Abstract
Purpose
To examine the association between dispositional optimism and all-cause mortality after esophageal cancer surgery and whether pathological tumor stage and the COVID-19 pandemic modified this association.
Methods
This nationwide, population-based prospective cohort study included 335 patients undergoing esophageal cancer surgery in Sweden between January 1, 2013, and December 31, 2019. Dispositional optimism was measured 1 year post-surgery using Life Orientation Test-Revised (LOT-R). A higher LOT-R sum score represents higher dispositional optimism. Mortality information was obtained from the Swedish Register of the Total Population. All patients were followed up until death or until December 31, 2020, whichever occurred first. Cox regression with adjustments for confounders was used.
Results
The median follow-up was 20.8 months, during which 125 (37.3%) patients died. Among the included 335 patients, 219 (65.4%) patients had tumor pathologically staged Tis-II, and 300 (89.6%) patients entered the cohort before the COVID-19 pandemic. Both tumor stage and the COVID-19 pandemic were effect modifiers. For each unit increase in LOT-R sum score, the risk of all-cause mortality decreased by 11% (HR 0.89, 95% CI 0.81 to 0.98) among patients with tumor staged Tis-II before the COVID-19 pandemic. This association was non-significant in patients with tumor staged III–IV (HR 0.99, 95% CI 0.92 to 1.07) and during the COVID-19 pandemic (HR 1.08, 95% CI 0.94 to 1.25).
Conclusion
Assessing dispositional optimism may help predict postoperative survival, especially for patients with early and intermediate esophageal cancer. Increasing dispositional optimism might be a potential intervention target to improve survival after esophageal cancer surgery.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Background
Esophageal cancer is the seventh most commonly diagnosed cancer worldwide [1], which carries a poor prognosis with an overall 5-year survival below 20% [2, 3]. Surgical resection (esophagectomy) is the mainstay of curative treatment. However, the 5-year survival after esophagectomy is still less than 50% as reported by a Swedish nationwide cohort study [2]. Identified prognostic factors for survival after esophageal cancer surgery include sociodemographic, clinical, and surgeon-related factors such as age, tumor stage, and surgeon volume [4,5,6,7,8,9]. However, these predictors cannot fully explain the variation of postoperative survival, and most of them are not modifiable after surgery. Thus, identifying other potentially modifiable predictors is important.
Dispositional optimism is a personality trait defined as generalized positive expectations for the future [10]. It is relatively stable but can be increased via psychological interventions [11]. Higher dispositional optimism has been found to be associated with better physical health [12] and with lower all-cause mortality and cardiovascular death in the general and elderly populations [13,14,15,16,17,18,19,20,21]. However, studies conducted in patients with cancer have reported inconsistent results. Higher dispositional optimism was associated with lower mortality in patients with recurrent or metastatic cancer treated with palliative radiation, head and neck cancer, and ovarian cancer [22,23,24]. Contrarily, there was no association in patients with metastatic colorectal cancer, wherein the reported hazard ratio (HR) equaled 0.98 based on 429 subjects [25]. To date, it remains unclear whether such an association exists in patients with esophageal cancer.
The present study aimed to use a Swedish nationwide, population-based, prospective cohort to examine whether dispositional optimism predicted all-cause mortality after esophageal cancer surgery. In addition, given that esophageal cancer is an aggressive tumor and the COVID-19 pandemic severely affects the society and healthcare system, the effect of dispositional optimism on survival might be overridden by the effect of the strongest prognostic factor, tumor stage, and of the hazard event, the COVID-19 pandemic. Therefore, we also aimed to examine the potential effect modifications by pathological tumor stage and the COVID-19 pandemic.
Material and methods
Study design
Data for the present study were drawn from a prospective, ongoing, Swedish nationwide, and population-based cohort study entitled Oesophageal Surgery on Cancer patients-Adaptation and Recovery (OSCAR). Details of the OSCAR study have been described elsewhere [26, 27]. In brief, all 1-year survivors without cognitive impairment undergoing esophagectomy for cancer in Sweden from January 1, 2013, and onwards are invited (approximate response rate 66%) [26]. Follow-up data included patient-reported outcomes as well as regular assessments of vital status [26]. The OSCAR study was approved by the Regional Ethical Review Board in Stockholm, Sweden (diary number 2013/844–31/1), and informed consents were obtained from all participants before inclusion.
The present study included OSCAR study participants undergoing esophageal cancer surgery in Sweden between January 1, 2013, and December 31, 2019. Patients with a psychiatric history were excluded.
Exposure: dispositional optimism
After consenting to participate in the OSCAR study, patients self-reported their dispositional optimism on the Swedish version of Life Orientation Test-Revised (LOT-R) [28, 29] during the first interview (i.e., 1 year after esophageal cancer surgery). LOT-R consists of three positively worded items and three negatively worded items [28, 29], and the extent of agreement on these six items are indicated on a five-point Likert scale ranging from 0 (“strongly disagree”) to 4 (“strongly agree”) [28, 29].
Given the inconclusive dimensionality of LOT-R [30] and the absence of psychometric research among patients with esophagectomy for cancer, we have previously conducted a series of confirmatory factor analyses and presented the detailed results elsewhere [31]. Because the first negatively worded item had bimodal response distribution, equivocal correlations with both positively and negatively worded items, and negative loading in the best-fitting model [31], which suggest that a considerable proportion of patients most likely misread it, we removed this item [31]. The final adopted model assumes one dimension (dispositional optimism) with correlated errors between the two reversed negatively worded items [31].
A sum score of the remaining five items was computed with a higher score representing higher dispositional optimism. The internal reliability estimated by McDonald’s omega was 0.49, with 95% bootstrapped confidence interval (CI) 0.31 to 0.62 [31].
Outcome: all-cause mortality
All patients were followed from 1 year post-surgery (identified as the date of the first interview after consenting to participate in the OSCAR study) and until the date of death or December 31, 2020, whichever occurred first. Mortality information was obtained from the Swedish Register of the Total Population, which has 100% complete ascertainment of death [32].
Covariates
Four sociodemographic variables including age at surgery (continuous), sex (female or male), education level (9-year compulsory school, upper secondary school, or higher education), and cohabitation status (non-cohabitating or cohabitating) were considered as confirmed confounders because they are associated with both dispositional optimism and mortality [4, 33]. The following seven clinical variables were considered as potential but unconfirmed confounders as they are predictors of survival [4,5,6,7] but no available evidence has suggested that they can affect dispositional optimism [34, 35]: (1) pathological tumor stage (Tis-II or III–IV), (2) Charlson Comorbidity Index (0, 1, or ≥ 2), (3) neoadjuvant therapy (yes or no), (4) histology (squamous cell carcinoma or adenocarcinoma), (5) postoperative Clavien–Dindo complication score (none, I–II, or III–IV), (6) resection margin status (R0 or non-R0), and (7) surgical approach (minimally invasive, hybrid, or open surgery).
The sociodemographic data were collected from the patient-reported questionnaires and the Swedish Longitudinal Integration Database for Health Insurance and Labor Market Studies. Clinical data were obtained from medical records, the Swedish Patient Register, and the Swedish Cancer Register.
Statistical analysis
The overall mean of the LOT-R sum scores between patients with different characteristics were compared using t-test or analysis of variance. Multivariable Cox proportional hazards model was used to calculate the HR with 95% CI for the all-cause mortality with one unit increase in the LOT-R sum score. Time since surgery was used as the time scale. The four sociodemographic variables were adjusted for in all Cox models because they were confounders based on prior knowledge [4, 33]. Given that we could not decide whether clinical variables were confounders because of the absence of prior knowledge, we used data-driven forward selection, and only covariates leading to ≥ 10% change in the estimated HR could be included in the final Cox models. The estimated HR was almost unchanged after further adjusting for clinical variables (Supplementary Table S1), and thus none of them was considered as confounders in the current study. Potential effect modification was examined by adding related interaction terms into Cox models.
We treated the COVID-19 pandemic as a time-varying variable, and the period from March 1, 2020, was regarded as within the COVID-19 pandemic in Sweden. Given that most person-time in this study was before the COVID-19 pandemic, we might only have statistical power for the analysis involving period before the COVID-19 pandemic but not period within the COVID-19 pandemic. Therefore, we conducted a subgroup analysis limiting the follow-up ending date before the occurrence of the COVID-19 pandemic in Sweden (i.e., March 1, 2020) to further examine the association during the period without the COVID-19 pandemic.
In addition, to account for the potential reverse causality that approaching death might affect dispositional optimism, we excluded patients who survived less than 2 months after the dispositional optimism measurement and re-estimated all Cox models.
The assumption of proportional hazards was tested in all Cox models using Schoenfeld residuals and it was met for all analyses. All hypothesis tests were two-sided with significance level 0.05. Stata 13 (StataCorp, College Station, Texas, USA) and SAS 9.4 (SAS Institute, Cary, NC, USA) were used for the statistical analyses.
Results
Study participants
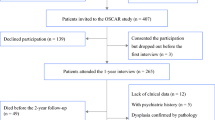
In total, 921 patients underwent esophageal cancer surgery between January 1, 2013, and December 31, 2019, in Sweden. After excluding 221 patients who died within 1 year after surgery and 131 patients who were uncontactable, 569 patients were invited to participate in the OSCAR study. Of these, 376 patients consented to the participation and accomplished the first interview. Non-participation was mainly associated with poor health, cancer recurrence, and unwillingness [26]. After further excluding 41 patients with a psychiatric history or missing data, the final study cohort included 335 patients. The detailed process of patient selection is displayed in Fig. 1.
Characteristics of participants
Table 1 presents the characteristics of the 335 patients included in the present study. The average age at surgery was 67.4 years with a standard deviation (SD) of 8.2 years. Most patients were male (90.8%), married/cohabitating (75.4%), had pathological tumor stage Tis-II (65.4%), and entered the OSCAR study before the COVID-19 pandemic (89.6%). The mean of LOT-R sum scores was 15.2 ± 3.3 (range 5 to 20). The dispositional optimism score was comparable between patients with different characteristics (Supplementary Table S2).
All-cause mortality in the whole cohort with follow-up containing the COVID-19 pandemic
Among the 335 patients included in the whole cohort with follow-up until December 31, 2020, 125 deaths occurred. Patients were followed up for a median of 20.8 months (interquartile range 10.9 to 43.7 months) with a total of 9305.0 person-months.
The model without any interaction terms showed that the overall HR was 0.96 (95% CI 0.91 to 1.02). The model containing an interaction term between dispositional optimism and pathological tumor stage showed that HR for patients with tumor staged Tis-II was 0.93 (95% CI 0.86 to 1.01; Fig. 2) and for patients with tumor staged III–IV was 1.00 (95% CI 0.93 to 1.08; Fig. 2). The p-value for the interaction term was 0.24. The model containing an interaction term between dispositional optimism and the COVID-19 pandemic showed that HR for the period before the COVID-19 pandemic was 0.94 (95% CI 0.89 to 1.00; Fig. 2) and for the period within the COVID-19 pandemic was 1.08 (95% CI 0.94 to 1.25; Fig. 2). The p-value for the interaction term was 0.08.
Given that the above results suggested that both pathological tumor stage and the COVID-19 pandemic were potential effect modifiers, we built a model with interaction terms among dispositional optimism, pathological tumor stage, and the COVID-19 pandemic. It showed that in the period before the COVID-19 pandemic and among patients with tumor staged Tis-II, higher dispositional optimism was significantly associated with better overall survival. For each unit increase in the LOT-R sum score, the risk of all-cause mortality decreased by 11% (HR 0.89, 95% CI 0.81 to 0.98; Fig. 2). However, during the COVID-19 pandemic and for patients with tumor staged III–IV, the associations between dispositional optimism and all-cause mortality were not statistically significant (Fig. 2).
All-cause mortality in the sub-cohort with follow-up before the COVID-19 pandemic
In the subgroup analysis limiting the follow-up before the COVID-19 pandemic occurred in Sweden (i.e., March 1, 2020), 300 patients were included, of which 102 patients died during the follow-up with a median of 18.0 months (interquartile range 9.4 to 36.1 months) and a total of 7221.0 person-months.
The model without any interaction terms showed that the overall HR was 0.94 (95% CI 0.89 to 1.00). The model with an interaction term between dispositional optimism and pathological tumor stage generated similar results as the above model using the whole cohort. Higher dispositional optimism was significantly associated with lower all-cause mortality for patients with tumor staged Tis-II (HR 0.89, 95% CI 0.81 to 0.98), but not for patients with tumor staged III–IV (HR 0.99, 95% CI 0.92 to 1.07). The p-value for the interaction term was 0.08. Figure 3 presents the predicted cumulative survival.
Predicted cumulative survival curves after esophageal cancer surgery for patients with average dispositional optimism level (LOT-R sum score equaling to 15) and patients with higher dispositional optimism (LOT-R sum score equaling to 16), with follow-up period before the COVID-19 pandemic and pathological tumor stage as an effect modifier. Note. LOT-R, Life Orientation Test-Revised; HR, hazard ratio; CI, confidence interval
In the sensitivity analyses excluding patients who survived less than 2 months after entering the OSCAR study, the association between dispositional optimism and all-cause mortality remained almost unchanged (results not shown).
Discussion
This Swedish nationwide population-based cohort study found that in the period before the COVID-19 pandemic, higher dispositional optimism was associated with lower all-cause mortality after esophageal cancer surgery among patients with tumor staged Tis-II. However, among those with advanced tumor stage (III–IV) and during the COVID-19 pandemic, non-significant associations were observed.
The current study adds to the growing evidence of the beneficial effect of dispositional optimism on survival [13,14,15,16,17,18,19,20,21,22,23,24]. Several potential biological, behavioral, and psychosocial pathways may explain the observed protective effect. Higher dispositional optimism has been found to be associated with healthier levels of several aging biomarkers including telomere, fibrinogen, interleukin-6, interleukin-10, homocysteine, and lipid profile [36,37,38,39], and thus, it may be related to lower risks of developing and dying from age-related diseases such as cardiovascular diseases, which is the leading non-cancer cause of death in patients with esophageal cancer in Sweden [40]. Moreover, people with higher dispositional optimism tend to be non-smokers, exercise more, and have healthier dietary habits [41]. Such healthier lifestyles may further protect them from developing comorbidities and slow the progression of already diagnosed diseases. In addition, more optimistic people are less likely to develop psychological comorbidities and feel lonely [31, 42], which may decrease the risks of mortality [8, 43]. Moreover, higher dispositional optimism predicts better health-related quality of life [44], and the latter further predicts better survival after esophageal cancer surgery [45].
The prognosis for advanced esophageal cancer is much less controllable compared with early and intermediate stages of cancer, which may explain the observed effect modification by tumor stage. It is difficult for patients with advanced esophageal cancer to prolong survival due to the aggressive tumor itself (5-year survival below 15%) [3]. However, it is increasingly possible for patients with early and intermediate esophageal cancer to achieve long-term survival (5-year survival 30–56%) [3], so they can have the chance to benefit from dispositional optimism via fewer comorbidities and slower disease progression. Future studies with larger sample sizes examining disease-specific cause of death and examining whether dispositional optimism relates to cancer recurrence might be useful to verify this speculation.
The observed predictive effect of dispositional optimism on survival may be beneficial, especially for patients with early and intermediate stages of esophageal cancer. Assessing dispositional optimism may help identify patients with a higher risk of premature death, thus leading to timely and tailored interventions. Also, given that dispositional optimism can be improved via psychological interventions like Best Possible Self exercise and cognitive behavioral therapy [11], it might be a potential intervention target to improve survival after esophageal cancer surgery.
We did not find a statistically significant association between dispositional optimism and all-cause mortality for the period within the COVID-19 pandemic. The analyzed person-time within the COVID-19 pandemic in this study was limited, which led to unstable point estimates and wide confidence intervals. Thus, this result should be interpreted with great caution and needs to be examined further by future studies using larger sample sizes. However, it makes conceptual sense that the protective effect of dispositional optimism on survival disappeared during the COVID-19 pandemic. It has been reported that individuals aged above 60 have up to 70% excess mortality during the first wave of the COVID-19 pandemic in Sweden, compared with corresponding calendar weeks in previous years [46]. Patients with esophageal cancer are one of the most vulnerable populations because of their diseases and relatively old age (mean age in the current study was 67.4 years). Moreover, lots of routine healthcare and scheduled surgeries have been postponed during the COVID-19 pandemic in Sweden [47]. Such a healthcare shortage implies that the prognosis of esophageal cancer became less controllable for all patients during the COVID-19 pandemic, which might thus nullify the protective effect of dispositional optimism. In addition, one study has found that more optimistic people tend to underestimate the severity of the COVID-19 pandemic and engage less in protective behaviors [48], such a side effect of optimism during the COVID-19 pandemic may cancel out the protective effect of optimism, thus leading to a non-significant observed association.
This study has several strengths. First, to the best of our knowledge, it is the first study examining the association between dispositional optimism and all-cause mortality after esophageal cancer surgery. Second, the comprehensive data collection allowed for adjustments for confirmed confounders as well as a selection of potential but unproven confounders, which reduced the risk of confounding bias. Third, the predefined interaction and subgroup analyses addressed the effect modifications by pathological tumor stage and the COVID-19 pandemic, and thus presented a more comprehensive picture of the association between dispositional optimism and postoperative survival. Fourth, complete follow-up based on register reduced the risk of selection bias. Lastly, the nationwide population-based prospective cohort study design enhanced the generalizability of the results.
This study also has some limitations. Because the OSCAR study focuses on patients who have survived for at least 1 year after esophagectomy, all patient-reported outcomes were started to be collected at 1 year after surgery. The results of this study might not be applicable to the period within 1 year after surgery and should be interpreted in light of the measurement time point of dispositional optimism. However, previous studies have shown that dispositional optimism keep almost unchanged before and after cancer diagnosis and surgery [34, 35]. Additionally, the analysis was conditioned on participation in the OSCAR study, which might induce selection bias, and findings of this study should be generalized cautiously to non-participants. In addition, although the sensitivity analysis excluding patients surviving less than 2 months after LOT-R measurement generated similar results as the main analysis, the possibility of reverse causality cannot be entirely ruled out as the choice of 2 months was arbitrary, albeit predefined. Lastly, although adjusting for sociodemographic variables may have partly controlled for some unmeasured confounders including genetic factors [49] and early life events [50], there is still a risk of residual confounding. Moreover, association is not equal to causation, and whether increasing dispositional optimism could improve survival needs to be further examined by future interventional research.
In conclusion, this study showed that higher dispositional optimism was associated with lower all-cause mortality after esophageal cancer surgery in patients with early and intermediate tumor stages in the period before the COVID-19 pandemic. Future studies with larger sample sizes are needed to validate the predictive effect of dispositional optimism and to identify the underlying mechanisms. Intervention studies are warranted to examine whether dispositional optimism could be a modifiable target to help improve survival after esophageal cancer surgery.
Data availability
The data that support the findings of this study are stored at the server of Karolinska Institutet and are available from the corresponding author on reasonable request. The data are not publicly available due to privacy and ethical restrictions.
References
Sung H, Ferlay J, Siegel RL et al (2021) Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 71(3):209–249
Kauppila JH, Mattsson F, Brusselaers N et al (2018) Prognosis of oesophageal adenocarcinoma and squamous cell carcinoma following surgery and no surgery in a nationwide Swedish cohort study. BMJ Open 8(5):e021495
Office for National Statistics. Cancer survival in England — adults diagnosed 2019 [Jun 27, 2021]. Available from: https://www.ons.gov.uk/peoplepopulationandcommunity/healthandsocialcare/conditionsanddiseases/datasets/cancersurvivalratescancersurvivalinenglandadultsdiagnosed
Xie S-H, Santoni G, Mälberg K et al (2021) Prediction model of long-term survival after esophageal cancer surgery. Ann Surg 273(5):933–939
Backemar L, Lagergren P, Johar A et al (2015) Impact of co-morbidity on mortality after oesophageal cancer surgery. Br J Surg 102(9):1097–1105
Rutegard M, Lagergren P, Rouvelas I et al (2012) Surgical complications and long-term survival after esophagectomy for cancer in a nationwide Swedish cohort study. Eur J Surg Oncol 38(7):555–561
Gottlieb-Vedi E, Kauppila JH, Malietzis G et al (2019) Long-term survival in esophageal cancer after minimally invasive compared to open esophagectomy: a systematic review and meta-analysis. Ann Surg 270(6):1005–1017
Wikman A, Ljung R, Johar A et al (2015) Psychiatric morbidity and survival after surgery for esophageal cancer: a population-based cohort study. J Clin Oncol 33(5):448–454
Markar SR, Lagergren J (2020) Surgical and surgeon-related factors related to long-term survival in esophageal cancer: a review. Ann Surg Oncol 27(3):718–723
Carver CS, Scheier MF, Segerstrom SC (2010) Optimism. Clin Psychol Rev 30(7):879–889
Malouff JM, Schutte NS (2016) Can psychological interventions increase optimism? A meta-analysis. J Posit Psychol 12(6):594–604
Rasmussen HN, Scheier MF, Greenhouse JB (2009) Optimism and physical health: a meta-analytic review. Ann Behav Med 37(3):239–256
Lee LO, James P, Zevon ES et al (2019) Optimism is associated with exceptional longevity in 2 epidemiologic cohorts of men and women. Proc Natl Acad Sci U S A 116(37):18357–18362
Kim ES, Hagan KA, Grodstein F et al (2017) Optimism and cause-specific mortality: a prospective cohort study. Am J Epidemiol 185(1):21–29
Whitfield JB, Zhu G, Landers JG et al (2020) Pessimism is associated with greater all-cause and cardiovascular mortality, but optimism is not protective. Sci Rep 10(1):12609
Anthony EG, Kritz-Silverstein D, Barrett-Connor E (2016) Optimism and mortality in older men and women: The Rancho Bernardo Study. J Aging Res 2016:5185104
Engberg H, Jeune B, Andersen-Ranberg K et al (2013) Optimism and survival: does an optimistic outlook predict better survival at advanced ages? A twelve-year follow-up of Danish nonagenarians. Aging Clin Exp Res 25(5):517–525
Tindle HA, Chang YF, Kuller LH et al (2009) Optimism, cynical hostility, and incident coronary heart disease and mortality in the Women’s Health Initiative. Circulation 120(8):656–662
Giltay EJ, Kamphuis MH, Kalmijn S et al (2006) Dispositional optimism and the risk of cardiovascular death: the Zutphen Elderly Study. Arch Intern Med 166(4):431–436
Giltay EJ, Geleijnse JM, Zitman FG et al (2004) Dispositional optimism and all-cause and cardiovascular mortality in a prospective cohort of elderly dutch men and women. Arch Gen Psychiatry 61(11):1126–1135
Rozanski A, Bavishi C, Kubzansky LD et al (2019) Association of optimism with cardiovascular events and all-cause mortality: a systematic review and meta-analysis. JAMA Netw Open 2(9):e1912200
Schulz R, Bookwala J, Knapp JE et al (1996) Pessimism, age, and cancer mortality Psychol Aging. 11(2):304–309
Allison PJ, Guichard C, Fung K et al (2003) Dispositional optimism predicts survival status 1 year after diagnosis in head and neck cancer patients. J Clin Oncol 21(3):543–548
Price MA, Butow PN, Bell ML et al (2016) Helplessness/hopelessness, minimization and optimism predict survival in women with invasive ovarian cancer: a role for targeted support during initial treatment decision-making? Support Care Cancer 24(6):2627–2634
Schofield PE, Stockler MR, Zannino D et al (2016) Hope, optimism and survival in a randomised trial of chemotherapy for metastatic colorectal cancer. Support Care Cancer. 24(1):401–408
Schandl A, Johar A, Anandavadivelan P et al (2020) Patient-reported outcomes 1 year after oesophageal cancer surgery. Acta Oncol 59(6):613–619
Liu YJ, Schandl A, Markar S et al (2021) Psychological distress and health-related quality of life up to 2 years after oesophageal cancer surgery: nationwide population-based study. BJS Open 5(1):zraa038
Scheier MF, Carver CS, Bridges MW (1994) Distinguishing optimism from neuroticism (and trait anxiety, self-mastery, and self-esteem): a reevaluation of the Life Orientation Test. J Pers Soc Psychol 67(6):1063–1078
Muhonen T, Torkelson EVA (2013) Kortversioner av frågeformulär inom arbets-och hälsopsykologi—om att mäta coping och optimism. Nordisk Psykologi. 57(3):288–297
Carver CS, Scheier MF. Optimism. 2019 Positive psychological assessment: a handbook of models and measures, 2nd ed. Washington, DC, US: American Psychological Association. p. 61–76.
Liu Y, Pettersson E, Schandl A et al (2022) Psychological distress after esophageal cancer surgery and the predictive effect of dispositional optimism: a nationwide population-based longitudinal study. Support Care Cancer 30(2):1315–1322
Ludvigsson JF, Almqvist C, Bonamy AK et al (2016) Registers of the Swedish total population and their use in medical research. Eur J Epidemiol 31(2):125–136
Glaesmer H, Rief W, Martin A et al (2012) Psychometric properties and population-based norms of the Life Orientation Test Revised (LOT-R). Br J Health Psychol 17(2):432–445
Bredal IS, Ekeberg Ø (2016) The stability of dispositional optimism in relation to receiving or not receiving a cancer diagnosis. Psychology 07(06):806–814
Schou I, Ekeberg O, Sandvik L et al (2005) Stability in optimism-pessimism in relation to bad news: a study of women with breast cancer. J Pers Assess 84(2):148–154
Ikeda A, Schwartz J, Peters JL et al (2014) Pessimistic orientation in relation to telomere length in older men: the VA normative aging study. Psychoneuroendocrinology 42:68–76
Roy B, Diez-Roux AV, Seeman T et al (2010) Association of optimism and pessimism with inflammation and hemostasis in the Multi-Ethnic Study of Atherosclerosis (MESA). Psychosom Med 72(2):134–140
Kohut ML, Cooper MM, Nickolaus MS et al (2002) Exercise and psychosocial factors modulate immunity to influenza vaccine in elderly individuals. J Gerontol A Biol Sci Med Sci 57(9):M557–M562
Boehm JK, Williams DR, Rimm EB et al (2013) Relation between optimism and lipids in midlife. Am J Cardiol 111(10):1425–1431
Xie S-H, Wahlin K, Lagergren J (2017) Cause of death in patients diagnosed with esophageal cancer in Sweden: a population-based study. Oncotarget 8(31):51800–51809
Boehm JK, Chen Y, Koga H et al (2018) Is optimism associated with healthier cardiovascular-related behavior? Meta-analyses of 3 health behaviors. Circ Res 122(8):1119–1134
Rius-Ottenheim N, Kromhout D, van der Mast RC et al (2012) Dispositional optimism and loneliness in older men. Int J Geriatr Psychiatry 27(2):151–159
Stek ML, Vinkers DJ, Gussekloo J et al (2005) Is depression in old age fatal only when people feel lonely? Am J Psychiatry 162(1):178–180
Liu Y, Pettersson E, Schandl A et al (2021) Higher dispositional optimism predicts better health-related quality of life after esophageal cancer surgery: a nationwide population-based longitudinal study. Ann Surg Oncol 28(12):7196–7205
Djarv T, Metcalfe C, Avery KN et al (2010) Prognostic value of changes in health-related quality of life scores during curative treatment for esophagogastric cancer. J Clin Oncol 28(10):1666–1670
Modig K, Ahlbom A, Ebeling M (2021) Excess mortality from COVID-19: weekly excess death rates by age and sex for Sweden and its most affected region. Eur J Public Health 31(1):17–22
Ludvigsson JF (2020) The first eight months of Sweden’s COVID-19 strategy and the key actions and actors that were involved. Acta Paediatr 109(12):2459–2471
Fragkaki I, Maciejewski DF, Weijman EL et al (2021) Human responses to COVID-19: the role of optimism bias, perceived severity, and anxiety. Pers Individ Dif 176:110781
Mosing MA, Medland SE, McRae A et al (2012) Genetic influences on life span and its relationship to personality: a 16-year follow-up study of a sample of aging twins. Psychosom Med 74(1):16–22
Lee LO, Aldwin CM, Kubzansky LD et al (2019) The long arm of childhood experiences on longevity: testing midlife vulnerability and resilience pathways. Psychol Aging 34(7):884–899
Acknowledgements
We thank all patients in the OSCAR study and the patient representatives in our research partnership group for their participation. We thank our OSCAR project team for their contributions to the data collection and validation.
Funding
Open access funding provided by Karolinska Institutet. This study was supported by the Robert Lundberg Memorial Foundation under grant number 2021–00658, the Swedish Cancer Society under grant number 180685, Region Stockholm under grant number LS 2018–1157, and the Cancer Research Funds of Radiumhemmet under grant number 171103. Yangjun Liu is supported by the Karolinska Institutet and China Scholarship Council joint program. Pernilla Lagergren is supported by the NIHR Imperial Biomedical Research Centre for her position at Imperial College London, London, UK.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation and data collection were performed by PL. Data analysis was performed by YL and was supervised by AJ. The first draft of the manuscript was written by YL and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Liu, Y., Pettersson, E., Schandl, A. et al. Dispositional optimism and all-cause mortality after esophageal cancer surgery: a nationwide population-based cohort study. Support Care Cancer 30, 9461–9469 (2022). https://doi.org/10.1007/s00520-022-07311-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00520-022-07311-z