Abstract
Background
To study the clinical condition of poor-grade aneurysmal subarachnoid hemorrhage (aSAH) patients alive at 3 years after neurointensive care.
Methods
Of the 769 consecutive aSAH patients from a defined population (2005–2015), 269 (35%) were in poor condition on admission: 145 (54%) with H&H 4 and 124 (46%) with H&H 5. Their clinical lifelines were re-constructed from the Kuopio Intracranial Aneurysm Database and Finnish nationwide registries. Of the 269 patients, 155 (58%) were alive at 14 days, 125 (46%) at 12 months, and 120 (45%) at 3 years.
Results
The 120 H&H 4–5 patients alive at 3 years form the final study population. On admission, 73% had H&H 4 but only 27% H&H 5, 59% intracerebral hematoma (ICH; median 22 cm3), and 26% intraventricular blood clot (IVH). The outcome was favorable (mRS 0–1) in 45% (54 patients: ICH 44%; IVH clot 31%; shunt 46%), moderate (mRS 2–3) in 30% (36 patients: ICH 64%; IVH clot 19%; shunt 42%), and unfavorable (mRS 4–5) in 25% (30 patients: ICH 80%; IVH clot 23%; shunt 50%). A total of 46% carried a ventriculoperitoneal shunt. ICH volume was a significant predictor of mRS at 3 years.
Conclusions
Of poor-grade aSAH patients, 45% were alive at 3 years, even 27% of those extending to pain (H&H 5). Of the survivors, 75% were at least in moderate condition, while only 2.6% ended in hospice care. Consequently, we propose non-selected admission to neurointensive care (1) for a possibility of moderate outcome, and (2), in case of brain death, possibly improved organ donation rates.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Aneurysmal subarachnoid hemorrhage (aSAH), in most cases from a ruptured saccular intracranial aneurysm (sIA), is the third most frequent form of stroke [26, 50]. The mechanisms of acute brain injury include intracerebral hemorrhage (ICH), intraventricular hemorrhage (IVH), acute brain ischemia, acute hydrocephalus, increased intracranial pressure (ICP), and seizures [7, 9, 46]. Further deterioration may be due to uncontrollable ICP, delayed ischemic brain injury, electrolyte disturbances, cardiopulmonary complications, and CNS or systemic infections [7, 9, 12, 40, 46].
Poor condition on admission is usually expressed as Hunt and Hess (H&H) scale 4 or 5, or World Federation of Neurosurgical Societies (WFNS) grade 4 or 5 [6, 48]. The extent of intracranial bleeding is expressed as CT scales, including the Fisher scale, while computed bleeding volume and site analyses are emerging [13, 40]. Poor condition predicts high early mortality, and aSAH patients brain dead within 14 days are a significant group of organ donors [21]. In Finland, with national presumed consent (opt-out) since 2010 [21, 45], aSAH patients with dismal prognosis can be admitted to neurointensive care as potential organ donors, also at high age. Non-selected admission to neurointensive care would increase organ donation [21, 45]—but is long-term hospice care survival (mRS 5) increasing as well? The long-term mortality is often presented at 12 months [47]: 49% for H&H 4 and 92% for H&H 5 in our previous study [22]. A minority of H&H 4–5 or WFNS 4–5 patients will become true long-term survivors, alive e.g., at 5 years or 10 years, albeit being stroke-risk carriers and prone to further vascular events [16]. They have remained an unrecognized group among stroke survivors for obvious reasons: prospective databases with long recruitment and outpatient follow-up periods would be virtually impossible to maintain, while much of literature is focused on “aneurysm treatment” [43].
We constructed the clinical lifelines of 269 consecutive aSAH patients with H&H 4 or 5 on admission to the Neurointensive Care Unit of the Kuopio University Hospital (KUH) from a defined population between 2005 and 2015 by using the nationwide registries and personal identity codes. Our aim is to present a cross-sectional analysis of the 120 (45%) H&H 4–5 patients alive at 3 years. The 120 primary CT scans (ICH; IVH) and the interventions (ICH evacuation; decompressive craniectomy; shunt) are compiled together, according to the modified Rankin Scale (mRS) at 3 years.
Methods and materials
Kuopio University Hospital (KUH) and aSAH management protocol in Eastern Finland
KUH, one of the five university hospitals in Finland, is an academic, non-profit, publicly funded tertiary center, which serves a defined population (about 842,000 in 2015) in Eastern Finland. The KUH area contains four central hospitals, each with 24/7 neuroacutology, CT services, intensive care, neurology, and neurorehabilitation. The KUH area is served by ambulance and (since 2002) by helicopter transfer. At KUH Neurosurgery, at least two neurosurgeons are on duty at all times. During the study period from 2005 to 2015, all cases of SAH were acutely transferred to KUH for neurointensive care, neuroradiology (4-vessel angiography and/or CT angiography), and neurosurgery [21]. Neurointensive care was provided to virtually all patients regardless of the age or condition on admission, including H&H 4–5 patients [21]. A dedicated team of neurointensivists, neurosurgeons, and interventional neuroradiologists coordinated the aSAH treatment. The KUH Neurovascular Group provided microsurgical or endovascular occlusion of the ruptured aneurysm and evacuated significant ICHs with immediate microsurgery. The KUH aSAH neurointensive care protocol in 2005–2015 followed international recommendations in detail [9, 27, 29, 44, 46], and it was presented in our previous study of organ donors after aSAH [21, 45]. Briefly, the protocol aimed to prevent further brain damage due to rebleeding, increased intracranial pressure (ICP), hydrocephalus, electrolyte disturbances, seizures, cardiac and pulmonary dysfunction, fever, hyperglycemia, and development of delayed brain ischemia. The protocol included, when appropriate, e.g., external ventricular drainage (EVD), parenchymal ICP monitoring, endovascular procedures, and intra-arterial nimodipine infusion in case of delayed brain ischemia, as well as decompressive craniectomy (DC).
Kuopio Intracranial Aneurysm Patient and Family Database
The database, prospective since 1995, contains all cases of unruptured and ruptured IAs admitted to KUH since 1980. A dedicated, full-time nurse administrates the database, interviews all new IA patients, including their family history, and arranges the follow-ups. The clinical data, including prescribed medicines, hospital diagnosis, and causes of death, have been fused from the national registries, using the Finnish personal codes. We have characterized the aSAH patients, e.g., for the 14-day mortality and organ donation [21], 12-month [22] and long-term excess mortality [17], shunt-dependent hydrocephalus [1], depression [19], epilepsy [18], pain [33], anti-psychotics [39], diabetes mellitus [31], hypertension [25, 32], as well as, polycystic kidney disease [36]. Three first-degree relatives with a diagnosed sIA disease form an sIA family.
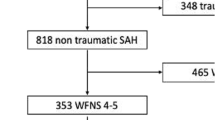
Basic study population of 269 aSAH patients in poor condition on admission
A total of 769 consecutive aSAH patients were acutely admitted to the KUH Neurointensive Care Unit from 2005 to 2015 (Fig. 1). The six patients lost to follow-up were excluded. A total of 269 (35%) aSAH patients had H&H 4 (n=145) or H&H 5 (n=124; extension to pain) on arrival or before intubation before arrival to the KUH (Fig. 1). Their clinical lifelines were re-constructed from their clinical data in the Kuopio database and from the national clinical registries until death (n=149) or July 2019 (Table 1). The patients who deteriorated from H&H 1–3 on admission to H&H 4–5 during the neurointensive care were excluded.
Final study population of 120 aSAH patients alive at 3 years
A total of 120 H&H 4 (n=88) or H&H 5 (n=32) patients were alive at 3 years after admission (Tables 1 and 2). Their 120 primary digital CT scans were analyzed for the presence or absence of ICH, IVH blood clot, IVH sediment, and hydrocephalus (Table 2). A representative CT slice of each patient was compiled together and sorted according to the modified Rankin Scale (mRS 0 to 5) at 3 years. The ICH volumes were calculated from the CT scans using the formula π × (a × b × c) / 6 where a, b, and c are the three perpendicular diameters of ICH. All available clinical data were obtained from the KUH electronic health care records, purchases of prescripted drugs, and all hospital and primary health care diagnoses from the national databases. By our definition, the use of anti-epileptic, anti-depressive, or anti-psychotic medications excluded mRS 0.
Literature review
Firstly, PubMed was searched in November 2020 for English articles on human aSAH published between 2010 and 2020 with the words (Aneurysm* AND “subarachnoid hemorrhage”) AND ((poor-grade) OR (poor grade)) AND (long-term AND (mortality OR survival OR outcome OR prognosis)) AND (12 months OR twelve-month). This gave 14 hits.
Secondly, we identified the original clinical aSAH patient cohorts, excluding case reports, duplicate publications, systematic reviews, and meta-analyses.
Thirdly, we approved the aSAH cohorts fulfilling the following criteria:
-
1.
Poor condition on acute admission, expressed as H&H 4 or 5, or WFNS 4 or 5, or GCS 3 to 12.
-
2.
Over 200 poor-grade aSAH patients to have a significant group of long-term survivors (mortality rate at 50%).
-
3.
Median or mean follow-up time over 12 months.
-
4.
Outcome expressed at more than 12 months using the modified Rankin Scale (mRS), or Glasgow Outcome Scale (GOS), or Extended Glasgow Outcome Scale (GOSE).
Finally, none of the articles fulfilled the criteria.
Statistical methods
The categorical variables were expressed in proportions, and the χ2 test was used in comparisons. The continuous variables were expressed in medians, quartiles, and ranges, and the non-parametric tests were used in comparisons. The Kaplan-Meier analysis was used to calculate the cumulative mortality rates, and the log-rank test was used to test for differences between groups. Independent risk factors for the clinical condition at 3 years of the 120 patients were searched with logistic regression analysis. P values < 0.05 were considered significant. We used the SPSS 27 statistical software (SPSS, Inc., Chicago, IL).
Results
Basic study population of 269 aSAH patients in poor condition on admission
A total of 269 consecutive aSAH patients were in poor condition (H&H 4, n=145; H&H 5, n=124) on admission to the KUH Neurointensive Care Unit (flowchart in Fig. 1, Table 1). Table 1 presents their clinical characteristics on admission, as well as those alive at 12 months and 3 years. Of the 269 patients, 155 (58%) were alive at 14 days, 125 (46%) at 12 months, and 120 (45%) at 3 years, significantly more often the H&H 4 patients (Fig. 2). Decompressive craniectomy (DC) was performed in 35 (13%) patients in a median of 2 (IQR 1–4) days; 13 (37%) of them died within 14 days (Table 1).
The cumulative survival rates for the H&H 4 (n=145) patients and the H&H 5 (n=124) patients after admission for acute aSAH. Cumulative survival rates at 14 days, 12 months, and 3 years of 269 consecutive aneurysmal subarachnoid hemorrhage (aSAH) patients admitted acutely in poor condition (H&H scale 4 or 5) to the Neurointensive Care Unit of the Kuopio University Hospital from its Eastern Finnish catchment population from 2005 to 2015. The follow-up time is logarithmic to emphasize the early high mortality. H&H, Hunt & Hess scale
Final study population of 120 aSAH patients alive at 3 years
At 3 years, 120 (45%) patients were alive, 88 (73%) after H&H 4 and 32 (27%) after H&H 5 on admission (Table 1). Their clinical condition at 3 years is distributed as follows (Table 2, Fig. 3): 14% mRS 0; 31% mRS 1; 17% mRS 2; 13% mRS 3; 19% mRS 4; and 6% mRS 5 (nursing home or hospital). By 3 years, 33 (28%) patients had started new anti-epileptic (AE) medication, 33 (28%) new anti-depressive (AD) medication, and 16 (13%) new anti-psychotic (AP) medication (Table 2). A total of 55 (46%) patients carried a ventriculoperitoneal shunt (Tables 1 and 2, Fig. 3).
Primary CT scans of the 120 aSAH patients alive at 3 years, arranged according to their mRS. A total of 269 aSAH patients had been acutely admitted in poor condition (H&H 4; n=145 or H&H 5; n=124) to the neurointensive care between 2005 and 2015 from Eastern Finnish catchment population (Fig. 1). At 3 years, 120 (45%) of them were alive. This presents one representative CT slice of each patient (age in years) on admission, arranged according to the patient’s mRS from 0 to 5 at 3 years. There were 71 (59%) intracerebral hematomas (ICHs) on admission, 29 (41%) of them microsurgically evacuated (ICHE). There were 31 (26%) intraventricular hematomas (IVHs). Extraventricular drainage (EVD) was used in 105 (88%) patients and not used in 15 (13%). A total of 16 (13%) decompressive craniectomies (DC) were performed. In two patients in poor condition (mRS 5), cranioplasty was not performed. A ventriculoperitoneal shunt (Sh) was subsequently inserted in 55 (46%) patients. The collection is zoomable to observe details in different CT slices. By using the Find command, the following can be simultaneously identified in the whole collection: mRS, ICH, ICHE, IVH, DC, or Sh, or their different combinations. aSAH, aneurysmal subarachnoid hemorrhage; H&H, Hunt and Hess scale; CT, computed tomography; mRS, modified Rankin Scale; ICH, intracerebral hematoma; ICHE, intracerebral hematoma microsurgically evacuated; IVH, intraventricular hematoma (blood clot); EVD, extraventricular drainage; DC, decompressive craniectomy; Sh, shunt. Small blood sedimentations in occipital tips of the lateral ventricles were not considered as intraventricular clots (see above IVH)
Primary CT scans of 120 aSAH patients and their mRS at 3 years
Figure 3 presents one representative CT slice of each patient, together with the surgical interventions (see below), arranged according to the patient’s modified Rankin Scale (mRS) from 0 to 5 at 3 years. There were 71 (59%) intracerebral hematomas (ICH) with a median volume of 22 cm3 (IQR 11–56), 47 (66%) from the middle cerebral artery (MCA) sIA, and 12 (17%) from the anterior communicating artery (AComA) sIA. There were 31 (26%) intraventricular blood clots (IVH), 15 (48%) from the AComA sIA, and 10 (32%) from the MCA sIA. Mere intraventricular blood sediments were seen in 59 (49%). Acute hydrocephalus was present in 76 (63%) patients.
Neurointensive care and surgical interventions of 120 aSAH patients
Extraventricular drainage (EVD) was started in 105 patients (88%). Table 3 cross-tabulates the 12 combinations of microsurgical evacuation of ICH (n=29), microsurgical clipping (n=55) or endovascular occlusion (n=64) of ruptured sIA, and decompressive craniectomy (n=16). In each of the 12 patient groups, median mRS at 3 years is presented. Figure 3 shows the representative CT slices for each of the 12 patient groups. Intraventricular blood clots, present in 31 (26%) patients (Fig. 3), were not evacuated microsurgically or endoscopically; one patient had intraventricular alteplase (t-PA) thrombolysis.
Shunt dependency among 120 aSAH patients
A total of 55 (46%) patients had a ventriculoperitoneal shunt (Fig. 3), inserted in a median of 17 days (IQRs 9 and 64) after aSAH. In the 55 shunted patients, the ventricles on admission contained IVH clot in 18 (33%) (Fig. 3), mere blood sedimentation in 34 (62%), and no blood in 3 (5%). Until 3 years, 32 (58%) patients had no shunt revisions (median mRS 1). A total of 23 (42%) patients had shunt revisions (median mRS 3), 11 once, and 12 two to five times. At 3 years, the 65 non-shunted patients had a median mRS of 2.
Cranioplasty after decompressive craniectomy among 120 aSAH patients
DC was performed in 16 (13%) patients (Table 1 and 3, Fig. 3) in a median of 2 (IQR 0–5) days after admission. In 14 of the 16 DC patients, cranioplasty was performed in a median of 94 (IQR 46–127) days after DC, always with the own frozen bone flap; 4 flaps were later replaced with an artificial flap.
Favorable condition (mRS 0 or 1)
At 3 years, 54 patients (median 55 years) had mRS 0 or 1, 45% of the 120 three-year survivors but only 20% of all the 269 original aSAH patients with H&H 4–5 on admission (Table 2). As many as 17 (31%) patients had no symptoms (mRS 0; no AE, AD, or AP drug use) despite minor ICH in five, IVH blood clot in five, or shunt in six (Table 2, Fig. 3).
Unfavorable condition (mRS 4 or 5)
At 3 years, 30 patients (median 57 years) had mRS 4 or 5, 25% of the 120 three-year survivors but only 11% of the 269 original H&H 4–5 aSAH patients (Table 2). As many as 24 (80%) patients had ICH (14 evacuated) while only 6 had no ICH at all, 7 (23%) had an IVH clot, and 15 (50%) had a shunt (Tables 2 and 3, Fig. 3). In multivariate testing of available data, the ICH volume stood out as a significant predictor of mRS at 3 years.
Discussion
Long-term survivors among “poor-grade” aSAH patients
After acute aSAH, “poor condition” or “poor grade” on admission to the first hospital for CT and after transfer to the neurointensive care predicts early mortality so high that aSAH patients brain dead within 14 days are a significant group of organ donors [21, 37, 45]. The acute effect may be so devastating that some aSAH patients do not reach alive the first hospital and diagnostic CT [30]. The long-term mortality, often presented at 12 months [47], is also high: 37% in H&H 4 and 73% in H&H 5 in our present study (Fig. 2). A minority of H&H 4–5 or WFNS 4–5 patients will become true long-term survivors, in the present study 120 (45%) patients alive at 3 years (Table 2), albeit being stroke-risk carriers and prone to further vascular events [16]. They have remained an unrecognized group among stroke survivors for obvious reasons: prospective databases with long recruitment and outpatient follow-up periods would be virtually impossible to maintain, while much of literature is focused on “aneurysm treatment” [43]. We have retrospectively analyzed their clinical condition during very long follow-ups: excess mortality (median 12 years) [17]; epilepsy (median 6 years) [18]; anti-depressants (median 9 years) [19]; anti-psychotics (median 9 years) [39]; and shunt dependency (median 8 years) [1].
Visualization of a complex dataset with clinical lifelines
In the present study, we constructed the clinical date point calendaric lifelines for the 269 consecutive aSAH patients in poor condition (54% with H&H 4 and 46% with H&H 5) on admission to the Neurointensive Care Unit of the Kuopio University Hospital (KUH) from the defined KUH catchment population from 2005 to 2015 (Fig. 1, Table 1). For the lifeline analysis, each patients’ medical data, using the Finnish personal identification code, was obtained from the nationwide registries, and fused into the Kuopio Intracranial Aneurysm Patient and Family Database. The cross-sectional study population consisted the 120 consecutive patients alive at 3 years, 73% with H&H 4 and 27% with H&H 5 on admission (Table 1, Table 2). We found it impossible, using sentences and risk factors only, to present a comprehensive view of the associations of multiple intertwining factors during the neurointensive care vs. the clinical conditions at 3 years. For the same reason, the patients who deteriorated from H&H 1–3 to H&H 4–5 during the neurointensive care were excluded. For visual estimation and browsing, one representative slice of each of the 120 primary CT scans (ICH; IVH; hydrocephalus) and the interventions (extraventricular drainage; sIA occlusion; ICH evacuation; decompressive craniectomy; shunt) were compiled together, according to the mRS (0 to 5) at 3 years (Fig. 3) (see also 45 asymptomatic meningiomas by Näslund et al. in 2020 in Acta Neurochirurgica [35]). Furthermore, the interventions and the presence or absence of ICH were cross-tabulated into 12 groups (Table 3).
Aneurysmal intracerebral hemorrhage and the brain connectome
Of the 120 H&H 4 or 5 patients alive at 3 years, 71 (59%) had an intracerebral hematoma (ICH) with a median volume of 22 cm3, mostly (47; 66%) from a middle cerebral artery (MCA) sIA, and (12; 17%) from an anterior communicating artery (AComA) sIA. Aneurysmal ICH, depending on its volume and location, is the single most damaging and deadly complication of acute aSAH [51]. In the present study, ICH volume was a significant predictor of mRS at 3 years (Table 2). Arterial blood yet from the ruptured aneurysm wall tears and enters the adjacent brain tissue, forming a permanent brain cavity, filled with ICH clot. MRI (diffusion/perfusion/tractography) may give further prognostic information on brain edema [42], early brain ischemia [11, 14], and injury to white matter tracts and nuclei, e.g., in relation to motor function [4] or impaired consciousness [20]. The local injury of the brain connectome is permanent; i.e., no axonal connections will redevelop across the ICH cavity that later in neuroimaging appears as a CSF cavity. Convalescence, e.g., from acute hemiparesis or aphasic disorder, depends on the functional re-organization of the connectome around the cavity.
Acute evacuation of aneurysmal ICH
In our series, 71 (59%) of the 120 patients presented with ICH, and 29 ICHs (median volume 56 cm3) were microsurgically evacuated. Acute evacuation relieves expansion and pressure against the cavity wall, but the extent of brain tear remains unchanged. Only gentle subtotal evacuation is realistic as aneurysmal ICHs are rooted locally among cortical and perforating arteries [49]. Nevertheless, the ICH clot inducing neuroinflammation becomes smaller [53]. However, there is not yet concrete evidence on long-term benefits of acute aneurysmal ICH evacuation [14]. Notably, in spontaneous ICHs and IVHs, there is increasing interest in acute endoscopic and stereotactic evacuation [3, 34].
Decompressive craniectomy and cranioplasty
In our series, DC was performed in 35 (13%) of the 269 aSAH patients with H&H 4 or 5 on admission: 16 of them (46%) were among the 120 three-year survivors. After this series, we abandoned the frozen bone flaps for artificial ones, because 19% of own bone flaps were removed due to complications in a Finnish study [24]. In a systematic review of 407 aSAH patients with WFNS 4 or 5, the effect of DC on functional outcome remained unknown because of the lack of robust control groups [2].
Aneurysmal intraventricular hemorrhage
Aneurysmal IVHs of various volumes are frequent in acute aSAH patients [38, 44]. In our basic series, IVHs ranged from mere blood sediments (38%) in the occipital horns to clots (34%), some casting one or both lateral ventricles. IVH clots may co-exist with adjacent ICH, in 64 (24%) cases in our series, adding instant brain tissue injury to multiple potentially harmful effects of IVH. With pathobiology poorly understood, they include neuroinflammation [10]; alteration in ependymal cells, ciliary beat, arachnoid villi, CSF production and resorption, and glymphatic circulation; and clinically manifest hydrocephalus (acute; subacute; latent). IVH with enlarged ventricles often require prolonged EVD with ICP and CSF outflow monitoring, with the risk of catheter occlusions and exchanges, and eventual bacterial meningitis [1]. In principle, it would make sense to reduce clots and casts in the lateral ventricles, even III and IV, with neuroendoscopy as soon as feasible. In spontaneous IVH, recent meta-analysis suggested that endoscopy improved the survival and prognosis with the lowest rate of shunts and infection [34].
Secondary hydrocephalus and shunt dependency
Secondary hydrocephalus is frequent among aSAH survivors, and several risk scores have been published [1]. The pathobiology is poorly understood but neuroinflammation is a candidate [10], activating in the acute phase and possibly exerting a long-term dysfunction in the CSF environment, with tendency of valves and catheters to occlude, as well as shunt infection. EVD is an instant indicator of shunt dependency, and concomitant bacterial meningitis or ventriculitis increases the risk [1]. Surprisingly, in our series and other cohorts, even normal ventricle size and sediments of IVH only could be followed by latent shunt dependency [1, 8, 52]. The long-term outcome and quality of life of shunted aSAH survivors is virtually unknown according to our literature review. Long-term follow-up studies will show whether shunt-dependent post-aSAH hydrocephalus is a degenerative brain disease, such as idiopathic normal pressure hydrocephalus (iNPH) after temporary improvement with a shunt [23].
Outcome algorithms for H&H 4 or 5 aSAH patients on admission
Our pilot study, aimed to analyze a significant group of long-term aSAH survivors, did not include enough data (e.g., previous conditions of patients; ICP monitoring; EVD outflow; acute or delayed ischemic brain injury; electrolyte disturbances; cardiopulmonary complications; CNS or systemic infections; and complications of management) for computerized (e.g., machine learning) prediction on admission or during neurointensive care for subsequent mortality and outcome. Still, some aspects concerning triage on admission can be extracted from the 120 H&H 4–5 survivors at 3 years, as visualized in Fig. 3 and Table 3.
Firstly, in 49 patients (41%) of 120 patients with H&H 4 or 5 on admission, the primary CT scan did not show immediate brain injury by ICH. Their mRS range at 3 years from 0 to 5 indicates that coma and extension to pain are unreliable triage predictors of dismal outcome [28], interfered, e.g., by seizures, hydrocephalus, brain herniation, sedation, intubation, and ventilator care. Acute MRI would add information on possible acute ischemic brain injury [14].
Secondly, large ICHs in eventual mRS 3 to 5 survivors (maximum 200 cm3 in our series) may question chances of survival at the initial triage for neurointensive care. Large ICHs raise the question whether lives saved with neurointensive care are “worth living” with mRS 4 to 5 in the long run, i.e., whether such logistics would be ethically acceptable in neuroacutology. With the opt-out system for brain death and organ donation in Finland since 2010, we also admit “hopeless” aSAH patients, provided that the National Transplantation Service does not by phone exclude the possibility of organ donation due to concomitant diseases or conditions [21, 45]. The donors of kidneys included brain dead aSAH patients even at their 80’s. In Finland, the annual costs of dialysis (about 40,000 euros) far exceed the costs after the first year of a kidney transplant patient [15]. Eventually, evaluating CT scans in Fig. 3, the lives saved with large ICH and dismal (“unacceptable”) long-term condition (mRS 4 or 5) were few: 23 (9%) of the 269 H&H 4 or 5 patients on admission (Fig. 3).
Thirdly, it was surprising, e.g., how the six mRS 0 or 1 patients (median 58 years) with ICH over 27 cm3 (the largest quartile) managed to recover so well at 3 years (Tables 1 and 2, Fig. 3). This unexpected resilience to brain injury requires further investigations.
Strengths and limitations of the study
The strengths derive from the free tax-paid Finnish health care system, as well as the automatic archival of clinical data, using the Finnish identity codes, in the national clinical registries. Finland is divided into exclusive catchment areas among the 5 university hospitals which allows the creation of disease cohorts that are minimally selected and biased. The Kuopio Intracranial Aneurysm Patient and Family Database reliably reflects aSAH in the Eastern Finnish population and allows reconstruction of the clinical date point lifelines of all diagnosed aSAH patients, also using data from the national clinical registries. Our study is retrospective although the database prospectively collected all aSAH patients through the study period.
There are also limitations. The evaluation of the clinical condition of the 120 three-year survivors was based on the available case reports and the data available in the national registries. In this pilot study, the patients who deteriorated from H&H 1–3 on admission to H&H 4–5 later were excluded from the final study population. Finally, the extensive digital neurointensive care monitoring data, available in the national database, was not used [41]; such data are indispensable in further studies with machine learning algorithms.
Suggested further research
Firstly, retrospective re-reconstruction of clinical lifelines for individual aSAH patients, or any neuroacutology patients, is arduous patchwork at present, for clinical research or quality control. The IT systems of the hospital catchment area should automatically provide individual digital timelines (in minute scale) from the first contact through all phases to, e.g., the neurointensive care. Such quality control practice would aid to detect and prevent delays and deviations in aSAH logistics that might risk the final outcome, including time of intubation or release of tentorial herniation.
Secondly, the long-term course of consequences of acute aSAH on the brain has not been adequately studied. Long-term understanding would require consecutive MRI scans (diffusion/perfusion and tractography), at acute phase (acute hemorrhagic injuries, severing of tracts, hydrocephalus, and ischemia), at subacute and late subacute phases, at 12 months (permanent hemorrhagic and ischemic injuries), and after a few years (neurodegeneration).
Thirdly, neuroinflammation might function as an umbrella for cellular and molecular biology research of several calamities of the aSAH brain: early brain ischemia; ICH; IVH; ischemic events during occlusive IA therapy; delayed ischemia; shunt-dependent hydrocephalus—also postulated neurodegeneration in the post-aSAH brain [5].
Clinical conclusions
-
1.
Poor clinical condition (H&H 4–5, WFNS 4–5) on admission does not reliably predict the long-term outcome, particularly so if the patient survives the early excess mortality (14 days and 12 months) caused by aSAH. The risk of ending in poor long-term condition is relatively small, e.g., 8.6% at mRS 4 and 2.6% at mRS 5 (hospice care) at 3 years in our series.
-
2.
We propose that virtually all acute aneurysmal subarachnoid hemorrhage (aSAH) patients in poor condition on admission are immediately transferred to neurointensive care. In the countries with national presumed consent (opt-out), aSAH patients with dismal prognosis can be admitted to neurointensive care as potential organ donors, also at high age.
-
3.
Better understanding of the consequences of acute aSAH on the brain would require consecutive MRI scans (diffusion/perfusion and tractography), at acute phase (acute hemorrhagic injuries, severing of tracts, hydrocephalus, and ischemia), at subacute and late subacute phases, at 12 months (permanent hemorrhagic and ischemic injuries), and after a few years (neurodegeneration).
Abbreviations
- ACA:
-
Anterior cerebral artery and peripheral segments
- AComA:
-
Anterior communicating artery
- aSAH:
-
Aneurysmal subarachnoid hemorrhage
- BA:
-
Basilar artery trunk and bifurcation
- CNS:
-
Central nervous system
- CT:
-
Computed tomography
- DC:
-
Decompressive craniectomy
- EVD:
-
Extraventricular drainage
- H&H:
-
Hunt and Hess scale
- ICA:
-
Internal carotid artery trunk and bifurcation
- ICH:
-
Intracerebral hematoma
- ICHE:
-
Intracerebral hematoma microsurgically evacuated
- ICP:
-
Intracranial pressure
- IQR:
-
25% and 75% range
- IVH:
-
Intraventricular hematoma (blood clot)
- KUH:
-
Kuopio University Hospital
- MCA:
-
Middle cerebral artery and peripheral segments
- mRS:
-
Modified Rankin Scale
- PICA:
-
Posterior inferior cerebellar artery
- Sh:
-
Shunt
- sIA:
-
Saccular intracranial aneurysm
- WFNS:
-
World Federation of Neurosurgical Societies
References
Adams H, Ban VS, Leinonen V, Aoun SG, Huttunen J, Saavalainen T, Lindgren A, Frosen J, Fraunberg M, Koivisto T, Hernesniemi J, Welch BG, Jaaskelainen JE, Huttunen TJ (2016) Risk of shunting after aneurysmal subarachnoid hemorrhage: a collaborative study and initiation of a consortium. Stroke 47:2488–2496
Alotaibi NM, Elkarim GA, Samuel N, Ayling OGS, Guha D, Fallah A, Aldakkan A, Jaja BNR, de Oliveira Manoel AL, Ibrahim GM, Macdonald RL (2017) Effects of decompressive craniectomy on functional outcomes and death in poor-grade aneurysmal subarachnoid hemorrhage: a systematic review and meta-analysis. J Neurosurg 127:1315–1325
Awad IA, Polster SP, Carrión-Penagos J, Thompson RE, Cao Y, Stadnik A, Money PL, Fam MD, Koskimäki J, Girard R, Lane K, McBee N, Ziai W, Hao Y, Dodd R, Carlson AP, Camarata PJ, Caron JL, Harrigan MR, Gregson BA, Mendelow AD, Zuccarello M, Hanley DF, Trial Investigators MISTIEIII (2019) Surgical performance determines functional outcome benefit in the minimally invasive surgery plus recombinant tissue plasminogen activator for intracerebral hemorrhage evacuation (MISTIE) procedure. Neurosurgery 84:1157–1168
Cho SH, Kim SH, Choi BY, Cho SH, Kang JH, Lee CH, Byun WM, Jang SH (2007) Motor outcome according to diffusion tensor tractography findings in the early stage of intracerebral hemorrhage. Neurosci Lett 421:142–146
Corraini P, Henderson VW, Ording AG, Pedersen L, Horváth-Puhó E, Sørensen HT (2017) Long-term risk of dementia among survivors of ischemic or hemorrhagic stroke. Stroke 48:180–186
Damani R, Mayer S, Dhar R, Martin RH, Nyquist P, Olson DM, Mejia-Mantilla JH, Muehlschlegel S, Jauch EC, Mocco J, Mutoh T, Suarez JI, Unruptured Intracranial Aneurysms and SAH CDE Project Investigators (2019) Common data element for unruptured intracranial aneurysm and subarachnoid hemorrhage: recommendations from assessments and clinical examination workgroup/subcommittee. Neurocrit Care 30:28–35
de Oliveira Manoel AL, Goffi A, Marotta TR, Schweizer TA, Abrahamson S, Macdonald RL (2016) The critical care management of poor-grade subarachnoid haemorrhage. Crit Care 20:1–19
Diesing D, Wolf S, Sommerfeld J, Sarrafzadeh A, Vajkoczy P, Dengler NF (2018) A novel score to predict shunt dependency after aneurysmal subarachnoid hemorrhage. J Neurosurg 128:1273–1279
Diringer MN, Bleck TP, Claude Hemphill J 3rd, Menon D, Shutter L, Vespa P, Bruder N, Conolly ES Jr, Citerio G, Gress D, Hänggi D, Hoh BL, Lanzino G, Le Roux P, Rabinstein A, Schmutzhard E, Stocchetti N, Suarez JI, Treggiari M, Tseng MY, Vergouwen MD, Wolf S, Zipfel G, Neurocritical Care Society (2011) Critical care management of patients following aneurysmal subarachnoid hemorrhage: recommendations from the Neurocritical Care Society’s Multidisciplinary Consensus Conference. Neurocrit Care 15:211–240
Fam MD, Zeineddine HA, Eliyas JK, Stadnik A, Jesselson M, McBee N, Lane K, Cao Y, Wu M, Zhang L, Thompson RE, John S, Ziai W, Hanley DF, Awad IA, Trial Investigators CLEARIII (2017) CSF inflammatory response after intraventricular hemorrhage. Neurology 89:1553–1560
Fragata I, Alves M, Papoila AL, Nunes AP, Ferreira P, Canto-Moreira N, Canhão P (2017) Early prediction of delayed ischemia and functional outcome in acute subarachnoid hemorrhage: role of diffusion tensor imaging. Stroke 48:2091–2097
Frontera JA, Ahmed W, Zach V, Jovine M, Tanenbaum L, Sehba F, Patel A, Bederson JB, Gordon E (2015) Acute ischaemia after subarachnoid haemorrhage, relationship with early brain injury and impact on outcome: a prospective quantitative MRI study. J Neurol Neurosurg Psychiatry 86:71–78
García S, Torné R, Hoyos JA, Rodríguez-Hernández A, Amaro S, Llull L, López-Rueda A, Enseñat J (2018) Quantitative versus qualitative blood amount assessment as a predictor for shunt-dependent hydrocephalus following aneurysmal subarachnoid hemorrhage. J Neurosurg 131:1743–1750
Gonçalves B, Turon R, Mendes A, Melo N, Lacerda P, Brasil P, Bozza FA, Kurtz P, Righy C (2018) Effect of early brain infarction after subarachnoid hemorrhage: a systematic review and meta-analysis. World Neurosurg 115:292–298
Helanterä I, Isola T, Lehtonen TK, Åberg F, Lempinen M, Isoniemi H (2019) Association of clinical factors with the costs of kidney transplantation in the current era. Ann Transplant 24:393–400
Huhtakangas J, Lehto H, Seppä K, Kivisaari R, Niemelä M, Hernesniemi J, Lehecka M (2015) Long-term excess mortality after aneurysmal subarachnoid hemorrhage: patients with multiple aneurysms at risk. Stroke 46:1813–1818
Huttunen J, Kurki MI, von Und Zu Fraunberg M, Koivisto T, Ronkainen A, Rinne J, Jääskeläinen JE, Kälviäinen R, Immonen A (2015) Epilepsy after aneurysmal subarachnoid hemorrhage: a population-based, long-term follow-up study. Neurology 84:2229–2237
Huttunen J, Lindgren A, Kurki MI, Huttunen T, Frösen J, von Und Zu Fraunberg M, Koivisto T, Kälviäinen R, Räikkönen K, Viinamäki H, Jääskeläinen JE, Immonen A (2016) Antidepressant Use after aneurysmal subarachnoid hemorrhage: a population-based case-control study. Stroke 47:2242–2248
Huttunen T, von und zu Fraunberg M, Koivisto T, Ronkainen A, Rinne J, Sankila R, Seppä K, Jääskeläinen JE (2011) Long-term excess mortality of 244 familial and 1502 sporadic one-year survivors of aneurysmal subarachnoid hemorrhage compared with a matched Eastern Finnish catchment population. Neurosurgery 68:20–27
Jang SH, Chang CH, Jung YJ, Kim JH, Kwon YH (2019) Relationship between impaired consciousness and injury of ascending reticular activating system in patients with intracerebral hemorrhage. Stroke 50:2234–2237
Kämäräinen OP, Huttunen J, Lindgren A, Lång M, Bendel S, Uusaro A, Parviainen I, Koivisto T, Isoniemi H, Jääskeläinen JE (2018) Identification of potential organ donors after aneurysmal subarachnoid hemorrhage in a population-based neurointensive care in Eastern Finland. Acta Neurochir 160:1507–1514
Karamanakos PN, von Und Zu Fraunberg M, Bendel S, Huttunen T, Kurki M, Hernesniemi J, Ronkainen A, Rinne J, Jaaskelainen JE, Koivisto T (2012) Risk factors for three phases of 12-month mortality in 1657 patients from a defined population after acute aneurysmal subarachnoid hemorrhage. World Neurosurg 78:631–639
Koivisto AM, Alafuzoff I, Savolainen S, Sutela A, Rummukainen J, Kurki M, Jääskeläinen JE, Soininen H, Rinne J, Leinonen V, Kuopio NPH Registry (2013) Poor cognitive outcome in shunt-responsive idiopathic normal pressure hydrocephalus. Neurosurgery 72:1–8
Korhonen TK, Tetri S, Huttunen J, Lindgren A, Piitulainen JM, Serlo W, Vallittu PK, Posti JP, Finnish National Cranial Implant Registry (FiNCIR) study group (2019) Predictors of primary autograft cranioplasty survival and resorption after craniectomy. J Neurosurg 130:1672–1679
Kotikoski S, Huttunen J, Huttunen TJ, Helin K, Frösen J, Koivisto T, Kurki MI, von Und Zu Fraunberg M, Kunnamo I, Jääskeläinen JE, Lindgren AE (2018) Secondary hypertension in patients with saccular intracranial aneurysm disease: a population based study. PLoS One 13:1–10
Krishnamurthi RV, Barker-Collo S, Parag V, Parmar P, Witt E, Jones A, Mahon S, Anderson SC, Barber PA, Feign VL (2018) Stroke incidence by major pathological type and ischemic subtypes in the Auckland regional community stroke studies: changes between 2002 and 2011. Stroke 49:3–10
Lång M, Raj R, Skrifvars MB, Koivisto T, Lehto H, Kivisaari R, von Und Zu Fraunberg M, Reinikainen M, Bendel S (2016) Early moderate hyperoxemia does not predict outcome after aneurysmal subarachnoid hemorrhage. Neurosurgery 78:540–545
Lashkarivand A, Sorteberg W, Rosseland LA, Sorteberg A (2020) Survival and outcome in patients with aneurysmal subarachnoid hemorrhage in Glasgow coma score 3–5. Acta Neurochir 162:533–544
Levine JM (2009) Critical care management of subarachnoid hemorrhage. Curr Treat Options Neurol 11:126–136
Lindbohm JV, Kaprio J, Jousilahti P, Salomaa V, Korja M (2017) Risk factors of sudden death from subarachnoid hemorrhage. Stroke 48:2399–2404
Lindgren AE, Kurki MI, Riihinen A, Koivisto T, Ronkainen A, Rinne J, Hernesniemi J, Eriksson JG, Jääskeläinen JE, von und zu Fraunberg M (2013) Type 2 diabetes and risk of rupture of saccular intracranial aneurysm in eastern Finland. Diabetes Care 36:2020–2026
Lindgren AE, Kurki MI, Riihinen A, Koivisto T, Ronkainen A, Rinne J, Hernesniemi J, Eriksson JG, Jääskeläinen JE, von und zu Fraunberg M (2014) Hypertension predisposes to the formation of saccular intracranial aneurysms in 467 unruptured and 1053 ruptured patients in Eastern Finland. Ann Med 46:169–176
Lindlöf M, Lindgren A, Paavola J, Väntti N, von Und Zu Fraunberg M, Koivisto T, Jääskeläinen JE, Kämäräinen OP, Huttunen J (2019) Analgesic use after aneurysmal subarachnoid hemorrhage: a population-based case-control study of 1187 patients. World Neurosurg 126:1276–1286
Mei L, Fengqun M, Qian H, Dongpo S, Zhenzhong G, Tong C (2020) Exploration of efficacy and safety of interventions for intraventricular hemorrhage: a network meta-analysis. World Neurosurg 136:382–389
Näslund O, Skoglund T, Farahmand D, Bontell TO, Jakola AS (2020) Indications and outcome in surgically treated asymptomatic meningiomas: a single-center case-control study. Acta Neurochir 162:2155–2163
Nurmonen HJ, Huttunen T, Huttunen J, Kurki MI, Helin K, Koivisto T, von Und Zu Fraunberg M, Jääskeläinen JE, Lindgren AE (2017) Polycystic kidney disease among 4,436 intracranial aneurysm patients from a defined population. Neurology 89:1852–1859
Ojha M, Finnis ME, Heckelmann M, Raith EP, Moodie S, Chapman MJ, Reddi B, Maiden MJ (2020) Outcomes following grade V subarachnoid haemorrhage: a single-centre retrospective study. Anaesth Intensive Care 48:289–296
Oppong MD, Gembruch O, Herten A, Frantsev R, Chihi M, Dammann P, El Hindy N, Forsting M, Sure U, Jabbarli R (2018) Intraventricular hemorrhage caused by subarachnoid hemorrhage: does the severity matter? World Neurosurg 111:693–702
Paavola JT, Väntti N, Junkkari A, Huttunen TJ, von Und Zu Fraunberg M, Koivisto T, Kämäräinen OP, Lång M, Meretoja A, Räikkönen K, Viinamäki H, Jääskeläinen JE, Huttunen J, Lindgren AE (2019) Antipsychotic use among 1144 patients after aneurysmal subarachnoid hemorrhage. Stroke 50:1711–1718
Panni P, Colombo E, Donofrio CA, Barzaghi LR, Albano L, Righi C, Scomazzoni F, Simionato F, Mortini P, Falini A, Anzalone N (2019) Hemorrhagic burden in poor-grade aneurysmal subarachnoid hemorrhage: a volumetric analysis of different bleeding distributions. Acta Neurochir 161:791–797
Raj R, Bendel S, Reinikainen M, Hoppu S, Laitio R, Ala-Kokko T, Curtze S, Skrifvars MB (2018) Costs, outcome and cost-effectiveness of neurocritical care: a multi-center observational study. Crit Care 22:1–10
Rass V, Ianosi BA, Wegmann A, Gaasch M, Schiefecker AJ, Kofler M, Lindner A, Addis A, Almashad SS, Rhomberg P, Pfausler B, Beer R, Gizewski ER, Thomé C, Helbok R (2019) Delayed resolution of cerebral edema is associated with poor outcome after nontraumatic subarachnoid hemorrhage. Stroke 50:828–836
Rinkel GJ, Algra A (2011) Long-term outcomes of patients with aneurysmal subarachnoid haemorrhage. Lancet Neurol 10:349–356
Rosen DS, Macdonald RL, Huo D, Goldenberg FD, Novakovic RL, Frank JI, Rosengart AJ (2007) Intraventricular hemorrhage from ruptured aneurysm: clinical characteristics, complications, and outcomes in a large, prospective, multicenter study population. J Neurosurg 107:261–265
Scholz N (2020) Organ donation and transplantation facts, figures and European Union action. EPRS European Parliamentary Research Service. https://www.europarl.europa.eu/thinktank/en/document.html?reference=EPRS_BRI(2020)649363. Accessed 6 Jan 2021
Steiner T, Juvela S, Unterberg A, Jung C, Forsting M, Rinkel G, European Stroke Organization (2013) European Stroke Organization guidelines for the management of intracranial aneurysms and subarachnoid haemorrhage. Cerebrovasc Dis 35:93–112
Stienen MN, Visser-Meily JM, Schweizer TA, Hänggi D, Macdonald RL, Vergouwen MDI, Unruptured Intracranial Aneurysms and SAH CDE Project Investigators (2019) Prioritization and timing of outcomes and endpoints after aneurysmal subarachnoid hemorrhage in clinical trials and observational studies: proposal of a multidisciplinary research group. Neurocrit Care 30:102–113
Suwatcharangkoon S, Meyers E, Falo C, Schmidt JM, Agarwal S, Claassen J, Mayer SA (2016) Loss of consciousness at onset of subarachnoid hemorrhage as an important marker of early brain injury. JAMA Neurol 73:28–35
Talamonti G, Nichelatti M, Al Mashni AA, D’Aliberti G (2016) Life-threatening cerebral hematoma owing to aneurysm rupture. World Neurosurg 85:215–227
van Gijn J, Kerr RS, Rinkel GJ (2007) Subarachnoid haemorrhage. Lancet 369:306–318
Wan A, Jaja BN, Schweizer TA, Macdonald RL, on behalf of the SAHIT collaboration (2016) Clinical characteristics and outcome of aneurysmal subarachnoid hemorrhage with intracerebral hematoma. J Neurosurg 125:1344–1351
Wilson CD, Safavi-Abbasi S, Sun H, Kalani MY, Zhao YD, Levitt MR, Hanel RA, Sauvageau E, Mapstone TB, Albuquerque FC, McDougall CG, Nakaji P, Spetzler RF (2017) Meta-analysis and systematic review of risk factors for shunt dependency after aneurysmal subarachnoid haemorrhage. J Neurosurg 126:586–595
Zhou Y, Wang Y, Wang J, Stetler RA, Yang QW (2014) Inflammation in intracerebral hemorrhage: from mechanisms to clinical translation. Prog Neurobiol 115:25–44
Acknowledgements
The authors thank Research Nurse Katariina Helin for the data acquisition in the Kuopio Intracranial Aneurysm Patient and Family Database.
Data and code availability
All data and coding are available for corresponding author.
Funding
Open access funding provided by University of Eastern Finland (UEF) including Kuopio University Hospital. This study is funded by the Maire Taponen Foundation, Pro Humanitate Foundation, the Päivikki and Sakari Sohlberg Foundation, the Kuopio University Hospital, and the Academy of Finland. The sponsors had no role in the design or conduct of this research.
Author information
Authors and Affiliations
Contributions
Anniina H. Autio, BM: Design and conceptualized study; acquisition of data; analyzed the data; drafted the manuscript for intellectual content, Kuopio University Hospital
Juho Paavola, BM: Acquisition of data; revised the manuscript for intellectual content, Kuopio University Hospital
Joona Tervonen, MD: Acquisition of data; revised the manuscript for intellectual content, Kuopio University Hospital
Maarit Lång, MD: Revised the manuscript for intellectual content, Kuopio University Hospital
Terhi J. Huuskonen, MD, PhD: Revised the manuscript for intellectual content, Kuopio University Hospital
Jukka Huttunen, MD, PhD: Revised the manuscript for intellectual content, Kuopio University Hospital
Virve Kärkkäinen, PhD: Revised the manuscript for intellectual content, Kuopio University Hospital
Mikael von und zu Fraunberg, MD, PhD: Revised the manuscript for intellectual content, Kuopio University Hospital
Antti E. Lindgren, MD, PhD: Revised the manuscript for intellectual content, Kuopio University Hospital
Timo Koivisto MD, PhD: Revised the manuscript for intellectual content, Kuopio University Hospital
Juha E. Jääskeläinen, MD, PhD: Design and conceptualized study; acquisition of data; revised the manuscript for intellectual content; study supervision, Kuopio University Hospital
Olli-Pekka Kämäräinen, MD, PhD: Design and conceptualized study; acquisition of data; revised the manuscript for intellectual content; study supervision, Kuopio University Hospital
Corresponding author
Ethics declarations
Compliance with ethical standards
The Kuopio University Hospital Research Ethics Committee approved the study and the study was performed in accordance with the ethical standards as laid down in the 1964 Declaration of Helsinki and its later amendments. KUH Neurosurgery IA Study Group received a written informed consent from all patients in the database. The Ministry of Social Affairs and Health of Finland approved the data fusion from the national registries. The patients of the study cohort were not contacted during the study. A notable part of our data is acquired from Finnish national health registries, which are regulated by the Finnish Institute of Health and Welfare. Due to this, we are not allowed to share our datasets openly in any form, because of privacy requirements of Finnish law. Interested researchers can be granted permissions to our datasets, but this requires individual evaluation of the intended use of the data and the final permission will be granted by Institute of Health and Welfare.
Ethical approval
This study was performed in line with the principles of the Declaration of Helsinki. Approval was granted by the Ethics Committee of Kuopio University Hospital.
Consent to participate
Written informed consent was obtained from all individual participants included in the study.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Vascular Neurosurgery - Aneurysm
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Autio, A.H., Paavola, J., Tervonen, J. et al. Clinical condition of 120 patients alive at 3 years after poor-grade aneurysmal subarachnoid hemorrhage. Acta Neurochir 163, 1153–1166 (2021). https://doi.org/10.1007/s00701-021-04725-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00701-021-04725-2