Abstract
How drought affects tree and stand growth is an old question, but is getting unprecedented relevance in view of climate change. Stress effects related to drought have been mostly studied at the individual tree level, mostly investigating dominant trees and using their responses as indicator for the impact at the stand level. However, findings at tree and stand level may differ, as the stand responses include interactions and feedbacks that may buffer or aggravate what is observed at the individual tree level. Here, we trace drought effects on growth and development from tree to the stand scale. Therefore, we analyse annually measured data from long-term experiments in temperate and Mediterranean forests. With this analysis, we aim to disclose how well results of dominant tree growth reflect stand-level behaviour, hypothesizing that drought resistance of dominant trees’ can strongly deviate from the overall sensitivity of the stand. First, we theoretically derive how drought responses at the stand level emerge from the tree-level behaviour, thereby considering that potential drought resistance of individual trees is modulated by acclimation and tree–tree interactions at the stand level and that the overall stress response at the stand level results from species-specific and size-dependent individual tree growth and mortality. Second, reviewing respective peer-reviewed literature (24 papers) and complementing findings by own measurements (22 experiments) from temperate and Mediterranean monospecific and mixed-species forests, we are able to reveal main causes for deviations of tree-level and stand-level findings regarding drought stress responses. Using a long-term experiment in Norway spruce (Picea abies (L.) KARST.) and European beech (Fagus sylvatica L.), we provide evidence that the species-dependent and size-dependent reactions matter and how the size–frequency distribution affects the scaling. We show by examples that tree-level derived results may overestimate growth losses by 25%. Third, we investigate the development of the growth dominance coefficient based on measurements gathered at the Bavarian forest climate stations. We show that drought changes stand biomass partitioning in favour of small trees, reduce social differentiation, and homogenize the vertical structure of forests. Finally, we discuss the drought-related consequences of the social class-specific growth reaction patterns for inventory and monitoring and highlight the importance of these findings for understanding site-specific stand dynamics, for forest modelling, and for silvicultural management.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction: social status and drought stress response
How tree growth and the underlying resources are partitioned between trees of different social positions and how the partitioning is modulated by drought are essential for monitoring tree growth and vitality, for the understanding and modelling stand dynamics, and for climate smart forest management (Bowditch et al. 2020). Nevertheless, monitoring of growth and vitality is often based on observations of dominant trees only (Dobbertin 2005). Their growth reaction is thus used as an indicator for the stress at the stand level. However, responses at the tree compared to the stand level may differ, because various feedback processes may buffer or aggravate the development at the individual tree level. For instance, growth of predominant trees may be more sensitive to drought and thus is decreasing more strongly than the majority of subdominant or suppressed trees in a stand (Grote et al. 2016). Not considering such effects has been shown to potentially result in an overestimation of growth losses (Pretzsch et al. 2018a, b; Zang et al. 2012).
For ecosystem management and planning, the vitality, stability, and resilience of a stand as a whole are of primary interest, whereas tree-level traits and responses provide the means for understanding and explaining stand-level reaction patterns (Bauhus et al. 2017; Ulrich 1983). Differences in tree-level responses—or individual growth—can be characterized by the growth partitioning between small and big trees. Heat and drought may, for instance, reduce the growth of exposed tall trees in relation to small trees and lead to a more equal size distribution and a more homogeneous vertical structure of the stand (Grote et al. 2016). Such a general pattern is not necessarily true in specific cases, where precipitation intensity which determines the water distribution in the root profile, or the species-specific size and distribution of fine roots may be important. However, it indicates that water availability may be the prevailing factor not only for individual growth but also for stand structural development (Olivar et al. 2015). In contrast, it can be expected that ample water and light supply will result in a more heterogeneous vertical structure because tall trees can then profit from their advantage in light competition without the negative feedback of limited water resources (Pretzsch and Biber 2010; Wichmann 2001; Schwinning and Weiner 1998). The importance of individual tree size and competition on growth, particularly during drought years, has been demonstrated empirically (Gleason et al. 2017; Ding et al. 2017) as well as in model simulations that considered resource acquisition and stress at the individual level (Holtmann et al. 2021).
Consequently, silvicultural design should consider the prevailing resource and growth-partitioning pattern. Assuming that drought disproportionately affects growth and vitality of tall trees, the selection of future crop trees among the predominant trees may become questionable. In such cases, small trees may counterbalance growth losses of tall neighbours (Steckel et al. 2020a, b; Pretzsch et al. 2018a, b), or the elimination of small trees may cause a density reduction that is beneficial for the remaining tall trees and future crop trees (Pretzsch 2021a, b, c; Gebhardt et al. 2014; Aussenac 2000). The particular best measures may be site- and species-specific, highlighting the importance of further research about this topic. In addition, it may be important to differentiate between drought events of different timing, intensity, and frequency (Mérian and Lebourgeois 2011; Clark et al. 2016).
Drought responses are mostly investigated at dominant or dominant and suppressed trees in monospecific stands (e.g. Eilmann and Rigling 2012, Bogino and Bravo 2008, Martín Benito et al. 2008). Other studies investigate the size dependency of drought reactions in size classes (Stephenson and Das 2020) or in mixed-species stands (Pretzsch et al. 2013; Teets et al. 2018). Hardly any study evaluates the impact at stand level, even when comparing different social classes. This is particularly true for the most severe impact which is provoking tree death (e.g. Archambue et al. 2020) as it has been rarely recorded on an annual basis and is also difficult to assign to a specific cause (Trugman et al. 2021; Stephenson et al. 2019). However, since an increased drought-related mortality has been reported for many regions worldwide (e.g. Allen et al. 2010; Zhang et al. 2017; Obladen et al. 2021), also the importance of tree size and forest structure is increasingly highlighted (Bennett et al. 2015, Colangelo 2017, Venturas et al. 2021).
Scaling from individual tree drought responses to growth reactions at the stand level requires a multiplication of the size-related reaction patterns (growth, mortality) by the frequency of trees per size class or social class. Thus, for upscaling to the stand level, the size dependency of stress reactions and frequency distribution of the trees are indispensable. As latter information is missing in most studies, level-overarching insight about stand-level behaviour emerging from tree-level reactions is still poor (Strigul 2012; Binkley et al. 2002; Cermák et al. 1998). Therefore, we here trace drought effects from the tree to the stand growth, using a mini-review (24 studies) and own findings (22 experiments) in temperate and Mediterranean mono- and mixed-species forests. The papers considered covered tree species worldwide and the experiments include the most ecologically and economically relevant tree species in Europe. Own findings were derived from experimental plots in temperate mono- and mixed-species stands of Norway spruce (Picea abies (L.) H. Karst.), European beech (Fagus sylvatica L.), Sessile oak (Quercus petraea (Matt.) Liebl.), Scots pine (Pinus sylvestris L.), silver fir (Abies alba Mill.), European larch (Larix decidua Mill.), European ash (Fraxinus excelsior L.), Maritime pine (Pinus pinaster Ait.), Scots pine (Pinus sylvestris L.), and Pyrenean oak (Quercus pyrenaica Will.).
In general, we hypothesize that dominant tree drought responses can strongly deviate from stand-level responses. More specifically, our objectives are to derive how the stand-level drought responses emerge from the tree-level behaviour and, in particular, how size–frequency distribution and mortality affect this scaling. We hypothesize that in this context, species- and size-dependent reactions matter, and that drought may change the growth partitioning in favour of the small trees, reduce the social inequality, and homogenize the size and vertical structure of forests. Finally, we will discuss the consequences of considering the size-specific growth reaction patterns for forest mensuration, ecology, and management.
Methodology: theoretical considerations
Quantifying drought resistance of individual trees
Figure 1 illustrates the three possible response patterns of growth in individual trees to drought. Suppose a period (ref period) of normal growing conditions is followed by a dry period (drought year). In this case, the drought resistance (res) in terms of growth (\(i_{{{\text{drought}}}}\)) in the drought period may be divided by the growth (\(i_{{{\text{ref}}}}\)) in the reference period (\({\text{res}} = i_{{{\text{drought}}}} /i_{{{\text{ref}}}}\)). In principle, the growth in the drought period may be (Fig. 1a) reduced, (1b) neutral, or (1c) increased due to the drought. The resistance ratio res may be used to analyse any relationship between drought-related growth reactions and tree traits such as species, size, and competition.
Courses of annual tree growth with a negative, b neutral, and c positive response of tree growth in a drought year (dotted line) compared with the growth in a preceding period of normal growth (grey area). The ratio of annual growth within the drought year to that during the reference period \({\text{res}} = i_{{{\text{drought}}}} /i_{{{\text{ref}}}}\) indicates the growth resistance and is suitable for analysing any size dependencies of the trees’ growth reactions (see Fig. 2)
Suppose that the stem basal area growth in the extreme drought year 2003 divided by the mean stem basal area growth in the preceding normal growth period 2000–2002 results in a ratio \({\text{res}} = {\text{iba}}_{{{\text{drought}}}} /{\text{iba}}_{{{\text{ref}}}} = 0.80\). Then, the resistance to drought is 0.80; that is, in the year 2003 the growth is only 80% of the normal growth and the growth loss is 20%. The response ratio res has been widely used to quantify tree growth reactions during dry periods (e.g. Thurm et al. 2016; Pretzsch et al. 2013; Lloret et al. 2011).
Characterizing drought responses of different tree sizes or social positions
To analyse drought-related growth responses of individual tree properties, basal area is used as a substitute for tree size or social position within the stand. Figure 2 shows three principle relationships between res and tree size, assuming a linear relationship; later, we will see that the relationships between resistance and size can also be nonlinear.
Growth resistance to drought (relative tree growth \({\text{res}}_{{i{\text{drought}}/i{\text{ref}}}}\) in dry compared to normal reference years) in dependence on the social position of trees within a stand. The three main reaction patterns are visualized by a decreasing, b constant, and c increasing \({\text{res}}_{{i{\text{drought}}/i{\text{ref}}}}\) values with increasing social rank of individual trees. The horizontal line (1.0-line represents, size independence) represents parity of growth in dry compared with reference years
In the first case (Fig. 2a), small trees hardly suffer or even benefit by drought, but growth losses increase with increasing dominance and size of the tree. In the second case (2b), all trees have the same resistance and growth loss, while in the third case (2c) small trees suffer the most and losses are smaller for dominant trees. The actual relationships may be derived by regression analyses; the statistical characteristics may be used for the revelation of significant decreasing or increasing patterns or constant growth–size reaction patterns, respectively.
Growth resistance to drought can be calculated at the tree and the stand level, at the species or social class level. In this paper, we used capital RES for the resistance at the stand level and res for the resistance at the tree level. Depending on the available data, we calculate the resistance at tree and stand level based on the basal area (iba, IBA) or stem volume growth (iv, IV) at the tree or stand level, respectively (\({\text{res}}_{{{\text{iba}}}}\), \({\text{res}}_{{{\text{iv}}}}\), or \({\text{RES}}_{{{\text{IBA}}}}\), \({\text{RES}}_{{{\text{IV}}}}\)). Any size dependency will then be revealed by the model \(\ln \left( {{\text{res}}} \right) = a_{0} + a_{1} \times \ln \left( {ba} \right)\) using individual tree basal area as a substitute for tree size or social position. The coefficient a1 of the model will indicate whether the growth resistance is invariant to tree size (\(a_{1} = 0\)), increases (\(a_{1} > 0\)), or decreases (\(a_{1} < 0\)) with the social position of the tree.
Characterizing individual size-related tree mortality due to drought
Mortality can be interpreted as an ultimate growth reduction caused by drought. It may preferentially affect small trees (Fig. 3a), may be size-invariant (3b), or preferentially eliminate larger trees (3c). To quantify the mode of mortality, the arithmetic mean stem diameter of trees that have died (\(\overline{d}_{{{\text{mort}}}}\)) can be divided by the initial mean stem diameter of the whole population (\(\overline{d}_{{{\text{total}}}}\)). The ratio \({\text{modemort}} = \overline{d}_{{{\text{mort}}}} /\overline{d}_{{{\text{total}}}}\) indicates the mode of mortality. In case of \({\text{modemort}} < 1\), the mortality is concentrated on small trees, in case of \({\text{modemort}} = 1\), the mortality is invariant, and \({\text{modemort}} > 1\) would indicate an increased drop out of tall trees. For the sake of completeness, we address how to consider mortality when upscaling to the stand level; however, in the empirical part of this review (“Empirical evidence” section) the database was too small to apply these characteristics.
Different size-related mortality. The mortality may be a concentrated on small trees (\({\text{modemort}} < 1\)), b similar for trees of all sizes (\({\text{modemort}} = 1\)), or c concentrated on large trees (\({\text{modemort}} > 1\)). The hatched area indicates the tree number reduction by drought-induced mortality
Relevance of tree size–frequency distribution and mortality
Figure 4a–c schematically shows the three main components for scaling up the drought reaction from tree to stand growth. We already introduced the size-related growth reaction at the individual tree level that can be derived from the annual growth rates (Fig. 4a) in normal and dry years to calculate the drought resistance and relate it to tree size (Fig. 4b). In addition, upscaling requires information about the size–frequency distribution of the remaining and the removed (due to silvicultural management or natural mortality) trees (Fig. 4c). Stand growth then results as the product of individual tree growth and tree number per size class (Fig. 4d).
Upscaling the effect of drought from the tree to the stand level. Analyses at the individual tree level show a the dependency of tree growth in normal and dry years on tree size, and b the ratio between tree growth in dry and normal years. Upscaling furthermore requires information on c tree number over tree size classes for remaining and dead trees. The product of reaction patterns and their frequency in size classes finally results in d the stand growth over tree size for normal and dry conditions
We use the schematic representation in Fig. 4 to explain the relationships. In this example, small trees suffer less than large trees and some even benefit from the drought (Fig. 4a, b). As small trees are mostly much more frequent in the stand than tall trees (Fig. 4c), they contribute to a larger degree to stand basal area growth and can thus compensate growth losses of larger trees to a certain degree (Fig. 4d). The black line in Fig. 4d shows the stand growth plotted over basal area in normal years and the red line the same relationship in drought years. The hatched area between the two curves represents the size-dependent gains (in low size classes) and losses (in high size classes) of stand growth. The presented example demonstrates that the frequency of tree size classes (quantity) can potentially matter when scaling the resistance (quality of the growth reaction) from tree to stand level.
Drought impact on growth distribution among the trees within forest stands
The pattern of growth resistance in dependence of tree size does not consider the size class frequency and therefore the growth partitioning within the stand (Fig. 4). In order to analyse whether drought modifies the stand growth allocation between small and tall trees, we applied the growth dominance coefficient, GDC (Binkley et al. 2002). The GDC is based on the Gini coefficient for cumulative growth of trees and the Gini coefficient for cumulative stock of trees, which both are generally calculated as follows (see de Camino 1976; Kramer 1988, p 82):
Variables xi and xj denote size or growth (or other tree characteristics) for the ith and the jth tree in the stand with i and j = 1…n trees. Here, we calculate both the Gini coefficients of the cumulative growth of trees (\({\text{GC}}_{{{\text{growth}}}}\)) and the Gini coefficients of the cumulative stock of trees (\({\text{GC}}_{{{\text{stock}}}}\)) based on individual tree basal areas and derive the growth dominance coefficient as \({\text{GDC}} = {\text{GC}}_{{{\text{growth}} - {\text{GC}}_{{{\text{stock}}}} }}\). It also can be calculated directly based on the individual tree records of stem basal area and stem basal area growth of all trees of a population sorted by size as:
The Gini coefficients of cumulative tree basal area (\({\text{GC}}_{{{\text{stock}}}}\)) and basal area growth (\({\text{GC}}_{{{\text{growth}}}}\)) can be visualized by plotting cumulative tree basal area or basal area growth, respectively, over cumulative tree number (Fig. 5a, b). The growth dominance coefficient can be visualized by the cumulative distribution of tree basal area growth over tree basal area, e.g. tree basal area growth over initial tree basal area (Fig. 5c). For this purpose, all trees of a stand are ranked from smallest to largest basal area, the cumulative basal area of the trees is registered on the abscissa, and their cumulative basal area growth is registered on the ordinate. The resulting curves illustrate how tree size distribution contribute to total stand growth. The lines in Fig. 5c indicate a growth dominance of tall trees (lower curve, GDC > 0), small trees (upper curve, GDC < 0), or a proportional contribution of growth according to their size (straight line, GDC = 0) (Binkley et al. 2006).
Schematic representation of the Gini coefficients based on basal area, GCba, GCiba, and the growth dominance coefficient, GDC, for quantifying the growth distribution among the trees in dry and normal growth years. a The Gini coefficient of tree basal area distribution, GCba, indicates the degree of size equality of trees in a forest stand, b the Gini coefficient of tree basal area growth distribution, GCiba, indicates the degree of size growth equality of the trees in a forest stand, c the growth dominance coefficient, GDC, indicates the relative contribution of small compared with tall trees to stand growth. In all cases, the bisectoral line (broken) represents equality of size, growth, and growth contribution in relation to size
Any differences of the GDC in dry compared with normal years indicate a change of the growth-partitioning pattern between differently sized trees. A constant GDC in dry and normal years would indicate that drought affected the growth of all trees in the population (small and large ones) equally (although the absolute growth rate of the stand may have changed). An increase in the GDC (e.g. a shift from the upper to the lower line in Fig. 5c) would mean that large trees increase their share of stand basal area growth. In contrast, a decreasing GDC (e.g. a shift from the lower to the upper line in Fig. 5c) would indicate an increasing share of the small trees. The consequences of such a shift of growth allocation will be shown by example in “Climate impact on the contribution of different tree size classes to stand growth” section and discussed in “Observed patterns and underlying processes” section.
Data requirement
Insight into the size-dependent growth reactions on drought years requires annual growth records, which are often not available from long-term experiments due to their measurement intervals of 3–10-years (Johann 1993; Prodan 1965). In addition, annual measurements of stem diameter by calliper or circumference tape are of limited use due to their low accuracy (Zöhrer 1980, Loetsch and Zöhrer 1973). Retrospective annual growth detection based on increment cores may be a makeshift to get annual data (Heym et al. 2018, 2017). However, such retrospective measurements have the disadvantages that mortality and thinning in the past may be unknown, and measurements refer in most cases to a (biased) subset of trees only (e.g. dominant tress or sample trees with the quadratic mean diameter). Exceptions may be increment corings on long-term experiments from which thinnings and mortalities are known. A very useful measurement setup for such analyses is to fully equip a whole stand with permanent band dendrometers with readings taken at least annually over longer time periods. If such stands are under long-term observation, they also provide information about tree heights, mortality, etc. In this study, we used measurements with permanent band dendrometer from plots in fully stocked monospecific and mixed-species temperate forests in Germany as well as Mediterranean forests in Spain. We included only fully stocked stands in order not to confuse drought effects with stand density effects (e.g. spacing, thinning, regeneration cuts).
Empirical evidence
Mini-review of size-dependent growth reactions
We used the keywords “drought effects”, “low-growth years”, “growth partitioning”, “social rank”, “upscaling from tree to stand”, “sampling bias”, “buffering of growth losses” for searching in Web of Science, Google Scholar, and Scopus. We also excluded all investigations that did not provide sufficient information about tree dimensional distribution or did not investigate drought responses. Very helpful for getting a wide overview of this topic was the international authoring group with special expertise covering boreal, temperate, and Mediterranean forest research as well as the respective relevant publications. Table 1 summarizes the remaining 24 studies that provide all required information. These studies cover many different commercial tree species and climate conditions from tropical to boreal. Most of them refer to fully stocked even-aged monospecific stands. Studies in mixed stands evaluated the resistance–size relationship for each species separately.
In general, the objective of the majority of studies was to analyse the drought resistance of dominant versus suppressed trees, others sample systematically over all size classes, while some studies compare different social classes, but without covering the full dimensional range (e.g. dominant and intermediate). Only few studies performed a complete inventory; however, none of these have analysed the growth decline due to drought at both the tree (analysing social classes) and the stand level.
We found several studies on the relationship between mortality and drought. These were not considered, however, since they were not consequently combined with growth analyses or do not provide inventory information about the size–frequency distribution, necessary for scaling from the tree to the stand level. We found only one study that analyses growth resistance to drought at tree as well as stand level (Bottero et al 2021), but it is not included in Table 1 as it does not compare tree growth reaction by social classes.
The majority of the studies (17 out of 24) indicate a decrease in the drought resistance (higher drought susceptibility) with increasing tree size and dominance within forest stands. Shade-tolerant tree genera such as Fagus, Abies, or Picea nearly always showed a significant decrease in the resistance with increasing size, whereas light-demanding genera such as Pinus, Larix, or Quercus showed more often size-invariant drought resistance (although there are also few examples showing opposite findings).
Own studies on size-related growth reactions in temperate forest stands
For analysing the drought effects in temperate forests, we used the long-term observational plots of the forest climate stations (Intensive Forest Monitoring, Level II) in Bavaria (Ferretti and Fischer 2013) that cover the most relevant tree species in Central Europe. In addition, we used the mono- and mixed-species plots of the long-term experiment in Norway spruce and European beech Freising 813/1 (Grams et al. 2021). According to the different geological situations in Bavaria, the soil types at the experimental plots differ slightly. Dominant soil type in main root layer in most cases is loam as listed in Supplementary Table 1. The site conditions at all included observation sites are representative for the respective growth region. All included plots are fully equipped with band dendrometers that record long-term developments of circumferences and are continuously recording climatic boundary conditions. Supplementary Table 1 gives an overview of the main characteristics of the included long-term experimental plots. Supplementary Tables 2 and 3 show the characteristics of all statistical analyses regarding the relationship between tree size and drought resistance on all included experimental plots in temperate and Mediterranean climate.
We used the long-term observational plot Flossenbuerg in monospecific and even-aged Norway spruce stand as model example for analysing the relationship between drought stress reaction and tree site. The plot Flossenbuerg in Norway spruce belongs to the network of long-term forest climate stations, is fully equipped with band dendrometers that were read off since 1996. The location is N 49° 45′ 40'', E 12° 23'', stand age 100 years, plot size 0.26 ha, stand basal area 38.8 m2 ha−1 and the standing stem volume 515,6 m3 ha−1. We chose the drought years 2003 and 2018 (see Fig. 6). The stem diameter range was rather wide (15.6–61.5 cm in 2003 and 20.5–58.7 cm in 2018) and underpins the suitability of this plot for revelation of any relationship between drought reaction and tree size. For more stand characteristics, see Supplementary Table 1.
Drought reaction of the annual basal growth on the monospecific Norway spruce observational plot Flossenbuerg, South Germany. Annual growth records based on band dendrometers (data from 1997 to 2020; n = 117 and 77 sample trees available for the analysis in 2003 and 2018, respectively). a Absolute annual stem basal area growth of individual trees. b Growth resistance in 2003 based on the ratio between basal area growth in 2003 and in the preceding two years. c, d Resistance in 2003 and 2018 plotted over stem basal area at the beginning of the growth periods and respective regression lines based on the model \(\ln \left( {{\text{res}}} \right) = a_{0} + a_{1} \times \ln \left( {ba} \right)\). The two vertical broken lines indicate the drought years 2003 and 2018 (left and right, respectively), and the horizontal black lines indicate the reference periods (for statistical characteristics, see Supplementary Table 2)
The long-term observational plot Flossenbuerg thus provides evidence for a size-dependent growth reaction of Norway spruce on the extreme drought events in 2003 and 2018. Figure 6a shows the plunges of annual tree basal area growth in 2003 and 2018. We chose the proceeding periods 2000–2002 and 2016–2017 for calculation of the growth resistance \({\text{res}}_{2003}\) and \({\text{res}}_{2018}\), respectively; in case of 2018, we chose a 2-years reference period to exclude the drought year 2015 from the reference period. Figure 6b shows the \({\text{res}}_{2003}\) values resulting as ratio between annual growth in 2003 and the average growth in the period 2000–2002; analogously we calculated the \({\text{res}}_{2018}\) values (not shown). Figures 6c, d present the \({\text{res}}_{2003}\) and \({\text{res}}_{2018}\) values plotted over stem basal area at the beginning of the respective growth periods, and the resulting regression curves based on the model \(\ln \left( {{\text{res}}} \right) = a_{0} + a_{1} \times \ln \left( {ba} \right)\). In both drought years, resistance values slightly increased with increasing tree basal area. For statistical characteristics of the regression analyses, see Supplementary Table 2.
Figure 7 illustrates the relationships between tree basal area and growth resistance in the drought year 2003 for 14 long-term observational plots in monospecific stands. Here, we used the years 2000–2002 as reference. In case of Norway spruce, only one out of six plots showed a significant decrease in the resistance with increasing tree size, and none of them a significant increase (Fig. 7a). In European beech stands, the resistance was always decreasing with increasing tree size (Fig. 7b), while the development in Scots pine stands showed no consistent or significant relation between resistance and size (Fig. 7c). Finally, in the only European larch stand investigated, we found a significant increase in the resistance with increasing tree size (Fig. 7d). On average, the resistance was lower for Norway spruce and Scots pine than for European beech and European larch. In essence, the level of resistance and its change with tree size was species-specific.
Relationship between tree basal area and growth resistance in the drought year 2003 for long-term observational plots in South Germany for monospecific stands of a Norway spruce, b European beech, c Scots pine, and d European larch. Curves with slopes significantly different from \(a_{1} = 0\) are represented by solid lines; slopes not different from \(a_{1} = 0\) are indicated by broken lines. For statistical characteristics of the underlying model \(\ln \left( {{\text{res}}} \right) = a_{0} + a_{1} \times \ln \left( {ba} \right)\), see Supplementary Table 2
For spruce and beech stands, we used the long-term experiment Freising 813/1 (see Supplementary Table 1). This includes monospecific as well as mixed-species stands, which were composed of both species. However, there was only a small (European beech) or no (Norway spruce) effect of mixture (Fig. 8). In other words, the tendency of reduced resistance of larger trees is consistent in Norway spruce (small decline) as well as for European beech (large decline). For statistical characteristics of the regression analyses, see Supplementary Table 2.
Relationship between tree basal area in 2000–2002 and growth resistance in the drought year 2003 for a Norway spruce and b European beech on the mixed-species experiment Freising 813/1. The relationship between basal area and growth resistance was fitted for trees in monospecific stands (red), in mixed stands (green), and for all Norway spruces and European beeches together (black). For statistical characteristics of the underlying model \(\ln \left( {{\text{res}}} \right) = a_{0} + a_{1} \times \ln \left( {ba} \right)\), see Supplementary Table 2. (Color figure online)
Growth partitioning in drought years in Mediterranean forests
We used the long-term experiments Demanda (Lόpez-Marcos et al. 2021; Riofrío et al. 2017) and Lubia (Aldea et al 2017, 2018) in Central Spain to represent the drought reactions in Mediterranean forests. On the first field trial in Demanda (41° 47′ 35″ N and 41° 53′ 41″ N latitude and 2° 56′ 12″ W and 3° 20′ 46″ W longitude), the mean annual precipitation is around 600 mm, the mean annual temperature 10 °C, and in 2019, the precipitation was about 20–50% below the average. The 44–121-year-old stands consist of Pinus sylvestris and Pinus pinaster growing in mixed-species conditions (López-Marcos et al. 2021). At the mixed-species trials, the drought year 2019 reduced stem basal area growth of Pinus pinaster more than that of Pinus sylvestris. The overall regression \(\ln \left( {{\text{res}}} \right) = a_{0} + a_{1} \times \ln \left( {ba} \right)\) in Fig. 9c includes both species on all plots and shows no significant slope. A calculation for each species separately shown in Fig. 9d also revealed non-significant slopes (represented by broken curves) that are different in trend.
Annual stem basal area growth of Pinus sylvestris and Pinus pinaster on the mixed plots in Demanda (Spain) (a, b, with the drought year 2019 indicated with broken vertical lines), and growth resistance res calculated for the drought year 2019 in relation to the mean of the preceding period 2016–2018 (c and d). The overall regression \(\ln \left( {{\text{res}}} \right) = a_{0} + a_{1} \times \ln \left( {ba} \right)\) includes both species (c) and shows no significant slope, while a calculation for each species separately reveals non-significant slopes (represented by broken curves) that are different in trend (d)
The second field site, Lubia (2° 28′ 41″ W, 41° 39′ 22″ N) in central Spain, was selected to also include the effect of drought on deciduous trees (Aldea et al. 2017, 2018, 2021b). Here, the mean annual precipitation is 512 mm and the mean annual temperature 10 °C. In 2012, the precipitation was about 30% below average (362 mm) and not only spring 2012 but also autumn 2011 was very dry. The 51-year-old stand (2021) consists of Pinus pinaster and Quercus pyrenaica. The soils are regosols andarenosols, characterized by a slightly acidic pH (5–6), sandy texture and low fertility, composed of predominately tertiary and quaternary materials (Aldea 2018).
Overall, we mostly found a decrease in the drought resistance with increasing tree size and a species dependency. In particular, the investigation at Lubia provides evidence for a species-specific behaviour under drought, which can be explained with differences in growth phenology and species-specific physiological stress responses (Aldea et al 2018, 2021b). It should be noted, however, that most of the oaks at this site are suppressed or subdominant, whereas the pines are dominant and subdominant. (Fig. 10c, d). A separate evaluation for both species were not significant, but indicated that subdominant oaks hardly suffered by drought and showed a decrease in the drought resistance with increasing size. Pines showed a strong growth loss, but at a rather similar level for trees of all sizes. The results indicate that subdominant oaks in a stand may even benefit from the strong drought stress exposition of the neighbouring pines. At the stand level, an increase in the oak growth can compensate to some extent for the losses of the pines.
Annual stem basal area growth of Pinus pinaster and Quercus pyrenaica at the mixed plots in Lubia (Spain) (a, b, with the drought year 2012 indicated with broken vertical lines), and growth resistance res calculated for the drought year 2012 in relation to the mean of the preceding period 2010–2011 (c, d). The overall regression includes both species (c) and shows a significantly positive intercept and a negative slope while a calculation for each species separately reveals a significant intercept but non-significant decreasing slopes (represented by broken curves). For statistical characteristics, see Supplementary Table 2
Upscaling of drought reactions from the tree to the stand level
We use the approximately 70–90-year-old stands of Norway spruce and European beech at the study site Freising 813/1 to show the upscaling of drought reactions from the tree to the stand level. From these plots, we had both a complete inventory of the drought reactions of all individual trees and also the stand growth response based on the repeated inventories, including tree mortality.
Table 2 shows the drought resistance at the stand level calculated from the growth of all trees on the monospecific plots in 2003 divided by the mean growth in the reference period 2000–2002. We used the volume growth which yielded in case of Norway spruce a resistance of res = 0.479 and for European beech res = 0.776. This means that growth of Norway spruce in the drought year 2003 was only 47.9% of that in the years 2000–2002—a growth reduction of 52.1%. European beech grew 77.6% compared with the reference period, which means a growth reduction in 2003 of only 22.4%. If assessed by the growth of the 100 tallest trees (with respect to stem diameter) per hectare, i.e. based on the dominant trees, the resistance would be 0.359 in case of Norway spruce and 0.789 for European beech. This means an underestimation of resistance of Norway spruce by about 25% (0.359/0.479 = 0.749). In case of European beech, the 100 tallest trees represent the stand reaction very well (0.778/0.776 = 1.003). The ratio between resistance values derived from subsample at the stand level has also been calculated for other selected samples of tall and small trees as shown in Table 2. In general, using only dominant Norway spruces underestimates the stand resistance and using only subdominant Norway spruces overestimate the stand-level resistance, underestimating the growth losses. In contrast, calculating stand-level resistance from dominant beeches only reflects the stand resistance quite well, while using subdominant trees will overestimate the stand-level resistance (and underestimate growth losses). Overall, the resistance of subdominant spruce trees in the drought period 2003 was 23–35% of the stand-level resistance, and for beeches, it was 37–131%.
The calculations in Table 2 also show that growth losses of tall beeches were larger than those of small trees, leading to some degree to a compensation effect at the stand level and stabilizing stand productivity under drought. It is also demonstrated that an unbiased sampling of drought reactions and growth losses at the stand level requires sampling across all size classes. For details of the statistical test of the differences between tree- and stand-level resistance of Norway spruce and European beech, see Supplementary Table 3.
Pitfalls for upscaling from tree to stand level
Assuming n trees on a plot show a tree-level resistance \({\text{res}}_{1} \ldots {\text{res}}_{n}\), a simple scaling to the stand-level growth can be done as a calculation of the arithmetic mean \(\overline{{{\text{res}}}} = \left( {{\text{res}}|1 \ldots {\text{res}}_{n} } \right)/n\). This would, however, require that the resistance values of all tree sizes are equal (Table 3, Example 1) or that all trees have the same growth rate resistance (Table 3, Example 2). In all other cases, individual tree resistance values need a weighting by responses.
To illustrate common pitfalls, Table 3 shows four simplified examples always based on the same three trees, but assuming different values for their resistance and volume growth for a 3-year reference period. The analysed experimental plots showed that a similar resistance as well as similar growth rates across all tree sizes and social classes is unlikely. Both Norway spruce and European beech have shown a decrease in the resistance and increase in the reference growth rate with increasing tree size. This is reflected in Example 3 where the arithmetic mean results in an overestimation of stand-level resistance (arithmetic mean of 0.5 versus weighted mean of 0.44 means an overestimation of resistance by 13%). Example 4 demonstrates the effect of the opposite tendency—although unlikely. Here, large trees are assumed to be more resistant than small trees, which results in an arithmetic mean of 0.50 and a weighted mean of 0.56—indicating an underestimation of stand-level resistance by 10%. Many studies reported a higher growth rate of tall trees under normal conditions (e.g. Stephenson et al. 2014) and/or a lower resistance under drought stress (see Table 1 and especially Aldea et al. 2021b, Pretzsch et al. 2018a, b, Trouvé et al. 2017). As a result, the use of an arithmetic mean resistance will likely lead to assessments that overestimate the growth reduction by drought at the stand level. This is especially relevant when the direct assessment of stand growth resistance is not possible because not all trees are sampled (i.e. sampling by size classes).
Climate impact on the contribution of different tree size classes to stand growth
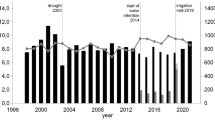
We investigate the development of GDC again based on measurements gathered at the Bavarian forest climate stations (see “Own studies on size-related growth reactions in temperate forest stands” section). In Fig. 11, the development of annual GDC values from three selected sites is presented, in particular, the long-term trend and the response to the conditions in dry years (2003 and 2015, vertical lines). The drought years often triggered a longer lasting decrease in GDC values in the subsequent years. This indicates that drought can change the partitioning of the stand growth in favour of small trees at the expense of tall trees and equalizes the size distribution (see introduction of GDC in “Drought impact on growth distribution among the trees within forest stands” section).
Development of GDC since the late 1990s at three Bavarian sites (out of 18) with adjacent climate stations. On most of the analysed plots, the GDC values decrease in drought years, i.e. the stand growth allocation changes in favour of small trees and on the expense of the tall trees. The vertical lines indicate the drought years 2003 and 2015. The horizontal lines indicate GDC = 0.0, i.e. the basal area growth contribution of the trees is equal to their basal area contribution to the stand. Curve sections above the 0-line line indicate superiority of tall trees, and curve sections below the 0-line indicate superiority of small trees. A decrease from one year to the next indicates a change in the growth distribution pattern in favour of small trees
In order to demonstrate differences between GDC values in dry vs. normal years, we calculated the mean GDC coefficient of all sites available from the Bavarian forest climate stations in the three reference years before a drought (2000–2002 and 2012–2014) and compared them with the GDC values in subsequent drought years (2003 and 2015). Any changes indicate a modification of the stand growth partitioning between small and tall trees. Figure 12 summarizes for all the experimental plots at the forest climate stations in Bavaria the effect of the drought on the GDC in the years (a) 2003) and (b) 2015. The GDC values in 2003 and 2015 are plotted against the respective mean GDC values in the reference periods. In case of a neutral effect of drought on the GDC, the points should lie on the bisector line. That the majority of the points are located below the line indicates that in dry years GDC is lower than in normal years and stand growth is preferentially allocated to the small tree at the expense of tall trees. A two-sided t-test showed significant differences (p < 0.05) between the GDC values in drought compared to normal years for both drought events (2003 and 2015).
Comparison of growth dominance coefficients for dry and reference years (calculated as described in the text). a GDC in 2003 compared to the normal growth period 2000–2000 and b GDC in 2015 compared to the normal growth period 2012–2014. Most of the points are below the bisectoral line, which means that on average drought reduced the GDC, i.e. the growth of small trees was less reduced than the growth of tall trees
Discussion and conclusions
Relevance of drought stress reactions at the tree and the stand level
In general, smaller scales determine fluxes and states at larger scales, but larger scales in turn determine boundary conditions for small-scale processes. For forest ecosystems, this general principle has been postulated by Ulrich (1994) and Robinson and Ek (2000), who also defined a number of relevant processes to be considered for stand-level development. Regarding responses to stress, primary species-specific individual impacts can be expected on the organ level which are directed to mitigate the respective stress (e.g. Niinemets 2010). Such responses in relation to drought are for example stomatal closure to reduce transpiration, root growth to improve water acquisition, or allocation to sapwood to strengthen or repair its conductivity. Since the benefits and costs of all reactions are different and depend on size, social status, or species properties, also the individual growth development can be expected to differ between trees (e.g. Grote et al. 2016; McGregor et al. 2021).
A further complication results from changing interactions between trees based on the individually different responses. For example, competition on water resources can be increased due to differences in uptake efficiency although the opposite effect of facilitation is also possible in case of hydraulic lift and water redistribution (e.g. Zapater et al. 2011; Magh et al. 2020). On medium term, differences in individual tree growth responses and mortality result in stand structural changes and new species compositions (Caspersen et al. 2011), which then feed back to the growing conditions of individuals. We have tried to summarize these mechanisms in Fig. 13, which illustrates that tree-level traits are essential, but not sufficient for understanding stand-level growth responses. It also shows, how management activities can have an influence, namely due to species selection (for example by planting, nursing and protecting young trees) and thinning (i.e. selective thinning). Similarly, disturbances also impact stand density, structure and species composition by acting selectively on species (e.g. insect attacks) or size (e.g. storm damage).
It should be noted that drought and other stresses interact with each other with enhancing (e.g. the increased transpiration demand during heatwaves is increasing drought stress, e.g. von Buttlar et al. 2018) or in some cases also compensating effects (e.g. drought-imposed stomatal closure may reduce the uptake of air pollutants such as ozone, Matyssek et al. 2006). In general, it can be expected that stand-level stress can be mitigated by trait diversification (e.g., variation of tree size, provenance and species) because this is likely to reduce stress-related risks (e.g. barrier against pathogen spread, balance by predatory herbivores). Over longer periods, stress mitigation also occurs due to acclimation responses that are related to tissue properties (e.g. wood density or leaf stomata density) or tree allometry (i.e. more root- relative to foliage biomass), also known as legacy effects (Kannenberg et al. 2019).
Future analyses may also address the size dependence of growth recovery after extreme drought, i.e. the drought resilience. Latter can also depend on social classes and affect the upscaling from tree to the stand level (Pretzsch et al. 2020). For instance, Martin-Benito et al. (2008) found that dominant trees showed more plastic response and a better recovery after drought. This trade-off between resistance and recovery requires further consideration.
Observed patterns and underlying processes
In this study, we could show the different drought responses by tree size and social classes, and their potential impact on stand-level behaviour. We are not differentiating into size- and age although small trees of high age might respond differently from younger ones (Aakala et al. 2013), but we could not find any consistent pattern regarding drought stress in this regard. In most of the investigated stands, drought resistance was lower for tall trees or similar for trees of all sizes and social classes (see, for example, Figures 7, 8, 10). In a minor number of cases drought resistance increased with tree size in the case of Norway spruce (Fig. 6), European larch (Fig. 7d), or Scots pine and Maritime pine stands in Spain (Fig. 9). Overall, most of our findings were in line with the literature where 17 out of 24 studies indicated a decreasing or invariant relationship between tree size and drought resistance (Table 1), particularly for European beech and Norway spruce. In addition, Norway spruce and Scots pine react more homogeneous across different size classes and sites than European beech and European larch. Moreover, mixed stands were more variable in individual tree growth responses than monospecific stands (Figs. 8).
We could not determine the underlying causes of the pattern that were observed, but can hypothesize based on the general considerations from the previous chapter. It is likely that the shading effect is greatest in European beech and Norway spruce stands as they have the highest leaf area and stand density. Scots pine and European larch stands have lower densities due to their higher light demand. Thus, a cooler microclimate and less transpiration demand can be expected in the shade-tolerant species, protecting from heat damage and decreasing drought stress (Grote et al. 2016). Consequently, we assume that the decrease in the resistance with tree size develops in parallel with maximum stand density and leaf area in the order European beech > Norway spruce > Scots pine > European larch (Ellenberg and Leuschner 2010). It is possible that the minority of findings that indicate an increase in the resistance with size originate from statistical problems because the number of stands investigated was not sufficient to generalize the results. This might be the case particularly for Scots pine (both decreasing and increasing with size, Fig. 7c) and European larch (Fig. 7d). Another possible explanation originates from the fact that we only refer to responses of aboveground (stem) growth, although partitioning between above and belowground tissues is known to depend nonlinearly on soil water availability (Phillips et al. 2016). Since drought stress is differently experienced in small and large trees, and indications exist that such responses might be more expressed in mixed stands (Schäfer et al. 2019), overall biomass growth responses might be obscured or even seem to be reversed when only looking at aboveground growth observations.
Finally, stand-level reactions result from tree-level traits and interactions. Species-specific drought resistance may result from anatomical (Saadaoui et al. 2017; Pellizzari et al. 2016), morphological (Bussotti and Pollastrini 2015; Seiler and Johnson 1988), or physiological acclimation (Lemaire et al. 2021; Anderegg and HilleRisLambers 2016) of trees. In Central Europe, sessile oak and Scots pine, for example, seem to be more resistant to drought events than European beech and Norway spruce (Pretzsch et al. 2013). Many studies provide evidence that drought resistance may decrease with progressive tree size due to a more difficult water transport to leaves (e.g. Bennett et al. 2015; Olson et al. 2018; McGregor et al. 2021). This is also well reflected by the global correlation between site index and water supply (Koch et al. 2004; Schäfer et al. 2000; Ryan and Yoder 1997), but see also (Becker et al. 2000). Competitive and facilitative neighbourhood interactions can modify the drought stress response in monospecific (Bottero et al. 2017; Steckel et al. 2020a, b) and mixed-species stands (Alba et al. 2019; Grossiord 2020). In monospecific stands, it is mainly stand density that can increase drought stress due to a reduction in the water availability per tree (Toraño Caicoya and Pretzsch 2021; Sohn et al. 2016; Giuggiola et al. 2013). However, density reduction by thinning, especially if realized too abruptly and strongly, can also cause drought stress as trees may be not acclimated to the new microclimate, or forest floor vegetation may become an even stronger competitor for water than the former neighbours (Gebhardt et al. 2014). An additional finding which is supported by several studies is that mixing tree species can mitigate drought stress (Pardos et al. 2021; Forrester et al. 2016; Thurm et al. 2016; Pretzsch et al. 2013; Vitali et al. 2018; Steckel et al. 2020a, b). This has been demonstrated to occur particularly by the facilitative effect of deep-rooting trees on more shallow-rooting neighbours by hydraulic lift (Pretzsch 2021a, b, c; Dawson 1993).
Small size and low social position may have disadvantages under normal conditions that can turn into advantages in years with heat or drought stress (Bose et al. 2014; Pretzsch et al. 2018a, b). Ample water supply of a stand may enable tall trees to take advantage of their predominant social position and to better exploit radiation, shade their smaller neighbours and thus strengthen their preferential, size-asymmetric competitive status (Schwinning and Weiner 1998). However, in hot and dry years, a smaller size can turn into an advantage because shaded trees transpiration demand is lower and water supply may be easier to facilitate with less height (Dordel et al. 2011). In particular, shade-tolerant tree species which are able to grow in the understory and endure high stand densities such as European beech may be able to use this advantage, whereas light-demanding species such as Scots pine or European larch may be not.
System dynamics and modelling
How can tree and stand modelling consider the dependence of growth partitioning between small and tall trees and of tree mortality on annual weather conditions? This is only possible with process-based ecophysiological models that depend on environmental conditions, and with explicit consideration of individual trees or at least differently sized social classes (e.g. Grote and Pretzsch 2002; Deckmyn et al. 2008; Grote et al. 2020; Jonard et al. 2020). Only then, carbon assimilation and stand development can be dynamically represented based on temporally shifting light and/or water limitations. The range from size-asymmetric competition (promotion of tall trees) under ample water supply to size-symmetric competition (growth proportional to size) or even overproportioned growth of small trees under drought is then inherently integrated.
Statistical dendrometry models that estimate tree growth depending on competition indices and additional tree covariables such as stem size or crown dimension (e.g. Pretzsch et al. 2002, Le Moguedec et al. 2012, Thurnher et al. 2017) assume ceteris paribus a strong exponential decrease in the growth with increasing competition index. Our results suggest that this may apply in years without drought stress, i.e. when light is the growth limiting factor and competition is asymmetric. In such years, growth rate may strongly decrease with decreasing social position and competitive status (Fig. 14a). In dry years, however, being tall and predominant may become less advantageous, the slope of the growth–competition relation becomes less steep, and the growth difference between small and tall trees smaller.
Consequences of the drought-related modification of tree growth for modelling the growth–competition relationship in a individual tree models and b diameter distribution and size–class models. Solid lines represent growth under moist conditions (asymmetric in favour of tall trees), broken lines growth under normal conditions (size-symmetric growth of small and tall trees), and dotted lines represent growth under dry and hot conditions (asymmetric in favour of small trees)
Most size–class and diameter distribution models predict the tree growth depending on size, often assuming a linear growth–size relationship (Fig. 14b, straight broken line in the middle). Our results suggest that in moist years the growth–size relationship may change to a progressively increasing (Fig. 14b, broken line, favouring tall trees) while in dry years to a degressively increasing relationship (Fig. 14b, dotted line, favouring small trees).
Certainly, models based on average dimensions for trees and aggregated stand-level values such as yield tables (Weiskittel et al. 2011; Assmann and Franz 1965) are not appropriate for reflecting the mentioned structural changes of the stand by drought effects. The relative growth reduction of tall compared to small trees in drought years means a tendency to the homogenizing size differences, especially if drought years become more frequent. This tendency may be reinforced if drought triggers mortality preferentially of tall trees (e.g. Bennett et al. 2015). This homogenizing effect may be stronger in monospecific stands than in mixed-species stands, because in the latter a wider differentiation of ecophysiological traits may also result in a more expressed social ranking (Aldea et al. 2021b). It should be noted that already slight advantages of small and medium sized trees under dry conditions may strongly affect stand-level developments, because their frequency is generally unproportionable higher. A further positive feedback of such an influence is that a relatively larger exploitation of resources in dry years can also have a negative feedback on the growth on dominant trees (Toraño Caicoya and Pretzsch 2021; Knapp 1991).
Consequences for measurement and monitoring
The vitality of a stand is generally monitored at the stand level, considering drought stress in terms of volume production, crown density and mortality rates. These indicators include interactions at individual tree level that may have buffered or aggravated the observed features. Individual mortality or tree growth losses are detrimental at any rate and should be minimized. However, if the growth or volume reduction of more susceptible trees is compensated by trees of other social positions or sizes, productivity or other forest functions and services may be less reduced and the need of mitigating or restoring activities may be avoided. The capacity for a compensation of losses can at least for some species combination be assumed to be higher in more diversely structured or composed forests (Forrester et al. 2019; Aldea et al. 2021b).
Thus, studies of the reaction of small versus tall trees that start at the tree and end at the stand level are a relevant addition to the otherwise mostly tree-based stress analyses. They provide basic information about the resilience and sensitivity of a forest by integrating the individual tree stress reactions which are likely to differ from findings based on average or only dominant individual trees. The size-dependent growth response to drought strongly suggests sampling across the whole diameter range in order to correctly monitor growth responses and vitality. An approved methodology to do this is the installation of band dendrometers that give a very precise and highly resolved indication of basal area growth in normal as well as dry years. A possible drawback is that volume growth estimates can only be approached with pre-defined taper functions, which may, however, change due to drought (Sterba 1981, 1984; Mette et al. 2015; Pretzsch et al. 2010a, b; Rais et al. 2014). Rubio-Cuadrado et al. (2018) showed how also tree height growth could be affected by drought. More specifically, it is assumed that growth losses at breast height are much more pronounced than in the upper parts of the stem (Pretzsch et al. 2010a, b; Wipfler et al. 2005; Sterba 1981). Thus, biomass growth reactions and reported percentages of growth losses based on basal area may be overestimated. This also indicates that the sensitivity to drought stress of specific tree species may be overestimated (such has been measured for beech, Schäfer et al. 2019). Overall, this adds to the uncertainty that is related to drought-imposed allocation shifts, demanding either more measurements or an improved consideration of environmental conditions for biomass expansion factors (Temesgen et al. 2015).
Relevance for ecosystem management
Our evaluations indicate that silvicultural prescriptions need to consider the response of differently sized, positioned or ranked trees on drought stress separately. This is important since for example thinning from below would be contra-productive under dry conditions if smaller trees are less affected and are able to compensate for growth losses of large trees. This should be kept in mind when designing silvicultural prescriptions while otherwise their capacity of stabilization or buffering drought stress might be “thinned away”.
Drought stress reactions and growth losses may be less detrimental in widely size-structured and/or mixed stands where individual trees area differently affected by drought. In this case, specific trees may be threatened, but not necessarily the whole stand. Remaining, more resistant trees may partly buffer the losses and provide stand ecological functions and services. Thus, forest regeneration as well as selective thinning should be directed to increase the number of suitable species in view of climate change, drought, and stress in general. As important criteria, both the individual susceptibility which can be remarkably different (e.g. Gazol et al. 2018) and the stand-level interactions should be considered (see, for example, Vergarechea et al. 2019).
Knowledge of the behaviour of individual trees under drought stress is essential but not sufficient for the design of resilient forest stands. For ecosystem management and planning, also tree-level traits may provide evidence for understanding and influencing stand-level reaction patterns. The drought susceptibility of different species, their acclimation capacity (e.g. anatomical, morphological acclimation), and their facilitative interaction (e.g. shading, hydraulic lift) should therefore be important selection criteria. Since these might differ between social classes and sizes, the forest design in terms of space per tree as well as species and size distribution can strongly affect tree–tree interaction in order to increase stability at the stand level.
The desire to mitigate drought and other climate change-related stress by means of silvicultural treatments is intensively discussed (Camarero et al. 2015, 2018; Ogle et al. 2015). As indicated above, thinning from below may remove particularly those trees that may play a growth-stabilizing role in drought years. However, reducing stand density may reduce drought stress of the remaining stand by improving the water availability per tree (D’Amato et al. 2013; Andrews et al. 2020). In addition, other effects of thinning are an increase in the temperature and wind as well as a decrease in air humidity, which exacerbates transpiration demand and thus drought stress.
Trees respond to low stand density by acclimation of their tissue properties and anatomical features, which results in strong legacy effects of past treatments on the resilience to present or future conditions (Pretzsch 2021a, b, c; Ogle et al. 2015). In particular, strong and abrupt thinnings may be detrimental regarding drought resistance, because trees are only acclimating slowly to new environmental conditions. For instance, initial wide spacing or repeated strong thinning favours the formation of large xylem vessels that enhance hydraulic conductivity and thus transpiration and growth under normal conditions, but may increase the risk of embolism under drought (Saunders and Drew 2022). Together with the effects on physical conditions, this might explain contradicting results that either found an improvement (Aldea et al. 2017; Martín-Benito et al. 2010; Sohn et al. 2016) or a decline (D'Amato et al. 2013; Steckel et al. 2020a, b) of drought resistance with density reduction, or showed that thinning may have positive effects in the short term, followed by negative effects in the long term (D'Amato et al. 2013; Calama et al. 2019). These discrepancies may be resolved when analysing the past tree ring structure and could help re-evaluate and improve stand density regulation as recommended by Sohn et al. (2016).
References
Aakala T, Fraver S, D’Amato A, Palik B (2013) Influence of competition and age on tree growth in structurally complex old-growth forests in northern Minnesota, USA. For Ecol Manage 308:128–135
Alba C, Fahey C, Flory SL (2019) Global change stressors alter resources and shift plant interactions from facilitation to competition over time. Ecology 100(12):e02859
Aldea J, Bravo F, Bravo-Oviedo A, Ruiz-Peinado R, Rodríguez F, del Río M (2017) Thinning enhances the species-specific radial increment response to drought in Mediterranean pine-oak stands. Agric for Meteorol 237:371–383
Aldea J, Bravo F, Vázquez Piqué J, Rubio Cuadrado Á, del Río M (2018) Species-specific weather response in the daily stem variation cycles of Mediterranean pine-oak mixed stands. Agric for Meteorol 256:220–230
Aldea J, Ruiz-Peinado R, del Río M, Pretzsch H, Heym M, Brazaitis G, Jansons A, Metslaid M, Barbeito I, Bielak K, Granhus A, Holm S-O, Nothdurft A, Sitko R, Löf M (2021b) Species stratification and weather conditions drive tree growth in Scots pine and Norway spruce mixed stands along Europe. For Ecol Manage 481:118697
Allen CD, Macalady AK, Chenchouni H, Bachelet D, McDowell N, Vennetier M, Kitzberger T, Rigling A, Breshears DD, Hogg EH, Gonzalez P, Fensham R, Zhang Z, Castro J, Demidova N, Lim JH, Allard G, Running SW, Semerci A, Cobb N (2010) A global overview of drought and heat-induced tree mortality reveals emerging climate change risks for forests. For Ecol Manage 259:660–684. https://doi.org/10.1016/j.foreco.2009.09.001
Anderegg LD, HilleRisLambers J (2016) Drought stress limits the geographic ranges of two tree species via different physiological mechanisms. Glob Change Biol 22(3):1029–1045
Andrews CM, D’Amato AW, Fraver S, Palik B, Battaglia MA, Bradford JB (2020) Low stand density moderates growth declines during hot droughts in semi-arid forests. J Appl Ecol 57(6):1089–1102
Archambeau J, Ruiz-Benito P, Ratcliffe S, Fréjaville T, Changenet A, Muñoz Castañeda JM, Lehtonen A, Dahlgren J, Zavala MA, Benito Garzón M (2020) Similar patterns of background mortality across Europe are mostly driven by drought in European beech and a combination of drought and competition in Scots pine. Agric for Meteorol 280:107772. https://doi.org/10.1016/j.agrformet.2019.107772
Assmann E (1970) The principles of forest yield study. Pergamon Press, Oxford, p 506
Assmann E, Franz F (1965) Vorläufige Fichten-Ertragstafel Für Bayern. Forstw Cbl 84(1):13–43
Aussenac G (2000) Interactions between forest stands and microclimate: ecophysiological aspects and consequences for silviculture. Ann for Sci 57(3):287–301
Bauhus J, Forrester DI, Gardiner B, Jactel H, Vallejo R, Pretzsch H (2017) Ecological stability of mixed-species forests. In: Mixed-species forests. Springer, Berlin, pp 337–382
Becker P, Meinzer FC, Wullschleger SD (2000) Hydraulic limitation of tree height: a critique. Funct Ecol 14(1):4–11
Bennett AC, McDowell NG, Allen CD, Anderson-Teixeira KJ (2015) Larger trees suffer most during drought in forests worldwide. Nat Plants 1(10):1–5
Binkley D, Stape JL, Ryan MG, Barnard HR, Fownes J (2002) Age-related decline in forest ecosystem growth: an individual-tree, stand-structure hypothesis. Ecosystems 5(1):58–67
Bogino SM, Bravo F (2008) Growth response of Pinus pinaster Ait. to climatic variables in central Spanish forests. Ann for Sci 65(5):506–506
Bose AK, Brais S, Harvey BD (2014) Trembling aspen (Populus tremuloides Michx.) volume growth in the boreal mixedwood: Effect of partial harvesting, tree social status, and neighborhood competition. For Ecol Manage 327:209–220
Bottero A, D’Amato AW, Palik BJ, Bradford JB, Fraver S, Battaglia MA, Asherin LA (2017) Density-dependent vulnerability of forest ecosystems to drought. J Appl Ecol 54(6):1605–1614
Bottero A, Forrester DI, Cailleret M, Kohnle U, Gessler A, Michel D, Bose AK, Bauhus J, Bugmann H, Cuntz M, Gillerot L, Hanewinkel M, Lévesque M, Ryder J, Sainte-Marie J, Schwarz J, Yousefpour R, Zamora-Pereira JC, Rigling A (2021) Growth resistance and resilience of mixed Silver fir and Norway spruce forests in central Europe: Contrasting responses to mild and severe droughts. Glob Change Biol 27(18):4403–4419
Bowditch E, Santopuoli G, Binder F, Del Rio M, La Porta N, Kluvankova T, Tognetti R (2020) What is Climate-Smart Forestry? A definition from a multinational collaborative process focused on mountain regions of Europe. Ecosyst Serv 43:101113
Bussotti F, Pollastrini M (2015) Evaluation of leaf features in forest trees: Methods, techniques, obtainable information and limits. Ecol Ind 52:219–230
Calama R, Conde M, de Dios-García J, Madrigal G, Vázquez-Piqué J, Gordo FJ, Pardos M (2019) Linking climate, annual growth, and competition in a Mediterranean forest: Pinus pinea in the Spanish northern plateau. Agric for Meteorol 264:309–321
Camarero JJ, Gazol A, Sangüesa-Barreda G, Oliva J, Vicente-Serrano SM (2015) To die or not to die: early warnings of tree dieback in response to a severe drought. J Ecol 103(1):44–57
Camarero JJ, Gazol A, Sangüesa-Barreda G, Cantero A, Sánchez-Salguero R, Sánchez-Miranda A, Ibáñez R (2018) Forest growth responses to drought at short-and long-term scales in Spain: squeezing the stress memory from tree rings. Front Ecol Evol 6:9
Carnwath G, Nelson C (2017) Effects of biotic and abiotic factors on resistance versus resilience of Douglas fir to drought. PLoS ONE 12(10):e0185604
Caspersen JP, Vanderwel MC, Cole WG, Purves DW (2011) How stand productivity results from size- and competition-dependent growth and mortality. PLoS ONE 6(12):e28660
Castagneri D, Nola P, Cherubini P, Motta R (2012) Temporal variability of size–growth relationships in a Norway spruce forest: the influences of stand structure, logging, and climate. Can J for Res 42(3):550–560
Cermák J, Riguzzi F, Ceulemans R (1998) Scaling up from the individual tree to the stand level in Scots pine. I. Needle distribution, overall crown and root geometry. In: Annales des sciences forestières. EDP Sciences, vol 55, no 1–2, pp 63–88
Clark JS, Iverson L, Woodall CW, Allen CD, Bell DM, Bragg DC, Zimmermann NE (2016) The impacts of increasing drought on forest dynamics, structure, and biodiversity in the United States. Glob Change Biol 22(7):2329–2352
Colangelo M, Camarero JJ, Borghetti M, Gazol A, Gentilesca T, Ripullone F (2017) Size matters a lot: drought-affected italian oaks are smaller and show lower growth prior to tree death. Front Plant Sci. https://doi.org/10.3389/fpls.2017.00135
D’Amato AW, Bradford JB, Fraver S, Palik BJ (2013) Effects of thinning on drought vulnerability and climate response in northern temperate forest ecosystems. Ecol Appl 23(8):1735–1742
Dănescu A, Kohnle U, Bauhus J, Sohn J, Albrecht AT (2018) Stability of tree increment in relation to episodic drought in uneven-structured, mixed stands in southwestern Germany. For Ecol Manage 415:148–159
Dawson TE (1993) Hydraulic lift and water use by plants: implications for water balance, performance and plant-plant interactions. Oecologia 95(4):565–574
Deckmyn G, Verbeeck H, Op de Beeck M, Vansteenkiste D, Steppe K, Ceulemans R (2008) ANAFORE: A stand-scale process-based forest model that includes wood tissue development and labile carbon storage in trees. Ecol Model 215(4):345–368
De Camino R (1976) Zur Bestimmung der Bestandeshomogenität. Allgemeine Forst- Und Jagdzeitung 147:54–58
Diaconu D, Kahle HP, Spiecker H (2017) Thinning increases drought tolerance of European beech: a case study on two forested slopes on opposite sides of a valley. Eur J for Res 136(2):319–328
Ding H, Pretzsch H, Schütze G, Rötzer T (2017) Size-dependence of tree growth response to drought for Norway spruce and European beech individuals in monospecific and mixed-species stands. Plant Biol 19(5):709–719
Dobbertin M (2005) Tree growth as indicator of tree vitality and of tree reaction to environmental stress: a review. Eur J for Res 124(4):319–333
Dordel J, Seely B, Simard SW (2011) Relationships between simulated water stress and mortality and growth rates in underplanted Toona ciliata Roem. in subtropical Argentinean plantations. Ecol Model 222(17):3226–3235
Eilmann B, Rigling A (2012) Tree-growth analyses to estimate tree species’ drought tolerance. Tree Physiol 32(2):178–187
Ellenberg H, Leuschner C (2010) Vegetation Mitteleuropas mit den Alpen: in ökologischer, dynamischer und historischer Sicht (Vol. 8104). Utb
Ferretti M, Fischer R (2013) Forest Monitoring: methods for terrestrial investigations in Europe with an overview of North America and Asia. Developments in Environmental Science. Elsevier, Oxford, vol 12, p 507
Forrester DI, Bonal D, Dawud S, Gessler A, Granier A, Pollastrini M, Grossiord C (2016) Drought responses by individual tree species are not often correlated with tree species diversity in E uropean forests. J Appl Ecol 53(6):1725–1734
Forrester DI (2019) Linking forest growth with stand structure: tree size inequality, tree growth or resource partitioning and the asymmetry of competition. For Ecol Manage 447:139–157
Gazol A, Camarero JJ, Sangüesa-Barreda G, Vicente-Serrano SM (2018) Post-drought Resilience after forest die-off: shifts in regeneration, composition, growth and productivity. Front Plant Sci 9:1546
Gebhardt T, Häberle KH, Matyssek R, Schulz C, Ammer C (2014) The more, the better? Water relations of Norway spruce stands after progressive thinning. Agric for Meteorol 197:235–243
Giuggiola A, Bugmann H, Zingg A, Dobbertin M, Rigling A (2013) Reduction of stand density increases drought resistance in xeric Scots pine forests. For Ecol Manage 310:827–835
Gleason KE, Bradford JB, Bottero A, D’Amato AW, Fraver S, Palik BJ, Battaglia MA, Iverson L, Kenefic L, Kern CC (2017) Competition amplifies drought stress in forests across broad climatic and compositional gradients. Ecosphere 8(7):e01849
Grams TE, Hesse BD, Gebhardt T, Weikl F, Rötzer T, Kovacs B, Pritsch K (2021) The Kroof experiment: realization and efficacy of a recurrent drought experiment plus recovery in a beech/spruce forest. Ecosphere 12(3):e03399
Grossiord C (2020) Having the right neighbors: how tree species diversity modulates drought impacts on forests. New Phytol 228(1):42–49
Grote R, Pretzsch H (2002) A model for individual tree development based on physiological processes. Plant Biol 4(2):167–180
Grote R, Gessler A, Hommel R, Poschenrieder W, Priesack E (2016) Importance of tree height and social position for drought-related stress on tree growth and mortality. Trees 30(5):1467–1482
Grote R, Kraus D, Weis W, Ettl R, Göttlein A (2020) Dynamic coupling of allometric ratios to a process-based forest growth model for estimating the impacts of stand density changes. Forestry 93(5):601–615
He JS, Zhang QB, Bazzaz FA (2005) Differential drought responses between saplings and adult trees in four co-occurring species of New England. Trees 19:442–450
Heym M, Bielak K, Wellhausen K, Uhl E, Biber P, Perkins D, Pretzsch H (2018) A new method to reconstruct recent tree and stand attributes of temporary research plots: new opportunity to analyse mixed forest stands. IntechOpen, Conifers, Chapter 2. Conifers. IntechOpen, pp 25–45
Heym M, Ruíz-Peinado R, Del Río M, Bielak K, Forrester DI, Dirnberger G, Pretzsch H (2017) EuMIXFOR empirical forest mensuration and ring width data from pure and mixed stands of Scots pine (Pinus sylvestris L.) and European beech (Fagus sylvatica L.) through Europe. Ann for Sci 74(3):1–9
Holtmann A, Huth A, Pohl F, Rebmann C, Fischer R (2021) Carbon sequestration in mixed deciduous forests: the influence of tree size and species composition derived from model experiments. Forests 12(6):726. https://doi.org/10.3390/f12060726
Johann K (1993) DESER-Norm 1993. Normen der Sektion Ertragskunde im Deutschen Verband Forstlicher Forschungsanstalten zur Aufbereitung von waldwachstumskundlichen Dauerversuchen. Proc Dt Verb Forstl Forschungsanst, Sek Ertragskd, in Unterreichenbach-Kapfenhardt, pp 96–104
Jonard M, André F, de Coligny F, de Wergifosse L, Beudez N, Davi H, Ligot G, Ponette Q, Vincke C (2020) HETEROFOR 1.0: a spatially explicit model for exploring the response of structurally complex forests to uncertain future conditions—Part 1: carbon fluxes and tree dimensional growth. Geosci Model Dev 13(3):905–935
Kannenberg SA, Novick KA, Alexander MR, Maxwell JT, Moore DJP, Phillips RP, Anderegg WRL (2019) Linking drought legacy effects across scales: from leaves to tree rings to ecosystems. Glob Change Biol 25(9):2978–2992
Knapp E (1991) Zur Wuchsleistung der Unterbaubuche im ungleichaltrigen Kiefern-Buchen-Mischbestand vor und nach ihrer Übernahme als Hauptbestand auf Standorten des nordostdeutschen Tieflandes, Deutscher Verband Forstlicher Forschungsanstalten. Sektion Ertragskunde Jahrestagung, pp 96–110
Koch GW, Sillett SC, Jennings GM, Davis SD (2004) The limits to tree height. Nature 428(6985):851–854
Kramer H (1988) Waldwachstumslehre. Paul Parey, Hamburg, p 374
Le Moguedec G, Dhote JF (2012) Fagacees: a tree-centered growth and yield model for sessile oak (Quercus petraea L.) and common beech (Fagus sylvatica L.). Ann for Sci 69(2):257–269
Lemaire C, Blackman CJ, Cochard H, Menezes-Silva PE, Torres-Ruiz JM, Herbette S (2021) Acclimation of hydraulic and morphological traits to water deficit delays hydraulic failure during simulated drought in poplar. Tree Physiol. https://doi.org/10.1093/treephys/tpab086
Liu Y, Muller RN (1993) Effect of drought and frost on radial growth of overstory and understory stems in a deciduous forest. Am Midl Nat 129:19–25
Lloret F, Keeling EG, Sala A (2011) Components of tree resilience: effects of successive low-growth episodes in old ponderosa pine forests. Oikos 120(12):1909–1920
Looney CE, D’Amato AW, Jovan S (2021) Investigating linkages between the size-growth relationship and drought, nitrogen deposition, and structural complexity in western US Forests. For Ecol Manag 497:119494
Loetsch F, Zöhrer F, Haller KE (1973) Forest inventory, vol 2: Inventory data collected by terrestrial measurements and observations, data processing in forest inventory. The sample plot, plotless sampling and regenerations survey. List sampling with unequal probabilities and planning, performance and field checking of forest inventories. BLV-Verlagsgesellschaft, München, Bern, Wien, p 469
Lόpez-Marcos D, Turriόn M-B, Bravo F, Martínez-Ruiz C (2021) Characterization of mixed and monospecific stands of Scots pine and Maritime pine: soil profile, physiography, climate and vegetation cover data. Ann for Sci 78(2):28
Magh R-K, Eiferle C, Burzlaff T, Dannenmann M, Rennenberg H, Dubbert M (2020) Competition for water rather than facilitation in mixed beech-fir forests after drying-wetting cycle. J Hydrol 587:124944
Martín-Benito D, Cherubini P, Del Río M, Cañellas I (2008) Growth response to climate and drought in Pinus nigra Arn. trees of different crown classes. Trees 22(3):363–373
Martín-Benito D, Del Río M, Heinrich I, Helle G, Cañellas I (2010) Response of climate-growth relationships and water use efficiency to thinning in a Pinus nigra afforestation. For Ecol Manage 259(5):967–975
Martínez-Vilalta J, López BC, Loepfe L, Lloret F (2012) Stand-and tree-level determinants of the drought response of Scots pine radial growth. Oecologia 168(3):877–888
Matyssek R, Le Thiec D, Löw M, Dizengremel P, Nunn AJ, Häberle KH (2006) Interactions between drought and O3 stress in forest trees. Plant Biol 8(1):11–17
McGregor IR, Helcoski R, Kunert N, Tepley AJ, Gonzalez-Akre EB, Herrmann V, Zailaa J, Stovall AEL, Bourg NA, McShea WJ, Pederson N, Sack L, Anderson-Teixeira KJ (2021) Tree height and leaf drought tolerance traits shape growth responses across droughts in a temperate broadleaf forest. New Phytol 231(2):601–616
Mérian P, Lebourgeois F (2011) Size-mediated climate–growth relationships in temperate forests: a multi-species analysis. For Ecol Manage 261(8):1382–1391
Merlin M, Perot T, Perret S, Korboulewsky N, Vallet P (2015) Effects of stand composition and tree size on resistance and resilience to drought in sessile oak and Scots pine. For Ecol Manage 339:22–33
Metsaranta JM, Lieffers VJ (2010) Patterns of inter-annual variation in the size asymmetry of growth in Pinus banksiana. Oecologia 163(3):737–745
Mette T, Falk W, Uhl E, Biber P, Pretzsch H (2015) Increment allocation along the stem axis of dominant and suppressed trees in reaction to drought-results from 123 stem analyses of Norway spruce, Scots pine and European beech. Austrian Journal of Forest Science. 132. Jahrgang (2015). Heft 4:185–254
Niinemets Ü (2010) Responses of forest trees to single and multiple environmental stresses from seedlings to mature plants: Past stress history, stress interactions, tolerance and acclimation. For Ecol Manage 260(10):1623–1639
Obladen N, Dechering P, Skiadaresis G, Tegel W, Keßler J, Höllerl S, Kaps S, Hertel M, Dulamsuren C, Seifert T, Hirsch M, Seim A (2021) Tree mortality of European beech and Norway spruce induced by 2018–2019 hot droughts in central Germany. Agric for Meteorol 307:108482
Ogle K, Barber JJ, Barron-Gafford GA, Bentley LP, Young JM, Huxman TE, Loik ME, Tissue DT (2015) Quantifying ecological memory in plant and ecosystem processes. Ecol Lett 18(3):221–235
Olivar J, Rathgeber C, Bravo F (2015) Climate change, tree-ring width and wood density of pines in Mediterranean environments. IAWA J 36(3):257–269
Olson ME, Soriano D, Rosell JA, Anfodillo T, Donoghue MJ, Edwards EJ, León-Gómez C, Dawson T, Camarero Martínez JJ, Castorena M, Echeverría A, Espinosa CI, Fajardo A, Gazol A, Isnard S, Lima RS, Marcati CR, Méndez-Alonzo R (2018) Plant height and hydraulic vulnerability to drought and cold. PNAS 115(29):7551–7556
Orwig DA, Abrams MD (1997) Variation in radial growth responses to drought among species, site, and canopy strata. Trees 11:474–484
Pardos M, del Río M, Pretzsch H, Jactel H, Bielak K, Bravo F, Calama R (2021) The greater resilience of mixed forests to drought mainly depends on their composition: analysis along a climate gradient across Europe. For Ecol Manag 481:118687
Pellizzari E, Camarero JJ, Gazol A, Sangüesa-Barreda G, Carrer M (2016) Wood anatomy and carbon-isotope discrimination support long-term hydraulic deterioration as a major cause of drought-induced dieback. Glob Change Biol 22(6):2125–2137
Phillips RP, Ibáñez I, D’Orangeville L, Hanson PJ, Ryan MG, McDowell NG (2016) A belowground perspective on the drought sensitivity of forests: towards improved understanding and simulation. For Ecol Manage 380:309–320
Pichler P, Oberhuber W (2007) Radial growth response of coniferous forest trees in an inner Alpine environment to heat-wave in 2003. For Ecol Manage 242:688–699
Piutti E, Cescatti A (1997) A quantitative analysis of the interactions between climatic response and intraspecific competition in European beech. Can J for Res 27(3):277–284
Pretzsch H, Dieler J, Matyssek R, Wipfler P (2010a) Tree and stand growth of mature Norway spruce and European beech under long-term ozone fumigation. Env Poll 158:1061–1070
Pretzsch H (2021a) Facilitation and competition reduction in tree species mixtures in Central Europe: consequences for growth modelling and forest management. Ecol Model 464:109812
Pretzsch H (2021b) Trees grow modulated by the ecological memory of their past growth. Consequences for monitoring, modelling, and silvicultural treatment. For Ecol Manag 487:18982
Pretzsch H (2021c) Mixing intensity, stand density, and water supply can increase the overyielding of mixed versus mono-specific stands until advanced ages. For Ecol Manag 503:119741
Pretzsch H, Biber P (2010) Size-symmetric versus size-asymmetric competition and growth partitioning among trees in forest stands along an ecological gradient in central Europe. Can J for Res 40(2):370–384
Pretzsch H, Dieler J, Matyssek R, Wipfler P (2010b) Tree and stand growth of mature Norway spruce and European beech under long-term ozone fumigation. Environ Pollut 158(4):1061–1070
Pretzsch H, Grams T, Häberle KH, Pritsch K, Bauerle T, Rötzer T (2020) Growth and mortality of Norway spruce and European beech in monospecific and mixed-species stands under natural episodic and experimentally extended drought. Results of the KROOF throughfall exclusion experiment. Trees 34(4):957–970
Pretzsch H, Schütze G, Biber P (2018a) Drought can favour the growth of small in relation to tall trees in mature stands of Norway spruce and European beech. For Ecosyst 5(1):1–19
Pretzsch H, Schütze G, Uhl E (2013) Resistance of European tree species to drought stress in mixed versus pure forests: evidence of stress release by inter-specific facilitation. Plant Biol 15(3):483–495
Pretzsch H, Biber P, Dursky J (2002) The single tree-based stand simulator SILVA: construction, application and evaluation. For Ecol Manag 162:3–21
Pretzsch H, Schütze G, Biber P (2018b) Drought can favour the growth of small in relation to tall trees in mature stands of Norway spruce and European beech. For Ecosyst 5(1):20
Prodan M (1965) Holzmeßlehre. JD Sauerländer’s Verlag, Frankfurt am Main, p 644
Rais A, van de Kuilen JWG, Pretzsch H (2014) Growth reaction patterns of tree height, diameter, and volume of Douglas-fir (Pseudotsuga menziesii [Mirb.] Franco) under acute drought stress in Southern Germany. Eur J for Res 133(6):1043–1056
Riofrío J, del Río M, Pretzsch H, Bravo F (2017) Changes in structural heterogeneity and stand productivity by mixing Scots pine and Maritime pine. For Ecol Manag 405:219–228
Robinson AP, Ek AR (2000) The consequences of hierachy for modeling in forest ecosystems. Can J for Res 30:1837–1846
Rubio-Cuadrado Á, Camarero JJ, Del Rio M, Sánchez-González M, Ruiz-Peinado R, Bravo-Oviedo A, Montes F (2018) Drought modifies tree competitiveness in an oak-beech temperate forest. For Ecol Manage 429:7–17
Ryan MG, Yoder BJ (1997) Hydraulic limits to tree height and tree growth. Bioscience 47(4):235–242
Saadaoui E, Yahia KB, Dhahri S, Jamaa MLB, Khouja ML (2017) An overview of adaptative responses to drought stress in Eucalyptus spp. For Stud 67(1):86
Saunders A, Drew DM (2022) Measurements done on excised stems indicate that hydraulic recovery can be an important strategy used by Eucalyptus hybrids in response to drought. Trees 36(1):139–151
Schäfer KVR, Oren R, Tenhunen JD (2000) The effect of tree height on crown level stomatal conductance. Plant Cell Environ 23(4):365–375
Schäfer C, Rötzer T, Thurm EA, Biber P, Kallenbach C, Pretzsch H (2019) Growth and tree water deficit of mixed norway spruce and European beech at different heights in a tree and under heavy drought. Forests 10(7):577
Schmitt A, Trouvé R, Seynave I, Lebourgeois F (2020) Decreasing stand density favors resistance, resilience, and recovery of Quercus petraea trees to a severe drought, particularly on dry sites. Ann for Sci 77:1–21
Schwinning S, Weiner J (1998) Mechanisms determining the degree of size asymmetry in competition among plants. Oecologia 113(4):447–455
Seiler JR, Johnson JD (1988) Physiological and morphological responses of three half-sib families of loblolly pine to water-stress conditioning. For Sci 34(2):487–495
Sohn JA, Saha S, Bauhus J (2016) Potential of forest thinning to mitigate drought stress: a meta-analysis. For Ecol Manage 380:261–273
Steckel M, del Río M, Heym M, Aldea J, Bielak K, Brazaitis G, Pretzsch H (2020a) Species mixing reduces drought susceptibility of Scots pine (Pinus sylvestris L.) and oak (Quercus robur L., Quercus petraea (Matt.) Liebl.)–Site water supply and fertility modify the mixing effect. For Ecol Manag 461:117908
Steckel M, Moser WK, del Río M, Pretzsch H (2020b) Implications of reduced stand density on tree growth and drought susceptibility: a study of three species under varying climate conditions. Forests 11(6):627
Stephenson NL, Das AJ (2020) Height-related changes in forest composition explain increasing tree mortality with height during an extreme drought. Nat Commun 11(1):1–4
Stephenson NL, Das AJ, Ampersee NJ, Bulaon BM, Yee JL (2019) Which trees die during drought? The key role of insect host-tree selection. J Ecol 107(5):2383–2401
Stephenson NL, Das AJ, Condit R, Russo SE, Baker PJ, Beckman NG, Coomes DA, Lines ER, Morris WK, Ruger N, Alvarez E, Blundo C, Bunyavejchewin S, Chuyong G, Davies SJ, Duque A, Ewango CN, Flores O, Franklin JF, Grau HR, Hao Z, Harmon ME, Hubbell SP, Kenfack D, Lin Y, Makana JR, Malizia A, Malizia LR, Pabst RJ, Pongpattananurak N, Su SH, Sun IF, Tan S, Thomas D, van Mantgem PJ, Wang X, Wiser SK, Zavala MA (2014) Rate of tree carbon accumulation increases continuously with tree size. Nature 507:90–93
Sterba H (1981). Radial increment along the bole of trees: problems of measurement and interpretation. IUFRO Symposium, Sep 9–12, 1980; Mitteilungen d. Forstl. Bundesversuchsanstalt Wien 142 (1): 67–74
Sterba H (1984) Theoretische Überlegungen zur ertragskundlichen Auswirkung von Waldschäden. Forstarchiv 55(1):34–36
Strigul N (2012) Individual-based models and scaling methods for ecological forestry: implications of tree phenotypic plasticity. Sustain For Manag:359–384
Teets A, Fraver S, Weiskittel AR, Hollinger DY (2018) Quantifying climate–growth relationships at the stand level in a mature mixed-species conifer forest. Glob Change Biol 24(8):3587–3602
Temesgen H, Affleck D, Poudel K, Gray A, Sessions J (2015) A review of the challenges and opportunities in estimating above ground forest biomass using tree-level models. Scand J for Res 30(4):326–335
Thurm EA, Uhl E, Pretzsch H (2016) Mixture reduces climate sensitivity of Douglas-fir stem growth. For Ecol Manage 376:205–220
Thurnher C, Klopf M, Hasenauer H (2017) MOSES: A tree growth simulator for modelling stand response in Central Europe. Ecol Model 352:58–76
Toraño Caicoya A, Pretzsch H (2021) Stand density biases the estimation of the site index especially on dry sites. Can J for Res 51(7):1050–1064
Trouvé R, Bontemps J-D, Collet C, Seynave I, Lebourgeois F (2017) Radial growth resilience of sessile oak after drought is affected by site water status, stand density, and social status. Trees-Struct Funct 31(2):517–529
Trugman AT, Anderegg LD, Anderegg WR, Das AJ, Stephenson NL (2021) Why is tree drought mortality so hard to predict? Trends Ecol Evol 36:520–532
Ulrich B (1983) A concept of forest ecosystem stability and of acid deposition as driving force for destabilization. In: Effects of accumulation of air pollutants in forest ecosystems, Springer, Dordrecht, pp 1–29
Ulrich B (1994) Process Hierarchy in Forest Ecosystems: An Integrative Ecosystem Theory P. 353–398 in Effects of acid rain on forest processes. In: Godbold DL, Hüttermann A (eds) Wiley, Lissabon
Venturas MD, Todd HN, Trugman AT, Anderegg WRL (2021) Understanding and predicting forest mortality in the western United States using long-term forest inventory data and modeled hydraulic damage. New Phytol 230:1896–1910. https://doi.org/10.1111/nph.17043
Vergarechea M, Calama R, Fortin M, del Río M (2019) Climate-mediated regeneration occurrence in Mediterranean pine forests: a modeling approach. For Ecol Manage 446:10–19
Vitali V, Forrester DI, Bauhus J (2018) Know your neighbours: drought response of norway spruce, silver fir and douglas fir in mixed forests depends on species identity and diversity of tree neighbourhoods. Ecosystems 21(6):1215–1229
Von Buttlar J, Zscheischler J, Rammig A, Sippel S, Reichstein M, Knohl A, Jung M, Menzer O, Arain MA, Buchmann N, Cescatti A, Gianelle D, Kiely G, Law BE, Magliulo V, Margolis H, McCaughey H, Merbold L, Migliavacca M, Montagnani L, Oechel W, Pavelka M, Peichl M, Rambal S, Raschi A, Scott RL, Vaccari FP, van Gorsel E, Varlagin A, Wohlfahrt G, Mahecha MD (2018) Impacts of droughts and extreme-temperature events on gross primary production and ecosystem respiration: a systematic assessment across ecosystems and climate zones. Biogeosciences 15(5):1293–1318
Vose JM, Swank WT (1994) Effects of long-term drought on the hydrology and growth of a white-pine plantation in the southern Appalachians. For Ecol Manage 64:25–33
Wichmann L (2001) Annual variations in competition symmetry in even-aged Sitka spruce. Ann Bot 88(1):145–151
Weiskittel AR, Hann DW, Kershaw JA Jr, Vanclay JK (2011) Forest growth and yield modeling. Wiley
Wipfler P, Seifert T, Heerdt C, Werner H, Pretzsch H (2005) Growth of adult Norway spruce (Picea abies [L.] Karst.) and European beech (Fagus sylvatica L.) under free-air ozone fumigation. Plant Biol 7(06):611–618
Zang C, Pretzsch H, Rothe A (2012) Size-dependent responses to summer drought in Scots pine, Norway Spruce and Common Oak. Trees 26(2):557–569
Zapater M, Hossann C, Bréda N, Bréchet C, Bonal D, Granier A (2011) Evidence of hydraulic lift in a young beech and oak mixed forest using 18O soil water labelling. Trees-Struct Funct 25(5):885
Zhang Q, Shao MA, Jia X, Wei X (2017) Relationship of climatic and forest factors to drought- and heat-induced tree mortality. PLoS ONE 12:e0169770. https://doi.org/10.1371/journal.pone.0169770
Zöhrer F (1980) Forstinventur: ein Leitfaden für Studium und Praxis; mit 19 Tabellen. Parey, Hamburg und Berlin
Acknowledgements
The publication is part of the CARE4C project that has received funding from the European Union’s HORIZON 2020 research and innovation program under the Marie Skłodowska-Curie grant Agreement No 778322. Thanks go also to the Bayerische Staatsforsten (BaySF) for supporting the establishment and maintenance of the underlying long-term experiments and to the Bavarian State Ministry for Nutrition, Agriculture and Forestry for funding the project "Adaptability of trees and forest stands to drought stress" (klifW006 # Z3-7831-1/1015) and for the permanent support of the project W007, entitled “Long-term experimental plots for forest growth and yield research” (# 7831-22209-2013). Thanks go furthermore to the Junta de Castilla y León, Spain, and the European Union for funding the Projects VA183P20 (SMART—Bosques mixtos: Selvicultura, Mitigación, Adaptación, Resiliencia y Trade-offs) and CLU-2019-01—iuFOR Institute Unit of Excellence of the University of Valladolid through the ERDF "Europe drives our growth". We also thank anonymous reviewers for their constructive criticism.
Funding
Open Access funding enabled and organized by Projekt DEAL.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Peter Biber.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visithttp://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Pretzsch, H., del Río, M., Grote, R. et al. Tracing drought effects from the tree to the stand growth in temperate and Mediterranean forests: insights and consequences for forest ecology and management. Eur J Forest Res 141, 727–751 (2022). https://doi.org/10.1007/s10342-022-01451-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10342-022-01451-x