Health Econ 2:217–227
Queiroz MA, Chien HF, Sekeff-Sallem FA, Barbosa ER (2012) Physical therapy program for cervical dystonia: a study of 20 cases. Funct Neurol 27:187–192
Zetterberg L, Halvorsen K, Farnstrand C, Aquilonius SM, Lindmark B (2008) Physiotherapy in cervical dystonia: six experimental single-case studies. Physiother Theory Pract 24:275–290. https://doi.org/10.1080/09593980701884816
Faircloth S, Reid S (2006) A cognitive−behavioural approach to the management of idiopathic cervical dystonia. J Behav Ther Exp Psychiatry 37:239–246. https://doi.org/10.1016/j.jbtep.2005.07.003
Wadon ME, MacIver C, Winter M, Peall KJ (2021) Internet-based cognitive behavioural therapy as a feasible treatment of adult-onset, focal, isolated, idiopathic cervical dystonia. Clin Park Relat Disord 5:100121. https://doi.org/10.1016/j.prdoa.2021.100121
Loudovici-Krug D, Derlien S, Best N, Gunther A (2022) Physiotherapy for Cervical Dystonia: A Systematic Review of Randomised Controlled Trials. Toxins 14:784. https://doi.org/10.3390/toxins14110784
Costanzo M, Belvisi D, Berardelli I, Maraone A, Baione V, Ferrazzano G, Cutrona C, Leodori G, Pasquini M, Conte A, Fabbrini G, Defazio G, Berardelli A (2021) Effect of Botulinum Toxin on Non-Motor Symptoms in Cervical Dystonia. Toxins 13 https://doi.org/10.3390/toxins13090647
Acknowledgements
We would like to thank Laura J Wright (MA) and Susanna Langeloh (MA) for their assistance with accessing the Dystonia Coalition database as well as the patients for contributing to the study.
Funding
Open Access funding enabled and organized by Projekt DEAL. This work was supported in part by grants to the Dystonia Coalition, a consortium of the Rare Diseases Clinical Research Network (RDCRN) that is supported by the Office of Rare Diseases Research (ORDR) at the National Center for Advancing Clinical and Translational Studies (NCATS; U54 TR001456) in collaboration with the National Institute for Neurological Diseases and Stroke (NINDS; U54 NS065701 and U54 NS116025).
Author information
Authors and Affiliations
Consortia
Contributions
Johanna Junker, acquisition of data, study concept and design, statistical analysis, analysis and interpretation of data, drafting of manuscript. James Hall, statistical analysis, critical revision of manuscript for intellectual content. Brian D. Berman, acquisition of data, critical revision of manuscript for intellectual content. Marie Vidailhet, acquisition of data, critical revision of manuscript for intellectual content. Emmanuel Roze, acquisition of data, critical revision of manuscript for intellectual content. Tobias Bäumer, acquisition of data, critical revision of manuscript for intellectual content. Irene A Malaty, acquisition of data, critical revision of manuscript for intellectual content. Aparna Wagle Shukla, acquisition of data, critical revision of manuscript for intellectual content. Joseph Jankovic, acquisition of data, critical revision of manuscript for intellectual content. Stephen G. Reich, acquisition of data, critical revision of manuscript for intellectual content. Alberto J. Espay, acquisition of data, critical revision of manuscript for intellectual content. Kevin R. Duque, acquisition of data, critical revision of manuscript for intellectual content. Neepa Patel, acquisition of data, critical revision of manuscript for intellectual content. Joel S. Perlmutter, acquisition of data, critical revision of manuscript for intellectual content. H. A. Jinnah, acquisition of data, critical revision of manuscript for intellectual content. Valerie Brandt, statistical analysis, drafting of manuscript, critical revision of manuscript for intellectual content. Norbert Brüggemann, study concept and design, analysis and interpretation of data, study supervision, critical revision of manuscript for intellectual content.
Statistical Analysis conducted by James Hall, DPhil, and Valerie Brandt, PhD, University of Southampton.
Corresponding author
Ethics declarations
Conflict of interest
Johanna Junker is supported by the Michael J. Fox Foundation and received a Family Mobility Grant from the University of Luebeck. James Hall has received grant support from: Oxford University Press; The Australian Research Council; The Centre for British Teachers Education Trust; & The UK Government (Department for Education; UK Research and Innovation). Has received travel compensation and/or honoraria from: Oxford Risk; Oxfordshire Educational Psychology Service; The Moscow School of Social and Economic Sciences; The University of Macau; Westminster Education Forum. He is an advisor to the UK Government Department Ofsted via membership of their Early Education Curriculum Forum. Brian D. Berman has received research grant support from the Dystonia Coalition (receives the majority of its support through NIH grant NS065701 from the Office of Rare Diseases Research in the National Center for Advancing Translational Science and National Institute of Neurological Disorders and Stroke), the Parkinson’s Foundation, the VCU School of Medicine, the Administration for Community Living, and the Dystonia Medical Research Foundation. He has received honoraria from the International Parkinson and Movement Disorder Society and serves on the Medical and Scientific Advisory Council of the Dystonia Medical Research Foundation as well as the Medical Advisory Board of the Benign Essential Blepharospasm Research Foundation and the National Spasmodic Torticollis Association. Marie Vidailhet: The author declares that she has no conflict of interest. Emmanuel Roze received honorarium for speech from Orkyn, Aguettant, Elivie and for participating in advisory boards from Merz-Pharma, Ipsen, Teva, and BIAL. He received research support from Merz-Pharma, Orkyn, Aguettant, Elivie, Ipsen, Everpharma, Enjoysharing, Fondation Desmarest, AMADYS, ADCY5.org, Fonds de dotation Patrick Brou de Laurière, Agence Nationale de la Recherche, Dystonia Medical Research Foundation. Tobias Bäumer has received funding from the German Research Foundation (DFG, BA 6375/2–1). He received speaker and consultant fees from Pelzerhaken Children's Centre, Allergan/Abbvie, Ipsen Pharma and Merz Pharmaceuticals. He has received research funding from: Allergan/Abbvie, Ipsen Pharma and Merz Therapeutical. He was supported with exhibition ultrasound equipment on loan from Cannon and ESAOTE. Irene A. Malaty has participated in research funded by the Parkinson Foundation, Tourette Association, Dystonia Coalition, AbbVie, Emalex, Neuroderm, Revance, and Sage but has no owner interest in any pharmaceutical company. She has received consulting fees from Abbvie. She has received travel compensation or honoraria from the Tourette Association of America, Parkinson Foundation and Cleveland Clinic, and royalties from Robert Rose publishers. Aparna Wagle Shukla reports grant support from the NIH R01NS122943 as PI and Ro1 NS121120-01 as a Co-I. She reports past funding from Benign Essential Blepharospasm Research foundation, Dystonia coalition, Dystonia Medical Research foundation, National Organization for Rare Disorders. AWS has received consultant fees from Merz, Jazz and Acadia. She is the current Vice President for the Tremor Research Group and recent advisor for Supernus and Biogen-Sage. Joseph Jankovic has received research/training funding from: AbbVie Inc; Acadia Pharmaceuticals; Allergan, Inc; Biotek; Cerevel Therapeutics; CHDI Foundation; Dystonia Coalition; Emalex Biosciences, Inc; F. Hoffmann-La Roche Ltd; Huntington Study Group; Medtronic Neuromodulation; Merz Pharmaceuticals; Michael J Fox Foundation for Parkinson Research; National Institutes of Health; Neuraly, Inc.; Neurocrine Biosciences; Parkinson’s Foundation; Parkinson Study Group; Prilenia Therapeutics; Revance Therapeutics, Inc; Teva Pharmaceutical Industries Ltd. Stephen G. Reich: NINDS: Data Safety Monitoring Committee; Book Royalties: Springer, Informa, Oxford; Consultant: Best Doctors; Reviewer: UpToDate. Alberto J. Espay has received grant support from the NIH and the Michael J Fox Foundation; personal compensation as a consultant/scientific advisory board member for Neuroderm, Amneal, Acadia, Avion Pharmaceuticals, Acorda, Kyowa Kirin, Sunovion, Supernus (formerly, USWorldMeds), and Herantis Pharma; personal honoraria for speakership for Avion, Amneal, and Supernus; and publishing royalties from Lippincott Williams & Wilkins, Cambridge University Press, and Springer. He cofounded REGAIN Therapeutics and is co-inventor of the patent “Compositions and methods for treatment and/or prophylaxis of proteinopathies.” Kevin R. Duque has received honoraria from Parkinson's Foundation for translation support. Neepa Patel has received honoraria as a consultant for Abbott Laboratories, Abbvie Pharmaceuticals and a speaker for Boston Scientific. Joel S. Perlmutter has received research funding from National Institutes of Health (NCATS, NINDS, NIA), Michael J Fox Foundation, Barnes-Jewish Hospital Foundation (Elliot Stein Family Fund and Parkinson disease research fund), American Parkinson Disease Association (APDA) Advanced Research Center at Washington University, Greater St. Louis Chapter of the APDA, Paula and Rodger Riney Fund, Jo Oertli Fund, the Murphy Fund, Huntington Disease Society of America, and CHDI. He has received honoraria from CHDI, Huntington Disease Study Group, Parkinson Study Group, Beth Israel Hospital (Harvard group), U Pennsylvania, Stanford U, St. Lukes Hospital, Boston University. He also is co-director for the Dystonia Coalition, which has received the majority of its support through the NIH (grants NS116025, NS065701 from the National Institutes of Neurological Disorders and Stroke TR 001456 from the Office of Rare Diseases Research at the National Center for Advancing Translational Sciences). Dr. Perlmutter serves as Director of Medical and Scientific Advisory Committee of the Dystonia Medical Research Foundation, Chair of the Professional Standards and Ethics Committee of the Huntington Study Group, member of the Scientific Advisory Board of the APDA, Chair of the Scientific and Publication Committee for ENROLL-HD, and member of the Education Committee of the Huntington Study Group. H. A. Jinnah has active or recent grant support from the US government (National Institutes of Health), private philanthropic organizations (Cure Dystonia Now), academically-oriented institutions (the Dystonia Study Group), and industry (Cavion Therapeutics, Ipsen Pharmaceuticals, Retrophin Inc.). Dr. Jinnah has also served on advisory boards or as a consultant for Allergan Inc., CoA Therapeutics, Cavion Therapeutics, and Retrophin Inc. He has received honoraria or stipends for lectures or administrative work from the American Academy of Neurology, the American Neurological Association, the Dystonia Medical Research Foundation, the International Neurotoxin Society, and the International Parkinson’s Disease and Movement Disorders Society. Dr. Jinnah serves on the Scientific Advisory Boards for several private foundations including the Benign Essential Blepharospasm Research Foundation, Cure Dystonia Now, the Dystonia Medical Research Foundation, the Tourette Association of America, and Tyler's Hope for a Cure. He also is principle investigator for the Dystonia Coalition, which has received the majority of its support through the NIH (grants NS116025, NS065701 from the National Institutes of Neurological Disorders and Stroke TR 001456 from the Office of Rare Diseases Research at the National Center for Advancing Translational Sciences). The Dystonia Coalition has received additional material or administrative support from industry sponsors (Allergan Inc. and Merz Pharmaceuticals) as well as private foundations (The American Dystonia Society, Beat Dystonia, The Benign Essential Blepharospasm Foundation, Cure Dystonia Now, Dystonia Europe, Dystonia Inc., Dystonia Ireland, The Dystonia Medical Research Foundation, The Foundation for Dystonia Research, The National Spasmodic Dysphonia Association, and The National Spasmodic Torticollis Association). Valerie Brandt receives funding from the Academy of Medical Sciences. Dr. Brüggemann received honaria from Abbott, Abbvie, Biogen, Biomarin, Bridgebio, Centogene, Esteve, Zambon. Dr. Brüggemann was supported by the DFG (BR4328.2–1, GRK1957), the Michael J Fox Foundation, the Collaborative Center for X-linked Dystonia-Parkinsonism and the EU Joint Programme—Neurodegenerative Disease Research (JPND).
Supplementary Information
Below is the link to the electronic supplementary material.
Online Resource 1.
Coinvestigators of the Dystonia Coalition Study Group, that had a major role in the acquisition of data 1 (PDF 115 kb)
Online Resource 2.
Direct effects of the cross-lagged path model (stylized illustration). Direct paths of the cross-lagged path model for the physical functioning, physical role functioning, pain, energy / fatigue, emotional well-being, emotional role functioning and the social functioning subscales of HR-QoL are presented. Dashed lines illustrate the effect of age and sex on the different variables. Bonferroni corrected alpha is ≤ 0.006. Significant paths are marked in red, grey paths did not reach significance level 2 (PDF 686 kb)
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Junker, J., Hall, J., Berman, B.D. et al. Longitudinal predictors of health-related quality of life in isolated dystonia. J Neurol 271, 852–863 (2024). https://doi.org/10.1007/s00415-023-12022-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-023-12022-4