Abstract
Purpose
The role of thoracolumbar fascia (TLF) in the development of chronic low back pain (CLBP) has growing evidence in the literature. Although CLBP is reported in individuals with idiopathic scoliosis (IS), its relationship with the TLF has yet not been established. This study aims to evaluate the TLF and its relationship with CLBP in IS.
Methods
A total of 60 individuals were included in the study. They were divided into three groups as follows: painful scoliosis (n = 20, age: 17.1 ± 3.7 years, Cobb angle: 15–43°), non-painful scoliosis (n = 20, age: 16.4 ± 3.4 years, Cobb angle: 15–45°), and healthy group (n = 20, age: 16.4 ± 4.7 years). Pain was evaluated using the short form of the McGill Pain Questionnaire. TLF thickness was evaluated on the lumbar region using ultrasonography. Trunk range of motion was assessed using a universal goniometer, and flexibility was assessed with sit-and-reach test.
Results
The thickness of the right TLF was greatest in the painful group, followed by non-painful (p = 0.007) and healthy (p < 0.001) groups. The thickness of the left TLF in the non-painful and painful groups was greater compared to the healthy group (p < 0.001). In the painful group, right TLF thickness was negatively correlated with trunk flexion/extension (r = −0.540, p = 0.014/r = −0.514, p = 0.020) and left rotation (r = −0.499, p = 0.025) but positively correlated with pain (r = 0.562, p = 0.01).
Conclusions
Thickening of the TLF was observed in IS, whereby, in the presence of CLBP, it was further intensified. We suggest considering fascial thickening as a potential contributing factor to both pain and limited motion in relevant patients.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Idiopathic scoliosis (IS) is a three-dimensional spinal deformity characterized by lateral deviation and rotation of the spine, often associated with a reduction in its normal kyphotic curvature [1, 2]. Biomechanical and morphological variations are prevalent among individuals with IS [3]. They encompass discrepancies in limb–trunk asymmetry, postural malalignment, compensatory motion patterns, alterations in rib, pelvis, arm, and skull morphology, as well as reduction in the overall body stability/mobility. These changes result in postural deformity of the spine with chronic repetitive asymmetric stress on it [4]. Eventually, various problems such as muscle imbalance, functional limitation, low back pain, and their negative effects on the psychosocial well-being and body image might ensure [5].
IS has also been identified as a potential risk factor for the development of chronic low back pain (CLBP) in adolescents [6, 7]. The latter was reported with prevalence rates ranging from 27.5% to 72% in adolescents with IS [8, 9] having negative impact on physical and psychological functions as well as quality of life [10]. The etiology of CLBP includes asymmetric facet joint loading (resulting in arthritis/synovitis), muscular pain due to eccentric load on the curvature apex, and intervertebral disk problems [11, 12]. However, the relationship between CLBP and IS has yet not been well established [13].
The thoracolumbar fascia (TLF) is an anatomical structure composed of dense connective tissue layers in the lumbar region. It comprises aponeurotic and fascial planes united around the spinal muscles. The TLF plays an essential role in postural alignment and stability/movement of the spinal column [14, 15]. There is growing evidence as regards the role of TLF in individuals with CLBP [16]. For instance, it has been found to be thicker in subject with CLBP [17]. Moreover, any pathology in the TLF can impair its flexibility and elastic characteristics, thereby restricting the trunk range of motion [18]. Herewith, the TLF thickness has not been examined in individuals with IS. To this end, the hypothesis of our study was that there could be a relationship between CLBP and TLF in individuals with IS, whereby the thickness might affect trunk motion and flexibility. Accordingly, the present study aimed to evaluate the TLF and its relationship with CLBP in IS.
Materials and Methods
Study design
The data were collected between July–October 2023. The participants were recruited from individuals who were referred to the Department of Physical Therapy and Rehabilitation at university hospital. All individuals were informed prior to inclusion in the study, and written informed consent was collected from participants aged 18 and above. For participants under the age of 18, informed consent was also obtained from the parents.
Participants
A total of 60 individuals with IS were enrolled and divided into three groups: The painful group consisted of individuals who had pain for more than three months, with a severity score of 3 or higher based on the visual analog scale (VAS); the non-painful group comprised individuals with no pain; and the healthy group had no spine pathology or low back pain.
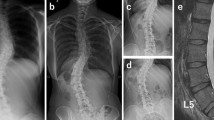
Individuals with IS (above the age of 10) were included if they had a left lumbar curve with a Cobb angle of more than 15 degrees. Healthy individuals (above the age of 10) were recruited if they had no back pain (VAS score = 0), orthopedic, neurological, or rheumatic problems that could affect spinal health. Individuals with a history of lower extremity or spinal region surgery/trauma were excluded.
Outcome measures
Age, gender, height, weight, and body mass index were collected from all participants. Cobb angle and axial trunk rotation were noted in the scoliosis group.
Pain severity was evaluated using the Turkish version of the short form of the McGill Pain Questionnaire (SF-MPQ) to assess the sensory/affective characteristics of pain and its intensity [19]. The primary component comprises 15 descriptors (11 sensory; four affective) that are assessed on an intensity scale, with ratings ranging from 0 (none) to 3 (severe). The second section includes five words designed to determine the intensity of pain. The third section entails the participant’s evaluation of the current pain intensity using VAS. Three pain scores are computed based on the sum of the intensity rank values assigned to the chosen words for sensory, affective, and overall descriptors.
Thickness of the TLF was measured at the L3 lumbar vertebra level by ultrasonography and using a previously described standardized protocol. Bilateral measurements (mm) were taken as subjects lay relaxed in prone position, and the transducer was localized vertical to the spine, approximately 2–3 cm lateral to the L3 spinous process. In order to mitigate the potential impact of thickness variations, three equidistant-spaced points were assessed per image for TLF and averaged for analysis (Fig. 1) [20]. A single physiatrist with 25-year experience in musculoskeletal ultrasound performed all the measurements.
Active trunk range of motions were assessed during flexion/extension, rotation, and lateral flexion using the universal goniometer. Prior to each evaluation, individuals were provided with an explanation of the motion and given an opportunity to perform a trial. During the assessment of trunk flexion and extension, the therapist stood on the lateral side of the individual. The goniometer’s pivot rested on the hip at the lateral projection of the lumbosacral joint. The stable arm was perpendicular to the ground, while the moving arm traced the lateral projection of the trunk toward the axilla. Special attention was given to preventing any hip joint motion throughout the measurement. For lateral flexion measurement, the therapist stood behind the individual. The goniometer’s pivot was at the midpoint of the lumbosacral joint, with the stable arm parallel to the posterior superior iliac spine and the ground. The moving arm followed the spinal processes of the lumbar vertebrae toward the cranial side. The therapist ensured no rotation, flexion, or extension of the trunk during the measurements. During the assessment for lumbar rotation, the therapist stood behind the seated individual. The pivot point was centered on the head, with the fixed goniometer arm parallel to the ground. The moving arm followed the acromion in the opposite direction of rotation. In both right and left rotation measurements, careful attention was paid to prevent any cranial rotation by the subject [21, 22].
Flexibility of the trunk was evaluated using the sit-and-reach test [23]. A tape measure was placed on a table at the soles of the feet. The individuals leaned forward slowly reaching as far as possible while keeping their hands adjacent to each other. After a familiarization test, the best of three trials was recorded (cm).
Statistical analysis
Data were analyzed using IBM SPSS 27 for Windows software. Descriptive statistical methods were initially employed. Normal distribution was assessed using the Kolmogorov–Smirnov test. To assess differences among the demographic characteristics of the groups, chi-square test was employed for gender, ANOVA test for age, height, and body weight, and Mann–Whitney U test for Cobb angle and rotation. When comparing trunk motions between groups, ANOVA and Kruskal–Wallis tests were used, where appropriate. Correlation analyses were performed using Pearson and Spearman correlation coefficients, as appropriate. To determine significant differences among groups, Bonferroni correction was employed for post-hoc analysis.
In assessing intra-rater reliability, we employed the intra-class correlation coefficient (ICC; two-way mixed effect model (absolute agreement)) based on three repeated ultrasound measurements. The interpretation of ICC values followed established criteria: poor (< 0.40), fair (0.40–0.59), good (0.60–0.74), and excellent (0.75–1.00) [24]. Additionally, we computed Cronbach’s alpha, Standard Error of Measurement (SEM), and minimum detectable changes (MDC = √2 × 1.96 × SEM) in accordance with the recommendations of Bland and Altman [25].
For determining an adequate sample size, a power analysis was conducted using G*Power Version 3.1.9.2 software. The effect size (f) obtained from a pilot study with 30 participants on scoliotic and healthy individuals was found to be 0.56. With a 95% power and type I error rate of 5% (α = 0.05), the study aimed to include 54 participants. However, considering equal distribution among groups and accounting for environmental and unforeseen factors, 60 individuals who voluntarily agreed to participate were enrolled.
Results
Demographics of the participants are presented in Table 1. No significant differences were observed among the groups in terms of age, height, body weight, and body mass index (all p > 0.05). There was no significant difference in Cobb angle and axial trunk rotation degrees between the non-painful and painful groups (both p > 0.05).
Differences in right and left TLF thicknesses were observed among the groups (Table 2). Post-hoc tests revealed that the right TLF thickness of the painful group was greater than the non-painful (p = 0.007) and the healthy (p < 0.001) groups. The right TLF thickness of the non-painful group was also greater than that of the healthy group (p = 0.015). The left TLF thickness of both the non-painful (p = 0.005) and painful (p = 0.001) groups was greater than that of the healthy group, whereby the scoliosis groups were similar in this aspect (p > 0.05). Regarding trunk motions, left lateral flexion was decreased in the painful (vs. healthy) group (p = 0.025).
With respect to the relationship between TLF thickness, trunk motion, flexibility, and pain (Table 3), no correlation was found in either the non-painful or the healthy group (all p > 0.05). In the painful group, moderate and negative correlations were found between the right TLF thickness and trunk flexion (r = −0.540, p = 0.01), trunk extension (r = −0.514, p = 0.02), and left rotation (r = −0.499, p = 0.02). There was also a moderate negative correlation between the left TLF thickness and trunk extension (r = −0.528, p = 0.02) and a moderate positive correlation between the right TLF thickness and MPQ (r = 0.562, p = 0.01).
Intra-rater reliability
The ultrasound outcome measurements exhibited consistently excellent reliability across three repeated assessments. This evaluation was based on key statistical indicators, including Cronbach’s α, Intra-class Correlation Coefficient (ICC), Standard Error of Measurement (SEM), and Minimal Detectable Change (MDC), all thoroughly summarized in Table 4.
Discussion
This study revealed that the TLF thicknesses were greater on either side of the curve in individuals with left-sided lumbar scoliosis (vs. healthy peers). Additionally, between individuals with IS, those who had CLBP had greater fascia thickness on the concave side when compared to non-painful individuals. Although TLF thickness was found to be positively correlated with pain, there was no association between fascial thickness and trunk motion in healthy or non-painful IS subjects. However, TLF thickness was particularly observed to be related to trunk motion limitation in the sagittal and horizontal planes in IS with CLBP.
Studies in the literature have confirmed asymmetries of the paravertebral and abdominal wall muscles in individuals with IS. Using ultrasound measurements, Karina et al. [26] found significantly greater muscle thickness on the concave side of patients with mild IS (vs. controls). An observational study with magnetic resonance imaging and histology showed that muscle degeneration was more common on the concave side in patients with degenerative lumbar scoliosis [27]. However, studies focused on muscles and did not examine any TLF abnormality. In our study we found that in individuals with IS, fascia thickness was significantly different from healthy individuals on both the convex and concave sides. It is noteworthy that the fascia thickens in individuals with IS. We speculate that the TLF might have undergone remodeling over time in response to repetitive stress due to compensatory patterns of motion, postural deviation, or asymmetric loading.
Another ultrasonographic study conducted in individuals with CLBP lasting more than 12 months reported increased thickness and echogenicity of the perimuscular connective tissues forming the TLF [17]. Similarly, Pirri et al. [20] and Gumruk et al. [28] showed that the TLF was thicker in patients with non-specific CLBP, whereby the latter also found positive correlation between TLF thickness and pain intensity. Our results yielded greater concave TLF thickness in painful (vs. non-painful) IS and positive correlation between TLF thickness and pain on the concave side. At this point, although a causal relationship between thicker TLF and CLBP cannot be ascertained, the former can be considered as a predisposing factor—also in light of the pertinent literature.
In previous cross-sectional (ultrasonographic) studies, thicker TLF with decreased mobility was identified in individuals with non-specific CLBP vs. non-painful peers [17, 20, 29, 30]. The potential role of fascia in CLBP was proposed by Langevin et al. [31] where pain-related fear was thought to induce a cycle of decreased motion. In turn, it was proposed that an altered motion pattern may result in connective tissue remodeling of TLF—leading to inflammation, nervous tissue sensitization, and further decreased mobility. Moreover, changes in the properties of fascia, such as thickness, may restrict muscular extensibility and range of motion [32]. A follow-up investigation found that TLF shear strain during passive trunk flexion was reduced in people with CLBP by 56% [29]. These changes alter/limit daily motion, determining the flaccidity of the spine and modifying the pattern of motion [33]. According to our results, in the painful group, a negative correlation was observed between the thickness of the concave TLF and trunk flexion–extension and left rotation. Structural and functional abnormalities of the TLF could have contributed to motion restriction, which could also/actually be worsened by pain.
Main strength of the current study is the inclusion of individuals with IS of the single lumbar curve pattern. This enabled evaluations to be conducted specifically for a certain type of curvature. In both painful and non-painful groups, individuals were found to have similar curve magnitudes and axial rotation degrees. Therefore, the potential impact of curve magnitude and rotation degree on the results has been prevented. Additionally, the use of ultrasound provided reliable/objective morphological assessment of the TLF thickness. On the other hand, this study has some limitations, yet, the results cannot be generalized to individuals who have other curve types or magnitudes. Further, the fact that TLF thickness measurements were taken at a single (L3 vertebra) level could be another drawback. Nonetheless, our study results can add to the growing evidence of data as regards the association between TLF and CLBP in subjects with IS.
Conclusion
This study showed that when comparing individuals with IS to healthy counterparts, the thickness of the TLF exhibits a notable increase. Additionally, in individuals with CLBP, the fascia in the concave region is found to be thicker than that of non-painful peers. TLF thickness is also associated with limitation of trunk motion in the sagittal/horizontal planes in painful individuals. Last but not least, to our best notice, our study is the first to have investigated the TLF in individuals with IS and examined its relationship with pain. Future research should investigate the cause and effect relationship of the aforementioned findings in a wider group of IS patients with different disease features.
Abbreviations
- IS:
-
Idiopathic scoliosis
- TLF:
-
Thoracolumbar fascia
- CLBP:
-
Chronic low back pain
- SF-MPQ:
-
Short form of the McGill Pain Questionnaire
- VAS:
-
Visual analog scale
References
Choudhry MN, Ahmad Z, Verma R (2016) Adolescent idiopathic scoliosis. Open J Orthop 10:143. https://doi.org/10.2174/1874325001610010143
Trobisch P, Suess O, Schwab F (2010) Idiopathic scoliosis. Dtsch Arztebl Int 107(49):875. https://doi.org/10.3238/arztebl.2010.0875
Yagci G, Ayhan C, Yakut Y (2018) Effectiveness of basic body awareness therapy in adolescents with idiopathic scoliosis: a randomized controlled study. J Back Musculoskelet Rehabil 31(4):693–701. https://doi.org/10.3233/BMR-170868
Stehbens WE (2003) Pathogenesis of idiopathic scoliosis revisited. Exp Mol Pathol 74(1):49–60. https://doi.org/10.1016/S0014-4800(03)80008-4
Yagci G, Yakut Y, Simsek E (2018) The effects of exercise on perception of verticality in adolescent idiopathic scoliosis. Physiother Theory Pract 34(8):579–588. https://doi.org/10.1080/09593985.2017.1423429
Sato T, Hirano T, Ito T, Morita O, Kikuchi R, Endo N et al (2011) Back pain in adolescents with idiopathic scoliosis: epidemiological study for 43,630 pupils in Niigata City, Japan. Eur Spine J 20:274–279. https://doi.org/10.1007/s00586-010-1657-6
Théroux J, Le May S, Hebert JJ, Labelle H (2017) Back pain prevalence is associated with curve-type and severity in adolescents with idiopathic scoliosis. Spine 42(15):E914–E919. https://doi.org/10.1097/BRS.0000000000001986
Theroux J, Stomski N, Hodgetts CJ, Ballard A, Khadra C, Le May S et al (2017) Prevalence of low back pain in adolescents with idiopathic scoliosis: a systematic review. Chiropr Man Ther 25(1):1–6. https://doi.org/10.1186/s12998-017-0143-1
Grauers A, Topalis C, Möller H, Normelli H, Karlsson MK, Danielsson A et al (2014) Prevalence of back problems in 1069 adults with idiopathic scoliosis and 158 adults without scoliosis. Spine 39(11):886–892. https://doi.org/10.1097/BRS.0000000000000312
Théroux J, Le May S, Fortin C, Labelle H (2015) Prevalence and management of back pain in adolescent idiopathic scoliosis patients: a retrospective study. Pain Res Manag 20:153–157. https://doi.org/10.1155/2015/674354
Buttermann GR, Mullin WJ (2008) Pain and disability correlated with disc degeneration via magnetic resonance imaging in scoliosis patients. Eur Spine J 17(2):240–249. https://doi.org/10.1007/s00586-007-0530-8
G. Bisson D, Lama P, Abduljabbar F, Rosenzweig DH, Saran N, Ouellet JA et al (2018) Facet joint degeneration in adolescent idiopathic scoliosis. JOR Spine 1(2):e1016. https://doi.org/10.1002/jsp2.1016
López-Torres O, Mon-López D, Gomis-Marzá C, Lorenzo J, Guadalupe-Grau A (2021) Effects of myofascial release or self-myofascial release and control position exercises on lower back pain in idiopathic scoliosis: A systematic review. J Bodyw Mov Ther 27:16–25. https://doi.org/10.1016/j.jbmt.2021.02.017
Gatton M, Pearcy M, Pettet G, Evans J (2010) A three-dimensional mathematical model of the thoracolumbar fascia and an estimate of its biomechanical effect. J Biomech 43(14):2792–2797. https://doi.org/10.1016/j.jbiomech.2010.06.022
Benetazzo L, Bizzego A, De Caro R, Frigo G, Guidolin D, Stecco C (2011) 3D reconstruction of the crural and thoracolumbar fasciae. Surg Radiol Anat 33:855–862. https://doi.org/10.1007/s00276-010-0757-7
De Coninck K, Hambly K, Dickinson JW, Passfield L (2018) Measuring the morphological characteristics of thoracolumbar fascia in ultrasound images: an inter-rater reliability study. BMC Musculoskelet Disord 19(1):1–6. https://doi.org/10.1186/s12891-018-2088-5
Langevin HM, Stevens-Tuttle D, Fox JR, Badger GJ, Bouffard NA, Krag MH et al (2009) Ultrasound evidence of altered lumbar connective tissue structure in human subjects with chronic low back pain. BMC Musculoskelet Disord 10(1):1–9. https://doi.org/10.1186/1471-2474-10-151
Wilke J, Macchi V, De Caro R, Stecco C (2019) Fascia thickness, aging and flexibility: is there an association? J Anat 234(1):43–49. https://doi.org/10.1111/joa.12902
Melzack R (1987) The short-form McGill pain questionnaire. Pain 30(2):191–197. https://doi.org/10.1016/0304-3959(87)91074-8
Pirri C, Pirri N, Guidolin D, Macchi V, Porzionato A, De Caro R et al (2023) Ultrasound imaging of thoracolumbar fascia thickness: chronic non-specific lower back pain versus healthy subjects; A sign of a “Frozen Back”? Diagnostics 13(8):1436. https://doi.org/10.3390/diagnostics13081436
Apti A, Çolak T, Akçay B (2023) Normative values for cervical and lumbar range of motion in healthy young adults. J Turk Spinal Surg 34(3). https://doi.org/10.4274/jtss.galenos.2023.33042
Johnson M, Mulcahey M (2021) Interrater reliability of spine range of motion measurement using a tape measure and goniometer. J Chiropr Med 20(3):138–147. https://doi.org/10.1016/j.jcm.2021.09.003
Hui S-C, Yuen PY (2000) Validity of the modified back-saver sit-and-reach test: a comparison with other protocols. Med Sci Sports Exerc 32(9):1655–1659. https://doi.org/10.1097/00005768-200009000-00021
Hallgren KA (2012) Computing inter-rater reliability for observational data: an overview and tutorial. Tutor Quant Methods Psychol 8(1):23. https://doi.org/10.20982/tqmp.08.1.p023
Bland JM, Altman D (1986) Statistical methods for assessing agreement between two methods of clinical measurement. The Lancet 327(8476):307–310. https://doi.org/10.1016/S0140-6736(86)90837-8
Zapata KA, Wang-Price SS, Sucato DJ, Dempsey-Robertson M (2015) Ultrasonographic measurements of paraspinal muscle thickness in adolescent idiopathic scoliosis: a comparison and reliability study. Pediatr Phys Ther 27(2):119–125. https://doi.org/10.1097/PEP.0000000000000131
Shafaq N, Suzuki A, Matsumura A, Terai H, Toyoda H, Yasuda H et al (2012) Asymmetric degeneration of paravertebral muscles in patients with degenerative lumbar scoliosis. Spine 37(16):1398–1406. https://doi.org/10.1097/BRS.0b013e31824c767e
Gumruk Aslan S, Koylu Uyar S, Gurcay E (2023) Potential role of thoracolumbar fascia in younger middle-aged patients with chronic low back pain. Int J Neurosci 1–7. https://doi.org/10.1080/00207454.2023.2251671
Langevin HM, Fox JR, Koptiuch C, Badger GJ, Greenan-Naumann AC, Bouffard NA et al (2011) Reduced thoracolumbar fascia shear strain in human chronic low back pain. BMC Musculoskelet Disord 12:1–11. https://doi.org/10.1186/1471-2474-12-203
Liu Y, Pan A, Hai Y, Li W, Yin L, Guo R (2019) Asymmetric biomechanical characteristics of the paravertebral muscle in adolescent idiopathic scoliosis. Clin Biomech 65:81–86. https://doi.org/10.1016/j.clinbiomech.2019.03.013
Langevin HM, Sherman KJ (2007) Pathophysiological model for chronic low back pain integrating connective tissue and nervous system mechanisms. Med Hypotheses 68(1):74–80. https://doi.org/10.1016/j.mehy.2006.06.033
Wilke J, Krause F, Vogt L, Banzer W (2016) What is evidence-based about myofascial chains: a systematic review. Arch Phys Med Rehabil 97(3):454–461. https://doi.org/10.1016/j.apmr.2015.07.023
Hodges PW, Danneels L (2019) Changes in structure and function of the back muscles in low back pain: different time points, observations, and mechanisms. J Orthop Sports Phys Ther 49(6):464–476. https://www.jospt.org / https://doi.org/10.2519/jospt.2019.8827
Funding
Open access funding provided by the Scientific and Technological Research Council of Türkiye (TÜBİTAK).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest and source of funding
There are no conflicts of interest for any author.
Ethical approval and clinical trials
The study was ethically approved by the Hacettepe University Non-Interventional Clinical Research Ethics Committee with the decision dated 06.20.2023 and numbered GO 23/516 and registered to ClinicalTrials.gov with ID number NCT06055140.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Yerli, S., Yinanç, S.B., Yağcı, G. et al. Thoracolumbar fascia and chronic low back pain in idiopathic lumbar scoliosis: an ultrasonographic study. Eur Spine J 33, 2469–2475 (2024). https://doi.org/10.1007/s00586-024-08266-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00586-024-08266-x