Abstract
The research of Mendel, born two centuries ago, still has many direct implications for our everyday clinical work. He introduced the terms “dominant” and “recessive” characters and determined their 3:1 ratio in the offspring of heterozygous “hybrid” plants. This distribution allowed calculation of the number of the phenotype-determining “elements,” i.e., the alleles, and has been used ever since to prove the monogenic origin of a disorder. The Mendelian inheritance of monogenic kidney disorders is still of great help in distinguishing them from those with multifactorial origin in clinical practice. Inheritance of most monogenic kidney disorders fits to Mendel’s observations: the equal contribution of the two parents and the complete penetrance or the direct correlation between the frequency of the recessive character and the degree of inbreeding. Nevertheless, beyond the truth of these basic concepts, several observations have expanded their genetic characteristics. The extreme genetic heterogeneity, the pleiotropy of the causal genes and the role of modifiers in ciliopathies, the digenic inheritance and parental imprinting in some tubulopathies, and the incomplete penetrance and eventual interallelic interactions in podocytopathies, reflect this expansion. For all these reasons, the transmission pattern in a natural setting may depend not only on the “character” but also on the causal gene and the variant. Mendel’s passion for research combined with his modest personality and meticulous approach can still serve as an example in the work required to understand the non-Mendelian universe of genetics.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Genetic research has revolutionized our understanding of the etiology, pathophysiology, and inheritance of kidney disorders during the last decades. The path from domesticating animals to the recent identification of hundreds of causal genes in monogenic kidney disorders was nevertheless long. One of the most important steps in this evolution was to understand that binomial traits are determined by two alleles of a gene, thanks to the work of Johann Gregor Mendel. Over 200 years after his birth of July 20, 1822, in Heinzendorf, Silesia, it is a good time to contemplate the influence of his observations on pediatric nephrology.
Humans have been domesticating animals for 12 millennia by selecting advantageous traits to be passed on to successive generations [1]. Thus, heredity of various traits has been known and consciously utilized for a long time by mankind. How transmission of traits from one generation to another occurs was nevertheless a matter of debate even in the nineteenth century.
The problem was formulated by Cyrill Franz Napp, the abbot of the Augustinian monastery in Brno (known as Brünn in German) between 1824 and 1867 as follows: “The question for discussion should not be the theory and process of breeding, but what is inherited and how?” [2]. Napp was also the one who welcomed Gregor Mendel into the Augustinian community as a young novice, little knowing that many years later Mendel would provide the answer to his fundamental question.
At this time, it was already recognized that all living organisms are made up of cell(s) and that during fertilization a specific cellular organelle (named nucleus by Robert Brown in 1833 [3]) is indispensable. However, the real significance and role of Brown’s nuclei were not yet clear.
The questions of this period are well reflected in the speculation of the Czech physiologist Jan Evangelista Purkyně (Purkinje, 1787–1868) about fertilization in 1834 when his wife was pregnant with their second son. He suggested that “parental characteristics are reduced in the’germs’ to’pure quality’ by a process he defined as’involution’… The meeting of the parental germs leads to’evolution’ in which the embryo develops to a form revealing the traits of both parents” [2, 4]. Purkinje was the first head of the first Physiology Department in the world in Breslau (now Wrocław, Poland). He visited the Augustinian convent on several occasions between 1835 and 1850, and may have thus influenced the work of the young Mendel [2].
The prerequisites of Mendel’s results: the thoroughly planned research design
Crossbreeding experiments had been carried out by many predecessors of Mendel. The British Thomas Andrew Knight studied a variety of plants using artificial pollination, including the garden pea, and made similar observations to Mendel [5]. What made Mendel’s work unique is the meticulous elaboration of his research method, which allowed him to perform calculations and gave him reproducible results. As Mendel wrote in 1865: “The value and utility of any experiment are determined by the fitness of the material to the purpose for which it is used” [6]. He selected the pea (Pisum sativum) because it possesses easily recognizable physical characteristics; its flowers are predominantly self-pollinating, i.e., well protected from foreign pollens; it has a relatively short life cycle and is easily cultured. Furthermore, its artificial fertilization, though painstakingly elaborate, was nearly always a successful process.
Once the pea was selected, Mendel devoted 2 years selecting the traits to study. Brilliantly, out of the 34 initially studied characteristics, he excluded those which had a “‘more or less’ nature” and focused only those that “stand out clearly and definitely” and were uniform in the hybrids [6]. Selecting such binomial traits was essential for Mendel to achieve his results later.
To study the effects of inheritance as purely as possible, Mendel had to successfully overcome a number of biasing factors: natural selection, foreign pollination, and the major enemy, the pea weevil (Bruchus pisi). He even used controls: “For each experiment a number of pot plants were placed during the blooming period in a greenhouse, to serve as control plants for the main experiment in the open as regards possible disturbance by insects.” To counterbalance the effect of natural selection, he lifted the undersized plants and transferred them to a special bed. “This precaution was necessary, as otherwise they would have perished through being overgrown by their tall relatives” [6]. Finally, he covered the flowers with paper bags to prevent unwanted fertilization.
Introduction of the terms ‘dominant’ and ‘recessive’
For all seven selected traits, Mendel crossed peas that differed in a single trait and found that the offspring of the two pure lines — the “hybrids” — were uniform. Moreover, not only were they uniform but they all exhibited the same trait as one of the parental plants. “In the case of each of the seven crosses the hybrid-character resembles that of one of the parental forms so closely that the other escapes observation completely…” [6]. Mendel introduced the terms “dominant” and “recessive” to describe the “power relations” of the two forms: “Henceforth in this paper those characters which are transmitted entire, or almost unchanged in the hybridization, and therefore in themselves constitute the characters of the hybrid, are termed the dominant, and those which become latent in the process recessive. The expression recessive has been chosen because the characters thereby designated withdraw or entirely disappear in the hybrids, but nevertheless reappear unchanged in their progeny.” The terms “dominant” and “recessive” were thus initially introduced to typify the phenotypes. In medical terms, both the disease phenotype and the disease-causing allele can be labelled dominant (over the wild type), but only the phenotype may be transmitted in a dominant fashion. Each allele is transmitted with a 50% probability to the offspring, and the disease (and not the allele) is transmitted in function of the allele’s dominant or recessive nature.
The equal contribution of the two parents
In order to compare the parental roles in phenotype determination, Mendel performed reciprocal crossings. He found that it was irrelevant whether the dominant character belonged to the seed or to the pollen plant, and thus concluded that each sex contributes equally to the characteristics of the offspring [6]. In perfect accordance with Mendel’s observations, there is no difference between paternal and maternal origin in determining the risk of most of the kidney diseases, since the two alleles of the causal autosomal genes are in general equally expressed. Some autosomal genes are nevertheless expressed predominantly or even exclusively from one allele, which can be either randomly assigned [7] or parental-specific, as recognized in Prader-Willi and Beckwith-Wiedemann syndromes [8, 9]. Parental imprinting can lead to unequal parental effect, like the kidney abnormalities linked to the maternal loss of the 11p15.5 region in Beckwith-Wiedemann syndrome [10], or the resistance to parathyroid hormone in the proximal tubules, mainly due to the loss of the maternal GNAS expression or function in pseudohypoparathyroidism type I [11,12,13].
From the 3:1 ratio to diploidy
Mendel was the first who concentrated on the ratio of the different forms that appeared among the offspring of the hybrids after self-pollination. His method was meticulous and precise enough to properly determine the ratio of the recessive form that reappeared in the second generation. This is how he documented the famous 3:1 ratio of plants exhibiting dominant and recessive trait, which was the same in the case of all seven traits [6]. The dominant form never reappeared in the progeny of plants with the recessive form, even after six generations of self-pollination, indicating that they do not contain any dominant “‘elements,” and thus, the recessive form only appears if the “embryo is formed from two similar cells,” i.e., if only recessive ‘elements’ are present in the parental generation [6]. It could be thus posited that if the characters (1) are transmitted unchanged to the hybrids and to their progeny, (2) the two parents contribute equally to the offspring, (3) the recessive phenotype cannot appear unless only recessive alleles are present, and (4) the recessive phenotype is present in ¼ of the offspring of the hybrid generation, then the number of the phenotype-determining “elements” (= alleles) must be two, since (½)n = ¼, where ½ is the chance of inheriting the recessive allele in the offspring of the hybrids (which contain the two alleles in equal quantity), ¼ is the proportion of the recessive phenotype among the offspring of the hybrids, and “n” is the number of the alleles. Accordingly, Mendel used two letters as symbols of alleles, to demonstrate the crossing [6]. The Mendelian ratio thus became a synonym of the 3:1 distribution, and indicates that the character is determined by a single gene of a diploid organism. Whenever a new animal model of an autosomal recessive disease is established, the first step is to check the Mendelian ratio of the disease phenotype among the offspring of the heterozygous “hybrids” to confirm monogenic transmission, complete penetrance, and to exclude in utero lethality.
The changing face of dominance
Mendel crossed “pure” lines, each likely to be homozygous for the associated allele. One of the two alleles was dominant over the other, as shown by the phenotype of the hybrids. In the natural setting of Mendelian disorders, each gene has thousands of pathogenic and benign alleles. Despite the heterogeneity of alleles, they are similar in their “behavior”: pathogenic alleles of a gene responsible for an autosomal dominant disorder tend to be dominant over the wild-type allele. As always, there are exceptions. A dominant disorder may turn into recessive when hypomorphic variants in a gene of an autosomal dominant (AD) disorder do not cause symptoms on their own, but only when present on both alleles. Such an example is the PKD1 R2220W- or R3277C-associated “ADPKD” [14, 15]. These variants are recessive compared with the wild-type allele, and the severe PKD caused by them follows an autosomal recessive (AR) transmission pattern.
It is less likely that a typical AR disorder becomes dominant, as most AR disorders develop as a result of a loss-of-function that is rarely severe enough when only one of the two alleles is mutant. This has nevertheless been observed in some nephropathies, like the IFT140, NEK8, or NPHS2-associated ones. While their biallelic loss-of-function variants cause Mainzer-Saldino syndrome [16], renal-hepatic-pancreatic dysplasia [17,18,19], or steroid-resistant nephrotic syndrome [20], respectively, monoallelic variants of IFT140 and NEK8 were found causal in rare forms of ADPKD [21,22,23] and those of NPHS2 in focal segmental glomerulosclerosis (FSGS) [24]. The monoallelic variants were specific in NEK8 (i.e., p.R45W) [21, 22] and in NPHS2 (p.L330Vfs*15 with a premature stop in the last exon) [24], suggesting a dominant negative effect [21], but the IFT140 variants in mild ADPKD are intriguingly loss-of-function variants [23]. Such transformations of dominant or recessive phenotypes would have reversed the two forms of a trait in Mendel’s experiment, but would not have changed the 3:1 ratio of the dominant and recessive forms in the second generation.
The dominant/recessive nature of a variant could thus far be determined on the basis of their interaction with the wild-type allele. However, a variant that is dominant over one allele can be recessive compared with another one. An example for such interactions between alleles causing human disorders is the NPHS2 R229Q that is benign in the homozygous state [25], recessive compared with the A284V and dominant over the R138Q pathogenic variant. As a result, the [R229Q];[A284V] combination is pathogenic and the [R138Q];[R229Q] is benign [26, 27]. Interallelic interactions which render benign variants pathogenic change not only the priority of the alleles but also the ratio of the recessive and the dominant phenotype among the offspring of the heterozygous individuals. Accordingly, healthy individuals who are compound heterozygous for a benign and a pathogenic allele: [R138Q];[R229Q] and [wt];[A284V] may have offspring that show a 1:1 proportion of the healthy and diseased phenotype instead of the Mendelian 3:1.
Monogenic vs. polygenic
The seven phenotypic characters selected initially by Mendel were all monogenic [6]. His first observations thus corresponded to the monogenic inheritance, and “Mendelian” has become the synonym of monogenic in genetics. Later on, however, he also found characters, e.g., the flower colors of the runner bean (Phaseolus multiflorus) which did not conform to the 3:1 ratio:”…from the union of a white and a purple–red coloring a whole series of colors results, from purple to pale violet and white, the circumstance is a striking one that among 31 flowering plants only one received the recessive character of the white color.” Mendel nevertheless recognized that the variable characters can also be explained by the combined effect of multiple “characters”: “Even these enigmatic results, however, might probably be explained by the law governing Pisum if we might assume that the color of the flowers is a combination of two or more entirely independent colors, which individually act like any other constant character in the plant” [6].
Accordingly, whenever the transmission rate of a human disorder is not Mendelian, i.e., siblings with the same genotype at the primary locus present with discordant phenotypes, such as in PLCE1-associated nephrotic syndrome [28,29,30], an oligogenic inheritance can be assumed. Positing such inheritance is straightforward, but the identification of the additional loci can be challenging.
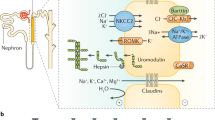
Digenic inheritance in kidney diseases has been rarely found. This would mean that the defect of a single gene is not pathogenic, but that of two is pathogenic with a complete penetrance. The hearing impairment associated with classic Bartter syndrome follows this inheritance pattern: patients with biallelic CLCNKB mutations develop hearing impairment if they also harbor biallelic mutations in the neighboring CLCNKA gene [31]. Hearing impairment develops only when the function of both chloride channels, encoded by the two genes, is lost. Mutations of both CLCNKB and CLCNKA also aggravate the severity of the tubular reabsorption defect, but this latter is penetrant already if any of the two genes is altered.
A similar inheritance was suggested in Bardet-Biedl syndrome (BBS) [32]. In a few families, biallelic mutations of a BBS gene were found to be penetrant only in siblings with a heterozygous mutation in a second BBS gene, leading to the proposition of “triallelic inheritance” [32]. Since several subsequent studies failed to confirm the widespread existence of this phenomenon, BBS is considered in the clinical practice to be a Mendelian, autosomal recessive disease [33,34,35,36,37].
A digenic inheritance was also described in Alport syndrome, even though the mutation in the second locus alters only the severity and the transmission risk of the disorder, but — apparently — not the penetrance [38, 39].
An oligogenic inheritance has been suggested in several extra-renal involvements, like the NPHP1-associated retinopathy and Joubert syndrome [40, 41], and even in less frequent subtypes of nephronophthisis itself [42]. While the extra-renal phenotype is clearly incompletely penetrant in siblings [40], nephronophthisis itself is considered to be autosomal recessive in clinical practice.
Overall, a causal oligogenic inheritance was more often suspected than actually demonstrated. Several misconceptions lay behind this discordance. First, the pathogenicity assessment of the sequence variants was more challenging at the beginning of this century, given the lack of large population reference sequences. The frequency of predicted-to-be pathogenic variants carried by each individual, therefore, was yet unknown [43]. This led to the classification of many benign variants as pathogenic. Furthermore, the extreme genetic heterogeneity of rare autosomal recessive disorders (BBS, nephronophthisis) [44, 45] was unexpected. And the higher the number of the causal genes of a disease with a given prevalence, the higher the rate of heterozygous individuals in the general population. Thus, despite the rarity of these disorders, the rate of heterozygous individuals with a truly causal pathogenic variant is in the order of percentage. It is thus perfectly possible to identify accidentally heterozygous well-known pathogenic variants in a second locus without a causal role, not even speaking about the more frequent, predicted-to-be-pathogenic, but in reality benign variants. Whenever predicted or known pathogenic variants were identified in multiple genes, it was tempting to speculate that they act in concert, and the disorder has an oligogenic nature. However, when the mutational load was properly compared between the patient and the general population, it turned out that the second-locus variants are not more frequent in BBS or Joubert syndrome, indicating that oligogenism is at least not a common mechanism in these disorders [46, 47].
An example of a truly polygenic/multifactorial kidney disorder was nevertheless provided by APOL1-associated FSGS, a disease typical in populations with West African ancestry. The APOL1 [S342G;I384M] (G1) and N388_Y389del (G2) alleles have evolved as the result of an evolutionary struggle against sleeping sickness caused by the parasite Trypanosoma brucei rhodesiense. Heterozygous individuals can neutralize this parasite, but at the cost of a lifetime FSGS risk of 4% in people with biallelic G1/G2 alleles [48, 49]. The risk is thus high, but substantially lower than the 100% seen in a Mendelian disease. In the case of an untreated HIV infection, the risk of HIV-associated nephropathy increases to 50% [49], demonstrating the decisive role of an environmental factor, unlike in a Mendelian disease.
The characteristics of Mendelian, oligogenic, and multifactorial disorders are summarized in Table 1.
Mendelian vs. non-Mendelian in clinical practice
There is an inverse relationship between the number of the causal alleles and the risk of disease transmission. Since Mendelian disorders are determined by only one or two alleles, at-risk relatives are frequently affected. The significant familial clustering, giving the typical dominant and recessive transmission patterns, made the monogenic origin of specific disorders recognizable long before the identification of the causal genes.
In contrast, uncommon multifactorial disorders, determined by numerous alleles (and also by environmental factors), rarely cluster in families, as the risk of disease transmission is significantly lower. The different familial clustering of monogenic and rare multifactorial disorders can help their differentiation in clinical practice. Distinguishing the monogenic Alport from the multifactorial IgA nephropathy or the monogenic podocytopathy from the immune-mediated nephrotic syndrome [50] can be markedly improved by knowing the family history or potentially by investigating the first-degree relatives. Their affected status is strongly supportive of a monogenic origin.
Importantly, more frequent polygenic disorders, like CAKUT with an incidence of 1:300–2400 [51,52,53], may affect several members of a family even without a monogenic origin. Influenced by genetic, epigenetic, and environmental factors [54], its higher incidence together with a monogenic origin in at least 15% of the families [54] explains the relatively high proportion of affected first-degree relatives (4–25%) of patients with CAKUT [55, 56].
Inbreeding and risk of homozygosity
Mendel performed cross-pollination only when crossing two pure lines. Their offspring, the hybrids, and the subsequent generations self-pollinated themselves, increasing the degree of inbreeding with each generation. Thus, the proportion of the “hybrids” decreased with each generation and the recessive phenotype became practically as frequent as the dominant after a few generations [6].
Inbreeding (self-pollination) does not change the allele frequency of the dominant and recessive alleles (assuming no natural selection), but does increase the relative frequency of the recessive phenotype from ¼ (q2) in the offspring of the hybrids (no inbreeding) to ~ ½ (F × q) in complete inbreeding, where q (ratio of the recessive allele) = ½ (since the two alleles were equally frequent in Mendel’s experiments), and F is the degree of inbreeding. After several self-pollinating generations (complete inbreeding), the value of F approaches 1.
In Mendel’s experiments, the proportion of the recessive phenotype only doubled secondary to inbreeding, since q was high (½), and therefore, the value of q2 and that of (F × q) did not differ by orders of magnitude. Similarly, in frequent autosomal recessive disorders, like in ARPKD, where q ~ 1/70 [57], the risk to the offspring of first-degree cousins (F = 1/16) to be affected secondary to inbreeding (F × q = 1/16 × 1/70) is only four times higher than the risk in a non-inbred population (q2 = 1/702). Accordingly, most of the children with ARPKD are compound heterozygous (and not homozygous) for the causal variants in populations with a low rate of consanguinity. In contrast, in extremely rare autosomal recessive disorders, where q < 0.001, parental consanguinity increases the risk of the child by orders of magnitude (F × q ≫ q2). This explains why most patients with the extremely rare PLCE1-associated nephrotic syndrome are homozygous for the causal variant [28,29,30]. Thus, the less frequent an autosomal recessive disease is, the higher is the increase in the risk caused by parental consanguinity and the more important it is to explore the potential presence of inbreeding. Therefore, whenever a child is consulted with an unknown (= extremely rare) and potentially autosomal recessive disorder, one of the first questions should aim at parental consanguinity.
Mendelian or non-Mendelian at the variant level
Transmission pattern is generally considered to depend on the causal gene and the phenotype. However, as several examples show above, pathogenic variants of the same gene may lead to different transmission patterns and may even change whether the disease is Mendelian (= completely penetrant) or non-Mendelian (= incompletely penetrant) (Table 2). It is also important to notice that different phenotypes related to the same variant may have different penetrances; i.e., while the nephronophthisis caused by homozygous NPHP1 deletion is considered to be completely penetrant, retinitis pigmentosa or Joubert syndrome affects only a minority of the patients [40, 58, 59]. In summary, the proper description of the transmission pattern requires the knowledge of the gene, the variant, and the phenotype (Table 2).
Distinction of Mendelian and non-Mendelian variants
For genetic counseling and risk calculation of a Mendelian disorder, it is essential to distinguish the incompletely penetrant (non-Mendelian) variants and to determine their penetrance. This is easier to implement in dominant disorders where segregation analysis of affected families can help in the rough estimation of the penetrance, based on the proportion of unaffected family members with the causal variant.
Penetrance estimation is more challenging in autosomal recessive disorders for several reasons. Segregation analysis is less indicative of incomplete penetrance given the low number of at-risk family members. The underlying pathophysiology, a complete or almost complete loss-of-function makes the chance for an incomplete penetrance lower. Accordingly, incomplete penetrance with biallelic loss-of-function variants is exceptional: the early-onset PLCE1-associated nephrotic syndrome was only accepted after the identification of several families with non-penetrant phenotypes [28,29,30]. This suggested a non-Mendelian, oligogenic origin.
In the rare occasions where an autosomal recessive disorder is incompletely penetrant, it is generally associated with hypomorphic variants [60, 67, 68]. The identification of the incompletely penetrant variants can be greatly aided by knowing their allele frequencies in the general population. The term “maximum credible allele frequency” was proposed based on the general population allele frequency of the major pathogenic variant in a specific gene [69]. Pathogenic variants that are more frequent in the general population than the major variant of the patient cohort are expected to be benign or non-Mendelian. Nevertheless, this approach has limitations, both because rare variants may also be incompletely penetrant, and frequent variants can also be completely penetrant when being subject to interallelic interactions as in case of NPHS2 R229Q, the allele frequency of which exceeds by more than an order of magnitude that of the major NPHS2 R138Q variant in the general population.
We recently introduced a population-genetic algorithm to identify incompletely penetrant variants in autosomal recessive disorders and to calculate their penetrance based on the relative allele frequency of specific pathogenic variants to the loss-of-function variants in the patient and the general population [60]. The sensitivity of this approach strongly depends on the patient cohort size, and will be especially informative in large national patient cohorts. We identified novel incompletely penetrant variants in several autosomal recessive disorders, also the PKHD1 S1833L or I2331K variants in ARPKD, supporting the notion that a non-negligible proportion of frequent variants in autosomal recessive disorders are in fact not Mendelian [60]. Knowledge and further investigation of these variants are essential for proper genetic counseling.
Nevertheless, whenever the degree of penetrance is estimated based on general population data, it is important to keep in mind that it can be dramatically different in families with affected children. Accordingly, the penetrance of severe liver fibrosis in patients with alpha1-antitrypsin deficiency secondary to SERPINA1 homozygous p.E366K (often referred to as “Z” allele) was found in 3/122 (2.5%) in a prospective study screening 200,000 newborns [70], but an order of magnitude higher (8/20, 40%) in siblings of a severely affected child [71], reflecting the enrichment of additional risk factors in families with affected children. Thus, different penetrance data apply to families with and without affected children.
The less frequent a variant, the more challenging to estimate its penetrance, but it is expected to be greatly helped by the rapidly growing patient cohorts and exome and genome sequence data in the general population.
The recessive researcher
Mendel had a shy, introverted character. He had a reputation for self-deprecating jokes. He published only two major scientific papers in his life, he had no pupils, and his work did not give rise to a novel school of science. No one during his lifetime cited his research, and his work was only discovered several years after his death. He never qualified as a teacher and was thus categorized as a substitute teacher for all his career [2]. Darwin, the other great biologist of the nineteenth century, was famous already in his own lifetime. While he was in the forefront of nineteenth-century science, Mendel was hiding in the background with no patent, no citation, no reputation, and no “research grant.” And even if scientific grants had existed and he had had the opportunity to apply for one, who would have financed the research of a scientist with two publications and zero citations? Mendel was thus only recompensed by his own intellectual excitement, a currently slowly fading source of pleasure. However, pure intellectual excitement was enough reward for Mendel to complete a highly meticulous work that laid the foundations of genetics.
After his election as abbot of the Augustinian monastery, he had less time to continue his research [2]. The pea weevil regained his territories in the evolutionary struggle, and thus, the Mendelian ratios of the characters must have been lost. The work was however done, even if its importance was discovered more than a decade after Mendel’s death [2].
Mendel suffered from “chronic nephritis,” and according to his recently obtained genome sequence data, he carried some risk variants for “kidney diseases” [72]. These variants must be unique in the field of nephrology, as they are both Mendelian and non-Mendelian.
The recessive (reclusive) Mendel transmitted his dominant findings to the next generations on a few pieces of paper allowing them to understand the basics of genetics. Genetic research has undergone a major burst afterwards and 100 years after Mendel’s landmark publication (1866), the genetic code was already cracked [73].
Key summary points
-
1.
The constant 3:1 ratio among the offspring of the hybrids allowed for Mendel to understand that binomial traits are determined by distinct elements.
-
2.
The dominant or the recessive terms, as introduced by Mendel, in general apply to all disease alleles of a specific gene. Several exceptions have been however identified. Therefore, the transmission pattern should be described in function of the causal variants and the specific organ involvement.
-
3.
Familial clustering can help the differentiation of Mendelian and rare multifactorial disorders in clinical practice.
-
4.
The less frequent an autosomal recessive disease is, the more important is the effect of inbreeding on its occurrence.
Multiple choice questions with answers
-
1.
What are the major consequences of Mendel’s work in the clinical practice? (select all that apply)
-
a)
The monogenic origin of a disorder can be established based on the transmission pattern.
-
b)
The number of the phenotype determining ‘elements’ (alleles) in an individual could be determined.
-
c)
The role of inbreeding in the rate of the recessive phenotype could be understood.
-
d)
Examples of digenic inheritance have been provided.
-
a)
-
2.
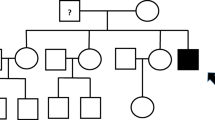
A boy is diagnosed with nephrotic syndrome at the age of 8 months. His twin sister is also found to have nephrotic range proteinuria. The healthy parents are first-degree cousins. What would you do?
-
a)
As the siblings are twins, their affected status should not be considered familial clustering. Steroid therapy should be started, and genetic test to be done only in case of steroid-resistance.
-
b)
Based on the familiar clustering in a consanguineous family, the monogenic origin is evident. Steroid therapy is useless, as well as the genetic test.
-
c)
Start a genetic test and steroid treatment of the index child.
-
d)
A genetic test is necessary, but as the monogenic origin is highly suspicious, no steroid therapy to be initiated.
-
a)
-
3.
An 8-year-old girl is diagnosed with isolated nephronophthisis secondary to homozygous NPHP1 deletion. What is the risk of her 1-month-old asymptomatic brother to develop nephronophthisis and what is his risk of Joubert syndrome?
-
a)
50% and negligible.
-
b)
25% and < 3%
-
c)
25% and < 10%
-
d)
10% and 5%
-
a)
-
4.
A 30-year-old woman was clinically diagnosed with IgA nephropathy based on subnephrotic proteinuria (0.5–1 g/d) and microscopic hematuria. Her two-year-old son was recently diagnosed with similar degree of proteinuria and hematuria and the twin sister with isolated hematuria. No complaint of hearing impairment in any of them. What would you do? (select all that apply)
-
a)
Reassessment of the mother’s diagnosis. Monogenic origin is suggested.
-
b)
Genetic test for Alport syndrome.
-
c)
Kidney biopsy in both children.
-
d)
Start immunosuppression in the son.
-
a)
References
Driscoll CA, Macdonald DW, O’Brien SJ (2009) From wild animals to domestic pets, an evolutionary view of domestication. Proc Natl Acad Sci U S A 106(Suppl 1):9971–9978
Mawer S, Field Museum of Natural History (2006) Gregor Mendel: planting the seeds of genetics. Abrams, in association with the Field Museum, Chicago, New York
Brown R (1833) Observations on the organs and mode of fecundation in Orchideae and Asclepiadeae. Trans Linn Soc 16:685–738
Wood RJ, Vtz O (2001) Genetic prehistory in selective breeding: a prelude to Mendel. Oxford University Press, Oxford England;New York
Zirkle C (1951) Gregor Mendel & his precursors. Isis 42:97–104
Mendel G (1865) Versuche über Plflanzenhybriden. Verhandlungen des naturforschenden Vereines in Brünn 4:3–47
Reinius B, Sandberg R (2015) Random monoallelic expression of autosomal genes: stochastic transcription and allele-level regulation. Nat Rev Genet 16:653–664
Nicholls RD, Knoll JH, Butler MG, Karam S, Lalande M (1989) Genetic imprinting suggested by maternal heterodisomy in nondeletion Prader-Willi syndrome. Nature 342:281–285
Henry I, Bonaiti-Pellie C, Chehensse V, Beldjord C, Schwartz C, Utermann G, Junien C (1991) Uniparental paternal disomy in a genetic cancer-predisposing syndrome. Nature 351:665–667
Goldman M, Smith A, Shuman C, Caluseriu O, Wei C, Steele L, Ray P, Sadowski P, Squire J, Weksberg R, Rosenblum ND (2002) Renal abnormalities in beckwith-wiedemann syndrome are associated with 11p15.5 uniparental disomy. J Am Soc Nephrol 13:2077–2084
Bastepe M, Frohlich LF, Linglart A, Abu-Zahra HS, Tojo K, Ward LM, Juppner H (2005) Deletion of the NESP55 differentially methylated region causes loss of maternal GNAS imprints and pseudohypoparathyroidism type Ib. Nat Genet 37:25–27
Liu J, Litman D, Rosenberg MJ, Yu S, Biesecker LG, Weinstein LS (2000) A GNAS1 imprinting defect in pseudohypoparathyroidism type IB. J Clin Invest 106:1167–1174
Turan S, Bastepe M (2013) The GNAS complex locus and human diseases associated with loss-of-function mutations or epimutations within this imprinted gene. Horm Res Paediatr 80:229–241
Vujic M, Heyer CM, Ars E, Hopp K, Markoff A, Orndal C, Rudenhed B, Nasr SH, Torres VE, Torra R, Bogdanova N, Harris PC (2010) Incompletely penetrant PKD1 alleles mimic the renal manifestations of ARPKD. J Am Soc Nephrol 21:1097–1102
Rossetti S, Kubly VJ, Consugar MB, Hopp K, Roy S, Horsley SW, Chauveau D, Rees L, Barratt TM, van’t Hoff WG, Niaudet P, Torres VE, Harris PC (2009) Incompletely penetrant PKD1 alleles suggest a role for gene dosage in cyst initiation in polycystic kidney disease. Kidney Int 75:848–855
Perrault I, Saunier S, Hanein S, Filhol E, Bizet AA, Collins F, Salih MA, Gerber S, Delphin N, Bigot K, Orssaud C, Silva E, Baudouin V, Oud MM, Shannon N, Le Merrer M, Roche O, Pietrement C, Goumid J, Baumann C, Bole-Feysot C, Nitschke P, Zahrate M, Beales P, Arts HH, Munnich A, Kaplan J, Antignac C, Cormier-Daire V, Rozet JM (2012) Mainzer-Saldino syndrome is a ciliopathy caused by IFT140 mutations. Am J Hum Genet 90:864–870
Otto EA, Trapp ML, Schultheiss UT, Helou J, Quarmby LM, Hildebrandt F (2008) NEK8 mutations affect ciliary and centrosomal localization and may cause nephronophthisis. J Am Soc Nephrol 19:587–592
Frank V, Habbig S, Bartram MP, Eisenberger T, Veenstra-Knol HE, Decker C, Boorsma RA, Gobel H, Nurnberg G, Griessmann A, Franke M, Borgal L, Kohli P, Volker LA, Dotsch J, Nurnberg P, Benzing T, Bolz HJ, Johnson C, Gerkes EH, Schermer B, Bergmann C (2013) Mutations in NEK8 link multiple organ dysplasia with altered Hippo signalling and increased c-MYC expression. Hum Mol Genet 22:2177–2185
Grampa V, Delous M, Zaidan M, Odye G, Thomas S, Elkhartoufi N, Filhol E, Niel O, Silbermann F, Lebreton C, Collardeau-Frachon S, Rouvet I, Alessandri JL, Devisme L, Dieux-Coeslier A, Cordier MP, Capri Y, Khung-Savatovsky S, Sigaudy S, Salomon R, Antignac C, Gubler MC, Benmerah A, Terzi F, Attie-Bitach T, Jeanpierre C, Saunier S (2016) Novel NEK8 mutations cause severe syndromic renal cystic dysplasia through YAP Dysregulation. PLoS Genet 12:e1005894
Boute N, Gribouval O, Roselli S, Benessy F, Lee H, Fuchshuber A, Dahan K, Gubler MC, Niaudet P, Antignac C (2000) NPHS2, encoding the glomerular protein podocin, is mutated in autosomal recessive steroid-resistant nephrotic syndrome. Nat Genet 24:349–354
Claus LR, Chen C, Stallworth J, Turner JL, Slaats GG, Hawks AL, Mabillard H, Senum SR, Srikanth S, Flanagan-Steet H, Louie RJ, Silver J, Lerner-Ellis J, Morel C, Mighton C, Sleutels F, van Slegtenhorst M, van Ham T, Brooks AS, Dorresteijn EM, Barakat TS, Dahan K, Demoulin N, Goffin EJ, Olinger E, Genomics England Research C, Larsen M, Hertz JM, Lilien MR, Obeidova L, Seeman T, Stone HK, Kerecuk L, Gurgu M, YousefYengej FA, Ammerlaan CME, Rookmaaker MB, Hanna C, Rogers RC, Duran K, Peters E, Sayer JA, van Haaften G, Harris PC, Ling K, Mason JM, van Eerde AM, Steet R (2023) Certain heterozygous variants in the kinase domain of the serine/threonine kinase NEK8 can cause an autosomal dominant form of polycystic kidney disease. Kidney Int 104:995–1007
Mehawej C, Chouery E, Ghabril R, Tokajian S, Megarbane A (2023) NEK8-associated nephropathies: do autosomal dominant forms exist? Nephron 147:229–233
Senum SR, Li YSM, Benson KA, Joli G, Olinger E, Lavu S, Madsen CD, Gregory AV, Neatu R, Kline TL, Audrezet MP, Outeda P, Nau CB, Meijer E, Ali H, Steinman TI, Mrug M, Phelan PJ, Watnick TJ, Peters DJM, Ong ACM, Conlon PJ, Perrone RD, Cornec-Le Gall E, Hogan MC, Torres VE, Sayer JA, Genomics England Research Consortium, the HALT PKD, CRISP, DIPAK, ADPKD Modifier, and TAME PKD studies, Harris PC (2022) Monoallelic IFT140 pathogenic variants are an important cause of the autosomal dominant polycystic kidney-spectrum phenotype. Am J Hum Genet 109:136–156
Suvanto M, Patrakka J, Jahnukainen T, Sjostrom PM, Nuutinen M, Arikoski P, Kataja J, Kestila M, Jalanko H (2016) Novel NPHS2 variant in patients with familial steroid-resistant nephrotic syndrome with early onset, slow progression and dominant inheritance pattern. Clin Exp Nephrol 21:677–684
Kerti A, Csohany R, Wagner L, Javorszky E, Maka E, Tory K (2013) NPHS2 homozygous p. R229Q variant: potential modifier instead of causal effect in focal segmental glomerulosclerosis. Pediatr Nephrol 28:2061–2064
Tory K, Menyhard DK, Woerner S, Nevo F, Gribouval O, Kerti A, Straner P, Arrondel C, Huynh Cong E, Tulassay T, Mollet G, Perczel A, Antignac C (2014) Mutation-dependent recessive inheritance of NPHS2-associated steroid-resistant nephrotic syndrome. Nat Genet 46:299–304
Miko A, Menyhárd DK, Kaposi A, Antignac C, Tory K (2018) The mutation-dependent pathogenicity of NPHS2 p. R229Q: a guide for clinical assessment. Hum Mutat 39:1854–1860
Boyer O, Benoit G, Gribouval O, Nevo F, Pawtowski A, Bilge I, Bircan Z, Deschenes G, Guay-Woodford LM, Hall M, Macher MA, Soulami K, Stefanidis CJ, Weiss R, Loirat C, Gubler MC, Antignac C (2010) Mutational analysis of the PLCE1 gene in steroid resistant nephrotic syndrome. J Med Genet 47:445–452
Hinkes B, Wiggins RC, Gbadegesin R, Vlangos CN, Seelow D, Nurnberg G, Garg P, Verma R, Chaib H, Hoskins BE, Ashraf S, Becker C, Hennies HC, Goyal M, Wharram BL, Schachter AD, Mudumana S, Drummond I, Kerjaschki D, Waldherr R, Dietrich A, Ozaltin F, Bakkaloglu A, Cleper R, Basel-Vanagaite L, Pohl M, Griebel M, Tsygin AN, Soylu A, Muller D, Sorli CS, Bunney TD, Katan M, Liu J, Attanasio M, O’Toole JF, Hasselbacher K, Mucha B, Otto EA, Airik R, Kispert A, Kelley GG, Smrcka AV, Gudermann T, Holzman LB, Nurnberg P, Hildebrandt F (2006) Positional cloning uncovers mutations in PLCE1 responsible for a nephrotic syndrome variant that may be reversible. Nat Genet 38:1397–1405
Gilbert RD, Turner CL, Gibson J, Bass PS, Haq MR, Cross E, Bunyan DJ, Collins AR, Tapper WJ, Needell JC, Dell B, Morton NE, Temple IK, Robinson DO (2009) Mutations in phospholipase C epsilon 1 are not sufficient to cause diffuse mesangial sclerosis. Kidney Int 75:415–419
Schlingmann KP, Konrad M, Jeck N, Waldegger P, Reinalter SC, Holder M, Seyberth HW, Waldegger S (2004) Salt wasting and deafness resulting from mutations in two chloride channels. N Engl J Med 350:1314–1319
Katsanis N, Ansley SJ, Badano JL, Eichers ER, Lewis RA, Hoskins BE, Scambler PJ, Davidson WS, Beales PL, Lupski JR (2001) Triallelic inheritance in Bardet-Biedl syndrome, a Mendelian recessive disorder. Science 293:2256–2259
Abu-Safieh L, Al-Anazi S, Al-Abdi L, Hashem M, Alkuraya H, Alamr M, Sirelkhatim MO, Al-Hassnan Z, Alkuraya B, Mohamed JY, Al-Salem A, Alrashed M, Faqeih E, Softah A, Al-Hashem A, Wali S, Rahbeeni Z, Alsayed M, Khan AO, Al-Gazali L, Taschner PE, Al-Hazzaa S, Alkuraya FS (2012) In search of triallelism in Bardet-Biedl syndrome. Eur J Hum Genet 20:420–427
Mykytyn K, Nishimura DY, Searby CC, Shastri M, Yen HJ, Beck JS, Braun T, Streb LM, Cornier AS, Cox GF, Fulton AB, Carmi R, Luleci G, Chandrasekharappa SC, Collins FS, Jacobson SG, Heckenlively JR, Weleber RG, Stone EM, Sheffield VC (2002) Identification of the gene (BBS1) most commonly involved in Bardet-Biedl syndrome, a complex human obesity syndrome. Nat Genet 31:435–438
Slavotinek AM, Searby C, Al-Gazali L, Hennekam RC, Schrander-Stumpel C, Orcana-Losa M, Pardo-Reoyo S, Cantani A, Kumar D, Capellini Q, Neri G, Zackai E, Biesecker LG (2002) Mutation analysis of the MKKS gene in McKusick-Kaufman syndrome and selected Bardet-Biedl syndrome patients. Hum Genet 110:561–567
Hichri H, Stoetzel C, Laurier V, Caron S, Sigaudy S, Sarda P, Hamel C, Martin-Coignard D, Gilles M, Leheup B, Holder M, Kaplan J, Bitoun P, Lacombe D, Verloes A, Bonneau D, Perrin-Schmitt F, Brandt C, Besancon AF, Mandel JL, Cossee M, Dollfus H (2005) Testing for triallelism: analysis of six BBS genes in a Bardet-Biedl syndrome family cohort. Eur J Hum Genet 13:607–616
Forsyth R, Gunay-Aygun M (1993) Bardet-Biedl syndrome overview. In: Adam MP, Mirzaa GM, Pagon RA, Wallace SE, Bean LJH, Gripp KW, Amemiya A (eds) GeneReviews((R)), Seattle (WA)
Fallerini C, Baldassarri M, Trevisson E, Morbidoni V, La Manna A, Lazzarin R, Pasini A, Barbano G, Pinciaroli AR, Garosi G, Frullanti E, Pinto AM, Mencarelli MA, Mari F, Renieri A, Ariani F (2017) Alport syndrome: impact of digenic inheritance in patients management. Clin Genet 92:34–44
Mencarelli MA, Heidet L, Storey H, van Geel M, Knebelmann B, Fallerini C, Miglietti N, Antonucci MF, Cetta F, Sayer JA, van den Wijngaard A, Yau S, Mari F, Bruttini M, Ariani F, Dahan K, Smeets B, Antignac C, Flinter F, Renieri A (2015) Evidence of digenic inheritance in Alport syndrome. J Med Genet 52:163–174
Tory K, Lacoste T, Burglen L, Moriniere V, Boddaert N, Macher MA, Llanas B, Nivet H, Bensman A, Niaudet P, Antignac C, Salomon R, Saunier S (2007) High NPHP1 and NPHP6 mutation rate in patients with Joubert syndrome and nephronophthisis: potential epistatic effect of NPHP6 and AHI1 mutations in patients with NPHP1 mutations. J Am Soc Nephrol 18:1566–1575
Louie CM, Caridi G, Lopes VS, Brancati F, Kispert A, Lancaster MA, Schlossman AM, Otto EA, Leitges M, Grone HJ, Lopez I, Gudiseva HV, O’Toole JF, Vallespin E, Ayyagari R, Ayuso C, Cremers FP, den Hollander AI, Koenekoop RK, Dallapiccola B, Ghiggeri GM, Hildebrandt F, Valente EM, Williams DS, Gleeson JG (2010) AHI1 is required for photoreceptor outer segment development and is a modifier for retinal degeneration in nephronophthisis. Nat Genet 42:175–180
Hoefele J, Wolf MT, O’Toole JF, Otto EA, Schultheiss U, Deschenes G, Attanasio M, Utsch B, Antignac C, Hildebrandt F (2007) Evidence of oligogenic inheritance in nephronophthisis. J Am Soc Nephrol 18:2789–2795
Abecasis GR, Altshuler D, Auton A, Brooks LD, Durbin RM, Gibbs RA, Hurles ME, McVean GA (2010) A map of human genome variation from population-scale sequencing. Nature 467:1061–1073
Heon E, Kim G, Qin S, Garrison JE, Tavares E, Vincent A, Nuangchamnong N, Scott CA, Slusarski DC, Sheffield VC (2016) Mutations in C8ORF37 cause Bardet Biedl syndrome (BBS21). Hum Mol Genet 25:2283–2294
Macia MS, Halbritter J, Delous M, Bredrup C, Gutter A, Filhol E, Mellgren AEC, Leh S, Bizet A, Braun DA, Gee HY, Silbermann F, Henry C, Krug P, Bole-Feysot C, Nitschke P, Joly D, Nicoud P, Paget A, Haugland H, Brackmann D, Ahmet N, Sandford R, Cengiz N, Knappskog PM, Boman H, Linghu B, Yang F, Oakeley EJ, Saint Mezard P, Sailer AW, Johansson S, Rodahl E, Saunier S, Hildebrandt F, Benmerah A (2017) Mutations in MAPKBP1 cause juvenile or late-onset cilia-independent nephronophthisis. Am J Hum Genet 100:323–333
Shaheen R, Szymanska K, Basu B, Patel N, Ewida N, Faqeih E, Al Hashem A, Derar N, Alsharif H, Aldahmesh MA, Alazami AM, Hashem M, Ibrahim N, Abdulwahab FM, Sonbul R, Alkuraya H, Alnemer M, Al Tala S, Al-Husain M, Morsy H, Seidahmed MZ, Meriki N, Al-Owain M, AlShahwan S, Tabarki B, Salih MA, Ciliopathy W, Faquih T, El-Kalioby M, Ueffing M, Boldt K, Logan CV, Parry DA, Al Tassan N, Monies D, Megarbane A, Abouelhoda M, Halees A, Johnson CA, Alkuraya FS (2016) Characterizing the morbid genome of ciliopathies. Genome Biol 17:242
Phelps IG, Dempsey JC, Grout ME, Isabella CR, Tully HM, Doherty D, Bachmann-Gagescu R (2018) Interpreting the clinical significance of combined variants in multiple recessive disease genes: systematic investigation of Joubert syndrome yields little support for oligogenicity. Genet Med 20:223–233
Genovese G, Friedman DJ, Ross MD, Lecordier L, Uzureau P, Freedman BI, Bowden DW, Langefeld CD, Oleksyk TK, Uscinski Knob AL, Bernhardy AJ, Hicks PJ, Nelson GW, Vanhollebeke B, Winkler CA, Kopp JB, Pays E, Pollak MR (2010) Association of trypanolytic ApoL1 variants with kidney disease in African Americans. Science 329:841–845
Kopp JB, Nelson GW, Sampath K, Johnson RC, Genovese G, An P, Friedman D, Briggs W, Dart R, Korbet S, Mokrzycki MH, Kimmel PL, Limou S, Ahuja TS, Berns JS, Fryc J, Simon EE, Smith MC, Trachtman H, Michel DM, Schelling JR, Vlahov D, Pollak M, Winkler CA (2011) APOL1 genetic variants in focal segmental glomerulosclerosis and HIV-associated nephropathy. J Am Soc Nephrol 22:2129–2137
Benoit G, Machuca E, Antignac C (2010) Hereditary nephrotic syndrome: a systematic approach for genetic testing and a review of associated podocyte gene mutations. Pediatr Nephrol 25:1621–1632
Wiesel A, Queisser-Luft A, Clementi M, Bianca S, Stoll C, Group ES (2005) Prenatal detection of congenital renal malformations by fetal ultrasonographic examination: an analysis of 709,030 births in 12 European countries. Eur J Med Genet 48:131–144
Tain YL, Luh H, Lin CY, Hsu CN (2016) Incidence and risks of congenital anomalies of kidney and urinary tract in newborns: a population-based case-control study in Taiwan. Medicine (Baltimore) 95:e2659
Andres-Jensen L, Jorgensen FS, Thorup J, Flachs J, Madsen JL, Maroun LL, Norgaard P, Vinicoff PG, Olsen BH, Cortes D (2016) The outcome of antenatal ultrasound diagnosed anomalies of the kidney and urinary tract in a large Danish birth cohort. Arch Dis Child 101:819–824
Nicolaou N, Renkema KY, Bongers EM, Giles RH, Knoers NV (2015) Genetic, environmental, and epigenetic factors involved in CAKUT. Nat Rev Nephrol 11:720–731
Bulum B, Ozcakar ZB, Ustuner E, Dusunceli E, Kavaz A, Duman D, Walz K, Fitoz S, Tekin M, Yalcinkaya F (2013) High frequency of kidney and urinary tract anomalies in asymptomatic first-degree relatives of patients with CAKUT. Pediatr Nephrol 28:2143–2147
Manoharan A, Krishnamurthy S, Sivamurukan P, Ananthakrishnan R, Jindal B (2020) Screening for renal and urinary tract anomalies in asymptomatic first degree relatives of children with congenital anomalies of the kidney and urinary tract (CAKUT). Indian J Pediatr 87:686–691
Liebau MC (2021) Early clinical management of autosomal recessive polycystic kidney disease. Pediatr Nephrol 36:3561–3570
Salomon R, Saunier S, Niaudet P (2009) Nephronophthisis. Pediatr Nephrol 24:2333–2344
Petzold F, Billot K, Chen X, Henry C, Filhol E, Martin Y, Avramescu M, Douillet M, Moriniere V, Krug P, Jeanpierre C, Tory K, Boyer O, Burgun A, Servais A, Salomon R, Benmerah A, Heidet L, Garcelon N, Antignac C, Zaidan M, Saunier S, INSERM–Necker Hospital NPH collaborative group, (2023) The genetic landscape and clinical spectrum of nephronophthisis and related ciliopathies. Kidney Int 104:378–387
Miko A, Kaposi A, Schnabel K, Seidl D, Tory K (2021) Identification of incompletely penetrant variants and interallelic interactions in autosomal recessive disorders by a population-genetic approach. Hum Mutat 42:1473–1487
Zurowska AM, Bielska O, Daca-Roszak P, Jankowski M, Szczepanska M, Roszkowska-Bjanid D, Kuzma-Mroczkowska E, Panczyk-Tomaszewska M, Moczulska A, Drozdz D, Hadjipanagi D, Deltas C, Ostalska-Nowicka D, Rabiega A, Taraszkiewicz J, Taranta-Janusz K, Wieczorkiewicz-Plaza A, Jobs K, Mews J, Musial K, Jakubowska A, Nosek H, Jander AE, Koutsofti C, Stanislawska-Sachadyn A, Kuleszo D, Zietkiewicz E, Lipska-Zietkiewicz BS (2021) Mild X-linked Alport syndrome due to the COL4A5 G624D variant originating in the Middle Ages is predominant in Central/East Europe and causes kidney failure in midlife. Kidney Int 99:1451–1458
Balogh E, Chandler JC, Varga M, Tahoun M, Menyhard DK, Schay G, Goncalves T, Hamar R, Legradi R, Szekeres A, Gribouval O, Kleta R, Stanescu H, Bockenhauer D, Kerti A, Williams H, Kinsler V, Di WL, Curtis D, Kolatsi-Joannou M, Hammid H, Szocs A, Perczel K, Maka E, Toldi G, Sava F, Arrondel C, Kardos M, Fintha A, Hossain A, D’Arco F, Kaliakatsos M, Koeglmeier J, Mifsud W, Moosajee M, Faro A, Javorszky E, Rudas G, Saied MH, Marzouk S, Kelen K, Gotze J, Reusz G, Tulassay T, Dragon F, Mollet G, Motameny S, Thiele H, Dorval G, Nurnberg P, Perczel A, Szabo AJ, Long DA, Tomita K, Antignac C, Waters AM, Tory K (2020) Pseudouridylation defect due to DKC1 and NOP10 mutations causes nephrotic syndrome with cataracts, hearing impairment, and enterocolitis. Proc Natl Acad Sci U S A 117:15137–15147
Saunier S, Calado J, Heilig R, Silbermann F, Benessy F, Morin G, Konrad M, Broyer M, Gubler MC, Weissenbach J, Antignac C (1997) A novel gene that encodes a protein with a putative src homology 3 domain is a candidate gene for familial juvenile nephronophthisis. Hum Mol Genet 6:2317–2323
Simon DB, Bindra RS, Mansfield TA, Nelson-Williams C, Mendonca E, Stone R, Schurman S, Nayir A, Alpay H, Bakkaloglu A, Rodriguez-Soriano J, Morales JM, Sanjad SA, Taylor CM, Pilz D, Brem A, Trachtman H, Griswold W, Richard GA, John E, Lifton RP (1997) Mutations in the chloride channel gene, CLCNKB, cause Bartter’s syndrome type III. Nat Genet 17:171–178
Moriniere V, Dahan K, Hilbert P, Lison M, Lebbah S, Topa A, Bole-Feysot C, Pruvost S, Nitschke P, Plaisier E, Knebelmann B, Macher MA, Noel LH, Gubler MC, Antignac C, Heidet L (2014) Improving mutation screening in familial hematuric nephropathies through next generation sequencing. J Am Soc Nephrol 25:2740–2751
Jais JP, Knebelmann B, Giatras I, De Marchi M, Rizzoni G, Renieri A, Weber M, Gross O, Netzer KO, Flinter F, Pirson Y, Dahan K, Wieslander J, Persson U, Tryggvason K, Martin P, Hertz JM, Schroder C, Sanak M, Carvalho MF, Saus J, Antignac C, Smeets H, Gubler MC (2003) X-linked Alport syndrome: natural history and genotype-phenotype correlations in girls and women belonging to 195 families: a “European Community Alport Syndrome Concerted Action” study. J Am Soc Nephrol 14:2603–2610
Thauvin-Robinet C, Munck A, Huet F, Genin E, Bellis G, Gautier E, Audrezet MP, Ferec C, Lalau G, Georges MD, Claustres M, Bienvenu T, Gerard B, Boisseau P, Cabet-Bey F, Feldmann D, Clavel C, Bieth E, Iron A, Simon-Bouy B, Costa C, Medina R, Leclerc J, Hubert D, Nove-Josserand R, Sermet-Gaudelus I, Rault G, Flori J, Leroy S, Wizla N, Bellon G, Haloun A, Perez-Martin S, d’Acremont G, Corvol H, Clement A, Houssin E, Binquet C, Bonithon-Kopp C, Alberti-Boulme C, Morris MA, Faivre L, Goossens M, Roussey M, Collaborating Working Group on R117H, Girodon E (2009) The very low penetrance of cystic fibrosis for the R117H mutation: a reappraisal for genetic counselling and newborn screening. J Med Genet 46:752–758
Chu CS, Trapnell BC, Curristin S, Cutting GR, Crystal RG (1993) Genetic basis of variable exon 9 skipping in cystic fibrosis transmembrane conductance regulator mRNA. Nat Genet 3:151–156
Whiffin N, Minikel E, Walsh R, O’Donnell-Luria AH, Karczewski K, Ing AY, Barton PJR, Funke B, Cook SA, MacArthur D, Ware JS (2017) Using high-resolution variant frequencies to empower clinical genome interpretation. Genet Med 19:1151–1158
Sveger T (1988) The natural history of liver disease in alpha 1-antitrypsin deficient children. Acta Paediatr Scand 77:847–851
Cox DW, Mansfield T (1987) Prenatal diagnosis of alpha 1 antitrypsin deficiency and estimates of fetal risk for disease. J Med Genet 24:52–59
Greenfieldboyce N (2022) Why scientists dug up the father of genetics, Gregor Mendel, and analyzed his DNA. https://www.npr.org/sections/health-shots/2022/12/30/1142202365/gregor-mendel-genetics-dna-analyzed
Nirenberg M, Caskey T, Marshall R, Brimacombe R, Kellogg D, Doctor B, Hatfield D, Levin J, Rottman F, Pestka S, Wilcox M, Anderson F (1966) The RNA code and protein synthesis. Cold Spring Harb Symp Quant Biol 31:11–24
Acknowledgements
I thank Corinne Antignac, my affectionately respected genetics mentor, and Julianna Beáta Vig and Máté Varga, two great colleagues from the F1 generation, for their Mendelian thorough reading and correction of the manuscript. Financial support of the Nephrogenetic Research Group is provided by the National Research, Development and Innovation Office (NKFIA/OTKA K135798) and by the Ministry of Innovation and Technology of Hungary from the National Research, Development and Innovation Fund, financed under the TKP2021-EGA and TKP2021-NVA funding schemes (TKP2021-EGA-24, TKP2021-NVA-15). The author of this publication is a member of the European Reference Network for Rare Kidney Diseases (ERKNet).
Funding
Open access funding provided by Semmelweis University.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The author declares no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Answers: 1. a, b, c; 2. d; 3. b; 4. a, b.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Tory, K. The dominant findings of a recessive man: from Mendel’s kid pea to kidney. Pediatr Nephrol 39, 2049–2059 (2024). https://doi.org/10.1007/s00467-023-06238-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00467-023-06238-9