Abstract
Rewetting drained agricultural peatlands aids in restoring their original ecosystem functions, including carbon storage and sustaining unique biodiversity. 30–60 cm of topsoil removal (TSR) before rewetting for Sphagnum establishment is a common practice to reduce nutrient concentrations and greenhouse gas emissions, and increase water conductivity. However, the topsoil is carbon-dense and preservation in situ would be favorable from a climate-mitigation perspective. The effect of reduced TSR on Sphagnum establishment and nutrient dynamics on degraded and rewetted raised bogs remains to be elucidated. We conducted a two-year field experiment under Sphagnum paludiculture management with three TSR depths: no-removal (TSR0), 5–10 cm (TSR5), and 30 cm (TSR30) removal. We tested the effects of TSR on Sphagnum establishment and performance, nutrient dynamics, and hotspot methane emissions. After two years, TSR5 produced similar Sphagnum biomass as TSR30, while vascular plant biomass was highest in TSR0. All capitula nitrogen (N > 12 mg/g) indicated N-saturation. Phosphorus (P) was not limiting (N/P < 30), but a potential potassium (K) limitation was observed in year one (N/K > 3). In TSR0, ammonium concentrations were > 150 µmol/l in year one, but decreased by 80% in year two. P-concentrations remained high (c. 100 µmol/l) at TSR0 and TSR5, and remained low at TSR30. TSR30 and TSR5 reduced hotspot methane emissions relative to TSR0. We conclude that all TSR practices have their own advantages and disadvantages with respect to Sphagnum growth, nutrient availability and vegetation development. While TSR5 may be the most suitable for paludiculture, its applicability for restoration purposes remains to be elucidated. Setting prioritized targets when selecting the optimal TSR with peatland rewetting is pivotal.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Peatlands are unique ecosystems, because they cover only 3% of Earth’s surface and store almost a third of terrestrial carbon, making them the carbon-densest ecosystem worldwide (Gorham 1991; Leifeld & Menichetti 2018; Scharlemann et al. 2014; Temmink et al. 2022). Despite this importance, approximately half of the peatlands in Europe and more than half in Southeast Asia have been drained to be used for agricultural purposes, turning them from carbon sinks into major sources of carbon dioxide (CO2) through accelerated peat oxidation (Bonn et al. 2016; Fluet-Chouinard et al. 2023; Hooijer et al. 2010). Peatland drainage further leads to severe and long-term land subsidence and increased flood risk in coastal areas, susceptibility to peat fires, and loss of unique biodiversity (Page & Baird 2016; Renger et al. 2002; Tapio-Biström et al. 2012).
To mitigate these adverse effects, rewetting is a long-term option to restore the carbon storage function of peatlands, stop land subsidence, bring back the water retention capacity and restore lost biodiversity (Günther et al. 2020; Joosten et al. 2017; Nugent et al. 2018; Temmink et al. 2023a). Furthermore, rewetting rapidly reduces CO2 emissions and can lead to the return of the carbon sequestration function of the peatland on the long-term (Günther et al. 2020; Zerbe et al. 2013). However, rewetted formerly intensively-drained peatlands that were in agricultural use can be susceptible to nutrient mobilization (especially phosphorus and nitrogen) and high methane (CH4) emissions (Harpenslager et al. 2015; Hemes et al. 2018; Zak & Gelbrecht 2007). In addition, in bogs—peatlands predominantly fed by precipitation (Joosten et al. 2017)—high nutrient levels typically favor more common and fast-growing plants, as they easily outcompete peatland-specific species, such as Sphagnum (Barkman 1992; Grünig 1994). As such, the natural development of peatland-specific vegetation is a slow process, especially if no other management measures are taken, such as reprofiling and/or removal of the existing vegetation (Kreyling et al. 2021; Mackin et al. 2017).
To promote fast establishment of a Sphagnum lawn in paludiculture (sustainable agriculture and forestry on rewetted peatlands) or to facilitate rapid establishment of target species in bog restoration, it is common practice to remove 30 to 60 cm top peat layer before rewetting (Allison & Ausden 2004; Gaudig et al. 2018; Harpenslager et al. 2015; Quadra et al. 2023). Topsoil removal (TSR) aims to level the surface and remove the degraded, rooted, low water-conducting and nutrient-rich grass sod layer (~ 10 cm) (Allison & Ausden 2004; Gaudig et al. 2018; Harpenslager et al. 2015; Quadra et al. 2023). Moreover, TSR strives to create better hydraulic conductivity of the peat surface by exposing the underlying peat that is generally less decomposed, as maintaining the water table level constantly a few centimeters below Sphagnum surface—which is important for Sphagnum paludiculture (Brust et al. 2018). TSR also removes the nutrient-rich top layer, which creates more suitable (i.e., nutrient-poor(er)) growth conditions for Sphagnum and reduces the potential for internal eutrophication and downstream nutrient pollution (Allison & Ausden 2004; Quadra et al. 2023; Zak & Gelbrecht 2007). Quadra et al. (2023) showed that the removal of 20 cm of topsoil results in 25% reduction of pore water phosphorus (P) on intensively managed agricultural peatlands. Temmink et al. (2023a) showed in a Sphagnum paludiculture that the nutrient legacy decreased over 10 years with depth, particularly the concentrations of ammonium (NH4+).
Sphagnum naturally thrives in acidic and oligotrophic conditions and their reintroduction on eutrophic and more alkaline peat often leads them to be outcompeted by vascular plants (Bergen et al. 2020; Gunnarsson & Rydin 2000; Heijmans et al. 2002). Under eutrophic conditions, the balance of macronutrients, particularly nitrogen (N), P and potassium (K), can be easily disrupted, especially due to substantially high N availability (Bragazza et al. 2004; Temmink et al. 2017; Vroom et al. 2020; Temmink et al. 2023a). For instance, Sphagnum growth can be hindered by excess of N, leading to saturation or reduced N-uptake efficiency (Limpens et al. 2011). However, recent research demonstrated that Sphagnum can thrive under high N conditions when there is sufficient P and K available, i.e. optimal NPK stoichiometry (e.g. Gaudig et al. (2020), Käärmelahti et al. (2023) and Temmink et al. (2017)). Bragazza et al. (2004) suggested that a N/K and N/P quotient in Sphagnum capitula below 3 and between 15 and 30, respectively, are optimal for Sphagnum growth.
Under N-rich conditions, vascular plants gain a competitive edge over mosses, because they can utilize N that leeches through the upper moss layer for their growth (Lamers et al. 2000; Limpens et al. 2003; Tomassen et al. 2003). If vascular plants are too abundant, they often reduce Sphagnum growth through water and nutrient consumption, litterfall and shading (Limpens et al. 2003, 2011; Malmer et al. 1994). Specifically, shading of more than 50% of Sphagnum by plants already negatively affects their growth (Clymo & Hayward 1982). At the same time, however, a small amount of vascular plants can be beneficial to Sphagnum growth, because shading creates an improved microclimate with less evaporation and more constant temperature, and the plants themselves provide a structuring effect that stimulates vertical moss growth (Pouliot et al. 2011). In the shade, Sphagnum cells have a higher water content that has been linked to greener plants (Breemen 1995), with better photosynthetic function, especially during drought periods (Laing et al. 2014). Furthermore, high abundance of vascular plants has also been linked to longer Sphagnum shoots (higher lawn), as taller Sphagnum are exposed to more light (Pouliot et al. 2011). In addition, Sphagnum cover indicates the success of a Sphagnum lawn; once green Sphagnum covers more than 90% of the surface, the productivity of a Sphagnum paludiculture greatly increases (Gaudig et al. 2017).
Rewetting nutrient-rich peat dominated by plants adapted to drained conditions will result in easily decomposable roots and plant biomass, fueling CH4 production and emissions (Bridgham & Richardson 1992; Schrier-Uijl et al. 2011). Although TSR has been found to reduce CH4 emissions post-rewetting, it also removes the stored carbon, which greatly affects the carbon budget of the site at high costs (Daun et al. 2023; Huth et al. 2022; Wichmann et al. 2020; Segers 1998). While TSR of 25 cm can lower CH4 emission by 99% (Harpenslager et al. 2015; Quadra et al. 2023), Quadra et al. (2023) found that removing as little as 5 cm already reduced CH4 emissions by 80%. Taking multiple perspectives into account, it seems that less than 30 cm of TSR can be sufficient for better nutrient conditions for Sphagnum establishment, while being carbon-conscious when restoring agricultural bogs (Huth et al. 2022; Quadra et al. 2023). However, it remains to be elucidated how TSR affects nutrient availability and the establishment and productivity of Sphagnum and vascular plants in nutrient-rich environment.
To unravel the effects of topsoil removal on Sphagnum establishment, we studied how different depths of TSR affect nutrient dynamics, Sphagnum performance and biomass accumulation under field paludiculture conditions. We aim to determine whether a Sphagnum lawn can successfully established with less TSR in relatively nutrient-rich field conditions. Therefore, we applied three levels of TSR (0, 5–10 and 30 cm) in a large-scale (c. 3000 m2) two-year field experiment. Next to this, we provide a snapshot of hotspot CH4 fluxes in the different treatments. We examined how less TSR affects nutrient availability, Sphagnum nutrient stoichiometry, Sphagnum performance, amount of Sphagnum and vascular plants, and hotspot CH4 fluxes. We hypothesized that (i) nutrient mobilization will be higher with less topsoil removal; (ii) 5–10 cm topsoil removal already substantially reduces the competition between vascular plants and Sphagnum; (iii) 5–10 cm topsoil removal will show a better nutrient stoichiometry for Sphagnum establishment and performance compared to no removal; (iv) 5–10 cm topsoil removal is enough to reduce hotspot fluxes of CH4.
Materials and methods
Study site and experimental setup
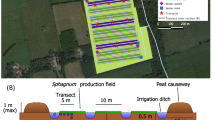
Our study site is part of a 17-hectare Sphagnum paludiculture study site located in the peatland Hankhauser Moor in NW Germany (53° 15.80′ N, 08° 16.05 ′E, Fig. 1). The bog, with a 2–2.5 m thick peat layer overlaying sand, has been drained and used as an agricultural grassland for six decades until land use changed to paludiculture. In November 2020, three fields of 20 × 50 m were installed by either only milling the surface layer i.e. no TSR (named TSR0), or removing the top layer of peat by an excavator in two different depths: 5–10 cm of TSR (named TSR5) and 30 cm of TSR (named TSR30) (Fig. 1). Each field is surrounded by a ring ditch of 0.5 × 0.5 m (width × depth) and is divided by a ditch in the centre, resulting in two 10 × 50 m subfields (Fig. 1). A mix of six Sphagnum species (S. papillosum, S. cuspidatum, S. palustre, S. divinum, S. medium, S. rubellum, S. fallax) or pure S. fallax (at the other subfields) was spread over the fields to achieve c. 65% initial cover following (Gaudig et al. 2017). Water tables were kept at c. 5 cm below the peat moss surface through active water management (i.e., pumping, outlet to prevent flooding, details see Brust et al. 2018). During the growing season, vascular plants were mown 4–6 times (all fields at the same time) a few centimetres above Sphagnum surface.
A The location of the field site in Germany indicated by a blue dot, B aerial view of the topsoil removal (TSR) treatments and locations of the sampling transects indicated by yellow lines, and C schematic cross-section of the TSR fields. NB: the schematic figure in C is not scaled; the remaining peat thickness is c. 2 m. Picture B: R.J.M. Temmink
Transect setup and porewater collection
Surface water and porewater samples were collected in four transects from the ditch to the centre of a subfield in each treatment (Fig. 1). At each transect, one ditch surface water sample was collected using a vacuum syringe attached to a ceramic cup sampler (Eijkelkamp Agrisearch Equipment, Giesbeek, The Netherlands; n = 4 per field). Per transect, four porewater samples were collected using syringes under vacuum attached to a rhizon (Rhizosphere Research Products, Wageningen, the Netherlands), placed at 0–10 cm depth relative to the surface. The rhizons were placed at 0.5, 1, 2.5, and 5 m from the ditch (n = 4 per distance per treatment) following Temmink et al. (2017, 2023a) and Vroom et al. (2020). Water samples were collected five times in 2021 (March, April, June, August, and November) and six times in 2022 (March, May, June, August, September and November). To prevent oxygenation of the samples, syringes were directly closed, stored at 4 °C and processed within two days. Upon processing (see method below) samples were split into two 10 mL subsamples, either stored at − 20 °C or after adding 0.1 mL of 65% nitric acid (HNO3) at room temperature, until further chemical analyses.
Biomass, cover and lawn height analyses
Sampling of Sphagnum and vascular plants for nutrient analyses and biomass of 2021 (one year after installation) followed methods published previously (Temmink et al. 2017; Vroom et al. 2020), where the samples were collected using quadrats of 10 by 10 cm. The samples were collected from the Sphagnum lawn down to the old initial peat surface, which was easily recognized by a much stronger degree of decomposition. These samples were collected at each transect at 0.5 and 5 m from the ditch in August 2021 and November 2022 for nutrient analyses and November 2021 for biomass. In November 2022, 10 additional biomass samples of 15 by 15 cm per treatment field were collected randomly (Hurlbert 1984) to be analysed for final biomass accumulation two years after installation. The amount of initial Sphagnum founder material was determined right after installation in November 2020 using similar random sampling method with 20 samples for each treatment.
For nutrient analyses, vascular plants, Sphagnum capitula and stems, and Sphagnum founder material were processed separately. All biomass fractions were weighed (fresh weight), dried for 72 h at 70 °C and weighed again (dry weight). Dried Sphagnum samples were homogenized at 18,000 rounds per minute for 2 min using 5 mm bullets (bullet grinder Mixer Mill MM 400, Retsch, Haan, Germany). Biomass samples were stored in dry conditions until chemical analyses.
Sphagnum (total and green) and vascular plant cover was monitored visually four times during the experiment: October–November 2020, May 2021, April 2022 and November 2022 using the scale of Londo (1976) at 20 randomly located plots (25 × 25 cm) per treatment (cf. Hurlbert 1984). Additionally, the Sphagnum lawn height was recorded at five points per plot.
Greenness index analysis
For Sphagnum greenness analyses, 10 × 10 samples were collected in June 2022 at the transects (see method for biomass sampling) from which three representative shoots were selected by hand and photographed on a white background, in a standardized manner. Subsequently, colour analyses of the Sphagnum shoots were performed with the image analysis software ImageJ (Schneider et al. 2012). Specifically, the capitula from each of the shoots was selected by drawing an elliptical shape around it, and the analyses were performed with the Colour Histogram command (three measurements per Sphagnum sample). The greenness index of the samples was calculated from the green, red and blue light values of the output, following the formula:
The higher the greenness index the greener the plant (high chlorophyll content), which is an indication of better photosynthetic function, and thus growth performance.
Chemical analyses
Water pH and alkalinity were determined the day after sampling using an Ag/AgCl electrode (Orion Research, Beverly, MA, USA) and a TIM 840 Titration Manager (Radiometer Analytical SAS, Villeurbanne, France). Concentrations of K and P in the water samples preserved with acid were measured using inductively coupled plasma optical emission spectrometry (ICP-OES ARCOS Spectro Analytical; Kleve, Germany). Ammonium (NH4+) concentrations of water samples stored at – 20 °C were determined by colorimetric methods following NEN protocols (NEN-EN-ISO 13395:1997; NEN-EN-ISO 11732:2005) adapted to the Seal Auto Analyzer System (SEAL Analytical Ltd., Norderstedt, Germany).
Total N and C in the Sphagnum capitula were determined from 4–5 mg of each milled biomass fraction with an elemental CNS analyser (Vario MICRO cube; Elementar Analysensysteme, Langenselbold, Germany). Total P and K were determined by digesting c. 200 mg dried moss material in 4 mL of HNO3 (65%) and 1 mL hydrogen peroxide (H2O2; 30%) in Teflon vessels, followed by heating in a microwave oven (EthosD, Milestone, Sorisole Lombardy, Italy). Elemental content was determined by inductively coupled plasma optical emission spectrophotometry (ICP-OES, Thermo Fischer Scientific, Bremen, Germany).
Methane flux measurements
CH4 fluxes were measured three times (once in May and twice in June) during 2022 from partly flooded parts of the field. As Daun et al. (in preparation) already conducted a GHG study on relatively dry fixed sublocations on these fields for annual GHG budget calculations, we selected wetter sub-sites to study CH4 dynamics on potential emission hotspots. With this we want to highlight the need for measurements with better spatial resolution, as the fluxes during these peak moments can be quite substantial. Gas measurements were carried out using an automatic chamber system with three PVC chambers per field (volume of 60 L, area of 0.1 m2), which were connected to a Li-COR Trace Gas Analyzer (LI-7810, LI-COR Corporate, Nebraska, USA). The automated system was developed by the Technocentrum at Radboud University in the Netherlands. The chambers have a built-in fan to allow constant airflow. The measurements lasted for 180 s per chamber, with 30 s of flushing time. In total, all treatments had 32 measurements (daytime only). Simultaneously with the gas measurements, the air temperature was logged using a HOBO logger (Onset Computer Corporation, Bourne, MA, USA). CH4 fluxes (mg/m2/d) were calculated following previous publications (Almeida et al. 2016; Paranaíba et al. 2020; Quadra et al. 2023). Briefly, the calculation considers the slope of the relationship between the gas over time; the volume and area of the chamber; atmospheric pressure and air temperature; the gas constant; and the molecular weight of CH4 (16 g/mole).
Statistical analyses
Statistical analyses were performed using R version 4.1.2 (R Core Team 2021). The effect of TSR and time (factors) on NH4+, P, K concentrations in porewater; plant biomass; Sphagnum lawn height N, P and K concentrations in Sphagnum biomass; C/N, N/P and N/K quotients; N, P and K capitula/stem nutrient allocation; green Sphagnum cover; Sphagnum lawn height; greenness index and CH4 fluxes (variables) were tested using linear mixed-effects models (lme) or generalized least squares (gls), from package ‘nlme’ (Pinheiro et al. 2023). Main effects of treatment, year and their interaction were included in the model. Model selection was done by first removing non-significant interactions and then main effects from the model in a backwards stepwise analysis. Normality and homogeneity of the residuals were visually assed for the best fitting model (Zuur et al. 2009). Non-normally distributed data were log10 or square root transformed (Table S1). Sphagnum biomass, cover and lawn height were tested only from November 2022. Distance from the irrigation ditch was included as a random effect and, in case of porewater concentrations, sampling time was included as a nested random effect. When differences were significant, a post-hoc test was applied (Tukey multiple comparisons of means) using ‘emmeans’ (Lenth 2018). Significance was assumed at p ≤ 0.05. Plots were created using ggplot2 (Wickham 2009). All results are shown with their standard error of the arithmetic mean (± SE).
Results
Biomass and Sphagnum greenness
The initial amount of the spread Sphagnum founder material in November 2020 was 77.4 ± 8.8 g DW/m2 (average ± SE, Fig. 2). Sphagnum biomass increased in all treatments from 2021 to 2022 (p < 0.05, Table S1, Fig. 2). The average Sphagnum biomass productivity two years after installation (November 2022) was highest at TSR5 and TSR30 with 740 ± 120 and 740 ± 78 g DW/m2, respectively (p > 0.05, Table S1). TSR0 had nearly six times lower Sphagnum biomass than TS5 and TS30 with 130 ± 110 g DW/m2 (Fig. 2). TSR0 had the highest percentage (70%) of plots without any Sphagnum biomass. In contrast, vascular plant biomass of 770 ± 120 g DW/m2 was the highest at TSR0 compared to the other fields with 330 ± 71 at TSR5 and 110 ± 25 g DW/m2 at TSR30 (p < 0.05, Table S1, Fig. 2). The greenness index in Sphagnum capitula was the highest at TSR0 (31 ± 2.4) and lowest at TSR5 (21 ± 1.9; Fig. 2).
A Sphagnum (grey) and vascular plant (white) biomass (g DW/m2) in November 2021 (n = 8) and 2022 (n = 10), the dashed horizontal line represents the average amount of Sphagnum founder material (g DW/m2) spread for installation, sampled in November 2020. B Greenness index in Sphagnum capitula in June 2022 (n = 8). Bars represent means with ± standard error (SE)
Sphagnum cover and lawn height
Total Sphagnum cover increased at TSR5 and TSR30 since installation, but decreased at TSR0 (Fig. 3). In November 2022, two years after the installation, green Sphagnum cover at TSR5 and TSR30 plots was similarly high (TSR5 83 ± 4.4%; TSR30 89 ± 3.1%), but much lower at TSR0 (10 ± 5.1%; p < 0.05, Fig. 3). At TSR0, green Sphagnum cover decreased during the first spring (May 2021) to 12 ± 3.5% and remained low until November 2022, whereas the vascular plant cover increased to 64 ± 4.7% (Fig. 3). Sphagnum lawn was higher at TSR5 in November 2022 with 15 ± 0.9 cm and at TSR0 with 12 ± 1.0 cm than at TSR30 with 8.0 ± 0.6 cm (p < 0.05, Table S1, Fig. 3).
Elemental contents and quotients in Sphagnum
Sphagnum capitula and stem N content were highest at TSR0 in 2021 with 16 ± 1.6 mg/g, but lower and similar to the other treatments in 2022 with 14 ± 0.7 mg/g (p < 0.05, Fig. 4, Table S1). Surprisingly, these N capitula concentrations were moderate, despite considerable NH4+ porewater concentrations (see 3.3), high atmospheric N deposition (21 kg ha−1 year−1; UBA 2023) and limited biomass accumulation (Fig. 2, 4, 5). Capitula K content was highest at TSR0 in both years, and decreased by 40% between 2021 and 2022 at TSR0 from 8.3 ± 0.6 to 4.9 ± 0.6 mg/g and increased by 160% at TSR30 from 1.8 ± 0.2 to 4.7 ± 0.4 mg/g (p < 0.05, Fig. 4, Table S1). The same trend was observed for K content in the stems, although they were lower than capitula content and the increase was not significant at TSR30 (Fig. 3, Table S1). P content both in capitula and stems decreased from 2021 to 2022 in all treatments (p < 0.05), but were similar between the treatments (average of all treatments 2021: 2.7 ± 0.3 and 2022: 1.3 ± 0.1 mg/g; Fig. 4, Table S1).
Ammonium (NH4+), phosphorus (P) and potassium (K) concentrations (µmol/l) in the porewater at different distances (m) from the irrigation ditch (0 represents ditch surface water, which was excluded from the model) in 2021 (n = 20) and 2022 (n = 24). Bars represent means ± standard error (SE). The vertical dotted line separates the ditch surface water and the porewater in the field
C/N quotients in capitula and stems followed an opposite pattern than N content, being the lowest at TSR0 with 28 ± 2.3 in 2021, but higher and similar to others with 32 ± 1.5 in 2022 (Fig. 4, Table S1). The mean C/N capitula quotient at TSR30 decreased by 30% from with 46 ± 2.9 in 2021 to 31 ± 1.2 in 2022 (p < 0.05, Fig. 4, Table S1). N/P quotients in both capitula and stems were generally low at all treatments but increased considerably at both TSR0 from 6.8 ± 0.9 to 15 ± 1.7 and TSR30 from 5.2 ± 0.6 to 15 ± 2.2 between 2021 and 2022. In 2022, N/P quotients were the lowest at TSR5 with 9.2 ± 0.4 (Fig. 4, Table S1). Both capitula and stem N/K quotients in 2021 were higher at TSR30 with 5.7 ± 0.6 compared to TSR5 with 3.0 ± 0.4 and TSR0 with 2.0 ± 0.3. All capitula N/K quotients were higher than 3 and similar between treatments in 2022 (average of all 3.3 ± 0.1; Fig. 4, Table S1). Capitula N/K quotient at TSR0 increased and in TSR30 decreased from 2021 to 2022, opposite from the trend for K contents (p < 0.05, Fig. 4, Table S1). N/K quotients in the stems were higher than in capitula in all treatments ranging from 3.8 ± 0.3 at TSR5 to 4.9 ± 0.6 in 2022 (Fig. 4). N and P were mostly equally distributed between capitula and stem (quotient close to 1), but K capitula/stem quotient increased from 2021 to 2022, ranging from 1.5 ± 0.1 in TSR30 to 1.6 ± 0.1 in TSR0 and TSR5 in 2022.
Surface and porewater nutrient concentrations
Topsoil removal resulted in lower nutrient concentrations in the porewater. Specifically, in the first year the average NH4+, P and K concentrations were highest at TSR0 with values of 160 ± 27, 500 ± 52 and 110 ± 13 µmol/l, respectively (Fig. 5). The average porewater P concentrations were approximately four times lower at TSR5 with 97 ± 9.5 µmol/l and virtually absent at TSR30 with 3.6 ± 0.4 µmol/l in both years. Mean NH4+, P and K concentrations were also higher at TSR5 than at TSR30 in both years, except for NH4+ in 2021 (p < 0.05, Table S2). There was a decrease in NH4+ and K concentrations between the years within TSR0 (p < 0.05, Table S2). Specifically, NH4+ concentrations decreased largely from 160 ± 27 in 2021 to 25 ± 1.2 µmol/l in 2022 (p < 0.05, Table S2, Fig. 5). Similarly, K concentrations in TSR0 decreased from 110 ± 13 in 2021 to 39 ± 2.9 µmol/l in 2022 (p < 0.05, Table S2). P porewater concentrations did not differ between years within any of the treatments (p > 0.05, Table S2).
The irrigation water could generally be characterized with low pH (5.4 ± 0.1) and very low alkalinity (0.33 ± 0.03 meq/l). NH4+ concentrations in the surface water of the irrigation ditches were similar between treatments (16 ± 1.2 µmol/l, p > 0.05, Table S2). However, on average over the two-year period, P and K concentrations in the ditches were higher at TSR0 than at TSR5 and TSR30. The mean surface water K concentration of 89 ± 12.0 µmol/l and P concentration of 58 ± 9.3 µmol/l at TSR0 were 1.7 and 2.9 times higher (respectively, p < 0.05, Table S2) than in the other treatments with near-detectable concentrations of P. Ditches were influenced by activities outside of the treatment fields which limits the information value of a direct comparison.
Hotspot methane fluxes
Spring/summer daytime CH4 fluxes from partly flooded lawns increased from spring to summer, with average (per treatment) fluxes ranging from 1.5 ± 0.6 to 18 ± 7.7 mg/m2/day in early May, 12 ± 3.6 to 110 ± 22 mg/m2/day in early June, and 3.4 ± 0.3 to 190 ± 40 mg/m2/day in mid-June (Fig. 6). We measured a reduction in CH4 emissions in TSR5 and TSR30 compared to TSR0 at the days with highest fluxes, in mid-June (p < 0.05, Fig. 6, Table S2).
Discussion
Our experiment revealed that topsoil removal affects Sphagnum establishment and nutrient content in Sphagnum and porewater nutrient concentrations. Specifically, Sphagnum establishment was rapid and its growth was the highest at both TSR5 and TSR30 with near-optimal nutrient supply. Nutrient availability was the highest at TSR0, which promoted the growth of vascular plants. On the other hand, removing 30 cm of topsoil possibly removed too many nutrients, which reduced the growth of vascular plants, but also reduced Sphagnum growth. Lastly, we found first indications that TSR5 slightly reduced hotspot CH4 emissions, but this warrants frequent monitoring of carbon fluxes spanning two years or longer (Huth et al. 2020).
Biomass
5–10 cm topsoil removal seems to have promoted balanced nutrient stoichiometry in Sphagnum, and led to substantial Sphagnum biomass (c. 740 g DW/m2) in only two years after establishment, which is in line with our hypothesis. Biomass at TSR5 was similar to TSR30, which is the current common practice on this site (Wichmann et al. 2017). Vroom et al. (2020) reported Sphagnum biomass of c. 900 g DW/m2 after 2.5 years of Sphagnum cultivation with 30 cm topsoil removal on this site, which is similar yearly growth as in our study for both TSR5 and TSR30. Interestingly, the TSR0 field did still produce Sphagnum biomass—though nearly six times less than the other treatments—with c. 11% green Sphagnum cover remaining after two years. Vascular plants dominated TSR0, however, this field produced healthy green (high greenness index) Sphagnum biomass with no clear signs of macronutrient limitation. However, without the regular mowing of the vascular plants it is likely that Sphagnum would have completely lost the competition for light as Sphagnum growth is reduced when shading exceeds 50% (Clymo & Hayward 1982). As the N deposition on this site is already very high (21 kg ha−1 year−1; UBA 2023)—more than twice as high as the critical load threshold defined by Bragazza et al. (2004) for natural bogs—the competitive equilibrium has already shifted to be more beneficial to vascular plants. Since not all N can be taken up by the mosses, it can leach through the moss layer and can become available for vascular plants (Lamers et al. 2000).
After two years of growth, we observed the greatest Sphagnum lawn height at TSR0 and TSR5. At higher N availability (also combined with high P), length growth of Sphagnum might be promoted to compete with vascular plants, while growing in less dense (Fritz et al. 2014; Bengtsson et al. 2021), although some reported results show that increased N availability does not change length growth (e.g., Li et al. 2018). Still, it is possible that the higher N concentrations at TSR5 relative to TSR30, and the lower cover of vascular plants (and, thus, less competition for resources) relative to TSR0, yet enough vascular plants to provide structuring effect (Malmer et al. 1994), might have created the most favorable conditions for Sphagnum growth. Length increment does reflect the ability to reach light, which is crucial for Sphagnum to compete with vascular plants (Bengtsson et al. 2021; Li et al. 2018). That was the case in an experiment carried out by Ma et al. (2015), where shading had no effect on overall biomass production, but caused an increase in length in all studied Sphagnum species. This might partly explain the higher lawn at TSR5 and TSR0.
To support Sphagnum growth and generate good quality biomass under nutrient-rich conditions in Sphagnum paludiculture, it is crucial to manage vascular plants and water level (Gaudig et al. 2017, 2018; Wichmann et al. 2017). For restoration, after an initial intervention, the area would (nearly) not be managed and left to develop naturally. After two years of regular mowing, peat moss growth was comparable in TSR5 and TSR30, not forgetting that Sphagnum biomass did also increase in TSR0. However, the long-term development of peat moss growth on natural succession (without mowing and water management) still needs to be investigated for all of these TSR depths.
Nutrient stoichiometry
Despite the high N input, Sphagnum did not display clear signs of nutrient imbalance or toxicity in Sphagnum paludiculture setting with vascular plant management. This could explain their successful establishment and rapid growth, especially at TSR5. The mean Sphagnum capitula N concentrations remained moderate though saturated (> 12 mg/g, Lamers et al. 2000) within all treatments. Only in 2021 at TSR0, capitula N concentration was slightly more elevated (> 15 mg/g), which van der Heijden et al. (2000) proposed to indicate N pollution stress. This treatment also had highest C/N quotients (more N relative to C than in TSR5 and TSR30). The higher concentration of N in the first year could be explained by higher porewater NH4+ concentrations and limited biomass accumulation (Figs. 2, 5). Interestingly, in 2022 mean N concentrations in the Sphagnum capitula at TSR0 were lower (13–14 mg/g) and similar to the other treatments in 2022, after two years of growth, comparable to those reported by Temmink et al. (2017) and Käärmelahti et al. (2023) from this same site. If environmental N load is increasing gradually, it has been shown that Sphagnum can adapt to this by decreasing N uptake efficiency and so avoid N toxicity (Fritz et al. 2014). However, the moderate capitula N contents and absence of N toxicity is most likely explained by sufficient supply of other macronutrients, namely P and K (Chapin et al. 1986).
Capitula N/P quotients remained low in all the treatments indicating no evident P limitation in any of the treatments. Capitula N/P quotient was the lowest at TSR5 (< 10) implying high uptake of P at medium to high N concentrations (optimal ratio of these nutrients) (Arróniz-Crespo et al. 2008; Granath et al. 2009, 2012). On the other hand, N/K quotients in the capitula were high (5.7) at TSR30 in 2021. This is clearly above capitula N/K quotient of 3, which Bragazza et al. (2004) proposed as a threshold for K limitation. The high N/K quotient in TSR30 indicates suboptimal potassium supply directly after installation. In 2022, in all treatments capitula N/K quotients decreased but remained elevated (N/K > 3) potentially reducing growth in high N environments. Moreover, in the stems, especially at TSR0 and TSR30, N/K quotients were well above 3 (4.5 and 4.9, respectively) suggesting a lower relative amount of K in the stems. This is supported by high capitula/stem K quotients in 2022, suggesting a translocation of K from the stems to capitula in order to have enough K to sustain growth probably supplied by the irrigation water (Bridgham et al. 1995; Vitousek 1982). Further research is necessary to find out whether clear K-limitation would occur in a longer term and if the topsoil removal of 30 cm could affect Sphagnum growth due to limited supply of K. In addition, nutrient addition experiments would be needed to quantify the effect of potassium on Sphagnum establishment and growth under high supply of N and P. Furthermore, it remains to be elucidated what would be the effects on Sphagnum stoichiometry without vascular plant management.
Nutrients in the porewater
We show that TSR prior to rewetting is very effective in reducing N, P and K and thereby potentially preventing leaching of these nutrients. Removing 5–10 cm of topsoil already reduced NH4+ and K porewater concentrations after two years (73% and 64% decrease, respectively). However, relatively high porewater P concentrations (97 ± 10 µmol/l) remained at TSR5, indicating fairly large P leaching potential. These results are in line with Quadra et al. (2023), who also found 54–76% reduction in porewater NH4+ concentrations with as little as 5 cm of topsoil removal, but it required 25 cm of TSR to reduce most of the stored P in Dutch peat soils drained for agriculture. Interestingly, in TSR0 a substantial decrease of NH4+ was observed from 2021 to 2022, where the average NH4+ concentrations were more than six times lower in the second year, suggesting efficient uptake by vascular plants. However, longer term monitoring would reveal whether the P mobilization reduces overtime.
Topsoil removal is a potent method to remove nutrients, but does not allow the removal of selective nutrients. Even though it may be necessary to remove excess N and P, this may lead to the loss of K, which is essential for Sphagnum growth and even more so for vascular plants, in particular with excess availability of N. However, as Sphagnum can thrive with lower K availability than most vascular plants, it can succeed better with low K-availability (Bragazza et al. 2004; Wassen et al. 2020). Especially mowing and removing the mowed biomass has been linked to reduced K availability (Koerselman et al. 1990). It is important to take into account that the removed topsoil is not completely removed from the system, but used for the construction of causeways next to the fields, allowing possible leaching of the nutrients into the water of the irrigation ditch. However, the ditch water concentrations of NH4+ and P were similar between the treatments, and K concentrations were only higher in the irrigation ditch of TSR0.
Porewater K concentrations at TSR30 in 2022 (9 ± 1 µmol/l) were lower than in previous studies with a similar amount of topsoil removal from this site after one year (39 ± 2 µmol/l, Käärmelahti et al. 2023) and 2.5 years (15 ± 4 µmol/l; Vroom et al. 2020) of Sphagnum cultivation. At TSR5, K concentrations of 2022 were higher (24 ± 2 µmol/l), indicating higher availability of potassium. It is likely that the topsoil removal together with wetter year of 2021 (less K in the irrigation water due to dilution) caused K-limitation at TSR30 during the first year. In the long term, K availability in the irrigation water and weather conditions are crucial to predict potential nutrient shortages. For example, Temmink et al. (2023a) showed that K concentrations in Sphagnum lawns gradually increased over 5 years. Overall, low porewater K concentration together with the higher stem N/K ratios at TSR30 at the end of the experiment indicate potential K limitation during the 2-year establishment period.
Hotspot methane fluxes
In line with previous studies, we also found that TSR reduced CH4 fluxes, even during hotspot moments in wetter parts of the field, with very low fluxes at TSR30 (i.e., Daun et al. 2023; Huth et al. 2020, 2022; Quadra et al. 2023). Our results are a pilot in the site to investigate the magnitude of CH4 fluxes under different TSR in a field scale experiment in partly flooded sublocations on the fields. Our data is limited, disregarding winter- and night time measurements, being unrealistic to draw robust conclusions or reliable annual estimates. However, this data does still provide a snapshot to CH4 fluxes on this site in favourable conditions for CH4 emissions (i.e., warmer periods under wet conditions). Within the scope of the present work, we showed that TSR reduced CH4 fluxes during these hotspots and hot moments, either visually observable (early May/June) or statistically significant (mid-June). For future studies and greenhouse gas budget calculations, it is important to consider the wintertime fluxes, as well as oxidation potential of the removed peat (Daun et al. in preparation). For instance, Daun et al. (2023) found that topsoil-made causeways were the major source of CO2 at this site. Although TSR30 reduced CH4 fluxes the most, for climate mitigation the TSR5 treatment may be a preferred option, because it minimizes carbon export through TSR.
Conclusions and implications
Our study shows the effects of TSR on nutrient dynamics, nutrient stoichiometry and Sphagnum performance on rewetted agricultural peatland under Sphagnum paludiculture management for two years directly after installation in the establishment phase. In terms of Sphagnum paludiculture with continuous management of water and vascular plants, 5–10 cm of TSR is likely to be the best cost–benefit option considering Sphagnum productivity and yields. However, in case of reduced or discontinuous management for bog restoration, Sphagnum establishment should be tested in the absence of vascular plant (and water) management and in the long term to verify whether Sphagnum is able to outcompete vascular plants. In addition, further long-term studies to monitor the greenhouse gas emissions with reduced amount of TSR are needed.
In a broader context, the worldwide decline of peatlands is causing adverse environmental effects such as loss of unique biodiversity, land subsidence, downstream pollution and long-term high greenhouse gas emissions. This highlights the urgent need for extensive restoration efforts to reinstate essential peatland functions on rewetted peatlands through bog restoration or Sphagnum paludiculture (i.e. wetscapes; Temmink et al. 2023b). Removing less topsoil could lower the threshold to establish Sphagnum paludiculture in terms of costs and labor intensity, which, in turn, can contribute significantly to the goals of the Paris Agreement and the UN Decade on Ecosystem Restoration. Productive use of wetlands could efficiently enhance the ecological functioning of peatlands while reducing the challenges associated with more extensive restoration efforts.
Data availability
Data available via Archiving and Networked Services (DANS) EASY: https://doi.org/10.17026/dans-zgm-2hz4.
Change history
03 February 2024
A Correction to this paper has been published: https://doi.org/10.1007/s10533-024-01119-1
References
Allison M, Ausden M (2004) Successful use of topsoil removal and soil amelioration to create heathland vegetation. Biol Cons 120(2):221–228. https://doi.org/10.1016/j.biocon.2004.02.017
Almeida RM, Nóbrega GN, Junger PC, Figueiredo AV, Andrade AS, de Moura CGB, Tonetta D, Oliveira ES, Araújo F, Rust F, Piñeiro-Guerra JM, Mendonça JR, Medeiros LR, Pinheiro L, Miranda M, Costa MRA, Melo ML, Nobre RLG, Benevides T, Kosten S (2016) High primary production contrasts with intense carbon emission in a eutrophic tropical reservoir. Front Microbiol. https://doi.org/10.3389/fmicb.2016.00717
Arróniz-Crespo M, Leake JR, Horton P, Phoenix GK (2008) Bryophyte physiological responses to, and recovery from, long-term nitrogen deposition and phosphorus fertilisation in acidic grassland. New Phytol 180(4):864–874. https://doi.org/10.1111/j.1469-8137.2008.02617.x
Barkman JJ (1992) Plant communities and synecology of bogs and heath pools in the Netherlands. In: Verhoeven JTA (ed) Fens and Bogs in the Netherlands. Springer Netherlands, Dordrecht, pp 173–235. https://doi.org/10.1007/978-94-015-7997-1_5
Bengtsson F, Rydin H, Baltzer JL, Bragazza L, Bu Z, Caporn SJM, Dorrepaal E, Flatberg KI, Galanina O, Gałka M, Ganeva A, Goia I, Goncharova N, Hájek M, Haraguchi A, Harris LI, Humphreys E, Jiroušek M, Kajukało K, Granath G (2021) Environmental drivers of Sphagnum growth in peatlands across the Holarctic region. J Ecol 109(1):417–431. https://doi.org/10.1111/1365-2745.13499
Bergen TJHM, Temmink RJM, Tweel-Groot L, Bakker WJ, Rehlmeyer K, Koks AHW, Waajen AC, Roelofs JGM, Grootjans AP, Heide T, Lamers LPM (2020) Self-facilitation and negative species interactions could drive microscale vegetation mosaic in a floating fen. J Veg Sci 31(2):343–354. https://doi.org/10.1111/jvs.12851
Bonn A, Allott T, Evans M, Joosten H, Stoneman R (2016) Peatland restoration and ecosystem services: an introduction. In: Bonn A, Joosten H, Evans M, Stoneman R, Allott T (eds) Peatland restoration and ecosystem services: science, policy and practice. Cambridge University Press, Cambridge, pp 1–16. https://doi.org/10.1017/CBO9781139177788.002
Bragazza L, Tahvanainen T, Kutnar L, Rydin H, Limpens J, Hájek M, Grosvernier P, Hájek T, Hajkova P, Hansen I, Iacumin P, Gerdol R (2004) Nutritional constraints in ombrotrophic Sphagnum plants under increasing atmospheric nitrogen deposition in Europe. New Phytol 163(3):609–616. https://doi.org/10.1111/j.1469-8137.2004.01154.x
Bridgham SD, Richardson CJ (1992) Mechanisms controlling soil respiration (CO2 and CH4) in southern peatlands. Soil Biol Biochem 24(11):1089–1099. https://doi.org/10.1016/0038-0717(92)90058-6
Bridgham SD, Pastor J, McClaugherty CA, Richardson CJ (1995) Nutrient-use efficiency: a litterfall index, a model, and a test along a nutrient-availability gradient in North Carolina peatlands. Am Nat 145(1):1–21. https://doi.org/10.1086/285725
Brust KM, Wahren KA, Gaudig G, Joosten H (2018) The water balance of a Sphagnum farming site in north-west Germany. Mires Peat 20:1–12
Chapin FS, Vitousek PM, Van Cleve K (1986) The nature of nutrient limitation in plant communities. Am Nat 127(1):48–58. https://doi.org/10.1086/284466
Clymo RS, Hayward PM (1982) The ecology of sphagnum. In: Smith AJE (ed) Bryophyte ecology. Springer Netherlands, Dordrecht, pp 229–289. https://doi.org/10.1007/978-94-009-5891-3_8
Daun C, Huth V, Gaudig G, Günther A, Krebs M, Jurasinski G (2023) Full-cycle greenhouse gas balance of a Sphagnum paludiculture site on former bog grassland in Germany. Sci Total Environ 877:162943. https://doi.org/10.1016/j.scitotenv.2023.162943
Daun C, Huth V, Gaudig G, Günther A, Krebs M, Jurasinski G. Minimising topsoil removal and ditch area are viable options to further optimise the greenhouse gas balance of Sphagnum paludiculture. Biogeochemistry (in prep)
Fritz C, Lamers LPM, Riaz M, van den Berg LJL, Elzenga TJTM (2014) Sphagnum mosses - masters of efficient N-uptake while avoiding intoxication. PLoS ONE 9:e79991
Fluet-Chouinard E, Stocker BD, Zhang Z, Malhotra A, Melton JR, Poulter B, Kaplan JO, Goldewijk KK, Siebert S, Minayeva T, Hugelius G, Joosten H, Barthelmes A, Prigent C, Aires F, Hoyt AM, Davidson N, Finlayson CM, Lehner B, McIntyre PB (2023) Extensive global wetland loss over the past three centuries. Nature 614(7947):281–286. https://doi.org/10.1038/s41586-022-05572-6
Gaudig G, Krebs M, Joosten H (2017) Sphagnum farming on cut-over bog in NW Germany: long-term studies on Sphagnum growth. Mires and Peat 20:1–19. https://doi.org/10.19189/MaP.2016.OMB.238
Gaudig G, Krebs M, Prager A, Wichmann S et al (2018) Sphagnum farming from species selection to the production of growing media: a review. Mires and Peat 20:1–30. https://doi.org/10.19189/MaP.2018.OMB.340
Gorham E (1991) Northern peatlands: role in the carbon cycle and probable responses to climatic warming. Ecol Appl 1(2):182–195. https://doi.org/10.2307/1941811
Granath G, Strengbom J, Breeuwer A, Heijmans MMPD, Berendse F, Rydin H (2009) Photosynthetic performance in Sphagnum transplanted along a latitudinal nitrogen deposition gradient. Oecologia 159(4):705–715. https://doi.org/10.1007/s00442-008-1261-1
Granath G, Strengbom J, Rydin H (2012) Direct physiological effects of nitrogen on Sphagnum: a greenhouse experiment: direct effects of N application on Sphagnum. Funct Ecol 26(2):353–364. https://doi.org/10.1111/j.1365-2435.2011.01948.x
Grünig A (1994). Mires and man: Mire conservation in a densely populated country -- the Swiss experience : excursion guide and symposium proceedings of the 5th Field Symposium of the International Mire Conservation Group (IMCG) to Switzerland 1992. https://www.academia.edu/64137918/Mires_and_man_mire_conservation_in_a_densely_populated_country_the_Swiss_experience_excursion_guide_and_symposium_proceedings_of_the_5th_Field_Symposium_of_the_International_Mire_Conservation_Group_IMCG_to_Switzerland_1992
Gunnarsson U, Rydin H (2000) Nitrogen fertilization reduces Sphagnum production in bog communities. New Phytol 147(3):527–537. https://doi.org/10.1046/j.1469-8137.2000.00717.x
Günther A, Barthelmes A, Huth V, Joosten H, Jurasinski G, Koebsch F, Couwenberg J (2020) Prompt rewetting of drained peatlands reduces climate warming despite methane emissions. Nat Commun 11(1):1644. https://doi.org/10.1038/s41467-020-15499-z
Harpenslager SF, van den Elzen E, Kox MAR, Smolders AJP, Ettwig KF, Lamers LPM (2015) Rewetting former agricultural peatlands: topsoil removal as a prerequisite to avoid strong nutrient and greenhouse gas emissions. Ecol Eng 84:159–168. https://doi.org/10.1016/j.ecoleng.2015.08.002
Heijmans MMPD, Klees H, Berendse F (2002) Competition between Sphagnum magellanicum and Eriophorum angustifolium as affected by raised CO2 and increased N deposition. Oikos 97(3):415–425. https://doi.org/10.1034/j.1600-0706.2002.970311.x
Hemes KS, Chamberlain SD, Eichelmann E, Knox SH, Baldocchi DD (2018) A biogeochemical compromise: the high methane cost of sequestering carbon in restored wetlands. Geophys Res Lett 45(12):6081–6091. https://doi.org/10.1029/2018GL077747
Hooijer A, Page S, Canadell JG, Silvius M, Kwadijk J, Wösten H, Jauhiainen J (2010) Current and future CO<sub>2</sub> emissions from drained peatlands in Southeast Asia. Biogeosciences 7(5):1505–1514. https://doi.org/10.5194/bg-7-1505-2010
Hurlbert SH (1984) Pseudoreplication and the design of ecological field experiments. Ecol Monogr 54(2):187–211. https://doi.org/10.2307/1942661
Huth V, Günther A, Bartel A, Hofer B, Jacobs O, Jantz N, Meister M, Rosinski E, Urich T, Weil M, Zak D, Jurasinski G (2020) Topsoil removal reduced in-situ methane emissions in a temperate rewetted bog grassland by a hundredfold. Sci Total Environ 721:137763. https://doi.org/10.1016/j.scitotenv.2020.137763
Huth V, Günther A, Bartel A, Gutekunst C, Heinze S, Hofer B, Jacobs O, Koebsch F, Rosinski E, Tonn C, Ullrich K, Jurasinski G (2022) The climate benefits of topsoil removal and sphagnum introduction in raised bog restoration. Restor Ecol. https://doi.org/10.1111/rec.13490
Joosten H, Tanneberger F, Moen A (eds) (2017) Mires and peatlands of Europe: Status, distribution and conservation. Schweizerbart Science Publishers, Stuttgart
Käärmelahti SA, Temmink RJM, van Dijk G, Prager A, Kohl M, Gaudig G, Koks AHW, Liu W, Vroom RJE, Gerwing K, Peters R, Krebs M, Fritz C (2023) Nutrient dynamics of 12 Sphagnum species during establishment on a rewetted bog. Plant Biol. https://doi.org/10.1111/plb.13534
Koerselman W, Bakker SA, Blom M (1990) Nitrogen, phosphorus and potassium budgets for two small fens surrounded by heavily fertilized pastures. The Journal of Ecology 78(2):428. https://doi.org/10.2307/2261122
Kreyling J, Tanneberger F, Jansen F, van der Linden S, Aggenbach C, Blüml V, Couwenberg J, Emsens W-J, Joosten H, Klimkowska A, Kotowski W, Kozub L, Lennartz B, Liczner Y, Liu H, Michaelis D, Oehmke C, Parakenings K, Pleyl E, Jurasinski G (2021) Rewetting does not return drained fen peatlands to their old selves. Nat Commun 12(1):5693. https://doi.org/10.1038/s41467-021-25619-y
Laing CG, Granath G, Belyea LR, Allton KE, Rydin H (2014) Tradeoffs and scaling of functional traits in Sphagnum as drivers of carbon cycling in peatlands. Oikos 123(7):817–828. https://doi.org/10.1111/oik.01061
Lamers LPM, Bobbink R, Roelofs JGM (2000) Natural nitrogen filter fails in polluted raised bogs: nitrogen filter fails in polluted raised bogs. Glob Change Biol 6(5):583–586. https://doi.org/10.1046/j.1365-2486.2000.00342.x
Leifeld J, Menichetti L (2018) The underappreciated potential of peatlands in global climate change mitigation strategies. Nat Commun 9(1):1071. https://doi.org/10.1038/s41467-018-03406-6
Lenth R (2018) Package ‘lsmeans.’ Amer Stat 34:216–221
Li T-T, Lei Y, Dai C, Yang L-F, Li Z-Q, Wang Z-X (2018) Effects of both substrate and nitrogen and phosphorus fertilizer on Sphagnum palustre growth in subtropical high-mountain regions and implications for peatland recovery. Wetlands Ecol Manage 26(4):651–663. https://doi.org/10.1007/s11273-018-9598-7
Limpens J, Berendse F, Klees H (2003) N deposition affects N availability in interstitial water, growth of Sphagnum and invasion of vascular plants in bog vegetation. New Phytol 157(2):339–347. https://doi.org/10.1046/j.1469-8137.2003.00667.x
Limpens J, Granath G, Gunnarsson U, Aerts R, Bayley S, Bragazza L, Bubier J, Buttler A, van den Berg LJL, Francez A, Gerdol R, Grosvernier P, Heijmans MMPD, Hoosbeek MR, Hotes S, Ilomets M, Leith I, Mitchell EAD, Moore T, Xu B (2011) Climatic modifiers of the response to nitrogen deposition in peat-forming Sphagnum mosses: a meta-analysis. New Phytologist 191(2):496–507. https://doi.org/10.1111/j.1469-8137.2011.03680.x
Londo G (1976) The decimal scale for releves of permanent quadrats. Vegetatio 33(1):61–64. https://doi.org/10.1007/BF00055300
Ma, J. Z., Bu, Z., Zheng, X. X., Ge, J., & Wang, S. (2015). Effects of shading on relative competitive advantage of three species of Sphagnum. Mires and Peat. https://www.semanticscholar.org/paper/Effects-of-shading-on-relative-competitive-of-three-Ma-Bu/c329b0ab080c590cd5aac1321a1654053cb09ae4
Mackin, F., Barr, A., Rath, P., Eakin, M., Ryan, J., Jeffrey, R., & Valverde, F. F. (2017). Best practice in raised bog restoration in Ireland [Report]. National Parks and Wildlife Service, Department of Culture, Heritage and the Gaeltacht. http://www.tara.tcd.ie/handle/2262/82140
Malmer N, Svensson BM, Wallén B (1994) Interactions between Sphagnum mosses and field layer vascular plants in the development of peat-forming systems. Folia Geobotanica Et Phytotaxonomica 29(4):483–496. https://doi.org/10.1007/BF02883146
Nugent KA, Strachan IB, Strack M, Roulet NT, Rochefort L (2018) Multi‐year net ecosystem carbon balance of a restored peatland reveals a return to carbon sink. Global Change Biol 24:5751–5768
Page SE, Baird AJ (2016) Peatlands and global change: response and resilience. Annu Rev Environ Resour 41(1):35–57. https://doi.org/10.1146/annurev-environ-110615-085520
Paranaíba JR, Quadra G, Josué IIP, Almeida RM, Mendonça R, Cardoso SJ, Silva J, Kosten S, Campos JM, Almeida J, Araújo RL, Roland F, Barros N (2020) Sediment drying-rewetting cycles enhance greenhouse gas emissions, nutrient and trace element release, and promote water cytogenotoxicity. PLoS ONE 15(4):e0231082. https://doi.org/10.1371/journal.pone.0231082
Pinheiro J, Bates D, DebRoy S, Sarkar D (2023) Linear and nonlinear mixed effects models. R Core Team
Pouliot R, Rochefort L, Karofeld E, Mercier C (2011) Initiation of Sphagnum moss hummocks in bogs and the presence of vascular plants: Is there a link? Acta Oecologica 37(4):346–354. https://doi.org/10.1016/j.actao.2011.04.001
Quadra GR, Boonman CCF, Vroom RJE, Temmink RJM, Smolders AJP, Geurts JJM, Aben RCH, Weideveld STJ, Fritz C (2023) Removing 10 cm of degraded peat mitigates unwanted effects of peatland rewetting: a mesocosm study. Biogeochemistry 163(1):65–84. https://doi.org/10.1007/s10533-022-01007-6
R Core Team (2021) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria
Renger M, Wessolek G, Schwärzel K, Sauerbrey R, Siewert C (2002) Aspects of peat conservation and water management. J Plant Nutr Soil Sci 165(4):487. https://doi.org/10.1002/1522-2624(200208)165:4%3c487
Scharlemann JP, Tanner EV, Hiederer R, Kapos V (2014) Global soil carbon: understanding and managing the largest terrestrial carbon pool. Carbon Management 5(1):81–91. https://doi.org/10.4155/cmt.13.77
Schneider CA, Rasband WS, Eliceiri KW (2012) NIH Image to ImageJ: 25 years of image analysis. Nat Methods 9(7):671–675. https://doi.org/10.1038/nmeth.2089
Schrier-Uijl AP, Veraart AJ, Leffelaar PA, Berendse F, Veenendaal EM (2011) Release of CO2 and CH4 from lakes and drainage ditches in temperate wetlands. Biogeochemistry 102(1–3):265–279. https://doi.org/10.1007/s10533-010-9440-7
Segers R (1998) Methane production and methane consumption: a review of processes underlying wetland methane fluxes. Biogeochemistry 41(1):23–51. https://doi.org/10.1023/A:1005929032764
Tapio-Biström M-L, Joosten H, Tol S (eds) (2012) Peatlands: guidance for climate change mitigation through conservation, rehabilitation and sustainable use, 2nd edn. Food and Agriculture Organization of the United Nations: Wetlands International, Location
Temmink RJM, Fritz C, van Dijk G, Hensgens G, Lamers LPM, Krebs M, Gaudig G, Joosten H (2017) Sphagnum farming in a eutrophic world: the importance of optimal nutrient stoichiometry. Ecol Eng 98:196–205. https://doi.org/10.1016/j.ecoleng.2016.10.069
Temmink RJM, Lamers LPM, Angelini C, Bouma TJ, Fritz C, van de Koppel J, Lexmond R, Rietkerk M, Silliman BR, Joosten H, van der Heide T (2022) Recovering wetland biogeomorphic feedbacks to restore the world’s biotic carbon hotspots. Science 376(6593):eabn1479. https://doi.org/10.1126/science.abn1479
Temmink RJM, Vroom RJE, van Dijk G, Käärmelahti SA, Koks AHW, Joosten H, Krebs M, Gaudig G et al (2023a) Restoring organic matter, carbon and nutrient accumulation in degraded peatlands: 10 years Sphagnum paludiculture. Biogeochemistry, 1–15. https://doi.org/10.1007/s10533-023-01065-4
Temmink RJM, Robroek BJM, van Dijk G, Koks AHW, Käärmelahti SA, Barthelmes A, Wassen MJ, Ziegler R et al. (2023b) Wetscapes: restoring and maintaining peatland landscapes for sustainable futures. Ambio 52:1519–1528. https://doi.org/10.1007/s13280-023-01875-8
Tomassen HBM, Smolders AJP, Lamers LPM, Roelofs JGM (2003) Stimulated growth of Betula pubescens and Molinia caerulea on ombrotrophic bogs: role of high levels of atmospheric nitrogen deposition. J Ecol 91(3):357–370. https://doi.org/10.1046/j.1365-2745.2003.00771.x
UBA (2023). Hintergrundbelastungsdaten Stickstoff. https://gis.uba.de/website/depo1/de/index.html
Van Breemen N (1995) How Sphagnum bogs down other plants. Trends Ecol Evol 10(7):270–275. https://doi.org/10.1016/0169-5347(95)90007-1
van der Heijden E, Verbeek SK, Kuiper PJC (2000) Elevated atmospheric CO2 and increased nitrogen deposition: effects on C and N metabolism and growth of the peat moss Sphagnum recurvum P. Beauv. Var. mucronatum (Russ.) Warnst: ELEVATED CO 2, N and SPHAGNUM RECURVUM. Global Change Biology 6(2):201–212. https://doi.org/10.1046/j.1365-2486.2000.00303.x
Vitousek P (1982) Nutrient cycling and nutrient use efficiency. Am Nat 119(4):553–572. https://doi.org/10.1086/283931
Vroom RJE, Temmink RJM, van Dijk G, Joosten H, Lamers LPM, Smolders AJP, Krebs M, Gaudig G, Fritz C (2020) Nutrient dynamics of Sphagnum farming on rewetted bog grassland in NW Germany. Sci Total Environ 726:138470. https://doi.org/10.1016/j.scitotenv.2020.138470
Wassen MJ, Schrader J, Van Dijk J, Eppinga MB (2020) Phosphorus fertilization is eradicating the niche of Northern Eurasia’s threatened plant species. Nat Ecol Evolut 5(1):67–73. https://doi.org/10.1038/s41559-020-01323-w
Wichmann S, Prager A, Gaudig G (2017) Establishing Sphagnum cultures on bog grassland, cut-over bogs, and floating mats: procedures, costs and area potential in Germany. Mires and Peat 20:1–19. https://doi.org/10.19189/MaP.2016.OMB.235
Wichmann S, Krebs M, Kumar S, Gaudig G (2020) Paludiculture on former bog grassland: profitability of Sphagnum farming in North West Germany. Mires and Peat 26:18. https://doi.org/10.19189/MaP.2019.SNPG.StA.1768
Wickham H (2009) Ggplot2: elegant graphics for data analysis. Springer, Berlin
Zak D, Gelbrecht J (2007) The mobilisation of phosphorus, organic carbon and ammonium in the initial stage of fen rewetting (a case study from NE Germany). Biogeochemistry 85(2):141–151. https://doi.org/10.1007/s10533-007-9122-2
Zerbe S, Steffenhagen P, Parakenings K, Timmermann T, Frick A, Gelbrecht J, Zak D (2013) Ecosystem service restoration after 10 years of rewetting peatlands in NE Germany. Environ Manage 51(6):1194–1209. https://doi.org/10.1007/s00267-013-0048-2
Zuur AF, Ieno EN, Walker NJ, Saveliev AA, Smith GM (eds) (2009) Mixed effects models and extensions in ecology with R. Springer, Berlin
Acknowledgements
The authors would like to thank Sebastian Krosse and Paul van der Ven from the General Instrumentation for help with chemical analyses; Roy Peters, Germa Verheggen, Ian Hermsen and Roy van Swam for sample preparation, and Keno Gerwing, Gina Maessen and Moritz Adam for sample collection and preparation. Christian Fritz got funded by WET HORIZONS GAP-101056848. Gabrielle Quadra received support from Interreg NWE Carbon Connects. The project was supported by NBank ‘Optimoos’ Project. This study has been facilitated by the German Federal Ministry of Food, Agriculture and Consumer Protection (BMEL) and Torfwerk Moorkultur Ramsloh Werner Koch GmbH & Co. KG, whose financial and in-kind support is gratefully acknowledged. The concept for this paper was developed at the workshop titled "Peatlands for climate change mitigation in agriculture" that took place in Aarhus, Denmark, on 4–5 October 2022, and which was sponsored by the Organisation for Economic Co-operation and Development (OECD) Co-operative Research Programme: Sustainable Agricultural and Food Systems. The opinions expressed and arguments employed in this publication are the sole responsibility of the authors and do not necessarily reflect those of the OECD or of the governments of its Member countries.
Funding
Christian Fritz got funded by WET HORIZONS GAP-101056848. Gabrielle Quadra received support from Interreg NWE Carbon Connects. The project was supported by NBank ‘Optimoos’ Project. This study has been facilitated by the German Federal Ministry of Food, Agriculture and Consumer Protection (BMEL) and Torfwerk Moorkultur Ramsloh Werner Koch GmbH & Co. KG.
Author information
Authors and Affiliations
Contributions
SAK, RJMT, GRQ and MEG wrote the original draft and all authors contributed to the subsequent drafts. All authors contributed to the methodology. SAK and GRQ analyzed and visualized the data. SAK, GRQ, MK and GG contributed to carrying out the field sample collection and/or analyses. MK and GG were responsible for project administration. CF was responsible for conceptualization and CF and RJMT for supervision. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors have no conflict of interest to declare.
Additional information
Responsible Editor: Klaus Butterbach-Bahl
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original online version of this article has been updated to correct the "n" value in Figure 3. The initial value was "n=100" which was corrected to "n=20".
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Käärmelahti, S.A., Fritz, C., Quadra, G.R. et al. Topsoil removal for Sphagnum establishment on rewetted agricultural bogs. Biogeochemistry 167, 479–496 (2024). https://doi.org/10.1007/s10533-023-01096-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10533-023-01096-x