Abstract
The purpose of this study was to clarify differences in skeletal morphologies between male and female orthodontic patients with and without agenesis of all four third molars. A total of 64 patients (32 males and 32 females) with agenesis of all four third molars without agenesis of other teeth were selected as the third molars agenesis group (group 1). In addition, 64 patients (32 males and 32 females) with all these teeth were selected as controls (group 2). Lateral cephalograms taken between the ages of 14 and 30 years were used to compare skeletal morphology between groups 1 and 2 and between sexes. Maxillary length (P < 0.001), lower facial height (P < 0.05), gonial angle (P < 0.001) and mandibular plane angle (P < 0.001) were significantly smaller in group 1 than in group 2. Irrespective of the presence or absence of all four third molars, males had significantly smaller lower facial height (P < 0.01) and mandibular plane angle (P < 0.001) and significantly greater total mandibular length (P < 0.001), mandibular body length (P < 0.001) and mandibular ramus height (P < 0.001) than females. Japanese orthodontic patients with agenesis of all four third molars had significantly small maxillary length, lower facial height, gonial angle and mandibular plane angle.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Agenesis of third molars (M3s) occurs most frequently in orthodontic populations [1, 2]. An abundance of the literature has reported a wide range in the prevalence rate of M3 agenesis in different populations [3,4,5], and most studies have indicated no significant differences in prevalence rates according to sex [2, 3, 5]. In Japanese populations, the prevalence of M3 agenesis has ranged from 22.2 [6] to 32.3% [2]. A recent study showed that the prevalence of subjects with of agenesis of all four M3s and without agenesis of other teeth was 4% of Japanese orthodontic patients [2].
Several studies have indicated significant associations between M3 agenesis and agenesis of other teeth, including maxillary lateral incisors and maxillary and mandibular second premolars [1, 2, 7,8,9,10,11]. Additionally, orthodontic subjects with M3 agenesis exhibit short maxillary length [12, 13] and mandibular length [14, 15], small gonial angle [16] and mandibular plane angle [14, 16, 17] and reduced lower facial height [14, 17]. Some studies have showed that orthodontic patients with skeletal Class II malocclusion have significantly lower prevalence of agenesis of M3s [6] and other teeth than M3s [18, 19] than those with skeletal Class III malocclusion, thus suggesting that skeletal pattern may have influence on tooth agenesis. In most of these studies on the associations of craniofacial morphology and agenesis of M3s, orthodontic patients were divided into groups according to the number or location of congenitally missing M3s [12, 13, 16, 17]. Woodworth et al. [14] and Tavajohi-Kermani et al. [15] selected orthodontic subjects with tooth agenesis without restriction to M3s. These studies did not assess the influence of sex differences on craniofacial morphology of patients with M3 agenesis [12,13,14,15,16,17].
To our knowledge, no studies have assessed in detail the skeletal morphology in orthodontic patients with agenesis of all this molar and without agenesis of other teeth. Therefore, the purpose of this study was to clarify skeletal morphologies in male and female orthodontic patients with agenesis of all four M3s and without agenesis of other teeth, relative to those patients with all these teeth.
Materials and methods
This study was approved by the Research Ethics Committee of our institution (Approval No. ECNG-R-306).
Sample
A total of 436 Japanese patients (163 males and 273 females) with and without M3 agenesis were retrospectively selected from orthodontic files of patients that had visited our institution between January 1994 and December 2015. Informed consent was obtained from all individual participants included in the study. The criteria for inclusion in this study were patients with availability of panoramic radiographs and lateral cephalograms taken between the ages of 14 and 30 years in the same day and with full eruption of all maxillary and mandibular teeth up to the second molars. The criteria for exclusion were patients with agenesis of other teeth than M3s, with congenital deformities, such as craniofacial syndrome or clefts, with history of extraction of a permanent tooth, and with history of orthodontic treatment prior to visiting our hospital. Patients with a Class I skeletal base had no lateral cephalograms taken when the orthopantomogram was taken if the orthodontist did not require it. These patients were immediately excluded from this study.
Among the 436 patients, 228 (102 males and 126 females) had agenesis of one or more M3s. The remaining 208 patients (61 males and 147 females) had all four M3s. A study subject of 74 patients (36 males and 38 females) without all four M3s was selected from 228 patients with agenesis of one or more M3s. One hundred fifty-four patients were excluded from the 228 patients initially selected from orthodontic patient files because they had M3 agenesis that did not fit the agenesis patterns in this study. Thirty-two males with a mean age of 18 years and 6 months (SD, 3 years and 4 months; range, 14 years 2 months–25 years 7 months) and 32 females with a mean age of 19 years and 1 month (SD, 3 years and 4 months; range, 14 years–25 years 9 months) were randomly selected from 74 patients without all four M3s as the M3 agenesis group (group 1). As the control group (group 2), 32 males with a mean age of 17 years and 10 months (SD, 2 years and 4 months; range, 14 years 4 months–23 years 2 months) and 32 females with a mean age of 17 years and 10 months (SD, 2 years and 4 months; range, 14 years 3 months–21 years 9 months) were randomly chosen from 208 patients with four M3s. To make the random selection, patients studied were coded and then selected in each group by a person who was not directly involved in this study.
Using G* Power version 3 (Heinrich Heine University, Dusseldorf, Germany), a post hoc power analysis was performed to determine the power of two-way analysis of variance (ANOVA) at an effect size of 0.25 (Cohen’s medium effect size), alpha error probability of 0.05, degree of freedom of 1, number of groups of 4 and a sample size of 128 [20]. The power was 0.80, thus demonstrating that the sample size in each group was sufficient for statistical comparisons.
Tooth agenesis
Panoramic radiographs were mainly used to examine tooth agenesis. All panoramic radiographs were taken with the same system (Veraview epocs X550, Morita Corporation, Kyoto, Japan). Tooth agenesis was diagnosed when there was no sign of crown mineralization on panoramic radiographs and no history of extraction of this tooth. A tooth was defined as not missing when more than 3/4 of its crown appeared to be mineralized on panoramic radiographs. If necessary, medical and dental records were examined to confirm any history of tooth extraction.
Cephalometric analysis
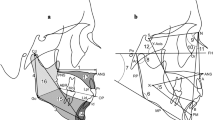
All cephalograms were taken using the same cephalostat (CX-150SK, Asahi roentgen, Kyoto, Japan) and with standardized settings. Cephalograms of each patient were coded by a person who was not directly involved in this study, and each was traced and measured. Sixteen reference points and four reference lines were selected, and four linear and eight angular measurements were taken to investigate the association between maxillofacial morphology and M3 agenesis (Fig. 1). Angular and linear measurements were made to the nearest 0.1° and 0.1 mm, respectively, with the aid of a computer system containing the WinCeph analysis software program (Rise Corp, Miyagi, Japan).
Reference points and lines used. S sella turcica, N nasion, Or orbitale, Po porion, Ar articulare, ANS anterior nasal spine, PNS posterior nasal spine, A point A, B point B, Pog pogonion, Gn gnathion, Me menton, Go gonion, Cd condylion, Xi Xi point, Pm protuberance menti, FH Frankfort horizontal plane, PP palatal plane, MP mandibular plane, RP ramus plane
To avoid any measurement bias, one investigator (YS), who was blinded to the groups, measured the coded cephalograms. The results of all measurements were subsequently sorted by group and sex for statistical comparison.
Statistical analysis
Statistical analyses were performed using a commercially available statistical package SPSS version 17.0 J (SPSS Japan Inc., Tokyo, Japan). Means and standard deviations (SDs) were calculated for each measurement within each group and each sex. Two-way ANOVA was performed to test the main effects of sex and group. P values < 0.05 were considered statistically significant.
Measurement error
To assess measurement errors, forty coded cephalograms were randomly selected and re-examined by the same investigator (YS) for a second time 3 months later. Student’s t test with a 95% confidence interval did not reveal any systematic errors. Random errors, calculated according to the Dahlberg formula [21], did not exceed ± 0.49° or ± 0.44 mm, which were unlikely to spoil the significant results in this study.
Results
Two-way ANOVA showed no significant differences in mean ages between the two groups or between sexes and no significant interactions between the two variables.
Table 1 shows the means and SDs for all measurements in the two groups.
As shown in Tables 2 and 3, two-way ANOVA demonstrated significant differences in the ANS–PNS dimension (maxillary length, P < 0.001, P < 0.001), the ANS–Xi–Pm angle (lower facial height, P < 0.05, P < 0.01) and the MP–FH angle (mandibular plane angle, P < 0.001, P < 0.001) between groups and between sexes, respectively, with no significant interactions between the two variables. Additionally, significant differences in the Cd–Gn dimension (total mandibular length, P < 0.001), the Go–Pog dimension (mandibular body length, P < 0.001) and the Cd–Go dimension (mandibular ramus height, P < 0.001) were observed between sexes (Table 2). A significant difference was also observed in the MP–RP angle (gonial angle, P < 0.001) between groups (Table 3). However, no significant differences were observed in the SNA angle (prognathism of maxillary alveolar bone), the SNB angle (mandibular alveolar bone), the RP–FH angle (ramus inclination), the ANB angle (sagittal jaw relationship angle) or the Y-axis angle (Y-axis) between groups or between sexes (Table 3).
Discussion
The panoramic radiographs taken between the ages of 14 and 30 years were used for diagnosis of tooth agenesis in this study. This minimum age of 14 years was based on the suggestion of Garn and Lewis [7] that the upper age limit for M3 agenesis is 14 years. Some studies have adopted a maximum age of 21 years [2, 22, 23]. The adaptation is based on the fact that eruption of M3s begins at 14–21 years of age [21]. However, Rakhshan [24] reported that there were no limits over the maximum age as long as positive extraction history is an exclusion criterion. Therefore, the maximum age of subjects in this study was 30 years.
Previous studies indicated that there were significant correlation coefficients between skeletal and dental maturity stages and that the second molars showed the highest correlation and the M3s showed the lowest relationship for both males and females [25, 26]. In this study, subjects with full eruption of all maxillary and mandibular teeth up to the second molars were selected and there were no significant differences in the mean ages between groups 1 and 2 or between sexes, as confirmed by two-way ANOVA. Therefore, subjects selected in this study were suitable for measuring skeletal morphology.
In this study, maxillary length, lower facial height, gonial angle and mandibular plane angle were significantly smaller in subjects without all four M3s (group 1, agenesis group) than in those with all four M3s (group 2, control group). These results were supported by previous studies that demonstrated the M3 agenesis group showed shorter maxillary length [12, 13] and decreased gonial angle [16], mandibular plane angle [16, 17] and lower facial height [17] than the control group. There were some discrepancies in sample selection between their agenesis groups and ours. Altan et al. [13] categorized their subjects with M3 agenesis into three groups: the bilateral maxillary M3 agenesis group, the bilateral mandibular M3 agenesis group and the all four M3 agenesis. Sanchez et al. [17] divided their subjects with M3 agenesis into two groups: the bilateral maxillary M3 agenesis group and the bilateral mandibular M3 agenesis group. Kajii et al. [12] also classified their subjects into two maxillary and mandibular agenesis groups, irrespective of unilateral or bilateral M3 agenesis. Moreover, Ramiro-Verdugo et al. [16] selected subjects with agenesis of at least one M3. Woodworth et al. [14] and Tavajohi-Kermani et al. [15] verified short maxillary length in subjects with agenesis of maxillary M3s and other teeth, as confirmed by the results of the present study.
Reduced maxillary length was considered to be due to inadequate apposition of bone to the tuberosity area in our M3 agenesis group. This consideration is based on a report that increased maxillary length was accomplished almost completely by apposition of bone to the maxillary tuberosity, which was associated with tooth eruption [27]. Decreased gonial angle, mandibular plane angle and lower facial height in the M3 agenesis group were presumably related to upward rotation of the mandible as a result of reduced vertical dimension of the alveolar process. This upward rotation of the mandible in the M3 agenesis group may be due to the fact that the vertical growth at the condyle was superior to the sum of the vertical growth components at facial sutures and alveolar processes in the subjects with agenesis of posterior teeth [28].
Our results demonstrated that there were no significant differences in the total mandibular length, mandibular body length, or mandibular ramus height between the subjects with and without agenesis of all four M3s. These results were consistent with the findings by Ades et al. [29] and Kaplan [30], who reported that mandibular growth pattern was no significantly different between the subject with and without M3, thus indicating that agenesis of all four M3s might not be associated with the mandibular length. Conversely, Altan et al. [13] reported that the total mandibular length was significantly smaller in patients with agenesis of all for M3s than in those with all four M3s.
From a genetic point of view, Msx1 over-expression during bone development affected craniofacial morphology and a deficiency of this expression resulted in a switch from a dolichofacial pattern to a mesiofacial pattern or brachyfacial pattern [31, 32]. Moreover, Pax9 deficiency was responsible for skeletal deficiency and agenesis of M3s [32, 33]. These pieces of evidence of Msx1 and PAX9 were in accord with our findings that skeletal morphological deviations occurred in the agenesis group.
Our results showed that irrespective of the presence or absence of all four M3s, lower facial height and mandibular plane angle were significantly smaller in males than in females, demonstrating that males had a greater tendency toward a brachyfacial pattern than females. Males have thicker masseter muscle and stronger occlusal force than females [34, 35], and subjects with strong occlusal force presented with a brachyfacial pattern [36, 37], as observed in our male subjects. Therefore, differences in the tested measurements between sexes observed in this study may be caused by the discrepancy in occlusal force generated by the masseter muscle. Conversely, Wu et al. [38] and Gu et al. [39] observed that males had larger lower facial height and increased mandibular plane angle compared with females.
In this study, total mandibular length, mandibular body length and mandibular ramus height were significantly shorter in females than in males, irrespective of the presence or absence of all four M3s. These results were consistent with findings of Gu et al. [39] and Daraze et al. [40] who reported that these mandibular dimensions were significantly shorter in females than in males. Growth at the condylar cartilage thrusts the mandible forward and downward, thus resulting in an increase in total mandibular length. Moreover, growth of the mandible by resorption along the anterior edge of the ramus and apposition along its posterior edge increases mandibular body length and mandibular ramus height [41]. Moreover, Franchi et al. [42] showed that both males and females experienced pubertal growth spurts in linear dimensions of the mandible. Mellion et al. [43] described that skeletal growth changes in the face of females slow and cease shortly after puberty; however, dimensional changes in males continue through the late adolescent period. Accordingly, sex differences in mandibular dimensions observed in this study were thought to be due to discrepancies in the mechanism of mandibular growth between sexes.
We can summarize our conclusions as follows:
-
Maxillary length, lower facial height, gonial angle and mandibular plane angle were significantly smaller in orthodontic subjects without all four M3s and with other teeth than in those with all these teeth.
-
Irrespective of the presence or absence of all four M3s, males had significantly smaller lower facial height and mandibular plane angle and significantly greater total mandibular length, mandibular body length and mandibular ramus height than females.
References
Celikoglu M, Bayram M, Nur M. Patterns of third-molar agenesis and associated dental anomalies in an orthodontic population. Am J Orthod Dentofacial Orthop. 2011;140:856–60.
Endo S, Sanpei S, Ishida R, Sanpei S, Abe R, Endo T. Association between third molar agenesis patterns and agenesis of other teeth in a Japanese orthodontic population. Odontology. 2015;103:89–96.
Hattab FN, Rawashdeh MA, Fahmy MS. Impaction status of third molars in Jordanian students. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 1995;79:24–9.
Celikoglu M, Miloglu O, Kazanci F. Frequency of agenesis, impaction, angulation, and related pathologic changes of third molar teeth in orthodontic patients. J Oral Maxillofac Surg. 2010;68:990–5.
Mok YY, Ho KK. Congenitally absent third molars in 12 to 16 year old Singaporean Chinese patients: a retrospective radiographic study. Ann Acad Med Singapore. 1996;25:828–30.
Kajii T, Imai T, Kajii S, Iida J. Presence of third molar germs in orthodontic patients in Japan. Am J Orthod Dentofacial Orthop. 2001;119:245–50.
Garn SM, Lewis AB. The relationship between third molar agenesis and reduction in tooth number. Angle Orthod. 1962;32:14–8.
Garib DG, Peck S, Gomes SC. Increased occurrence of dental anomalies associated with second-premolar agenesis. Angle Orthod. 2009;79:436–41.
Garib DG, Alencar BM, Lauris JRP, Baccetti T. Agenesis of maxillary lateral incisors and associated dental anomalies. Am J Orthod Dentofacial Orthop. 2010;137:732.e1–6.
Endo T, Sanpei S, Komatsuzaki A, Endo S, Takakuwa A, Oka K. Patterns of tooth agenesis in Japanese subjects with bilateral agenesis of mandibular second premolars. Odontology. 2013;101:216–21.
Sanpei S, Ishida R, Sanpei S, Endo S, Tanaka S, Endo T, et al. Patterns of bilateral agenesis of maxillary third molars and agenesis of other teeth. Odontology. 2016;104:98–104.
Kajii TS, Sato Y, Kajii S, Sugawara Y, Iida J. Agenesis of third molar germs depends on sagittal maxillary jaw dimensions in orthodontic patients in Japan. Angle Orthod. 2004;74:337–42.
Altan AB, Sinanoglu EA, Ucdemir E, Sandalci S, Karaman AI. Dentofacial morphology in third molar agenesis. Turkish J Orthod. 2015;28:7–12.
Woodworth DA, Sinclair PM, Alexander RG. Bilateral congenital absence of maxillary lateral incisors: a craniofacial and dental cast analysis. Am J Orthod. 1985;87:280–93.
Tavajohi-Kermani H, Kapur R, Sciote JJ. Tooth agenesis and craniofacial morphology in an orthodontic population. Am J Orthod Dentofacial Orthop. 2002;122:39–47.
Ramiro-Verdugo J, De Vicente-Corominas E, Montiel-Company JM, Gandía-Franco JL, Bellot-Arcís C. Association between third molar agenesis and craniofacial structure development. Am J Orthod Dentofacial Orthop. 2015;148:799–804.
Sánchez MJ, Vicente A, Bravo LA. Third molar agenesis and craniofacial morphology. Angle Orthod. 2009;79:473–8.
Chung CJ, Han JH, Kim KH. The pattern and prevalence of hypodontia in Koreans. Oral Dis. 2008;14:620–5.
Celikoglu M, Kazanci F, Miloglu O, Oztek O, Kamak H, Ceylan I. Frequency and characteristics of tooth agenesis among an orthodontic patient population. Med Oral Patol Oral Cir Bucal. 2010;15:797–801.
Faul F, Erdfelder E, Lang AG, Buchner A. G*Power 3: a flexible statistical power analysis program for the social, behavioral, and biomedical sciences. Behav Res Methods. 2007;39:175–91.
Dahlberg G. Statistical methods for medical and biological students. New York: Interscience Publications; 1940.
Otuyemi OD, Ugboko VI, Ndukwe KC, Adekoya-Sofowora CA. Eruption times of third molars in young rural Nigerians. Int Dent J. 1997;47:266–70.
Hirakata C, Sanpei S, Sanpei S, Tanaka S, Endo T, Sekimoto T. Symmetrical agenesis of the mandibular third molars and agenesis of other teeth in a Japanese orthodontic population. J Oral Sci. 2016;58:171–6.
Rakhshan V, Rakhshan A. Systematic review and meta-analysis of congenitally missing permanent dentition: sex dimorphism, occurrence patterns, associated factors and biasing factors. Int Orthod. 2016;14:273–94.
Uysal T, Sari Z, Ramoglu SI, Basciftci FA. Relationships between dental and skeletal maturity in Turkish subjects. Angle Orthod. 2004;74:657–64.
Krailassiri S, Anuwongnukroh N, Dechkunakorn S. Relationships between dental calcification stages and skeletal maturity indicators in Thai individuals. Angle Orthod. 2002;72:155–66.
Vucic S, Dhamo B, Kuijpers MA, Jaddoe VW, Hofman A, Wolvius EB, Ongkosuwito EM. Craniofacial characteristics of children with mild hypodontia. Am J Orthod Dentofacial Orthop. 2016;150:611–9.
Endo T, Yoshino S, Ozoe R, Kojima K, Shimooka S. Association of advanced hypodontia and craniofacial morphology in Japanese orthodontic patients. Odontology. 2004;92:48–53.
Ades AG, Joondeph DR, Little RM, Chapko MK. A long-term study of the relationship of third molars to changes in the mandibular dental arch. Am J Orthod Dentofacial Orthop. 1990;97:323–35.
Kaplan RG. Mandibular third molars and postretention crowding. Am J Orthod. 1974;66:411–30.
Nassif A, Senussi I, Meary F, Loiodice S, Hotton D, Robert B, Bensidhoum M, Berdal A, Babajko S. Msx1 role in craniofacial bone morphogenesis. Bone. 2014;66:96–104.
Cocos A, Halazonetis DJ. Craniofacial shape differs in patients with tooth agenesis: geometric morphometric analysis. Eur J Orthod. 2016;39(4):345–51.
Peters H, Neubüser A, Kratochwil K, Balling R. Pax9-deficient mice lack pharyngeal pouch derivatives and teeth and exhibit craniofacial and limb abnormalities. Genes Dev. 1998;12:2735–47.
Charalampidou M, Kjellberg H, Georgiakaki I, Kiliaridis S. Masseter muscle thickness and mechanical advantage in relation to vertical craniofacial morphology in children. Acta Odontol Scand. 2008;66:23–30.
Roldán SI, Restrepo LG, Isaza JF, Vélez LG, Buschang PH. Are maximum bite forces of subjects 7 to 17 years of age related to malocclusion? Angle Orthod. 2016;86:456–61.
Kitafusa Y. Application of “prescale” as an aid to clinical diagnosis in orthodontics. Bull Tokyo Dent Coll. 2004;45:99–108.
Custoid W, Gomes SG, Faot F, Garcia RC, Del Bel Cury AA. Occlusal force, electromyographic activity of masticatory muscles and mandibular flexure of subjects with different facial types. J Appl Oral Sci. 2011;19:343–9.
Wu J, Hägg U, Rabie AB. Chinese norms of McNamara’s cephalometric analysis. Angle Orthod. 2007;77:12–20.
Gu Y, McNamara JA Jr, Sigler LM, Baccetti T. Comparison of craniofacial characteristics of typical Chinese and Caucasian young adults. Eur J Orthod. 2011;33:205–11.
Daraze A, Delatte M, Bou Saba S, Majzoub Z. Craniofacial characteristics in the sagittal dimension: a cephalometric study in Lebanese young adults. Int Orthod. 2017;15:114–30.
Duthie J, Bharwani D, Tallents RH, Bellohusen R, Fishman L. A longitudinal study of normal asymmetric mandibular growth and its relationship to skeletal maturation. Am J Orthod Dentofacial Orthop. 2007;132:179–84.
Franchi L, Baccetti T, McNamara JA Jr. Thin-plate spline analysis of mandibular growth. Angle Orthod. 2001;71:83–9.
Mellion ZJ, Behrents RG, Johnston LE Jr. The pattern of facial skeletal growth and its relationship to various common indexes of maturation. Am J Orthod Dentofacial Orthop. 2013;143:845–54.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Sugiki, Y., Kobayashi, Y., Uozu, M. et al. Association between skeletal morphology and agenesis of all four third molars in Japanese orthodontic patients. Odontology 106, 282–288 (2018). https://doi.org/10.1007/s10266-017-0336-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10266-017-0336-z