Abstract
The energy demand to produce cellulose nanofibrils, CNFs, is high and additionally the cost of the starting material, the pulp, is substantial as high purity cellulose dissolving pulp is generally used. Pulps aimed for board and paper are produced at higher yield as they contain hemicelluloses and, in the case of unbleached pulp, lignin, and would be a more economical starting material for CNFs. It is of interest to understand how the presence of hemicellulose and lignin affects the fibrillation process and CNF properties. Kraft cooks of softwood were performed as well as kraft cooks with addition of polysulfide to increase the hemicellulose content. Part of the pulps were bleached to remove residual lignin, thus making it possible to compare pulps with and without lignin. Higher amount of hemicellulose had an obstructive effect on the enzymatic pre-treatment whereas lignin had no adverse effect on enzyme accessibility. Increased amount of charged groups improved the accessibility for enzymes. Both hemicellulose and lignin were carboxymethylated when pre-treatment by carboxymethylation was employed. However, carboxymethylation partly dissolved hemicelluloses. The tensile strength of CNF films was independent of the chemical composition of the pulp and the pre-treatment strategy. However, since the enzymatic pre-treatment decreased the cellulose DP more, CNF films from enzymatically pre-treated pulps had generally lower tensile strength.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Cellulose nanofibrils, CNFs, are obtained by submitting cellulosic pulp fibers to shearing forces until the fiber wall is fibrillated into individual fibrils. To reduce the energy demand in the fibrillation process, the pulp fibers are pre-treated to reduce the fiber wall strength, for example by enzymatic hydrolysis or by introducing charged groups by TEMPO oxidation or carboxymethylation. CNFs are mainly produced from dissolving pulps, which are characterized by high purity regarding cellulose. The other main wood constituents, hemicelluloses and lignin, have been dissolved in the cooking and bleaching stages and consequently the pulp yield is low. Kraft pulps, aimed for board and paper, have higher yield, as they contain hemicelluloses and, in the case of unbleached pulp, also lignin. Board and paper grade pulp would be a more economical starting material for CNFs, and it is of interest to understand how presence of hemicellulose and lignin remaining in the pulp affects the fibrillation process and the properties of the CNF films. There are indications that higher hemicellulose content improves the fibrillation yield (Chaker et al. 2013; Fall et al. 2014). However, Chaker et al. (2013) compared chlorite delignified samples with samples delignified by alkali and the different processes might have also influenced the fibers. Fall et al. (2014) compared fibers from different raw materials containing different amounts of hemicellulose. Comparing the production of CNFs from the fine materials fraction from either mechanical pulp or bleached chemical pulp has revealed that the mechanical pulp fines, rich in lignin and hemicellulose, were easier to fibrillate than the bleached chemical pulp fines (Osong et al. 2013). The presence of lignin in pulp fibers has been reported to lead to easier (Ferrer et al. 2012; Solala et al. 2012) and more homogeneous fibrillation (Laitinen et al. 2014) and the fibrils are thinner the higher the lignin content (Rojo et al. 2015; Jiang et al. 2018). Oliaei et al. (2019) obtained higher nanofibril yield from unbleached pulps with kappa number around 30 than from bleached pulps.
In the previous studies, chemical pulps have been compared with mechanical pulps or pulps with different degree of delignification. However, the difference in lignin content in previous studies was also accompanied by differences in hemicellulose content and raw material. In the present study, pulps have been manufactured with the aim to be able to distinguish between the influence of lignin and hemicellulose. Comparison was made between pulps with different lignin content but same hemicellulose content and between pulps with same lignin content but different hemicellulose content. The effect on pre-treatment stage, homogenization and CNF properties was evaluated.
Materials and methods
Industrially produced softwood chips of Norway spruce (Picea abies) were screened, keeping the fraction with a thickness of 4–8 mm. They were subsequently dried to a moisture content of approx. 8% after which bark and knots were removed by hand. The chemical composition of the chips was 44% cellulose, 17% galactoglucomannan, 6% xylan, 31% lignin and 1% extractives. NaOH pastilles of puriss grade (VWR International AB, Radnor, PA, USA) and Na2S technical grade flakes (VWR International AB) were dissolved in deionized water to obtain stock solutions of NaOH and Na2S for cooking.
Five pulps with different chemical composition were prepared according to Fig. 1.
Batches of 250 g o.d. (oven dry) chips were delignified in steel autoclaves of 2.5 dm3 volume. Air was removed from autoclaves by vacuum suction, followed by addition of 1 500 ml deionized water to each autoclave. A pressure of 5 bar was applied by nitrogen gas. After 30 min, the gas was released, water was drained from the chips and the water impregnated chips were weighed. Cooking liquor was added with an effective alkali charge of 22% and sulfidity of 35%. The liquor-to-wood was 5 l/kg o.d. wood and the initial hydroxide and hydrosulfide ion concentrations are presented in Table 1. The autoclaves were placed in a glycol bath at 25 °C and the temperature was ramped by 5 °C/min to 100 °C. To ensure good impregnation of the chips with cooking liquor prior to delignification, the autoclaves were kept at 100 °C for 30 min. Next, the temperature was increased by 5 °C/min to 160 °C. The kraft pulping was terminated after a certain cooking time, given as H-factor in Table 1.
Oxygen delignification was performed in Teflon-coated steel autoclaves at pulp consistency 12%, with 0.5% MgSO4 and 2.5% NaOH for the kraft pulps and 2.4% for the PS pulp. The temperature was 100 °C and the time was 18 min for the kraft pulp with kappa number 40 (end-pH 12.5) and 105 min (end-pH 11.2–11.5) for the pulps to be fully bleached. Bleaching was performed in plastic bags in a sequence starting with chlorine dioxide (D), followed by alkaline extraction (E) and a second D stage. For conditions in the bleaching stages, see Table 2. The pulps were washed with deionized water after each stage.
A commercial softwood dissolving pulp was used as reference.
Enzymatic treatment
Enzymatic CNFs were prepared similar to a previously described procedure including refining and endoglucanase enzymes pre-treatment steps and defibrillation using a high-pressure microfluidizer (Microfluidizer M-110EH, Microfluidics Corp., USA) (Pääkkö et al. 2007), but with modifications for the pulps in the present study. The pulps were PFI refined 6000 revolutions to increase the accessibility of the fiber wall. Subsequently, the endoglucanase was mixed with a buffer solution at pH 7 (KH2PO4/Na2HPO4 and added to the pulp. The enzymatic treatment was performed at 50 °C for 2 h. The enzyme was deactivated by increasing the temperature to 90 °C and keeping it at the temperature for 45 min followed by washing with deionized water. Prior to homogenization, the pulps were PFI refined again. The bleached pulps were refined 15 000 revolutions, the oxygen delignified pulp 25 000 revolutions and the unbleached pulps 20 000 revolutions.
Carboxymethylation
Carboxymethylated CNFs were prepared by a carboxymethylation pretreatment of fibers followed by a high-pressure homogenization technique similar to a previously described procedure (Wågberg et al. 2008). The pulps were dispersed in deionized water at 10,000 revolutions in a laboratory disintegrator and subsequently solvent-changed to methanol by washing in methanol four times with an intermediate filtration step. In the next step the pulps were impregnated for 30 min with a solution of monochloroacetic acid and isopropanol. The pulps were then added to a solution of NaOH in methanol mixed with isopropanol which had been preheated to below its boiling temperature. The carboxymethylation reaction was allowed to continue for 1 h after which the pulps were filtered and washed in three steps: first with deionized water, then acetic acid (0.1 M), and finally with deionized water. The fibers were then impregnated with a NaHCO3 solution (4 wt % solution) for 60 min in order to convert the carboxyl groups to their sodium form. Finally, the pulps were washed with deionized water and drained on a Buchner funnel. Total charge of the fibre DS ≈ 0.1
Homogenization
After the pretreatments, the pulps were homogenized using a high-pressure fluidizer (Microfluidizer M-110EH, Microfluidics Corp.). The fluidizer was equipped with two chambers of different sizes connected in series (200 and 100 µm). Full homogenization could be achieved with a single pass at a fiber consistency of 2 wt% and an operating pressure of 1650 bar.
Chemical composition
The kappa number, a measurement of lignin content in pulp, was analysed according to ISO 302:2004. The carbohydrate composition was performed according to SCAN-CM 71 by acid hydrolysis followed by ion chromatography; the measurement uncertainty was ± 2 mg/g for monosaccharides at the level of 1–10 mg/g and ± 20% for monosaccharides at a level > 10 mg/g. The xylan content was calculated as the sum of xylose and arabinose and a uronic acid content of 4.1%. The galactoglucomannan was calculated using a mannose to glucose ratio of 4.2.
Total amount of charged groups
The total charge (µeq/g) was determined by conductometric titration according to the SCAN-CM 65:02, using an automated Metrohm titrator. About 0.5 g (dry mass) of pulp was mixed in 80 ml of deionized water for 15 min using a magnetic stirrer. The pH of the suspension was adjusted to pH 2 addition of 1 M hydrochloric acid, and the suspension was allowed to equilibrate under stirring for 30 min. Deionized water was subsequently added to a total volume of 500 ml and titrated with 0.10 M sodium hydroxide. The amount of sodium hydroxide, corresponding to the plateau value in the titration curve was used to calculate the total charge. Experiments were performed in triplicate.
Degree of polymerization
The molecular weight (degree of polymerization (DP)) of the carboxymethylated pulps and CNFs were estimated from an average intrinsic viscosity value. The measurements were performed on fibers dissolved according to SCAN-CM 15:99 with cupriethylendiamine as solvent. Two samples were made for each pulp and CNF quality. The intrinsic viscosity, η (ml/g), is related to the degree of polymerization (DP) and an empirical relationship for this polymer–solvent system is suggested as η = 2.28 DP0.76 (Marx-Figini 1978). The presence of any hemicelluloses is not considered when calculating the DP.
Transmittance
The in-line transmittance of the CNF gels was measured in the UV–visible region with a PerkinElmer Lambda 265 UV–Vis spectrophotometer. The transmittance spectra were acquired using water as background.
Rheology
Rheology measurements were performed using a Kinexus stress-controlled rotational rheometer (Malvern Instruments, Malvern, UK) together with iSpace software (Malvern Instruments, Malvern, UK). A standard (ISO 3219/DIN 53019) metal concentric cylinder (bob and cup geometry) was used in the studies. The height and distance between the serrations were 300 and 1000 µm, respectively (supplier information, Malvern Instruments, Malvern, UK). The diameter and length of the used bob were 25 and 37.5 mm, respectively; the diameter and wall height of the cup were 27.5 and 62.5 mm, respectively. A working gap of 9.15 mm was employed in the measurements. The samples were loaded into the device at 2 wt% consistency and pre-sheared at 100 s−1 for 1 min and then allowed to equilibrate for 2 min before the measurements began. The experimental temperature was 25 °C; a protective hood was used during the measurements to minimize possible effects from the drying of the samples. The controlled shear rate measurements were conducted in the range of 0.01–1000 s−1. Integration time per measuring point was set to 30 s.
Nanofraction determination
The procedure for nanofraction analysis is described in Naderi et al. (2017).
Samples were prepared by blending the CNF dispersions of approx. 2wt% consistency with water to a consistency of about 0.02 wt% and stirred overnight. Part of the suspension was heated at 105 °C to determine solid content gravimetrically and the second part was centrifuged at 1000 g for 15 min, to remove the larger constituents (e.g. residual fibre fragments). After centrifugation, the supernatant was collected, weighed, and dried at 105 °C to determine its solid content. The dispersion concentrations before and after centrifugation were used to estimate the apparent fraction of nano-sized cellulosic materials in the dry content of the suspension.
It is noted that the nanofraction denotes the entities which have the ability to resist phase separation during centrifugation. It is further noted that this method of analysis is based on the assumption that the magnitude of the nanofraction increases with the increasing efficiency of the delamination process.
Turbidity
Turbidity was measured to qualitatively determine the presence of larger components in the dispersions. The sizes of these structures are 10–100 s micrometres. It is mainly these fragments that scatter light (scattering from the nanosized structures can be discarded), hence a decrease in turbidity at a fixed dry content indicates a reduction in the number and/or size of larger fragments.
A simple turbidity meter (2100P from Hach, USA) and 20 mL of dispersion was used to measure the turbidity. Measurements before and after filtration were made. Dispersions were diluted to a dry content close to 0.1wt%, in order to measure all dispersions at the same concentration.
Film preparation and mechanical testing
0.1 wt% CNF dispersions were prepared by diluting the 2 wt% dispersions by magnetic stirring for 24 h to ensure full mixing. CNF films were prepared through vacuum filtration, using 0.65 μm DVPP filters (supplied by Millipore), followed by drying under restrained conditions in an oven for 7 h at 50 °C. Target film grammage was 30–40 g/m2 (equal to a film thickness of 20–27 µm).
Tensile testing was performed at 23 °C and 50% relative humidity with an Instron 5944 MicroTester (Instron, Norwood, MA, USA), equipped with a 500 N load cell. Test pieces, 5 mm wide were clamped with a gauge length of 20 mm and strained at a rate of 2 mm/min.
Dynamic vapour sorption
A DVS apparatus (Q5000 SA, TA Instruments, US) was used to determine the isotherms and dynamic sorption behaviour of the CNF samples. The data such as the running time, real-time mass of the samples, and the actual RH at a constant temperature of 25 °C were obtained during the sorption process. The RH was set to change from 0 to 90% in 10% steps and then decrease to 0%. At every stage, the RH was kept constant for some time and then increased to the next stage as the changes in sample mass were smaller than 0.002% per minute.
The oxygen transmission rate (OTR) tests were performed with a Mocon Ox-Tran Model 2/21 apparatus (Mocon, Minneapolis, USA) in accordance with ASTM D 3985-06 and the test conditions were 23 °C at 50% RH. The sample area was 5 cm2 and the partial pressure of the oxygen was 1 atm (100% oxygen). The OTR was normalized with respect to material thickness to yield the oxygen permeability (OP). Two measurements were made for each film composition and the mean value is reported.
Water vapor transmission rate (WVTR)
The water vapor transmission rate (WVTR) tests were performed with a Mocon Permatran 3/33 MG apparatus (Mocon, Minneapolis, USA) in accordance with ASTM F 1249 and the test conditions were 23 °C at 50% RH. The sample area was 5 cm2 and the partial pressure of the water vapour was 0.01387 atm at 50% RH. The WVTR was normalized with respect to material thickness and partial pressure to yield the water vapour permeability (WVP). Two measurements were made for each film composition and the mean value is reported.
Results and discussion
The aim was to study the influence of lignin and hemicellulose content in pulp fibers on the behavior in pre-treatment and homogenization as well on the properties of fibrillated nanocellulose. Cellulose nanofibrils, CNFs, were first produced from dissolving pulp, which is highly pure with respect to cellulose (Turbak et al. 1983). Nanocellulose from lignin containing pulp has been referred to in the literature as lignocellulose nanofibrils, LCNFs. In the present study, fibrillated nanocellulose has been produced from pure cellulose (dissolving pulp) and pulps containing lignin and hemicellulose. The fibrillated nanocellulose will be referred to as CNFs, irrespective of chemical composition, since cellulose was the main component in all pulps.
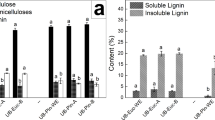
Pulps with different chemical composition were prepared, see Table 3. Unbleached pulps, consequently containing lignin, were compared with bleached pulps to see the effect of lignin. Pulps with higher hemicellulose content were prepared by kraft cooking with polysulfide and compared with conventional kraft pulps to see the effect of hemicellulose content. Additionally, an unbleached pulp was produced by kraft cooking to kappa number 40 and further delignified by oxygen to similar lignin content as the other two unbleached pulps to obtain a pulp with higher amount of charged groups.
As seen in Table 3, addition of polysulfide to kraft cooking resulted in increased glucomannan content in the pulp. The oxidation by polysulfide slightly increased the amount of charged groups, 96 µekv/g, compared to 81 µekv/g in the unbleached kraft pulp. An even higher amount of charges, 120 µekv/g, was obtained by terminating the kraft cooking at a higher kappa number and continuing the delignification by oxygen, thus oxidizing mainly the lignin (Unbleached high charge). Bleaching of chemical pulp removes the lignin, but the higher glucomannan content remained in the bleached polysulfide pulp, which also had slightly higher amount of charged groups. The unbleached pulps had a pulp viscosity of approx. 1250 ml/g while the viscosity level of the bleached pulps was approx. 150 units lower.
Pre-treatment
Prior to preparation of CNFs by homogenization, the pulp fibers need to be pre-treated, as the energy needed to fibrillate untreated pulp fibers is excessive and yields inhomogeneous CNFs (Pääkkö et al. 2007). The pre-treatments used were enzymatic pre-treatment and carboxymethylation.
Figure 2 shows the visual appearance of the CNF gels. From visual observation, the CNF gels prepared from carboxymethylation were rather transparent, whereas the transmittance of the aqueous dispersions prepared from enzymatic pre-treatment were translucent. Hence, pre-treatment process is the key factor influencing the degree of nanofiber conversion of pulp and the transparency of the final CNF gel.

source a unbleached kraft b unbleached polysulfide c unbleached high charge d bleached kraft e bleached polysulfide and f reference dissolving pulp
Photographs of sets of CNF gels produced from enzymatic pre-treatment (left) and carboxymethylation (right), respectively, followed by high-pressure homogenization. Pulp
Enzymatic pre-treatment
The pulp fibers were refined prior to the enzyme treatment to improve the accessibility for enzymes (Virtanen et al. 2015). Refining results in separation of the lamellae in fiber wall (Page and de Grace 1967; Nanko et al. 1989) and in a more open fiber surface (Nanko et al. 1989; de Assis et al. 2018). The refined fibers were subsequently treated with endoglucanases, which randomly select and cleave glycosidic bonds along the cellulose polymer, thereby decreasing the degree of polymerisation. Endoglucanases preferably degrade amorphous cellulose, present on cellulose fibril surfaces and in damaged areas. This weakens the interaction between elemental fibrils and increases the pore volume (Miao et al. 2014; Virtanen et al. 2015). Weak zones in cellulose molecules of lower DP are created, resulting in a more easily fibrillated fiber wall as the pulp is subjected to shearing forces in the homogenizer.
The efficiency of the enzyme treatment can be evaluated by the extent of reduction in the degree of polymerization of cellulose, measured as pulp viscosity. The pulps were subjected to enzymatic pre-treatment with endoglucanase at two dosage levels. In Fig. 3, the effect on pulp viscosity is shown. It is evident that a higher glucomannan content resulted in a less effective enzyme hydrolysis as the decrease in pulp viscosity was lower for the polysulfide pulps compared to the kraft pulps. Lignin on the other hand seems not to hinder enzymes to reach the cellulose chains, the decrease in degree of polymerization was quite similar for unbleached and corresponding bleached pulp. Of the chemical components in plant cell walls, glucomannan is most closely connected with cellulose (Åkerholm and Salmén 2001). A higher retention of glucomannan consequently means that more of the cellulose surface is covered by glucomannan and thus prevents enzymes from accessing the cellulose. Additionally, removal of glucomannan leads to enlargement of pores in the fiber wall (Suurnäkki et al. 1997), which improves enzyme accessibility. Consequently, a fiber wall with higher hemcellulose content will have smaller pores. Removal of lignin from the fiber wall will also create voids in the fiber wall. However, the kappa number (lignin content) of the unbleached pulps in the present study corresponds to a delignification degree of 95%. It can be assumed that at this high degree of lignin removal, only a minor part of the cellulose fibrils are covered by lignin. Previous studies have also shown that increased hemicellulose content is more detrimental to cellulose accessibility than increased lignin content (Mooney et al. 1998; Mussatto et al. 2008).
The best response to enzyme treatment was achieved by the unbleached pulp with high amount of charged groups. The charge groups are deprotonated at the pH prevailing during enzyme treatment, which means that the fiber wall is more swollen and thereby more accessible to the enzymes.
Pre-treatment by carboxymethylation
By substituting hydroxyl groups in cellulose with carboxylmethyl groups, the electrostatic repulsion forces between fibrils are increased, which improves swelling of the fiber wall and decreases the energy needed to fibrillate the pulp (Im et al. 2018; Wågberg et al. 1987). In Table 4, the amount of charged groups after carboxymethylation are shown. The level was similar to what was achieved for the dissolving pulp, consisting of 95% cellulose. The pulps could thus be carboxymethylated to obtain same amount of charges despite higher content of lignin and glucomannan. The sugar monomers in hemicellulose also have hydroxyl groups which can be substituted by carboxylmethyl groups (Peng et al. 2011; Bach et al. 1985; Xu et al. 2011). It has previously been shown that lignin does not prevent carboxymethylation of the carbohydrates, even at as high lignin content as in mechanical pulp (Bach et al. 1981). Lignin actually contains phenolic and aliphatic hydroxyl groups which can be carboxymethylated (Lange and Schweers 1980; Gan et al. 2013). DP values of the carboxymethylated pulps slightly decreased from that of the original pulp, regardless of the carboxylate contents. This observation was in contradiction to what has been observed for TEMPO/NaBr/NaClO oxidation of native celluloses in water at pH 10 (Isogai et al. 2011; Shinoda et al. 2012) where a large decrease in DP was associated with the oxidation procedure (from 1200 to 600). However, DP values of higher than 1000 were obtained from the TEMPO-oxidized celluloses at pH 6.8 (Isogai et al. 2011).
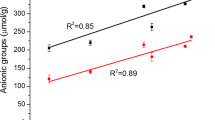
However, the chemical composition of the pulp appeared to affect the carboxymethylation. The total amount of charges in the dissolving pulp, containing practically pure cellulose, increased by almost 1600%, while the increase was only 500–700% for the other pulps. The total amount of charged groups seemed to correlate with the cellulose content in pulp, Fig. 4, except for the unbleached pulp with an originally higher charge content. Carboxymethylation can cause degradation of carbohydrates in the etherification reaction and by alkali, resulting in lower molecular weight (Peng et al. 2011; Im et al. 2018; Xu et al. 2011; Silva et al. 2004). As seen in Table 4, the pulp viscosity generally decreased with carboxymethylation except in the case of the polysulphide pulps where a slight increase was seen. The dissolution of low molecular weight compounds, i.e. hemicelluloses, from the pulps during carboxymethylation could explain this. In Table 5, the chemical composition in pulp before and after either carboxymethylation or enzymatic pre-treatment is presented for the unbleached pulp with higher amount of charged groups and the bleached polysulfide pulp. As seen, the relative amount of the hemicelluloses (xylan and glucomannan) decreased, indicating a loss of these substances. The decrease was most pronounced for the carboxymethylated pulps. Carboxymethylation lead to dissolution in particular of glucomannan while enzymes mainly dissolved xylan. Carboxymethylation of pulps containing substantial amounts of hemicellulose thus seems to be uneconomical, as the input of chemicals is wasted.
Interestingly, carboxymethylation of the unbleached pulp with a higher original pulp charge resulted in the highest amount of charged groups. During oxygen delignification, new aliphatic hydroxyl groups are created (Yang et al. 2007), thus providing more sites for carboxymethylation. Oxidation of unbleached pulps by oxygen prior to carboxymethylation might be an attractive choice as less chemicals would be needed to obtain a certain degree of substitution.
Homogenization
To evaluate the fibrillation of pulps with different lignin and hemicellulose content, the pre-treated pulps were passed once through the homogenizer. It was not possible to fibrillate the pulps pre-treated with the low enzyme dosage as the fibers clogged the entrance of the homogenizer. It is evident that the cohesive forces holding together the fiber wall in paper-grade pulps are much stronger than in the dissolving grade pulp. The cellulose polymers in the dissolving pulp were more extensively degraded already prior to pre-treatment, having a degree of polymerization only about half of the paper-grade pulps, Fig. 3. A more severe enzymatic pre-treatment was needed prior to homogenization, including both higher enzyme dosage and harsher mechanical treatment. Clogging may be caused by entanglement of long fibers and strong cohesive forces of the fiber wall, resisting the impact of shearing. However, fiber length per se seems not be an obstacle, as seen in Fig. 5a. The carboxymethylated fibers were significantly longer than the enzymatically pre-treated fibers and only 10% or less of the fibers were 0.5 mm or below. The reference dissolving pulp fibers were much shorter than the pulps containing lignin and hemicellulose, despite the fact that the raw material for the dissolving pulp also was softwood.
In Table 6, the nano-fraction after homogenization and the turbidity of the CNF suspension is shown. A higher turbidity implies that higher amount of coarser material is present in the suspension. The carboxymethylated pulps were easier to fibrillate as homogenization resulted in a higher nano-fraction and significantly lower turbidity compared to enzymatically pre-treated pulps. For enzymatically pre-treated pulps, the accessibility of the fiber wall for enzymes was important for the homogenization efficiency. Highest nano-fraction and lowest turbidity was obtained for the unbleached pulp with higher amount of charged groups. The lowest nano-fraction was obtained from the other two unbleached pulps, which also had significantly higher turbidity. Homogenization of the carboxymethylated polysulfide kraft pulps, with higher hemicellulose content, resulted in the highest nano-fraction.
The shear thinning behavior of the CNF suspensions with increasing shear rate was rather similar for all pulps, Fig. 6. Shear thinning is caused by the breakage of entanglements between cellulose particles and their alignment when shear rate increases. It was expected that the carboxymethylated CNFs would have higher shear viscosity than enzymatically pre-treated CNFs, due the higher surface charge and degree of fibrillation, i.e., nanofraction (Table 6). This was also the case for the reference dissolving pulp, which displayed a significantly higher viscosity for the carboxymethylated pulp. However, for the paper-grade pulps, the viscosity is practically same for enzymatically pre-treated and carboxymethylated CNFs at lower shear rates. At higher shear rates, the carboxymethylated CNFs were on a higher viscosity level compared to the enzymatically pre-treated CNFs. According to Schenker et al. (2019), the presence of coarser fibers hinders entanglement of fibrils. The low content of nanomaterial and consequently higher content of coarse fibers for both pre-treatment processes probably hinder the agglomeration of fibrillated material at low shear rates.
As seen in Fig. 7, the transmittance differed between the suspensions. Enzymatically treated pulps resulted in opaque CNF suspensions while CNF suspensions from carboxymethylated pulps showed higher transmittance than the carboxymethylated reference dissolving pulp. The highest transmittance and lowest turbidity was observed in CNF suspensions from unbleached polysulfide pulp. Transmittance correlated with turbidity at visible wave lengths. The reference dissolving pulp obtained the highest turbidity and the lowest transmittance. The UV–vis transmittance spectrum in Fig. 7 shows that the transmittance depends on the wavelength. According to previous studies, turbidity is related to the width of (Carr Jr et al. 1977; Carr Jr. and Hermans 1978).
Films
CNF films were prepared and the strength was evaluated. Generally, carboxymethylated pulps resulted in CNF films with higher tensile strength index, Table 7. Siró and co-workers (Siró et al. 2011) demonstrated that the more times the MFC gel passes through the high-pressure homogenizer, the greater the strength of the resulting films. That is, increased tensile properties with increased degree of fibrillation. In the present study, the films are prepared from one-pass homogenized CNFs, so it is not perceived that this plays a major role for the mechanical properties of the films. The strength of the films under strain is very sensitive to the DP value of the MFC, a higher DP being associated with higher strength properties. In the present study, the strength properties of the films are very well correlated with the DP of the pulp after pre-treatment, Fig. 8, which is in agreement with earlier published data (Naderi et al. 2017; Henriksson et al. 2008). It has been proposed that the highest theoretical tensile strength index value is 170 kNm/kg (Lindström 2016). The presence of lignin and hemicellulose did not have a harmful effect on the tensile strength of films made from CNFs from carboxymethylated pulps as the film strength was very close to the maximum value. Our results highlight the importance of pulp DP, optimized pulping and bleaching sequences, pre-treatment and homogenization conditions to suppress fibre aggregation and to allow thorough fibrillation of cellulose fibrils.
Table 8 shows the barriers properties for CNF films, which were quite similar for all films. A slight improvement was observed for bleached CNFs compared to lignin containing CNFs and a lower hemicellulose content resulted also in improved barrier properties. The values are on the same level as obtained by Rojo et al. (2015).
It would be expected that the higher nanofraction of carboxymethylated CNFs should form films with lower OTR than films from enzymatically pre-treated CNFs. Nevertheless, the nature of CNFs, including fibril size distribution and fibril charge density play a major role for the final barrier performance of the CNFs. Carboxymethylated CNFs exhibit a charge density almost an order of magnitude higher than the enzymatic pre-treated CNFs. This highly affects the hygroscopic behaviour and moisture sorption of the fibrils and hence the barrier properties of the films. The presence of moisture softens polymer chains and increases interfibrillar free volume, which can result in local deformation of the film and allow oxygen molecules to permeate through It has previously been shown that OTR increases significantly with charge density (Aulin et al. 2010; Wang et al. 2018).
The water vapor permeability (WVP) of the CNF films was higher for the carboxymethylated films compared with the enzymatic CNF films. CNFs from unbleached pulp with higher charges had the highest water vapor permeability and transmission rate.
It is well-known that the properties of polysaccharides such as cellulose are affected by water. The hydrophilicity depends on the fibre type and preparation and can be considerably influenced by surface modifications such as carboxymethylation. To examine the effect of the surrounding humidity on the moisture content of CNF films prepared from different pulp sources, water vapor sorption isotherm was measured as shown in Fig. 9. Higher moisture uptake was observed for the carboxymethylated CNF films compared with the enzymatic pre-treated CNF films. The driving force for moisture sorption increases with increased nanofibril charge. The shape of the curve for CNFs is characteristic of systems with strong polymer–polymer and polymer–solvent interactions (Kohler et al. 2006). The CNFs displayed an accelerating water uptake above 80% RH, due to partial replacement of CNF-CNF hydrogen bonds with CNF-water hydrogen bonds. The shape of the curve matches previously reported water vapor sorption isotherms of CNFs and Cellulose I (Guo et al. 2017; Kohler et al. 2006). As expected, typical sigmoidal isotherms were generated in both the adsorption and desorption processes. High sorption hysteresis was clearly observed in the high RH region. It has previously been considered that the hysteresis phenomenon of cellulosic materials results from the response delay caused by the collapse of nanopores in the interfibrillar matrix as the internal water molecules exit as well as the delay of structural deformation during the adsorption process (Guo et al. 2017).
Conclusions
Enzymatic pre-treatment of paper grade pulps needed a higher enzyme dosage and extensive PFI refining compared to the reference dissolving pulp to avoid clogging in the homogenizer. Carboxymethylation on the other hand was as efficient for paper-grade pulps as for dissolving pulp.
Effect of lignin
The enzymatic pre-treatment was as efficient for pulps containing lignin as for bleached pulps. The enzymatic hydrolysis was improved when a lignin containing pulp with a higher amount of charged groups was subjected to enzyme pre-treatment. The presence of charged groups increases the swelling of the fiber wall and thereby the accessibility for enzymes.
Carboxymethylation introduced charges also to lignin. The highest total amount of charged groups was introduced to the unbleached pulp subjected to a mild oxygen delignification stage. The oxygen treatment had most likely increased the number of sites in lignin available for carboxymethylation.
The lignin content had no significant effect on the homogenization or the properties of the CNF suspension, which were quite similar to the CNF suspension of the dissolving pulp. The tensile properties of films from lignin containing CNFs were similar to films from CNFs without lignin. Lignin decreased the barrier properties slightly.
Effect of hemicellulose
A higher content of hemicelluloses, mainly glucomannan, reduced the efficiency of the enzymatic pre-treatment as the cellulose accessibility was impaired. Charges were introduced to hemicelluloses by carboxymethylation. However, some hemicelluloses were dissolved by carboxymethylation pre-treatment, as the increased amount of charged groups made them soluble. The beneficial effect of increased hemicellulose content in pulp is thereby counteracted by an increased dissolution of hemicellulose in the pre-treatment. No significant effect of hemicelluloses on the homogenization or the properties of CNF suspensions was observed. The tensile properties of films were on same level regardless of hemicellulose content.
References
Åkerholm M, Salmén L (2001) Interactions between wood polymers studied by dynamic FT-IR spectroscopy. Polymer 42:963–969. https://doi.org/10.1016/S0032-3861(00)00434-1
Aulin C, Gällstedt M, Lindström T (2010) Oxygen and oil barrier properties of microfibrillated cellulose films and coatings. Cellulose 17:559–574. https://doi.org/10.1007/s10570-009-9393-y
Bach Tuyet L, Iiyama K, Nakano J (1985) Preparation of carboxymethylcellulose from refiner mechanical pulp. IV Analyses of carboxymethylated polysaccharides by the uses of 1H-NMR. Mokuzai Gakaishi 31:14–19. https://doi.org/10.2524/jtappij.35.798
Bach Tuyet L, Ishizu A, Nakano J (1981) Preparation of carboxymethylcellulose from refiner mechanical pulp. Jpn Tappi 35:798–804. https://doi.org/10.2524/jtappij.35.798
Carr M Jr, Shen L, Hermans J (1977) Mass-length ratio of fibrin fibers from gel permeation and light scattering. Biopolymers 16:1–5. https://doi.org/10.1002/bip.1977.360160102
Carr M Jr, Hermans J (1978) Size and density of fibrin fibers from turbidity. Macromolecules 11:46–50. https://doi.org/10.1021/ma60061a009
Chaker A, Alila S, Mutjé P, Rei Vilar M, Boufi S (2013) Key role of the hemicellulose content and the cell morphology on the nanofibrillation effectiveness of cellulose pulps. Cellulose 20:2863–2875. https://doi.org/10.1007/s10570-013-0036-y
de Assis T, Huang S, Driemeier C, Donohoe B, Kim C, Kim S, Park S (2018) Toward an understanding of the increase in enzymatic hydrolysis by mechanical refining. Biotechnol Biofuels 11:289. https://doi.org/10.1186/s13068-018-1289-3
Fall A, Burman A, Wågberg L (2014) Cellulosic nanofibrils from eucalyptus, acacia and pine fibers. Nordic Pulp Paper Res J 29:176–184. https://doi.org/10.3183/npprj-2014-29-01-p176-184
Ferrer A, Quintana E, Filpponen I, Solala I, Vidal T, Rodríguez A, Rojas O (2012) Effect of residual lignin and heteropolysaccharides in nanofibrillar cellulose and nanopaper from wood fibers. Cellulose 19:2179–2193. https://doi.org/10.1007/s10570-012-9788-z
Gan L, Zhou M, Yang D, Qiu X (2013) Preparation and evaluation of carboxymethylated lignin as dispersant for aqueous graphite suspension using turbiscan lab analyzer. J Dispersion Sci Technol 34:644–650. https://doi.org/10.1080/01932691.2012.686248
Guo X, Wu Y, Xie X (2017) Water vapor sorption properties of cellulose nanocrystals and nanofibers using dynamic vapor sorption apparatus. SciRep-UK 7:14207. https://doi.org/10.1038/s41598-017-14664-7
Henriksson M, Berglund L, Isaksson P, Lindström T, Nishino T (2008) Cellulose nanopaper structures of high toughness. Biomacromol 9:1579–1585. https://doi.org/10.1021/bm800038n
Im W, Lee S, Rajabi Abhari A, Youn H, Lee H (2018) Optimization of carboxymethylation reaction as a pretreatment for production of cellulose nanofibrils. Cellulose 25:3873–3883. https://doi.org/10.1007/s10570-018-1853-9
Isogai A, Saito T, Fukuzumi H (2011) TEMPO-oxidized cellulose nanofibers. Nanoscale 3:71–85. https://doi.org/10.1039/C0NR00583E
Jiang Y, Liu X, Yang Q, Song X, Qin C, Wang S, Li K (2018) Effects of residual lignin on mechanical defibrillation process of cellulosic fiber for producing lignocellulose nanofibrils. Cellulose 11:6479–6494. https://doi.org/10.1007/s10570-018-2042-6
Kohler R, Alex R, Brielmann R, Ausperger B (2006) A new kinetic model for water sorption isotherms of cellulosic materials. Macromol Sy 244:89–96. https://doi.org/10.1002/masy.200651208
Laitinen P, Liukkonen S, Pere J, Sneck A, Kangas H (2014) A comparative study of fibrillated fibers from different mechanical and chemical pulps. BioResources 9:2115–2127
Lange W, Schweers W (1980) The carboxymethylation of organosolv and kraft lignins. Wood Sci Technol 14:1–7. https://doi.org/10.1007/BF00353458
Lindström T (2016) From microfibrillar cellulose to nanocellulose applications—an account of the evolutionary developments. In: 9th Int Paper Coating Chem Symp/Int Paper Physics Conf Tokyo, Japan.
Marx-Figini M (1978) Significance of the intrinsic viscosity ratio of unsubstituted and nitrated cellulose in different solvents. Angew Makromol Chem 72:161–171. https://doi.org/10.1002/apmc.1978.050720114
Miao Q, Chen L, Huang L, Tian C, Zheng L, Ni Y (2014) A process for enhancing the accessibility and reactivity of hardwood kraft-based dissolving pulp for viscose rayon production by cellulase treatment. Bioresource Technol 154:109–113. https://doi.org/10.1016/j.biortech.2013.12.040
Mooney C, Mansfield S, Touhy M, Saddler J (1998) The effect of initial pore volume and lignin content on the enzymatic hydrolysis of softwoods. Bioresource Technol 64:113–119
Mussatto S, Fernandes M, Milagres A, Roberto I (2008) Effect of hemicellulose and lignin on enzymatic hydrolysis of cellulose from brewer’s spent grain. Enzyme Microb Tech 43:124–129. https://doi.org/10.1016/j.enzmictec.2007.11.006
Naderi A (2017) Nanofibrillated cellulose: properties reinvestigated. Cellulose 24:1933–1945. https://doi.org/10.1007/s10570-017-1258-1
Naderi A, Larsson T, Stevanic J, Lindström T, Erlandsson J (2017) Effect of the size of the charged group on the properties of alkoxylated NFCs. Cellulose 24:1307–1317. https://doi.org/10.1007/s10570-017-1190-4
Nanko H, Ohsawa J, Okagawa A (1989) How to see interfibre bonding in paper sheets. J Pulp Paper Sci 15:17–23
Oliaei E, Lindén P, Wu Q, Berthold F, Berglund L, Lindström T (2020) Microfibrillated lignocellulose (MFLC) and nanopaper films from unbleached kraft softwood pulp. Cellulose 27:2325–2341. https://doi.org/10.1007/s10570-019-02934-8
Osong S, Norgren S, Engstrand P (2013) An approach to produce nano-ligno-cellulose from mechanical pulp fine materials. Nordic Pulp Paper Res J 28:472–479. https://doi.org/10.3183/npprj-2013-28-04-p472-479
Page D, de Grace J (1967) The delamination of fiber walls by beating and refining. Tappi 50:489–495
Peng X, Ren J, Zhong L, Cao X, Sun R (2011) Microwave-induced synthesis of carboxylmethyl hemicelluloses and their rheological properties. J Agric Food Chem 59:570–576. https://doi.org/10.1021/jf1036239
Pääkkö M, Ankerfors M, Kosonen H, Nykänen A, Ahola S, Österberg M, Lindström T (2007) Enzymatic hydrolysis combined with mechanical shearing and high-pressure homogenization for nanoscale cellulose fibrils and strong gels. Biomacromol 8:1934–1941. https://doi.org/10.1021/bm061215p
Rojo E, Soledad Peresin M, Sampson W, Hoeger I, Vartiainen J, Laine J, Rojas O (2015) Comprehensive elucidation of the effect of residual lignin on the physical, barrier, mechanical and surface properties of nanocellulose films. Green Chem 17:1853–1866. https://doi.org/10.1039/C4GC02398F
Schenker M, Schoelkopf J, Gane P, Mangin P (2019) Rheology of microfibrillated cellulose (MFC) suspensions: influence of the degree of fibrillation and residual fibre content on flow and viscoelastic properties. Cellulose 26:845–860. https://doi.org/10.1007/s10570-018-2117-4
Shinoda R, Saito T, Okita Y, Isogai A (2012) Relationship between length and degree of polymerization of TEMPO-oxidized cellulose nanofibrils. Biomacromol 13:842–849. https://doi.org/10.1021/bm2017542
Silva D, de Paula R, Feitosa J, de Brito A, Maciel J, Paula H (2004) Carboxymethylation of cashew tree exudate polysaccharide. Carbohyd Polym 58:163–171. https://doi.org/10.1016/j.carbpol.2004.06.034
Siró I, Plackett D, Hedenqvist M, Ankerfors M, Lindström T (2011) Highly transparent films from carboxymethylated microfibrillated cellulose: the effect of multiple homogenization steps on key properties. J Appl Polym Sci 119:2652–2660. https://doi.org/10.1002/app.32831
Solala I, Volperts A, Andersone A, Dizhbite T, Mironova-Ulmane N, Vehniäinen A, Vuorinen T (2012) Mechanoradical formation and its effects on birch kraft pulp during preparation of nanofibrillated cellulose with Matsuko refining. Holzforschung 66:477–483. https://doi.org/10.1515/hf.2011.183
Suurnäkki A, Li T, Buchert J, Tenkanen M, Viikari L, Vuorinen T, Ödberg L (1997) Effects of enzymatic removal of xylan and glucomannan on the pore size distribution of kraft fibres. Holzforschung 51:27–33. https://doi.org/10.1515/hfsg.1997.51.1.27
Turbak A, Snyder F, Sandberg K (1983) Microfibrillated cellulose, a new cellulose product: properties, uses, and commercial potential. J Appl Polym Sci Appl Polym Symp 37:815–827
Virtanen T, Penttilä P, Maloney T, Grönqvist S, Kamppuri T, Vehviläinen M, Maunu S (2015) Impact of mechanical and enzymatic pretreatments on softwood pulp fiber wall structure studied with NMR spectroscopy and X-ray scattering. Cellulose 22:1565–1576. https://doi.org/10.1007/s10570-015-0619-x
Wågberg L, Decher G, Norgren M, Lindström T, Ankerfors M, Axnäs K (2008) The build-up of polyelectrolyte multilayers of microfibrillated cellulose and cationic polyelectrolytes. Langmuir 24:784–795. https://doi.org/10.1021/la702481v
Wågberg L, Winter L, Ödberg L, Lindström T (1987) On the charge stoichiometry upon adsorption of a cationic polyelectrolyte on cellulosic materials. Colloids Surfaces 27:163–173. https://doi.org/10.1016/0166-6622(87)80140-3
Wang J, Gardner D, Stark N, Bousfield D, Tajvidi M, Cai Z (2018) Moisture and oxygen barrier properties of cellulose nanomaterial-based films. ACS Sustain Chem Eng 6:49–70. https://doi.org/10.1021/acssuschemeng.7b03523
Xu C, Eckerman C, Smeds A, Reunanen M, Eklund P, Sjöholm R, Willför S (2011) Carboxymethylated spruce galactoglucomannans: preparation, characterisation, dispersion stability, water-in-oil emulsion stability, and sorption on cellulose. Nordic Pulp Paper Res J 26:1–12. https://doi.org/10.3183/npprj-2011-26-02-p167-178
Yang R, Lucia L, Ragauskas A, Jameel H (2007) Oxygen delignification chemistry and its impact on pulp fibers. J Wood Chem Technol 23:13–29. https://doi.org/10.1081/WCT-120018613
Acknowledgments
The authors would like to acknowledge the partners of RISE Bioeconomy research program on nanocellulose; Stora Enso, Holmen, Suzano, Södra, Performance Biofilaments, Fortum and BillerudKorsnäs, for financial contribution.
Funding
Open access funding provided by RISE Research Institutes of Sweden. Industry consortium “Nanocellulose” within the RISE Innventia Research Program 2018–2020. This project has been co-financed with funds for strategic competence development to RISE from the Swedish state.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Human and animals rights
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Brännvall, E., Aulin, C. CNFs from softwood pulp fibers containing hemicellulose and lignin. Cellulose 29, 4961–4976 (2022). https://doi.org/10.1007/s10570-022-04585-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10570-022-04585-8