Abstract
Purpose
This study evaluated the effects of dietary nitrate (NO3 −) supplementation on physiological functioning and exercise performance in trained runners/triathletes conducting short and longer-distance treadmill running time-trials (TT).
Method
Eight trained male runners or triathletes completed four exercise performance tests comprising a 10 min warm up followed by either a 1500 or 10,000 m treadmill TT. Exercise performance tests were preceded 3 h before the exercise by supplementation with either 140 ml concentrated nitrate-rich (~12.5 mmol nitrate) (BRJ) or nitrate-deplete (~0.01 mmol nitrate) (PLA) beetroot juice.
Results
BRJ supplementation significantly elevated plasma [NO2 −] (P < 0.05). Resting blood pressure and exercise \(\dot{\text{V}}{{\text{O}}_{\text{2}}}\) were not significantly different between BRJ and PLA (P > 0.05). However, post-exercise blood [lactate] was significantly greater in BRJ following the 1500 m TT (6.6 ± 1.2 vs. 6.1 ± 1.5 mM; P < 0.05), but not significantly different between conditions in the 10,000 m TT (P > 0.05). Performance in the 1500 m TT was significantly faster in BRJ vs. PLA (319.6 ± 36.2 vs. 325.7 ± 38.8 s; P < 0.05). Conversely, there was no significant difference in 10,000 m TT performance between conditions (2643.1 ± 324. 1 vs. 2649.9 ± 319.8 s, P > 0.05).
Conclusion
Acute BRJ supplementation significantly enhanced 1500 m, but not 10,000 m TT performance. These findings suggest that BRJ might be ergogenic during shorter distance TTs which allow for a high work rate, but not during longer distance TTs, completed at a lower work rate.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Nitric oxide (NO) is a reactive gaseous signalling molecule that regulates a broad range of physiological processes, including muscle contraction (Reid 1998), mitochondrial respiration (Clerc et al. 2007), vasodilation (Kelm and Schrader 1990), and immune function (Bogdan 2001). NO can be generated by the NO synthase (NOS) family of enzymes from the precursors l-arginine and oxygen (O2), in a complex reaction that requires the presence of multiple cofactors (Moncada and Higgs 1993). A complementary pathway for NO production has also been elucidated, and entails the reduction of nitrate (NO3 −) into nitrite (NO2 −) by oral bacteria (Duncan et al. 1995), and subsequent reduction of NO2 − into NO throughout the body via multifarious enzymatic and non-enzymatic catalysts (Lundberg and Govoni 2004).
A strong body of evidence indicates that NO3 − supplementation can improve time to exhaustion (TTE) and/or time-trial (TT) performance during short duration (<30 min) exercise tests, which mandate a high relative (to the peak) work rate (Bailey et al. 2009, 2010; Vanhatalo et al. 2010; Lansley et al. 2011; Cermak et al. 2012a; Porcelli et al. 2014; McMahon et al. 2016), particularly in untrained and moderately-trained individuals (\(\dot{\text{V}}{{\text{O}}_{\text{2max}}}\) < ~60 ml kg−1 min−1). Notably, Lansley et al. (2011) reported a significant and comparable improvement in 4 km (TT duration: ~6 min; performance improvement: 2.8%) and 16.1 km (TT duration: ~ 28 min; performance improvement: 2.7%) cycle ergometry TT performance in moderately trained males (\(\dot{\text{V}}{{\text{O}}_{\text{2max}}}\) ~56 ml kg−1 min−1) consequent to NO3 − ingestion. Conversely, NO3 − supplementation has been reported to have a minimal effect on performance during longer duration (>30 min) exercise tests, which mandate a lower work rate (Bescós et al. 2012; Wilkerson et al. 2012; Cermak et al. 2012b; Lane et al. 2014; Glaister et al. 2015).
The precise reasons for this disparity are unclear, but might be related to differences in muscle recruitment patterns between short and longer duration exercise tests. Recent data from murine models suggest that NO3 − supplementation elevates blood flow and oxygenation (Ferguson et al. 2013, 2015), and increases calcium (Ca2+) handling protein expression and force production (Hernandez et al. 2012) in type II muscle only (for review, see Jones et al. 2016). Therefore, the beneficial effects of NO3 − supplementation might be restricted to, or most pronounced during, exercise where type II fibers are predominantly recruited (Breese et al. 2013; Bailey et al. 2015; Coggan et al. 2015), such as during short duration exercise where a higher relative work rate can be adopted (Essén 1978; Sale 1987). Additionally, hypoxia and acidosis—cellular conditions particularly prevalent at higher work rates (Richardson et al. 1995; Robergs et al. 2004)—are known to enhance the reduction of NO2 − into NO (Modin et al. 2001; Cosby et al. 2003; Castello et al. 2006). Consequently, NO3 − supplementation may be more effective at elevating NO2 − derived NO, and thus enhancing NO signalling, during short duration exercise tests where a high work rate is adopted, compared with longer duration exercise tests conducted at a lower work rate.
Previous investigations utilising longer duration (>30 min) exercise tests have employed trained subjects (Males: \(\dot{\text{V}}{{\text{O}}_{\text{2max}}}\) >60 ml kg−1 min−1, Females: \(\dot{\text{V}}{{\text{O}}_{\text{2max}}}\) >50 ml kg−1 min−1), who manifest a diminished response to NO3 − supplementation (Porcelli et al. 2014). This may be explained by the elevated baseline plasma [NO3 −] and [NO2 −] in this cohort (Vassalle et al. 2003), although multiple other factors may also be important (Jones 2014). It is therefore unclear whether the reduced effects of NO3 − supplementation reported during longer vs. shorter duration exercise tests are related to the exercise duration (and requisite work rate) or the high training status of subjects in these investigations (Bescós et al. 2012; Wilkerson et al. 2012; Cermak et al. 2012b; Lane et al. 2014; Glaister et al. 2015). Direct comparison between the effects of NO3 − supplementation on short and longer duration (< or >30 min) exercise performance where other methodological variables are fixed is necessary, yet presently scarce.
Therefore, the purpose of this investigation was to evaluate the effects of NO3 − supplementation on short duration high work rate (1500 m; ~5 min) versus longer duration lower work rate (10,000 m; ~45 min) treadmill running TT performance in a group of trained men following an identical NO3 − supplementation regimen. We hypothesized that NO3 − supplementation would enhance short, but not longer duration exercise performance.
Methods
Subjects
Eight trained male runners or triathletes (age: 28.3 ± 5.8 years, body mass: 74.7 ± 10.1 kg, height 179.1 ± 2.4 cm, \(\dot{\text{V}}{{\text{O}}_{\text{2max}}}\): 62.3 ± 8.1 ml kg−1 min−1) volunteered to take part in this study. The subjects all had experience of competing in running events. The procedures and any associated risks and benefits of the study were explained to the subjects, after which they gave fully informed written consent. The study received institutional ethical clearance, and was conducted in accordance with the Declaration of Helsinki.
Experimental design
Each subject visited the laboratory on six separate occasions. On the first visit, subjects completed an incremental exercise test to exhaustion to determine their maximal rate of oxygen uptake (\(\dot{\text{V}}{{\text{O}}_{\text{2max}}}\)) and elucidate suitable sub-maximal warm up exercise intensities for the experimental trials. Following a rest period of approximately 20 min, subjects then completed familiarization of the 1500 m TT. On the second visit to the laboratory, subjects completed familiarization of the 10,000 m TT. The third to sixth visits included each of the following performance trials: (1) 1500 m TT preceded by nitrate-rich beetroot juice (BRJ) supplementation; (2) 1500 m TT preceded by nitrate-deplete beetroot juice (PLA) supplementation; (3) 10,000 m TT preceded by BRJ supplementation; (4) 10,000 m TT preceded by PLA supplementation. Performance trials were conducted in a randomized order, with 140 ml concentrated BRJ (~12.5 mmol nitrate) or PLA (~0.01 mmol nitrate) (Beet It, James White Ltd., Ipswich, UK) administered double blind, 3 h before the start of exercise. The exact NO3 − content of the beetroot juice was determined via ozone-based chemiluminescence, as previously described (Shannon et al. 2016b). Subjects were asked to complete a food and exercise diary for 24 h prior to the first experimental trial, and used this to replicate their diet and activity as closely as possible for subsequent visits. During this time period, subjects were asked not to perform strenuous exercise, or consume caffeine and alcohol. Subjects were also asked not to consume anything except for water in the 3 h before testing. Antibacterial mouthwash and chewing gum were also avoided throughout the testing period, as these are known to destroy the oral bacteria responsible for nitrate reduction into nitrite (Govoni et al. 2008). Each testing sessions was conducted at the same time of the day to minimise the influence of circadian variance.
Maximal exercise testing
Subjects completed a two-part incremental running test on a motorized treadmill (Woodway, Cranlea, Birmingham, UK) (Jones 2007). The first part of the test comprised five to eight, 3-min sub-maximal stages, separated with 1 min recovery periods. Running speed was increased by 1 km h−1 each stage. A finger-tip blood sample was also obtained between stages to determine blood [lactate] (YSI 2300 STAT plus, Yellow Springs, Ohio, USA). At the end of each stage, heart rate (HR) was monitored via a chest worn strap (Polar Electro, Oy, Finland), and ratings of perceived exertion (RPE) was assessed using a 15-point (6–20) scale. A 1% treadmill gradient was applied to replicate the energetic cost of running outdoors (Jones and Doust 1996). Exercise was stopped when blood [lactate] exceeded 4 mM. Subjects then rested for approximately 5 min, after which the second phase of the test commenced. A fixed running speed was applied, equal to the final speed achieved during the first part of the test, minus 2 km h−1. Treadmill gradient was increased by 1% every minute, until volitional exhaustion. An online gas analysis system (MedGraphics Ultima CPX, MGC Diagnostics, MN, USA), calibrated before each trial according to the manufacturer’s instructions, was used to monitor expired gas throughout exercise. Expired gas data was used to define \(\dot{\text{V}}{{\text{O}}_{\text{2max}}}\) (greatest 30 s mean value) and the relevant sub-maximal running speeds for the experimental trials. All subjects were deemed to have attained \(\dot{\text{V}}{{\text{O}}_{\text{2max}}}\), as they achieved ≥2 of the following criteria: a plateau in \(\dot{\text{V}}{{\text{O}}_{\text{2}}}\) in the final exercise stage (Taylor et al. 1955), RER ≥1.15 (Issekutz et al. 1962), HR within 10 b min−1 of age-predicted maximum (220—age), RPE ≥19, blood [lactate] ≥8 mM (Midgley et al. 2007).
Performance trials
Approximately 1 week after the 10,000 m familiarization trial, subjects completed the first of four performance trials. All performance trials were conducted over a 3–5-week period, with a minimum of 5 days and maximum of 10 days between visits. These consisted of 10 min of continuous treadmill running as a warm up period, split as 5 min bouts at 45 and 65% \(\dot{\text{V}}{{\text{O}}_{\text{2max}}}\), respectively. After a 5-min passive rest period, subjects then completed either a 1500 or a 10,000 m TT. Each TT commenced with a 30 s rolling start at a running speed approximating 80% \(\dot{\text{V}}{{\text{O}}_{\text{2max}}}\), after which subjects could freely adjust their speed. Running speed and time were not visible to subjects during the TT. Subjects were verbally informed of the distance they had covered at 200 m (1500 m TT) or 500 m (10,000 m TT) intervals. The treadmill gradient was set to 1% throughout the exercise (Jones and Doust 1996).
Measurements
Prior to the start of each performance trial, subjects were required to sit for a 10-min period, after which blood pressure (BP) of the brachial artery was measured using an automated sphygmomanometer (Omron Healthcare Ltd., Kyoto, Japan). Four measures were obtained, and the mean value of the final three measurements was used for data analysis. An 8 ml venous blood sample was then collected via venepuncture from a vein in the arm into lithium heparin containing tubes. Blood was immediately centrifuged at 5000 rpm for 3 min. Plasma was extracted into opaque cryotubes (Argos Technologies, IL, USA), which were pre-treated with 6.5 mM N-ethylmaleimide (NEM) and 0.1 mM Diethylenetriaminepentaacetic acid (DTPA) to prevent the interchange between NO metabolites (Nagababu and Rifkind 2010). Cryotubes were immediately placed in a freezer at −80 °C, for later analysis of plasma [NO2 −] via ozone-based chemiluminescence, as previously described (Shannon et al. 2016b). A blood sample was also collected from the fingertip, to determine blood [lactate] (YSI 2900, Yellow Springs, OH, USA). A further fingertip blood sample was obtained immediately after exercise, and an additional venous blood sample was collected within 5 min of TT completion to monitor the change in plasma [NO2 −]. HR and RPE were monitored immediately following completion of the TT, as previously described. Expired gas was monitored continuously throughout TT exercise, as previously described.
Data analysis
Data analysis was conducted using SPSS version 22. An α level of P < 0.05 was accepted for significance. Between-supplement differences in plasma [NO2 −], the transient change in pulmonary gas data, blood [lactate], and TT splits and running speed were compared in the 1500 and 10,000 m exercise testing sessions using a two-way (time × condition) repeated measures ANOVA. In the 1500 m condition, pulmonary gas data, TT splits, and running speed were averaged over 200 m intervals to 1400 m, and then during the final 100 m. Alternatively, these data were averaged over 1000 m intervals for the 10,000 m TT. Post-hoc analysis was conducted using t-tests with a Bonferroni correction. Between-supplement differences in BP, post-exercise HR and RPE, mean (average during the entire TT) \(\dot{\text{V}}{{\text{O}}_{\text{2}}}\), \(\dot{\text{V}}\text{C}{{\text{O}}_{\text{2}}}\), RER, and TT performance were compared in the 1500 and 10,000 m exercise testing sessions using a paired t-test. A statistical spreadsheet was also applied to derive qualitative probabilistic inferences for performance data (Hopkins 2007). Comparisons were made against a smallest worthwhile change (SWC) of 0.5% derived from previously published data (Russell et al. 2004; Shannon et al. 2016a). Verbal descriptors were used to express the chance that the true value of the effect was beneficial, trivial, or harmful, according to the following scale: < 0.5%, ‘almost certainly not’; 0.5–5%, ‘very unlikely not’; 5–25%, ‘unlikely’; 25–75%, ‘possibly’; 75–95%, ‘likely’; ‘95–99.5%’, ‘very likely’; >99.5%, ‘almost certainly’. The effect was deemed unclear when an odds ratio of benefit to harm of <66 was identified, corresponding to a 25% chance of benefit and 0.5% risk of harm. Data are presented as mean ± SD, unless otherwise stated.
Results
Plasma [NO2 −] and blood pressure
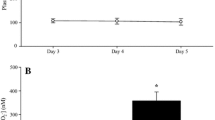
Plasma [NO2 −] data for the 1500 and 10,000 m exercise test protocols is presented in Fig. 1. Pre-exercise plasma [NO2 −] was significantly elevated in BRJ compared with PLA prior to both the 1500 and 10,000 m TTs (P < 0.05). Pre-exercise plasma [NO2 −] tended to be greater in the 1500 m compared to 10,000 m BRJ condition, and was significantly greater prior to the 1500 m compared with the 10,000 m exercise testing session in PLA (P < 0.05). Plasma [NO2 −] remained significantly elevated in both BRJ conditions post-exercise compared with PLA (P < 0.05). In the 1500 m exercise test protocol, post-exercise plasma [NO2 −] was significantly decreased in BRJ relative to pre-exercise levels (P < 0.05). In contrast, plasma [NO2 −] was not significantly different to pre-exercise levels in PLA (P > 0.05). In the 10,000 m exercise test protocol, post-exercise plasma [NO2 −] was not significantly different to pre-exercise levels in either BRJ (P > 0.05) or PLA (P > 0.05). Post-exercise plasma [NO2 −] was not significantly different between the two BRJ conditions, or between the two PLA conditions (P > 0.05).
Group mean ± SEM pre and post-exercise plasma [NO2 −] in BRJ (closed circles/triangles) and PLA (open circles/triangles). BRJ significantly elevated plasma [NO2 −] above PLA in both 1500 and 10,000 m conditions (*) (P < 0.05). Pre-exercise plasma [NO2 −] was significantly greater in 1500 m PLA compared to 10,000 m PLA (+) (P < 0.05). Plasma [NO2 −] significantly decreased in 1500 m BRJ (#) (P < 0.05)
There was no difference in mean arterial blood pressure (MAP) in BRJ compared with PLA prior to the 1500 m (BRJ 92 ± 5 vs. PLA 92 ± 5 mmHg; P > 0.05) nor 10,000 m (BRJ 94 ± 6 vs. PLA 94 ± 6 mmHg; P > 0.05) exercise test protocols.
Heart rate and ratings of perceived exertion
HR and RPE assessed immediately following the TTs did not differ significantly between BRJ and PLA (P > 0.05; Table 1).
Pulmonary gas exchange
Mean pulmonary \(\dot{\text{V}}{{\text{O}}_{\text{2}}}\), \(\dot{\text{V}}\text{C}{{\text{O}}_{\text{2}}}\), and RER data throughout the 1500 and 10,000 m TTs did not differ significantly between BRJ and PLA (P > 0.05; Table 1). The transient change in these data at 200 m (1500 m TT) and 1000 m (10,000 m TT) intervals throughout the TT is presented in Fig. 2. These variables did not differ significantly between BRJ and PLA at any time point (P > 0.05).
The transient changes in pulmonary gas-exchange data with BRJ (closed circles/triangles) and PLA (open circles/triangles) throughout the 1500 m (a–c) and 10,000 m (d–f) TTs. Data for the 1500 m TT represents average values over 200 m intervals up to 1400 m, and during the final 100 m. Data for the 10,000 m TT represents average values over 1000 m intervals
Blood [lactate]
Pre-exercise blood [lactate] was not significantly different between BRJ and PLA in either exercise test protocol (P > 0.05). In the 1500 m exercise test protocol, post-exercise blood [lactate] was significantly greater in BRJ compared with PLA (6.6 ± 1.2 vs. 6.1 ± 1.5 mM; P < 0.05), however, not different between BRJ and PLA in the 10,000 m exercise test protocol (4.5 ± 1.5 vs. 4.2 ± 0.8 mM; P > 0.05).
Time trial performance
Individual performance times and running speed for the 1500 and 10,000 m TT are presented in Fig. 3.
Individual performance times and running speed during the 1500 and 10,000 m TT tests. Performance in the 1500 m TT was significantly faster (asterisk) in BRJ (filled circles) compared with PLA (open circles) (a) (P < 0.05). There was no significant difference in 10,000 m TT performance between BRJ (filled triangles) and PLA (open triangles) (c). Running speed did not significantly differ between BRJ (closed circles/triangles) and PLA (open circles/triangles) in the 1500 m (b) and 10,000 m (d) TT tests
1500 m TT
Completion time was significantly faster in BRJ (319.6 ± 36.2 s) compared with PLA (325.7 ± 38.8 s; P < 0.05) for the 1500 m TT. Magnitude based inferences indicated a ‘very likely beneficial’ effect of BRJ on group mean 1500 m TT performance. Six of eight subjects exceeded the SWC for an improvement in 1500 m TT performance in BRJ vs. PLA (Fig. 3a). There was no significant difference in running speed at any of the 200 m (up to 1400 m) and final 100 m (1400–1500 m) splits (P > 0.05, Fig. 3b).
10,000 m TT
There was no significant difference in 10,000 m TT completion time between BRJ (2643.1 ± 324.1 s) compared to PLA (2649.9 ± 319.8 s, P > 0.05). Magnitude-based inferences indicated an ‘unclear’ effect of BRJ on group mean 10,000 m TT performance. However, four of eight subjects exceeded the SWC for an improvement in 10,000 m TT performance in BRJ vs. PLA (Fig. 3c). There was no significant difference in running speed at any of the 1000 m splits (P > 0.05, Fig. 3d). However, when the TT was divided into two, 5000 m segments for analysis, subjects had a significantly faster running speed (BRJ 14.0 ± 1.6 vs. PLA 13.7 ± 1.5 km h−1, P < 0.05) and quicker time (BRJ 1287.5 ± 153 vs. PLA 1317.6 ± 149.9 s, P < 0.05) in the first 5000 m with BRJ compared to PLA. There was no difference in running speed (BRJ 13.3 ± 1.8 vs. PLA 13.5 ± 1.8 km·h−1, P > 0.05) nor time (BRJ 1355.6 ± 189.5 vs. PLA 1332.3 ± 173.3 s, P > 0.05) during the second 5000 m split. Magnitude based inferences indicated a ‘very likely beneficial’ effect of BRJ during the first 5000 m, and an ‘unclear’ effect of BRJ during the second 5000 m of the 10,000 m TT.
Discussion
The main finding of the present investigation was that acute supplementation with BRJ significantly enhanced 1500 m treadmill TT performance, but not 10,000 m treadmill TT performance. This suggests that BRJ supplementation might improve performance in trained males conducting shorter distance TTs which allow for a high work rate, but not necessarily during longer distance TTs where a lower work rate is mandated.
Effects of BRJ on plasma [NO2 −] and blood pressure
In agreement with the findings of a number of earlier investigations (e.g. Bailey et al. 2009, 2010; Vanhatalo et al. 2010; Lansley et al. 2011; Cermak et al. 2012a; Wylie et al. 2013; Muggeridge et al. 2014; Shannon et al. 2016b; McMahon et al. 2016), BRJ supplementation significantly elevated pre-exercise plasma [NO2 −], which is a highly sensitive marker of NO bioavailability (Lundberg et al. 2008). Interestingly, despite close replication of diet and exercise for 24 h preceding each trial, administration of identical batch checked supplements, and a consistent time-frame for post-supplement blood sampling, there was some variation in pre-exercise plasma [NO2 −] between the two BRJ conditions, and between the two PLA conditions. Given pre-exercise plasma [NO2 −] was ~5–6-fold greater in both BRJ conditions compared to PLA, and remained elevated above PLA post-exercise, it is likely that the difference in the performance response to BRJ between 1500 and 10,000 m exercise test protocols is not attributable to insufficient availability of NO2 − as a ‘substrate’ for NO generation in the 10,000 m BRJ trial. Plasma [NO2 −] did not significantly decrease post-exercise in the BRJ 10,000 m condition, yet declined significantly in the BRJ 1500 m condition. This suggests that it is differences in the ‘use’ rather than the availability of NO2 − that is important. This might be related to greater acidosis and hypoxia, and thus NO2 − reduction (Modin et al. 2001; Cosby et al. 2003; Castello et al. 2006), in the 1500 vs. 10,000 m conditions, as a consequence of the relatively greater work rate adopted (~86 vs. 78% \({\dot{\text{V}}}{{\text{O}}_{\text{2max}}}\); ~6.5 vs. 4.5 mM blood [Lactate]).
The increase in plasma [NO2 −] with BRJ did not translate into a significant reduction in BP, as evident by similar MAP values between conditions. Previous investigations indicate that the magnitude of the decline in BP typically observed with NO3 − supplementation is related to the initial BP of the subject (Ashworth et al. 2015). Thus, it is possible that the relatively low BP in these subjects minimized the effects of NO3 − supplementation on this parameter.
Effects of BRJ on 1500 m TT performance
In the cohort of trained men recruited for this study, it was hypothesized that BRJ would enhance 1500 m TT performance. In agreement with previous literature and our experimental hypothesis, BRJ supplementation significantly enhanced 1500 m running performance, with a mean improvement of 1.9%—an effect deemed ‘very likely beneficial’ via magnitude based inferential statistics. These data are comparable to previous studies which have demonstrated that the ingestion of dietary NO3 − significantly enhances short duration (<30 min) TTE and/or TT performance in untrained and moderately-trained individuals (\(\dot{\text{V}}{{\text{O}}_{\text{2max}}}\) < ~60 ml kg−1 min−1) (Bailey et al. 2009, 2010; Vanhatalo et al. 2010; Lansley et al. 2011; Cermak et al. 2012a, b; Porcelli et al. 2014). In contrast, limited effects of NO3 − supplementation have been reported for similar duration exercise tests in individuals with a greater training status (i.e. well-trained and elite athletes) (\(\dot{\text{V}}{{\text{O}}_{\text{2max}}}\) >70 ml·kg−1·min−1) (Peacock et al. 2012; Bescós et al. 2012; Christensen et al. 2013; Porcelli et al. 2014; Sandbakk et al. 2015). These highly trained athletes manifest a host of different characteristics likely to diminish their responsiveness to NO3 − supplementation (for review, see Jones 2014), including elevated endogenous NO3 − and NO2 − reserves (Vassalle et al. 2003).
Regular splits were obtained throughout TTs, which allowed us to compare the pacing strategy between BRJ and PLA conditions, and identify the points at which BRJ might have enhanced running speed. Although there were no significant differences in running speed at any of the splits, subjects tended to run faster during the first 1000 m of the 1500 m TT in BRJ compared with PLA, such that the majority of time ‘gained’ in BRJ occurred during this period. Subjects achieved similar running speeds between 1000 and 1400 m, although this represented a greater percentage of average pace in PLA which could not be maintained. Consequently, subjects were typically unable to employ a sprint finish in PLA, unlike in BRJ. The apparent benefits of BRJ during the first and last phases of the 1500 m TT could be of tactical advantage during competition.
A range of mechanisms might explain the ergogenic effect of NO3 − supplementation on 1500 m TT performance observed here. Multiple previous studies have reported a reduction in the O2 cost of exercise with NO3 − supplementation (Larsen et al. 2007; Bailey et al. 2009; Lansley et al. 2011; Cermak et al. 2012a; Wylie et al. 2013; Pawlak-Chaouch et al. 2016), consequent to either an improvement in the efficiency of mitochondrial respiration (Larsen et al. 2011) and/or muscle contraction (Bailey et al. 2010). However, this mechanism cannot explain the improvement in performance observed in this study, as BRJ supplementation did not reduce O2 consumption during exercise. Instead, the ergogenic effect of BRJ may be better explained by other physiological changes. Interestingly, recent murine model studies have identified effects of NO3 − which appear to be selective to type II muscle (for review, see Jones et al. 2016), and may be of mechanistic relevance in the present study given the likely high recruitment of type II fibers during the 1500 m TT (Essén 1978; Sale 1987). NO3 − supplementation was reported to augment blood flow and thus O2 delivery to type II muscle in exercising rats (Ferguson et al. 2013, 2015). It was suggested that these local blood flow changes might be beneficial by increasing the microvascular partial pressure of O2 (PO2), and concomitantly decreasing muscle metabolic perturbations (e.g. PCr degradation, ADP and Pi accumulation)—effects previously demonstrated to extend TTE with NO3 − supplementation in humans (Bailey et al. 2010; Vanhatalo et al. 2011). The enhanced 1500 m exercise performance subsequent to BRJ in the present study may thus, at least partly, be underpinned by changes to type II muscle blood flow and O2 delivery. Additionally, Hernandez et al. (2012) observed an increased expression, and therefore content, of the Ca2+ handling protein calsequestrin 1 and the dihydropyridine receptor. These effects, which were reported in predominantly type II muscle, were accompanied by an increased muscle force production. Subsequent studies have confirmed the beneficial effect of NO3 − on force production in humans (Haider and Folland 2014; Coggan et al. 2015). It is therefore possible that elevated type II muscle Ca2+ handling and muscle contractile function may have further contributed to the improved 1500 m performance observed in the present investigation. Furthermore, several recent studies have observed improvements in physiological functioning and exercise performance in situations where type II muscle recruitment is likely to be high (Breese et al. 2013; Bailey et al. 2015; Coggan et al. 2015), which further supports the findings of this investigation.
Effects of BRJ on 10,000 m TT performance
There was no significant difference in 10,000 m TT performance between BRJ and PLA in this study. Magnitude based inferences also indicated an ‘unclear’ practical effect of BRJ on 10,000 m TT performance. Nevertheless, four subjects exceeded the SWC in performance following BRJ in the present study, supporting the notion of a responder vs. non-responder phenomenon (Wilkerson et al. 2012).
Interestingly, subjects appeared to adopt disparate pacing strategies in BRJ and PLA conditions. In BRJ, subjects ran the first 5000 m significantly faster, yet the pace gradually declined throughout the TT before a final sprint finish. Conversely, in PLA subjects adopted a more even paced strategy prior to a similar paced sprint finish. While it is important to acknowledge that overall 10,000 m performance was no different between conditions, the faster first 5000 m with BRJ might offer tactical advantages in competition. In contrast, Wilkerson et al. (2012) observed no difference in power output during the earlier stages of a 50 mile (total exercise duration: >2 h) cycle ergometry TT, yet the final 10 miles was faster with NO3 − supplementation. Together, these findings might suggest a potential application of BRJ during higher work rate periods within a longer duration event, perhaps specifically those which require greater type II muscle recruitment or markedly decrease the muscle pH and PO2 and hence, augment NO2 − reduction into NO (Modin et al. 2001; Cosby et al. 2003; Castello et al. 2006). This might include facilitating a fast start or finish during competition, or responding to a surge by another competitor. Investigations into the performance effects of NO3 − supplementation during ‘real world’ competition would provide interesting insight.
The effects of NO3 − supplementation are likely determined by an interaction between multiple variables, rather than individual factors, and hence the results of this study cannot easily be generalised to other populations or experimental conditions. Aerobic fitness strongly moderates the effects of NO3 − supplementation (Porcelli et al. 2014), such that the beneficial effects of NO3 − supplementation on 1500 m TT performance might not be applicable to individuals with a higher training status (Boorsma et al. 2014). Likewise, it is possible that untrained individuals may derive a beneficial effect of NO3 − supplementation during longer duration (>30 min) lower work rate exercise tests, which remains to be explored. Additionally, there is some evidence to suggest that chronic dosing might elicit more robust physiological and performance changes (Vanhatalo et al. 2010). Therefore, it cannot be ruled out that NO3 − supplementation may be beneficial for longer duration lower work rate exercise tests or in trained individuals when more protracted supplementation strategies are applied. Future investigations exploring the potential interaction between these and other variables are warranted, to help elucidate the precise conditions where NO3 − supplementation might be beneficial.
In conclusion, the results of the present study suggest that acute BRJ supplementation increases plasma [NO2 −], and enhances 1500 m treadmill TT performance in trained men. In contrast, BRJ supplementation did not significantly enhance 10,000 m TT performance. These findings provide important insight into the specific conditions where BRJ supplementation may be ergogenic, and evidence that the ergogenic effect of NO3 − supplementation is moderated by the duration and requisite work rate of a TT.
Abbreviations
- ANOVA:
-
Analysis of variance
- ATP:
-
Adenosine triphosphate
- BP:
-
Blood pressure
- BRJ:
-
Nitrate-rich beetroot juice
- Ca2+ :
-
Calcium
- DTPA:
-
Diethylenetriaminepentaacetic acid
- HR:
-
Heart rate
- MAP:
-
Mean arterial pressure
- NEM:
-
N-Ethylmaleimide
- NO:
-
Nitric oxide
- NO2 − :
-
Nitrite
- NO3 − :
-
Nitrate
- NOS:
-
Nitric oxide synthase
- O2 :
-
Oxygen
- PCr:
-
Phosphocreatine
- PO2 :
-
Partial pressure of oxygen
- PLA:
-
Nitrate-deplete beetroot juice
- RER:
-
Respiratory exchange ratio
- RPE:
-
Ratings of perceived exertion
- SWC:
-
Smallest worthwhile change
- TT:
-
Time-trial
- TTE:
-
Time to exhaustion
- \(\dot{\text{V}}\text{C}{{\text{O}}_{\text{2}}}\) :
-
Carbon dioxide production
- \(\dot{\text{V}}{{\text{O}}_{\text{2}}}\) :
-
Oxygen uptake
- \(\dot{\text{V}}{{\text{O}}_{\text{2max}}}\) :
-
Maximal oxygen uptake
References
Ashworth A, Mitchell K, Blackwell JR, Vanhatalo A, Jones AM (2015) High-nitrate vegetable diet increases plasma nitrate and nitrite concentrations and reduces blood pressure in healthy women. Public Health Nutr 18:2669–2678. doi:10.1017/S1368980015000038
Bailey SJ, Winyard P, Vanhatalo A, Blackwell JR, DiMenna FJ, Wilkerson DP, Tarr J, Benjamin N, Jones AM (2009) Dietary nitrate supplementation reduces the O2 cost of low-intensity exercise and enhances tolerance to high-intensity exercise in humans. J Appl Physiol 107:1144–1155. doi:10.1152/japplphysiol.00722.2009
Bailey SJ, Fulford J, Vanhatalo A, Winyard PG, Blackwell JR, DiMenna FJ, Wilkerson DP, Benjamin N, Jones AM (2010) Dietary nitrate supplementation enhances muscle contractile efficiency during knee-extensor exercise in humans. J Appl Physiol 109:135–148. doi:10.1152/japplphysiol.00046.2010
Bailey SJ, Varnham RL, DiMenna FJ, Breese BC, Wylie LJ, Jones AM (2015) Inorganic nitrate supplementation improves muscle oxygenation, O2 uptake kinetics, and exercise tolerance at high but not low pedal rates. J Appl Physiol 118:1396–1405. doi:10.1152/japplphysiol.01141.2014
Bescós R, Ferrer-Roca V, Galilea PA, Roig A, Drobnic F, Sureda A, Martorell M, Cordova A, Tur JA, Pons A (2012) Sodium nitrate supplementation does not enhance performance of endurance athletes. Med Sci Sports Exerc 44:2400–2409. doi:10.1249/MSS.0b013e3182687e5c
Bogdan C (2001) Nitric oxide and the immune response. Nat Immunol 2:907–916. doi:10.1038/ni1001-907
Boorsma RK, Whitfield J, Spriet LL (2014) Beetroot juice supplementation does not improve performance of elite 1500-m runners. Med Sci Sports Exerc 46:2326–2334. doi:10.1249/MSS.0000000000000364
Breese BC, McNarry MA, Marwood S, Blackwell JR, Bailey SJ, Jones AM (2013) Beetroot juice supplementation speeds O2 uptake kinetics and improves exercise tolerance during severe-intensity exercise initiated from an elevated metabolic rate. Am J Physiol Regul Integr Comp Physiol 305:R1441–R1450. doi:10.1152/ajpregu.00295.2013
Castello PR, David PS, McClure T, Crook Z, Poyton RO (2006) Mitochondrial cytochrome oxidase produces nitric oxide under hypoxic conditions: implications for oxygen sensing and hypoxic signalling in eukaryotes. Cell Metab 3:277–287. doi:10.1016/j.cmet.2006.02.011
Cermak NM, Gibala MJ, van Loon LJ, others (2012a) Nitrate supplementation’s improvement of 10-km time-trial performance in trained cyclists. Int J Sport Nutr Exerc Metab 22:64–71
Cermak NM, Res P, Stinkens R, Lundberg JO, Gibala MJ, van Loon L JC (2012b) No improvement in endurance performance after a single dose of beetroot juice. Int J Sport Nutr Exerc Metab 22:470–478
Christensen PM, Nyberg M, Bangsbo J (2013) Influence of nitrate supplementation on VO2 kinetics and endurance of elite cyclists. Scand J Med Sci Sports 23:e21–e31. doi:10.1111/sms.12005
Clerc P, Rigoulet M, Leverve X, Fontaine E (2007) Nitric oxide increases oxidative phosphorylation efficiency. J Bioenerg Biomembr 39:158–166. doi:10.1007/s10863-007-9074-1
Coggan AR, Leibowitz JL, Kadkhodayan A, Thomas DP, Ramamurthy S, Spearie CA, Waller S, Farmer M, Peterson LR (2015) Effect of acute dietary nitrate intake on maximal knee extensor speed and power in healthy men and women. Nitric Oxide 48:16–21. doi:10.1016/j.niox.2014.08.014
Cosby K, Partovi KS, Crawford JH, Patel RP, Reiter CD, Martyr S, Yang BK, Waclawiw MA, Zalos G, Xu X, Huang KT, Shields H, Kim-Shapiro DB, Schechter AN, Cannon RO, Gladwin MT (2003) Nitrite reduction to nitric oxide by deoxyhemoglobin vasodilates the human circulation. Nat Med 9:1498–1505. doi:10.1038/nm954
Duncan C, Dougall H, Johnston P, Green S, Brogan R, Leifert C, Smith L, Golden M, Benjamin N (1995) Chemical generation of nitric oxide in the mouth from the enterosalivary circulation of dietary nitrate. Nat Med 1:546–551
Essén B (1978) Glycogen depletion of different fibre types in human skeletal muscle during intermittent and continuous exercise. Acta Physiol Scand 103:446–455. doi:10.1111/j.1748-1716.1978.tb06239.x
Ferguson SK, Hirai DM, Copp SW, Holdsworth CT, Allen JD, Jones AM, Musch TI, Poole DC (2013) Impact of dietary nitrate supplementation via beetroot juice on exercising muscle vascular control in rats. J Physiol 591:547–557. doi:10.1113/jphysiol.2012.243121
Ferguson SK, Holdsworth CT, Wright JL, Fees AJ, Allen JD, Jones AM, Musch TI, Poole DC (2015) Microvascular oxygen pressures in muscles comprised of different fiber types: impact of dietary nitrate supplementation. Nitric Oxide 48:38–43. doi:10.1016/j.niox.2014.09.157
Glaister M, Pattison JR, Muniz-Pumares D, Patterson SD, Foley P (2015) Effects of dietary nitrate, caffeine, and their combination on 20-km cycling time trial performance. J Strength Cond Res 29:165–174. doi:10.1519/JSC.0000000000000596
Govoni M, Jansson EA, Weitzberg E, Lundberg JO (2008) The increase in plasma nitrite after a dietary nitrate load is markedly attenuated by an antibacterial mouthwash. Nitric Oxide Biol Chem Off J Nitric Oxide Soc 19:333–337. doi:10.1016/j.niox.2008.08.003
Haider G, Folland JP (2014) Nitrate supplementation enhances the contractile properties of human skeletal muscle. Med Sci Sports Exerc 46:2234–2243. doi:10.1249/MSS.0000000000000351
Hernandez A, Schiffer TA, Ivarsson N, Cheng AJ, Bruton JD, Lundberg JO, Weitzberg E, Westerblad H (2012) Dietary nitrate increases tetanic [Ca2+]i and contractile force in mouse fast-twitch muscle. J Physiol 590:3575–3583. doi:10.1113/jphysiol.2012.232777
Hopkins WG (2007) A spreadsheet for deriving a confidence interval, mechanistic inference and clinical inference from a P value. Sportscience 11:16–20.
Issekutz B, Birkhead NC, Rodahl K (1962) Use of respiratory quotients in assessment of aerobic work capacity. J Appl Physiol 17:47–50
Jones AM (2007) Middle- and long-distance running. In: Winter EM, Jones AM, Davison R, Bromley PD, Mercer TH (eds) Sport and exercise physiology testing guidelines: volume I—sport testing. The British Association of Sport and Exercise Sciences Guide. Routledge, London
Jones AM (2014) Influence of dietary nitrate on the physiological determinants of exercise performance: a critical review. Appl Physiol Nutr Metab 39:1019–1028. doi:10.1139/apnm-2014-0036
Jones AM, Doust JH (1996) A 1% treadmill grade most accurately reflects the energetic cost of outdoor running. J Sports Sci 14:321–327. doi:10.1080/02640419608727717
Jones AM, Ferguson SK, Bailey SJ, Vanhatalo A, Poole DC (2016) Fiber-type specific effects of dietary nitrate. Exerc Sport Sci Rev. doi:10.1249/JES.0000000000000074
Kelm M, Schrader J (1990) Control of coronary vascular tone by nitric oxide. Circ Res 66:1561–1575. doi:10.1161/01.RES.66.6.1561
Lane SC, Hawley JA, Desbrow B, Jones AM, Blackwell JR, Ross ML, Zemski AJ, Burke LM (2014) Single and combined effects of beetroot juice and caffeine supplementation on cycling time trial performance. Appl Physiol Nutr Metab 39:1050–1057. doi:10.1139/apnm-2013-0336
Lansley KE, Winyard PG, Bailey SJ, Vanhatalo A, Wilkerson DP, Blackwell JR, Gilchrist M, Benjamin N, Jones AM (2011) Acute dietary nitrate supplementation improves cycling time trial performance. Med Sci Sports Exerc 43:1125–1131. doi:10.1249/MSS.0b013e31821597b4
Larsen FJ, Weitzberg E, Lundberg JO, Ekblom B (2007) Effects of dietary nitrate on oxygen cost during exercise. Acta Physiol 191:59–66. doi:10.1111/j.1748-1716.2007.01713.x
Larsen FJ, Schiffer TA, Borniquel S, Sahlin K, Ekblom B, Lundberg JO, Weitzberg E (2011) Dietary inorganic nitrate improves mitochondrial efficiency in humans. Cell Metab 13:149–159. doi:10.1016/j.cmet.2011.01.004
Lundberg JO, Govoni M (2004) Inorganic nitrate is a possible source for systemic generation of nitric oxide. Free Radic Biol Med 37:395–400. doi:10.1016/j.freeradbiomed.2004.04.027
Lundberg JO, Weitzberg E, Gladwin MT (2008) The nitrate–nitrite–nitric oxide pathway in physiology and therapeutics. Nat Rev Drug Discov 7:156–167. doi:10.1038/nrd2466
McMahon NF, Leveritt MD, Pavey TG (2016) The effect of dietary nitrate supplementation on endurance exercise performance in healthy adults: A systematic review and meta-analysis. Sports Med. doi:10.1007/s40279-016-0617-7
Midgley AW, McNaughton LR, Polman R, Marchant D (2007) Criteria for determination of maximal oxygen uptake: a brief critique and recommendations for future research. Sports Med 37:1019–1028
Modin A, Björne H, Herulf M, Alving K, Weitzberg E, Lundberg JO (2001) Nitrite-derived nitric oxide: a possible mediator of “acidic-metabolic” vasodilation. Acta Physiol Scand 171:9–16. doi:10.1046/j.1365-201X.2001.00771.x
Moncada S, Higgs A (1993) The L-arginine-nitric oxide pathway. N Engl J Med 329:2002–2012. doi:10.1056/NEJM199312303292706
Muggeridge DJ, Howe CCF, Spendiff O, Pedlar C, James PE, Easton C (2014) A single dose of beetroot juice enhances cycling performance in simulated altitude. Med Sci Sports Exerc 46:143–150. doi:10.1249/MSS.0b013e3182a1dc51
Nagababu E, Rifkind JM (2010) Measurement of plasma nitrite by chemiluminescence. In: Uppu RM, Murthy SN, Pryor WA, Parinandi NL (eds) Free radicals and antioxidant protocols. Humana, Totowa, pp 41–49
Pawlak-Chaouch M, Boissière J, Gamelin FX, Cuvelier G, Berthoin S, Aucouturier J (2016) Effect of dietary nitrate supplementation on metabolic rate during rest and exercise in human: a systematic review and a meta-analysis. Nitric Oxide 53:65–76. doi:10.1016/j.niox.2016.01.001
Peacock O, Tjønna AE, James P, Wisløff U, Welde B, Böhlke N, Smith A, Stokes K, Cook C, Sandbakk Ø (2012) Dietary nitrate does not enhance running performance in elite cross-country skiers. Med Sci Sports Exerc 44:2213–2219. doi:10.1249/MSS.0b013e3182640f48
Porcelli S, Ramaglia M, Bellistri G, Pavei G, Pugliese L, Montorsi M, Rasica L, Marzorati M (2014) Aerobic fitness affects the exercise performance responses to nitrate supplementation. Med Sci Sports Exerc. doi:10.1249/MSS.0000000000000577
Reid MB (1998) Role of nitric oxide in skeletal muscle: synthesis, distribution and functional importance. Acta Physiol Scand 162:401–409. doi:10.1046/j.1365-201X.1998.0303f.x
Richardson RS, Noyszewski EA, Kendrick KF, Leigh JS, Wagner PD (1995) Myoglobin O2 desaturation during exercise. Evidence of limited O2 transport. J Clin Invest 96:1916–1926
Robergs RA, Ghiasvand F, Parker D (2004) Biochemistry of exercise-induced metabolic acidosis. Am J Physiol Regul Integr Comp Physiol 287:R502–R516. doi:10.1152/ajpregu.00114.2004
Russell RD, Redmann SM, Ravussin E, Hunter GR, Larson-Meyer DE (2004) Reproducibility of endurance performance on a treadmill using a preloaded time trial. Med Sci Sports Exerc 36:717–724
Sale DG (1987) Influence of exercise and training on motor unit activation. Exerc Sport Sci Rev 15:95–151
Sandbakk SB, Sandbakk Ø, Peacock O, James P, Welde B, Stokes K, Böhlke N, Tjønna AE (2015) Effects of acute supplementation of L-arginine and nitrate on endurance and sprint performance in elite athletes. Nitric Oxide 48:10–15. doi:10.1016/j.niox.2014.10.006
Shannon O, Barlow M, Duckworth L, Woods DR, Barker T, Grindrod A, Griffiths A, O’Hara JP (2016a) The reliability of a pre-loaded treadmill time-trial in moderate normobaric hypoxia. Int J Sports Med 37:825–830. doi:10.1055/s-0042-108651
Shannon OM, Duckworth L, Barlow MJ, Woods D, Lara J, Siervo M, O’Hara JP (2016b) Dietary nitrate supplementation enhances high-intensity running performance in moderate normobaric hypoxia, independent of aerobic fitness. Nitric Oxide 59:63–70. doi:10.1016/j.niox.2016.08.001
Taylor HL, Buskirk E, Henschel A (1955) Maximal oxygen intake as an objective measure of cardio-respiratory performance. J Appl Physiol 8:73–80
Vanhatalo A, Bailey SJ, Blackwell JR, DiMenna FJ, Pavey TG, Wilkerson DP, Benjamin N, Winyard PG, Jones AM (2010) Acute and chronic effects of dietary nitrate supplementation on blood pressure and the physiological responses to moderate-intensity and incremental exercise. AJP Regul Integr Comp Physiol 299:R1121–R1131. doi:10.1152/ajpregu.00206.2010
Vanhatalo A, Fulford J, Bailey SJ, Blackwell JR, Winyard PG, Jones AM (2011) Dietary nitrate reduces muscle metabolic perturbation and improves exercise tolerance in hypoxia. J Physiol 589:5517–5528. doi:10.1113/jphysiol.2011.216341
Vassalle C, Lubrano V, Domenici C, L’Abbate A (2003) Influence of chronic aerobic exercise on microcirculatory flow and nitric oxide in humans. Int J Sports Med 24:30–35. doi:10.1055/s-2003-37202
Wilkerson DP, Hayward GM, Bailey SJ, Vanhatalo A, Blackwell JR, Jones AM (2012) Influence of acute dietary nitrate supplementation on 50 mile time trial performance in well-trained cyclists. Eur J Appl Physiol 112:4127–4134. doi:10.1007/s00421-012-2397-6
Wylie LJ, Kelly J, Bailey SJ, Blackwell JR, Skiba PF, Winyard PG, Jeukendrup AE, Vanhatalo A, Jones AM (2013) Beetroot juice and exercise: Pharmacodynamic and dose-response relationships. J Appl Physiol 115:325–336. doi:10.1152/japplphysiol.00372.2013
Acknowledgements
The authors would like to thank Rachael Bradley for assistance with data collection. We also would like to thank Long Xie for his technical support in the measurement of plasma nitrite concentrations. Finally, we would like to thank the reviewers of this paper for their detailed and insightful comments.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Communicated by Peter Krustrup.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Shannon, O.M., Barlow, M.J., Duckworth, L. et al. Dietary nitrate supplementation enhances short but not longer duration running time-trial performance. Eur J Appl Physiol 117, 775–785 (2017). https://doi.org/10.1007/s00421-017-3580-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00421-017-3580-6