Abstract
Wild boar is among the most abundant ungulates in Europe and its spread is locally creating concerns as a major threat to biodiversity. However, through their rooting activity, wild boars could play an effective role in the creation of specific microhabitat resources for plants and animals. Here, we tested the hypothesis that wild boar affects the habitat suitability to threatened butterflies, by evaluating the influence of rooting on multiple key aspects of the biology and ecology of the Mediterranean endemic Zerynthia cassandra. Namely, we used Z. cassandra as a model to test the effects of wild boar rooting on adult foraging opportunities, host plant occurrence, and oviposition site selection. We found that herbaceous communities disturbed by wild boar rooting have a higher proportion of plants representing nectar resources for early-flying butterflies. We also discovered that wild boar rooting positively influences the occurrence and abundance of the larval host plant of Z. cassandra, as well as the butterfly site selection for egg-laying. Our results indicate that wild boars may locally prove beneficial to endangered butterflies by favoring habitat quality and availability, and their role as ecosystem engineers should thus be further investigated to improve species and habitat management and conservation actions.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Habitat degradation and loss are recognized among the primary causes for the widespread decline of many butterfly populations in Europe (Maes and Van Dyck 2001; van Swaay et al. 2010; Fox 2012). In this regard, the role of ecosystem engineers such as large ungulates can be locally predominant, as they can alter the availability of resources by modifying the physical state of biotic or abiotic materials (Jones et al. 1994) and, therefore, they can destroy, create and maintain habitats (Boogert et al. 2006; Byers et al. 2006; Streitberger and Fartmann 2013).
Wild ungulates have significantly spread throughout Europe in the last decades, as a response to a combination of reforestation, rural and agricultural-land abandonment, legislative changes and active reintroductions (Linnell et al. 2020), sometimes establishing overabundant populations (Valente et al. 2020). The activity of large ungulates is known to drive complex changes on plant populations and communities (Rooney and Waller 2003; Heckel et al. 2010; Royo et al. 2010; Rutherford and Schmitz 2010; Jensen et al. 2011; Murray et al. 2016) that may extend to other trophic levels (Nuttle et al. 2011; Lessard et al. 2012; Wheatall et al. 2013; Shelton et al. 2014). Among wild ungulates, wild boar (Sus scrofa) is a widespread ecosystem engineer affecting soil structure and composition, due to its rooting activity i.e., overturning vegetation in search for belowground plant parts, invertebrates and fungi (Baubet et al. 2003; Barrios-Garcia and Ballari 2012; Sandom et al. 2013b). Therefore, wild boar rooting has a distinctive impact on plant communities by setting back succession (Sandom et al. 2013a; Sims et al. 2014; Burrascano et al. 2015) with likely, yet poorly known to date, impacts on invertebrate communities (Carpio et al. 2014). Recent studies reported effects on butterfly oviposition and larval requirements due to soil disturbances by ecosystem engineers, e.g., European mole (Streitberger and Fartmann 2013; Streitberger et al. 2014) and meadow ant (Streitberger and Fartmann 2016). However, only few researches were performed on the significance of wild boar rooting effects on butterfly habitat requirements (de Schaetzen et al. 2018).
For a better understanding of the effects of habitat perturbations on butterfly populations, particular attention should be given to the availability of food sources, as these provide a defining feature of habitat quality and set an upper limit on carrying capacity (Brown et al. 2004; Krauss et al. 2005; Dennis 2010). Butterflies may require a variety of microhabitat conditions to provide all the resources needed during their life stages (Dennis et al. 2006; Wynhoff et al. 2008; Curtis et al. 2015). The ecological requirements of adult butterfly species have long been missed in the scientific literature, since most studies only assessed the relationships between the target species and its host plant, an aspect directly connected to oviposition and larval survival (e.g., López Munguira et al. 2009; García-Barros and Fartmann 2009; Cini et al. 2021). Specific larval habitat requirements should be generally met at a small spatial scale, such as the availability, abundance and quality of specific host plant species (López Munguira et al. 2009; Curtis et al. 2015; de Schaetzen et al. 2018). Yet, in most cases, occurrence or abundance of the host plant is irrelevant to adult butterflies’ survival, since these opportunistically feed upon the nectar of plants flowering during their adult-stage phenological phase (Curtis et al. 2015; Stefanescu and Traveset 2009). Effects of habitat changes are even stronger on species with low dispersal ability, including many terrestrial invertebrates (Thomas et al. 2004). Among butterflies, range-restricted species such as local endemics are severely threatened by habitat modification and loss (Bonelli et al. 2018), also due to several ecological and biological traits that make them more prone to extinction risk e.g., fewer generations per year, lower diversity of suitable host plants, earlier and shorter flying period (López-Villalta 2010). The Italian Peninsula in particular represents a butterfly biodiversity hotspot within the Mediterranean biome (Menchetti et al. 2021), with highest rates of endemic species that urgently need a deeper understanding of their threats to secure proper management, such as the Italian endemic butterfly Zerynthia cassandra (Lepidoptera, Papilionidae).
Z. cassandra was recently recognized as a sister species of the European-listed butterfly Zerynthia polyxena (Dapporto 2010; Zinetti et al. 2013) and has seen declining populations during the last decades, with documented local extinctions (Bonelli et al. 2011; Vovlas et al. 2014). Z. cassandra is a single brooded species with very limited dispersal abilities, and strictly dependent on the occurrence of host plants of the genus Aristolochia, such as A. rotunda and A. lutea (Vovlas et al. 2014; Camerini et al. 2018; Ghesini et al. 2018; Cini et al. 2019, 2021; Cagnetta et al. 2020). Recent studies provided important insights on the microhabitat requirements of the sedentary and oligophagous Z. cassandra, underlining its marked selectivity in terms of plant and site features for oviposition and larval growth (Vovlas et al. 2014; Cini et al. 2019). All these traits make Z. cassandra highly susceptible to extinction due to micro-habitat changes (e.g., disappearance of the host plant, grassland encroachment: Bonelli et al. 2011, Camerini et al. 2018, Ghesini et al. 2018), and thus represent an excellent candidate model to assess the effects of ungulate activities on the habitat of diurnal lepidopterans.
Here we test the hypothesis that wild boars act as environmental engineers in grasslands by changing micro-habitat characteristics when rooting, in turn affecting suitability to threatened butterflies, using the endemic Z. cassandra as a model. We disentangled the direct and indirect effects of wild boars on Z. cassandra by evaluating the influence of rooting on multiple key aspects of Z. cassandra biology and ecology, namely adult foraging opportunities, host plant occurrence, and oviposition site selection, according to the following hypotheses and predictions:
-
1)
Wild boar rooting increases foraging opportunities to adult Z. cassandra; we predict that changes in plant species composition within a grassland landscape due to rooting will favor early flowering and entomophilous species, in turn providing suitable nectar resources to adult Z. cassandra;
-
2)
Wild boar rooting favors the host plant of Z. cassandra; we expect the host plant (A. clusii) to be favored by rooting due to its low palatability and capability to grow in disturbed soil i.e., we predict a spatial association between rooting and A. clusii;
-
3)
Wild boar rooting influences oviposition site selection by Z. cassandra; by changing plant community structure (1) and favoring the host plant (2), we predict that rooting will increase the probability of a potentially suitable site to be chosen by Z. cassandra for egg-laying.
Materials and methods
Study area and species
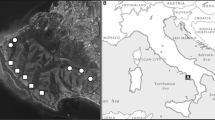
The study was carried out within a network of Natura 2000 sites belonging to Murgia calcareous plateau in Apulia region (southeast Italy; Fig. 1a-b), namely ZSC/SPA IT9120007 “Murgia Alta” and ZSC/ZPS IT9130007 “Area delle Gravine” (41.185°–40.530° N, 16.085°–17.225° E).
Ranging from 100 to 700 m a.s.l., this area is characterized by its compact limestone plateau, with lack of surface watercourses. The climate is mediterranean, with annual temperatures from 7 to 9 °C in January to 25–27 °C in July/August, and rainfall of 500–700 mm*yr− 1 mostly in autumn–winter, with occasional snowfall above 500 m a.s.l.
The landscape is characterized by a gradient of vegetation and land use influenced by elevation, geography and historical features. From northwest to southeast, the wide extensions of dry grasslands and cereal crops, with residual patches of downy oak (Quercus pubescens s.l.) woodland, become gradually richer in sub-mediterranean forests with Trojan oak (Quercus trojana Webb) and evergreen Mediterranean forests with Aleppo pine (Pinus halepensis Mill.) and holm oak (Quercus ilex L.).
Wild boar (S. scrofa) represents the only wild ungulate species locally, with a population density of about 20–50 individuals/100 ha (Gaudiano et al. 2022). Wild boar population levels are not maintained by hunting policies, although little sport hunting occurs outside protected areas.
Zerynthia cassandra is widespread in the agro-forestry mosaic (Cagnetta et al. 2020; Labadessa et al. 2021), especially in the southeastern part of the study area, and is associated with the occurrence of Aristolochia clusii as a primary host plant. The distribution of other potential host plants, such as A. rotunda and A. lutea, is restricted to few sites at higher elevations and in forest areas.
At a smaller scale (Fig. 1c), 6 forest clearings of various shapes and sizes (from 0.06 to 0.49 ha) were selected in the southeastern portion of the studied area (40.6570°N – 17.1035°E – 400 m a.s.l.). These sites were selected as locally representing important sites for Z. cassandra and its host plant A. clusii, in conjunction with the occurrence of noticeable activity by wild boar, and in absence of domestic livestock and recent human land uses. These sites consist of previously cultivated lands, now covered with semi-natural herbaceous vegetation with scattered encroachment of shrubs and small trees (Pyrus spinosa, Prunus spinosa, Quercus ilex).
Sampling design
To evaluate the potential effects of wild boar rooting on factors that may influence the habitat use by adults of Z. cassandra, we carried out vegetation relevés from 2018 to 2021 in different semi-natural herbaceous communities in the study area (Fig. 1b). Within each grassland site, we sampled plant species cover in 1 × 1 m plots placed in surfaces that were recently (less than 1 year) disturbed by wild boar rooting. Recent wild boar rooting was readily distinguished as large disturbed patches (> 1 m2) with a lower microtopography, similar to sod cutting (Welander 2000; Burrascano et al. 2015; Sandom et al. 2013b; Sims et al. 2014). These patches were created in the season prior to this study, generally during autumn-winter (Welander 2000; Sims et al. 2014), and were partially recolonized by vegetation. Rooting events were not observed during the duration of this study. With each plot affected by rooting, we paired a 1 × 1 m control plot in the closest portion of the adjoining undisturbed vegetation, i.e. presenting similar environmental conditions, as a reference for plant communities before rooting. In total, we sampled 18 rooted and 18 non-rooted plots.
For each vegetation plot, we quantified the relative abundance (cover) and relative species richness of potential nectar resource plants, upon the overall plant cover and richness, respectively. Among the sampled plant species, we selected those species that represent potential nectar resources for adults of Z. cassandra. Since Z. cassandra is known to feed on several herbaceous nectar resources (Ghesini et al. 2018; Cini et al. 2021), and thus is not strictly associated with peculiar plant traits, we selected 38 plant species whose flower morphology allows feeding by butterflies and whose flowering period overlaps with the phenology of adult Z. cassandra (i.e., between March and May; see Supplementary table S1 for the list of species).
In order to assess the effects of rooting on Z. cassandra host plant, in May 2020 we exhaustively assessed the distribution of Aristolochia clusii plants and wild boar rooting in the study sites (Fig. 1c). For this purpose, we overlaid a regular 2 × 2 m mesh grid on the selected forest clearings, being these considered fully suitable for the occurrence of A. clusii. In total, we considered 5,368 cells. In each cell, we independently estimated the surface occupied by host plant patches and wild boar rooting. In each cell we also recorded the average grass height, shadow degree (0 = fully irradiated; 0.5 = half shady; 1 = completely shady) and the percentage cover of grass layer, rocks and stones.
In order to understand the habitat features promoting oviposition, we adopted a sampling scheme based on previous studies (Vovlas et al. 2014; Ghesini et al. 2018; Cini et al. 2019, 2021). Knowing the exact distribution of A. clusii in the study sites (Fig. 1.c), for each plant patch we measured: (i) the occurrence of Z. cassandra eggs or early-staged larvae; (ii) the extent of the plant patch (m2); (iii) an indication of the growing state of the plant patch (0.5 = depleted and/or withered patch; 1 = dense and luxuriant patch). Since there was a little difference in the number of eggs per plant (range: 0–2 eggs), we considered egg presence/absence as a mere indication of oviposition event. As plant stems grow in clusters (mainly clonal or belonging to the same tuber), each plant cluster was treated as a single sample in our data set regardless of the number of batches it carried, while we measured the extent of each plant cluster. In the area of 1 m radius around each plant we also measured the following parameters: (i) occurrence of recent wild boar rooting adjoining the host plant patch; (ii) height of the surrounding vegetation; percentage of coverage of (iii) herbaceous layer, (iv) rocks and (v) stones; (vi) shadow degree. In total, we sampled 138 patches.
Statistical analyses
Wild boar rooting and foraging opportunities to Z. cassandra
To evaluate the potential effect of wild boar rooting on the availability of nectar resource for adults of Z. cassandra, we compared the rate of flowering plants between paired vegetation plots, i.e. disturbed vs. undisturbed by wild boar rooting. We used paired Student t-tests to assess differences between paired plots (N = 18). T-test was run using SPSS 16.0 (SPSS, Chicago, IL, US).
Wild boar rooting and the host plant of Z. cassandra
To test for the environmental factors that drive the occurrence and abundance of A. clusii within each grid cell, we run Generalized Linear Mixed Models (GLMMs) using a binomial (occurrence) and normal (percent cover) error distributions, and log-link functions. We modeled the occurrence and percent cover by A. clusii within each sampled grid cell as a function of the percent of rock and grass cover, degree of shadow, and percent of wild boar rooting activity; we used clearing (site) identity as a random effect to account for potential inter-site differences. In each model, significant effects were considered those with p < 0.05 and 0.95 confidence intervals of the effect size not including 0. Models were run using the lme4 package for R (Bates 2014).
Wild boar rooting and oviposition site selection by Z. cassandra
To quantify the direct and indirect effects of wild boar rooting and plot-level environmental factors on the reproduction of Z. cassandra, we performed a path analysis using the lavaan package (Rosseel et al. 2022). Path analysis is a variant of Structural Equation Modeling (SEM) that allows to test an a priori hypothesis about causal relationships among variables (Wootton 1994), and that essentially performs a multiple regression test to a set of relationships, based on pre-defined hypotheses among the variables of interest. As a result, this approach allows to decompose and estimate the relative strengths of direct and indirect effects of factors upon a specific response variable. The designed path in our case study included the effects of wild boar activity, A. clusii abundance and status, land cover variables (grass and rock cover percent), shadow degree and grass height, on the reproduction of Z. cassandra, assessed by the presence/absence of eggs and caterpillars, within a given A. clusii patch. Relationships among the explaining variables were also tested, together with the indirect effects on Z. cassandra, based on both published evidence and potential effects that may be expected (Fig. 2; Supplementary Table S2). Recent studies documented that the oviposition of Z. cassandra is positively influenced by the abundance and growing conditions of its host plants, and by solar irradiation (Vovlas et al. 2014; Camerini et al. 2018; Ghesini et al. 2018; Cini et al. 2019, 2021), while it is negatively affected by grass cover (Cini et al. 2021). Wild boar rooting directly halters the structure and composition of herbaceous vegetation (Burrascano et al. 2015; Sandom et al. 2013a; Sims et al. 2014), and thus is deemed to have potential effects on host plant growth and oviposition patterns. Overall model fit was assessed with a chi-squared goodness-of-fit test, with the root-mean-square error of approximation index (RMSEA), and the comparative fit index (CFI). For all the tested relationships, we evaluated standardized coefficients and associated p-values, considering as significant those with values > 0.1 and < 0.05, respectively.
Results
Wild boar rooting and foraging opportunities to Z. cassandra
When compared to undisturbed herbaceous vegetation, plant communities affected by wild boar rooting showed a significantly higher cover of species representing nectar resource for Z. cassandra (Fig. 3). The proportion of the number of species richness representing foraging opportunities did not significantly change in rooted sites (Fig. 3).
Box-plot showing the proportion of cover (a: t = -3.20; dF = 17.00; p = 0.005) and richness (b: t = -0.47; dF = 17.00; p = 0.65) of feeding resources in not-rooted and rooted vegetation. Relative values of cover (a) and species richness (b) of potential nectar resource plants are quantified upon the overall plant cover and richness, respectively
Wild boar rooting and the host plant of Z. cassandra
Both the occurrence and abundance of A. clusii were significantly influenced by plot-level environmental conditions, with models explaining 75% and 66% of the observed variance, respectively. The amount of wild boar rooting had significant positive effects on both occurrence and abundance of A. clusii (Fig. 4). Similarly, percent cover of rocks within the grid cell influenced both occurrence and abundance of A. clusii, yet negatively. Conversely, the percent amount of herbaceous species and the degree of shadow at plot level negatively influenced only A. clusii occurrence.
Linear relationships between the occurrence (a-d) and abundance (approximated as the percent cover at 2 × 2 m grid square; e-h) of Aristolochia clusii at grid-square level (N = 5,368) and a set of plot-level environmental factors. Significance: **=p < 0.01; ***=p < 0.001; n.s.=p > 0.05. Rooting = percent of ground with evidence of wild boar rooting activity; Shadow degree = measure of shadowing due to tree crowns (0 = full sun; 1 = full shadow); Rock cover = percent of ground covered in rocks; Grass cover = percent of ground covered by herbaceous vegetation (A. clusii excluded). Regression lines estimated by generalized linear models with binomial (occurrence) and normal (abundance) error distributions. Shaded areas indicate 95% confidence intervals. Estimate ± standard errors of significant predictors: (a) 0.06 ± 0.01; (b) -0.02 ± 0.01; (c) -0.06 ± 0.01; (d) -0.10 ± 0.03; (e) 0.04 ± 0.01; g) -0.01 ± 0.00
Wild boar rooting and oviposition site selection by Z. cassandra
Among the 138 A. clusii patches sampled, 48 (32.4%) hosted eggs or early staged larvae of Z. cassandra (Fig. 5). Overall, the structural model satisfactorily fit the data (χ2 = 0.02, p = 0.09; RMSEA < 0.1, CFI = 0.90), highlighting that the occurrence of eggs and larvae of Z. cassandra was directly favored by wild boar rooting activity (standardized coefficient: 0.21, p < 0.01), A. clusii cover (0.24, p < 0.001), and percent cover by herbaceous species (0.20, p < 0.05), and negatively affected by grass height (-0.19, p < 0.001). Interestingly, indirect effects were also evident, with rooting activity showing positive effects on the cover of A. clusii (0.21, p < 0.05) and negative on the percent cover of herbaceous species (-0.78, p < 0.001). The degree of shadow only showed positive yet negligible effect on grass cover (0.07, p < 0.05), and on grass height (0.22, p < 0.001), as well as on the status of A. clusii individuals (0.12, p < 0.05), yet the latter was also weak and had no further significant effect on the considered variables (Fig. 5).
Path analysis explaining oviposition site selection by Zerynthia cassandra on Aristolochia clusii patches, based on presence/absence of eggs or larvae on each patch (N = 138). Reported values for each relationship are standardized regression coefficients. Significance: * = p < 0.05; ** = p < 0.01; *** = p < 0.001
Discussion
Wild boar rooting and foraging opportunities to Z. cassandra
Our findings show that herbaceous communities disturbed by wild boar rooting have a significantly higher proportion of plants representing nectar resources for Z. cassandra, thus confirming our first hypothesis. Despite rooting did not modify the richness of flowering sources, it promoted their abundance compared to the overall herbaceous cover. By altering soil structure and reducing the cover of well-established competitive species, such as perennial grasses and forbs, wild boar rooting is able to set successional pathways back to early-staged communities, therefore favoring short-lived pioneer species (Burrascano et al. 2015; Sandom et al. 2013a; Sims et al. 2014). Due to their adaptation to ephemeral and xeric conditions, Mediterranean therophyte-rich communities are frequently characterized by an early phenology (Grime 1979), thus providing a high abundance of flowers at the beginning of spring. The lack of distinct changes in plant species richness is not surprising as an effect of the balance between positive and negative responses of single species e.g., when new colonizers replace formerly existing species, thus resulting in an overall neutral response in terms of species richness (Davis et al. 2000; Ries et al. 2004; Labadessa et al. 2017).
Similarly to Z. cassandra, other insects directly associated with early flowering species are also likely to benefit from wild boar rooting, i.e. many Lepidoptera and Hymenoptera (Munguía-Rosas et al. 2011; Riedinger et al. 2014).
Wild boar rooting and the host plant of Z. cassandra
The occurrence and abundance of A. clusii in our study area were positively influenced by the amount of wild boar rooting, as we predicted in our second hypothesis. Within homogeneous sites similarly suitable for A. clusii, patches of this species tended to be more common and abundant in association with rooting activities. This evidence could be due to a set of peculiar traits of A. clusii, i.e. associated with (i) vegetative propagation, (ii) gamic reproduction, (iii) plant phenology, and (iv) toxic/repulsive compounds. First, this species often grows in clonal clusters derived by the fragmentation of tubers, which can benefit from shallow soil perturbation that moves and disperses vegetative propagules. Similarly positive effects of rooting were also observed in the UK (Sims 2005), where geophytes such as Anemone nemorosa, Ranunculus ficaria and Hyacinthoides non-scripta were significantly more abundant in rooted plots than in control ones, presumably due to rhizome fragmentation by wild boar rooting activity and subsequent stem re-growth. Such effects on vegetation were though only detectable with 1–2 years of time lag (Sims et al. 2014), indicating that short-term responses to rooting may be misleading when assessing the impacts of wild boars on vegetation, and consequently on invertebrates (Scandurra et al. 2016). Second, like other herbaceous European Aristolochia species (Örvössy et al. 2014), A. clusii is known to prefer moderately nitrogen-rich soil, such as abandoned cultivations, roadsides and forest clearings. These traits are both favored by wild boar rooting, which increases nutrient availability in the soil (Singer et al. 1984; Siemann et al. 2009; Cuevas et al. 2012). Third, if compared to the surrounding annual grassland plants, A. clusii shows a relatively late development of epigean stems, in April-May. The effect of rooting, which is mostly concentrated in the wet season (from winter to early spring), may be highly negative on early-developing plants, while nearly negligible for A. clusii. Consequently, A. clusii may thus benefit from the reduction of competition, promptly occupying the place cleared of more competitive species. Fourth, based on the general knowledge of toxic and odorous terpenoids contained in the genus Aristolochia (Wu et al. 2004), which were also demonstrated to be toxic to ungulates (Barakat et al. 1983), it is likely that A. clusii have a repulsive effect on wild boar.
Wild boar rooting and oviposition site selection by Z. cassandra
We documented that wild boar rooting, coupled with local biotic and abiotic conditions, influences the oviposition of Z. cassandra, as predicted by our third hypothesis. The importance of suitable host plants and microhabitat conditions for egg-laying was confirmed in several populations of Z. cassandra (Vovlas et al. 2014; Camerini et al. 2018; Ghesini et al. 2018; Cini et al. 2019) and other endangered grassland butterflies (Wiklund 1984; de Schaetzen et al. 2018). However, our findings first demonstrate that the occurrence of eggs and larvae of Z. cassandra was directly favored by wild boar rooting activity. When compared to a set of environmental variables that are well known to be key for the selection of oviposition site, i.e. sun exposure, vegetation height and cover, and host plant growing condition (Vovlas et al. 2014; Camerini et al. 2018; Ghesini et al. 2018; Cini et al. 2019), rooting influence was even more significant at the local scale. Similarly positive effects of soil perturbation by ecosystem engineers were also reported with regard to the oviposition patterns of other butterfly species, i.e. the case of mound-building mammals (Streitberger and Fartmann 2013; Streitberger et al. 2014) and arthropods (Streitberger and Fartmann 2016). Besides the possible indirect effects on host plant occurrence and abundance, direct effects of rooting can be explained in terms of increase of accessibility and/or visibility of oviposition sites. Host plants adjoining rooted surfaces are less sheltered by taller grasses, with stems more easily reachable by flying females. This condition can be especially important for a weak flier such as Z. cassandra (Vovlas et al. 2014), and in the case of A. clusii, which often grows shorter than the surrounding herbs and grasses. The reduction of grasses leaning against the host plant also results in a more direct exposure to sun, locally determining warmer microclimatic conditions. Indeed, warmer temperature is deemed to favor larval development as effect of increased metabolism (Ghesini et al. 2018; Cini et al. 2019, 2021), as also recorded in immature stages of other Papilionidae butterflies (Scriber and Lederhouse 1983; Valimaki and Itamies 2005).
Moreover, our study first documents the importance of the neglected Italian endemic species A. clusii as a food resource for the larvae of Z. cassandra. So far, studies on Z. cassandra host plant were based on observations regarding A. rotunda and A. lutea (Altini et al. 2007; Vovlas et al. 2014; Camerini et al. 2018; Ghesini et al. 2018; Cini et al. 2019). However, A. clusii represents the primarily available host plant in many sites within the distributional range of Z. cassandra, e.g. in most of the peninsular portion of Apulia region (Nardi 1984). This evidence, partially due to a possible misidentification of Aristolochia species in the field, implies reconsidering some of the previous observations in Apulia region, which should be partially referred to A. clusii rather than A. rotunda (Vovlas et al. 2014; Altini et al. 2007).
Conclusion
Taken together, our results indicate that, at least within the conditions met in our study area, wild boar rooting activity may prove beneficial to the habitat quality and availability of early-flying butterflies by increasing food resources, favoring host plants and influencing oviposition site selection, as exemplified by the effects we recorded on the endangered Italian endemic Z. cassandra. Wild boars are still on the rise throughout Europe (Melis et al. 2006), sometimes creating concerns due to overabundant populations that damage crops or threaten wildlife (Valente et al. 2020). In our case, boars’ density was high, yet rooting intensity was relatively low, as typical for grassland habitats (de Schaetzen et al. 2018; Ferretti et al. 2021), and restricted to the winter season, two factors that should suggest caution before generalizing our results to the great variety of ecological contexts where the species occurs. The spread of the wild boar as environmental engineer may thus locally prevent some of the threats that hamper the persistence and conservation of invertebrates living in open habitats e.g., grassland encroachment (Bonelli et al. 2018). Yet, high ungulate densities may also provide other negative effects (e.g., by overgrazing and trampling; Ramirez et al. 2021) that may counteract the positive effects of rooting. More research is thus certainly needed to assess the rooting pressure optimum levels that lead to positive effects on endangered grassland butterflies and other protected taxa potentially affected by this ungulate (e.g., orchids). Such positive cascading effects on habitat quality may though represent key assets in the local management of wild boars and invertebrates of conservation concern, and should thus be included in the decision-making process when boar management actions are planned.
References
Altini E, Tarasco E, Gallo M, Triggiani O (2007) I lepidotteri diurni della Riserva naturale regionale orientata dei ‘‘Laghi di Conversano e Gravina di Monsignore’’ e note di biologia di Zerynthia polyxena (Lepidoptera, Papilionidae). Atti XXI Congresso Nazionale Italiano di Entomologia, Campobasso 11–16 giugno:74
Barakat SEM, Wasfi IA, Adam SEI (1983) The toxicity of Aristolochia bracteata in Goats. Vet Pathol 20:611–616
Barrios-Garcia MN, Ballari SA (2012) Impact of wild boar (Sus scrofa) in its introduced and native range: a review. Biol Invasions 14(11):2283–2300
Bates D (2014) Package ‘lme4’. Available at: https://cran.r-project.org/web/packages/lme4/index.html
Baubet E, Ropert-Coudert Y, Brandt S (2003) Seasonal and annual variations in earthworm consumption by wild boar (Sus scrofa L.). Wildl Res 30(2):179–186
Bonelli S, Casacci LP, Barbero F, Cerrato C, Dapporto L, Sbordoni V, Scalercio S, Zilli A, Battistoni A, Teofili C, Rondinini C, Balletto E (2018) The first red list of italian butterflies. Insect Conserv Divers 11:506–521
Bonelli S, Cerrato C, Loglisci N, Balletto E (2011) Population extinctions in the italian diurnal lepidoptera: an analysis of possible causes. J Insect Conserv 15:879–890. https://doi.org/10.1007/s10841-011-9387-6
Boogert NJ, Paterson DM, Laland KN (2006) The implications of niche construction and ecosystem engineering for conservation biology. Bioscience 56(7):570–578
Brown JH, Gillooly JF, Allen AP, Savage VM, West GB (2004) Toward a metabolic theory of ecology. Ecology 85:1771–1789
Burrascano S, Copiz R, Del Vico E, Fagiani S, Giarrizzo E, Mei M, Mortellilti A, Sabatini FM, Blasi C (2015) Wild boar rooting intensity determines shifts in understorey composition and functional traits. Community Ecol 16(2):244–253
Byers JE, Cuddington K, Jones CG, Talley TS, Hastings A, Lambrinos JG, Crooks JA, Wilson WG (2006) Using ecosystem engineers to restore ecological systems. TREE 21(9):493–500
Cagnetta G, Labadessa R, Altini E, Clemente D, Vovlas A (2020) New records and an updated checklist of the butterfly fauna (Lepidoptera: Papilionoidea) of Puglia, south-eastern Italy. Phegea 48:113–121
Camerini G, Groppali R, Minerbi T (2018) Observations on the ecology of the endangered butterfly Zerynthia cassandra in a protected area of Northern Italy. J Insect Conserv 22:41–49. https://doi.org/10.1007/s10841-017-0036-6
Carpio AJ, Castro-López J, Guerrero-Casado J, Ruiz-Aizpurua L, Vicente J, Tortosa FS (2014) Effect of wild ungulate density on invertebrates in a Mediterranean ecosystem. Anim Biodivers Conserv 37(2):115–125
Cini A, Benetello F, Platania L, Bordoni A, Boschi S, Franci E, Ghisolfi G, Pasquali L, Negroni R, Dapporto L (2021) A sunny spot: habitat management through vegetation cuts increases oviposition in abandoned fields in an endemic Mediterranean butterfly. Insect Conserv Divers 14:582–596. https://doi.org/10.1111/icad.12489
Cini A, Bordoni A, Ghisolfi G, Lazzaro L, Platania L, Pasquali L, Negroni R, Benetello F, Coppi A, Zanichelli F, Dapporto L (2019) Host plant selection and differential survival on two Aristolochia L. species in an insular population of Zerynthia cassandra. J Insect Conserv 23:239–246
Cuevas MF, Mastrantonio L, Ojeda RA, Jaksic FM (2012) Effects of wild boar disturbance on vegetation and soil properties in the Monte Desert. Argentina Mamm 77:299–306
Curtis RJ, Brereton TM, Dennis RLH et al (2015) Butterfly abundance is determined by food availability and is mediated by species traits. J Appl Ecol 52:1676–1684. https://doi.org/10.1111/1365-2664.12523
Dapporto L (2010) Speciation in Mediterranean refugia and postglacial expansion of Zerynthia polyxena (Lepidoptera, Papilionidae). J Zool Syst Evol Res 48:229–237
Davis MA, Grime PJ, Thompson K (2000) Fluctuating resources in plant communities: a general theory of invisibility. J Ecol 88:528–534
Dennis RLH (2010) A resource-based habitat view for conservation: butterflies in the british landscape. Wiley, Chichester
Dennis RLH, Shreeve TG, Van Dyck H (2006) Habitats and resources: the need for a resource-based definition to conserve butterflies. Biodivers Conserv 15(6):1943–1966
de Schaetzen F, van Langevelde F, De Wallis MF (2018) The influence of wild boar (Sus scrofa) on microhabitat quality for the endangered butterfly Pyrgus malvae in the Netherlands. J Insect Conserv 22:51–59
Ferretti F, Lazzeri L, Mori E, Cesaretti G, Calosi M, Burrini L, Fattorini N (2021) Habitat correlates of wild boar density and rooting along an environmental gradient. J Mammal 102:1536–1547
Fox R (2012) The decline of moths in Great Britain: a review of possible causes. Insect Conserv Divers 6:5–19
Garcia Barros E, Fartmann T (2009) Butterfly oviposition: sites, behaviour, modes. In: Settele J, Shreeve T, Konvicka M, Van Dyck H (eds) Ecology of butterflies in Europe. Cambridge University Press, Cambridge
Gaudiano L, Pucciarelli L, Frassanito AG, Mori E, Morimando F, Silvestri FM, Sorino R, Viviano A, Corriero G (2022) Spatio–temporal behaviour of female wild boar in an agro–forestry–pastoral landscape of Southern Italy. Mammal Res 67:163–172
Ghesini S, Magagnoli S, Marini M (2018) Biology and conservation of Zerynthia cassandra (Lepidoptera, Papilionidae) in semi-natural environments and agricultural landscapes. J Insect Conserv 22:151–161. https://doi.org/10.1007/s10841-018-0049-9
Grime JP (1979) Plant strategies and vegetation processes. Wiley & Sons, Chichester
Heckel CD, Bourg NA, McShea WJ, Kalisz S (2010) Nonconsumptive effects of a generalist ungulate herbivore drive decline of unpalatable forest herbs. Ecology 91:319–326
Jensen NR, Webster CR, Witt JC, Grant JB (2011) Ungulate winter habitat selection as a driver of herbaceous-layer heterogeneity in northern temperate forests. Ecosphere 2:67
Jones CG, Lawton JH, Shachak M (1994) Organisms as ecosystem engineers. Oikos 69(3):373–386
Krauss J, Steffan-Dewenter I, Müller CB, Tscharntke T (2005) Relative importance of resource quantity, isolation and habitat quality for landscape distribution of a monophagous butterfly. Ecography 28:465–474. https://doi.org/10.1111/j.0906-7590.2005.04201.x
Labadessa R, Alignier A, Cassano S, Forte L, Mairota P (2017) Quantifying edge influence on plant community structure and composition in semi-natural dry grasslands. Appl Veg Sci 20:572–581
Labadessa R, Cagnetta G, Désaphy J-F, Bonifacino M, Dodaro G, Festa D, Monterastelli E, Papa VE, Zollo L, Balletto E, Dapporto L (2021) Using occurrence data to evaluate extinction reveals a strong resilience of butterflies in a National Park of Southern Europe (Alta Murgia National Park). J Insect Biodivers 28:001–012
Lessard J-P, Reynolds N, Bunn WA, Genung MA, Cregger MA, Felker-Quinn E, Barrios-Garcia N, Stevenson ML, Lawton M, Brown CB, Patrick M, Rock J, Jenkins MA, Bailey JK, Schweitzer JA (2012) Equivalence in the strength of deer herbivory on above and below ground communities. Basic App Ecol 13:59–66
Linnell JD, Cretois B Nilsen EB, Rolandsen CM, Solberg EJ, Veiberg V, Kaczensky P, Van Moorter B, Panzacchi M, Rauset GR, Kaltenborn B (2020) The challenges and opportunities of coexisting with wild ungulates in the human-dominated landscapes of Europe’s Anthropocene. Biol Conserv 244:108500
López Munguira M, García-Barros E, Martín Cano J (2009) Butterfly herbivory and larval ecology. In: Van Dyck H, Settele J, Shreeve T, Konvička M (eds) Ecology of butterflies in Europe. Cambridge University Press, Cambridge
Lopez-Villalta JS (2010) Ecological trends in endemic Mediterranean butterflies. Bull Insectology 63:161–170
Maes D, van Dyck H (2001) Butterfly diversity loss in Flandres (north Belgium): Europe’s worst case scenario? Biol Conserv 99:263–276
Melis C, Szafrańska PA, Jędrzejewska B, Bartoń K (2006) Biogeographical variation in the population density of wild boar (Sus scrofa) in western Eurasia. J Biogeogr 33:803–811
Menchetti M, Talavera G, Cini A, Salvati V, Dincă V, Platania L, Bonelli S, Balletto E, Vila R, Dapporto L (2021) Two ways to be endemic. Alps and Apennines are different functional refugia during climatic cycles. Mol Ecol 30:1297–1310
Munguía-Rosas MA, Ollerton J, Parra-Tabla V, De‐Nova JA (2011) Meta-analysis of phenotypic selection on flowering phenology suggests that early flowering plants are favoured. Ecol 14:511–521
Murray BD, Webster CR, Jenkins MA, Saunders MR, Haulton GS (2016) Ungulate impacts on herbaceous-layer plant communities in even-aged and uneven-aged managed forests. Ecosphere 6:e01378
Nardi E (1984) The genus «Aristolochia» L. (Aristolochiaceae) in Italy. Webbia 38:221–300. https://doi.org/10.1080/00837792.1984.10670308
Nuttle T, Yerger EH, Stoleson SH, Ristau TE (2011) Legacy of top-down herbivore pressure ricochets back up multiple trophic levels in forest canopies over 30 years. Ecosphere 2:1–11. https://doi.org/10.1890/ES10-00108.1
Örvössy N, Kőrösi Á, Batary P, Vozar A, Peregovits L (2014) Habitat requirements of the protected southern festoon (Zerynthia polyxena); adult, egg and larval distribution in a highly degraded habitat complex. Acta Zool Acad Sci Hung 60:371–387
Ramirez JI, Jansen PA, den Ouden J, Moktan L, Herdoiza N, Poorter L (2021) Above-and below-ground cascading effects of wild ungulates in temperate forests. Ecosyst 24:153–167
Riedinger V, Renner M, Rundlöf M, Steffan-Dewenter I, Holzschuh A (2014) Early mass-flowering crops mitigate pollinator dilution in late-flowering crops. Landsc Ecol 29:425–435
Ries L, Fletcher RJ, Battin J, Sisk TD (2004) Ecological responses to habitat edges: mechanisms, models, and variability explained. Annu Rev Ecol Evol Syst 35:491–522
Rooney TP, Waller DM (2003) Direct and indirect effects of white-tailed deer in forest ecosystems. For Ecol Manag 181:165–176
Rosseel Y, Oberski D, Byrnes J, Vanbrabant L, Savalei V, Merkle E, Hallquist M, Rhentulla M, Katsikatsou M, Barendse M, Scharf F, Du H (2022) Package ‘lavaan’. Available at: https://cran.r-project.org/web/packages/lavaan/index.html
Royo AA, Collins R, Adams MB, Kirschbaum C, Carson WP (2010) Pervasive interactions between ungulate browsers and disturbance regimes promotes temperate forest herbaceous diversity. Ecology 91:93–105
Rutherford AC, Schmitz OJ (2010) Regional-scale assessment of deer impacts on vegetation within western Connecticut, USA. J Wildl Manag 74:1257–1263
Sandom CJ, Hughes J, Macdonald DW (2013a) Rewilding the scottish highlands: do wild boar, Sus scrofa, use a suitable foraging strategy to be effective ecosystem engineers? Restor Ecol 21(3):336–343
Sandom CJ, Hughes J, Macdonald DW (2013b) Rooting for rewilding: quantifying wild boar’s Sus scrofa rooting rate in the scottish highlands. Restor Ecol 21(3):329–335
Shelton AL, Henning JA, Schultz P, Clay K (2014) Effects of abundant white-tailed deer on vegetation, animals, mycorrhizal fungi, and soil. For Ecol Manag 320:39–49
Scandurra A, Magliozzi L, Fulgione D, Aria M, D’Aniello B (2016) Lepidoptera Papilionoidea communities as a sentinel of biodiversity threat: the case of wild boar rooting in a Mediterranean habitat. J Insect Conserv 20:353–362
Scriber JM, Lederhouse RC (1983) Temperature as a factor in the development and feeding ecology of tiger swallowtail caterpillars, Papilio glaucus (Lepidoptera). Oikos 95–102. https://doi.org/10.2307/3544203
Siemann E, Carrillo JA, Gabler CA, Zipp R, Rogers WE (2009) Experimental test of the impacts of feral hogs on forest dynamics and processes in the southeastern US. For Ecol Manag 258:546–553
Sims NK, John EA, Stewart AJA (2014) Short-term response and recovery of bluebells (Hyacinthoides non-scripta) after rooting by wild boar (Sus scrofa). Plant Ecol 215(12):1409–1416
Sims NKE (2005) The ecological impacts of wild boar rooting in East Sussex. Ph.D. dissertation, School of Biological Sciences, University of Sussex, Brighton, UK
Singer FJ, Swank WT, Clebsch EEC (1984) Effects of wild pig rooting in a deciduous forest. J Wildl Manag 48:464–473
Stefanescu C, Traveset A (2009) Factors influencing the degree of generalization in flower use by Mediterranean butterflies. Oikos 118:1109–1117
Streitberger M, Fartmann T (2013) Molehills as important larval habitats for the Grizzled Skipper (Pyrgus malvae) in calcareous grasslands. Eur J Entomol 110:643–648. https://doi.org/10.14411/eje.2013.087
Streitberger M, Fartmann T (2016) Vegetation heterogeneity caused by an ecosystem engineer drives oviposition-site selection of a threatened grassland insect. Arthropod Plant Interact 10:545–555. https://doi.org/10.1007/s11829-016-9460-x
Streitberger M, Rose S, Hermann G, Fartmann T The role of a mound-building ecosystem engineer for a grassland butterfly. J Insect Conserv 18:745–751., Cameron A, Green RE, Bakkenes M, Beaumont LJ, Collingham YC, Erasmus BFN, de Ferreira M, Grainger A, Hannah L, Hughes L, Huntley B, van Jaarsveld AS, Midgley GF, Miles L, Ortega-Huerta MA, Townsend Peterson A, Phillips OL, Williams SE (2014) (2004) Extinction risk from climate change. Nature 427:145–148
Thomas CD, Cameron A, Green RE, Bakkenes M, Beaumont LJ, Collingham YC, Erasmus BFN, Ferreira de Siqueira M, Grainger A, Hannah L, Hughes L, Huntley B, van Jaarsveld AS, Midgley GF, Miles L, Ortega-Huerta MA, Townsend Peterson A, Phillips OL, Williams SE (2004) Extinction risk from climate change. Nature 427:145–148
Valente AM, Acevedo P, Figueiredo AM, Fonseca C, Torres RT (2020) Overabundant wild ungulate populations in Europe: management with consideration of socio-ecological consequences. Mammal Rev 50:353–366
Valimaki P, Itamies J (2005) Effects of canopy coverage on the immature stages of the Clouded Apollo butterfly [Parnassius mnemosyne (L.)] with observations on larval behaviour. Entomol Fenn 16:117–123
Van Swaay CAM, Cuttelod A, Collins S, Maes D, López Munguira M, Šašić M, Settele J, Verovnik R, Verstrael T, Warren M, Wiemers M, Wynhof I (2010) European red list of butterfies. Publications Office of the European Union, Luxembourg
Vovlas A, Balletto E, Altini E, Clemente D, Bonelli S (2014) Mobility and oviposition site-selection in Zerynthia cassandra (Lepidoptera, Papilionidae): implications for its conservation. J Insect Conserv 18:587–597. https://doi.org/10.1007/s10841-014-9662-4
Wheatall L, Nuttle T, Yerger E (2013) Indirect Effects of pandemic deer overabundance inferred from Caterpillar-Host relations. Conserv Biol 27:1107–1116
Wiklund C (1984) Egg-laying patterns in butterflies in relation to their phenology and the visual apparency and abundance of their host plants. Oecologia 63:23–29
Wu T-S, Damu AG, Su C-R, Kuo P-C (2004) Terpenoids of Aristolochia and their biological activities. Nat Prod Rep 21:594–624
Wynhoff I, Grutters M, van Langevelde F (2008) Looking for the ants: selection of oviposition sites by two myrmecophilous butterfly species. Anim Biol 58:371–388
Wootton JT (1994) Predicting Direct and Indirect Effects: An Integrated Approach Using Experiments and Path Analysis. Ecol 75:151-165
Welander J (2000) Spatial and temporal dynamics of wild boar (Sus scrofa) rooting in a mosaic landscape. JZool 252:263-271
Zinetti F, Dapporto L, Vovlas A, Chelazzi G, Bonelli S, Balletto E, Ciofi C (2013) When the rule becomes the exception. No evidence of gene flow between two Zerynthia cryptic butterflies suggests the emergence of a new model group. PLoS ONE 8:e65746. https://doi.org/10.1371/journal.pone.0065746
Acknowledgements
We acknowledge the staff of “WWF Trulli e Gravine – OdV” for their support during data collection in Oasi WWF Monte Sant’Elia. We also thank Francesca Aquilino for the help provided during field activities. Data collection has been partially carried out within the project “Azioni per la tutela di flora e di fauna di interesse conservazionistico nel SIC/ZPS IT9130007 Area delle Gravine - POR-PUGLIA 2014–2020 - Asse VI - Azione 6.5.1”.
Funding
Data collection has been partially carried out with the financial support of Comune di Massafra (TA), within the project “Azioni per la tutela di flora e di fauna di interesse conservazionistico nel SIC/ZPS IT9130007 Area delle Gravine - POR-PUGLIA 2014–2020 - Asse VI - Azione 6.5.1”.
Author information
Authors and Affiliations
Contributions
RL and LA contributed to the study conception and design. RL collected original data. Both authors performed analyses, wrote and revised the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Communicated by Jens Dauber.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Labadessa, R., Ancillotto, L. Beauty and the beast: multiple effects of wild boar rooting on butterfly microhabitat. Biodivers Conserv 32, 1189–1204 (2023). https://doi.org/10.1007/s10531-023-02545-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10531-023-02545-7