Abstract
Background
Hydrocephalus is one of the major complications of aneurysmal subarachnoid haemorrhage (aSAH). In the acute setting, an external ventricular drain (EVD) is used for early management. A cisternal drain (CD) coupled with the micro-surgical opening of basal cisterns can be an alternative when the aneurysm is clipped. Chronic hydrocephalus after aSAH is managed with ventriculo-peritoneal (VP) shunt, a procedure associated with a wide range of complications. The aim of this study is to analyse the impact of micro-surgical opening of basal cisterns coupled with CD on the incidence of VP shunt, compared to patients treated with EVD.
Methods
The authors conducted a retrospective review of 89 consecutive cases of patients with aSAH treated surgically and endovascularly with either EVD or CD between January 2009 and September 2021. Patients were stratified into two groups: Group 1 included patients with EVD, Group 2 included patients with CD. Subgroup analysis with only patients treated surgically was also performed. We compared their baseline characteristics, clinical outcomes and shunting rates.
Results
There were no statistically significant differences between the two groups in terms of epidemiological characteristics, WFNS score, Fisher scale, presence of intraventricular hemorrhage (IVH), acute hydrocephalus, postoperative meningitis or of clinical outcomes at last follow-up. Cisternostomy with CD (Group 2) was associated with a statistically significant reduction in VP-shunt compared with the use of an EVD (Group 1) (9.09% vs 53.78%; p < 0.001). This finding was confirmed in our subgroup analysis, as among patients with a surgical clipping, the rate of VP shunt was 43.7% for the EVD group and 9.5% for the CD group (p = 0.02).
Conclusions
Cisternostomy with CD may reduce the rate of shunt-dependent hydrocephalus. Cisternostomy allows the removal of subarachnoid blood, thereby reducing arachnoid inflammation and fibrosis. CD may enhance this effect, thus resulting in lower rates of chronic hydrocephalus.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Aneurysmal subarachnoid haemorrhage (aSAH) has an incidence of 6.1–9 per 100,000 persons/year [10, 13] and is still associated with high morbidity and mortality. Fatality rate varies from 27 to 44% and it leaves up to 20% of patients dependent [27]. Hydrocephalus is one of the major complications of aSAH, together with vasospasm, delayed cerebral ischemia, epilepsy and cognitive impairment [8].
Acute hydrocephalus, defined as occurring up to 72 h post SAH [35], is generally managed with placement of an external ventricular drain (EVD) [8]. Positioning of a cisternal drain (CD), coupled with micro-surgical opening of basal cisterns, is a potential alternative treatment when surgical treatment of the aneurysm is performed [28, 32]. Chronic or delayed hydrocephalus, defined as occurring after 14 days from SAH [35], is seen in up to 30% of patients [1, 9, 12, 15] and is managed with ventriculoperitoneal (VP) shunt insertion [8]. Readmission rate after aSAH is 11.4% and hydrocephalus accounts for about one-third of it [19]. Moreover, VP shunting is associated with up to 33.4% complication rate, with a high rate of infections and need for surgical revisions (22–51.9%) [24, 29, 31].
Several studies investigated the potential risk factors for developing chronic hydrocephalus after aSAH [23, 25, 37, 39]. Surgical fenestration of the lamina terminalis (LT) and membrane of Liliequist (MoL) during open microsurgical clipping was associated with a reduced risk of chronic hydrocephalus [2, 3, 11, 20, 22, 33, 34, 38, 40]. This open ventriculostomy enhances CSF circulation, thus reducing arachnoid fibrosis and vascular inflammation leading to decreased risk of developing chronic hydrocephalus [23].
The aim of the study is to report our experience with cisternostomy and placement of a cisternal drain at the time of surgical clipping of ruptured aneurysms, and study its impact on the prevalence of VP shunts for chronic hydrocephalus, when compared to a similar population of patients treated with EVD.
Methods
Patient population
We performed a retrospective analysis of our consecutive surgical series including all patients with aSAH admitted at the Neurosurgical Department of the University Hospital of Lausanne between January 2009 and September 2021. Inclusion criteria were: adults (age > or 18) presenting with aSAH (all localisations, Fisher and WFNS were included) treated either surgically or endovascularly and with concomitant placement of an EVD or CD for acute hydrocephalus, with at least 3 months follow-up after the haemorrhage. We excluded from the analysis patients that did not need a drain, those who did not survive the initial hospitalization as well as those not having a 3-month follow-up (Fig. 1).
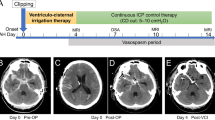
The criteria for drain placement were radiological evidence of ventricular dilation in patients with clinical symptoms of hydrocephalus. We used an EVD for these patients until 2016, after which we started the systematic use of CD after cisternostomy for surgically treated-patients, after observing the benefit of this procedure for traumatic brain injury [5,6,7, 16,17,18] and aneurysmal subarachnoid haemorrhage [28, 32].
Shunt dependency was defined as unsuccessful weaning from EVD/CD: first the drain was progressively raised, then clamped for 24 h and a CT was performed to control the ventricular size. If the patient presented a neurological degradation during these 24 h and/or the CT showed a significant increase in ventricular size, the weaning process was considered unsuccessful. The process was then repeated within a few days of interval to consider if a patient was shunt-dependent.
Postoperative meningitis was defined as a positive CSF gram stain/culture [26].
Cisternal drain technique
The surgical technique for the opening of basal cisterns or cisternostomy has been previously described for aSAH [28, 32] and traumatic brain injuries [18]. The head is fixed in a Mayfield clamp and rotated 30° to the contralateral side and extended. A standard pterional craniotomy with drilling of the sphenoid ridge and flattening of the orbital roof is mandatory. Frontotemporal durotomy is performed in a curvilinear fashion close to the basal dura. A lateral subfrontal approach allows early identification of the olfactory nerve and then medially of the optic nerve. This allows the opening of the optico-carotid cistern and of the membrane of Liliequist. The posterior circulation can be visualized through this window. More medially, the interoptic space anteriorly and the lamina terminalis posteriorly can be opened to obtain a direct communication with the third ventricle. A standard ventricular drain is then usually placed in the optico-carotid cistern, secured to the dura with a simple loose stich to assure smooth removal, and tunneled through the scalp as an EVD. Figure 2 illustrates the relevant anatomy of this procedure.
A Illustration depicting microsurgical technique for cisternostomy with the opening of the LT and MoL to promote circulation of CSF following aSAH. A cisternal drain is positioned in the optico-carotid cistern and is left for 7–10 days to drain blood products and debris and avoid arachnoid fibrosis. B Cadaveric specimen, right side. Head turned to the left and fixed with a clamp. Standard right pterional craniotomy with lateral subfrontal approach. Cisternostomy has been performed with opening of the optico-carotid cistern, Membrane of Lillequist (MoL), interoptic space and lamina terminalis (LT), not visible. A ventricular catheter is placed at the level of the opticocarotid cistern. I = olfactory nerve; II = optic nerve; III = oculomotor nerve; ICA = internal carotid artery; M1 = first segment of middle cerebral artery; A1 = first segment anterior cerebral artery, ACOM = anterior communicating artery, PCOM = posterior communicating artery; AchorA = anterior choroidal artery; LT = Lamina Terminalis; MoL = Membrane of Liliequist
Data collection and analysis
Epidemiological data, clinical and radiological characteristics were collected through retrospective review of the electronic medical records.
The modified Fisher scale was used [14] and it was assigned through analysis of the pre-operative non-contrast CT or CT angiogram. Fisher score I–II were defined as low scores and III–IV as high scores. The localization and size of the aneurysm were defined in preoperative angio-CT or angio-MR. Surgical data were collected such as the modality of treatment (endovascular treatment (ET) or surgical treatment (ST)) and the EVD or CD placement. Post-procedural complications were also collected.
Interval of follow-up was defined as time from the aSAH to the last documented neurosurgical or neuroradiological consult. Clinical outcomes were evaluated using the Glasgow Outcome Scale (GOS) and the modified Rankin Scale (mRS) at hospital discharge, at 3 months and at last follow-up. During the same timeline, a regular radiological evaluation was performed to detect chronic hydrocephalus or other delayed surgical complications.
Patients were divided into two groups: Group 1 included patients with an EVD, while Group 2 included patients with CD. To limit confounding factors, we further divided the patients undergoing a surgical treatment into Group 1.1, including patients with EVD, and Group 2.1, including patients with CD.
Statistical analysis
Statistical analysis of the data was performed with Stata/IC 16.1 software (StataCorp, Texas, USA). Continuous variables are presented as mean ± standard deviation (SD), and categorical variables as number and percentage. Univariate comparisons between Groups 1 and 2 were performed with a t-test study for the continuous variables. For categorical variables, Fisher’s exact test and Chi-square test were used. Point-biserial correlation was used to analyse the correlation between two variables. Significance was assessed at p-value < 0.05.
Results
Between January 2009 and September 2021, 236 patients with aSAH were admitted at the Neurosurgical Department and 89 fulfilled our inclusion criteria and were included in our analysis (Fig. 1). Sixty-seven patients had an EVD (Group 1) and twenty-two had a CD (Group 2).
Epidemiological, clinical, and radiological data are summarized in Table 1. No statistically significant differences were found between the two groups with respect to age (p 0.22), sex (p 0.43), WFNS score (p 0.24), Fisher scale (p 0.83), presence of intraventricular hemorrhage (IVH) (p 0.37), or acute hydrocephalus (p 0.06). As the CD group (Group 2) included only patients where a surgical clipping was performed, coupled with cisternostomy, significant differences were found between the two groups for the modality of aneurysm treatment and for aneurysm location (p 0.03). One exception was made for a giant carotid-ophthalmic aneurysm that was treated with embolization and subsequently had surgery for hematoma evacuation along with cisternostomy and cisternal drainage.
Overall shunting rate was 42.7% (38/89 patients). Cisternostomy and CD positioning were associated with a statistically significant risk reduction of shunt-dependent hydrocephalus rate, as VP-shunt rate was of 9.09% in CD group versus 53.78% in the EVD group (p < 0.001) (Table 2, Fig. 3). Mean timing from aneurysm rupture to VP-shunt is 60 days (Table 2). No significant differences were found between the two groups concerning the timing to surgery. The absolute risk reduction of shunt dependency with CD was 44.7%, with a number needed to treat of 2.3 patients. No specific complications related to the opening of the skull base cisterns and cisternal drain positioning were reported. In the EVD group instead, a drain revision was necessary in 7 cases (10.4%) secondary to a permanent drain occlusion.
No statistically significant differences were found in the shunting rate of hydrocephalus comparing patients treated surgically versus endovascularly. Nevertheless, in order to reduce the possible confounding factor of surgical opening of basal cisterns itself, we compared only patients who underwent surgical treatment and had either and EVD or a CD. The EVD group (n = 16) presented a rate of VP-shunting of 43.7% (7/16), while CD group (n = 21) had a rate of 9.5% (2/21), and this difference was statistically significant (p = 0.02) (Table 3, Fig. 3).
The presence of postoperative meningitis was reported in 13.4% of cases (9/67) of group 1 (EVD), and in 4.5% (1/22) in group 2 (CD) (p = 0.44). Five out of the 9 patients with an EVD and meningitis had a VP-shunt. Nevertheless, we could not find a statistically significant correlation between meningitis and need for VP-shunt, probably because of the small sample size considered.
The mean length of drainage for the EVD group was 19.4 days and 15.2 days for the CD group: this difference was not statistically significant. Nevertheless, longer drainage was correlated with the need for VP-shunting in the EVD group (p = 0.001).
No significant differences were found for the clinical outcomes calculated with GOS and mRS at last follow-up between the EVD and the CD group (median follow-up 24 months; range between 6 and 260 months), as shown in Fig. 4 and Table 1. No difference was found between the two cohorts if we divide patients into good outcome (GOS 5 or mRS 0–2) versus bad outcome (GOS 1–4 or mRS 3–6). The two cohorts showed similar functional outcomes for GOS (p = 1) and mRS (p = 0.44).
Discussion
The pathophysiology of hydrocephalus after aSAH is complex and yet to be fully understood. Two main mechanisms were discussed: first, the impaired absorption of CSF from fibrosed arachnoid granulations and second, an anatomic obstruction of ventricles and cisterns by blood products [4, 15]. Yasargil et al. specifically described thickening and inflammation of the MoL after aSAH blocking CSF flow at the level of the interpeduncular and prepontine area, causing hydrocephalus [41]. Fibrosis and partial obstruction of the fourth ventricle outflow have also been evoked [15].
Given this knowledge, it is logical to hypothesize that the microsurgical opening of the basal cisterns would help in restoring CSF flow and preventing hydrocephalus. Therefore, multiple papers have studied the relationship between the intra-operative opening of the LT and MoL and the development of shunt-dependent hydrocephalus [2, 3, 11, 20,21,22, 33, 34, 38, 40]. First, the fenestration of the LT alone was introduced and investigated by several groups with heterogeneous results. Komotar et al. [23] published a systematic review analysing the efficacy of LT fenestration in reducing shunt-dependent hydrocephalus, but they failed to reveal a significant reduction in shunting. More recently, Winkler et al. [38] studied the systematic tandem fenestration of LT and MoL during microsurgical aneurysm repair and achieved a significant reduction in shunt-dependent hydrocephalus. They attributed this finding to the fact that the isolated opening of the LT fails to allow an appropriate communication between the supra and infra-tentorial compartments and that the opening of MoL is mandatory to reduce shunting rates.
Since 2016, we routinely position a cisternal drain after cisternostomy for all cases of ruptured aneurysms with acute hydrocephalus and treated surgically, and we obtained significant reduction in VP shunting for chronic hydrocephalus. In our study, the positive effects of cisternostomy, thus the connection between the supra and infra-tentorial cisterns as well as the ventricular system, are enhanced by leaving a cisternal drain. The drain allows extensive evacuation of blood clots and debris from both cisternal and ventricular compartments, with a double beneficial implication: the reduction of the “CSF shift edema” and brain swelling and a decreased risk of developing chronic hydrocephalus [7, 23]. In our subgroup analysis comparing only patients with microsurgical clipping, we also found a significant difference between the EVD and CD groups, hence supporting the hypothesis that the cisternal drain enhances the effect of the opening of the basal cisterns more than the performance of cisternal fenestration with no drain.
The two groups were homogenous with respect to epidemiological data, clinical and radiological presentation (Table 1). No statistically significant differences were found for age, Fisher scale, presence of intraventricular haemorrhage or acute hydrocephalus, all known to be major predicting factors for developing chronic hydrocephalus [30, 36, 39]. Our analysis reports a shunt dependency rate of 42.7%, higher than described in literature [1, 9, 15]. This is mainly due to the patient population selected, all patients being with acute hydrocephalus at presentation and with a high rate of IVH, all needing CSF diversion in the acute period.
Even if the VP shunt rate was significantly reduced in the CD group when compared to the EVD group, the timing between aSAH and VP shunt for chronic hydrocephalus was similar between the two groups, so the CD does not seem to impact the timing of shunting. Furthermore, the functional outcome evaluated as GOS and mRS at last follow-up was not different between the two groups, probably secondary to the fact that an appropriately treated hydrocephalus has no major impact on the long-term functional outcome, as the final prognosis is multifactorial.
Despite these promising findings, the study has its limitations, mainly related to its retrospective observational design and limited number of patients included in the analysis and some contributing factors may be underestimated. The surgeries in this series were performed by the senior vascular surgeon in our department with extensive vascular and skull base experience and this might account for the absence of complications reported.
Further clinical studies with a prospective design and a larger cohort of patients are needed to validate these findings.
Conclusion
Cisternostomy coupled with cisternal drainage at the time of microsurgical clipping of ruptured aneurysm is associated with a reduced rate of VP-shunting for chronic hydrocephalus, when compared to a cohort treated with an EVD. No increase in complications was observed in relation to cisternostomy or drainage. These results may be related to the removal of subarachnoid blood, thereby reducing arachnoid inflammation and fibrosis, and the cisternal drainage may enhance this effect.
Abbreviations
- EVD:
-
External ventricular drain
- CD:
-
Cisternal drain
- VP-shunt:
-
Ventriculo-peritoneal shunt
- LT:
-
Lamina terminalis
- MoL:
-
Membrane of Liliequist
- ST:
-
Surgical treatment
- ET:
-
Endovascular treatment
- GOS:
-
Glasgow Outcome Scale
- mRS:
-
Modified Rankin Scale
References
Adams H, Ban VS, Leinonen V et al (2016) Risk of shunting after aneurysmal subarachnoid hemorrhage: a collaborative study and initiation of a consortium. Stroke 47(10):2488–2496. https://doi.org/10.1161/STROKEAHA.116.013739
Akyuz M, Tuncer R (2006) The effects of fenestration of the interpeduncular cistern membrane arousted to the opening of lamina terminalis in patients with ruptured ACoA aneurysms: a prospective, comparative study. Acta Neurochir (Wien). 148(7):725–3. https://doi.org/10.1007/s00701-006-0738-0. (discussion 731-2)
Andaluz N, Zuccarello M (2004) Fenestration of the lamina terminalis as a valuable adjunct in aneurysm surgery. Neurosurgery 55(5):1050–1059. https://doi.org/10.1227/01.neu.0000140837.63105.78
Black PM, Tzouras A, Foley L (1985) Cerebrospinal fluid dynamics and hydrocephalus after experimental subarachnoid hemorrhage. Neurosurgery 17(1):57–62. https://doi.org/10.1227/00006123-198507000-00009
Cherian I, Bernardo A, Grasso G (2016) Cisternostomy for traumatic brain injury: pathophysiologic mechanisms and surgical technical notes. World Neurosurg 89:51–57. https://doi.org/10.1016/j.wneu.2016.01.072
Cherian I, Grasso G, Bernardo A, Munakomi S (2016) Anatomy and physiology of cisternostomy. Chin J Traumatol 19(1):7–10. https://doi.org/10.1016/j.cjtee.2016.01.003
Cherian I, Beltran M, Landi A, Alafaci C, Torregrossa F, Grasso G (2018) Introducing the concept of “CSF-shift edema” in traumatic brain injury. J Neurosci Res 96(4):744–752. https://doi.org/10.1002/jnr.24145
Connolly ES, Rabinstein AA, Carhuapoma JR et al (2012) Guidelines for the management of aneurysmal subarachnoid hemorrhage. Stroke 43(6):1711–1737. https://doi.org/10.1161/str.0b013e3182587839
de Oliveira JG, Beck J, Setzer M et al (2007) Risk of shunt-dependent hydrocephalus after occlusion of ruptured intracranial aneurysms by surgical clipping or endovascular coiling: a single-institution series and meta-analysis. Neurosurgery. 61(5):924–33. https://doi.org/10.1227/01.neu.0000303188.72425.24. (discussion 933-4)
de Rooij NK, Linn FH, van der Plas JA, Algra A, Rinkel GJ (2007) Incidence of subarachnoid haemorrhage: a systematic review with emphasis on region, age, gender and time trends. J Neurol Neurosurg Psychiatry 78(12):1365–1372. https://doi.org/10.1136/jnnp.2007.117655
Dehdashti AR, Rilliet B, Rufenacht DA, de Tribolet N (2004) Shunt-dependent hydrocephalus after rupture of intracranial aneurysms: a prospective study of the influence of treatment modality. J Neurosurg 101(3):402–407. https://doi.org/10.3171/jns.2004.101.3.0402
Dorai Z, Hynan LS, Kopitnik TA, Samson D (2003) Factors related to hydrocephalus after aneurysmal subarachnoid hemorrhage. Neurosurgery. 52(4):763–9. https://doi.org/10.1227/01.neu.0000053222.74852.2d. (discussion 769-71)
Etminan N, Chang HS, Hackenberg K et al (2019) Worldwide incidence of aneurysmal subarachnoid hemorrhage according to region, time period, blood pressure, and smoking prevalence in the population: a systematic review and meta-analysis. JAMA Neurol 76(5):588–597. https://doi.org/10.1001/jamaneurol.2019.0006
Frontera JA, Claassen J, Schmidt JM et al (2006) Prediction of symptomatic vasospasm after subarachnoid hemorrhage: the modified fisher scale. Neurosurgery. 59(1):21–7. https://doi.org/10.1227/01.NEU.0000218821.34014.1B. (discussion 21-7)
Germanwala AV, Huang J, Tamargo RJ (2010) Hydrocephalus after aneurysmal subarachnoid hemorrhage. Neurosurg Clin N Am 21(2):263–270. https://doi.org/10.1016/j.nec.2009.10.013
Giammattei L, Messerer M, Cherian I et al (2018) Current perspectives in the surgical treatment of severe traumatic brain injury. World Neurosurg 116:322–328. https://doi.org/10.1016/j.wneu.2018.05.176
Giammattei L, Messerer M, Oddo M, Borsotti F, Levivier M, Daniel RT (2018) Cisternostomy for refractory posttraumatic intracranial hypertension. World Neurosurg 109:460–463. https://doi.org/10.1016/j.wneu.2017.10.085
Giammattei L, Starnoni D, Maduri R et al (2020) Implementation of cisternostomy as adjuvant to decompressive craniectomy for the management of severe brain trauma. Acta Neurochir (Wien) 162(3):469–479. https://doi.org/10.1007/s00701-020-04222-y
Greenberg JK, Washington CW, Guniganti R, Dacey RG Jr, Derdeyn CP, Zipfel GJ (2016) Causes of 30-day readmission after aneurysmal subarachnoid hemorrhage. J Neurosurg 124(3):743–749. https://doi.org/10.3171/2015.2.JNS142771
Kim JM, Jeon JY, Kim JH et al (2006) Influence of lamina terminalis fenestration on the occurrence of the shunt-dependent hydrocephalus in anterior communicating artery aneurysmal subarachnoid hemorrhage. J Korean Med Sci 21(1):113–118. https://doi.org/10.3346/jkms.2006.21.1.113
Komotar RJ, Olivi A, Rigamonti D, Tamargo RJ (2002) Microsurgical fenestration of the lamina terminalis reduces the incidence of shunt-dependent hydrocephalus after aneurysmal subarachnoid hemorrhage. Neurosurgery. 51(6):1403–12 (discussion 1412-3)
Komotar RJ, Hahn DK, Kim GH et al (2008) The impact of microsurgical fenestration of the lamina terminalis on shunt-dependent hydrocephalus and vasospasm after aneurysmal subarachnoid hemorrhage. Neurosurgery. 62(1):123–32. https://doi.org/10.1227/01.NEU.0000311069.48862.C8. (discussion 132-4)
Komotar RJ, Hahn DK, Kim GH et al (2009) Efficacy of lamina terminalis fenestration in reducing shunt-dependent hydrocephalus following aneurysmal subarachnoid hemorrhage: a systematic review. Clinical article. J Neurosurg. 111(1):147–54. https://doi.org/10.3171/2009.1.JNS0821
Merkler AE, Ch’ang J, Parker WE, Murthy SB, Kamel H (2017) The rate of complications after ventriculoperitoneal shunt surgery. World Neurosurg 98:654–658. https://doi.org/10.1016/j.wneu.2016.10.136
Motiei-Langroudi R, Adeeb N, Foreman PM et al (2017) Predictors of shunt insertion in aneurysmal subarachnoid hemorrhage. World Neurosurg 98:421–426. https://doi.org/10.1016/j.wneu.2016.11.092
Munoz-Gomez S, Wirkowski E, Cunha BA (2015) Post craniotomy extra-ventricular drain (EVD) associated nosocomial meningitis: CSF diagnostic criteria. Heart Lung Mar-Apr 44(2):158–160. https://doi.org/10.1016/j.hrtlng.2015.01.003
Nieuwkamp DJ, Setz LE, Algra A, Linn FH, de Rooij NK, Rinkel GJ (2009) Changes in case fatality of aneurysmal subarachnoid haemorrhage over time, according to age, sex, and region: a meta-analysis. Lancet Neurol 8(7):635–642. https://doi.org/10.1016/S1474-4422(09)70126-7
Ogura K, Hara M, Tosaki F, Hirai N (1988) Effect of cisternal drainage after early operation for ruptured intracranial aneurysms. Surg Neurol 30(6):441–444. https://doi.org/10.1016/0090-3019(88)90028-6
O’Kelly CJ, Kulkarni AV, Austin PC, Urbach D, Wallace MC (2009) Shunt-dependent hydrocephalus after aneurysmal subarachnoid hemorrhage: incidence, predictors, and revision rates. Clinical article. J Neurosurg. 111(5):1029–35. https://doi.org/10.3171/2008.9.JNS08881
Paisan GM, Ding D, Starke RM, Crowley RW, Liu KC (2018) Shunt-dependent hydrocephalus after aneurysmal subarachnoid hemorrhage: predictors and long-term functional outcomes. Neurosurgery 83(3):393–402. https://doi.org/10.1093/neuros/nyx393
Reddy GK (2012) Ventriculoperitoneal shunt surgery and the incidence of shunt revision in adult patients with hemorrhage-related hydrocephalus. Clin Neurol Neurosurg 114(9):1211–1216. https://doi.org/10.1016/j.clineuro.2012.02.050
Sakaki S, Ohta S, Kuwabara H, Shiraishi M (1987) The role of ventricular and cisternal drainage in the early operation for ruptured intracranial aneurysms. Acta Neurochir (Wien) 88(3–4):87–94. https://doi.org/10.1007/BF01404143
Sindou M (1994) Favourable influence of opening the lamina terminalis and Lilliequist’s membrane on the outcome of ruptured intracranial aneurysms. A study of 197 consecutive cases. Acta Neurochir (Wien). 127(1–2):15–6. https://doi.org/10.1007/BF01808539
Tomasello F, d’Avella D, de Divitiis O (1999) Does lamina terminalis fenestration reduce the incidence of chronic hydrocephalus after subarachnoid hemorrhage? Neurosurgery. 45(4):827–31. https://doi.org/10.1097/00006123-199910000-00018. (discussion 831-2)
Vale FL, Bradley EL, Fisher WS 3rd (1997) The relationship of subarachnoid hemorrhage and the need for postoperative shunting. J Neurosurg 86(3):462–466. https://doi.org/10.3171/jns.1997.86.3.0462
Walcott BP, Iorgulescu JB, Stapleton CJ, Kamel H (2015) Incidence, timing, and predictors of delayed shunting for hydrocephalus after aneurysmal subarachnoid hemorrhage. Neurocrit Care 23(1):54–58. https://doi.org/10.1007/s12028-014-0072-y
Wilson CD, Safavi-Abbasi S, Sun H et al (2017) Meta-analysis and systematic review of risk factors for shunt dependency after aneurysmal subarachnoid hemorrhage. J Neurosurg 126(2):586–595. https://doi.org/10.3171/2015.11.JNS152094
Winkler EA, Burkhardt JK, Rutledge WC et al (2018) Reduction of shunt dependency rates following aneurysmal subarachnoid hemorrhage by tandem fenestration of the lamina terminalis and membrane of Liliequist during microsurgical aneurysm repair. J Neurosurg 129(5):1166–1172. https://doi.org/10.3171/2017.5.JNS163271
Xie Z, Hu X, Zan X, Lin S, Li H, You C (2017) Predictors of shunt-dependent hydrocephalus after aneurysmal subarachnoid hemorrhage? A systematic review and meta-Analysis. World Neurosurg 106(844–860):e6. https://doi.org/10.1016/j.wneu.2017.06.119
Yonekawa Y, Imhof HG, Ogata N et al (1998) Aneurysm surgery in the acute stage: results of structured treatment. Neurol Med Chir (Tokyo) 38(Suppl):45–49. https://doi.org/10.2176/nmc.38.suppl_45
Yaşargil MG (1984) Microsurgical anatomy of the basal cisterns and vessels of the brain, diagnostic studies, general operative techniques and pathological considerations of the intracranial aneurysms. In: Yaşargil MG (ed) Microneurosurgery: microsurgical anatomy of the basal cisterns and vessels of the brain. Georg Thieme, Stuttgart, vol I, pp 346–347
Funding
Open access funding provided by University of Lausanne
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics approval and consent to participate
All procedures performed in studies involving human participants were in accordance with the ethical standards of the local Ethical Committee (Geneva Ethics Committee Board no. 11-233R, NAC 11-085R) and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. For this retrospective type of study, formal consent is not required.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Vascular Neurosurgery—Aneurysm
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Garvayo, M., Messerer, M., Starnoni, D. et al. The positive impact of cisternostomy with cisternal drainage on delayed hydrocephalus after aneurysmal subarachnoid hemorrhage. Acta Neurochir 165, 187–195 (2023). https://doi.org/10.1007/s00701-022-05445-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00701-022-05445-x