Abstract
Purpose
We hypothesised that, during a light-to-moderate exercise transient, compared to an equivalent rest-to-exercise transient, (1) a further baroreflex sensitivity (BRS) decrease would be slower, (2) no rapid heart rate (HR) response would occur, and (3) the rapid cardiac output (CO) response would have a smaller amplitude (A1). Hence, we analysed the dynamics of arterial baroreflexes and the HR and CO kinetics during rest-to-50 W (0–50 W) and 50-to-100 W (50–100 W) exercise transients.
Methods
10 subjects performed three 0–50 W and three 50–100 W on a cycle ergometer. We recorded arterial blood pressure profiles (photo-plethysmography) and R-to-R interval (RRi, electrocardiography). The former were analysed to obtain beat-by-beat mean arterial pressure (MAP) and stroke volume (SV). CO was calculated as SV times HR. BRS was measured by modified sequence method.
Results
During 0–50 W, MAP transiently fell (− 9.0 ± 5.7 mmHg, p < 0.01) and BRS passed from 15.0 ± 3.7 at rest to 7.3 ± 2.4 ms mmHg−1 at 50 W (p < 0.01) promptly (first BRS sequence: 8.1 ± 4.6 ms mmHg−1, p < 0.01 vs. rest). During 50–100 W, MAP did not fall and BRS passed from 7.2 ± 2.6 at 50 W to 3.3 ± 1.3 ms mmHg−1 at 100 W (p < 0.01) slowly (first BRS sequence: 5.3 ± 3.1 ms mmHg−1, p = 0.07 vs. 50 W). A1 for HR was 9.2 ± 6.0 and 6.0 ± 4.5 min−1 in 0–50 W and 50–100 W, respectively (p = 0.19). The corresponding A1 for CO were 2.80 ± 1.54 and 0.91 ± 0.55 l∙min−1 (p < 0.01).
Conclusion
During 50–100 W, with respect to 0–50 W, BRS decreased more slowly, in absence of a prompt pressure decrease. BRS decrease and rapid HR response in 50–100 W were unexpected and ascribed to possible persistence of some vagal tone at 50 W.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Exercise requires a tight control of arterial blood pressure to provide an optimal cerebral perfusion. This control is provided by several mechanisms, the most important being the arterial baroreflex. Although the arterial baroreflex has been widely investigated during exercise (Potts et al. 1993; Papelier et al. 1994; Iellamo et al. 1997; Norton et al. 1999; Ogoh et al. 2005; Peçanha et al. 2017; Porta et al. 2018), little is known about its responses during the exercise transients. When the neck suction-neck pressure technique is applied at the exercise steady-state, a shift of the entire baroreflex logistic curve upward and rightward (baroreflex resetting) and a displacement of the operating point (OP) toward the baroreflex threshold are observed (Ogoh et al. 2005; Raven et al. 2006). When the sequence method is applied instead, a decrease of spontaneous baroreflex sensitivity (BRS) around OP appears, coherently with the observed displacement of the OP toward the baroreflex threshold with the aforementioned technique. Moreover, the baroreflex resetting appears as a displacement of the baroreflex sequences, and thus OP, toward higher values of both pressure and heart rate (HR) with respect to rest (Rowell and O’Leary 1990; Iellamo et al. 1997; Raven et al. 2006; Porta et al. 2018).
Different mechanisms have been called upon as determinants of the baroreflex alterations at exercise: a feedforward central command, and feedback loops such as the exercise pressor reflex (Ogoh et al. 2002, 2007; Fadel et al. 2003; Gallagher et al. 2006; Raven et al. 2006). More recently, Bringard et al. (Potts et al. 1993; Iellamo et al. 1997; Norton et al. 1999; Raven et al. 2006; Vallais et al. 2009; Bringard et al. 2017; Fontolliet et al. 2018) analysed the dynamics of baroreflex resetting upon exercise onset with a closed-loop approach. They found that, as exercise started, the BRS decreased almost immediately, whereas OP resetting was slower and delayed. Bringard et al. (Potts et al. 1993; Iellamo et al. 1997; Norton et al. 1999; Raven et al. 2006; Vallais et al. 2009; Bringard et al. 2017; Fontolliet et al. 2018) interpreted the former result as being an effect of sudden withdrawal of vagal tone (Fagraeus and Linnarsson 1976; Lador et al. 2006) when exercise begins, and concluded that their results were compatible with a central command mechanism. Their interpretation is coherent with the subsequent demonstration of very low BRS under parasympathetic blockade with atropine (Fontolliet et al. 2018).
The hypothesis of vagal withdrawal stemmed from experiments in which the kinetics of the rapid component (phase I) of the HR response at exercise onset was analysed (Fagraeus and Linnarsson 1976). In fact, at the very beginning of exercise, HR increases abruptly at first, the so-called phase I, and then more slowly. When the vagal activity was blocked with atropine, the phase I was completely abolished and only a slow HR increase was observed (Fagraeus and Linnarsson 1976). The vagal withdrawal hypothesis could at least partially explain also the phase I of cardiac output (CO) responses to exercise onset, i.e., the rapid initial response (Lador et al. 2006). Further results, obtained during experiments in acute hypoxia (Lador et al. 2008), under lower body negative pressure condition (Fagoni et al. 2020), and particularly during vagal blockade with atropine (Fontolliet et al. 2021), were also compatible with the hypothesis.
If the vagal withdrawal hypothesis was correct, then we would expect that during a light-to-moderate exercise transient, with respect to an equivalent rest-to-exercise transient, (1) a further BRS decrease at increasing workload would not be immediate, (2) no phase I HR response would occur, and (3) the phase I CO response would have a smaller amplitude, the size of which would be determined by the size of SV response only. The aim of the present study is to test the above hypothesis, by analysing the dynamics of arterial baroreflexes (Bringard et al. 2017) and the HR and CO kinetics (Lador et al. 2006; Fontolliet et al. 2021) during the rest-to-50 W and the 50-to-100 W exercise transients.
Materials and methods
Subjects
Ten healthy, physically active subjects were enlisted (9 males and 1 female). Age, height, and body mass were 31 ± 6 years, 174 ± 9 cm, and 69 ± 11 kg, respectively. Their maximal oxygen consumption measured during a ramp test was 3.3 ± 0.4 l min−1. None reported history of cardiovascular, pulmonary, or neurological diseases; none was taking medications or was pregnant at the time of the study. The subjects were asked to refrain from drinking coffee or from smoking for 24 h before the experiments. All subjects gave their informed consent after having received a detailed description of the methods and experimental procedures of the study. Every subject was aware of the right of withdrawing from the study at any time without jeopardy. This study was performed in line with the principles of the Declaration of Helsinki. Approval was granted by the Commission Cantonale d’Éthique de la Recherche, Canton de Genève, CH (Date 11th July 2018-No. 2018-00913).
Experimental procedure
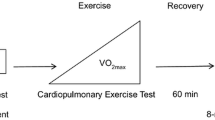
The subjects came to the laboratory on one occasion, at least 2 h after a light meal. Room temperature was 24 ± 2 °C. After instrumentation and wearing cycling shoes, the subject took place on an electromagnetically braked cycle ergometer (Lode Corival, Lode B.V., Groningen, The Netherlands). The experimental protocol comprised two sessions, one for rest to 50 W transients (0–50 W) and one for 50-to-100 W transients (50–100 W), which were administered in random order and were separated by 2–3 h, to allow resting, drinking, and a light meal or snacks. The two experimental sessions are depicted in Fig. 1. Both 0–50 W and 50–100 W sessions consisted of three trials to obtain three repetitions of the two exercise transients to be superimposed to decrease the signal-to-noise ratio.
In the 0–50 W session, after calibration of the instruments with the subject quietly resting on the cycle ergometer, 5 min of rest were recorded. At the 4th min, a 20 µl capillary blood sample was taken from an earlobe for blood lactate concentration [(La)] measurement. Afterwards, the subject started to pedal at 50 W at 60 rpm (trial 1). The subject continued to exercise for 10 min to reach and ensure at least 5 min of steady-state condition (Ferretti et al. 2017); a 20 µl capillary blood sample was taken from an earlobe for [La] measurement at the 9th min. At the end of this trial, the subject underwent further 6 min of resting recovery, followed by 5 min exercising at 50 W (trial 2). This same sequence was repeated for trial 3. At the end of trial 3, 5 min resting was allowed. During 0–50 W, the flywheel was not pre-accelerated; this usually implies that the mechanical power necessary for the flywheel acceleration may compensate for the delayed activation of the magnetic brake of the cycle ergometer (Hibi et al. 1996).
In the 50–100 W session, after calibration of the instruments with the subject quietly resting on the cycle ergometer, 5 min of rest was recorded. Then, the subject pedalled at 50 W at 60 rpm for 10 min, to ensure at least 5 min of steady state condition (Ferretti et al. 2017); a 20 µl capillary blood sample was taken from an earlobe for [La] measurement at the 9th min. Afterwards, the workload was increased to 100 W for 10 min for the same reasons explained above and at the 9th min [La] was measured (trial 1). Afterwards, the subject continued pedalling for further 6 min at 50 W, followed by 5 min at 100 W (trial 2). This same sequence was repeated for trial 3. At the end of trial 3, 5 min resting was allowed.
Measurements
Continuous non-invasive arterial blood pressure profiles were recorded at the medium finger of the left arm (Portapres, Finapres® Medical Systems, Enschede, The Netherlands). The left arm was positioned on a rigid support at the heart level. Beat-by-beat HR was recorded by electrocardiography (ECG100C module, BIOPAC® Systems Inc., Goleta, CA, USA). Applied workload and pedalling speed were continuously monitored by analogic outputs from the cycle ergometer which were digitalised (UIM100C module, BIOPAC® Systems Inc., Goleta, CA, USA) and used to provide a precise time alignment of the three repetitions of the two exercise transients. All signals were collected and sampled at 400 Hz (MP150 system with AcqKnowledge acquisition and analysis software, BIOPAC® Systems Inc., Goleta, CA, USA) and stored on a personal computer for subsequent analysis. [La] was measured by an enzymatic-amperometric method (Biosen C-Line Glucose and Lactate analyser, EKF Diagnostics, Cardiff, UK) on 20 μl capillary blood samples.
Data treatment
Arterial blood pressure profiles were analysed to obtain beat-by-beat values of systolic (SAP), diastolic (DAP), and mean (MAP) blood arterial pressure using the Beatscope® software (Finapres® Medical Systems, Enschede, The Netherlands). The same software provided a beat-by-beat calculation of the stroke volume (SV) through the Modelflow method (Wesseling et al. 1993). Beat-by-beat CO was calculated as the SV times the corresponding HR and total peripheral resistances (TPR) as the ratio between MAP and CO.
The BRS during steady states was calculated with the sequence method (Bertinieri et al. 1988) using MAP and R-to-R interval (RRi) as independent and dependent variable, respectively (Taboni et al. 2018). A phase shift of one beat between MAP and RRi was introduced (Steptoe and Vogele 1990), and then, sequences of three or more consecutive beats characterised by consensual increase or decrease in MAP and RRi were identified. Within each sequence, the relationship between RRi and MAP was analysed by linear regression to compute the slope and the coefficient of determination (R2). When R2 > 0.85, the slope was retained (Iellamo et al. 1997). In steady-state conditions, the mean slope of the RRi versus MAP relationship was considered representative of the BRS for each subject and the mean RRi and MAP value was considered as the corresponding OP.
For 0–50 W, data in steady-state conditions were computed on the last 5 min of the initial resting period and of the 10-min-lasting 50 W exercise bout. For 50–100 W, data in steady-state conditions were computed on the last 5 min of the 10-min-lasting 50 W and 100 W exercise bouts.
The baroreflex patterns during 0–50 W and 50–100 W were analysed with the same approach as previously proposed (Bringard et al. 2017). Briefly, at the very beginning of a rest-to-exercise transient, MAP decreases and successively recovers, and the minimum value during this initial MAP readjustment was retained and defined as minimum MAP. Then, the BRS was calculates as follows: when at least three consecutive beats were characterised by consensual increase or decrease of MAP and RRi, these beats were treated as baroreflex sequences on which the sequence method could be applied, as described above.
Dynamics of the CO and HR changes over time (f(t)) during the two exercise transients were analysed through bi exponential model (Barstow and Molé 1987; Lador et al. 2006)
where b is the baseline, A is the amplitude, d is the time delay between the two components, and τ is the time constant. The subscripts 1 and 2 refer to the initial (phase I) and the primary components, respectively. For every subject, the three repetitions were time aligned at exercise onset or workload increase. When the amplitude of one phase resulted equal to 0 l min−1 for CO or 0 bpm for HR, the corresponding time constant was not considered for the statistical analysis.
Statistical analysis
Data are presented as mean ± standard deviation. Student T test for paired samples was used to compare pre- with post-transient steady-state data in the two experimental conditions. One-way ANOVA for repeated measures was used to investigate differences among MAP and BRS measured at different time points during exercise transients. Tukey’s multiple comparisons test was used to isolate differences when necessary. Differences were considered significant when p < 0.05; otherwise, they were considered non-significant. The statistical software Prism (version 8, GraphPad®, La Jolla, CA, USA) was used. Data fitting with Eq. 1 was performed on the superimposition of the three repetitions for each exercise transient for each subject; MATLAB (version 9.5.0.944444 with Curve Fitting Toolbox, The MathWorks, Inc., Natick, MA, USA) was used for this aim. The resulting Eq. 1 parameters obtained in the two exercise transients were compared by Student T test.
Results
The steady-state cardiovascular parameters, BRS, and [La] are reported in Table 1. The 50 W steady-state data did not differ between the two protocols. When the 50 W steady state was compared to the corresponding resting data, all cardiovascular parameters resulted different, except for DAP. When the 100 W was compared to the corresponding 50 W steady state, all cardiovascular parameters resulted different, except for DAP and SV. Moreover, the BRS at steady state decreased with increasing the workload.
The time course of the main cardiovascular parameters is shown in Fig. 2. During 50–100 W (grey line), with respect to 0–50 W (black line), CO and HR increased less sharply (a, b, respectively), SV showed a smaller increase (c), and TPR decreased slowly and by a lesser amount (f). Moreover, during 0–50 W, SAP showed an initial notch (d) which corresponded to an MAP transient fall (e). This fall of MAP was not observed in 50–100 W.
Time course of the cardiac output (CO), heart rate (HR), stroke volume (SV), systolic and mean arterial pressure (SAP and MAP, respectively), and total peripheral resistances (TPR) during the rest to 50 W (black line) and the 50 to 100 W (grey line) exercise transients. Mean values from all subjects. The time scale represents the time elapsed since the change of the workload
Parameters obtained from the analysis of the CO kinetics are reported in Table 2. Compared to 0-to-50 W, 50–100 W was characterised by higher b, smaller A1, higher τ1, similar A2, and higher τ2. Furthermore, in 0–50 W and 50–100 W, A1 accounted for 69 ± 23% and 35 ± 19% of the total amplitude, respectively (p = 0.0022). Parameters obtained from the analysis of the HR kinetics are reported in Table 3. In this case, both A1 and A2 were similar in the two investigated transients, whereas b, τ1, and τ2 resulted higher in 50–100 W than in 0–50 W. Additionally, A1 accounted for the 55 ± 26% and the 33 ± 24% of the total amplitude in 0–50 W and 50–100 W, respectively (p = 0.0593). Of note, the wide standard deviation observed for τ1 and τ2 results from a subject, whose data are more than 2 SD far from the mean value, and thus who can be considered an outlier (see Table S2 on Supplementary material). After exclusion of this subject, τ1 and τ2 for HR turn out equal to 0.9 ± 1.0 s and 2.9 ± 2.0 s, respectively.
During 0–50 W, MAP showed a sudden decrease at the very beginning of exercise (Fig. 2e, black line). Minimum MAP was 74 ± 11 mmHg (p = 0.0002 and p < 0.0001 vs. rest and 50 W steady state, respectively) and appeared after 8.2 ± 1.9 s from exercise onset. A similar decrease was not observed during 50–100 W (Fig. 2e, grey line). This implied that the relationship between MAP and RRi resulted different in the two transients, as shown in Figs. 3a and 4a.
a Contour plot of the relationship between R-to-R interval (RRi) and mean arterial pressure (MAP) during the rest-to-50 W exercise transient. A time shift of 0.1 s was applied between MAP and RRi. Mean values from all subjects from 10 s before (dashed line) to 60 s after (continuous line) exercise onset (black arrowhead). Black dot: rest steady state; black square: 50 W steady state. b Tukey representation of the baroreflex sensitivity measured during the indicated conditions. *: significantly different vs. rest steady state (***p < 0.001; ****p < 0.0001)
a contour plot of the relationship between R-to-R interval (RRi) and mean arterial pressure (MAP) during the 50 to 100 W exercise transient. A time shift of 0.1 s was applied between MAP and RRi. Mean values from all subjects from 10 s before (dashed line) to 60 s after (continuous line) workload change (black arrowhead). Black square: 50 W steady state; black diamond: 100 W steady state. b Tukey representation of the baroreflex sensitivity measured during the indicated conditions. #: significantly different vs. 50 W steady state (###p < 0.001)
In 0–50 W, during the initial MAP decrease, also RRi decreased (Fig. 3a). During this period and before, a minimum MAP was reached, it was possible to detect a BRS sequence which appeared 4.8 ± 2.2 s after exercise onset and was 3.4 ± 0.3 beats long. The measured slope of these sequences is reported in Fig. 3b, before minimum MAP. After attainment of minimum MAP, the pattern of the RRi versus MAP relationship changed, as long as a decrease in RRi was associated with an increase in MAP. The subsequent BRS sequences were identified after this second period, when approaching the steady state. The slope of the first BRS sequence observed after minimum MAP is also reported in Fig. 3b. These sequences appeared 16.1 ± 7.3 s after exercise onset.
During 50–100 W, no significant MAP decrease was detected following the workload increase (Fig. 2e, grey line and Fig. 4a). Consequently, it was not possible to identify BRS sequences which may correspond to those found during 0–50 W. Therefore, to analyse the dynamic changes in BRS, we considered the first two BRS sequences after workload increase. The first BRS sequence was characterised by consensual decrease in MAP and RRi in 22 out of 30 exercise transients and took place within 6.4 ± 5.5 s after the workload change, and the second sequence was characterised by consensual decrease in MAP and RRi in 17 out of 30 exercise transients and appeared 17.5 ± 11.5 s after the workload change. The slopes of both these BRS sequences are reported in Fig. 4b.
Discussion
The results are in line with the tested hypothesis. As forecasted, the BRS decreased following a different pattern, and the CO kinetics had smaller A1 and slower τ1 during 50–100 W than during 0–50 W. Moreover, during 50–100 W, the HR showed a phase I; thus, it participates in the CO phase I, which had a similar A1 and a slower τ1 than during 0–50 W. This is in line with the concept that some degree of vagal activity is still present at 50 W (White and Raven 2014). We suggest that the withdrawal of the vagal tone is responsible for the HR phase I and BRS decrease both during the 0–50 W and the 50–100 W.
Cardiovascular response kinetics
The data obtained from the analysis of the kinetics of CO and HR during 0-50 W are in line with previous findings (Lador et al. 2006, 2013; Fagoni et al. 2020; Fontolliet et al. 2021), indicating that A1 of CO results from the sudden increase of both HR and SV. The A1 of HR was proposed to have a neural origin and be mediated by rapid vagal withdrawal (Fagraeus and Linnarsson 1976; Lador et al. 2006, 2008; McNarry et al. 2014; Fontolliet et al. 2018, 2021; Fagoni et al. 2020). At variance, the prompt SV increase has been hypothesised to have mechanical origin, due to (1) a displacement of blood from the periphery toward the thorax (Chung et al. 1997; Sundblad et al. 2000, 2014; Naeije and Badagliacca 2017) and (2) a sudden decrease in TPR as a consequence of a strong vasodilation in the contracting muscles (Elstad et al. 2002), as shown in Fig. 2f. Nevertheless, neural mechanisms have also been evoked in the interpretation of the SV increase, in so far as this increase is at least partially sustained by incurring sympathetic activation, which, however, may have a response time too slow to act during the phase I (Fontolliet et al. 2021). In addition, some authors have proposed that also vagal withdrawal may contribute to the SV increase (DeGeest et al. 1965; Machhada et al. 2016).
If indeed the SV response has mechanical origin, the impact of SV on the CO response in phase I may be different in the 0–50 W from the 50–100 W. In fact, the 0–50 W starts with relaxed leg muscles; hence, the abrupt initiation of the contraction–relaxation cycles determines the activation of the muscle pump that, in turn, displaces the venous blood toward the thorax and the right heart. After passing through the pulmonary circulation, the blood arrives to the left heart thus contributing to the CO increase. Moreover, starting from rest, leg exercise is capable to increase the muscle blood flow rapidly up to fourfold (MacDonald et al. 1998). Conversely, during the 50–100 W, the muscle pump is already active and the muscle capillaries are already open, so that their effect on the CO, if any, is expected to be of a lesser amount than during the 0–50 W.
During 50–100 W, both CO and HR showed an initial phase which resulted slower than during 0–50 W (Table 3). It has been recently found (Fontolliet et al. 2021) that complete vagal blockade with atropine abolishes the phase I of the HR response to a rest-to-exercise transient, but not the phase I of the CO response. This is the best and clearest evidence supporting the hypothesis that a rapid HR response may be mediated by sudden withdrawal of the vagal tone. This being the case, the presence of an A1 of HR during 50–100 W would be in line with the concept that during exercise, at least at 50 W, some degree of vagal activity is still present (White and Raven 2014), vagal withdrawal thus being incomplete. This is coherent with previous results obtained at steady state under selective β1-adrenergic blockade, demonstrating that HR progressively increases with increasing workload, despite the lack of sympathetic control of the sinus node (Ferretti et al. 2005). Moreover, previous analysis of the HR variability showed that its total power, which can be considered an indirect measure of the vagal activity (Warren et al. 1997; Montano et al. 1998; Challapalli et al. 1999; Formes et al. 2005), in control conditions at exercise steady state (80 W) was higher than under vagal blockade with atropine at rest (Fontolliet et al. 2018), further sustaining the concept of an incomplete vagal withdrawal at least in the light–moderate intensity domain.
Although during 0–50 W, both HR and SV clearly contribute to the CO phase I, the results are not so straightforward for 50–100 W. In fact, if we assume that SV does not change during the initial phase of 50–100 W, the CO asymptote of the initial phase (COI) should be equal to
From the data of the present study, COI turns out equal to 9.66 ± 1.89 l min−1. The assumption behind Eq. 2 implies that COI should correspond to the sum of baseline CO plus A1 for CO. This last is equal to 10.05 ± 2.15 l min−1 (see Table S1), a value which, although similar to COI, yet appears to be significantly different from it (p = 0.0407). In this context, the difference between these two values (~ 0.39 l min−1) would provide the portion of CO increase during the initial phase due to SV increase. However, if we adopt a different approach and calculate the percentage increase in CO during the initial phase, we can compare it to the percentage HR increase during the same phase. The former resulted to be 10 ± 6% and the latter 6 ± 5% (p = 0.1191); the difference between these two values (~ 3%), which should correspond to the percentage increase in SV during the initial phase, was not significantly different from zero. Therefore, no clear-cut conclusion can be attained concerning the lack of a role for SV as a determinant of the A1 of CO in 50–100 W. Of note, the slower kinetics of CO during 50–100 W than 0–50 W (see τ1 values in Table 2) may leave the door open to the simultaneous contribution of slower neural and/or mechanical effects to the CO increase.
Baroreflex sensitivity dynamics
During 0–50 W, MAP decreased abruptly. This was interpreted as being a consequence of the muscular vasodilation (Rådegran and Saltin 1998; Saltin et al. 1998) sustaining the increase in muscle blood flow at exercise onset (Ferretti et al. 1995; DeLorey et al. 2003; Clifford 2007; Chin et al. 2010). In fact, this MAP decrease was coupled with a fast decrease in TPR (Fig. 2f), in line with previous findings (Wieling et al. 1996; Elstad et al. 2002; Lador et al. 2006, 2008). During this MAP decrease, also RRi consensually decreased, as in a baroreflex relationship. This was interpreted as a baroreflex attempt to correct for the MAP fall at exercise onset (Bringard et al. 2017) and, indeed, we identified a long baroreflex sequence upon which it was possible to compute the BRS. As expected, the BRS measured during this initial fall in MAP was lower than that computed during the preceding resting steady state and similar to that measured during the successive 50 W steady state. This is in line with previous observations by Bringard et al. (Bringard et al. 2017), who put forward the hypothesis that the immediate fall of BRS upon exercise start may be due to vagal withdrawal. However, open-loop studies of baroreflexes at exercise steady state showed that the maximal baroreflex gain, measured around the centring point of a baroreflex logistic curve, does not change with exercise (Potts et al. 1993; Norton et al. 1999; Fadel et al. 2001; Ogoh et al. 2003, 2005; Raven et al. 2006). Nevertheless, some authors (Potts et al. 1993; Norton et al. 1999; Fadel et al. 2001; Ogoh et al. 2003, 2005; Raven et al. 2006) reported also a progressive shift of the OP toward the threshold of the open loop baroreflex relationship, which is more marked, the higher the workload. Moreover, Ogoh et al. (Ogoh et al. 2005) found that this OP shift toward the threshold at exercise is present both in control conditions and during sympathetic blockade with metoprolol. This suggests that this OP displacement is sustained by a mechanism acting through modulation of the vagal activity. Since, the BRS corresponds to the baroreflex gain around the OP, an OP shift toward the threshold, i.e., toward a flatter portion of the baroreflex curve, is coherent with the BRS decrease found in this and in a previous study (Bringard et al. 2017). This being so, both the open-loop and the closed-loop findings appear compatible with the hypothesis that vagal withdrawal is the cause of the BRS decrease at exercise. The closed-loop approach made it possible to identify that the BRS decrease upon 0–50 W occurs immediately at exercise start. Thus, we cannot exclude that a central command mechanism may be at the origin of vagal withdrawal. Moreover, since also the initial HR response is extremely rapid, and is likely sustained by vagal withdrawal (Fontolliet et al. 2021), the same mechanism may well cause both the BRS fall and the initial HR increase.
During 50–100 W, which is the most significant and novel aspect of the present study, the abrupt decrease in MAP, which occurred in 0–50 W, was not observed (Fig. 2e). This reflects a slower decrease in TPR (Fig. 2f) and, possibly, a slower regulation of the increase in muscle blood flow upon workload increase in 50–100 W than in 0–50 W. The analysis of the MAP-RRi dynamic relationship revealed a progressive shift toward the successive 100 W steady-state OP (Fig. 4a).
This analysis showed that at the time of the first sequence (6.4 ± 5.5 s after the workload change) the BRS was still similar to that computed during the previous 50 W steady state. Nonetheless, the BRS of the second sequence resulted lower than the BRS at 50 W and similar to that at 100 W steady state. This suggests that the BRS adjustment was slower in this case than during 0–50 W. If the decrease of the BRS around the OP is due to vagal withdrawal, then a further withdrawal of vagal tone may determine (1) the decrease in BRS and (2) the HR phase I response to workload increase also in 50–100 W, yet with slower kinetics.
Operating point dynamics
In addition to the shift of the OP toward the threshold, as previously discussed, when passing from rest to exercise and with increasing workload, the open-loop analysis also showed that the entire baroreflex relationship moves toward higher values of blood pressure and lower values of RRi (Potts and Raven 1995; Ogoh et al. 2005; Raven et al. 2006). This last baroreflex relationship displacement was defined as baroreflex resetting. Coherently, in the present closed-loop analysis, the OP measured at steady state (Table 1) was characterised by higher MAP values and lower RRi values at 50 W than at rest and at 100 W than at 50 W, indicating progressive displacement of the entire baroreflex relationship downward and leftward. However, at variance with the previously cited studies, we were also able to observe the dynamic displacement of the OP during the two investigated transients, which is precluded with open-loop approach.
During 0–50 W, the OP showed a two-phase movement (Fig. 3a): (1) a decrease in MAP and RRi following a baroreflex relationship until the minimum MAP value was reached, followed by (2) a continuous decrease in RRi coupled with an increase in MAP. The former started at exercise onset and finished at the minimum MAP value, thus lasting 7.9 ± 3.5 s, the latter started at minimum MAP and ended when approaching the 50 W steady state. Our interpretation is that the first phase of the OP displacement may reflect the shift of the OP toward the threshold of the resting baroreflex curve, whereas the second phase may represent the resetting of the entire baroreflex response curve.
During 50–100 W, a baroreflex resetting took place, since both MAP and RRi were, respectively, higher and lower at 100 W than at 50 W steady states (Table 1). However, the OP did not show a two-phase pattern as during its 0–50 W counterpart (Fig. 4a). The baroreflex resetting, in fact, appeared as a progressive displacement of the OP from the 50 W to the 100 W steady state. We propose that this is a consequence of the lack of the sudden large fall in TPR in 50–100 W, and thus of the lack of sudden MAP fall at the beginning of the exercise transient.
Methodological consideration
In the investigation of the arterial baroreflex, two approaches can be adopted: the open-loop and the closed-loop approaches. In both cases, the HR or blood pressure response (output) to some stimuli capable to activate the arterial baroreflex (input) is recorded and analysed. However, the techniques adopting an open-loop approach assume that any output cannot serve as an input, whereas those adopting a closed-loop approach accept that any output, in terms of HR or blood pressure, is capable to influence the arterial baroreflex and hence to serve as an input.
The currently available open-loop methods, e.g., the neck suction-neck pressure techniques or the use of vasoactive drugs, may provide a more complete description of the baroreflex response both at rest and at exercise. However, their application requires strict steady-state conditions. Therefore, they cannot be applied during the exercise transients. Hence, if we would like to investigate the dynamics of baroreflex readjustments upon exercise start, we are obliged to use a closed-loop approach. Previous literature showed that a modified sequence method can be utilised to this aim (Bringard et al. 2017; Taboni et al. 2021b, a).
Conclusions
For the first time, the dynamics of the baroreflex adjustments during an exercise transient from light-to-moderate intensity domain have been investigated and compared to a rest-to-light exercise transient. The results showed that during the exercise-to-exercise, with respect to the rest-to-exercise transient, the spontaneous baroreflex sensitivity decreased more slowly and the operating point displacement was not characterised by an initial prompt pressure decrease. Moreover, data suggest that minimal vagal activity is still present in the light intensity domain, and that the withdrawal of the vagal tone could be responsible for the further decrease of the baroreflex sensitivity around the operating point in the transition from light to moderate exercise. The same mechanism may explain also the further rapid increase of heart rate.
Overall, this study represents a step forward in understanding how the arterial baroreflex readjusts to exercise. This understanding appears crucial to explain the exercise intolerance in those conditions characterised by arterial baroreflex impairment, as in spinal cord injuries, Parkinson’s disease, or hypertension. In this context, this study set the basis for further investigations aimed at developing countermeasures to exercise and orthostatic intolerance in those patients coping with blood pressure dysregulation.
Data availability
The datasets generated and analysed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- 0–50 W:
-
Rest-to-50 W exercise transients
- 50–100 W:
-
50-to-100 W exercise transients
- A 1 :
-
Amplitude of the initial phase of the cardiovascular responses
- A 2 :
-
Amplitude of the primary phase of the cardiovascular responses
- b :
-
Baseline
- BRS:
-
Spontaneous baroreflex sensitivity
- CO:
-
Cardiac output
- COI :
-
Cardiac output asymptote of the initial phase
- d :
-
Time delay between the initial and the primary phases of the cardiovascular responses
- DAP:
-
Diastolic arterial pressure
- HR:
-
Heart rate
- MAP:
-
Mean arterial pressure
- OP:
-
Operating point
- RRi:
-
R-to-R interval
- SAP:
-
Systolic arterial pressure
- SV:
-
Stroke volume
- TPR:
-
Total peripheral resistances
- τ1 :
-
Time constant of the initial phase of the cardiovascular responses
- τ2 :
-
Time constant of the primary phase of the cardiovascular responses
- [La]:
-
Blood lactate concentration
References
Barstow TJ, Molé PA (1987) Simulation of pulmonary O2 uptake during exercise transients in humans. J Appl Physiol 63:2253–2261. https://doi.org/10.1152/jappl.1987.63.6.2253
Bertinieri G, Di Rienzo M, Cavallazzi A et al (1988) Evaluation of baroreceptor reflex by blood pressure monitoring in unanesthetized cats. Am J Physiol 254:H377–H383. https://doi.org/10.1152/ajpheart.1988.254.2.H377
Bringard A, Adami A, Fagoni N et al (2017) Dynamics of the RR-interval versus blood pressure relationship at exercise onset in humans. Eur J Appl Physiol 117:619–630. https://doi.org/10.1007/s00421-017-3564-6
Challapalli S, Kadish AH, Horvath G, Goldberger JJ (1999) Differential effects of parasympathetic blockade and parasympathetic withdrawal on heart rate variability. J Cardiovasc Electrophysiol 10:1192–1199. https://doi.org/10.1111/j.1540-8167.1999.tb00295.x
Chin LMK, Heigenhauser GJF, Paterson DH, Kowalchuk JM (2010) Pulmonary O2 uptake and leg blood flow kinetics during moderate exercise are slowed by hyperventilation-induced hypocapnic alkalosis. J Appl Physiol 108:1641–1650. https://doi.org/10.1152/japplphysiol.01346.2009
Chung DC, Niranjan SC, Clark JW et al (1997) A dynamic model of ventricular interaction and pericardial influence. Am J Physiol-Heart Circ Physiol 272:H2942–H2962. https://doi.org/10.1152/ajpheart.1997.272.6.h2942
Clifford PS (2007) Skeletal muscle vasodilatation at the onset of exercise. J Physiol 583:825–833
DeGeest H, Levy MN, Zieske H, Lipman RI (1965) Depression of ventricular contractility by stimulation of the vagus nerves. Circ Res 17:222–235. https://doi.org/10.1161/01.RES.17.3.222
DeLorey DS, Kowalchuk JM, Paterson DH (2003) Relationship between pulmonary O2 uptake kinetics and muscle deoxygenation during moderate-intensity exercise. J Appl Physiol 95:113–120. https://doi.org/10.1152/japplphysiol.00956.2002
Elstad M, Toska K, Walløe L (2002) Model simulations of cardiovascular changes at the onset of moderate exercise in humans. J Physiol 543:719–728. https://doi.org/10.1113/jphysiol.2002.019422
Fadel PJ, Ogoh S, Watenpaugh DE et al (2001) Carotid baroreflex regulation of sympathetic nerve activity during dynamic exercise in humans. Am J Physiol-Heart Circ Physiol 280:H1383–H1390. https://doi.org/10.1152/ajpheart.2001.280.3.H1383
Fadel PJ, Strømstad M, Wray DW et al (2003) New insights into differential baroreflex control of heart rate in humans. Am J Physiol-Heart Circ Physiol 284:H735–H743. https://doi.org/10.1152/ajpheart.00246.2002
Fagoni N, Bruseghini P, Adami A et al (2020) Effect of lower body negative pressure on phase I cardiovascular responses at exercise onset. Int J Sports Med 41:209–218. https://doi.org/10.1055/a-1028-7496
Fagraeus L, Linnarsson D (1976) Autonomic origin of heart rate fluctuations at the onset of muscular exercise. J Appl Physiol 40:679–682. https://doi.org/10.1152/jappl.1976.40.5.679
Ferretti G, Binzoni T, Hulo N et al (1995) Kinetics of oxygen consumption during maximal exercise at different muscle temperatures. Respir Physiol 102:261–268. https://doi.org/10.1016/0034-5687(95)00071-2
Ferretti G, Fagoni N, Taboni A et al (2017) The physiology of submaximal exercise: the steady state concept. Respir Physiol Neurobiol 246:76–85. https://doi.org/10.1016/j.resp.2017.08.005
Ferretti G, Licker MJ, Anchisi S et al (2005) The effects of β1-adrenergic blockade on cardiovascular oxygen flow in normoxic and hypoxic humans at exercise. Eur J Appl Physiol 95:250–259. https://doi.org/10.1007/s00421-005-1393-5
Fontolliet T, Bringard A, Adami A et al (2021) Vagal blockade suppresses the phase I heart rate response but not the phase I cardiac output response at exercise onset in humans. Eur J Appl Physiol 121:3173–3187. https://doi.org/10.1007/s00421-021-04769-3
Fontolliet T, Pichot V, Bringard A et al (2018) Testing the vagal withdrawal hypothesis during light exercise under autonomic blockade: a heart rate variability study. J Appl Physiol 125:1804–1811. https://doi.org/10.1152/japplphysiol.00619.2018
Formes KJ, Wray DW, O-Yurvati AH, et al (2005) Sympathetic cardiac influence and arterial blood pressure instability. Auton Neurosci 118:116–124. https://doi.org/10.1016/j.autneu.2005.01.002
Gallagher KM, Fadel PJ, Smith SA et al (2006) The interaction of central command and the exercise pressor reflex in mediating baroreflex resetting during exercise in humans. Exp Physiol 91:79–87. https://doi.org/10.1113/expphysiol.2005.032110
Hibi N, Fujinaga H, Ishii K (1996) Work and power outputs determined from pedalling and flywheel friction forces during brief maximal exertion on a cycle ergometer. Eur J Appl Physiol 74:435–442. https://doi.org/10.1007/BF02337724
Iellamo F, Legramante JM, Raimondi G, Peruzzi G (1997) Baroreflex control of sinus node during dynamic exercise in humans: effects of central command and muscle reflexes. Am J Physiol-Heart Circ Physiol 272:H1157–H1164. https://doi.org/10.1152/ajpheart.1997.272.3.H1157
Lador F, Azabji Kenfack M, Moia C et al (2006) Simultaneous determination of the kinetics of cardiac output, systemic O2 delivery, and lung O2 uptake at exercise onset in men. Am J Physiol-Regul Integr Comp Physiol 290:R1071–R1079. https://doi.org/10.1152/ajpregu.00366.2005
Lador F, Tam E, Adami A et al (2013) Cardiac output, O2 delivery and VO2 kinetics during step exercise in acute normobaric hypoxia. Respir Physiol Neurobiol 186:206–213. https://doi.org/10.1016/J.RESP.2013.01.017
Lador F, Tam E, Azabji Kenfack M et al (2008) Phase I dynamics of cardiac output, systemic O2 delivery, and lung O2 uptake at exercise onset in men in acute normobaric hypoxia. Am J Physiol-Regul Integr Comp Physiol 295:R624–R632. https://doi.org/10.1152/ajpregu.00797.2007
MacDonald MJ, Shoemaker JK, Tschakovsky ME, Hughson RL (1998) Alveolar oxygen uptake and femoral artery blood flow dynamics in upright and supine leg exercise in humans. J Appl Physiol 85:1622–1628. https://doi.org/10.1152/jappl.1998.85.5.1622
Machhada A, Marina N, Korsak A et al (2016) Origins of the vagal drive controlling left ventricular contractility. J Physiol 594:4017–4030. https://doi.org/10.1113/JP270984
McNarry MA, Kingsley MIC, Lewis MJ (2014) Relationship between changes in pulmonary VO2kinetics and autonomic regulation of blood flow. Scand J Med Sci Sports 24:613–621. https://doi.org/10.1111/sms.12041
Montano N, Cogliati C, Porta A et al (1998) Central vagotonic effects of atropine modulate spectral oscillations of sympathetic nerve activity. Circulation 98:1394–1399. https://doi.org/10.1161/01.CIR.98.14.1394
Naeije R, Badagliacca R (2017) The overloaded right heart and ventricular interdependence. Cardiovasc Res 113:1474–1485
Norton KH, Boushel R, Strange S et al (1999) Resetting of the carotid arterial baroreflex during dynamic exercise in humans. J Appl Physiol 87:332–338. https://doi.org/10.1152/jappl.1999.87.1.332
Ogoh S, Fadel PJ, Nissen P et al (2003) Baroreflex-mediated changes in cardiac output and vascular conductance in response to alterations in carotid sinus pressure during exercise in humans. J Physiol 550:317–324. https://doi.org/10.1113/jphysiol.2003.041517
Ogoh S, Fisher JP, Dawson EA et al (2005) Autonomic nervous system influence on arterial baroreflex control of heart rate during exercise in humans. J Physiol 566:599–611. https://doi.org/10.1113/jphysiol.2005.084541
Ogoh S, Fisher JP, Raven PB, Fadel PJ (2007) Arterial baroreflex control of muscle sympathetic nerve activity in the transition from rest to steady-state dynamic exercise in humans. Am J Physiol Heart Circ Physiol 293:H2202–H2209. https://doi.org/10.1152/ajpheart.00708.2007
Ogoh S, Wasmund WL, Keller DM et al (2002) Role of central command in carotid baroreflex resetting in humans during static exercise. J Physiol 543:349–364
Papelier Y, Escourrou P, Gauthier JP, Rowell LB (1994) Carotid baroreflex control of blood pressure and heart rate in men during dynamic exercise. J Appl Physiol 77:502–506. https://doi.org/10.1152/jappl.1994.77.2.502
Peçanha T, Forjaz CLM, DavidA L (2017) Additive effects of heating and exercise on baroreflex control of heart rate in healthy males. J Appl Physiol 123:1555–1562. https://doi.org/10.1152/japplphysiol.00502.2017
Porta A, Bari V, de Maria B et al (2018) Peripheral resistance baroreflex during incremental bicycle ergometer exercise: characterization and correlation with cardiac baroreflex. Front Physiol. https://doi.org/10.3389/fphys.2018.00688
Potts JT, Raven PB (1995) Effect of dynamic exercise on human carotid-cardiac baroreflex latency. Am J Physiol-Heart Circ Physiol 268:H1208–H1214. https://doi.org/10.1152/ajpheart.1995.268.3.h1208
Potts JT, Shi XR, Raven PB (1993) Carotid baroreflex responsiveness during dynamic exercise in humans. Am J Physiol 265:H1928–H1938
Rådegran G, Saltin B (1998) Muscle blood flow at onset of dynamic exercise in humans. Am J Physiol-Heart Circ Physiol 274:H314–H322. https://doi.org/10.1152/ajpheart.1998.274.1.h314
Raven PB, Fadel PJ, Ogoh S (2006) Arterial baroreflex resetting during exercise: a current perspective. Exp Physiol 91:37–49. https://doi.org/10.1113/expphysiol.2005.032250
Rowell LB, O’Leary DS (1990) Reflex control of the circulation during exercise: chemoreflexes and mechanoreflexes. J Appl Physiol 69:407–418. https://doi.org/10.1152/jappl.1990.69.2.407
Saltin B, Rådegran G, Koskolou MD, Roach RC (1998) Skeletal muscle blood flow in humans and its regulation during exercise. Acta Physiol Scand 162:421–436
Steptoe A, Vogele C (1990) Cardiac baroreflex function during postural change assessed using non-invasive spontaneous sequence analysis in young men. Cardiovasc Res 24:627–632. https://doi.org/10.1093/cvr/24.8.627
Sundblad P, Haruna Y, Tedner B, Linnarsson D (2000) Short-term cardiovascular responses to rapid whole-body tilting during exercise. Eur J Appl Physiol 81:259–270. https://doi.org/10.1007/s004210050041
Sundblad P, Spaak J, Kaijser L (2014) Time courses of central hemodynamics during rapid changes in posture. J Appl Physiol 116:1182–1188. https://doi.org/10.1152/japplphysiol.00690.2013
Taboni A, Fagoni N, Fontolliet T et al (2021a) A closed-loop approach to the study of the baroreflex dynamics during posture changes at rest and at exercise in humans. Am J Physiol-Regul Integr Comp Physiol 321:R960–R968. https://doi.org/10.1152/ajpregu.00167.2021
Taboni A, Fagoni N, Vinetti G, Ferretti G (2018) Baroreflex sensitivity: an algebraic dilemma. Acta Physiol (oxf). https://doi.org/10.1111/apha.12979
Taboni A, Vinetti G, Fontolliet T et al (2021b) Baroreflex responses during dry resting and exercise apnoeas in air and pure oxygen. Eur J Appl Physiol 121:539–547. https://doi.org/10.1007/s00421-020-04544-w
Vallais F, Baselli G, Lucini D et al (2009) Spontaneous baroreflex sensitivity estimates during graded bicycle exercise: a comparative study. Physiol Meas 30:201–213. https://doi.org/10.1088/0967-3334/30/2/007
Warren JH, Jaffe RS, Wraa CE, Stebbins CL (1997) Effect of autonomic blockade on power spectrum of heart rate variability during exercise. Am J Physiol-Regul Integr Comp Physiol 273:R495–R502. https://doi.org/10.1152/ajpregu.1997.273.2.r495
Wesseling KH, Jansen JR, Settels JJ, Schreuder JJ (1993) Computation of aortic flow from pressure in humans using a nonlinear, three-element model. J Appl Physiol 74:2566–2573. https://doi.org/10.1152/jappl.1993.74.5.2566
White DW, Raven PB (2014) Autonomic neural control of heart rate during dynamic exercise: revisited. J Physiol 592:2491–2500. https://doi.org/10.1113/jphysiol.2014.271858
Wieling W, Harms MPM, ten Harkel ADJ et al (1996) Circulatory response evoked by a 3 s bout of dynamic leg exercise in humans. J Physiol 494:601–611. https://doi.org/10.1113/jphysiol.1996.sp021518
Acknowledgements
The authors thank all the subjects who participated in this study
Funding
Open access funding provided by Università degli Studi di Brescia within the CRUI-CARE Agreement. This study was supported by Swiss National Science Foundation, Berne, CH (Grant numbers 32003B_179448) to Guido Ferretti.
Author information
Authors and Affiliations
Contributions
GF conceived and designed the research. AT, NF, and TF conducted the experiments. AT and GV analysed data. AT wrote the manuscript. All authors read and approved the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Ethical approval
This study was performed in line with the principles of the Declaration of Helsinki. Approval was granted by the Commission Cantonale d’Éthique de la Recherche, Canton de Genève, CH (Date 11th July 2018-No. 2018-00913).
Consent to participate
All subjects gave their written informed consent after having received a detailed description of the methods and experimental procedures of the study.
Consent for publication
Informed consent was obtained from all participants included in the study.
Additional information
Communicated by Ellen Adele Dawson.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Taboni, A., Fagoni, N., Fontolliet, T. et al. Dynamics of cardiovascular and baroreflex readjustments during a light-to-moderate exercise transient in humans. Eur J Appl Physiol 122, 2343–2354 (2022). https://doi.org/10.1007/s00421-022-05011-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00421-022-05011-4