Abstract
Milk is an essential commodity whose demand far exceeds supply. However, dairy animal productivity is constantly hampered by parasitic diseases such as fasciolosis, affecting milk production. Despite the negative impact of liver fluke on milk production, there is little information on liver fluke infection and associated abattoir losses (body weight, condition score, liver pathology, and carcass quality) in culled dairy cattle. This study aimed to determine body condition scores, fluke intensity, liver pathology, and carcass quality of different cattle genotypes infected with Fasciola species at three commercial abattoirs. A longitudinal study was conducted from September 2019 to October 2020 to determine body condition score, liver fluke intensity, liver pathology in 3065 dairy cattle slaughtered in CA1, CA2, and CA3, of the Eastern Cape Province South Africa. Liver fluke intensity significantly increased with cattle age (P < 0.0001). Cattle ≥ 7 years old (59.93 ± 6.42) and those 4 to 6 years old (49.78 ± 9.98) had higher infection than those 2 to 3 years old (27.55 ± 13.68). The liver fluke infection was significantly (P < 0.001) the highest when sampling was conducted in summer, followed by autumn and winter, and least for spring. The differences in carcass weights or body condition scores decreased by 0.99 units (P < 0.0001) or 0.97 units (P < 0.0001) respectively. Therefore, this study suggests that fluke infection could be responsible for considerable economic and production losses mainly due to condemnation and weight loss in dairy cattle. This study recommended a combination of holistic and grazing management to control infection rates in dairy herds.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The dairy sector contributes about 4.2% to the South African gross domestic product (GDP) and is the largest agricultural sector (DAFF 2019; Lacto Data 2020). Dairy production is widely practiced across the country, with the Western Cape, Eastern Cape, and KwaZulu Natal being the leading provinces (Stats SA 2020; Lacto Data 2020). The South African dairy industry is divided into two sub-sectors, namely, commercial, characterized by large cattle herd sizes, enough land for practicing and nutrition, and small-holders, with small size herds and less land to produce (Lacto data 2017). The Eastern Cape commercial dairy sector contributes about 28% to the South African dairy industry and plays a vital role in skill development, economic sustainability, livelihoods, and food security (Lacto Data, 2016, 2019; DAFF, 2019; Stats SA, 2020). The increase of the human population in urban and rural communities has increased the demand for fresh milk and dairy products, thus requiring an increase in dairy cattle numbers to enhance milk production (Lacto Data, 2019; Stats SA, 2020). However, this production is heavily impacted by parasitic diseases (Rehman 2020).
Fasciolosis is a snail-borne disease chiefly driven by two dominant species; Fasciola gigantica and Fasciola hepatica. The snail hosts play a prominent role in the distribution and the epidemiology of fasciolosis (Pfukenyi et al. 2005; Malatji et al. 2019). Liver fluke is a growing and emerging threat to dairy cattle and human health in developing countries (Jaja et al. 2017a; Mas-Coma et al. 2020; Mehmood et al. 2017; Takeuchi-Storm et al. 2018). Dairy cattle infected by liver fluke may experience reduced feed conversion efficiency, loss of body weight and condition score, reduced milk quality and yield (milk volume), and inhibition of puberty in replacement heifers (Charlier et al. 2014; Bloemhoff et al. 2015; Howell et al. 2015). Liver fluke has been reported to directly affect the liver through the migratory action of flukes in ductular tracts, blood-sucking, and liver damage leading to metabolic diseases. Consequently, these effects often lead to the culling of poorly performing animals, liver condemnations, and substantial economic losses (Radfar et al. 2015; Jaja et al. 2017a, b, Mehmood et al. 2017; Mochankana and Robertson 2018; Zewde et al. 2019; Arias-Pacheco et al. 2020).
Severity index can be described as the degree of infection or fibrotic scores caused by parasites in the liver (Jaja et al. 2017a; Mochankana and Robertson 2018; Charlier et al. 2020). Several studies reported that fluke pathology is diagnosed through lesions and fibrotic tracks found in the livers during a proper inspection after slaughter (Kusumarini et al. 2020). Fluke burden and liver pathology are important parameters that should be determined to be incorporated in the estimation of the impact of liver fluke on dairy production (Howell et al. 2012; Mazeri et al. 2017; Zewde et al. 2019). Data on the severity of the infection and its association with body condition score and carcass quality in dairy cattle slaughtered at commercial abattoirs is scarce. Hence, the current study aimed to investigate the severity of Fasciola spp. infection in dairy cattle slaughtered in three commercial abattoirs of the Eastern Cape Province, South Africa.
Materials and methods
Ethical consideration
Experimental protocols for this study were reviewed and approved by the animal ethics committee of the University of Fort Hare (Ref: JAJ011SMPI01/19/A). All the experimental procedures were conducted as per moral standards of experimentation given by the ethics committee on animal use of the Society for the Prevention of Cruelty to Animals (SPCA).
Study site and design
The study sites are shown in Fig. 1. A longitudinal study of dairy cattle in three commercial abattoirs was conducted using ante-mortem and post-mortem inspection. The study was carried out from September 2019 to October 2020 to determine the severity index or fluke intensity and seasonality in dairy cattle slaughtered in the Eastern Cape. Prior to slaughter, body condition scores were observed by trained personnel. A five-point scale description was used to determine the body condition scores, where 1 denoted very poor or emaciated, 2 denoted poor, 3 denoted good, 4 denoted fat, and 5 denoted excessively fat animals (Nicholson and Butterworth 1986). However, it was re-arranged to fit into a 3-point scale (1–2) poor, (3–4) moderate, and (5) good (Jaja et al. 2017a, b). Age information was obtained using the records from farmers. Animals were grouped into two based on age: young denoting cattle less than 3 years of age, and old, meaning those 3 years of age and greater. The study focused only on females since few (3) bulls came from dairy farms. The place or geographic origins and farming systems were obtained from the abattoir records. Pasture-based feeding systems characterized the commercial dairy farms where all slaughtered cattle originated. Animal genotypes were identified by using abattoir records. In cases where the records were unavailable, breed phenotypes were used to determine the breed type (Dupuy et al. 2013; Mpakama et al. 2014; Soji et al. 2015).
Data collection
Liver inspection
During the study period, monthly visits were made to the selected three commercial abattoirs to gather samples of Fasciola-infected livers from dairy cattle. A typical case selection method was adopted and used for the survey. The sampling technique allows for the suitable inspection of all the condemned livers due to fasciolosis. The post-mortem meat inspection was carried out as outlined in the Meat Safety Act of 2000 (MSA 2000). During sampling, condemned livers and gall bladders were thoroughly inspected visually for the presence of liver flukes. Evidence of Fasciola infection was supported by liver enlargement with raised or depressed areas and a firm consistency on palpation. Soon after slaughter, the livers condemned due to fasciolosis were immediately weighed on the electronic scale (Ansutek M1/M2 Portable Crane Scale, Ansutek Commercial Ltd, New Zealand) as described elsewhere in the literature (Jaja et al. 2017a, b). A total of 3065 livers were inspected, and 98,875 adult flukes were recovered from the livers detained for fluke extraction and characterization. To determine the presence of Fasciola species, the bile duct, livers, and gall bladders were longitudinally incised with a sharp knife. The livers were incised into sections and squeezed to force out any fluke present from the bile duct. The gallbladder was also opened, drained, and inspected for any presence of flukes and eggs. Flukes were removed from the liver using blunt forceps, enumerated, and preserved in clean universal bottles containing 70% alcohol or formalin. The number of flukes in each liver was described as the severity index or fluke burden. The severity index was classified as follows: 1 mild infection (< 30 flukes), 2 moderately infection (31–50 flukes), and 3 heavily infection (51 flukes and above) (Ploeger et al. 2017). Carcass classes were obtained during post-slaughter-house inspection and were classified from 1 to 5 where: 1 denoted cattle with no fat cover on the back and pelvis, 2 denoted little fat cover, 3 denoted medium, 4 denoted fat cover on the back, and 5 denoted over fat carcasses (Soji et al. 2015). The fluke species were transported to the Department of Veterinary Tropical Diseases Parasitology Laboratory in Pretoria for further identification. Details regarding the geographical origin, type of management system, breed, age, and body condition score were recorded during the post-mortem or meat inspection for every dairy cow and heifer in each abattoir. Furthermore, six liver samples per fibrotic class (mild, moderate, and severe) were taken for further laboratory analyses to assess the level of pathology using a light microscope (Kaewkong et al. 2012; Jenkins et al. 2020; Kurzy´nska and Kurzy´nska-Kokorniak 2021).
Liver collection and preparation
A total of 20 g of livers was harvested from condemned livers due to Fasciola infection and immediately put in the universal bottle containing 70% of ethanol for fixation. The samples were then put in the cooler box fixed with ice and transported to the Botany Laboratory at the University of Fort Hare. Before the analysis, 0.5 cm2 liver was cut into two sections. The livers were fixed with cold buffer containing 2.5% glutaraldehyde and stored at 4 °C for 24 h. Then, the samples were washed with the cold phosphate buffer (0.5 m) four times. The samples were treated with 2% osmium tetroxide buffer for 24 h. Then, distilled water was used to wash and remove osmium tetroxide three times. The samples were dried and graded with ethanol for 20 min in ascending order of 30–100%, respectively. To improve electron conductivity, the sample surface in the scanning electron microscope (SEM) uses a sputter-coated palladium-gold film to enhance analysis. Use Hitachi Point Dryer HCP2 (Hitachi KOKI Co Ltd, Tokyo, Japan) to dry at critical points to avoid sample changes and promote good structure preservation. This was done by mounting aluminium rod samples with double-sided carbon tape and then spraying gold plating (EIKO engineering Co TD, Japan). The samples were viewed under the scanning electron microscope (SEM) (JEOL, JSM6390 LV, JEOL Ltd, Japan) to identify fibrotic lesions in the liver caused by Fasciola species.
Statistical analysis
Descriptive statistics were used to estimate each explanatory variable’s such as mean, standard deviation, and range. The negative binomial generalized linear mixed-effects model (GLMM) was performed to determine the influence of age (2 to 3 years, 4 to 6 years, ≥ 7 years), season (summer, autumn, winter, spring), and breed (Friesland, Jersey, crossbreed) as fixed effects, and origin of animal (coastal, inland) as the random effect, on the severity of fluke infection. The response variable, fluke infection (continuous variable), was, in this case, the number of liver flukes recovered from the carcass of each bovine. The effect of fluke infection on carcass weight (continuous data for each animal) or body condition (scale 1 to 5) was also determined using separate negative binomial regression models. Statistical analyses were performed at 5% significance level using R software version 4.0.5. Data for BCS was square-root transformed to get normal distribution and subsequently subjected to the PROC GLM. Outliers were set to missing, and the data was re-run to determine if any new outlier appeared. PROC FREQ was used to generate frequencies for the severity index of the condemned livers. The chi-squared test was employed to determine the relation between fluke severity index and geographic origin, age, genotype, and body condition scores. Pearson’s correlation was used to determine the relationship between categorical variables.
Results
Liver fluke severity significantly increased with age (P < 0.0001); cattle ≥ 7 years old (mean ± standard deviation: 59.93 ± 6.42) and those 4 to 6 years old (49.78 ± 9.98) had 2.7 times and 1.9 times, respectively, higher infection than those 2 to 3 years old (27.55 ± 13.56) (Table 1). Liver fluke infection was significantly the highest in summer, followed by autumn and winter, and least in spring (Table 1). Crossbred and Friesland cattle had 1.1 times higher (P < 0.0001) liver fluke infection than Jersey cattle.
Negative binomial regression analyses of the effect of liver fluke infection on carcasses weight or body condition scores showed that with one unit increase in fluke infection, carcass weight, and body condition score decreased by 0.99 units (1% reduction) (P < 0.0001) and 0.97 units (3% reduction) (P < 0.0001), respectively.
The highest prevalence was observed in cattle from the coastal regions East London, Tsitsikamma, and Port Elizabeth (73.6%), compared to those coming from the inland region (Queenstown) (16.4%). The highest prevalence was observed in cattle slaughtered in CA1 (53.5%), followed by CA2 (30.1%) and CA3 (16.4%) (Table 2). Fluke infection rate varied with season. High fluke infection rates were more frequently observed in summer (P < 0.05) than in other seasons, while moderate infection rates were more commonly observed in autumn (P < 0.05) compared with other seasons. Heavy infections were more in summer in all abattoirs (CA1: 55.1%, CA2: 48%, CA3: 46.2%). On the other hand, mild infections were more in winter in all abattoirs (CA1: 43.2%, CA2: 37.5%, CA3: 41.2%). In autumn, moderate infection was the highest (40.3%, 40.2%, and 52.5%), at CA1 and CA2, and CA3, respectively (Table 3).
Light infections were observed more frequently (P < 0.05) in crossbred (46.9%) followed by Friesland (39.5%), and then Jersey (30.9%) cattle. Moderate infections were observed more frequently (P < 0.05) in Jersey (28.0%), followed by Friesland (15.4%), and then crossbred (1.9%) cattle. Severe infections were more frequent (P < 0.05) in Friesland (45.1%), followed by Jersey (41.9%), and then crossbred (31.7%) cattle. The fluke infection rate was significantly higher (P < 0.05) in adult cattle (58.5%) compared with young ones (58.5%). Moderate infections were observed more frequently (P < 0.05) in young (60.8%) as compared to old (1.7%) cattle. Light infections were observed more frequently in old (39.8%) than young (28.7%) cattle (Table 4).
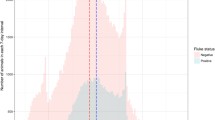
A moderate infection in cattle was observed in all abattoirs. However, infection was highly significant (P < 0.05) in cattle with good body condition scores in all three abattoirs (Fig. 2). Heavy infections were observed in cattle with poor body condition scores in CA3. Mild infections were observed more in cattle with moderate body condition scores in CA3 and CA1 and less in CA2 (Table 5). Moderate infections were observed frequently in grade B (61.5%) than AB (39.0) and C grades, respectively. Severe infections were observed more frequently (P < 0.01) in grade C cows than in AB and B grades (Table 6). Season was significantly (P < 0.05) positively correlated with BCS and carcass class and negatively correlated with severity index and liver pathology (Fig. 3A–C). Significant (P < 0.05) positive correlations were observed between carcass class and the animal’s age. A negative correlation (P < 0.05) was observed between liver pathology and body condition score (Table 7).
Discussion
Fluke infestation is a problem in many countries, including South Africa, leading to the culling and slaughter of dairy cattle (Jaja et al. 2017a, b). Abattoirs are surveillance points and serve as a critical source of knowledge of the prevalence of diseases such as parasitic illnesses (Yusuf et al. 2016; Jaja et al. 2017a; Mochankana and Robertson 2018; Tulu 2018; Villa-Mancera and Reynoso-Palomar 2019; Ahmad et al. 2020). The documented prevalence from the current study showed some differences in Fasciola infection among the three abattoirs. It was further noted that cattle slaughtered at East London abattoir had the highest Fasciola spp. than those slaughtered in Queenstown and Port Elizabeth abattoirs. The reason for such differences could be based on the source of animal origin or number of animals slaughtered per day. Most cattle come from different catchment areas (Jaja et al. 2017a). These catchment areas belong to different agro-climatic zones with fluctuating temperatures and rainfall. Rainfall, solar radiation, and global warming are common conditions promoting the distribution of Galba truncatula and Radix natalensis, the intermediate host of Fasciola spp. (Pfukenyi et al. 2005; Caron et al. 2017; Jaja et al. 2017a; Celi-Erazo et al. 2020; Malatji and Mukaratirwa 2020; Malatji et al. 2020).
The highest fasciolosis intensity observed in summer and autumn compared to winter and spring might be attributed to high temperatures and rainfall required for larval development and the swimming of metacercariae on pastures. Similar studies found that fluke burden was significantly higher during summer than other seasons (Jaja et al. 2017a; Byrne et al. 2018; Nyirenda et al. 2019; Isah 2019). These studies indicated that the summer season is characterized by high rainfall and relative humidity, providing excellent and favourable conditions for the intermediate host snail. High rainfall and relative humidity allow the replication of the snail hosts and infective stage of the disease’s life cycle (metacercariae) (Jaja et al. 2017a; Martin and Cabrera 2018; Kelley et al. 2020; Thi et al. 2021). Therefore cattle herds might be exposed to highly contaminated pastures during summer (Valero et al. 2018; Villa-Mancera and Reynoso-Palomar 2019). The current results differ from those reported in Northern Ireland, in which the fluke burden was very high in winter (Byrne et al. 2018). A related study did not find the significance of the season (Arias-Pacheco et al. 2020). Such knowledge gaps and findings from this study warrant further research on the influence of season on severity index in different production systems.
The overall prevalence of 39.1% observed for fluke infection in the current study was moderate in KwaZulu-Natal. It was also higher than other catchment areas of the Eastern Cape Province. KwaZulu-Natal has a humid subtropical climate and a subtropical oceanic highland climate while the ECP, which comprises parts of the study site (Queenstown and East London), has a warm and cold semi-arid climate and a temperate oceanic climate (Kottek et al. 2006; Jaja et al. 2017a). Hence, the differences in the fluke infection among the catchment areas in the current study could be attributed to different altitudes or ecological zones with high precipitation (Ortiz et al. 2013; Pinilla et al. 2020). Furthermore, the ECP has been reported to experience shortages of veterinarians thus compromising the quality of primary animal health programmes at the herd level (Jaja et al. 2017a). The current study results were in line with a study conducted in Malang district, Indonesia. The study reported that climatic factors such as rainfall and temperatures varied with regions. These conditions favour and influence the rate of distribution and prevalence of snail-borne disease in each catchment (Tulu and Gebeyehu 2018).
Interestingly, in the current study, adult cattle had a significantly higher infection rate compared to other age groups (4–6 and 2–3 years of age). This may reflect age-related resistance to fasciolosis, in which some young cattle are less susceptible to liver fluke infection than the adults. Adult cows’ high frequency of infections deserves attention as this can affect production and increase culling earlier in their productive years. These results align with those of Nyirenda et al. (2019), who reported a high level of pathology in matured cows. This might be due to prolonged exposure to metacercariae-contaminated pastures most likely causing heavy infections in adult cows compared to young cows. Another possible reason could be the decline in the level of immunity to liver fluke as the animal grows older. The findings obtained in this study agree with previous studies reported worldwide (Pfukenyi et al. 2005; Howell et al. 2015; Jaja et al. 2017a, b; Mochankana and Robertson 2018; de Costa et al., 2019; Kelly et al. 2019; Khan, 2020).
The current study revealed a higher occurrence of heavy fluke infections or severity index, which led to the high condemnation of the livers in dairy cattle with poor body condition scores and affected carcass quality or class and weights. Chronic fasciolosis infection has been reported to lead to liver damage, anaemia, and weight loss (Derso and Genet 2015; Yusuf et al. 2016; Jaja et al. 2017a; Mehmood et al. 2017; Alemu 2019; Ayad et al. 2019; Abdel Al-Hakeem and Omar 2020; Yesuf et al. 2020). Hence, association between poor body condition, carcass class, and high fluke intensity is not surprising. These results are similar to those reported in South Africa (Jaja et al. 2017a), Botswana (Mochankana and Robertson, 2018), Pakistan (Khan 2020), Zambia (Nyirenda et al. 2019), and Brazil (da Costa et al. 2019). These studies reported a positive association between heavily affected animals and poor body condition scores.
The current study also revealed that fluke infection rates varied with the breed. Friesland and crossbred cows had the highest fluke burden compared to Jersey cows. The association between breed and fluke intensity reported in the current study is in line with those reported in Denmark (Takeuchi-Storm et al. 2017; Ghodsian et al. 2019). The Danish studies reported a higher prevalence of fasciolosis in Danish Holstein than crossbreds and other breeds reared across Denmark. This could be attributed to breed intolerance and the environment. Moreover, the information mentioned above would influence breed selection and reduce veterinary costs. Moreover, the results from the current study are in line with those reported elsewhere in the UK, Peru, and Turkey (Bostanci and Ouz 2017; Hayward et al. 2021b; Diaz-Quevedo et al. 2021).
Conclusion
The present study confirmed a high fluke infection in dairy cattle with poor body condition scores in three abattoirs in Eastern Cape Province, South Africa. These results indicate that liver infection is responsible for losing body weight, reduced carcass weights, carcass quality, and often significant production losses. Several risk factors associated with the high prevalence were identified in the study, including geographic origin, age, genotype, and season. The resistance of trematode species to anthelmintic remains a concern in dairy farming in South Africa. Therefore, a combination of good pasture management and holistic grazing management is highly advised to control the infection rate in dairy herds. Furthermore, proper administration of chemoprophylaxis is also recommended to maintain animal health, and modern diagnostic tools to monitor field prevalence at the herd level are crucial. In addition, farmer training and on-farm studies should be conducted to determine the prevalence of fasciolosis and associated risk factors in dairy cattle.
References
Abdel Al-Hakeem S, Omar M (2020) Ovine fascioliasis: environmental epidemiology and meta-analysis of the prevalence, agro-ecological and economic factors in five provinces of the Nile Delta region of Egypt. Damanhour J Vet Sci 3:23–31
Ahmad I, Yakubu Y, Chafe UM et al (2020) Prevalence of fasciolosis (liver flukes) infection in cattle in Zamfara, Nigeria: a slaughterhouse surveillance data utilizing post-mortem examination. Vet Parasitol Reg Stud Reports 22:100483. https://doi.org/10.1016/j.vprsr.2020.100483
Alemu A (2019) Prevalence of dairy cattle fasciolosis in and around Wolayta Sodo, Southern Ethiopia. J Dairy Res Technol 2:1–6. https://doi.org/10.24966/drt-9315/100010
Arias-Pacheco C, Lucas JR, Rodríguez A et al (2020) Economic impact of the liver condemnation of cattle infected with Fasciola hepatica in the Peruvian Andes. Trop Anim Health Prod 52:1927–1932. https://doi.org/10.1007/s11250-020-02211-y
Ayad A, Benhanifia M, Balla E-H et al (2019) A retrospective survey of fasciolosis and hydatidosis in domestic ruminants based on abattoirs’ data in Bejaia province, Algeria. Retrospektivna Stud Fascioloze i Hidatidoze Domaćih Preživara Prema Pod Iz Klaonica u Prov Bejaia u Alžiru 68:47
Bloemhoff Y, Forbes A, Danaher M et al (2015) Determining the prevalence and seasonality of fasciola hepatica in pasture-based dairy herds in Ireland using a bulk tank milk ELISA. Ir Vet J 68:16. https://doi.org/10.1186/s13620-015-0042-5
Bostanci SB, Ouz A (2017) Prevalence of Fasciola hepatica in cattle in Van. Turkey Acta Sci Vet 45:1–7
Byrne AW, Graham J, Mcconville J et al (2018) Seasonal variation of Fasciola hepatica antibodies in dairy herds in Northern Ireland measured by bulk tank milk ELISA. Parasitol Res 117:2725–2733. https://doi.org/10.1007/s00436-018-5961-0
Caron Y, Celi-Erazo M, Hurtrez-Boussès S et al (2017) (Mollusca, Gastropoda) an intermediate host of Fasciola hepatica (Trematoda, Digenea) in Ecuador? Parasite 24:24. https://doi.org/10.1051/parasite/2017026
Celi-Erazo M, Alda P, Montenegro-Franco M et al (2020) Prevalence of Fasciola hepatica infection in Galba cousini and Galba schirazensis from an Andean region of Ecuador. Vet Parasitol Reg Stud Reports 20:100390. https://doi.org/10.1016/j.vprsr.2020.100390
Charlier J, van der Voort M, Kenyon F et al (2014) Chasing helminths and their economic impact on farmed ruminants. Trends Parasitol 30:361–367. https://doi.org/10.1016/j.pt.2014.04.009
Charlier J, Rinaldi L, Musella V, et al (2020) Initial assessment of the economic burden of major parasitic helminth infections to the ruminant livestock industry in Europe. Prev Vet Med 182. https://doi.org/10.1016/J.PREVETMED.2020.105103
da Costa RA, Corbellini LG, Castro-Janer E, Riet-Correa F (2019) Evaluation of losses in carcasses of cattle naturally infected with Fasciola hepatica: effects on weight by age range and on carcass quality parameters. Int J Parasitol 49:867–872. https://doi.org/10.1016/j.ijpara.2019.06.005
DAFF (2019) Department of agriculture, forestry of the Republic of South Africa “Animal Production” https//www.daff.gov.za/Branches/Agricultural Production/Health Food safety/ Animal Production/Livestock Production[Accessed April 16 2021]. https://static.pmg.org.za/DAFF_APP_2019_2020_WEB.pdf. Accessed 16 Apr 2021
Derso S, Genet A (2015) Prevalence of bovine fasciolosis and its associated risk factor in and around Dangila District, Awi Administration Zone, Northwestern Ethiopia. Eur J Biol Sci 7:114–119. https://doi.org/10.5829/idosi.ejbs.2015.7.03.94228
Diaz-Quevedo C, Frias H, Cahuana GM et al (2021) High prevalence and risk factors of fascioliasis in cattle in Amazonas. Peru Parasitol Int 85:102428. https://doi.org/10.1016/J.PARINT.2021.102428
Dupuy C, Morignat E, Maugey X et al (2013) Defining syndromes using cattle meat inspection data for syndromic surveillance purposes: a statistical approach with the 2005–2010 data from ten French slaughterhouses. BMC Vet Res 9:88. https://doi.org/10.1186/1746-6148-9-88
Elelu N, Eisler MC (2018) A review of bovine fasciolosis and other trematode infections in Nigeria. J Helminthol 92:128–141. https://doi.org/10.1017/S0022149X17000402
Ghodsian S, Rouhani S, Fallahi S et al (2019) Detection of spiked Fasciola hepatica eggs in stool specimens using LAMP technique. Iran J Parasitol 14:387–393
Hayward AD, Skuce PJ, McNeilly TN (2021a) Tolerance of liver fluke infection varies between breeds and producers in beef cattle | Elsevier Enhanced Reader. In: Th Int. J. Anim. Biosci. https://reader.elsevier.com/reader/sd/pii/S1751731120301282?token=0E8C660002591B1E3A11585E283B80044E8537157E7D44123E6F5FFA87696EDB21523B85D3238C7F0C1FD9DE8C095B82&originRegion=eu-west-1&originCreation=20210708100357. Accessed 8 Jul 2021
Hayward AD, Skuce PJ, McNeilly TN (2021) The influence of liver fluke infection on production in sheep and cattle: a meta-analysis. Int J Parasitol 51:913–924. https://doi.org/10.1016/j.ijpara.2021.02.006
Howell A, Baylis M, Smith R et al (2015) Epidemiology and impact of Fasciola hepatica exposure in high-yielding dairy herds. Prev Vet Med 121:41–48. https://doi.org/10.1016/j.prevetmed.2015.05.013
Howell A, Mugisha L, Davies J et al (2012) Bovine fasciolosis at increasing altitudes: Parasitological and malacological sampling on the slopes of Mount Elgon, Uganda. Parasites Vectors 5:196. https://doi.org/10.1186/1756-3305-5-196
Isah UM (2019) Studies on the prevalence of fascioliasis among ruminant animals in northern Bauchi state, north-eastern Nigeria. Parasite Epidemiol Control 5:e00090. https://doi.org/10.1016/j.parepi.2019.e00090
Jaja IF, Mushonga B, Green E, Muchenje V (2017) Seasonal prevalence, body condition score and risk factors of bovine fasciolosis in South Africa. Vet Anim Sci 4:1–7. https://doi.org/10.1016/j.vas.2017.06.001
Jaja IF, Mushonga B, Green E, Muchenje V (2017) Financial loss estimation of bovine fasciolosis in slaughtered cattle in South Africa. Parasite Epidemiol Control 2:27–34. https://doi.org/10.1016/j.parepi.2017.10.001
Jenkins D, Baker A, Porter M et al (2020) Wild fallow deer (Dama dama) as definitive hosts of Fasciola hepatica (liver fluke) in alpine New South Wales. Aust Vet J 98:546–549. https://doi.org/10.1111/AVJ.13001
Kaewkong W, Choochote W, Kanla P et al (2012) Chromosomes and karyotype analysis of a liver fluke, Opisthorchis viverrini, by scanning electron microscopy. Parasitol Int 61:504–507. https://doi.org/10.1016/J.PARINT.2012.03.008
Kelley JM, Rathinasamy V, Elliott TP et al (2020) Determination of the prevalence and intensity of Fasciola hepatica infection in dairy cattle from six irrigation regions of Victoria, South-eastern Australia, further identifying significant triclabendazole resistance on three properties. Vet Parasitol 277:109019. https://doi.org/10.1016/j.vetpar.2019.109019
Kelley JM, Stevenson MA, Rathinasamy V, et al (2021) Analysis of daily variation in the release of faecal eggs and coproantigen of Fasciola hepatica in naturally infected dairy cattle and the impact on diagnostic test sensitivity. Vet Parasitol 109504. https://doi.org/10.1016/j.vetpar.2021.109504
Kelly RF, Mazeri S, Hartley C, et al (2019) Assessing the performance of a Fasciola gigantica serum antibody ELISA to estimate prevalence in cattle in Cameroon. BMC Vet Res 15. https://doi.org/10.1186/s12917-018-1762-z
Khan NU (2020) Epidemiological study of bovine fasciolosis using coprological technique in district Mardan, Khyber Pakhtunkhwa, Pakistan. Pure Appl Biol 9:455–463. https://doi.org/10.19045/bspab.2020.90050
Kottek M, Grieser J, Beck C et al (2006) World map of the Köppen-Geiger climate classification updated. Meteorol Zeitschrift 15:259–263. https://doi.org/10.1127/0941-2948/2006/0130
Kurzy´nska A, Kurzy´nska- Kokorniak K (2021) Comparison of the benzanthrone luminophores: they are not equal for rapid examination of Parafasciolopsis fasciolaemorpha (Trematoda: Digenea) Ilze Rubenina *, Inese Gavarane, Elena Kirilova, Ligita Mezaraupe and Muza Kirjusina. Biomolecule 11:1–15. https://doi.org/10.3390/biom11040598
Kusumarini SR, Permata FS, Widyaputri T, Prasetyo D (2020) Prevalence of fasciolosis emphasis on age, origin, body condition and post mortem by geographic information systems on sacrificial examination in Malang District - East Java. J Phys Conf Ser 1430. https://doi.org/10.1088/1742-6596/1430/1/012025
Lacto Data (2016) A commodity value chain analysis of dairy. https://www.mpo.co.za/wp-content/uploads/2020/09/AA-P093-124-LACTO-DATA-MAY20-FINAL-TO-PRINT.pdf. Accessed 20 Dec 2020
Lacto data (2017) Lacto data. https://www.mpo.co.za/wp-content/uploads/2020/09/AA-P093-124-LACTO-DATA-MAY20-FINAL-TO-PRINT.pdf. Accessed 20 Dec 2020
Lacto Data (2019) Suid-Afrikaanse Melkprosesseer South African Milk Processors’ Organisation dersorganisasie A Milk SA publication compiled by the Milk Producers’ Organisation, https://www.mpo.co.za/wp-content/uploads/2020/09/AA-P093-124-LACTO-DATA-MAY20-FINAL-TO-PRINT.pdf. Accessed 20 Dec 2020
Lacto Data (2020) Suid-Afrikaanse Melkprosesseer South African Milk Processors’ Organisation dersorganisasie A Milk SA publication compiled by the Milk Producers’ Organisation. https://www.mpo.co.za/wp-content/uploads/2020/09/AA-P093-124-LACTO-DATA-MAY20-FINAL-TO-PRINT.pdf. Accessed 20 Dec 2020
Malatji MP, Mukaratirwa S (2020) Molecular detection of natural infection of Lymnaea (Pseudosuccinea) columella (Gastropoda: Lymnaeidae) with Fasciola gigantica (Digenea: Fasciolidae) from two provinces of South Africa. J Helminthol 94:1–694. https://doi.org/10.1017/S0022149X19000129
Malatji MP, Pfukenyi DM, Mukaratirwa S (2019) Fasciola species and their vertebrate and snail intermediate hosts in East and Southern Africa: a review. J Helminthol 94:1–11. https://doi.org/10.1017/S0022149X19000531
Martin GL, Cabrera EC (2018) Morphological characterization of emerging cercariae among Lymnaeid snails from Barangay Cawongan, Padre Garcia, Batangas. J Parasitol Res 5241217:1–13. https://doi.org/10.1155/2018/5241217
Mas-Coma S, Bargues MD, Valero MA (2020) Erratum: human fascioliasis infection sources, their diversity, incidence factors, analytical methods and prevention measures (Parasitology (2018) 145 (1665–1699) DOI: 10.1017/S0031182018000914). Parasitology 147:601. https://doi.org/10.1017/S0031182020000256
Mazeri S, Rydevik G, Handel I et al (2017) Estimation of the impact of Fasciola hepatica infection on time taken for UK beef cattle to reach slaughter weight. Sci Rep 7:1–14. https://doi.org/10.1038/s41598-017-07396-1
Meat Safety Act No.40 (2000) Department of Agriculture, Forestry and Fisheries, Republic of South Africa
Mehmood K, Zhang H, Sabir AJ et al (2017) A review on epidemiology, global prevalence and economical losses of fasciolosis in ruminants. Microb Pathog 109:253–262
Mochankana ME, Robertson ID (2018) Cross-sectional prevalence of Fasciola gigantica infections in beef cattle in Botswana. Trop Anim Health Prod 50:1355–1363. https://doi.org/10.1007/s11250-018-1568-9
Mpakama T, Chulayo AY, Muchenje V (2014) Bruising in slaughter cattle and its relationship with creatine kinase levels and beef quality as affected by animal related factors. Asian-Australasian J Anim Sci 27:717–726. https://doi.org/10.5713/ajas.2013.13483
Nicholson MJ, Butterworth MH (1986) A guide to body condition scoring of zebu cattle. International Livestock Center for Africa- ILCA, Addis Ababa, Ethiopia, pp 4–16
Nyirenda SS, Sakala M, Moonde L, et al (2019) Prevalence of bovine fascioliasis and economic impact associated with liver condemnation in abattoirs in Mongu district of Zambia. BMC Vet Res 15:33. https://doi.org/10.1186/s12917-019-1777-0
Ortiz P, Scarcella S, Cerna C et al (2013) Resistance of Fasciola hepatica against Triclabendazole in cattle in Cajamarca (Peru): a clinical trial and an in vivo efficacy test in sheep. Vet Parasitol 195:118–121. https://doi.org/10.1016/j.vetpar.2013.01.001
Pfukenyi DM, Mukaratirwa S, Willingham AL, Monrad J (2005) Epidemiological studies of amphistome infections in cattle in the highveld and lowveld communal grazing areas of Zimbabwe. Onderstepoort J Vet Res 72:67–86. https://doi.org/10.4102/ojvr.v72i1.224
Pinilla JC, Florez AA, Orlandoni G, et al (2020) Current status of prevalence and risk factors associated with liver fluke Fasciola hepatica in cattle raised in different altitudinal regions of Colombia. Vet Parasitol Reg Stud Reports 22:100487. https://doi.org/10.1016/j.vprsr.2020.100487
Ploeger HW, Ankum L, Moll L et al (2017) Presence and species identity of rumen flukes in cattle and sheep in the Netherlands. Vet Parasitol 243:42–46. https://doi.org/10.1016/J.VETPAR.2017.06.009
Radfar MH, Nourollahi-Fard SR, Mohammadyari N (2015) Bovine fasciolosis: prevalence, relationship between faecal egg count and worm burden and its economic impact due to liver condemnation at Rudsar abattoir, Northern Iran. J Parasit Dis 39:522–525. https://doi.org/10.1007/s12639-013-0389-z
Rehman A ur (2020) Prevalence of F. hepatica infestation and pathological examination in sheep (Ovis aries) in Dera Ismail Khan. Pure Appl Biol 9:105–111. https://doi.org/10.19045/bspab.2020.90013
Soji Z, Mabusela SP, Muchenje V (2015) Associations between animal traits, carcass traits and carcass classification in a selected abattoir in the Eastern Cape Province, South Africa. South African J Anim Sci 45:278–288. https://doi.org/10.4314/sajas.v45i3.6
Springer A, Jordan D, Kirse A, et al (2021) Seroprevalence of major pasture-borne parasitoses (gastrointestinal nematodes, liver flukes and lungworms) in German dairy cattle herds, association with management factors and impact on production parameters. 11:2078. https://doi.org/10.3390/ani11072078
Stats SA (2020) Abstract of Agricultural Statistics SA. https://www.dalrrd.gov.za/Portals/0/Statistics and Economic Analysis/Statistical Information/Abstract 2020_organized.pdf. Accessed 16 Apr 2021
Takeuchi-Storm N, Denwood M, Hansen TVA et al (2017) Farm-level risk factors for Fasciola hepatica infection in Danish dairy cattle as evaluated by two diagnostic methods. Parasit Vectors 10:1–11. https://doi.org/10.1186/s13071-017-2504-y
Takeuchi-Storm N, Denwood M, Petersen HH et al (2018) Patterns of Fasciola hepatica infection in Danish dairy cattle: implications for on-farm control of the parasite based on different diagnostic methods. Parasit Vectors 11:1–18. https://doi.org/10.1186/s13071-018-3248-z
Thi P, Nguyen X, Van HH et al (2021) Insights on foodborne zoonotic trematodes in freshwater snails in North and Central Vietnam. Parasitol Res 120:949–962. https://doi.org/10.1007/s00436-020-07027-1/Published
Tulu D (2018) Study of prevalence and associated risk factors of bovine fasciolosis in and around Ambo District Abattoir and field survey Western Ethiopia. Biomed J Sci Tech Res 11:8515–8520. https://doi.org/10.26717/bjstr.2018.11.002100
Tulu D, Gebeyehu S (2018) Study of prevalence and associated risk factors of bovine fasciolosis in Jimma Horro District of Kellem Wollega Zone, Western Ethiopia. Arch Vet Sci Med 01:9–18. https://doi.org/10.26502/avsm.002
Valero MA, Bargues MD, Calderón L et al (2018) First phenotypic and genotypic description of Fasciola hepatica infecting highland cattle in the state of Mexico, Mexico. Infect Genet Evol 64:231–240. https://doi.org/10.1016/j.meegid.2018.06.032
Villa-Mancera A, Reynoso-Palomar A (2019) High prevalence, potential economic impact, and risk factors of Fasciola hepatica in dairy herds in tropical, dry and temperate climate regions in Mexico. Acta Trop 193:169–175. https://doi.org/10.1016/j.actatropica.2019.03.005
Yesuf M, Erara M, Kenubih A et al (2020) Hemato-biochemical profiles of sheep infected with fasciolosis in comparison with healthy controls. Online J Anim Feed Res 10:71–75. https://doi.org/10.36380/scil.2020.ojafr10
Yusuf M, Ibrahim N, Tafese W, Deneke Y (2016) Prevalence of bovine fasciolosis in Municipal Abattoir of Haramaya, Ethiopia. 48:
Zewde A, Bayu Y, Wondimu A (2019) Prevalence of bovine fasciolosis and its economic loss due to liver condemnation at Wolaita Sodo Municipal Abattair, Ethiopia. Vet Med Int 9572373:1–7. https://doi.org/10.1155/2019/9572373
Acknowledgements
We are grateful to the participating abattoirs for granting permission to use their facility for data collection.
Funding
This study received funding from the National Research Foundation (NRF) (animal product safety: project grant no. 140702).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Section Editor: Dante Zarlenga
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Mpisana, Z., Jaja, I.F., Byaruhanga, C. et al. Body condition scores, fluke intensity, liver pathology, and carcass quality of different dairy cattle genotypes infected with Fasciola species at high throughput abattoirs in South Africa. Parasitol Res 121, 1671–1682 (2022). https://doi.org/10.1007/s00436-022-07504-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-022-07504-9