Abstract
Background
Non-small cell lung cancer (NSCLC) is the most common type of lung cancer (LC), which is the leading cause of tumor mortality. In recent years, compared with tissue biopsy, which is the diagnostic gold standard for tumor diagnosis, Liquid biopsy (LB) is considered to be a more minimally invasive, sensitive, and safer alternative or auxiliary diagnostic method. However, the current value of LB in early diagnosis of LC is not ideal, so it is particularly important to study the changes in blood composition during the process of tumorigenesis and find more sensitive biomarkers.
Purpose
Platelets are a type of abundant blood cells that carry a large amount of RNA. In the LC regulatory network, activated platelets play an important role in the process of tumorigenesis, development, and metastasis. In order to identify predictive liquid biopsy biomarkers for the diagnosis of NSCLC, we summarized the development and function of platelets, the interaction between platelets and tumors, the value of TEP RNA in diagnosis, prognosis, and treatment of NSCLC, and the method for detecting TEP RNA of NSCLC in this article.
Conclusion
The application of platelets in the diagnosis and treatment of NSCLC remains at a nascent stage. In addition to the drawbacks of low platelet count and complex experimental processes, the diagnostic accuracy of TEP RNA-seq for cancer in different populations still needs to be improved and validated. At present, a large number of studies have confirmed significant differences in the expression of TEP RNA in platelets between NSCLC patients and healthy individuals. Continuous exploration of the diagnostic value of TEP RNA in NSCLC is of utmost importance. The integration of NSCLC platelet-related markers with other NSCLC markers can improve current tumor diagnosis and prognostic evaluation systems, providing broad prospects in tumor screening, disease monitoring, and prognosis assessment.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Background of non-small cell lung cancer
Lung cancer is the leading cause of cancer death worldwide, with non-small cell lung cancer (NSCLC) accounting for more than 80% of all lung cancers (Siegel et al. 2023). Although significant progress has been made in the treatment of lung cancer in recent years, the five-year survival rate of this disease is still not high, mainly due to the lack of effective early diagnostic methods. At present, the gold standard for tumor diagnosis is still tissue biopsy and pathologic diagnosis. However, it has been found that many patients refuse to undergo puncture surgery. Therefore, this method has significant limitations. Low-dose computed tomography (LDCT), as a screening diagnostic method for lung cancer, has been widely used in clinical practice; however, it has drawbacks such as high false-positive rates and radiation exposure (National Lung Screening Trial Research Team et al. 2011). Therefore, finding highly sensitive and specific tumor markers for early diagnosis, guiding treatment, and evaluating curative effects and prognosis is the key method to overcome malignant tumors and solve clinical diagnosis and treatment problems.
Current status of liquid biopsy
Liquid biopsy, as a new kind of diagnostic technique, has shown great potential in the field of tumor diagnosis in recent years (Cagle et al. 2013). It is minimally invasive, rapid, and highly sensitive and accepted by most patients. With the rapid development of LB technology, the detection of tumor markers has shifted from static detection to dynamic monitoring, and the research content and direction are gradually enriched. LB samples include blood, urine, chest and abdominal fluid, and cerebrospinal fluid, with blood being most commonly used in clinical practice. LB technology can be used not only to predict diseases, but also to detect efficacy, cancer types, evaluate side effects, understand tumor heterogeneity, and find the correct treatment methods (Fig. 1b). The detection objects of LB mainly include circulating tumor DNA (ctDNA)/circulating free DNA (cfDNA), circulating tumor cells (CTCs), extracellular vesicle (EV) released into the bloodstream from the primary tumor and metastatic sites, RNA, proteins and others (Fig. 1a) (Nikanjam et al. 2022). The expression of platelet-derived growth factor and transforming growth factor gene in lung cancer cells has been studied since 1988 (Söderdahl et al. 1988; Heldin et al. 1993). However, the current value of LB in early diagnosis of LC is not ideal, so it is particularly important to study the changes in blood composition during the process of tumorigenesis and find more sensitive biomarkers.
The classification of a sample types, test items of liquid biopsy and b clinical applications of liquid biopsy in tumor. Liquid biopsy (LB) is applied in clinical treatment and diagnosis by collecting patients’ blood, urine, pleural and ascitic fluids, as well as cerebrospinal fluid, and detecting their ctDNA/cfDNA, RNA, proteins, methylation products, EVs, and CTCs. LB can be applied to efficacy monitoring, disease prognosis, early cancer detection, monitoring of side effects after surgery or drug treatment, evaluating difficult to biopsy cancers, understanding tumor heterogeneity, and seeking treatment options. Created with BioRender.com
Tumor-educated platelets (TEPs) as important biological sources for liquid biopsy
Platelets, as a type of abundant blood cells, play important roles in many physiological and pathological processes, including tumorigenesis. New therapeutic targets related to platelet function can be studied through platelet proteomics (Zufferey et al. 2012; Morris et al. 2022). Platelets are composed of fragments produced by megakaryocytes in the bone marrow. In the circulatory system, they function as highly reactive and secretory cell after produced by megakaryocytes for 5–7 days. In addition to its functions of hemostasis, thrombosis and innate immunity, platelets are also involved in the growth, extravasation, and metastasis of tumor cells, and have a significant impact on the treatment of tumor (Holinstat 2017; Haemmerle et al. 2018).
Furthermore, as observed in physical tests, the parameters of platelets can be affected by tumors. Platelet counts, which often tend to decrease during initial treatment and increase during relapse, reflect poor prognosis in solid tumors (Lin et al. 2014). For example, preoperative thrombocytosis is strongly associated with decreased survival rates in epithelial ovarian cancer (Hufnagel et al. 2020). By meta-analysis, Li et al. discovered a correlation between elevated platelet-to-lymphocyte ratio (PLR) and decreased overall survival (OS) or progression-free survival (PFS) among patients diagnosed with advanced cancer (Li et al. 2018). PLR can be used as a prognostic predictor of non-small cell lung cancer according to the study conducted by Liu et al. (Liu et al. 2019a). Recently, Li et al. undertook a retrospective analysis to investigate mean platelet volume (MPV), platelet count, survival rate, and risk of immune-related adverse events (irAEs) in patients treated with immune checkpoint inhibitors (ICIs). The results indicate a significant correlation between the alteration in MPV following one cycle of pembrolizumab treatment and both the overall survival and occurrence of irAEs in NSCLC patients (Li et al. 2023). MPV can also serve as a convenient prognostic diagnostic marker for locally advanced non-small cell lung cancer (Pyo et al. 2016; Sakin et al. 2019). Some scholars believe that there is an increase in MPV among patients with early-stage lung cancer, whereas it decreases in patients with advanced lung cancer (Sabrkhany et al. 2017; Goksel et al. 2021). Nevertheless, several scholars have discovered that the MPV among individuals diagnosed with lung cancer is comparatively lower than that observed in individuals who are unaffected by the disease (Şahin and Aslan 2018). The study discrepancy may be attributed to variances in cancer type and patient numbers across different stages. However, these parameters are influenced by additional inflammations. There is a requirement for an exploration aimed at identifying diagnostic markers that exhibit a higher degree of specificity.
The biogenesis and function of the TEP RNA
Platelets are non-nucleated and abundant cellular debris derived from megakaryocytes (MK) of bone marrow stem cells, participating in the process of hemostasis, promoting angiogenesis, and inducing inflammation and immune response. The production of platelets is driven by the interaction of several transcription factors (Grozovsky et al. 2015). When the number of platelets in the blood is low, megakaryocytes are activated. Thrombopoietin (TPO) produced by the liver induces platelets formation by stimulating the thrombopoietin receptor (TpoR/MPL) in megakaryocytes. In reactive thrombopoiesis, IL-6 promotes proplatelets formation by increasing TPO level (Kaser et al. 2001). Even in the absence of nuclear structures, platelets contain various RNAs which undergo changes when MK become proplatelet cells. In this process, RNA-binding protein (RBP) plays important roles (Singh 2021). The RNA-binding protein SRSF3 distributes MK RNA into platelets and mediates RNA processing for MK gene regulation and platelet biogenesis (Heazlewood et al. 2022). Platelets have no nuclei, no DNA, and are rich in coding RNA (mRNA) and non-coding RNA (ncRNA). Among them, mRNA, miRNA, circulating RNA, and long-strand RNA are often used in tumor diagnosis and treatment. Studies have shown that RNA can be transferred to hepatocytes, vascular endothelial cells, macrophages, and tumor cells through platelets, and the integrated RNA can be translated into protein (Xia et al. 2018). TEP miRNA regulates gene expression, leading to changes in receptor cell function (Tran et al. 2020). In addition, some studies have confirmed that changes in platelet miRNA expression can affect platelet aggregation, antibacterial ability, and endothelial cell function (Qu et al. 2020). In the tumor microenvironment, platelets act as highly dynamic cells that communicate with tumor-related RNA transfer and influence the environment (D’Ambrosi et al. 2021a).
In this article, we will introduce the application of TEPs with tumor high driving phenotype in the diagnosis of NSCLC. It mainly includes the following aspects: (1) the historical interactions between platelets and tumors; (2) the clinical applications of TEP RNA in tumors; (3) the diagnosis, prognosis and treatment of NSCLC and TEP RNA; and (4) the method for detecting TEP RNA of NSCLC.
The historical interplay between platelets and cancer
The role of TEPs in tumors
TEPs play direct and indirect roles in the occurrence, development, and metastasis of tumors (Fig. 2). Firstly, platelets are activated and directly interact upon encounter with circulating tumor cells (CTC). Then activated platelets aggregate around tumor cells, forming platelet–tumor cell complexes to evade immune system clearance. After determining the relationship between platelets and cancer cells by Dyngo4a treatment and flow cytometry detection, the researchers found that cancer cells could uptake platelets by phagocytosis and recycling platelet membrane proteins (Martins Castanheira et al. 2022).
Interactions and mechanisms between platelets and tumor cells in tumor patients. Platelets are composed of fragments of megakaryocytes, and can be activated and undergo TCIPA under the action of GPCR and CTC. On the one hand, ATP, TGF-β, LPA, GP IV, and NETs released by activated platelets can act on the tumor microenvironment and affect tumor metastasis. On the other hand, activated platelets release GITRL, MHC1, GARP, TGF-β, PF4, and CTAP-III which participate in the occurrence of tumor-related inflammation and immune escape of tumors. The high expression of CLEC2 in activated platelets affects the synthesis of podophyllotoxin and thrombosis. Created with BioRender.com
Second, activated platelets also release various factors, indirectly promoting tumor growth, angiogenesis, metastasis, and immune escape (Mezouar et al. 2014; Schlesinger 2018; Liu et al. 2021). Platelet factor 4 (PF-4/CXCL4) and connective tissue-activating peptide III (CTAP-III) are two platelet-related chemokines which regulate tumor angiogenesis and inflammation in the tumor microenvironment, thereby regulating tumor growth (Pilatova et al. 2013). Research has found that CLEC-2 is almost specifically expressed in human platelets/megakaryocytes. In B16F10 cells, CLEC-2 regulates the expression of podophyllotoxin and interacts with it, thereby regulating the formation of cancer-related thrombosis (Suzuki-Inoue 2019). Platelet TLR4 and ERK5 expression is significantly increased after surgical stress, and the platelet TLR4-ERK5 axis captures CTCs by releasing neutrophil extracellular traps (NETs) and promotes the distant metastasis of CTCs (Ren et al. 2021). Thyroid hormone T4, as a pro-metastasis factor, is a major ligand for the hormone receptor on tumor cells and platelet integrin αvβ3. As T4 activates integrins to promote platelet aggregation and degranulation, platelet ATP is released and promotes cell proliferation, invasion and metastasis (Davis et al. 2020). Furthermore, Platelet-derived ATP can also promote cancer cell metastasis through the P2Y2 receptor (Schumacher et al. 2013). Researchers found that platelet-derived TGF-β contacts with platelet-tumor cells and co-activates the TGF-β/Smad and NF-κB pathways in CTCs, leading to the transition to an invasive mesenchymal-like phenotype and enhancing the metastasis of cancer cells in vivo (Labelle et al. 2011). Platelet glycoprotein VI can also promote metastasis through interacting with cancer cell-derived galectin-3 (Mammadova-Bach et al. 2020). Therefore, it can be seen that we may explore the development of cancer cells by studying the proteome, transcriptome, and secretome of platelets ingested by cancer cells.
Finally, factors released by platelets not only promote the survival and migration of tumor cells in the bloodstream, but also provide a protective barrier for tumor cells to prevent immune surveillance. By generating GITRL, MHC1, GARP and TGF-β, TEPs can reduce the immunogenicity, immunoreactivity, and toxicity of NK cells, hence avoiding immune surveillance (Kataki et al. 2002; Placke et al. 2012; Li et al. 2022a).
The application of platelets in tumors is receiving increasing attention. Researchers have found that tumor-associated platelets (TAP) which block nanoparticles can alleviate cancer metastasis (Li et al. 2022b). Biomimetic nano-NOS mediated local NO can inhibit TEPs activation, destroy tumor’s vascular barrier, and exert anticancer effect (Ma et al. 2020). The TLR agonist, resiquimod (R848), enhances anti-tumor immunity in solid tumors through local delivery of platelet membrane-coated nanoparticles (PNP-R848) (Bahmani et al. 2021). TEPs has been proposed as a potential target for reducing the incidence rate, metastasis, and mortality of cancer (Mezouar et al. 2016).
The role of CTCs on TEPs
Platelets are one of the most abundant cells in the blood circulation, second only to red blood cells, and are known as “circulatory sentinels”. They absorb and store a large number of proteins, cytokines, and specific receptors during circulation (Tran et al. 2020). These “platelet reservoirs” affect platelet count, function, RNA, and proteome changes.
During the development and metastasis of cancer, platelets are activated and functioning. First of all, tumor cells can alter the characteristics of platelets in turn. For example, the predictive significance of platelet counts and platelet lymphocyte ratio (PLR) in cancer diagnosis has been fully confirmed (Li et al. 2018). In addition, with changes in the tumor microenvironment, cancer also affects platelet RNA and protein profiles through changes in MK (Stone et al. 2012). In the study of tumor and platelet proteome, we find that six proteins (RNF100, CTSG, PGLYRP8, RPL100, S9A1, S1A432) are up-regulated, and two proteins (GPX1, TNS1) are downregulated in cancer patients (Walraven et al. 2021). Numerous DEG and bioinformatics analyses have confirmed that platelet RNA expression profiles play an important role in lung cancer metastasis and can serve as a mechanism for further research on tumor metastasis.
Driven by cancer-related proteins and cytokines, highly sensitive platelets act as the "first responders" of cancer metastasis, constantly responding to the occurrence and development of tumors. Tumor host angiogenic regulatory proteins can be selectively absorbed by platelets and participate in the regulation of platelet angiogenesis (Klement et al. 2009). Immunosuppressive cells, especially tumor-associated macrophages (TAMs), exist in the tumor microenvironment (TME). The consumption of TAMs enhances local and systemic platelet-mediated anti-PD-1 delivery effects by affecting platelet activation and aggregation, thereby inhibiting post-operative tumor recurrence. In addition to numerical changes, tumors also induce platelet dysfunction, thereby promoting thrombosis, which is known as tumor cell-induced platelet aggregation (TCIPA). During the TCIPA process, platelets are "educated" by tumor cells, further promoting tumor progression. Researchers showed that that adhesion to tumor cells expressing GPCR (CD97) receptors can promote the activation of platelet and the release of lysophosphatidic acid (LPA). LPA increases the invasive potential and vascular permeability of tumor cells, promoting tumor development through endothelial cells migration (Ward et al. 2018). By constructing a mouse model of in situ cancer (injection of 4T1 cells) and conducting in vitro cell experiments, Kassassir et al. found that platelet activation and platelet cancer cell aggregation rate increased with tumor progression; in addition, platelet activation and reactivity are closely related to lung metastasis and extrahepatic hematopoiesis (Kassassir et al. 2020).
Clinical application of TEPs
Application of platelet proteins in tumors
Platelet proteins originate from various sources including proteins present in debris from megakaryocytes, proteins internalized in the extracellular environment, and proteins that undergo translation and synthesis within the platelet ribose. Due to the fact that tumor cells possess the ability to stimulate platelet activation and release a significant quantity of regulatory proteins, numerous scholars have undertaken research on the simultaneous investigation of platelet proteins for the purpose of tumor diagnosis and treatment.
The concurrent detection of vascular endothelial growth factor (VEGF), platelet-derived growth factor (PDGF), and PF4 in platelets has the potential to serve as a diagnostic tool for the detection of colorectal cancer (Peterson et al. 2012). In addition, the application of platelet protein expression profiling can also serve to differentiate benign adnexal lesions from the diagnoses of ovarian and lung cancer, as well as aid in prognosis monitoring and diagnosing pancreatic cancer (Lomnytska et al. 2018; Sabrkhany et al. 2018). These studies have established a strong groundwork for the clinical application of platelet-derived proteins in the context of tumors.
Moreover, the utilization of platelet proteins extends beyond just the detection of tumors; it also encompasses their therapeutic capabilities. The investigation of platelet proteins’ impact on tumor progression has emerged as a significant topic in the field of TEPs research. Kuznetsov et al. discovered that platelets possess the ability to absorb proteins secreted by tumors and subsequently disperse them among other “potential tumors” in order to facilitate the advancement of tumor development and metastasis (Kuznetsov et al. 2012). In addition, Kerr et al. discovered that bone metastatic proteins derived from tumors, such as transforming growth factor b-1 and matrix metalloproteinase-1, were absorbed by platelets and subsequently released into the bone microenvironment via platelet α particles (Kerr et al. 2013). In the future, tumors may be effectively treated or even thwarted by the exertion of platelet proteins.
Clinical monitoring of tumors by TEP RNA
Platelet RNA can additionally serve as a means for clinical monitoring during tumor therapy. Since the protection of tumor-derived transcripts by platelets from degradation by circulating plasma-derived ribonucleases and their continuous accumulation in TEPs, TEP RNA can be used to monitor dynamic changes in tumor activity. On the one hand, it can be used to monitor the progress of tumor treatment. On the other hand, it can also contribute to the exploration of acquired mutation sites that result in chemoresistance. In the diagnosis and treatment of glioblastoma, TEP RNA serves not only as a tool for monitoring the progression of glioblastoma, but also for distinguishing between false-positive progression and true progression (Sol et al. 2020). Since the effective crizotinib treatment for EML4-ALK has been observed to decrease the levels of EML4-ALK transcripts in TEPs during the treatment of NSCLC patients, it becomes possible to monitor TEP RNA to comprehend the drug therapy in these individuals with NSCLC (Nilsson et al. 2015). Park et al. has exhibited that the successive collection of TEP RNA adds predictive value in detecting EML4-ALK mutations, thereby enabling the prediction of favorable and unfavorable outcomes for ALK1 NSCLC patients who are undergoing treatment with alectinib or crizotinib (Park et al. 2019). Despite the fact that the current detection level of gene mutation of chemotherapy resistance by TEP RNA method is not as proficient as the conventional approach (Park et al. 2019; Brinkman et al. 2020), the utilization of the novel digital PCR technology can enhance both sensitivity and specificity in this domain of detection (Olmedillas-López et al. 2017).
Current studies of TEP RNA in tumors (excluding NSCLC)
Compared with traditional tumor markers and circulating tumor RNA, TEP RNA is tumor-specific RNA with higher accuracy and specificity. There is a wealth of research on the applications of TEP RNA in tumors other than NSCLC (Table 1). By sequencing and analyzing 283 platelet samples, Best et al. concluded that TEP RNA can accurately distinguish cancer patients from normal individuals (96% accuracy) and locate six different types of primary tumors (71% accuracy) (Best et al. 2015). Specifically, TEP-miR-4435-1 is highly expressed in HBC, glioblastoma and PAAD, and low expressed in breast cancer, so it can be used for screening different cancers. In 2020, he continued to analyze the diagnosis of TEP RNA in pan cancer and found that one can use the information provided by TEP splicing RNA patterns and cfDNA patterns of mutations or methylation in plasma to locate pan cancer (Wurdinger et al. 2020). Veld et al. established a platelet RNA library, developed and tested Seq algorithms for 18 pan cancer cells, and concluded that platelet RNA can be used for blood-based cancer detection, staging, and identification of tumor sites of origin (Veld et al. 2022). In cluster analysis and principal component analysis (PCA), it was found that MT-RNR2, MT-RNR1, TMSB4X, B2M, and MTND1 can distinguish ovarian cancer from healthy individuals (Gao et al. 2023). Sarcoma, as a rare heterogeneous tumor disease, can also be detected through highly sensitive and specific TEPs PSO enhanced thrombus sequence analysis (Heinhuis et al. 2020). SNORA58, SNORA68, and SNORD93 were significantly upregulated in TEPs of ESCA patients (Zhang et al. 2023). Recently, Miao et al. found that TEP miRNA affects platelet activity, PMPs production, thrombosis risk, immune cell function, and sensitivity to chemotherapy drugs and aspirin by regulating platelet activity and megakaryocyte maturation and differentiation (Miao et al. 2021). In addition, platelets are considered to be potential targets for cancer therapy. The platelet CLEC2–Podoplanin axis can be used as a target in the treatment of oral cancer (Hwang et al. 2021). Myeloproliferative neoplasm (MPN) is a rare malignant hematological disease with great difficulty in diagnosis. Girardot et al. took the first step in the progress of TEP RNA and MPN diagnosis. He found that miR-28 was a negative regulator of myeloid cytokine receptors, targeting not only the translation of MPL and E2F6 proteins, but also their terminal differentiation in CD34 derived megakaryocytes (Girardot et al. 2010). Notably, a few years later, he reported that TEP miR-28 was highly expressed in a subgroup of MPN patients, which may be related to the constitutive activation of STAT5 (Girardot et al. 2015). In ovarian cancer, PMPs play a role through sPLA2-IIa mediated miR-939 detachment, leading to epithelial–mesenchymal transition and tumor progression (Tang et al. 2017). Wang et al. found that TEP miR-34c-3p and TEP miR-18a-5p were highly expressed in nasopharyngeal carcinoma patients through real-time fluorescence quantitative PCR detection and ROC analysis (Wang et al. 2019).
Diagnosis, prognosis, and treatment of TEP RNA and NSCLC
The applications of TEP RNA in the diagnosis of NSCLC have been investigated since 2015 (Fig. 3). Zhang et al. performed RNA-seq on platelets from NSCLC patients and conducted differential gene expression analysis and subgroup analysis (Zhang et al. 2018). The results indicated that platelet RNA-seq can be used not only for distinguishing NSCLC from individuals, but also for the classification of lung cancer. In addition, Best et al. used a particle swarm optimization (PSO) enhanced algorithm to screen and verify TEP RNA for NSCLC patient staging (Best et al. 2017). Then Sheng et al. performed the IFS method to screen 48 biomarkers from TEP RNA-seq data of NSCLC patients and conducted cross validation. They found that liquid biopsy of 48-genes contributes to the early diagnosis and prolonged survival of NSCLC (Sheng et al. 2018). On the basis of previous sequencing, researchers conducted further research and validation on TEP RNA related genes and discovered their clinical applications. In NSCLC, multiple TEP RNAs are upregulated or downregulated, serving as both oncogenes and tumor suppressors. Table 2 lists important TEP RNAs related to the development of NSCLC.
The research progress of TEP RNA in NSCLC in the past decade. Since 2015, people have gradually begun to study the diagnostic applications of TEPs in NSCLC. Best et al. found in 2015 that miR-4435-1 and SNORD55 in TEPs were low expressed in NSCLC. Xue and Luo et al. found in 2018 that ACIN1 in TEPs was highly expressed in NSCLC, while LncRNA-MAGI2-AS3 and ZFAS1 were low expressed in NSCLC. In 2019, Liu and Xing et al. found that MAX, MTURN, HLA-B, and ITGA2B in TEPs were highly expressed in NSCLC. In the most recent year in 2021, Li and D’Ambrosi et al. found that Lnc-ST8SIA4-12 in TEPs was highly expressed, while circNRIP1, Linc GTF2H2-1, RP3-466P17.2 were low expressed in NSCLC. Created with BioRender.com
Research has found that TEP mRNA of MAX, MTURN, and HLA-B groups are highly expressed in NSCLC. All three groups of TEP RNA can be used to evaluate the effect of chemotherapy (Liu et al. 2019b). MAX helps members of the transcription factor network bind to DNA, and tissues lacking MAX-interacting protein MNT are prone to tumor formation (Hurlin and Huang 2006). Although MAX acts as a tumor suppressor in small cell lung cancer (Augert et al. 2020), it can also interact with NF-κB synergistically to promote Fas ligand expression in NSCLC and promote Fas related immune escape mechanism (Bennett et al. 1998; Kasibhatla et al. 2000; Wiener et al. 2004). Furthermore, MYC can serve as a therapeutic target for MAX mutated pheochromocytoma, and we can continue to study the application of MYC in NSCLC (Cascón and Robledo 2012; Massó-Vallés et al. 2020). MTURN is involved in MAPK/ERK, SAPK/JNK and NF-κB signaling pathways and is highly expressed in platelets of NSCLC patients (Sun et al. 2014). Consistent with this, the diagnostic efficiency of TEP MTURN in female patients is extremely high (Liu et al. 2019b). Lian et al. constructed a prognostic TF-methylation-gene network by single-factor Cox regression analysis, analyzed the methylation sites and genes of lung adenocarcinoma patients, and concluded that MTURN was downregulated in LUAD samples. However, the expression and regulatory mechanism of MTURN in NSCLC is still unclear and need to be further studied (Lian et al. 2022). It is reported that HLA is highly expressed in NCSLC and plays an important role in p300 mediated p53 acetylation (Goodman and Smolik 2000; Sasaki et al. 2007, p. 5; Tsukahara et al. 2009). In early stage of lung adenocarcinoma, ANIN1 interaction with AAC-11 and promotes tumor cell development and malignant transformation under high DNA methylation level (Shu et al. 2006; Rigou et al. 2009). We have learned from recent research that ITGA2B mutations can lead to changes in the PI3K signaling pathway, promote the occurrence of cancer and predict poor prognosis in large cell lung cancer (Guo et al. 2023). Besides, the high expression of ITGA2B in platelets of NSCLC patients has been confirmed by many researchers (Xing et al. 2019; Negahdari et al. 2020; Lecot et al. 2022). Dong et al. found that snRNA (U1, U2, U5) were low expressed in platelets of lung cancer patients, and this study is the only one that has applied TEP snRNA to the diagnosis of NSCLC (Dong et al. 2020). Although the mechanism is not completely clear, some scholars have pointed out that RNU2 is highly expressed in lung cancer patients and can be used as a bi-functional ncRNA to process a 19-22nt fragment for the detection of NSCLC (Mazières et al. 2013; Köhler et al. 2016). The discovery of anti-U1-A snRNP antibody in lung cancer patients has further promoted the application of RNU1 in the diagnosis of NSCLC (Zhang et al. 2005). lncRNA-MAGI2-AS3, as an oncogene, is low expressed in platelets of NSCLC patients (Luo et al. 2018). Researchers and scholars have studied the mechanism: lncRNA MAGI2-AS3 inhibits the proliferation, migration and invasion of NSCLC cells by regulating miR-374a/b-5p/CADM2, miRNA-23a-3p/PTEN and miR-629-5p/TXNIP axis and up-regulating RECK and cytokine signaling 1 in combination with miR-25 and miR-15583 (Hao and Yang 2019; He et al. 2020; Li et al. 2020). However, the lncRNA ZFAS1, which regulates NSCLC cell proliferation through ZFAS1/miR-590-3p axis, was found to be downregulated in TEPs of NSCLC patients (Luo et al. 2018; Zhou et al. 2020). This may be influenced by factors from different gene sources, and the same gene may exhibit different expression trends in pathological tissues and TEPs.
Although researchers have found high expression of NRC-ST8SIA4-12, and low expression of circNRIP1, linc-GTF2H2-1, RP3-466P17.2, miR-4435-1, and SNORD55 in NSCLC TEPs, the mechanism of action of these genes in NSCLC remains unclear (Table 2). We need to further investigate the mechanisms in order to better apply the biomarkers to the diagnosis and treatment of NSCLC.
Methods for detecting platelet RNA
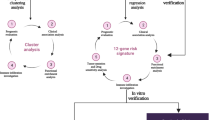
Based on existing studies and databases, we can further conduct confirmatory experiments on differentially expressed genes in NSCLC patients (Fig. 4). After collecting EDTA-K2 anticoagulants from a large number of NSCLC patients, platelets were isolated from plasma. The main extraction methods of platelets include washing, gradient density centrifugation, gel filtration, immunomagnetic bead separation, flow cytometry (FCM), etc. (Amisten 2012). Even if white blood cells can be removed by methods such as CD45 or magnetic bead sorting, they are still cumbersome, costly and may reduce platelet production. Therefore, gradient centrifugation has been included in thromboSeq protocol and can be used extensively in clinical trials (Rolf et al. 2005; Coppinger et al. 2007; Bray et al. 2013). To ensure platelet purity, an optical microscope was used to count platelet suspensions (Best et al. 2015) (There are less than 5 white blood cells per 1 million platelets). Platelet RNA was extracted and reverse-transcribed into cDNA for quantitative detection by RT-qPCR or NGS. Then, the differentially expressed genes in platelets will be analyzed based on Ct values or sequencing results.
Steps for TEP RNA study. Collect whole blood from the patient and store it with EDTA-K2 before separating platelets through two-step of low-speed centrifugation. Extract platelets RNA and perform NGS second-generation sequencing or real-time fluorescence quantitative PCR. Analyze the relationship between platelet RNA and NSCLC based on sequencing results or gene expression levels. Created with BioRender.com
Conclusions and future perspectives
There exists an intricate interplay between tumor cells and platelets. On the one hand, tumors and the surrounding environment exert an influence on the quantity, size, molecular omics, and function of platelets. On the other hand, platelets affect occurrence, development, and metastasis of tumor. Platelets and PMPS promote the development and progression of tumors by creating an inflammatory environment (Michael et al. 2017), stimulating angiogenesis, facilitating immune evasion, and modulating signaling pathways (Plantureux et al. 2020). Platelets also possess the capability to prevent tumor cell proliferation, indicating the necessity for comprehensive analysis of platelet function based on tumor type, disease stage, local microenvironment, and other pertinent factors. Currently, a large number of studies have confirmed significant differences in the expression of TEP RNA in platelets between NSCLC patients and healthy individuals. It is imperative to continuously explore the diagnostic value of TEP RNA in NSCLC.
Based on the bidirectional interaction between tumor cells and platelets, platelets are considered as emerging targets with significant potential in tumor diagnosis and treatment. The integration of NSCLC platelet-related markers with other NSCLC markers can enhance existing tumor diagnosis and prognostic evaluation systems, providing broad prospects in tumor screening, disease monitoring, and prognosis assessment. Beyond that, targeted inhibition of platelet function represents a feasible anti-tumor treatment strategy. In the development of anti-tumor nanomedicine, platelets exhibit excellent biocompatibility, tumor targeting, safety, and drug loading ability. The use of platelet simulation targeting agents can synergistically eradicate NSCLC cells through various methods, such as chemotherapy, phototherapy, radiotherapy, and calcium overload. Platelet-related tumor markers and targeted anti-tumor drugs have great potential in the field of tumor diagnosis and treatment, providing a promising pathway for achieving breakthroughs in precision oncology.
However, the application of platelets in the diagnosis and treatment of NSCLC is still in its early stages. In addition to the drawbacks of low platelet count and complex experimental processes, the diagnostic accuracy of TEP RNA-seq for cancer in different populations still needs to be improved and validated. For example, platelet RNA sequencing has poor diagnostic accuracy for detecting occult cancers in patients with unprovoked VTE (Best et al. 2015). We need to conduct a comprehensive analysis of TEP RNA from various aspects, including age, gender, and factors that contribute to NSCLC patients. It is important to analyze different patient situations separately, rather than just focusing on tumor phenotype. More importantly, in the clinical transformation process, we must pay attention to the risk of adverse reactions such as bleeding and off target of platelet-related anti-tumor drugs. To maximize the benefit of cancer patients, the molecular mechanism of platelet-tumor interaction needs to be further explored. Platelet-related tumor biomarkers and anti-tumor targeted drugs have broad prospects in tumor diagnosis and treatment, and are expected to bring significant breakthroughs for precise tumor treatment. Therefore, our future research direction will not be limited to the diagnostic value of TEP RNA in NSCLC, but should also extended to the intervention effect of TEP RNA.
Availability of data and materials
Not applicable.
Abbreviations
- NSCLC:
-
Non-small cell lung cancer
- LDCT:
-
Low-dose computed tomography
- LB:
-
Liquid biopsy
- CTCs:
-
Circulating tumor cells
- EVs:
-
Extracellular vesicle
- MKs:
-
Megakaryocytes
- RBP:
-
RNA-binding protein
- TEPs:
-
Tumor-educated platelets
- HBC:
-
Hepatobiliary cancer
- CRC:
-
Colorectal cancer
- GBM:
-
Glioblastoma
- PAAD:
-
Pancreatic cancer
- BC:
-
Breast cancer
- OC:
-
Ovarian cancer
- ESCA:
-
Esophageal cancer
- MPN:
-
Myeloproliferative neoplasms
- NPC:
-
Nasopharyngeal carcinoma
- LCC:
-
Lung and colon carcinoma.
- PLR:
-
Platelet-to-lymphocyte ratio
- OS:
-
Overall survival
- MPV:
-
Mean platelet volume
- PFS:
-
Progression-free survival
- irAEs:
-
Immune-related adverse events
- ICIs:
-
Immune checkpoint inhibitors
- VEGF:
-
Vascular endothelial growth factor
- PDGF:
-
Platelet-derived growth factor
- MAX:
-
MYC-associated protein X
- MTURN:
-
Maturin, neural progenitor differentiation regulator homolog
- HLA-B:
-
Human leukocyte antigen B
- ACIN1:
-
Apoptotic chromatin condensation inducer 1
- ITGA2B:
-
Integrin, alpha 2b (platelet glycoprotein IIb of IIb/IIIa complex, antigen CD41)
- ZFAS1:
-
ZNFX1 antisense RNA 1
- SNORD55:
-
Small nucleolar RNA C/D box 55
References
Amisten S (2012) A rapid and efficient platelet purification protocol for platelet gene expression studies. Methods Mol Biol 788:155–172. https://doi.org/10.1007/978-1-61779-307-3_12
Augert A, Mathsyaraja H, Ibrahim AH et al (2020) MAX functions as a tumor suppressor and rewires metabolism in small cell lung cancer. Cancer Cell 38:97-114.e7. https://doi.org/10.1016/j.ccell.2020.04.016
Bahmani B, Gong H, Luk BT et al (2021) Intratumoral immunotherapy using platelet-cloaked nanoparticles enhances antitumor immunity in solid tumors. Nat Commun 12:1999. https://doi.org/10.1038/s41467-021-22311-z
Bennett MW, O’Connell J, O’Sullivan GC et al (1998) The Fas counterattack in vivo: apoptotic depletion of tumor-infiltrating lymphocytes associated with Fas ligand expression by human esophageal carcinoma. J Immunol 160:5669–5675
Best MG, Sol N, Kooi I et al (2015) RNA-Seq of tumor-educated platelets enables blood-based pan-cancer, multiclass, and molecular pathway cancer diagnostics. Cancer Cell 28:666–676. https://doi.org/10.1016/j.ccell.2015.09.018
Best MG, Sol N, In’t Veld SGJG et al (2017) Swarm intelligence-enhanced detection of non-small-cell lung cancer using tumor-educated platelets. Cancer Cell 32:238-252.e9. https://doi.org/10.1016/j.ccell.2017.07.004
Bray PF, McKenzie SE, Edelstein LC et al (2013) The complex transcriptional landscape of the anucleate human platelet. BMC Genomics 14:1. https://doi.org/10.1186/1471-2164-14-1
Brinkman K, Meyer L, Bickel A et al (2020) Extracellular vesicles from plasma have higher tumour RNA fraction than platelets. J Extracell Vesicles 9:1741176. https://doi.org/10.1080/20013078.2020.1741176
Cagle PT, Allen TC, Olsen RJ (2013) Lung cancer biomarkers: present status and future developments. Arch Pathol Lab Med 137:1191–1198. https://doi.org/10.5858/arpa.2013-0319-CR
Cascón A, Robledo M (2012) MAX and MYC: a heritable breakup. Cancer Res 72:3119–3124. https://doi.org/10.1158/0008-5472.CAN-11-3891
Coppinger J, Fitzgerald DJ, Maguire PB (2007) Isolation of the platelet releasate. Methods Mol Biol 357:307–311. https://doi.org/10.1385/1-59745-214-9:307
D’Ambrosi S, Nilsson RJ, Wurdinger T (2021a) Platelets and tumor-associated RNA transfer. Blood 137:3181–3191. https://doi.org/10.1182/blood.2019003978
D’Ambrosi S, Visser A, Antunes-Ferreira M et al (2021b) The analysis of platelet-derived circRNA repertoire as potential diagnostic biomarker for non-small cell lung cancer. Cancers (basel) 13:4644. https://doi.org/10.3390/cancers13184644
Davis PJ, Mousa SA, Schechter GP, Lin H-Y (2020) Platelet ATP, thyroid hormone receptor on integrin αvβ3 and cancer metastasis. Horm Cancer 11:13–16. https://doi.org/10.1007/s12672-019-00371-4
Dong X, Ding S, Yu M et al (2020) Small nuclear RNAs (U1, U2, U5) in tumor-educated platelets are downregulated and act as promising biomarkers in lung Cancer. Front Oncol 10:01627
Gao Y, Liu C-J, Li H-Y et al (2023) Platelet RNA enables accurate detection of ovarian cancer: an intercontinental, biomarker identification study. Protein Cell 14:579–590. https://doi.org/10.1093/procel/pwac056
Girardot M, Pecquet C, Boukour S et al (2010) miR-28 is a thrombopoietin receptor targeting microRNA detected in a fraction of myeloproliferative neoplasm patient platelets. Blood 116:437–445. https://doi.org/10.1182/blood-2008-06-165985
Girardot M, Pecquet C, Chachoua I et al (2015) Persistent STAT5 activation in myeloid neoplasms recruits p53 into gene regulation. Oncogene 34:1323–1332. https://doi.org/10.1038/onc.2014.60
Goksel S, Ozcelik N, Telatar G, Ardic C (2021) The role of hematological inflammatory biomarkers in the diagnosis of lung cancer and in predicting TNM stage. Cancer Invest 39:514–520. https://doi.org/10.1080/07357907.2021.1938110
Goodman RH, Smolik S (2000) CBP/p300 in cell growth, transformation, and development. Genes Dev 14:1553–1577
Grozovsky R, Giannini S, Falet H, Hoffmeister KM (2015) Regulating billions of blood platelets: glycans and beyond. Blood 126:1877–1884. https://doi.org/10.1182/blood-2015-01-569129
Guo J-H, Ma Y-S, Lin J-W et al (2023) Whole-exome and targeted gene sequencing of large-cell lung carcinoma reveals recurrent mutations in the PI3K pathway. Br J Cancer 129:366–373. https://doi.org/10.1038/s41416-023-02301-2
Haemmerle M, Stone RL, Menter DG et al (2018) The platelet lifeline to cancer: challenges and opportunities. Cancer Cell 33:965–983. https://doi.org/10.1016/j.ccell.2018.03.002
Hao X-Z, Yang K (2019) LncRNA MAGI2-AS3 suppresses the proliferation and invasion of non-small cell lung carcinoma through miRNA-23a-3p/PTEN axis. Eur Rev Med Pharmacol Sci 23:7399–7407. https://doi.org/10.26355/eurrev_201909_18848
He J, Zhou X, Li L, Han Z (2020) Long noncoding MAGI2-AS3 suppresses several cellular processes of lung squamous cell carcinoma cells by regulating miR-374a/b-5p/CADM2 axis. Cancer Manag Res 12:289–302. https://doi.org/10.2147/CMAR.S232595
Heazlewood SY, Ahmad T, Mohenska M et al (2022) The RNA-binding protein SRSF3 has an essential role in megakaryocyte maturation and platelet production. Blood 139:1359–1373. https://doi.org/10.1182/blood.2021013826
Heinhuis KM, In’t Veld SGJG, Dwarshuis G et al (2020) RNA-sequencing of tumor-educated platelets, a novel biomarker for blood-based sarcoma diagnostics. Cancers (basel) 12:1372. https://doi.org/10.3390/cancers12061372
Heldin NE, Usuki K, Bergh J et al (1993) Differential expression of platelet-derived endothelial cell growth factor/thymidine phosphorylase in human lung carcinoma cell lines. Br J Cancer 68:708–711. https://doi.org/10.1038/bjc.1993.414
Holinstat M (2017) Normal platelet function. Cancer Metastasis Rev 36:195–198. https://doi.org/10.1007/s10555-017-9677-x
Hufnagel DH, Cozzi GD, Crispens MA, Beeghly-Fadiel A (2020) Platelets, thrombocytosis, and ovarian cancer prognosis: surveying the landscape of the literature. Int J Mol Sci 21:8169. https://doi.org/10.3390/ijms21218169
Hurlin PJ, Huang J (2006) The MAX-interacting transcription factor network. Semin Cancer Biol 16:265–274. https://doi.org/10.1016/j.semcancer.2006.07.009
Hwang B-O, Park S-Y, Cho ES et al (2021) Platelet CLEC2-podoplanin axis as a promising target for oral cancer treatment. Front Immunol 12:807600. https://doi.org/10.3389/fimmu.2021.807600
Kaser A, Brandacher G, Steurer W et al (2001) Interleukin-6 stimulates thrombopoiesis through thrombopoietin: role in inflammatory thrombocytosis. Blood 98:2720–2725. https://doi.org/10.1182/blood.v98.9.2720
Kasibhatla S, Beere HM, Brunner T et al (2000) A “non-canonical” DNA-binding element mediates the response of the Fas-ligand promoter to c-Myc. Curr Biol 10:1205–1208. https://doi.org/10.1016/s0960-9822(00)00727-2
Kassassir H, Karolczak K, Siewiera KM et al (2020) Time-dependent interactions of blood platelets and cancer cells, accompanied by extramedullary hematopoiesis, lead to increased platelet activation and reactivity in a mouse orthotopic model of breast cancer—implications for pulmonary and liver metastasis. Aging (albany NY) 12:5091–5120. https://doi.org/10.18632/aging.102933
Kataki A, Scheid P, Piet M et al (2002) Tumor infiltrating lymphocytes and macrophages have a potential dual role in lung cancer by supporting both host-defense and tumor progression. J Lab Clin Med 140:320–328. https://doi.org/10.1067/mlc.2002.128317
Kerr BA, McCabe NP, Feng W, Byzova TV (2013) Platelets govern pre-metastatic tumor communication to bone. Oncogene 32:4319–4324. https://doi.org/10.1038/onc.2012.447
Klement GL, Yip T-T, Cassiola F et al (2009) Platelets actively sequester angiogenesis regulators. Blood 113:2835–2842. https://doi.org/10.1182/blood-2008-06-159541
Köhler J, Schuler M, Gauler TC et al (2016) Circulating U2 small nuclear RNA fragments as a diagnostic and prognostic biomarker in lung cancer patients. J Cancer Res Clin Oncol 142:795–805. https://doi.org/10.1007/s00432-015-2095-y
Kuznetsov HS, Marsh T, Markens BA et al (2012) Identification of luminal breast cancers that establish a tumor-supportive macroenvironment defined by proangiogenic platelets and bone marrow-derived cells. Cancer Discov 2:1150–1165. https://doi.org/10.1158/2159-8290.CD-12-0216
Labelle M, Begum S, Hynes RO (2011) Direct signaling between platelets and cancer cells induces an epithelial-mesenchymal-like transition and promotes metastasis. Cancer Cell 20:576–590. https://doi.org/10.1016/j.ccr.2011.09.009
Lecot P, Ardin M, Dussurgey S et al (2022) Gene signature of circulating platelet-bound neutrophils is associated with poor prognosis in cancer patients. Int J Cancer 151:138–152. https://doi.org/10.1002/ijc.33991
Li B, Zhou P, Liu Y et al (2018) Platelet-to-lymphocyte ratio in advanced cancer: review and meta-analysis. Clin Chim Acta 483:48–56. https://doi.org/10.1016/j.cca.2018.04.023
Li F, Hu Q, Pang Z, Xu X (2020) LncRNA MAGI2-AS3 upregulates cytokine signaling 1 by sponging miR-155 in non-small cell lung cancer. Cancer Biother Radiopharm 35:72–76. https://doi.org/10.1089/cbr.2019.2898
Li X, Liu L, Song X et al (2021) TEP linc-GTF2H2-1, RP3-466P17.2, and lnc-ST8SIA4-12 as novel biomarkers for lung cancer diagnosis and progression prediction. J Cancer Res Clin Oncol 147:1609–1622. https://doi.org/10.1007/s00432-020-03502-5
Li A, Chang Y, Song N-J et al (2022a) Selective targeting of GARP-LTGFβ axis in the tumor microenvironment augments PD-1 blockade via enhancing CD8+ T cell antitumor immunity. J Immunother Cancer 10:e005433. https://doi.org/10.1136/jitc-2022-005433
Li S, Li L, Lin X et al (2022b) Targeted inhibition of tumor inflammation and tumor-platelet crosstalk by nanoparticle-mediated drug delivery mitigates cancer metastasis. ACS Nano 16:50–67. https://doi.org/10.1021/acsnano.1c06022
Li M, Zhao S, Lopez G et al (2023) Mean platelet volume, thrombocytosis, and survival in non-small cell lung cancer patients treated with first-line pembrolizumab alone or with chemotherapy. Cancer Immunol Immunother 72:2067–2074. https://doi.org/10.1007/s00262-023-03392-9
Lian D, Lian L, Zeng D et al (2022) Identification of prognostic values of the transcription factor-CpG-gene triplets in lung adenocarcinoma: a narrative review. Medicine (baltimore) 101:e32045. https://doi.org/10.1097/MD.0000000000032045
Lin RJ, Afshar-Kharghan V, Schafer AI (2014) Paraneoplastic thrombocytosis: the secrets of tumor self-promotion. Blood 124:184–187. https://doi.org/10.1182/blood-2014-03-562538
Liu J, Li S, Zhang S et al (2019a) Systemic immune-inflammation index, neutrophil-to-lymphocyte ratio, platelet-to-lymphocyte ratio can predict clinical outcomes in patients with metastatic non-small-cell lung cancer treated with nivolumab. J Clin Lab Anal 33:e22964. https://doi.org/10.1002/jcla.22964
Liu L, Song X, Li X et al (2019b) A three-platelet mRNA set: MAX, MTURN and HLA-B as biomarker for lung cancer. J Cancer Res Clin Oncol 145:2713–2723. https://doi.org/10.1007/s00432-019-03032-9
Liu Y, Zhang Y, Ding Y, Zhuang R (2021) Platelet-mediated tumor metastasis mechanism and the role of cell adhesion molecules. Crit Rev Oncol Hematol 167:103502. https://doi.org/10.1016/j.critrevonc.2021.103502
Lomnytska M, Pinto R, Becker S et al (2018) Platelet protein biomarker panel for ovarian cancer diagnosis. Biomark Res 6:2. https://doi.org/10.1186/s40364-018-0118-y
Luo C-L, Xu Z-G, Chen H et al (2018) LncRNAs and <em>EGFRvIII</em> sequestered in TEPs enable blood-based NSCLC diagnosis. CMAR 10:1449–1459. https://doi.org/10.2147/CMAR.S164227
Ma Z, Liu S, Ke Y et al (2020) Biomimetic nano-NOS mediated local NO release for inhibiting cancer-associated platelet activation and disrupting tumor vascular barriers. Biomaterials 255:120141. https://doi.org/10.1016/j.biomaterials.2020.120141
Mammadova-Bach E, Gil-Pulido J, Sarukhanyan E et al (2020) Platelet glycoprotein VI promotes metastasis through interaction with cancer cell-derived galectin-3. Blood 135:1146–1160. https://doi.org/10.1182/blood.2019002649
Martins Castanheira N, Spanhofer AK, Wiener S et al (2022) Uptake of platelets by cancer cells and recycling of the platelet protein CD42a. J Thromb Haemost 20:170–181. https://doi.org/10.1111/jth.15543
Massó-Vallés D, Beaulieu M-E, Soucek L (2020) MYC, MYCL, and MYCN as therapeutic targets in lung cancer. Expert Opin Ther Targets 24:101–114. https://doi.org/10.1080/14728222.2020.1723548
Mazières J, Catherinne C, Delfour O et al (2013) Alternative processing of the U2 small nuclear RNA produces a 19–22nt fragment with relevance for the detection of non-small cell lung cancer in human serum. PLoS ONE 8:e60134. https://doi.org/10.1371/journal.pone.0060134
Mezouar S, Mege D, Darbousset R et al (2014) Involvement of platelet-derived microparticles in tumor progression and thrombosis. Semin Oncol 41:346–358. https://doi.org/10.1053/j.seminoncol.2014.04.010
Mezouar S, Frère C, Darbousset R et al (2016) Role of platelets in cancer and cancer-associated thrombosis: experimental and clinical evidences. Thromb Res 139:65–76. https://doi.org/10.1016/j.thromres.2016.01.006
Miao S, Zhang Q, Chang W, Wang J (2021) New insights into platelet-enriched miRNAs: production, functions, roles in tumors, and potential targets for tumor diagnosis and treatment. Mol Cancer Ther 20:1359–1366. https://doi.org/10.1158/1535-7163.MCT-21-0050
Michael JV, Wurtzel JGT, Mao GF et al (2017) Platelet microparticles infiltrating solid tumors transfer miRNAs that suppress tumor growth. Blood 130:567–580. https://doi.org/10.1182/blood-2016-11-751099
Morris K, Schnoor B, Papa A-L (2022) Platelet cancer cell interplay as a new therapeutic target. Biochim Biophys Acta Rev Cancer 1877:188770. https://doi.org/10.1016/j.bbcan.2022.188770
National Lung Screening Trial Research Team, Aberle DR, Adams AM et al (2011) Reduced lung-cancer mortality with low-dose computed tomographic screening. N Engl J Med 365:395–409. https://doi.org/10.1056/NEJMoa1102873
Negahdari S, Zamani M, Seifi T et al (2020) Identification of three novel mutations in the FANCA, FANCC, and ITGA2B genes by whole exome sequencing. Int J Prev Med 11:117. https://doi.org/10.4103/ijpvm.IJPVM_462_19
Nikanjam M, Kato S, Kurzrock R (2022) Liquid biopsy: current technology and clinical applications. J Hematol Oncol 15:131. https://doi.org/10.1186/s13045-022-01351-y
Nilsson RJA, Karachaliou N, Berenguer J et al (2015) Rearranged EML4-ALK fusion transcripts sequester in circulating blood platelets and enable blood-based crizotinib response monitoring in non-small-cell lung cancer. Oncotarget 7:1066–1075
Olmedillas-López S, García-Arranz M, García-Olmo D (2017) Current and emerging applications of droplet digital PCR in oncology. Mol Diagn Ther 21:493–510. https://doi.org/10.1007/s40291-017-0278-8
Park C-K, Kim J-E, Kim M-S et al (2019) Feasibility of liquid biopsy using plasma and platelets for detection of anaplastic lymphoma kinase rearrangements in non-small cell lung cancer. J Cancer Res Clin Oncol 145:2071–2082. https://doi.org/10.1007/s00432-019-02944-w
Peterson JE, Zurakowski D, Italiano JE et al (2012) VEGF, PF4 and PDGF are elevated in platelets of colorectal cancer patients. Angiogenesis 15:265–273. https://doi.org/10.1007/s10456-012-9259-z
Pilatova K, Greplova K, Demlova R et al (2013) Role of platelet chemokines, PF-4 and CTAP-III, in cancer biology. J Hematol Oncol 6:42. https://doi.org/10.1186/1756-8722-6-42
Placke T, Salih HR, Kopp H-G (2012) GITR ligand provided by thrombopoietic cells inhibits NK cell antitumor activity. J Immunol 189:154–160. https://doi.org/10.4049/jimmunol.1103194
Plantureux L, Mège D, Crescence L et al (2020) The interaction of platelets with colorectal cancer cells inhibits tumor growth but promotes metastasis. Cancer Res 80:291–303. https://doi.org/10.1158/0008-5472.CAN-19-1181
Pyo J-S, Sohn JH, Kang G (2016) Diagnostic and prognostic roles of the mean platelet volume in malignant tumors: a systematic review and meta-analysis. Platelets 27:722–728. https://doi.org/10.3109/09537104.2016.1169265
Qu M, Zou X, Fang F et al (2020) Platelet-derived microparticles enhance megakaryocyte differentiation and platelet generation via miR-1915-3p. Nat Commun 11:4964. https://doi.org/10.1038/s41467-020-18802-0
Ren J, He J, Zhang H et al (2021) Platelet TLR4-ERK5 axis facilitates NET-mediated capturing of circulating tumor cells and distant metastasis after surgical stress. Cancer Res 81:2373–2385. https://doi.org/10.1158/0008-5472.CAN-20-3222
Rigou P, Piddubnyak V, Faye A et al (2009) The antiapoptotic protein AAC-11 interacts with and regulates Acinus-mediated DNA fragmentation. EMBO J 28:1576–1588. https://doi.org/10.1038/emboj.2009.106
Rolf N, Knoefler R, Suttorp M et al (2005) Optimized procedure for platelet RNA profiling from blood samples with limited platelet numbers. Clin Chem 51:1078–1080. https://doi.org/10.1373/clinchem.2005.049486
Sabrkhany S, Kuijpers MJE, van Kuijk SMJ et al (2017) A combination of platelet features allows detection of early-stage cancer. Eur J Cancer 80:5–13. https://doi.org/10.1016/j.ejca.2017.04.010
Sabrkhany S, Kuijpers MJE, Knol JC et al (2018) Exploration of the platelet proteome in patients with early-stage cancer. J Proteomics 177:65–74. https://doi.org/10.1016/j.jprot.2018.02.011
Şahin F, Aslan AF (2018) Relationship between inflammatory and biological markers and lung cancer. J Clin Med 7:160. https://doi.org/10.3390/jcm7070160
Sakin A, Secmeler S, Arici S et al (2019) Prognostic significance of mean platelet volume on local advanced non-small cell lung cancer managed with chemoradiotherapy. Sci Rep 9:3959. https://doi.org/10.1038/s41598-019-40589-4
Sasaki T, Gan EC, Wakeham A et al (2007) HLA-B-associated transcript 3 (Bat3)/Scythe is essential for p300-mediated acetylation of p53. Genes Dev 21:848–861. https://doi.org/10.1101/gad.1534107
Schlesinger M (2018) Role of platelets and platelet receptors in cancer metastasis. J Hematol Oncol 11:1–15. https://doi.org/10.1186/s13045-018-0669-2
Schumacher D, Strilic B, Sivaraj KK et al (2013) Platelet-derived nucleotides promote tumor-cell transendothelial migration and metastasis via P2Y2 receptor. Cancer Cell 24:130–137. https://doi.org/10.1016/j.ccr.2013.05.008
Sheng M, Dong Z, Xie Y (2018) Identification of tumor-educated platelet biomarkers of non-small-cell lung cancer. Onco Targets Ther 11:8143–8151. https://doi.org/10.2147/OTT.S177384
Shu Y, Iijima T, Sun W et al (2006) The ACIN1 gene is hypermethylated in early stage lung adenocarcinoma. J Thorac Oncol 1:160–167
Siegel RL, Miller KD, Wagle NS, Jemal A (2023) Cancer statistics, 2023. CA Cancer J Clin 73:17–48. https://doi.org/10.3322/caac.21763
Singh A (2021) RNA-binding protein kinetics. Nat Methods 18:335. https://doi.org/10.1038/s41592-021-01122-6
Söderdahl G, Betsholtz C, Johanson A et al (1988) Differential expression of platelet-derived growth factor and transforming growth factor genes in small- and non-small-cell human lung carcinoma lines. Int J Cancer 41:636–641. https://doi.org/10.1002/ijc.2910410426
Sol N, Int-Veld SGJG, Vancura A et al (2020) Tumor-educated platelet RNA for the detection and (pseudo)progression monitoring of glioblastoma. Cell Rep Med 1:100101. https://doi.org/10.1016/j.xcrm.2020.100101
Stone RL, Nick AM, McNeish IA et al (2012) Paraneoplastic thrombocytosis in ovarian cancer. N Engl J Med 366:610–618. https://doi.org/10.1056/NEJMoa1110352
Sun X, Lu B, Hu B et al (2014) Novel function of the chromosome 7 open reading frame 41 gene to promote leukemic megakaryocyte differentiation by modulating TPA-induced signaling. Blood Cancer J 4:e198. https://doi.org/10.1038/bcj.2014.18
Suzuki-Inoue K (2019) Platelets and cancer-associated thrombosis: focusing on the platelet activation receptor CLEC-2 and podoplanin. Hematol Am Soc Hematol Educ Progr 2019:175–181. https://doi.org/10.1182/hematology.2019001388
Tang M, Jiang L, Lin Y et al (2017) Platelet microparticle-mediated transfer of miR-939 to epithelial ovarian cancer cells promotes epithelial to mesenchymal transition. Oncotarget 8:97464–97475. https://doi.org/10.18632/oncotarget.22136
Tran JQD, Pedersen OH, Larsen ML et al (2020) Platelet microRNA expression and association with platelet maturity and function in patients with essential thrombocythemia. Platelets 31:365–372. https://doi.org/10.1080/09537104.2019.1636019
Tsukahara T, Kimura S, Ichimiya S et al (2009) Scythe/BAT3 regulates apoptotic cell death induced by papillomavirus binding factor in human osteosarcoma. Cancer Sci 100:47–53. https://doi.org/10.1111/j.1349-7006.2008.00991.x
Veld SGJGI, Arkani M, Post E et al (2022) Detection and localization of early- and late-stage cancers using platelet RNA. Cancer Cell 40:999-1009.e6. https://doi.org/10.1016/j.ccell.2022.08.006
Walraven M, Sabrkhany S, Knol JC et al (2021) Effects of cancer presence and therapy on the platelet proteome. Int J Mol Sci 22:8236. https://doi.org/10.3390/ijms22158236
Wang H, Wei X, Wu B et al (2019) Tumor-educated platelet miR-34c-3p and miR-18a-5p as potential liquid biopsy biomarkers for nasopharyngeal carcinoma diagnosis. Cancer Manag Res 11:3351–3360. https://doi.org/10.2147/CMAR.S195654
Ward Y, Lake R, Faraji F et al (2018) Platelets promote metastasis via binding tumor CD97 leading to bidirectional signaling that coordinates transendothelial migration. Cell Rep 23:808–822. https://doi.org/10.1016/j.celrep.2018.03.092
Wiener Z, Ontsouka EC, Jakob S et al (2004) Synergistic induction of the Fas (CD95) ligand promoter by Max and NFkappaB in human non-small lung cancer cells. Exp Cell Res 299:227–235. https://doi.org/10.1016/j.yexcr.2004.05.031
Wurdinger T, In’t Veld SGJG, Best MG (2020) Platelet RNA as pan-tumor biomarker for cancer detection. Cancer Res 80:1371–1373. https://doi.org/10.1158/0008-5472.CAN-19-3684
Xia L, Zeng Z, Tang WH (2018) The role of platelet microparticle associated microRNAs in cellular crosstalk. Front Cardiovasc Med 5:29. https://doi.org/10.3389/fcvm.2018.00029
Xing S, Zeng T, Xue N et al (2019) Development and validation of tumor-educated blood platelets integrin alpha 2b (ITGA2B) RNA for diagnosis and prognosis of non-small-cell lung cancer through RNA-seq. Int J Biol Sci 15:1977–1992. https://doi.org/10.7150/ijbs.36284
Xue L, Xie L, Song X, Song X (2018) Expression and significance of ACIN1 mRNA in platelets of lung cancer. Zhongguo Fei Ai Za Zhi 21:677–681. https://doi.org/10.3779/j.issn.1009-3419.2018.09.05
Zhang L, Liu J, Zhang H et al (2005) Discovery and identification of anti-U1-A snRNP antibody in lung cancer. Sci China C Life Sci 48:641–647. https://doi.org/10.1360/062005-82
Zhang Q, Hu H, Liu H et al (2018) RNA sequencing enables systematic identification of platelet transcriptomic alterations in NSCLC patients. Biomed Pharmacother 105:204–214. https://doi.org/10.1016/j.biopha.2018.05.074
Zhang Q, Bi Z, Song X et al (2023) Tumor-educated platelet SNORA58, SNORA68 and SNORD93 as novel diagnostic biomarkers for esophageal cancer. Future Oncol 19:651–661. https://doi.org/10.2217/fon-2023-0129
Zhou Y, Si L, Liu Z et al (2020) Long noncoding RNA ZFAS1 promotes progression of NSCLC via regulating of miR-590-3p. Cell Transplant 29:963689720919435. https://doi.org/10.1177/0963689720919435
Zufferey A, Fontana P, Reny J-L et al (2012) Platelet proteomics. Mass Spectrom Rev 31:331–351. https://doi.org/10.1002/mas.20345
Acknowledgements
Figures were created with BioRender.com by our team. We appreciate the graphic design materials and platform provided by BioRender.com (https://www.biorender.com/).
Funding
This work was funding by project ZR2023QC072 supported by Shandong Provincial Natural Science Foundation (Y.D.).
Author information
Authors and Affiliations
Contributions
Original draft preparation YW. Data collection AD and MJ. Review and editing SL. Conceptualization, review and editing YD. All authors have read and agreed to the published version of the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Ethical approval and consent to participate
Not applicable.
Patient consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Wang, Y., Dong, A., Jin, M. et al. TEP RNA: a new frontier for early diagnosis of NSCLC. J Cancer Res Clin Oncol 150, 97 (2024). https://doi.org/10.1007/s00432-024-05620-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00432-024-05620-w