Abstract
Introduction
Preserflo MicroShunt is a novel microinvasive bleb forming device for the treatment of primary open-angle glaucoma. The intermediate- and long-term success and the impact of this procedure on corneal endothelial cell density remain to be investigated.
Methods
In this prospective observational study, 62 eyes of 55 glaucoma patients (mean age ± SD: 67.0 ± 15.0 years) receiving a Preserflo MicroShunt were included. Corneal endothelial cell density, intraocular pressure and best corrected visual acuity were assessed preoperatively and at 3, 6, 9, 12, 18 and 24 months postoperatively. Success rates, bleb revision rates and complications were analysed. Complete success was defined as an intraocular pressure reduction of ≥ 20% and achieving a target pressure of ≤ 18, ≤ 15 or ≤ 12 mmHg without antiglaucoma medication. Qualified success indicated that the criteria were reached with or without medication.
Results
Corneal endothelial cells showed no significant decline over 24 months (p > 0.05). Intraocular pressure showed a substantial reduction postoperatively (p < 0.001), decreasing from 29.6 ± 8,3 mmHg to 13.0 ± 4.3 mmHg after 24 months (p < 0.001). Complete and qualified success with a target pressure ≤ 15 mmHg was achieved in 52.9% and 54.6% of cases after 24 months, respectively. Best corrected visual acuity did not change after 24 months.
Conclusion
Preserflo MicroShunt had no negative side effects on corneal endothelial cells and showed favourable success rates after 2 years in patients with open-angle glaucoma.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.

Purpose
Intraocular pressure (IOP) is the only modifiable risk factor of glaucoma progression [1]. When medical treatment with IOP-lowering eye drops is either insufficient or not possible due to adherence issues, surgery is necessary to prevent progressive visual field defects. Microinvasive bleb surgery with subconjunctival devices such as the XEN Gel Stent (AbbVie Inc., North Chicago, Illinois, USA) and the Preserflo MicroShunt (PMS) (Santen Pharmaceutical Co., Osaka, Japan) have recently gained popularity due to a better risk profile than trabeculectomy [2]. However, there are concerns regarding the impacts of the various stents on the corneal endothelium, due to the experience with the supraciliary CyPass Micro-Stent. This device was withdrawn from the market due to increased corneal endothelial cell (CEC) loss 5 years after implantation [3]. As a result, clinicians are showing considerable interest in determining the effectiveness and safety profile of stent-related filtering surgery, especially regarding CECs.
The PMS obtained its CE marking in 2012 [4] and received FDA approval in July 2020 [5]. The stent is made of a highly biocompatible and bioinert synthetic material called poly(styrene-block-isobutylene-block-styrene), abbreviated as SIBS. Measuring 8.5 mm in length, it has an outer diameter of 350 µm and an inner lumen diameter of 70 µm [6]. The device is implanted ab externo, creating a subconjunctival filtering bleb. Similar to other filtration surgeries, mitomycin C (MMC) is used intraoperatively to modulate the postoperative wound healing. The aim of our study was to investigate the intermediate-term efficacy and safety of the PMS in patients with open-angle glaucoma over a follow-up period of 2 years, with a focus on its potential effects on CECs.
Methods
This was a prospective, observational, nonrandomized study of patients who underwent PMS implantation. The study was conducted at a single tertiary centre. All surgeries were performed by a single, experienced surgeon (BV). None of the first 50 patients receiving the PMS at our centre were included in this study in order to reduce bias due to learning curve effects.
Inclusion criteria comprised the diagnosis of primary open-angle glaucoma, pigmentary glaucoma or pseudoexfoliative glaucoma, with above-target IOP despite the maximum tolerated IOP-lowering medication, and a minimum follow-up period of 12 months. Both pseudophakic and phakic patients were included, and all procedures were performed as solo operations without additional phacoemulsification.
Exclusion criteria were the diagnosis of narrow-angle glaucoma or normal tension glaucoma and a history of corneal diseases or corneal transplantation.
The study adhered to the principles of the Declaration of Helsinki of 1964. Prior to surgery, all patients provided informed consent, along with additional written consent for study participation. Ethical approval was granted by the local institutional ethics committee of the University of Tübingen (project number: 037/2021BO2).
Outcome measures
The primary outcome of the study was the mean change in IOP after 2 years. Secondary outcomes included change in CEC density, number of IOP-lowering medications, complete success rate, needling rate and complication rate after 2 years. Complete success was defined as an IOP reduction ≥ 20% from baseline and achieving a respective target IOP of ≤ 18 mmHg, ≤ 15 mmHg or ≤ 12 mmHg without IOP-lowering medications. If this IOP target was reached with or without IOP-lowering medication, the success was labelled as qualified. Hypotony was defined as IOP < 6 mmHg. Any further glaucoma surgery, except needling and incisional bleb revision, and the loss of light perception were defined as failure. Each active agent in combinations of IOP-lowering medications was counted as an individual treatment.
For an additional statistical analysis of success and bleb revision rates, we divided the study cohort into two groups: patients with previous glaucoma surgery (“previous surgery”, including prior trabeculectomy, XEN gel stent, canaloplasty, deep sclerectomy and trabeculotomy) and patients without previous surgery (“no previous surgery”). Patients who received only selective laser trabeculoplasty or diode laser cyclophotocoagulation prior to PMS implantation were also categorized as “no previous surgery”.
Surgical technique
The surgical procedure was performed under parabulbar anaesthesia. A traction suture was placed through the superior peripheral cornea. The conjunctiva was incised using two radial incisions, followed by blunt dissection of the subconjunctival space. Two round sponges soaked with 0.2 mg/mL MMC were placed under the conjunctiva for 3 min. Then, the sponges were removed, and the area was rinsed with balanced salt solution. A 1 mm-wide tunnel incision was made 3.5 mm from the limbus, and the PMS was introduced into the anterior chamber. After confirming the presence of fistulation through the PMS, the conjunctiva was closed with 10–0 nylon sutures. Dexamethasone and mezlocillin were injected subconjunctivally at the end of the procedure.
Pre- and postoperative management
IOP-lowering medication was stopped 2 weeks prior to surgery. Oral acetazolamide (250 mg) was administered twice daily until the night before surgery. Starting 1 week before surgery, unpreserved dexamethasone eye drops were instilled three times daily. No medication was administered on the day of surgery.
Postoperatively, moxifloxacin eye drops were initiated four times daily for 1 week beginning on the first postoperative day. Additionally, unpreserved dexamethasone eye drops were gradually tapered over a period of 7 weeks, starting with five drops per day on the first postoperative day.
Patients were examined on the first 2 postoperative days. Further follow-up visits were scheduled after 2 weeks and after 1, 3, 6, 12, 18 and 24 months.
In cases of inadequate IOP control or upon detecting clinical signs of bleb scarring, either a needling procedure or an incisional bleb revision was attempted. Needling was performed under topical anaesthesia using a needle to gently disrupt fibrotic tissue covering the PMS. A total of 25 µL of 0.2 mg/mL MMC (5 µg) was injected into the filtering bleb. Incisional bleb surgery was performed when the PMS was not visible subconjunctivally at the slit lamp. In these cases, the conjunctiva was incised, and the fibrotic tissue surrounding the PMS was dissected, and MMC was applied via soaked sponges. Moxifloxacin eye drops were administered four times daily for 3 days, and unpreserved dexamethasone eye drops were gradually tapered over a 5-week period after both needling and incisional bleb surgery.
Statistical analysis
The statistical analysis was performed using JMP 16.0 statistical software (SAS Institute Inc., Cary, NC, USA). Data were presented as frequency (percentage), mean (standard deviation [SD]), mean (standard error of the mean [SEM]), mean (95% confidence interval [95% CI]) or median (range), as appropriate. A linear mixed-effects model was used to analyse the effect of PMS implantation on best corrected visual acuity (BCVA), IOP and endothelial cell density (ECD). Dunnett’s test was used to compare pre- and postoperative BCVA, IOP and ECD. Changes in the number of IOP-lowering medications were assessed with the Wilcoxon signed-rank test. Kaplan–Meier analysis was used to calculate rates of complete and qualified success. A p-value of < 0.05 was considered to reflect a significant difference.
Results
Characteristics of the study population
Sixty-two eyes of 55 patients were included in the study. Patients’ baseline characteristics are shown in Table 1. The mean (± SD) age was 67 (± 15) years, and 54.8% of the studied eyes were from female patients. Thirty-eight eyes (61.3%) were pseudophakic and 24 eyes (38.7%) were phakic at the time of PMS implantation. The majority of eyes (60.3%) underwent no previous glaucoma surgery or laser treatment.
Development of IOP control, BCVA and medication
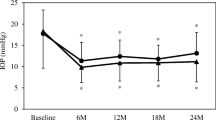
The mean (± SD) IOP decreased significantly from 29.6 (± 8.3) mmHg at baseline to 14.3 (± 5.3) mmHg after 12 months and to 13.0 (± 4.3) mmHg after 24 months (p < 0.001).
We observed average IOP reductions of 48.5% and 54.8% after 12 and 24 months, respectively. A comparison of pre- versus postoperative IOP values after 12 and 24 months is shown in Fig. 1.
BCVA declined significantly from 0.3 (± 0.3) logMAR at baseline to 0.5 (± 0.4) logMAR at day 1 and to 0.4 (± 0.4) logMAR at day 14 postoperatively (p < 0.001) but did not differ significantly from preoperative BCVA at any additional follow-up time points (p > 0.05). Twelve months after PMS implantation, BCVA was 0.2 (± 0.3) logMAR (p > 0.05), and it did not change significantly after 24 months (0.3 ± 0.3 logMAR; p > 0.05). The mean and SD BCVA at each follow-up time point are shown in Fig. 2.
The mean (± SD) number of IOP-lowering medications was reduced significantly from 3.2 (± 0.9) at baseline to 0.2 (± 0.7) after 12 months and to 0.2 (± 0.8) after 24 months (p < 0.001).
Endothelial cell density
Mean ECD did not change significantly from baseline to any postoperative follow-up time point (p > 0.05). At baseline, mean (± SD) ECD was 2240.8 ± 363.4 cells/mm2. ECD was 2175.6 ± 443.7 cells/mm2 (p > 0.05) and 2336.5 ± 388.6 cells/mm2 after 12 and 24 months, respectively. The mean ECD at all time points is shown in Fig. 3.
Success rates
The Kaplan–Meier analysis showed a complete success rate of 58.1% with a target IOP level of ≤ 15 mmHg after 12 months and 52.9% after 24 months. Qualified success with a target IOP level of ≤ 15 mmHg was achieved in 59.7% of eyes after 12 months and 54.6% after 24 months.
The Kaplan–Meier curves for each target IOP level are shown in Fig. 4.
In the additional analysis of the two subgroups "previous surgery" (n = 13) and "no previous surgery" (n = 49), the success rates tended to be higher for the "no previous surgery" group but without statistical significance (p > 0.05). In the "no previous surgery" group, 61.2% and 55.1% achieved complete success with IOP ≤ 15 mmHg at 12 and 24 months. Qualified success was achieved in 63.3% and 57.3% at 12 and 24 months. In contrast, in the "previous surgery" group, complete and qualified success with IOP ≤ 15 mmHg was achieved in 46.2% at both 12 and 24 months (p > 0.05).
The Kaplan–Meier curves for the two subgroups are shown in Fig. 5.
Kaplan–Meier survival plots illustrating the probability of success after PMS implantation for eyes with “previous surgery” and eyes with “no previous surgery”. a, c and e display the probabilities of “complete success” which was defined as an IOP reduction of ≥ 20% and achievement of the respective target IOP without IOP-lowering medication. b, d and f display the probabilities of “qualified success” with the respective target IOP. The survival probabilities with a target IOP ≤ 18 mmHg, ≤ 15 mmHg and ≤ 12 mmHg are shown. Differences were not statistically significant (p > 0.05)
Needling and incisional bleb revision
A needling procedure was performed at least once in 24 (38.7%) of the studied eyes. An incisional bleb revision was necessary in 16 (25.8%) of the eyes.
A primary incisional bleb revision without previous needling was performed in seven eyes (11.3% of all studied eyes). Of the 24 patients who had at least one needling, nine (14.5% of all studied eyes, 37.5% of eyes with needling) received an incisional bleb revision during the follow-up period. The average time between first needling and subsequent incisional bleb revision was 3.2 ± 3.0 months. In contrast, three eyes first received an incisional bleb revision and then a needling during the postoperative period, with an intervening interval of 2.0 ± 1.3 months. Thirty-four eyes (54.8%) did not require any bleb intervention after PMS implantation. The mean (± SD) numbers of needlings and incisional bleb revisions performed per eye were 0.6 ± 0.9 and 0.3 ± 0.6, respectively. The mean time to the first needling was 9.1 ± 7.0 months, and the mean time to first incisional bleb revision was 10.7 ± 6.8 months.
In the group of eyes with "previous surgery", the mean (± SD) numbers of needlings and incisional bleb revisions performed per eye were and 0.8 ± 0.9 and 0.4 ± 0.9, respectively, while in the "no previous surgery" group the mean (± SD) numbers were 0.5 ± 0.9 and 0.3 ± 0.6, respectively.
Complications
Six eyes (9.7%) showed a hypotony with choroidal detachment and a flat anterior chamber within the first 4 postoperative weeks. In five of those, the anterior chamber was filled with a high viscosity viscoelastic substance after a mean of 14.8 ± 8.2 days postoperatively. In three of the eyes, this was repeated after a mean of 22 ± 7.2 days after PMS implantation. This approach resulted in complete recovery of choroidal detachment and the flat anterior chamber in all eyes. One eye recovered spontaneously after 14 days. We did not observe any cases of persistent hypotony. No sight-threatening complications were observed throughout the follow-up period. None of the eyes required additional glaucoma surgery or laser treatment.
Discussion
The results of this study showed that PMS implantation achieved substantial IOP reductions of 48.5% and 54.8% after 12 and 24 months, respectively. Our study demonstrated that two thirds of the eyes retained an IOP level of ≤ 18 mmHg without any IOP-lowering medication after 24 months. These findings are consistent with those of previous studies demonstrating an effective decrease in IOP and the number of IOP-lowering medication after PMS implantation [7,8,9]. Fea et al. [8] observed complete and qualified success, with an IOP reduction of ≥ 20% and a target IOP ≤ 18 mmHg, in 26.0% and 58.7% of cases after 12 and 24 months, respectively, in a retrospective multicentre study of primary open-angle and pseudoexfoliative glaucoma. With a target IOP of ≤ 18 mmHg, our data show higher complete and qualified success rates of 74.2% and 75.8%, respectively, after 12 months. Compared to our results, Ibarz Barberá et al. [7] found a slightly lower mean IOP reduction of 38.7% at the final follow-up time point, with a mean follow-up period of 11 months. The authors defined complete and qualified success as reaching an IOP level of 6–17 mmHg and a minimum IOP reduction of 20% without or with medication, respectively, and observed complete and qualified success in 70.3% and 12.5% of patients, respectively, at the final visit [7]. Although the data are not directly comparable due to different success criteria and follow-up periods, the complete success rate appears to be similar to that in our study, as we observed a 74.2% complete success rate of IOP ≤ 18 mmHg after 12 months.
Interestingly, our sub-analysis showed that eyes with previous surgery tended to have lower success rates than those without previous surgery, even though the difference was not statistically significant. This may be due to pre-existing conjunctival scarring in the pre-operated eyes. It is also conceivable that these are patients who generally have a greater tendency to scar and have therefore already had failed glaucoma surgery in the past. The same reasons may also be responsible for the comparatively higher needling rate in eyes with previous surgery. The impact of previous surgery on the success rate of PMS deserves further investigation in a larger cohort.
Further aims of this study were to assess the safety and, specifically, the potential impact of PMS implantation on CECs. The complications observed in this study were manageable and had no long-term sequelae. Notably, ECD did not change significantly after 24 months.
The potential decline of ECD is one of the major concerns regarding the safety of glaucoma implants and drainage devices. Knowledge of the specific risks of the devices is crucial for the selection of the most favourable surgical procedure and for adequate patient counselling and education. It is worth noting that previous studies have reported concerns about endothelial cell loss (ECL) associated with other drainage devices, such as the CyPass Micro-Stent and the Ahmed glaucoma valve. In 2019, significant ECL was demonstrated up to 5 years after implantation of the CyPass Micro-Stent [3, 10]. This led to the withdrawal of the device from the market [3]. Significant decreases of CECs by 15.3% at 12 months and 18.6% at 24 months were also reported for the Ahmed glaucoma valve [11]. Similarly, the Ahmed glaucoma valve was independently associated with a significant ECL in children with uveitic glaucoma [12].
Ibarz Barberá et al. [13] reported that PMS implantation was associated with an ECL of 7.4% 12 months after surgery. ECL began immediately after surgery and continued over time, albeit at a slower rate after the first year. Position of the PMS, anterior chamber depth, hypotony and the presence of peripheral anterior synechia were factors influencing ECD changes. This study demonstrated the importance of proper positioning of the PMS. The proximity of the PMS to the corneal endothelium affected the ECL, with PMS implants located farther from the endothelium showing lower rates of ECL [13]. Similarly, the proximity of the tube was a major risk factor for ECL in patients with a Baerveldt drainage device or an Ahmed glaucoma valve [14, 15]. Baker et al. [16] reported mean rates of ECL of 5.2% and 6.9% 1 year after PMS implantation or trabeculectomy, respectively. Chamard et al. [5] reported significant ECL 5 years after PMS implantation in two cases. In one of these cases, anterior segment OCT and ultrasound biomicroscopy indicated a probable backward movement and a short intracameral portion of the device causing a corneal touch. In the second case, the authors assumed an inflammatory reaction. This case series indicates that ECL might be a slowly progressive process that takes years to become evident.
In contrast, we did not observe any significant ECL over the 24-month follow-up period of our study, which supports the intermediate-term safety profile of the PMS. A possible explanation for this discrepancy to the reported ECL in other studies is that at the time of the start of this study the learning curve of the surgeon was already completed, so that the risk of an ECL due to improper placement of the stent too close to the endothelium was minimized. On the other hand, no anterior segment OCT was performed in this study, so that no precise statement can be made about possible differences in the placement of the PMS in the anterior chamber. Further studies with anterior segment OCT and long-term observation and would be of great interest.
Similarly, Qidwai et al. [17] found no significant ECL over a 24-month study period; however, endothelial cell counts were available for only 12 of 48 patients. Furthermore, studies by Jamke et al. [18] and by Steindor et al. [19] found no significant ECL 12 months after PMS implantation. Notably, however, the current study is the only one investigating the impact of the PMS on the corneal endothelium with a follow-up period of at least 2 years.
Regarding pre- and postoperative complications, we did not observe any cases of permanent hypotony in our study. Filtering stent-based procedures are reported to have lower rates of long-term hypotony than trabeculectomy. A retrospective cohort study by Van Lancker et al. [20] reported no cases of persistent hypotony after PMS, in contrast to trabeculectomy. Chronic hypotony was the main reason for failure in the trabeculectomy patients in this study [20]. Several other studies showed a higher rate of prolonged hypotony with trabeculectomy than with PMS [21]. This is explained by the flow-limiting design of the PMS based on the Hagen–Poiseuille equation, which is intended to prevent permanent hypotony [22].
We observed that BCVA remained stable, without a significant reduction over the study period. This finding is reassuring, as it indicates that PMS implantation does not compromise visual acuity, which is crucial for maintaining the quality of life of glaucoma patients. Several studies found no significant alterations in BCVA after PMS implantation [23, 24]. In contrast, a cross-sectional, multicentre, retrospective study found a visual loss of > 2 Snellen lines in 5.6% of 428 studied eyes after trabeculectomy [25]. Another study reported 8.0% permanent vision loss after trabeculectomy in a large retrospective study [26].
We acknowledge that the single-centre design and some missing follow-up visits might introduce bias despite meticulous planning of this prospective study. In addition, the follow-up period of 24 months might have been too short to detect a significant rate of ECL. Consequently, we plan to prolong the study to gain a more comprehensive understanding of the safety and efficacy of the PMS.
Conclusion
Our study provides valuable insights into the safety and efficacy of the PMS in the treatment of medically uncontrolled open-angle glaucoma. Notably, ECD did not change significantly after 24 months. We found that 66.6% of eyes had an IOP of ≤ 18 mmHg and were free from IOP-lowering medication after 24 months. The complications were manageable or self-limiting and had no long-term sequelae.
References
Coleman AL, Kodjebacheva G (2009) Risk factors for glaucoma needing more attention. Open ophthalmol J 3:38–42. https://doi.org/10.2174/1874364100903020038
Gedde SJ, Feuer WJ, Lim KS, Barton K, Goyal S, Ahmed II, Brandt JD, Group PTVTS (2020) Treatment outcomes in the primary tube versus trabeculectomy study after 3 years of follow-up. Ophthalmology 127:333–345
Reiss G, Clifford B, Vold S, He J, Hamilton C, Dickerson J, Lane S (2019) Safety and effectiveness of CyPass supraciliary micro-stent in primary open-angle glaucoma: 5-year results from the COMPASS XT study. Am J Ophthalmol 208:219–225. https://doi.org/10.1016/j.ajo.2019.07.015
Gambini G, Carlà MM, Giannuzzi F, Caporossi T, De Vico U, Savastano A, Baldascino A, Rizzo C, Kilian R, Caporossi A, Rizzo S (2022) PreserFlo(®) MicroShunt: an overview of this minimally invasive device for open-angle glaucoma. Vision (Basel) 6. https://doi.org/10.3390/vision6010012
Chamard C, Hammoud S, Bluwol E, Lachkar Y (2022) Endothelial cell loss 5 years after Preserflo MicroShunt implantation: about two cases. Am J Ophthalmol Case Rep 25:101238
Pinchuk L, Riss I, Batlle JF, Kato YP, Martin JB, Arrieta E, Palmberg P, Parrish RK, Weber BA, Kwon Y (2017) The development of a micro-shunt made from poly (styrene-block-isobutylene-block-styrene) to treat glaucoma. J Biomed Mater Res B Appl Biomater 105:211–221
IbarzBarberá M, Martínez-Galdón F, Caballero-Magro E, Rodríguez-Piñero M, Tañá-Rivero P (2022) Efficacy and safety of the Preserflo Microshunt with mitomycin c for the treatment of open angle glaucoma. J Glaucoma 31:557–566. https://doi.org/10.1097/ijg.0000000000002052
Fea AM, Laffi GL, Martini E, Economou MA, Caselgrandi P, Sacchi M, Au L (2022) Effectiveness of MicroShunt in patients with primary open-angle and pseudoexfoliative glaucoma: a retrospective European multicenter study. Ophthalmol Glaucoma 5:210–218. https://doi.org/10.1016/j.ogla.2021.08.005
Batlle JF, Fantes F, Riss I, Pinchuk L, Alburquerque R, Kato YP, Arrieta E, Peralta AC, Palmberg P, Parrish RK (2016) Three-year follow-up of a novel aqueous humor microshunt. J Glaucoma 25:e58–e65
Lass JH, Benetz BA, He J, Hamilton C, Von Tress M, Dickerson J, Lane S (2019) corneal endothelial cell loss and morphometric changes 5 years after phacoemulsification with or without CyPass micro-stent. Am J Ophthalmol 208:211–218. https://doi.org/10.1016/j.ajo.2019.07.016
Lee E-K, Yun Y-J, Lee J-E, Yim J-H, Kim C-S (2009) Changes in corneal endothelial cells after Ahmed glaucoma valve implantation: 2-year follow-up. Am J Ophthalmol 148:361–367
KalininaAyuso V, Scheerlinck LM, de Boer JH (2013) The effect of an Ahmed glaucoma valve implant on corneal endothelial cell density in children with glaucoma secondary to uveitis. Am J Ophthalmol 155:530–535. https://doi.org/10.1016/j.ajo.2012.09.001
Ibarz-Barberá M, Morales-Fernández L, Corroto-Cuadrado A, Martinez-Galdón F, Tañá-Rivero P, Gómez de Liaño R, Teus MA (2022) Corneal endothelial cell loss after PRESERFLO™ MicroShunt implantation in the anterior chamber: anterior segment OCT tube location as a risk factor. Ophthalmol Ther 11:293–310. https://doi.org/10.1007/s40123-021-00428-0
Zhang Q, Liu Y, Thanapaisal S, Oatts J, Luo Y, Ying GS, Wang J, McLeod SD, Gedde SJ, Han Y (2021) The effect of tube location on corneal endothelial cells in patients with ahmed glaucoma valve. Ophthalmology 128:218–226. https://doi.org/10.1016/j.ophtha.2020.06.050
Tan AN, Webers CA, Berendschot TT, de Brabander J, de Witte PM, Nuijts RM, Schouten JS, Beckers HJ (2017) Corneal endothelial cell loss after Baerveldt glaucoma drainage device implantation in the anterior chamber. Acta Ophthalmol 95:91–96. https://doi.org/10.1111/aos.13161
Baker ND, Barnebey HS, Moster MR, Stiles MC, Vold SD, Khatana AK, Flowers BE, Grover DS, Strouthidis NG, Panarelli JF (2021) Ab-Externo MicroShunt versus trabeculectomy in primary open-angle glaucoma: one-year results from a 2-year randomized, multicenter study. Ophthalmology 128:1710–1721. https://doi.org/10.1016/j.ophtha.2021.05.023
Qidwai U, Jones L, Ratnarajan G (2022) A comparison of iStent combined with phacoemulsification and endocyclophotocoagulation (ICE2) with the PreserFlo MicroShunt and XEN-45 implants. Ther Adv Ophthalmol 14:25158414221125696. https://doi.org/10.1177/25158414221125697
Jamke M, Herber R, Haase MA, Jasper CS, Pillunat LE, Pillunat KR (2023) PRESERFLO ™ MicroShunt versus trabeculectomy: 1-year results on efficacy and safety. Graefes Arch Clin Exp Ophthalmol: 1–15. https://doi.org/10.1007/s00417-023-06075-4
Anton Steindor F, Trigaux C, Holtmann C, Spaniol K, Geerling G, Borrelli M (2023) Preserflo MicroShunt: efficacy and endothelial cell density. J Glaucoma 32:1018–1021. https://doi.org/10.1097/ijg.0000000000002325
Van Lancker L, Saravanan A, Abu-Bakra M, Reid K, Quijano C, Goyal S, Rodrigues I, Lascaratos G, Trikha S, Barwood C, Combe E, Kulkarni A, Lim KS, Low S (2023) Clinical outcomes and cost analysis of PreserFlo versus trabeculectomy for glaucoma management in the United Kingdom. Ophthalmol Glaucoma 6:342–357. https://doi.org/10.1016/j.ogla.2022.11.006
Pillunat KR, Herber R, Haase MA, Jamke M, Jasper CS, Pillunat LE (2022) PRESERFLO™ MicroShunt versus trabeculectomy: first results on efficacy and safety. Acta Ophthalmol 100:e779–e790. https://doi.org/10.1111/aos.14968
IbarzBarberá M, Hernández-Verdejo JL, Bragard J, Burguete J, Fernández LM, Rivero PT, de Liaño RG, Teus MA (2021) Evaluation of the ultrastructural and in vitro flow properties of the PRESERFLO MicroShunt. Transl Vis Sci Technol 10:26. https://doi.org/10.1167/tvst.10.13.26
Bhayani R, Martínez de la Casa JM, Figus M, Klabe K, Rabiolo A, Mercieca K (2023) Short-term safety and efficacy of Preserflo™ Microshunt in glaucoma patients: a multicentre retrospective cohort study. Eye 37:644–649
Quaranta L, Micheletti E, Carassa R, Bruttini C, Fausto R, Katsanos A, Riva I (2021) Efficacy and safety of PreserFlo® MicroShunt after a failed trabeculectomy in eyes with primary open-angle glaucoma: a retrospective study. Adv Ther 38:4403–4412
Kirwan JF, Lockwood AJ, Shah P, Macleod A, Broadway DC, King AJ, McNaught AI, Agrawal P (2013) Trabeculectomy in the 21st century: a multicenter analysis. Ophthalmology 120:2532–2539. https://doi.org/10.1016/j.ophtha.2013.07.049
Francis BA, Hong B, Winarko J, Kawji S, Dustin L, Chopra V (2011) Vision loss and recovery after trabeculectomy: risk and associated risk factors. Arch Ophthalmol 129:1011–1017
Acknowledgements
CJG was supported by the Junior TÜFF program of the Medical Faculty, Eberhard Karls University of Tübingen (application number: 2723- 0-0).
Funding
Open Access funding enabled and organized by Projekt DEAL. No funding was received for this research.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The study adhered to the principles of the Declaration of Helsinki of 1964. Prior to surgery, all patients provided informed consent, along with additional written consent for study participation. Ethical approval was granted by the local institutional ethics committee of the University of Tübingen (project number: 037/2021BO2).
Conflict of interest
CG, DW, EN and TS certify that they have no affiliations with or involvement in any organization or entity with any financial interest, or non-financial interest in the subject matter or materials discussed in this manuscript. BV has received speaker honoraria and advisory board compensation fees from AbbVie Deutschland GmbH, Novartis and Santen GmbH.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Gassel, C.J., Wenzel, D.A., Nasyrov, E. et al. Intermediate-term impact on corneal endothelial cells and efficacy of Preserflo MicroShunt implantation in patients with open-angle glaucoma – a prospective study over two years. Graefes Arch Clin Exp Ophthalmol (2024). https://doi.org/10.1007/s00417-024-06508-8
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00417-024-06508-8