Abstract
Humans have been altering the Mediterranean landscapes for millennia. To diminish the probability of encounters with domestic animals, humans and their activities, many species adjust their behavior to become more nocturnal. Even habitat-generalist species, such as red fox and stone marten that are somehow tolerant to environmental changes, might be affected by anthropic disturbances. Nevertheless, only a small number of studies were implemented in Iberia targeting these mesocarnivores’ activity patterns, and fewer have assessed the temporal ecology of these species in Eucalyptus plantations, the current main forest cover in Portugal. Based on camera traps, we aimed to analyze: 1) the temporal and spatio-temporal activity patterns of red fox and stone marten; and 2) how they are affected by distinct human disturbances (i.e., humans, livestock, dogs, plantations, and hunting). Foxes presented a higher crepuscular activity, while martens were entirely nocturnal, suggesting some avoidance behavior. Both mesocarnivores showed a higher overlap with dogs’ activity than with humans or livestock. Foxes’ activity patterns vary between seasons and habitats but were not influenced by the hunting period. Results suggest that both mesocarnivores, besides setting apart their activity from humans related disturbances, also show a tendency to temporally avoid each other. While the increase of nocturnality may indicate an anthropic disturbance impact, a reduction of activity overlap between mesocarnivores may be a strategy to reduce competition. These results may help support the sustainable management of landscapes by highlighting critical periods where activity overlaps may occur, and thus the anthropic impacts on wildlife are higher.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Species included in the same guild—i.e., that exploit similar categories of resources (Simberloff and Dayan 1991)—often show similar traits that force them to compete for resources (e.g., food resources; Blondel 2003). Nevertheless, to coexist, species must diverge in at least one of the three niche dimensions—temporal, spatial, and trophic (Schoener 1974), as a complete overlap in all three dimensions will lead to the local exclusion of the less efficient competitor. In terms of temporal segregation, species may adjust their activity patterns to reduce the overlap of the circadian activity or show similar activity patterns, being active in different areas/locations, i.e., distinct spatio-temporal patterns (Posillico et al. 1995). In anthropogenic areas, animals face extra challenges linked to the presence of humans and their activities (e.g., land-use changes, persecution; Reason et al. 1993; Frick et al. 2020), and while some move to other habitats/regions with lower human presence (Posillico et al. 1995; Lucherini et al. 2009), others tend to adapt and coexist with humans and their activities (Cruz et al. 2018; Smith et al. 2018; Li et al. 2019; Manlick and Pauli 2020). In such a scenario, animals must cope with the presence of competitors of the same guild, but also with humans and human-associated disturbances such as habitat destruction, land cover change, hunting (of predators and preys), domestic animals’ presence, among others (Gruber et al. 2019). Therefore, they may be forced to change their activity to avoid the most impactful disturbance, often of anthropic origin (e.g., hunt and agriculture; Posillico et al. 1995), and/or increase their levels of tolerance towards competitors allowing a higher overlap of activity between guild members to avoid higher anthropic disturbances (e.g., houses and human foot traffic; Smith et al. 2018).
Human populations and activities have been expanding toward natural areas since ancient times (Henn et al. 2012; Roser et al. 2013), enhancing the disturbance regimes. One recent example is the modification of natural and ancient habitats to install planted forests and wood production areas, which have been increasing in the last decades (FAO 2018) to fulfill demands on basic forest goods (e.g., wood, paper pulp) (Lamberg et al. 2012). Worldwide, the area of plantation forests has increased by 123 million ha in the last three decades (between 1990 and 2020), with, currently, over 293 million ha (FAO 2020). The majority of planted forests are composed of native species (Payn et al. 2015), aiming to enhance forest resources and protect watersheds (FAO 2020), control soil erosion and desertification (Fisher 1995), and maintain biodiversity (Humphrey et al. 2000; López‐Bedoya et al. 2022; Pietrek and Branch 2011). Nevertheless, they are still less efficient in delivering such ecosystems services than native forests (Hua et al. 2022). But others, are focused on wood/fiber production and used for commercial purposes, accounting for almost 45% (131 million ha) of the world’s planted forests, 5.5 million ha of which are present in Europe alone (FAO 2020). In Mediterranean Europe, landscapes have been altered by humans’ activities for millennia (Blondel 2006), which include establishing agroforestry systems (e.g., cork oak woodlands or “montado/dehesas”). These changes in habitats type, structure, and functionality have direct consequences on species’ ecological patterns, leading, for example, to regional/local extinctions (e.g., Chamberlain and Fuller 2000; Franco et al. 2006), population decline (e.g., Selwood et al. 2015) or simply to changes in their ecological processes and patterns (e.g., Verdade et al. 2011). However, even in natural areas that have not been highly altered by humans, their presence might be frequent (e.g., hunting, grazing livestock; personal observation), also inducing lower-scale changes in species’ ecological patterns (Oberosler et al. 2020). In order to avoid or reduce their interactions with human activities and consequent disturbance (e.g., noise), which are usually more concentrated in the daytime, many vertebrates, including carnivores, tend to increase their nocturnal activity. Agriculture (e.g., tractors and other agriculture machinery), forestry (e.g., clear-cutting), and hunting (e.g., gunshots and hunting dogs) (Cruz et al. 2018; Gaynor et al. 2018) can be listed as accountable for the disturbance.
Portugal can be considered a high carnivore diversity area in Europe (Bencatel et al. 2018), but mesocarnivores distribution varies greatly, with only 60% of the species being widely distributed (e.g., red fox, Vulpes vulpes L.; stone marten Martes foina Erxleben) (Bencatel et al. 2018; Álvares et al. 2019). All these mesocarnivores have been coping with humans’ presence in the Portuguese landscape for centuries and had to adapt to the disturbance contexts linked to this co-existence to find adequate resources to survive. Thus, to understand how species have adapted to anthropic environmental changes, especially to highly artificial systems such as planted exotic forests (e.g., Eucalyptus plantations), the use of habitat-generalist species as models often seems adequate. They have wider biological and physiological plasticity that allows them to be more tolerant to changes in environmental conditions (Richmond et al. 2005), which will allow them to change their ecological patterns to cope with different levels of disturbance. Within Portuguese mesocarnivores, two generalist species comply with this criterion, although showing distinct habits that may shape their ecological responses: the stone marten with a higher arboreal capacity and the red fox, a more cursorial mesocarnivore (Santos et al. 2007). The red fox is the most widespread and abundant mesocarnivore in Portugal (Bencatel et al. 2018) with a great ability to adjust to habitat transformation, occurring in most Portuguese native areas, but also in agroecosystems and more urbanized areas (Díaz-Ruiz et al. 2013; Alexandre et al. 2020). The stone marten is also a native species that occur in different types of habitats, from native forests to cities (Santos and Santos-Reis 2010; Pereira et al. 2012). Both mesocarnivores have been described as well adapted to humans and urban areas (Harris and Rayner 1986; Adkins and Stott 1998; Wilkinson and Smith 2001; Santos and Santos-Reis 2010), although there is a lack of data for Iberia. The ecology of red foxes and stone martens’ population in the Iberian Peninsula has been fairly described (e.g., Bencatel et al. 2018), with some information regarding these species’ activity patterns being already available in the literature (Santos and Santos-Reis 2010; Barrull et al. 2014; Monterroso et al. 2014, 2016; Díaz-Ruiz et al. 2016; Vilella et al. 2020). However, very little data exist on how both species adapt their activity pattern in production forests (but see Cruz et al. 2015), especially in Eucalyptus plantations, which in Portugal is the widest forested area, covering more territory than any individual type of native forest types (COS 2018) a de Uso e Ocupação do Solo de Portugal continental para 2018). The information available regarding these species’ activity patterns derive from studies implemented in native habitats or in historical and High Nature Value farmlands (e.g., Barrull et al. 2014; Monterroso et al. 2014). Those studies show that the red fox and the stone marten are mainly nocturnal mesocarnivores, but their activity peaks vary according to the season (Vilella et al. 2020), breeding season phase (Monterroso et al. 2014), and food availability (Barrull et al. 2014). Still, in none of those studies, the activity patterns are estimated and compared for different disturbance contexts to assess how specifically each disturbance factor may be shaping these generalist carnivores’ circadian cycle, nor the effect of inhabiting exotic Eucalyptus plantations on those patterns assessed.
Considering this information gap, our goal was to understand how two sympatric mesocarnivores may temporally coexist within the same guild and how they managed to adapt their activity to distinct human activities and disturbances contexts. Using camera trapping detections in a Portuguese landscape dominated by Eucalyptus plantations, also including native patches (in the central region of Portugal), we specifically studied the activity patterns of two Iberian mesocarnivore species, the red fox and the stone marten, by evaluating: (i) their daily activity pattern and corresponding overlaps; (ii) how each species activity pattern varies in different human-related disturbance contexts (i.e., in areas with and without the presence of humans, dogs, and livestock); (iii) how red fox activity patterns diverge between native areas and plantations, hunting periods and seasons; and (iv) the spatio-temporal pattern of both species, and each species with humans, dogs, and livestock.
We predicted that: (1) both mesocarnivores species will be more active during the nighttime (Vilella et al. 2020), sunrise and sunset period (Cruz et al. 2015), since in these periods humans are absent from plantations and native areas, leading to a lower encounter risk and (2) both mesocarnivores will overlap their activity patterns, considering they are nocturnal (Vilella et al. 2020); (3) the two species will show more nocturnal activities in areas where humans/human activities are present to reduce the probability of encountering those human-related disturbance drivers, since all occur more often during the day (Kight and Swaddle 2011); (4) red foxes will reduce their daytime activity in Eucalyptus plantations, when compared to native areas, due to forestry works that are usually implemented during daytime in these production areas (personal observation); (5) red foxes will show higher nocturnality during the hunting period to avoid being detected by hunters and hunting dogs, as hunting journeys can only legally target daytime periods (Hipólito et al. 2018); 6) red foxes will show a reduced daytime activity period during the dry season due to thermoregulation limitations, to avoid extreme daytime temperatures in the Iberian Mediterranean summer (Monterroso et al. 2014); 7) red foxes and stone martens will show an avoidance spatio-temporal pattern to reduce competition (Torretta et al. 2017); and 8) both species will present a spatio-temporal avoidance of humans, dogs and livestock as a strategy to minimize disturbance (Hipólito et al. 2016, 2018; Alexandre et al. 2020).
Material and methods
Study area
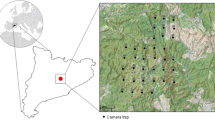
This study was carried out in eight different study areas in the Mediterranean region of central Portugal (Fig. 1), described by hot and dry summers and cold and wet winters, with an average annual temperature of 16 °C and an average annual rainfall of 758 mm. Six areas were located within Eucalyptus plantations dominated landscapes (Pampilhosa da Serra, Góis, and Mortágua; Fundão, Penamacor, and Penha-Garcia) and included patches of Mediterranean shrubland species and native woodlands. All Eucalyptus areas included in our study are managed by the same Portuguese forestry company and thus are managed similarly. Each area encompassed different production stages [initial (trees with ≤ 1.5 m); intermediate (> 1.5—< 10 m) and preharvest (≥ 10 m)], corresponding to a 12-years production cycle. Forestry workers normally start working after sunrise and all interventions on the plantation is limited to daylight hours and include: 1) the tree pruning process after 1.5–3.5 years of trees being planted and, sometimes, three years after the first pruning process; 2) soil and dirt roads maintenance to avoid erosion; 3) mechanical or chemical control of the understory (the use of the latter is limited to the first six years, and to the application of systemic herbicides with low/null toxicity to the environment); and 4) fertilization, implemented in the first four years of the plantation.
Location of the eight study areas in central Portugal where camera trap grid was implemented. The green area represents Eucalyptus plantations. This figure was produced using QGIS 3.10.8-A Coruña software (QGIS Development Team 2020)
Moreover, two areas corresponded to native vegetation areas used as control (Serra da Lousã and Serra da Malcata), dominated by native species such as Pinus pinaster Aiton., Quercus robur L., Quercus suber L. and native Mediterranean shrubland (e.g., Arbutus unedo L, Erica spp., Ulex spp., Genista spp.). Serra da Lousã is classified as a Site of Community Importance (SIC) within the Natura 2000 network (PTCON0060), and Serra da Malcata is a Natural Reserve, also SIC (PTZPE0007) (European Commission 2021).
Human activities (e.g., grazing, forestry operations, tracking, mountain biking, offroad 4WD, leisure activities) occurred in some parts of all the eight areas, including hunting that is permitted during the hunting season (October-February; Law-Decree n. º 202/2004—Official Journal (Diário da República) n.º 194/2004, Series I-A de 2004–08-18). In this period hunters use dogs to hunt game species, such as the red fox.
Sampling design
The eight study areas were located at least 10 km from the nearest one. For each study area, a grid containing 25 camera traps (Cuddeback 20 Megapixel IR H-1453 white series), 1 km apart, active for 24 h during 30 consecutive days for each sampling season (i.e., wet season from January to May 2019–2020; and dry season from June to September 2019–2020), totalizing 800 camera-trap stations. Camera trapping is a non-invasive methodology that enables the study and continuous monitoring of wildlife, while reducing the wildlife-observer interaction. The method is particularly efficient for providing evidence of an animal’s presence, especially for species that are difficult to observe (typically carnivores), due to their solitary and elusive behavior, low densities, and large home ranges (O’Connell et al. 2011; Rovero and Zimmermann 2016). It is widely used to study distinct ecological processes at different scales, such as individual behaviors (e.g., report specific behaviors, such as feeding), monitor populations (and estimate abundance, occupancy, density, etc.; Burton et al. 2015), or understand intra-community interactions (e.g., seed dispersal actors) (Trolliet et al. 2014). Even though, it still entangles some limitations, such as when applied to areas with dense vegetation cover (that reduces animals’ detectability due to a reduction of the camera’s field of view and detection zone; Hofmeester et al. 2019), when targeting species with specific movement behaviors [e.g., arboreal species such as the common genet (Genetta genetta L.)], or when individual identification is needed (which is not possible when the photographed individual does not have individual marks, such as fur patterns). Nevertheless, this methodology is commonly used, and highly efficient to study mammals’ activity patterns, and temporal overlap (i.e., the similarity between two activity patterns) between species (Li et al. 2019) since camera records allow accurate dates and times at which individuals are detected, and thus active. Camera time was set according to Coordinated Universal Time (UTC), and cameras were installed at 40 to 60 cm above ground level, attached to trees or wooden sticks, and programmed to take three consecutive photos every time the sensor was triggered, with a 30-s interval between any subsequent trigger event (O’Connell et al. 2011). We did not use bait or lures in any camera.
We identified the species detected in each photo and registered the trap location, day, and local time. However, to assure temporal independence between each event, multiple photo captures of the same species, less than 30 min apart, were collapsed into a single independent data point, and we only considered independent detection events in our analysis (Rovero and Zimmermann 2016).
Data analyses
Species activity patterns and overlap
To estimate red fox and stone marten activity patterns in the different environmental contexts, we converted the local time data of each mesocarnivore detection event to solar time using the “solaR” R package (Perpiñán 2012) to standardize the temporal data and allow inter-areas comparisons. After correction, the time was scaled into a 0–1 range and then transformed into radians (time*2π).
To visualize the daily activity patterns of species, we created probability density function plots for the global data set using the “overlap” R package (Meredith and Ridout 2021). We also estimated the activity overlap between both mesocarnivore species and between each species in the different human-related disturbance contexts (i.e., with and without humans, dogs, and livestock presence) by plotting both probability density functions. Then we compared activity patterns estimation between each data subset by calculating the coefficient of overlap Δ4, which is the recommended metric for sample sizes with more than 75 detections (Meredith and Ridout 2021), ranging from 0 to 1 (no overlap and complete overlap, respectively) (Ridout and Linkie 2009). We used a standard deviation of 999 bootstrap replicates (Linkie and Ridout 2011) to estimate Δ4 95% confidence intervals precision. Using the methodology adopted by Monterroso et al. (2014), we classify the overlapping activity in three categories: low overlap = Δ4 ≤ 50th sample percentile; moderate overlap = 50th percentile < Δ4 ≤ 75th percentile; high overlap = Δ4 > 75th percentile.
To test the uniformity of records’ distribution over the 24 h of the day, we used the Rayleigh Uniformity Test (Mardia and Jupp 2000), and to compare the detections’ distribution across the diel cycle for all the previous combinations of subsets data, we used the Watson-Wheeler test (W) (Zar 2010), both available in the “circular” R package (Agostinelli and Lund 2022). Rayleigh Test values close to 1 (records with a higher value) indicate that records are concentrated in a specific period of the day with a non-uniform distribution. Inversely, values close to 0 (records with a lower value) indicate that activity may be occurring during the 24 h of the day without a specific peak of activity. When the Watson-Wheeler test value is high, it indicates a bigger difference between the activity patterns of each data subset pair (which can be significant, i.e., p < 0.05).
As the red fox sample size was higher than that collected for the stone marten, we were able to test variations in activity between other environmental contexts for this canid. Therefore, we compared red fox activity patterns between seasons (i.e., dry vs wet season), locations (i.e., native areas vs Eucalyptus plantation), and hunting periods (i.e., hunting vs no-hunting period). When datasets were large enough, we also assessed activity variations between a combination of those contexts: hunting vs no-hunting period in Eucalyptus plantation and native areas.
Species spatio-temporal segregation—Time-to-encounter
To assess the spatio-temporal segregation among species in the different environmental contexts, we created a matrix of species encounters per hour for each camera-trap station. Then we calculated the proportion of sites, at each hourly interval, when the activity detection of any two species overlapped. We then estimated the time-to-encounter across each species pair (i.e., each mesocarnivore species with humans, dogs, and livestock presence and between the two mesocarnivores) using a multi-response permutation procedure (Mielke et al. 1976). For each encounter of one species at a specific camera location, we calculated the minimum time to encounter the co-pair. Hence, we obtained, for each species pair, a set of observed times-to-encounter. We produced expected statistical distributions of times-to-encounter by randomly (in 1000 simulations) attributing encounter times to camera-trap locations. Lastly, we compared the median observed time-to-encounter with the simulated distribution of expected times-to-encounter (Mielke et al. 1976; Karanth et al. 2017).
A smaller observed time-to-encounter than expected (i.e., simulated) implies species aggregation, while a larger observed time-to-encounter reflects species segregation. This method was used to test species spatio-temporal segregation, conditional on observed space-use and temporal activity patterns of the target species (Mielke et al. 1976; Karanth et al. 2017).
All analyses were made in R software (R Core Team 2021).
Results
In total, we ended with 765 fully operating cameras during the complete study period due to some malfunctions that restrained us from using the entire 800 cameras. We detected five mesocarnivore species: the red fox, the stone marten, the European badger, the common genet, and the Egyptian mongoose (Herpestes ichneumon L.). However, only for red foxes (N = 364) and stone martens (N = 42) we generated enough data to assure a robust analysis (> 30 independent records). We also registered records of humans, livestock (goat and sheep), and dogs (Table 1).
Species’ daily activity patterns and temporal overlap
Red foxes and stone martens presented a typically nocturnal pattern (with a statistically significant non-uniform pattern: Rayleighfox = 0.328, p < 0.001; Rayleighmarten = 0.675, p < 0.001), with most of their detections being concentrated in the night period. Nevertheless, some variation was detected, with red fox presenting a high crepuscular activity, with activity peaks around 20:00 h (sunset) and 05:00 h (sunrise), and stone martens being more nocturnal, with an activity peak near 22:00 h (Fig. 2). Livestock, dogs, and humans were typically diurnal with activity peaks closer to sunrise and sunset (also showing a significant non-uniform activity pattern: Rayleighlivestock = 0.631, p < 0.001; Rayleighdogs = 0.325, p < 0.001; Rayleighhumans = 0.704, p < 0.001). Humans’ activity at the study site increases sharply at the beginning of the day and continues relatively constant throughout the day until around sunset, where it decreases abruptly (Fig. 2).
Daily activity patterns of red foxes a, stone martens b, livestock c, domestic dogs d, and humans e in six Eucalyptus dominated landscapes and two native habitats of central Portugal, based on the Kernel density estimator. Vertical dashed lines represent the beginning and end of sunset (in red) and sunrise (in blue). R – Rayleigh test; n – number of independent events
The activity patterns of red fox and stone marten were clearly distinct (Watson's Test = 15.689, p < 0.001), with stone marten showing a unimodal and typically nocturnal pattern, while red fox evidence a bimodal pattern with two activity peaks, near sunset and sunrise (Fig. 3). Their activity overlap was moderate according to Monterroso et al. (2014) [Δ4 = 0.734 (0.633–0.836 CI)] (see Appendix I).
Activity overlap (shaded areas) patterns between red foxes and: stone martens a, domestic dogs b, livestock d, and humans f; and between stone martens and: domestic dogs c, livestock e and humans g in six Eucalyptus dominated landscapes and two native habitats of central Portugal. Vertical dashed lines represent the beginning and end of sunset (in red) and sunrise (in blue). Overlap coefficients (Δ4), their confidence interval, and the significance of the difference are also presented. (Color figure online)
There was a significant mismatch between the activity patterns of red foxes (Watson's Testhumans = 218.220, p < 0.001; Watson's Testdogs = 68.304, p < 0.001; Watson's Testlivestock = 137.050, p < 0.001) and stone martens (Watson's Testhumans = 87.275, p < 0.001; Watson's Testdogs = 58.795, p < 0.001; Watson's Testlivestock = 80.530, p < 0.001) with the activity of humans and domestic animals (Fig. 3), with Δ4 always < 0.50, indicating a low overlap. This mismatch seems to be higher for stone martens as they consistently show a lower overlap with humans, livestock, and domestic dogs than foxes (Fig. 3), as foxes show some overlap, especially during sunset and sunrise. Both carnivores showed a higher overlap with dogs’ activity than with humans or livestock.
Red fox activity patterns in different environmental and disturbance contexts
Red fox activity patterns showed some variation between the different analyzed environmental and disturbance contexts. In the wet season and native habitats, red foxes show a unimodal pattern with a peak of activity in the early evening, and then the activity decreases towards sunrise (Figs. 4a, b). In the dry season and on Eucalyptus plantations, the activity has a peak at sunset and another at sunrise (Figs. 4a, b). These variations between habitats and seasons are highlighted by the medium value of Δ4 and the significant differences in patterns (Δ4season = 0.778, Watson's Testseason = 14.113, p < 0.001; Δ4habitat = 0.779; Watson's Testhabitat = 19.327, p < 0.001).
Activity patterns and overlap (shaded areas) of red foxes in different environmental and disturbance contexts: a wet and dry seasons; b Eucalyptus plantations and native habitats; c hunting and no-hunting periods; d hunting and no-hunting periods in Eucalyptus plantations; and e hunting and no-hunting periods in native habitats. Vertical dashed lines represent the beginning and end of sunset (in red) and sunrise (in blue). Overlap coefficients (Δ4), their confidence interval, and the significance of the difference are also presented. (Color figure online)
Regarding the effect of disturbance contexts, we did not detect a significant difference in activity patterns between hunting and no-hunting periods, with a high pattern overlap (Fig. 4c; Δ4 = 0.840, Watson's Test = 1.208, p = 0.547). This pattern holds when the effect of hunting is analyzed in each specific habitat (Δ4Eucalyptus = 0.870, Δ4native = 0.757).
Spatio-temporal patterns between red foxes and stone martens
We examined the times-to-encounter patterns between red foxes and stone martens to test for spatio-temporal segregation. The results apparently show some avoidance behavior, although not significantly (p-value = 0.338; Fig. 5; Appendix II), as the frequency of the simulated time-to-encounter between the two mesocarnivores is mostly within 125 days (Fig. 5), while the median minimum observed is in 177 days.
Spatio-temporal interactions between red foxes and stone martens, as shown by the times-to-encounter between both species. The vertical red line represents the median minimum observed time-to-encounter between the two species, and the area under the curve the randomly simulated times-to-encounter. P—p-value
Our data highlights a tendency for the red fox to avoid people (p-value = 0.062 is almost significant) (Fig. 6), with the estimated time-to-encounter between foxes and humans being 84 days (Fig. 6), while the median minimum observed period is 120 days. Surprisingly, stone martens seem to show some aggregation behavior towards dogs (Fig. 6; observed period of 12 days, but the estimated values reached 457 days). However, this difference was not significant (p > 0.05). A similar non-significant effect was detected for both carnivores and livestock. Red fox-dogs and stone martens-human spatio-temporal patterns showed an almost perfect match between observed and estimated values (Fig. 6).
Spatio-temporal interactions between red foxes and stone martens, and domestic dogs (a and b), livestock (c and d), and humans (e and f), as shown by the times-to-encounter between species. The vertical red line represents the median minimum observed time-to-encounter between the two species, and the area under the curve the randomly simulated times-to-encounter. P—p-value
Discussion
Red foxes and stone martens were the most common species of mesocarnivores across our study areas and therefore were the focus species of our study. Overall, both species were mostly active during nighttime, which is in line with the mesocarnivores’ nocturnal character described by several authors (Barrull et al. 2014; Monterroso et al. 2014; Vilella et al. 2020), supporting our first hypothesis. Often, the concentration of activity in a specific period is driven by habitat structure (Díaz-Ruiz et al. 2016) and by the need to reduce competition (Torretta et al. 2017), disturbance (Kight and Swaddle 2011) or predation (Monterroso et al. 2013). Moreover, the studied populations seem to follow most of these ecological mechanisms. Red foxes and stone martens seem to be reducing their competition by slightly diverging their night activity. Red fox shows a typically crepuscular activity, being mostly active at sunset and sunrise, while stone martens concentrate their activity in the middle of the night. Such a strategy may be adopted to reduce the encounter probability between both species and thus any agonistic behavior that may induce injuries, which may be common in contexts of low food abundance (Papakosta et al. 2010). In Mediterranean landscapes, this temporal segregation seems to be the most important ecological mechanism facilitating mesocarnivores coexistence (Ferreiro-Arias et al. 2021). Nevertheless, these processes are usually multidimensional (Ferreiro-Arias et al. 2021) with a concurrent effect of spatio and/or trophic dimensions. However, the spatio-temporal avoidance pattern was not corroborated by our results (and thus our seventh hypothesis) which showed a non-significant avoidance. In Southwestern Mediterranean, both species focus their diet on the consumption of fruits and insects (Santos et al. 2007), which indicates some degree of trophic competition and that both species may be targeting similar areas where they can find these resources. In poor resource environments (such as plantations; da Silva et al. 2019), high interspecific competition for food resources may occur between red foxes and stone martens (Remonti et al. 2012). López-Bedoya et al. (2022) showed that globally the richness and abundance of coleopters (an important prey group for both carnivores; Santos et al. 2007) is lower in plantations than in natural forest. A similar pattern was detected by da Silva et al. (2019) for Portugal, which also showed that Eucalyptus plantation harbor a lower diversity of shrub species, which are often the most import fruit producing plants in plantations (as native trees are often absent). The detected temporal segregation behavior may be a strategy to allow the use of the same sites as feeding grounds (and where fruits and/or insects are available), but at different night periods to reduce encounter probabilities, corroborating our seventh hypothesis. Thus, the detected temporal segregation could be linked to a decrease in food availability and be a strategy to allow co-existence between competitors. Such avoidance between red foxes and stone martens was already detected in other Mediterranean environments, and competition was one of the highlighted drivers (Monterroso et al. 2014; Torretta et al. 2017).
Red foxes’ activity also changed according to seasonality, with activity being mostly concentrated in the sunset during the wet season, while in the dry season, red foxes were also highly active in the sunrise. The wet season is characterized by colder temperatures and rain, creating more challenging weather conditions for wildlife while searching for food. During this season, temperatures drop more drastically at night, which combined with the rain and wind, increases the animal’s thermoregulatory needs (Maurel and Boissin 1983). Therefore, as a more nocturnal animal (Monterroso et al. 2014), red foxes may concentrate their activity in the sunset and the first hours of the night and avoid later night periods that get progressively colder, as a strategy to reduce body heat loss (thermoregulation). In the dry summer, the night period is milder, and the most challenging period is the daytime, in which temperatures in the Mediterranean region can reach more than 40 °C (Santos and Miranda 2006). Thus, red foxes are more active throughout the night, showing two activity peaks but less day activity in the dry season (as hypothesized in number six).
Influence of anthropic disturbance on species activity
The habitat structure can be one of the drivers of species activity patterns (Díaz-Ruiz et al. 2016), and this is especially true if this effect is coupled with the anthropic disturbance linked to man-shaped habitats, such as plantations. In Eucalyptus plantations, red foxes showed two activity peaks: one at the beginning of the night and another at sunrise. Eucalyptus plantations are often a low diversity system (da Silva et al. 2019) and, therefore carnivore trophic resources may be scarcer (e.g., management action, such as thinning, can affect insect richness, abundance, and community structure; Taki et al. 2010). Usually, in low-abundance resource areas, carnivores increase their mobility and activity to optimize prey detection and hunting efforts (Karanth et al. 2017). Therefore, in plantations, red foxes must be active for longer periods to find enough food to fill their energy requirements. The second-night activity peak detected in plantations may be linked to this strategy. Inversely, in the native areas, a single activity peak was detected at the end of the sunset, with a decline in activity throughout the night. The monitored native areas have some degree of statutory protection (e.g., Natural Reserve and Sites of Community Importance; European Commission 2021) and are often used by the general public as recreation and sports areas, where outdoor activities occur throughout the day (e.g., mountain biking, hiking trails), often starting near sunrise. Accordingly, foxes may be concentrating their activity in periods where fewer people are active in the area, thus minimizing encounter risks (Reilly et al. 2017). This data does not corroborate our fourth hypothesis, as no lower diurnal activity was detected in Eucalyptus plantations, but is aligned with the third one (i.e., avoidance of periods where humans and their activity occur).
Another disturbance factor that may affect species activity patterns, especially those that can be legally hunted, such as the red fox, is the hunting pressure (Kämmerle et al. 2020). This disturbance is linked not only to the direct effect of shooting, but also to the indirect effects of hunters’ vehicles and hunting dogs. As hunting activities start around 6 a.m. (near sunrise), we forecasted a change in red foxes’ activity during the hunting period, being more nocturnal (fifth hypothesis). Although no overall difference was detected between hunting and no-hunting periods, some variation was detected depending on the habitat type. In native habitats, where most hunting raids occur, red foxes start their activity later in the day (already during nighttime), probably to avoid hunters and hunting dogs, as no hunting is allowed during the night, thus reducing disturbance. As hunting raids start earlier in the morning, we detected a lower peak of activity near sunrise. We hypothesize that such a peak may be linked to the presence of hunters and dogs that may disturb the species leading them to move in search of refuge. This result partially supports our fifth initial hypothesis, where we stated that the hunting period would influence red fox activity.
In landscapes shaped by humans and their activities, such as Mediterranean Europe (Blondel 2006), their presence will constrain wildlife ecological patterns (Oberosler et al. 2017), usually leading to a mismatch between wildlife activity and humans/domestic animals (Hipólito et al. 2016; Gaynor et al. 2018) to reduce disturbance. Our data corroborate this general pattern as a significantly low overlap between red foxes and stone martens with humans, livestock, and domestic dogs (always Δ4 < 0.45) was detected (eighth hypothesis). This pattern was, however, more evident for stone martens. Nevertheless, even if the activity pattern shows a similar shape, there is a mismatch. For instance, domestic dogs show two activity peaks, like the red fox, but foxes’ activity peaks occur later in the sunset and earlier in the sunrise, reducing the overlap. Red foxes are known to shift their activity in areas where domestic dogs are present to reduce encounter probability (Galvez et al. 2021). Most of the dogs detected in our study areas are hunters or shepherds’ dogs (Appendix III), usually large dogs that may pose a real threat to the red fox. Thus, being active in periods when dogs are not is an adaptive strategy. Carnivores may even present similar activity patterns to those of humans, domestic dogs or livestock, but may be active at distinct sites, which will contribute to preventing negative effects. Our data shows that even when detected in the same sites, foxes show an avoidance behavior towards humans, reinforcing the impact humans induce on this canid activity (Monterroso et al. 2014; Díaz-Ruiz et al. 2016).
Conclusion
Our results corroborate that generalist and resilient mesocarnivores, such as the red fox and the stone marten, tend to avoid humans and their related activities due to the associated disturbances, such as hunting, domestic animals, or forest management which may affect these species. Red fox can even modify their activity peaks according to habitat, seasons or hunting pressures. Since species can adjust their activity patterns to some extent, it is important to assess the impacts of anthropic drivers (including humans and their activities) on other mesocarnivores to generate evidence that can support the sustainable management of landscapes and mitigate the disturbances associated with anthropic activities. Providing insights on critical periods where activity overlaps may occur and anthropic impacts on wildlife may be higher, will contribute to achieve a healthier coexistence between the wildlife community and humans. Such evidence should be considered in land management plans to avoid and reduce disturbances related to human activities, such as forestry activities (e.g., clear-cutting), during periods when most species are most sensitive. For example, red foxes usually search for mates between December and February (Loureiro et al. 2012), being more mobile in this period when looking for reproductive partners. Therefore, they may extend their activity towards sunset/sunrise, increasing the overlap with forestry activities (Cavallini 1996). By reducing anthropic activities in this period, we can assure that those disturbances have a limited impact on the natural wildlife activity patterns and, consequently, on individuals’ fitness.
Data availability
The datasets generated during the current study are available from the corresponding author on reasonable request.
References
Adkins CA, Stott P (1998) Home ranges, movements and habitat associations of red foxes Vulpes vulpes in suburban Toronto, Ontario, Canada. J Zool 244:335–346. https://doi.org/10.1017/S0952836998003045
Agostinelli C, Lund U (2022) Package “circular”: circular statistics. https://r-forge.r-project.org/projects/circular/. Accessed 9 Jan 2022
Alexandre M, Hipólito D, Ferreira E et al (2020) Humans do matter: determinants of red fox (Vulpes vulpes) presence in a western Mediterranean landscape. Mammal Res 65:203–214. https://doi.org/10.1007/s13364-019-00449-y
Álvares F, Ferreira CC, Barbosa AM, et al (2019) Carnívoros. In: Bencatel J, Sabino-Marques H, Álvares F, et al. (eds) Atlas de mamíferos de Portugal, 2a edição. Universidade de Évora, Évora, pp 61–95
Barrull J, Mate I, Ruiz-Olmo J et al (2014) Factors and mechanisms that explain coexistence in a Mediterranean carnivore assemblage: an integrated study based on camera trapping and diet. Mamm Biol 79:123–131. https://doi.org/10.1016/j.mambio.2013.11.004
Bencatel J, Ferreira CC, Barbosa M et al (2018) Research trends and geographical distribution of mammalian carnivores in Portugal (SW Europe). PLoS One 13:1–20. https://doi.org/10.1371/journal.pone.0207866
Blondel J (2003) Guilds or functional groups: does it matter? Oikos 100:223–231. https://doi.org/10.1034/j.1600-0706.2003.12152.x
Blondel J (2006) The “design” of Mediterranean landscapes: a millennial story of humans and ecological systems during the historic period. Hum Ecol 34:713–729. https://doi.org/10.1007/s10745-006-9030-4
Burton AC, Neilson E, Moreira D, Ladle A, Steenweg R, Fisher JT, Bayne E, Boutin S (2015) Wildlife camera trapping: a review and recommendations for linking surveys to ecological processes. J Appl Ecol 52:675–685. https://doi.org/10.1111/1365-2664.12432
Cavallini P (1996) Ranging behaviour of red foxes during the mating and breeding seasons. Ethol Ecol Evol 8:57–65
Chamberlain DE, Fuller RJ (2000) Local extinctions and changes in species richness of lowland farmland birds in England and Wales in relation to recent changes in agricultural land-use. Agric Ecosyst Environ 78:1–17. https://doi.org/10.1016/S0167-8809(99)00105-X
COS (2018) Carta de Uso e Ocupação do Solo de Portugal continental para 2018. Direção de Serviços de Geodesia, Cartografia e Informação Geográfica. Direção-Geral do Território, Lisboa. https://snig.dgterritorio.gov.pt/rndg/srv/por/catalog.search#/metadata/b498e89c-1093-4793-ad22-63516062891b
Cruz J, Sarmento P, White P (2015) Influence of exotic forest plantations on occupancy and co-occurrence patterns in a Mediterranean carnivore guild. J Mammal 96:854–865. https://doi.org/10.1093/jmammal/gyv109
Cruz P, Iezzi ME, De Angelo C et al (2018) Effects of human impacts on habitat use, activity patterns and ecological relationships among medium and small felids of the Atlantic Forest. PLoS One 13:1–21. https://doi.org/10.1371/journal.pone.0200806
da Silva LP, Heleno RH, Costa JM et al (2019) Natural woodlands hold more diverse, abundant, and unique biota than novel anthropogenic forests: a multi-group assessment. Eur J for Res 138:461–472. https://doi.org/10.1007/s10342-019-01183-5
Díaz-Ruiz F, Delibes-Mateos M, García-Moreno JL et al (2013) Biogeographical patterns in the diet of an opportunistic predator: the red fox Vulpes vulpes in the Iberian Peninsula. Mamm Rev 43:59–70. https://doi.org/10.1111/j.1365-2907.2011.00206.x
Díaz-Ruiz F, Caro J, Delibes-Mateos M et al (2016) Drivers of red fox (Vulpes vulpes) daily activity: prey availability, human disturbance or habitat structure? J Zool 298:128–138. https://doi.org/10.1111/jzo.12294
European Commission (2021) Natura 2000. https://ec.europa.eu/environment/nature/natura2000/index_en.htm
FAO (2018) The State of the World’s Forests 2018—Forest pathways to sustainable development. Rome. Licence: CC BY-NC-SA 3.0 IGO.
FAO (2020) Global Forest Resources Assessment 2020: Main report. Food and Agriculture. Organization of The United Nations, Rome. https://www.fao.org/3/ca9825en/ca9825en.pdf
Ferreiro-Arias I, Isla J, Jordano P, Benítez-López A (2021) Fine-scale coexistence between Mediterranean mesocarnivores is mediated by spatial, temporal, and trophic resource partitioning. Ecol Evol 11:15520–15533. https://doi.org/10.1002/ece3.8077
Fisher RF (1995) Amelioration of degraded rain forest soils by plantations of native trees. Soil Sci Soc Am J 59:544–549
Franco AMA, Hill JK, Kitschke C et al (2006) Impacts of climate warming and habitat loss on extinctions at species’ low-latitude range boundaries. Glob Chang Biol 12:1545–1553. https://doi.org/10.1111/J.1365-2486.2006.01180.X
Frick WF, Kingston T, Flanders J (2020) A review of the major threats and challenges to global bat conservation. Ann N Y Acad Sci 1469:5–25. https://doi.org/10.1111/NYAS.14045
Galvez N, Meniconi P, Infante J, Bonacic C (2021) Response of mesocarnivores to anthropogenic landscape intensification: activity patterns and guild temporal interactions. J Mammal 102:1149–1164. https://doi.org/10.1093/JMAMMAL/GYAB074
Gaynor KM, Hojnowski CE, Carter NH, Brashares JS (2018) The influence of human disturbance on wildlife nocturnality. Science 360:1232–1235. https://doi.org/10.1126/science.aar7121
Gruber T, Luncz L, Mörchen J et al (2019) Cultural change in animals: a flexible behavioural adaptation to human disturbance. Palgrave Commun 51(5):1–9. https://doi.org/10.1057/s41599-019-0271-4
Harris S, Rayner JMV (1986) Urban fox (Vulpes vulpes) population estimates and habitat requirements in several british cities. J Anim Ecol 55:575. https://doi.org/10.2307/4740
Henn BM, Cavalli-Sforza LL, Feldman MW (2012) The great human expansion. Proc Natl Acad Sci 109:17758–17764. https://doi.org/10.1073/pnas.1212380109
Hipólito D, Santos-Reis M, Rosalino LM (2016) Effects of agro-forestry activities, cattle-raising practices and food-related factors in badger sett location and use in Portugal. Mamm Biol 81:194–200. https://doi.org/10.1016/j.mambio.2015.11.005
Hipólito D, Guedes D, Cabecinha D et al (2018) Drivers of sett site location by european badgers in Portugal. Biodivers Conserv 27:2951–2970. https://doi.org/10.1007/s10531-018-1580-4
Hofmeester TR, Cromsigt JPGM, Odden J, Andrén H, Kindberg J, Linnell JDC (2019) Framing pictures: a conceptual framework to identify and correct for biases in detection probability of camera traps enabling multi-species comparison. Ecol Evol 9:2320–2336. https://doi.org/10.1002/ece3.4878
Hua F, Bruijnzeel LA, Meli P, Martin PA, Zhang J, Nakagawa S, Miao X, Wang W, McEvoy C, Peña-Arancibia JL, Brancalion PHS, Smith P, Edwards DP, Balmford A (2022) The biodiversity and ecosystem service contributions and trade-offs of forest restoration approaches. Science 376:839–844. https://doi.org/10.1126/science.abl4649
Humphrey JW, Newton AC, Peace AJ, Holden E (2000) The importance of conifer plantations in northern Britain as a habitat for native fungi. Biol Conserv 96:241–252. https://doi.org/10.1016/S0006-3207(00)00077-X
Kämmerle JL, Rondeaux S, Storch I (2020) Circadian activity patterns of red foxes (Vulpes vulpes) in montane forests under different culling regimes. Mammal Res 65:615–619. https://doi.org/10.1007/S13364-020-00496-W/FIGURES/1
Karanth KU, Srivathsa A, Vasudev D et al (2017) Spatio-temporal interactions facilitate large carnivore sympatry across a resource gradient. Proc R Soc B Biol Sci. https://doi.org/10.1098/rspb.2016.1860
Kight CR, Swaddle JP (2011) How and why environmental noise impacts animals: an integrative, mechanistic review. Ecol Lett 14:1052–1061. https://doi.org/10.1111/J.1461-0248.2011.01664.X
Lamberg J-A, Ojala J, Peltoniemi M, Särkkä T (eds) (2012) The Evolution of Global Paper Industry 1800–2050. Springer, Dordrecht
Li Z, Wang T, Smith JLD et al (2019) Coexistence of two sympatric flagship carnivores in the human-dominated forest landscapes of Northeast Asia. Landsc Ecol 34:291–305. https://doi.org/10.1007/s10980-018-0759-0
Linkie M, Ridout MS (2011) Assessing tiger-prey interactions in Sumatran rainforests. J Zool 284:224–229. https://doi.org/10.1111/j.1469-7998.2011.00801.x
López-Bedoya PA, Cardona-Galvis EA, Urbina-Cardona JN, Edwards FA, Edwards DP (2022) Impacts of pastures and forestry plantations on herpetofauna: a global meta-analysis. J Appl Ecol 59:3038–3048. https://doi.org/10.1111/1365-2664.14299
Loureiro F, Pedroso NM, Santos MJ, Rosalino LM (2012) Um olhar sobre os carnívoros portugueses. Carnivora, Portugal
Lucherini M, Reppucci JI, Walker RS et al (2009) Activity pattern segregation of carnivores in the high andes. J Mammal 90:1404–1409. https://doi.org/10.1644/09-MAMM-A-002R.1
Manlick PJ, Pauli JN (2020) Human disturbance increases trophic niche overlap in terrestrial carnivore communities. Proc Natl Acad Sci USA 117:26842–26848. https://doi.org/10.1073/PNAS.2012774117/-/DCSUPPLEMENTAL
Mardia KV, Jupp PE (2000) Directional Statistics. John Wiley & Sons Inc, London
Maurel D, Boissin J (1983) Comparative Mechanisms of Physiological, Metabolical and Eco-Ethological Adaptation to the Winter Season in two Wild European Mammals: The European Badger (Meles meles L.) and the Red Fox (Vulpes vulpes L.). In: Margaris NS, Arianoutsou-Faraggitaki M, Reiter RJ (eds) Adaptations to Terrestrial Environments. Springer, Boston, MA
Meredith M, Ridout M (2021) Overlap. R Core Development Team. In: R Proj. https://cran.r-project.org/web/ packages/overlap/overlap.pdf. Accessed 9 Jan 2022
Mielke PW, Berry KJ, Johnson ES (1976) Multi-response permutation procedures for a priori classifications. Commun Stat—Theory Methods 5:1409–1424. https://doi.org/10.1080/03610927608827451
Monterroso P, Alves PC, Ferreras P (2013) Catch me if you can: diel activity patterns of mammalian prey and predators. Ethology 119:1044–1056. https://doi.org/10.1111/eth.12156
Monterroso P, Alves PC, Ferreras P (2014) Plasticity in circadian activity patterns of mesocarnivores in Southwestern Europe: implications for species coexistence. Behav Ecol Sociobiol 68:1403–1417. https://doi.org/10.1007/s00265-014-1748-1
Monterroso P, Rebelo P, Alves PC, Ferreras P (2016) Niche partitioning at the edge of the range: a multidimensional analysis with sympatric martens. J Mammal 97:928–939. https://doi.org/10.1093/jmammal/gyw016
O’Connell AF, Nichols JD, Karanth KU (2011) Camera Traps in Animal Ecology. Springer, Tokyo
Oberosler V, Groff C, Iemma A et al (2017) The influence of human disturbance on occupancy and activity patterns of mammals in the Italian Alps from systematic camera trapping. Mamm Biol 87:50–61. https://doi.org/10.1016/J.MAMBIO.2017.05.005
Oberosler V, Tenan S, Zipkin EF, Rovero F (2020) When parks work: effect of anthropogenic disturbance on occupancy of tropical forest mammals. Ecol Evol 10:3881–3894. https://doi.org/10.1002/ece3.6048
Papakosta M, Bakaloudis D, Kitikidou K et al (2010) Dietary overlap among seasons and habitats of red fox and stone marten in central greece. Eur J Sci Res 45:122–127
Payn T, Carnus JM, Freer-Smith P, Kimberley M, Kollert W, Liu S, Orazio C, Rodriguez L, Silva LN, Wingfield MJ (2015) Changes in planted forests and future global implications. For Ecol Manag 352:57–67. https://doi.org/10.1016/j.foreco.2015.06.021
Pereira P, da Silva AA, Alves J et al (2012) Coexistence of carnivores in a heterogeneous landscape: Habitat selection and ecological niches. Ecol Res 27:745–753. https://doi.org/10.1007/s11284-012-0949-1
Perpiñán O (2012) solaR: solar Radiation and photovoltaic systems with R. J Stat Softw 50:1–32. https://doi.org/10.18637/jss.v050.i09
Pietrek AG, Branch LC (2011) Native plantations as an important element for biodiversity in vanishing forested landscapes: a study of the near threatened araucaria tit spinetail (Leptasthenura setaria, Furnariidae). Austral Ecol 36:109–116. https://doi.org/10.1111/j.1442-9993.2010.02129.x
Posillico M, Serafini P, Lovari S (1995) Activity patterns of the stone marten Martes Foina Erxleben, 1777, in relation to some environmental factors. Hystrix 7:79–97
QGIS Development Team (2020) QGIS Geographic Information System. Open Source Geospatial Foundation Project. http://qgis.osgeo.org.
R Core Team (2021) R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing. Vienna, Austria. https://www.R-project.org/.
Reason P, Harris S, Cresswell P (1993) Estimating the impact of past persecution and habitat changes on the numbers of Badgers Meles meles in Britain. Mamm Rev 23:1–15. https://doi.org/10.1111/J.1365-2907.1993.TB00413.X
Reilly ML, Tobler MW, Sonderegger DL, Beier P (2017) Spatial and temporal response of wildlife to recreational activities in the San Francisco Bay ecoregion. Biol Conserv 207:117–126. https://doi.org/10.1016/J.BIOCON.2016.11.003
Remonti L, Balestrieri A, Ruiz-González A et al (2012) Intraguild dietary overlap and its possible relationship to the coexistence of mesocarnivores in intensive agricultural habitats. Popul Ecol 54:521–532. https://doi.org/10.1007/S10144-012-0326-5
Richmond CE, Breitburg DL, Rose KA (2005) The role of environmental generalist species in ecosystem function. Ecol Modell 188:279–295. https://doi.org/10.1016/j.ecolmodel.2005.03.002
Ridout MS, Linkie M (2009) Estimating overlap of daily activity patterns from camera trap data. J Agric Biol Environ Stat 14:322–337. https://doi.org/10.1198/jabes.2009.08038
Roser M, Ritchie H, Ortiz-Ospina E (2013) World Population Growth - Our World in Data. In: Publ. online OurWorldInData.org. https://ourworldindata.org/world-population-growth#citation. Accessed 2 May 2022
Rovero F, Zimmermann F (2016) Camera trapping for Wildlife Research. Pelagic Publishing, UK
Santos MJ, Pinto BM, Santos-Reis M (2007) Trophic niche partitioning between two native and two exotic carnivores in SW Portugal. Web Ecol 7:53–62. https://doi.org/10.5194/WE-7-53-2007
Santos MJ, Santos-Reis M (2010) Stone marten (Martes foina) habitat in a Mediterranean ecosystem: effects of scale, sex, and interspecific interactions. Eur J Wildl Res 56:275–286. https://doi.org/10.1007/s10344-009-0317-9
Santos FD, Miranda P (2006) Alterações climáticas em Portugal. Cenários, impactos e medidas de adaptação - Projecto SIAM II. Gradiva, Lisboa
Schoener TW (1974) Resouce partitioning in ecological communities: research on how similar species divide resources helps reveal the natural regulation of species diversity. Science 185:27–39
Selwood KE, Mcgeoch MA, Mac Nally R (2015) The effects of climate change and land-use change on demographic rates and population viability. Biol Rev 90:837–853. https://doi.org/10.1111/brv.12136
Simberloff D, Dayan T (1991) The guild concept and the structure of ecological communities. Annu Rev Ecol Syst 22:115–143. https://doi.org/10.1146/annurev.es.22.110191.000555
Smith JA, Thomas AC, Levi T et al (2018) Human activity reduces niche partitioning among three widespread mesocarnivores. Oikos 127:890–901. https://doi.org/10.1111/oik.04592
Taki H, Inoue T, Tanaka H, Makihara H, Sueyoshi M, Isono M, Okabe K (2010) Responses of community structure, diversity, and abundance of understory plants and insect assemblages to thinning in plantations. For Ecol Manag 259:607–613. https://doi.org/10.1016/j.foreco.2009.11.019
Torretta E, Mosini A, Piana M et al (2017) Time partitioning in mesocarnivore communities from different habitats of NW Italy: insights into martens’ competitive abilities. Behaviour 154:241–266. https://doi.org/10.1163/1568539X-00003420
Trolliet F, Huynen M-C, Vermeulen C, Hambuckers A (2014) Use of camera traps for wildlife studies. a review. Biotechnol Agron Soc Environ 18:446–454
Verdade LM, Rosalino LM, Gheler-Costa C, et al (2011) Adaptation of mesocarnivores (mammalia: carnivora) to agricultural landscapes in Mediterranean Europe and Southeastern Brazil: A trophic perspective. In: Middle-Sized Carnivores in Agricultural Landscapes. 1–39
Vilella M, Ferrandiz-Rovira M, Sayol F (2020) Coexistence of predators in time: effects of season and prey availability on species activity within a Mediterranean carnivore guild. Ecol Evol 10:11408–11422. https://doi.org/10.1002/ece3.6778
Wilkinson D, Smith GC (2001) A preliminary survey for changes in urban Fox (Vulpes vulpes) densities in England and Wales, and implication for rabies control. Mamm Rev 31:107–110. https://doi.org/10.1046/j.1365-2907.2001.00076.x
Zar JH (2010) Biostatistical Analysis, 5th edn. Pearson Prentice Hall, Upper Saddle River
Acknowledgements
We would like to thank The Navigator Company for providing logistical support crucial to developing our study. We also wish to thank Ana Manuel Figueiredo, Sofia Lino, Renato Hilário, and Nuno Negrões for help during fieldwork, and Margarida Teixeira for help with the revision of the manuscript.
Funding
Open access funding provided by FCT|FCCN (b-on). This work was financially supported by the project POCI-01–0145-FEDER-028204 (WildForests) funded by FEDER, through COMPETE2020—Programa Operacional Competitividade e Internacionalização (POCI), and by national funds (OE), through FCT/MCTES. Thanks are due for the financial support to CESAM (UIDP/50017/2020 + UIDB/50017/2020 + LA/P/0094/2020) and cE3c (UIDB/00329/2020), by FCT/MCTES through national funds, and the co-funding by the FEDER, within the PT2020 Partnership Agreement and Compete 2020. Daniela Teixeira was supported by a PhD Grant (SFRH/BD/131608/2017) from Foundation for Science and Technology (Fundação para a Ciência e a Tecnologia—FCT).
Author information
Authors and Affiliations
Contributions
Conceptualization: DFT, LMR. Methodology: DFT, LMR, CF. Formal analysis and investigation: DFT, GA-P, CC, CL, AM, GC. Writing — original draft preparation: DFT; Writing—review and editing: DFT, LMR, GA-P, CC, CL, AM, GC, CF. Funding acquisition: LMR, CF. Resources: LMR, CF. Supervision: LMR, CF. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
Not applicable.
Additional information
Communicated by Pedro Aragón.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Teixeira, D.F., Ares-Pereira, G., Camarinha, C. et al. Effect of anthropic disturbances on the activity pattern of two generalist mesocarnivores inhabiting Mediterranean forestry plantations. Biodivers Conserv 32, 1251–1270 (2023). https://doi.org/10.1007/s10531-023-02548-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10531-023-02548-4