Abstract
Background
Previous studies suggest a relationship between central nervous system inflammatory demyelinating diseases and anti-N-methyl-d-aspartate receptor (NMDAR) encephalitis. Also, the overlap between anti-NMDAR encephalitis and multiple sclerosis (MS) has been reported. However, the pathogenesis and clinical characteristics are still obscure.
Case presentation
A 33-year-old woman presented with diplopia and sensory ataxia at the onset. The cerebrospinal fluid (CSF) anti-NMDAR antibodies were positive (1:3.2), and nuclear magnetic resonance imaging (MRI) showed bilateral centrum ovale and lateral ventricle demyelinating lesions. Therefore, she was diagnosed with anti-NMDAR encephalitis. After administering intravenous immunoglobulin and oral prednisone, her lesions disappeared, and symptoms were relieved. The condition was maintained with a low dose of prednisone, but her lesions reappeared on MRI. Consequently, immunomodulatory therapy of mycophenolate mofetil was initiated. However, she developed dysarthria and right limb ataxia after 10 months with a positive CSF anti-NMDAR antibody (1:1) and positive oligoclonal band. The MRI showed symmetrical multiple demyelinating lesions. Considering the MS diagnosis, her neurological dysfunction again improved significantly after intravenous methylprednisolone. Unfortunately, her symptoms aggravated for the second time when teriflunomide was started. Finally, her condition was controlled again with oral prednisone.
Conclusions
Consistent with previous cases of overlapping anti-NMDAR encephalitis and MS, patients often show atypical symptoms on MRIs and immunological tests. The overlap cannot be arbitrarily treated because of the recurrence of previous diseases. Long-term follow-up, dynamic antibody monitoring, and MRI examination are crucial for these patients. The special dependency of the patient on glucocorticoids in this study has been rarely reported, which may guide the treatment of insensitivity to disease-modifying therapy in recurrent overlapping anti-NMDAR encephalitis and MS.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Background
Multiple sclerosis (MS) is an autoimmune inflammation-mediated demyelinating disease of the central nervous system (CNS). The incidence rate of MS has increased in recent years and has become an essential cause of nontraumatic disability in young people [1]. Dissemination in space and time (DIT) is a significant clinical feature of MS, where lesions often involve the optic nerve, brainstem, cerebellum, cerebral hemisphere, and spinal cord [2]. Generally, diagnosing MS is not difficult. However, a long-term clinical follow-up may be required for atypical cases with first onset to determine the final diagnosis.
Anti-NMDAR encephalitis is the most common diagnosis of autoimmune encephalitis. The main clinical manifestations of anti-NMDAR encephalitis include mental and behavioral disorders, cognitive dysfunction, epilepsy, dyskinesia, and autonomic dysfunction [3]. Previous studies have reported the overlaps between anti-NMDAR encephalitis and central demyelinating diseases such as neuromyelitis optica spectrum disorder, acute disseminated encephalomyelitis, optic neuritis, and MS. However, these reports are still rare and have unclear clinical features [4]. Therefore, this study aimed to report a case of overlapping anti-NMDAR encephalitis and MS. We also comprehensively gathered similar reports to analyze the clinical characteristics of overlapping anti-NMDAR encephalitis and MS.
Case presentation
First admission (August 2021)
A 33-year-old woman developed a transient fever 1 month after vaccination for the 2019 novel coronavirus disease (COVID-19), with a maximum temperature of 38.5 °C. She later developed diplopia and unstable gait, successively, and was admitted to our hospital on August 30, 2021. The neurological examination showed limitation of the left inferior movement of the left eyeball, a four grade of right lower limb muscle strength, and Romberg sign. Abnormalities were not detected on hematology and serum biochemistry, including infection, immunity, endocrine system, and tumor. Abdominal and pelvic ultrasound, electrocardiogram, and chest X-ray also showed no abnormalities. Brainstem auditory-evoked potential, visual-evoked potential, electroneurography, and electroencephalogram were normal. Her Hess screen examination suggested superior oblique muscle paralysis of the left eye. The fundus photography showed the left eyeball external rotation. A somatosensory evoked potential indicated that the N22-P40 interval in the right lower limbs was prolonged.
On September 3, 2021, magnetic resonance imaging (MRI) of the head, entire spinal cord, and brainstem was normal (Fig.1a). A cerebrospinal fluid (CSF) examination showed a protein level of 1157.61 mg/L and 46 × 106/L total nucleated cells with 97% lymphocytes. The serum and CSF central nervous demyelinating antibody, peripheral nerve ganglioside antibody, oligoclonal band (OCB), immunoglobulin G (IgG) index, and intrathecal synthesis index were all found to be within normal ranges. We considered a diagnosis of viral brainstem encephalitis, and she was treated with intravenous ganciclovir 0.25 g every 12 h. However, her symptoms did not improve after 10 days of treatment. On September 14, 2021, a CSF recheck showed 35 × 106/L total nucleated cells with 97% lymphocytes and a protein level of 963.47 mg/L. The CSF metagenomic next-generation sequencing (mNGS) did not show a pathogenic microbial gene sequence. Later, unfortunately, the patient’s right lower limb muscle strength progressively decreased.
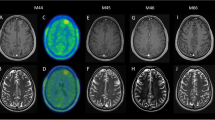
On September 26, 2021, the reexamination of the CSF suggested a nucleated cell count of 11 × 106/L with 90% lymphocytes and a protein level of 892 mg/L. At this point, further autoimmune encephalitis antibody detection suggested that anti-NMDAR antibody IgG in CSF was positive with a titer of 1:3.2, and the serum and CSF paratumor antibodies, anti-myelin oligodendrocyte glycoprotein (MOG) autoantibodies, were negative. Furthermore, on September 30, 2021, the patient’s head MRI showed abnormal signals in the centrum ovale majus and bilateral ventricle, indicating demyelinating lesions (Fig.1b). After considering a diagnosis of anti-NMDAR encephalitis, the patient received intravenous immunoglobulin (IVIG) (0.4 g/kg ⋅ d) for 5 days and oral prednisone 50 mg/d. After 1 month, her lower limb weakness and walking instability disappeared, and only diplopia partly remained. In addition, previous abnormal signals on MRI were completely absorbed (Fig.1c; on November 1, 2021). As a result, the dose of prednisone was reduced regularly.
Second admission (April 2022)
During the course of prednisone dose reduction in this patient, her diplopia was never completely relieved. When the prednisone dose was reduced to 15 mg/d, her walking instability recurred. The patient was readmitted, and a CSF follow-up examination showed normal nucleated cells and proteins. The titer of CSF anti-NMDAR antibody IgG was 1:1. At the same time, we examined the anti-MOG autoantibodies in CSF and serum, which were negative. In addition, the MRI manifested abnormal signals in the left lateral ventricle and centrum semiovale, and the lesions were diffusion limited without enhancement (Fig.2). She then received mycophenolate mofetil immunotherapy 1 g/d, and prednisone dose was gradually reduced to withdrawal.
Third admission (August 2022)
Mycophenolate mofetil was maintained in the patient, but she developed a series of new symptoms, including right limb stiffness, writing difficulties, and slurred speech after only 1 month of prednisone withdrawal. The neurological examination at readmission suggested dysarthria and right limb ataxia. The number of nucleated cells was 10 × 106/L with 90% lymphocytes in CSF, and anti-NMDAR antibody IgG was weakly positive (1:1). Oddly, we found positive CSF OCBs (10 bands) at this time. According to multiple MRI lesions, including enlarged left lateral ventricle and centrum semiovale lesions, symmetrical new lesions were found at the fourth ventricle, midbrain aqueduct, splenium of corpus callosum, and posterior horn of the lateral ventricle (Fig.3). We considered a diagnosis of MS. Consequently, the patient was given intravenous methylprednisolone (IVMP) (1 g/d) for 5 days, and her symptoms rapidly improved (Fig.4).
Follow-up
Later, the patient was put on immune modification therapy with teriflunomide. However, her previous neurologic impairment symptoms were again exacerbated. Based on the repeated deterioration of this patient’s condition during the reduction and discontinuation of prednisone or methylprednisolone, glucocorticoid dependence was speculated, and prednisone was given again. The patient’s symptoms again improved. She is still receiving sustained prednisone therapy with a minimum effective dose of 20 mg/d.
Literature review and discussion
We searched databases, such as PubMed, Embase, Sinomed, CNKI, and Wanfang, for case reports on MS and anti-NMDAR encephalitis published before August 2023. The search terms were “anti-N-methyl-D-aspartate receptor encephalitis” and “multiple sclerosis.” We found 18 related cases, and the clinical details are summarized in Table1. All patients were diagnosed with MS or anti-NMDAR encephalitis, successively, with an interval of 3 weeks to 15 years. It seemed that the diagnoses of many cases were converted from MS into anti-NMDAR encephalitis.
Vaccination and viral infection may be the inducing factors for anti-NMDAR encephalitis and MS [1, 5]. The patient in this study got a fever after receiving the COVID-19 vaccine. However, her diplopia and unstable gait did not conform to the characteristics of anti-NMDAR encephalitis. Consequently, we did not initially detect NMDAR antibodies. Her nervous system on MRI could not preclude NMDAR encephalitis. About 50% of patients with anti-NMDAR encephalitis have no abnormal signal on MRI [6]. Some studies suggested that CSF protein concentration and nucleated cell count might be normal or slightly elevated in MS and that a CSF protein level of more than 100 mg/dL or nucleated cell count of more than 50/mm3might indicate other diseases [7]. The first CSF examination of the patient in this study and negative OCBs did not support an MS diagnosis.
After antiviral therapy, a mild decrease in the patient’s protein level and nucleated cell count in CSF did not indicate viral meningoencephalitis sensitivity to antiviral therapy. In addition, her CSF mNGS, which had good diagnostic value for CNS infection, was also normal [8]. We later found that the CSF anti-NMDAR antibody was slightly positive (1:3.2). However, the patient’s MRI did not support anti-NMDAR encephalitis but supported MS. The MRIs of NMDA usually show high T2 and FLAIR signals in the hippocampus, cerebellum, cerebral cortex, frontal lobe, insula, and brainstem [5]. Unfortunately, we did not reinspect OCB due to previous negative results, but we later speculated that it might be positive at that time. We tentatively used IVIG and oral prednisone due to the patient’s refusal to IVMP, with effective results.
The inhibition of glucocorticoids for MS relapse may be transient [9,10]. Studies have suggested that IVMP can rapidly reduce the enhanced lesions of relapsing MS on MRI by about 96%. This benefit, however, may last for only 1 month and be followed by new or enlarged lesions [11]. As for anti-NMDAR encephalitis, delayed immunotherapy and high antibody titer may be the recurrent factors for anti-NMDAR encephalitis, and glucocorticoids can inhibit its recurrence [12,13]. The recurrent MRI lesions of the left lateral ventricle and centrum ovale majus after prednisone were reduced to 15 mg/d. The new neurological deficits and multiple lesions after prednisone withdrawal might support this conclusion. This also suggested that the inhibitory potency of glucocorticoids for the recurrence of MS might be related to its measurement [14].
This patient with MS seemed to be only sensitive to glucocorticoids. MS recurred during the treatment of mycophenolate mofetil and teriflunomide, possibly due to progressive MS [15]. In addition, disease-modifying therapy (DMT) can reduce the risk of recurrence of relapsing-remitting MS by 36%–85%, although different drugs may have different effects. Some studies suggested that MS patients who relapsed repeatedly, before or shortly after DMT, might relapse uncontrollably despite continuous DMT [16,17,18,19]. Anti-NMDAR encephalitis and CNS demyelinating diseases can overlap, but the underlying mechanism is still unclear [20]. For the patient described in this study, the pathogenesis may be a common autoimmune response [20]. The occurrence of anti-NMDAR encephalitis may cause immunologic imbalance and induce MS [21]. In addition, NMDARs are partially expressed on oligodendrocytes that form myelin, and demyelination is evoked when anti-NMDAR encephalitis occurs [22]. Conversely, MS can also expose NMDAR antigens [21, 23].
Conclusions
In conclusion, the mechanism of overlapping anti-NMDAR encephalitis and MS may be the coexistence of autoimmune reactions. The symptoms, MRIs, and immunological examinations of CSF or serum are often atypical. Thus, the positive antibody cannot be used as the only diagnostic basis. Furthermore, the overlap cannot be arbitrarily treated as the recurrence of a previous disease. Long-term follow-up, dynamic antibody monitoring, and MRI examination are crucial for these patients. For the patient described in this study, only glucocorticoids could control the recurrence. So, we speculate that this may be related to the short overlap time between the administration of glucocorticoids and DMT. Long-term use of the combination of low-dose effective glucocorticoids and DMT may be effective against the relapse of overlapping anti-NMDAR encephalitis and MS.
Data availability
The datasets are available from the corresponding author on reasonable request.
References
Dobson R, Giovannoni G (2019) Multiple sclerosis - a review. Eur J Neurol 26(1):27–40.https://doi.org/10.1111/ene.13819
Oh J, Vidal-Jordana A, Montalban X (2018) Multiple sclerosis: clinical aspects. Curr Opin Neurol 31(6):752–759.https://doi.org/10.1097/WCO.0000000000000622
Guasp M, Dalmau J (2018) Encephalitis associated with antibodies against the NMDA receptor. Encefalitis por anticuerpos contra el receptor de NMDA. Med Clin (Barc) 151(2):71–79.https://doi.org/10.1016/j.medcli.2017.10.015
Ramberger M, Bsteh G, Schanda K, Höftberger R, Rostásy K, Baumann M et al (2015) NMDA receptor antibodies: a rare association in inflammatory demyelinating diseases. Neurol Neuroimmunol Neuroinflamm 2(5):e141.https://doi.org/10.1212/NXI.0000000000000141
Dalmau J, Lancaster E, Martinez-Hernandez E, Rosenfeld MR, Balice-Gordon R (2011) Clinical experience and laboratory investigations in patients with anti-NMDAR encephalitis. Lancet Neurol 10(1):63–74.https://doi.org/10.1016/S1474-4422(10)70253-2
Dalmau J, Tüzün E, Wu HY, Masjuan J, Rossi JE, Voloschin A et al (2007) Paraneoplastic anti-N-methyl-D-aspartate receptor encephalitis associated with ovarian teratoma. Ann Neurol 61(1):25–36.https://doi.org/10.1002/ana.21050
Stangel M, Fredrikson S, Meinl E, Petzold A, Stüve O, Tumani H (2013) The utility of cerebrospinal fluid analysis in patients with multiple sclerosis. Nat Rev Neurol 9(5):267–276.https://doi.org/10.1038/nrneurol.2013.41
Ramachandran PS, Wilson MR (2020) Metagenomics for neurological infections-expanding our imagination. Nat Rev Neurol 16(10):547–556.https://doi.org/10.1038/s41582-020-0374-y
Brusaferri F, Candelise L (2000) Steroids for multiple sclerosis and optic neuritis: a meta-analysis of randomized controlled clinical trials. J Neurol 247(6):435–442.https://doi.org/10.1007/s004150070172
Vedula SS, Brodney-Folse S, Gal RL, Beck R (2007) Corticosteroids for treating optic neuritis. Cochrane Database Syst Rev (1):CD001430. Published 2007 Jan 24.https://doi.org/10.1002/14651858.CD001430.pub2
Thrower BW (2009) Relapse management in multiple sclerosis. Neurologist 15(1):1–5.https://doi.org/10.1097/NRL.0b013e31817acf1a
Zhao YY, Wang HX, Wang M, Yuan B, Sun TW, Xu YM (2022) Relapse factors of patients of anti-N-methyl-D-aspartate receptor encephalitis. Acta Neurol Scand 145(4):434–441.https://doi.org/10.1111/ane.13568
Zhong R, Chen Q, Zhang X, Zhang H, Lin W (2022) Relapses of Anti-NMDAR, anti-GABABR and anti-LGI1 encephalitis: a retrospective cohort study. Front Immunol 13:918396. Published 2022 Jun 9.https://doi.org/10.3389/fimmu.2022.918396
Oliveri RL, Valentino P, Russo C, Sibilia G, Aguglia U, Bono F et al (1998) Randomized trial comparing two different high doses of methylprednisolone in MS: a clinical and MRI study. Neurology 50(6):1833–1836.https://doi.org/10.1212/wnl.50.6.1833
Baldassari LE, Fox RJ (2018) Therapeutic advances and challenges in the treatment of progressive multiple sclerosis. Drugs 78(15):1549–1566.https://doi.org/10.1007/s40265-018-0984-5
Held U, Heigenhauser L, Shang C, Kappos L, Polman C, Sylvia Lawry Centre for MS Research (2005) Predictors of relapse rate in MS clinical trials. Neurology 65(11):1769–1773.https://doi.org/10.1212/01.wnl.0000187122.71735.1f
Jokubaitis VG, Li V, Kalincik T, Izquierdo G, Hodgkinson S, Alroughani R et al (2014) Fingolimod after natalizumab and the risk of short-term relapse. Neurology 82(14):1204–1211.https://doi.org/10.1212/WNL.0000000000000283
Rovaris M, Comi G, Ladkani D, Wolinsky JS, Filippi M, European/Canadian Glatiramer Acetate Study Group (2003) Short-term correlations between clinical and MR imaging findings in relapsing-remitting multiple sclerosis. AJNR Am J Neuroradiol 24(1):75–81
Sormani MP, Rovaris M, Comi G, Filippi M (2007) A composite score to predict short-term disease activity in patients with relapsing-remitting MS. Neurology 69(12):1230–1235.https://doi.org/10.1212/01.wnl.0000276940.90309.15
Titulaer MJ, Höftberger R, Iizuka T, Leypoldt F, McCracken L, Cellucci T et al (2014) Overlapping demyelinating syndromes and anti–N-methyl-D-aspartate receptor encephalitis. Ann Neurol 75(3):411–428.https://doi.org/10.1002/ana.24117
Hacohen Y, Deiva K, Pettingill P, Waters P, Siddiqui A, Chretien P et al (2014) N-methyl-D-aspartate receptor antibodies in post-herpes simplex virus encephalitis neurological relapse. Mov Disord 29(1):90–96.https://doi.org/10.1002/mds.25626
Lipton SA (2006) NMDA receptors, glial cells, and clinical medicine. Neuron 50(1):9–11.https://doi.org/10.1016/j.neuron.2006.03.026
Leypoldt F, Titulaer MJ, Aguilar E, Walther J, Bönstrup M, Havemeister S et al (2013) Herpes simplex virus-1 encephalitis can trigger anti-NMDA receptor encephalitis: case report. Neurology 81(18):1637–1639.https://doi.org/10.1212/WNL.0b013e3182a9f531
Johnston J, Ali K, Pearson OR, Rickards C, Vincent A (2010) POI14 Multiple sclerosis: a potential association with anti-N-methyl-d-aspartate receptor encephalitis. J Neurol Neurosurg Psychiatry 81(11):e56.https://doi.org/10.1136/jnnp.2010.226340.155
Uzawa A, Mori M, Takahashi Y, Ogawa Y, Uchiyama T, Kuwabara S (2012) Anti-N-methyl D-aspartate-type glutamate receptor antibody-positive limbic encephalitis in a patient with multiple sclerosis. Clin Neurol Neurosurg 114(4):402–404.https://doi.org/10.1016/j.clineuro.2011.10.047
Waschbisch A, Kallmünzer B, Schwab S, Gölitz P, Vincent A, Lee DH et al (2014) Demyelinating disease and anti-N-methyl-D-aspartate receptor immunoglobulin G antibodies: a case report. BMC Res Notes 7:948. Published 2014 Dec 23.https://doi.org/10.1186/1756-0500-7-948
Fleischmann R, Prüss H, Rosche B, Bahnemann M, Gelderblom H, Deuschle K et al (2015) Severe cognitive impairment associated with intrathecal antibodies to the NR1 subunit of the N-methyl-D-aspartate receptor in a patient with multiple sclerosis. JAMA Neurol 72(1):96–99.https://doi.org/10.1001/jamaneurol.2014.1817
Huang G, Zou XQ, Wang YM, Wu XM (2016) A case of recurrent anti-NMDA receptor encephalitis misdiagnosed as multiple sclerosis. Chin J Neuroimmunol Neurol 23(05):379–380
Baheerathan A, Brownlee WJ, Chard DT, Shields K, Gregory R, Trip SA (2017) Antecedent anti-NMDA receptor encephalitis in two patients with multiple sclerosis. Mult Scler Relat Disord 12:20–22.https://doi.org/10.1016/j.msard.2016.12.009
Suleman S, Javed Q (2018) NMDAR (N-methyl-D-aspartate receptor) encephalitis in a patient with MS (multiple sclerosis): a rare and challenging case. BMJ Case Rep 2018:bcr2017222086. Published 2018 Feb 17.https://doi.org/10.1136/bcr-2017-222086
Gulec B, Kurucu H, Bozbay S, Dikmen Y, Sayman H, Tuzun E et al (2020) Co-existence of multiple sclerosis and anti-NMDA receptor encephalitis: a case report and review of literature. Mult Scler Relat Disord 42:102075.https://doi.org/10.1016/j.msard.2020.102075
Huang Y, Wang Q, Zeng S, Zhang Y, Zou L, Fu X et al (2020) Case report: overlapping multiple sclerosis with anti-N-methyl-D-aspartate receptor encephalitis: a case report and review of literature. Front Immunol 11:595417. Published 2020 Dec 9.https://doi.org/10.3389/fimmu.2020.595417
Tappatà M, Riguzzi P, Volpi L, Albini Riccioli L, Pantieri R, Michelucci R (2021) Long-term persistence of NMDAR antibodies after encephalitis with de novo occurrence of demyelinating disorder. Neurol Sci 42(1):301–303.https://doi.org/10.1007/s10072-020-04729-3
Chahal K, Endeman T, Scapinello S, Sapieha M (2021) Anti-N-methyl-D-aspartate receptor encephalitis presenting as atypical psychosis in multiple sclerosis: a case report. BMC Psychiatry 21(1):347. Published 2021 Jul 12.https://doi.org/10.1186/s12888-021-03351-7
Seyed Ahadi M, Ghadiri F, Sahraian MA, Naser MA (2021) Anti-N-methyl-d-aspartate receptor encephalitis in a patient with multiple sclerosis on dimethyl fumarate: a case report. Neurol Sci 42(9):3929–3931.https://doi.org/10.1007/s10072-021-05385-x
Zhou R, Jiang F, Cai H, Zeng Q, Yang H (2021) Case Report: Antibodies to the N-methyl-D-aspartate receptor in a patient with multiple sclerosis. Front Immunol 12:664364. Published 2021 Apr 23.https://doi.org/10.3389/fimmu.2021.664364
Karunaratne K, Ahrabian D, Monoghan B, Campion T, Yousry T, Lunn MP et al (2021) Bortezomib for anti-NMDAR encephalitis following daclizumab treatment in a patient with multiple sclerosis. BMJ Neurol Open 3(1):e000096.https://doi.org/10.1136/bmjno-2020-00009
Etemadifar M, Nouri H, Salari M, Sedaghat N (2022) Detection of anti-NMDA receptor antibodies following BBIBP-CorV COVID-19 vaccination in a rituximab-treated person with multiple sclerosis presenting with manifestations of an acute relapse. Hum Vaccin Immunother 18(1):2033540.https://doi.org/10.1080/21645515.2022.2033540
Liu P, Yan H, Li H, Zhang C, Li Y (2023) Overlapping anti-NMDAR encephalitis and multiple sclerosis: a case report and literature review. Front Immunol 14:1088801. Published 2023 Jan 30.https://doi.org/10.3389/fimmu.2023.1088801
Acknowledgements
We appreciate the EditorBar for language editing service and the patient participating in this study.
Funding
The language editing service cost is provided by the Major Science and Technology Application Demonstration Project of Chengdu Science and Technology Bureau, Sichuan Province, 2020(2019-YF09-00142-SN).
Author information
Authors and Affiliations
Contributions
BY drafted the manuscript; BY and NWY interpreted the data and edited the manuscript. All authors have read and approved the final manuscript and ensured that this is the case. And we all agreed to submit the manuscript to Neurological Sciences.
Corresponding author
Ethics declarations
Ethical approval
No ethics approval is needed for this case report after reviewed by the local Ethics Committee of Sichuan Provincial People’s Hospital.
Informed consent
Written informed consent was obtained from the patient for publication of this case report and any accompanying images. And the consent form is available for reviewing by the editor when needed.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Yang, B., Yu, N. Glucocorticoid-dependent multiple sclerosis overlapping anti-NMDA receptor encephalitis: a case report and literature review update. Neurol Sci 45, 83–92 (2024). https://doi.org/10.1007/s10072-023-07034-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10072-023-07034-x