Abstract
Feline parvovirus infection, caused by feline parvovirus and canine parvovirus 2, is a highly contagious, life-threatening disease affecting cats. The available epidemiological data on parvovirus infection in cats in Egypt is limited. Therefore, the aim of the current study was to provide data concerning the epidemiological profile of cats infected with parvovirus, including the prevalence of parvovirus infection in cats in three Egyptian provinces (Sohag, Assiut, and Cairo) and the associated risk factors. Using rapid antigen tests of fecal samples and conventional PCR, the overall prevalence of parvovirus infection in cats was found to be 35% (35/100) and 43% (43/100), respectively. Anorexia, bloody diarrhea, severe dehydration, hypothermia, and vomiting were the most common clinical findings significantly associated with parvovirus-infected cats. The geographical location (Sohag) and the season (winter) were both statistically significant risk factors for parvovirus infection. These findings indicate that parvoviruses are circulating in different regions of Egypt. Our study provides baseline epidemiological data for future preventive and control measures against parvovirus infection, as well as highlighting the need for future genomic surveillance studies involving a large study population from various parts of Egypt in order to better shape the epidemiological picture of parvovirus infection.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Feline panleukopenia (FPL)/feline parvovirus (FPV) infection is a highly contagious, life-threatening disease affecting all members of the family Felidae, typically characterized by acute enteritis, severe dehydration, vomiting, and sepsis as a result of lymphoid depletion and pancytopenia [1,2,3]. Parvovirus infection in cats is commonly associated with FPV. However, it can also be caused by variants of canine parvovirus 2 (CPV-2), specifically CPV-2a, b, and c [3]. Collectively, FPV and CPV-2 are included within the species Carnivore protoparvovirus 1 (family Parvoviridae) and are found in a variety of carnivore and feline species [4,5,6].
Parvovirus infection occurs most frequently in young unvaccinated or incompletely vaccinated kittens [7, 8]. Importantly, even with treatment, the mortality rate of parvovirus infection ranges from 25% to 100%, depending on the severity of clinical signs [4]. Marked leucopenia or thrombocytopenia, as well as hypokalemia or hypoalbuminemia, are indicators of a poor prognosis [7, 8].
Despite the danger of parvovirus infection to cats and the growing interest in owning cats as pets, the epidemiological status of parvovirus infection is still unknown in most Egyptian localities. The only previous study of the prevalence of FPV in Egypt examined 165 diseased cats and found a prevalence of 40% and 45%, using ELISA and PCR, respectively [9]. Therefore, the aim of this study was to provide data concerning the epidemiological profile of cats infected with parvoviruses (FPV/CPV-2) in Egypt, including the prevalence of parvovirus infection among cats and the associated risk factors.
Materials and methods
Ethical approval
All procedures were performed following the ethical standards of the institutional ethics committee of Assiut University. All cat owners were informed about the objectives of the study and methodology, voluntary participation, and confidentiality of personal information. All cats were handled in compliance with Assiut University's regulations for animal research, and samples were collected with the owners' consent. The study was approved by the institutional ethics committee of Assiut University.
Study area
The current study was conducted in three Egyptian provinces: Cairo, Assiut, and Sohag, from November 2020 to December 2021. Cats were admitted to the Imbaba Veterinary Hospital and Pets Care Clinic (Cairo), Smile Pet Clinic (Assiut), and Vet Pet Clinic (Sohag).
Cats
This study included a total of 100 cats of different ages, sexes, and breeds. Only cats exhibiting one or more clinical signs of parvovirus infection (lethargy, anorexia, vomiting, and/or bloody diarrhea) were included.
Data collection and clinical examination
Data obtained from each cat owner included the age (1-6 months, >6-12 months, or >12 months), sex (male or female), breed (Persian, Siamese, Egyptian Mau, or mixed breed), location (Sohag, Assiut, or Cairo), vaccination status, and living conditions (indoors or outdoors) of the cat. Clinical signs such as anorexia, lethargy, vomiting, diarrhea, and mucoid or bloody diarrhea were also recorded. All cats were physically examined on admission as described by Gaskell et al. [10]. Physical examination included body temperature measurement, visual examination of mucous membranes, and checking for signs of respiratory illness, digestive disturbances, or dehydration.
Sampling
Stool samples were collected directly from the rectum using fecal swabs [11]. Each swab was submerged in 400 μl of sterile saline, and the suspension was divided into two parts; one was used immediately for rapid antigen tests, and the other was stored at -20 °C for molecular detection.
Screening of cats for the presence of FPV and CPV-2 antigen
SNAP Parvovirus Antigen Test Kits, including SNAP Parvo (IDEXX Laboratories, USA) and VDRG ®CPV Ag Rapid Kit (Medain, Korea), which are based on ELISA and immunochromatographic technology, respectively, were used according to the manufacturer's instructions to screen for parvovirus infection in cats.
Molecular detection
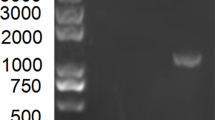
DNA was extracted from all fecal samples using a QIAamp Fast DNA Stool Mini Kit (QIAGEN, Germany) according to the manufacturer's instructions. Molecular screening was carried out by PCR using primers (Sigma-Aldrich) designed by Stephen Dunham for detection of the FPV VP-2 gene (felVP2-3820F, 5-TTGARGCRTCTACACAAGGG-3′; VP2-4247R, 5′-TGGTGCATTTACATGAAGTCTTGG-3′) [12]. The PCR reactions were carried out in a total volume of 25 µl, using the following PCR cycling conditions: initial denaturation at 95°C for 1 minute; 40 cycles of denaturation for 15 seconds at 95°C, primer annealing at 60°C for 15 seconds, and extension at 72°C for 15 seconds; and a final extension for 2 minutes at 72°C. The PCR products were visualized using 1.5% agarose gel electrophoresis and a UV transilluminator.
Statistical analysis
To measure the impact of each factor individually on the prevalence of the disease in cats (i.e., age, sex, breed, season, locality, vaccination status, and lifestyle) and the association of clinical signs with the occurrence of the disease (i.e., temperature, activity, appetite, vomiting, diarrhea, and dehydration), relative risk was calculated and chi-square tests were performed using SPSS statistics software (IBM Corp, USA, Version 29). A probability value (P-value) less than 0.05 was considered statistically significant when comparing two groups, but Bonferroni correction of the p-value using the formula anew = aoriginal /n was used when comparing more than two groups. P < 0.0167 was considered statistically significant when comparing three groups, while P < 0.0125 was considered statistically significant when comparing four groups.
Results
Using rapid antigen tests and conventional PCR, the overall prevalence of parvovirus infection in cats was found to be 35% (35/100) and 43% (43/100), respectively. The size of the PCR product amplified from the FPV VP-2 gene was 428 base pairs, which was similar to that of the positive control.
Among the parvovirus PCR-positive cats, 86.05% (37/43) were lethargic, compared to 68.42% (39/57) of parvovirus-negative cats; 88.37% (38/43) had anorexia, compared to 70.2% (40/57) of negative cats; 11.63% (5/6) had hypothermia, compared to 1.76% (1/6) of negative cats; 74.4% (32/43) showed persistent vomiting, compared to 49.1% (28/57) of negative cats; 58.14% (25/43) had mucoid to bloody diarrhea, compared to 57.9% (33/57) of negative cats; and 30.23% (13/43) showed severe dehydration, compared to 10.53% (6/57) of negative cats (Table 1). There was a significant association between PCR test results and clinical signs in cats (Table 1). Cats with anorexia, bloody diarrhea, severe dehydration, hypothermia, and vomiting had a significantly higher prevalence of parvovirus infection than cats with normal appetite, mucoid diarrhea, no dehydration, normal body temperature, and absence of vomiting (Table 1).
Among the risk factors analyzed, the geographic location and the season were statistically significant factors associated with parvovirus infection (Table 2). Cats from Sohag had a significantly higher prevalence rate (62.5%) than cats from Assiut and Cairo (27.3%, and 40%, respectively). Furthermore, cats sampled during the winter (54.5%) were significantly more frequently positive for parvovirus than cats sampled during the spring (46.1%), summer (15.8%), and autumn (41.7%). On the other hand, there was no significant relationship between the various risk factors analyzed (age, sex, breed, vaccination status, and lifestyle) and infection rate among the cats studied (Table 2).
Discussion
Feline parvovirus infection is a common disease that causes diarrhea in cats and can be life-threatening in severe cases. This study demonstrates that anorexia, bloody diarrhea, severe dehydration, hypothermia, and vomiting are the common clinical signs that are significantly associated with parvovirus infection in domestic cats in Egypt. These findings are similar to those of previous studies of FPV infection described by Mosallanejad et al. [13] and Miranda et al. [14]. Not all cats infected with FPV display clinical signs, and the disease severity varies with immune status, age, and concurrent infections [7].
Our survey showed that 35 cats were positive for parvovirus infection when using the rapid antigen test, while conventional PCR identified 43 positive cases. This difference may be attributed to false-negative results obtained when using fecal antigen tests. Due to its sensitivity and specificity, PCR is more reliable for detection of parvovirus in fecal samples [15, 16]. Notably, three out of the 35 positive samples (as determined by the rapid test) were found to be negative when tested by PCR. One of these three animals had recently been vaccinated. A similar result was also reported by Abd-Eldaim et al. [17], who also had one case in which the sample tested positive for parvovirus infection by the rapid test, but negative by PCR. False-positive results can be obtained by both the rapid test and PCR after vaccination because the virus is shed in feces [18], and therefore, the reason for the discrepancy between the results of the two tests in this case is still unknown.
False-positive results can occur in recently FPV-vaccinated kittens (within 14 days of vaccination) with modified live vaccines [19].
Furthermore, rapid assays can only detect parvoviruses in feces for up to 48 hours after infection, and their sensitivity and specificity vary among the different rapid tests and stages of infection [17, 20].
The overall prevalence of parvovirus infection among the examined cats was 43% when using PCR. This high prevalence rate could be attributed to environmental contamination with the virus (FPV or CPV) or the lack of vaccination against FPV infection (91% of the examined cats were unvaccinated), since unvaccinated cats are more vulnerable to infection [21, 22]. Additionally, infected homeless cats, which roam about in search of food, contribute to disease transmission [23, 24]. Similar rates of FPV prevalence in Giza, Egypt, were reported by Awad et al. [9], who found a prevalence of FPV infection of 45% among 165 diseased cats, using PCR. In contrast to our findings, a lower prevalence of parvovirus (either FPV or CPV) infection, 8.15% of fecal samples obtained from 1719 apparently healthy cats, was found in the United Kingdom, using PCR, whereas a higher prevalence of parvovirus infection, 72.7% of blood samples obtained from 11 cats, was found in Indonesia, also using PCR [25]. These divergent results might be explained by differences in the vaccination rates between countries, differences in natural exposure to the virus, the study populations chosen, the use of different diagnostic tests, housing conditions, and the immune status of the cats [7].
Regarding the prevalence of the disease in different Egyptian provinces, the significantly higher infection rate in Sohag province may be attributed to a lower rate of vaccination among the cats tested in Sohag than in Assiut and Cairo.
The prevalence of parvovirus infection was found to be inversely proportional to age, without a significant difference, which is consistent with the results reported by Kim et al. [26], who observed a higher prevalence of parvovirus infection in kittens (less than one year old) than adult cats (more than one year old). Young kittens are more vulnerable to parvovirus infection because they lack antibody protection [8]. Furthermore, early weaning, an improper practice of some cat breeders to reduce breeding costs, results in a gradual decline in maternally derived antibody titers [27]. On the other hand, older cats are protected either through subclinical infection or previous vaccination [8].
The prevalence rate of parvovirus infection in relation to sex and breed of cat showed no significant difference. These findings are consistent with previous studies that found no significant difference in disease prevalence based on breed or gender [11, 13, 21, 23].
In our study, cats were significantly more frequently infected with parvovirus in the winter season. A similar seasonality of parvovirus infection among cats has been reported in New Zealand and Australia, where case numbers peaked from December to May [28]. In contrast, outbreaks commonly occur during July, August, and September in the United States due to admission of kittens that were born in the spring to shelters at a time when maternal immunity is waning [29]. However, the season may be a confounding variable, because cats are both seasonal and prolific breeders; an average adult queen can produce two litters between spring and autumn [30].
Conclusions
The current study provides the first evidence that parvoviruses are circulating among cat populations in Sohag, Assiut, and Cairo provinces. Anorexia, bloody diarrhea, severe dehydration, hypothermia, and vomiting were the most common clinical findings in cats that were positive for parvovirus infection. The geographical location (Sohag) and the season (winter) were statistically significant risk factors associated with parvovirus infection. Our study provides baseline epidemiological data for future management of parvovirus infection and underscores the urgent need for cat vaccination programs based on the current international guidelines.
Limitations
Although the current study provides precise epidemiological data on parvovirus infection in cats in Egypt, the study population was small, and no molecular characterization was performed to determine whether the current parvovirus infections were caused by FPV or CPV-2. Therefore, genomic surveillance studies involving a large study population from various parts of Egypt are still needed to better shape the epidemiological picture of parvovirus infection in cats.
Data availability
The authors declare that the data supporting the findings of this study are available with the paper and confirm that all data generated or anaylsed during this study are included in the publised article.
References
Driciru M, Siefert L, Prager KC et al (2006) A serosurvey of viral infections in lions (Panthera leo), from Queen Elizabeth National Park, Uganda. J Wildl Dis 42:667–671. https://doi.org/10.7589/0090-3558-42.3.667
Nishimura Y, Goto Y, Yoneda K et al (1999) Interspecies transmission of feline immunodeficiency virus from the domestic cat to the Tsushima Cat ( Felis bengalensis euptilura ) in the wild. J Virol 73:7916–7921. https://doi.org/10.1128/jvi.73.9.7916-7921.1999
Hellard E, Fouchet D, Santin-Janin H et al (2011) When cats’ ways of life interact with their viruses: a study in 15 natural populations of owned and unowned cats (Felis silvestris catus). Prev Vet Med 101:250–264. https://doi.org/10.1016/j.prevetmed.2011.04.020
Barrs VR (2019) Feline panleukopenia: a re-emergent disease. Vet Clin N Am 49:651–670. https://doi.org/10.1016/j.cvsm.2019.02.006
Nakamura K, Ikeda Y, Miyazawa T et al (2001) Characterisation of cross-reactivity of virus neutralising antibodies induced by feline panleukopenia virus and canine parvoviruses. Res Vet Sci 71:219–222. https://doi.org/10.1053/rvsc.2001.0492
Cotmore SF, Agbandje-McKenna M, Chiorini JA et al (2014) The family Parvoviridae. Arch Virol 159:1239–1247. https://doi.org/10.1007/s00705-013-1914-1
Stuetzer B, Hartmann K (2014) Feline parvovirus infection and associated diseases. Vet J 201:150–155. https://doi.org/10.1016/j.tvjl.2014.05.027
Kruse BD, Unterer S, Horlacher K et al (2010) Prognostic factors in cats with feline panleukopenia. J Vet Intern Med 24:1271–1276. https://doi.org/10.1111/j.1939-1676.2010.0604.x
Awad R, Khalil W, Attallah A (2018) Epidemiology and diagnosis of feline panleukopenia virus in Egypt: clinical and molecular diagnosis in cats. Vet World 11:578–584. https://doi.org/10.14202/vetworld.2018.578-584
Gaskell RM, Tennant B, Bennett M, Willoughby K (1996) Feline and canine infectious diseases. Iowa State Press, Ames
Islam MA, Rahman MS, Rony SA et al (2010) Antigenic detection of feline panleukopenia virus in local breed cats at Tangail district in Bangladesh. Int J Biores 2:25–28
Hamed MI, Fins I, Daly JM, et al (2019) Prevalence of Feline and Canine Parvoviruses in Shelter - Housed Cats in The United Kingdom By. 99:251–271***
Mosallanejad B, Avizeh R, Ghorbanpoor Najafabadi M (2009) Antigenic detection of Feline panleukopenia virus (FPV) in diarrhoeic companion cats in Ahvaz area. Iran J Vet Res 10:289–293
Miranda C, Thompson G (2016) Canine parvovirus: The worldwide occurrence of antigenic variants. J Gen Virol 97:2043–2057. https://doi.org/10.1099/jgv.0.000540
Meason-Smith C, Diesel A, Patterson AP et al (2017) Characterization of the cutaneous mycobiota in healthy and allergic cats using next generation sequencing. Vet Dermatol 28:71-e17. https://doi.org/10.1111/vde.12373
Schunck B, Kraft W, Truyen U (1995) A simple touch-down polymerase chain reaction for the detection of canine parvovirus and feline panleukopenia virus in feces. J Virol Methods 55:427–433. https://doi.org/10.1016/0166-0934(95)00069-3
Abd-Eldaim M, Beall MJ, Kennedy MA (2009) Detection of feline panleukopenia virus using a commercial ELISA for canine parvovirus. Vet Ther 10:1–6
Bergmann M, Schwertler S, Speck S et al (2019) Faecal shedding of parvovirus deoxyribonucleic acid following modified live feline panleucopenia virus vaccination in healthy cats. Vet Rec 185:83. https://doi.org/10.1136/VR.104661
Patterson EV, Reese MJ, Tucker SJ et al (2007) Effect of vaccination on parvovirus antigen testing in kittens. J Am Vet Med Assoc 230:359–363
Mende K, Stuetzer B, Truyen U, Hartmann K (2014) Evaluation of an in-house dot enzyme-linked immunosorbent assay to detect antibodies against feline panleukopenia virus. J Feline Med Surg 16:805–811. https://doi.org/10.1177/1098612X14520812
Awad RA, Khalil WKB, Attallah AG (2018) Feline panleukopenia viral infection in cats: application of some molecular methods used for its diagnosis. J Genet Eng Biotechnol 16:491–497. https://doi.org/10.1016/j.jgeb.2018.08.001
Poole GM (1972) Stability of a modified, live panleucopenia virus stored in liquid phase. Appl Microbiol 24:663–664. https://doi.org/10.1128/aem.24.4.663-664.1972
Bayati A (2016) Detection of feline parvovirus (FPV) from cats infected with enteritis using rapid test and polymerase chain reaction in Iraq. Kufa J Vet Med Sci 7:61–70
Clegg SR, Coyne KP, Dawson S et al (2012) Canine parvovirus in asymptomatic feline carriers. Vet Microbiol 157:78–85. https://doi.org/10.1016/j.vetmic.2011.12.024
Raj VP, Haryanto A (2020) Clinical study and rapid detection of feline parvovirus in suspected cats by polymerase chain reaction method. Indones J Vet Sci 1:15–23. https://doi.org/10.22146/ijvs.v1i1.44835
Kim SG, Lee KI, Kim HJ, Park HM (2013) Prevalence of feline panleukopenia virus in stray and household cats in Seoul, Korea. J Vet Clin 30:333–338
Decaro N, Desario C, Campolo M et al (2004) Evaluation of lactogenic immunity to canine parvovirus in pups. New Microbiol 27:375–379
Van Brussel K, Carrai M, Lin C et al (2019) Distinct lineages of feline parvovirus associated with epizootic outbreaks in Australia, New Zealand and the United Arab Emirates. Viruses. https://doi.org/10.3390/V11121155
Reif JS (1976) Seasonally, natality and herd immunity in feline panleukopenia. Am J Epidemiol 103:81–87. https://doi.org/10.1093/oxfordjournals.aje.a112208
Jöchle W, Jöchle M (1993) Reproduction in a feral cat population and its control with a prolactin inhibitor, cabergoline. J Reprod Fertil Suppl 47:419–424
Funding
Open access funding provided by The Science, Technology & Innovation Funding Authority (STDF) in cooperation with The Egyptian Knowledge Bank (EKB). The authors have not disclosed any funding.
Author information
Authors and Affiliations
Contributions
Abdel-Baky MMM: writing–original draft, acquisition of data (sample collection and performing SNAP and PCR tests), analysis and interpretation of the data. El-Khabaz KAS: analysis and interpretation of the data (results), writing–reviewing and editing. Abdelbaset AE: writing–reviewing and editing. Hamed MI: conception and design of the study, acquisition of data, analysis and interpretation of the data, supervision, writing–reviewing and editing
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interest.
Additional information
Handling Editor: Sheela Ramamoorthy
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Abdel-Baky, M.M., El-Khabaz, K.A., Abdelbaset, A.E. et al. Clinico-epidemiological survey of feline parvovirus circulating in three Egyptian provinces from 2020 to 2021. Arch Virol 168, 126 (2023). https://doi.org/10.1007/s00705-023-05751-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00705-023-05751-4