Abstract
Limited data are available to guide the decision-making process for clinicians and their patients regarding palliative treatment options for patients with isolated synchronous colorectal cancer peritoneal metastases (CRC-PM). Therefore, the aim of this study is to analyze the outcome of the different palliative treatments for these patients. All patients diagnosed with isolated synchronous CRC-PM between 2009 and 2020 (Netherlands Cancer Registry) who underwent palliative treatment were included. Patients who underwent emergency surgery or curative intent treatment were excluded. Patients were categorized into upfront palliative primary tumor resection (with or without additional systemic treatment) or palliative systemic treatment only. Overall survival (OS) was compared between both groups and multivariable cox regression analysis was performed. Of 1031 included patients, 364 (35%) patients underwent primary tumor resection and 667 (65%) patients received systemic treatment only. Sixty-day mortality was 9% in the primary tumor resection group and 5% in the systemic treatment group (P = 0.007). OS was 13.8 months in the primary tumor resection group and 10.3 months in the systemic treatment group (P < 0.001). Multivariable analysis showed that primary tumor resection was associated with improved OS (HR 0.68; 95%CI 0.57–0.81; P < 0.001). Palliative primary tumor resection appeared to be associated with improved survival compared to palliative systemic treatment alone in patients with isolated synchronous CRC-PM despite a higher 60-day mortality. This finding must be interpreted with care as residual bias probably played a significant role. Nevertheless, this option may be considered in the decision-making process by clinicians and their patients.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Colorectal cancer (CRC) is one of the most common cancers worldwide, with a yearly incidence of almost two million cases [1]. Frequently, CRC has already metastasized at the time of diagnosis, with the peritoneum as the second most affected organ being present in approximately 23% of patients with metastatic CRC [2]. In one third of these patients these metastases are confined to the peritoneum [3, 4].
Treatment of CRC patients presenting with peritoneal metastases (PM) is challenging and depends on various factors including the condition of the patient, the presence of systemic metastases, symptoms of the primary tumor and extend of the peritoneal disease [5, 6]. A selected group of fit patients with limited peritoneal disease may undergo curative intent treatment such as cytoreductive surgery with or without hyperthermic intraperitoneal chemotherapy (CRS-HIPEC) [7]. Patients with a symptomatic primary tumor (e.g., obstruction or perforation) are usually treated with surgery in an emergency setting [8].
For fit patients that do not require emergency surgery and in whom curative intent treatment is not possible due to extensive disease, two palliative treatment options may be considered: resection of the primary tumor (with or without additional systemic treatment) or palliative systemic treatment only. Whether to resect an asymptomatic primary colorectal tumor in patients presenting with unresectable systemic metastases has been a highly debated issue for many years with various retrospective studies and recently published randomized trials reporting conflicting results [9,10,11,12,13,14,15,16,17,18,19,20,21].
However, it should be noted that both in these prospective trials and retrospective studies the vast majority of included patients suffered from liver metastases and/or lung metastases [9,10,11,12,13,14,15,16,17,18,19,20,21]. Patients with PM were either absent or represented only a very small proportion of the study population. Thus, these studies give no guidance regarding the treatment of patients with CRC and isolated PM. This is relevant as patients with colorectal cancer peritoneal metastases (CRC-PM) are known to have a different clinical outcome as compared to CRC patients with liver metastases or lung metastases with a markedly shorter survival [22]. This may be due to the observation that PM seem to respond less to systemic treatment as compared to other systemic metastases [23,24,25]. Therefore, the aim of the current study was to analyze the outcome of palliative primary tumor resection (with or without additional systemic treatment) and palliative systemic treatment only specifically in CRC patients with isolated synchronous PM who did not undergo emergency surgery or curative intent treatment.
Materials and methods
Data source
Data were extracted from the Netherlands Cancer Registry (NCR). The NCR registers all newly diagnosed malignancies in the Netherlands. Specially trained data managers of the NCR extract data on patient, tumor and treatment characteristics from the medical records. A yearly update of the vital status of patients is performed by linking the registry to the Dutch municipal administrative database, which contains information about all present, deceased and former inhabitants of the Netherlands. For the present study, the latest update was performed on January 31st, 2022. The International Classification of Diseases for Oncology (ICD-O) was used for the specification of the primary tumor location, location of synchronous metastases and for histological subtypes. The tumor node metastasis (TNM) classification was used for stage classification of the primary tumor, according to the edition valid at diagnosis. If pathological T or N stage was unknown, clinical T or N stage was used. The study is approved by the privacy review board of the NCR as well as the combined scientific committee of the NCR and Prospective Dutch ColoRectal Cancer Cohort (PLCRC) study of the Dutch Colorectal Cancer Group (DCCG).
Study population
All CRC patients with synchronous metastases diagnosed between 2009 and 2020 were evaluated. In patients with multiple primary tumors, the tumor which was first diagnosed was included or, if simultaneously diagnosed, the tumor with the highest TNM stage was included. Patients with extraperitoneal metastases were excluded. Patients were also excluded if they had a primary tumor in the appendix or a neuroendocrine primary tumor. In addition, patients were excluded if they had undergone curative intent treatment such as CRS-HIPEC, debulking surgery or metastasectomy, if they had only received best supportive care, if the primary tumor was resected after initial systemic treatment or neo-adjuvant chemoradiotherapy or if the treatment was unknown. The NCR records whether the primary tumor resection was performed in an elective setting or in an emergency setting. Patients who underwent an emergency resection were excluded. If no data regarding the clinical indication for surgery was registered, patients who had undergone surgery within 5 days after their initial diagnosis were considered to be emergency resection rather than primary tumor resection and were excluded.
Treatment allocation
For all analyses, treatment strategies were categorized as follows:
-
1.
Upfront palliative primary tumor resection with or without additional systemic treatment, comprising different types of resections (i.e., hemicolectomy, ileocecal resection, transverse colon resection, sigmoid resection, (sub)total colectomy, low anterior resection and rectum amputation).
-
2.
Palliative systemic treatment only.
Primary outcome
The primary outcome was overall survival (OS), compared between patients in the palliative primary tumor resection group and patients in the palliative systemic treatment group. Median OS was defined as the interval between date of diagnosis of CRC until date of death or loss to follow-up. Patients were censored if they were alive on January 31st, 2022.
Patient- and tumor characteristics
The location of the primary tumor was categorized according to the following sites: (1) right-sided colon (C18.0, C18.2-18.4: cecum, ascending colon, hepatic flexure, transverse colon); (2) left-sided colon (C18.5-18.7: splenic flexure, descending colon and sigmoid); and (3) rectum (C19.9-20.9: rectosigmoid and rectum). Primary tumor histology was categorized into the following subtypes: (1) adenocarcinoma (8000, 8010, 8020, 8140, 8144, 8210, 8211, 8220 8255, 8261, 8262, 8263, 8560); (2) mucinous adenocarcinoma (8480, 8481); and (3) signet ring cell carcinoma (8490). The following ICD-O codes were considered PM: C16.0-C16.9, C17.0-C17.9, C18.0-C18.9, C19.9, C20.9, C21.8, C23.9, C26.9, C48.0-C48.8, C49.4-C49.5, C52.9, C54.3-C54.9, C55.9, C56.9, C57.0-C57.8, C66.9, C67.0-C67.9, C76.2. Any other ICD-O code was considered to reflect extraperitoneal metastases. Patient- and tumor characteristics included in this study are sex, age, primary tumor location, tumor histology, differentiation of primary tumor, T stage, N stage and period of diagnosis.
Statistical analysis
Baseline characteristics of patients in the primary tumor resection group were compared to patients in the palliative systemic treatment group. Categorical variables were compared using χ2-test and presented as a No. (%), and continuous variables were compared with the unpaired t-tests and presented as mean (standard deviation [SD]). Missing data were excluded from comparative analyses. Sixty-day mortality was compared between patients in the palliative primary tumor resection group and the palliative systemic treatment group by using the χ2-test. Median OS of patients in the palliative primary tumor resection group and patients in the palliative systemic treatment group was estimated with the Kaplan Meier method and compared with the log-rank test.
Univariable cox regression analyses were performed to assess the association between palliative primary tumor resection and OS and to identify whether the following factors were associated with OS: sex, age, primary tumor location, tumor histology, tumor differentiation, T stage, N stage, period of diagnosis and the presence of a stoma. Subsequently, variables with a p-value lower than 0.10 in the univariable analyses, were combined in a multivariable cox regression model. To prevent overfitting, a minimum of 10 events per degree of freedom was used as limit for the number of variables of the multivariable model.
Finally, a subgroup analysis was performed in patients who underwent primary tumor resection. This subgroup analysis included uni- and multivariable cox regression analyses to identify factors associated with OS within this subgroup.
All tests were two-sided and a p-value lower than 0.05 was considered statistically significant. All analyses were performed with SAS statistical software (SAS system 9.4, SAS Institute, Cary, NC, United States).
Results
Study population
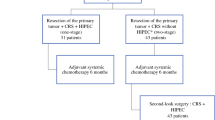
In total, 33.979 patients were diagnosed with metastasized CRC between 2009 and 2020. Of these patients, 8492 (25%) had synchronous PM of whom 3601 (11%) without concurrent extraperitoneal metastases. Of this latter group, 2215 (62%) patients were excluded because they had undergone curative treatment, best supportive care or an unknown treatment modality. An additional 328 patients undergoing primary tumor resection within five days of diagnosis of their primary CRC were excluded and 27 patients were excluded because their primary tumor was resected after initial systemic treatment or neo-adjuvant chemoradiotherapy. The remaining 1031 patients were included in this study, of whom 364 (35%) underwent primary tumor resection. In the palliative systemic treatment group (n = 667/1031, 65%), patients were exclusively treated with palliative systemic treatment. The primary tumor resection group (n = 364) comprised of 220 (60%) patients who underwent primary tumor resection only and 144 (40%) patients who underwent primary tumor resection followed by additional systemic treatment (Fig. 1).
In patients who underwent a primary tumor resection followed by additional systemic treatment (n = 144), 126 patients (88%) received chemotherapy only and 18 patients (12%) received both chemotherapy and targeted therapy. Details regarding the prescribed regimens were registered in 46 patients (32%). In these patients, capecitabine with oxaliplatin (CAPOX) (n = 20) and capecitabine monotherapy (n = 17) were the most used chemotherapeutic regimens.
In the palliative systemic treatment group (n = 667), 549 patients (82%) received chemotherapy only, 5 patients (1%) received targeted therapy only and 113 patients (17%) received both chemotherapy and targeted therapy. Details regarding the prescribed regimens were registered in 345 patients (52%), capecitabine with oxaliplatin (CAPOX) (n = 169), capecitabine monotherapy (n = 89) and 5-fluorouracil/leucovorin with oxaliplatin (FOLFOX) (n = 58) being the most used chemotherapeutic regimens. Panitumumab (n = 19) was the most used targeted therapy in these patients.
Older age, a right-sided tumor, a T4 tumor stage, positive lymph nodes and primary tumor diagnosis between 2009 and 2012 were more frequently present in patients who underwent palliative primary tumor resection than in those who received palliative systemic treatment (Table 1).
Survival
Sixty-day mortality was 9% in the primary tumor resection group and 5% in the palliative systemic treatment group (P = 0.007). Two-year survival was 32% in the primary tumor resection group and 14% in the palliative systemic treatment group (P < 0.001). The median OS was 13.7 (interquartile range [IQR] 6.4–29.4) months in the primary tumor resection group and 10.3 (IQR 5.5–17.0) months in the palliative systemic treatment group (P < 0.001) (Fig. 2). If a primary tumor resection was followed by systemic therapy, median OS was 18.0 months (IQR 8.9–33.4).
Univariable and multivariable analysis showed that primary tumor resection was significantly associated with improved OS (adjusted hazard ratio [aHR], 0.68; 95% CI, 0.57–0.81; P < 0.001). Factors that were associated with worse OS included a signet ring cell carcinoma histology (aHR, 1.38; 95% CI, 1.13–1.68; P = 0.001), a poor differentiated tumor (aHR, 1.49; 95% CI, 1.24–1.78; P < 0.001), a T4 tumor stage (aHR, 1.25; 95% CI, 1.07–1.46; P = 0.005) and nodal involvement (aHR, 1.28; 95% CI, 1.09–1.51; P < 0.001]) (Table 2).
Subgroup analyses in primary tumor resection group
In subgroup analyses of patients who underwent primary tumor resection, multivariable analysis showed that older age (aHR, 1.02; 95% CI, 1.01–1.03; P = 0.002), a signet ring cell carcinoma histology (aHR, 1.58; 95% CI, 1.08–2.31; P = 0.02), a poor differentiated tumor (aHR, 1.68; 95% CI, 1.31–2.15; P < 0.001), a T4 tumor stage (aHR, 1.46; 95% CI, 1.15–1.85; P = 0.002), nodal involvement (aHR, 1.87, 95% CI, 1.33–2.63; P < 0.001 ) and having a stoma (aHR, 0.67, 95%CI, 0.51–0.85; P = 0.002) were associated with worse OS (Table 3).
Discussion
In this nationwide observational cohort study of patients with isolated synchronous CRC-PM, primary tumor resection was associated with an improved OS when compared to palliative systemic treatment only (median 13.7 months vs. 10.3 months). However, primary tumor resection was associated with an increase in sixty-day mortality. Patients undergoing treatment with curative intent, patients undergoing best supportive care only and patients requiring emergency surgery were excluded in this study and therefore, the results from the current study apply to those in whom the choice whether to perform a palliative resection of the primary tumor could be considered in a non-emergency setting.
The role of primary tumor resection in the treatment of patients with unresectable synchronous metastatic CRC with an asymptomatic primary tumor has been a highly debated issue for many years [21, 26,27,28]. Various retrospective studies seem to suggest a survival benefit after primary tumor resection [9, 10, 12,13,14,15]. However, selection bias may be an important explanation for this finding with younger and fitter patients usually tending to undergo surgery instead of systemic treatment. To address this issue in a prospective manner, several randomized trials have been conducted over the recent past. The recently published randomized controlled iPACs trial (JCOG1007) showed that the OS of systemic metastatic CRC patients who underwent primary tumor resection followed by systemic treatment was comparable to that of patients treated with systemic treatment only (26.4 months versus 25.9 months, respectively), which was in line with recently presented results from the SYNCHRONOUS trial [19, 29, 30]. Also, the CAIRO4 trial (NCT01606098) recently published the short-term results and reported a significantly higher mortality after primary tumor resection as compared to systemic treatment only (11% vs. 3% respectively) in the first 60 days after randomization [18]. As such, both trials provide valid arguments for no longer removing the primary tumor in CRC patients with widespread systemic disease [27]. As a result, resection of an asymptomatic primary colorectal tumor in patients with systemic metastases is no longer advised in most clinical guidelines such as the National Comprehensive Cancer Network [31].
Up to 10% of patients with CRC will be diagnosed with PM during the course of their disease [3]. As such, the peritoneum is a very relevant metastatic site in CRC. In spite of this, patients with PM are usually underrepresented in clinical trials as PM are often not visible on radiological imaging that is required for response evaluation to treatment [32, 33]. Also, in both previously mentioned retrospective and prospective trials investigating the effect of primary tumor resection, patients with PM were virtually absent [9,10,11,12,13,14,15, 18,19,20,21].
As recent data suggests that PM almost exclusively derive from a specific molecular subtype of CRC, it is probably not appropriate to translate knowledge that has been obtained in trials, performed in patients with liver metastases and lung metastases, to clinical scenarios in which PM are involved [34]. One reason may be that the subtype that causes PM is known to be less sensitive to systemic treatment [23, 25]. Together with the typical clinical presentation of PM with frequent bowel obstructions resulting in malnourishment, this probably explains that the prognosis of patients with PM is markedly worse as compared to other metastatic sites. Therefore, it can be argued that surgical treatment may indeed be more effective in alleviating clinical symptoms in this specific patient category than treating chemo-resistant metastases with systemic treatment.
The present study reported a higher sixty-day mortality for patients in the primary tumor resection group (9%) than for patients in the palliative systemic treatment group (5%). This finding is in line with a recently published randomized controlled trial on this topic for patients with CRC and systemic metastases [18]. This increase in early mortality confirms that primary tumor resection in patients with systemic disease does not come without substantial risk in the early postoperative period. Regarding OS, older age, a signet ring cell carcinoma histology, a poor tumor differentiation, a T4 tumor stage, nodal involvement and having a stoma were associated with a worse survival within the primary tumor resection group. Early postoperative mortality and risk factors for decreased OS after surgical treatment should be taken into account when discussing treatment options in these patients.
Construction of a stoma was necessary in 21% of patients treated with systemic treatment. No significant difference in the number of stomas was observed as compared to the primary tumor resection group. This is important in the decision-making process as fear for a stoma may deter patients from undergoing primary tumor resection.
In this study, the proportion of patients who received chemotherapy alone or in combination with targeted therapy was comparable between the systemic treatment group and the primary tumor resection group. In both groups, CAPOX was the most frequently used chemotherapy regimen. Therefore, treatment with systemic chemotherapy is not expected to result in a significant difference in survival.
Patients who underwent primary tumor resection in the present study were significantly older, more frequently had a right-sided tumor and nodal involvement as compared to patients that received systemic treatment only. Although multivariable cox regression analyses aimed to correct for these confounders after which primary tumor resection remained associated with an improved OS, residual selection bias probably still plays an important role. Relevant in this respect is the fact that the extent of peritoneal disease was not known. It may be that patients with less extensive peritoneal disease were more prone to undergo a primary tumor resection, which may explain the more favorable outcome in these patients.
To our knowledge, this is the first nationwide population-based study to investigate the role of primary tumor resection in patients with isolated synchronous CRC-PM. The NCR provides highly accurate data on tumor and patients characteristics, strengthening the generalizability of the results [35]. However, the retrospective design is clearly a drawback of the current study as no data on extent of peritoneal disease, tumor biology (e.g., CMS subtype), mutational status, performance status, postoperative complications or toxicity of systemic therapy and clinical symptoms were available. The addition of these factors would have increased the accurateness of the multivariable model.
It is not likely that a randomized controlled trial will address the issue of primary tumor resection in CRC patients with PM in the near future. Therefore, in spite of its retrospective nature, we believe that the current study provides valuable information to guide decision making in current day clinical practice in this distinct and relevant category of metastatic patients.
Conclusions
In this retrospective nationwide cohort of patients with isolated synchronous CRC-PM, primary tumor resection appeared to be associated with an improved OS in comparison to those who received only systemic treatment, despite an increased sixty-day mortality rate after surgery. These findings must be interpreted with care as residual bias is likely to have played a significant role. Nevertheless, this finding may be considered in the decision-making process by clinicians and their patients regarding the different palliative treatment options in this specific patient category.
References
World Health Organisation. Global Cancer Observatory. Accessed April 1 (2022) https://gco.iarc.fr/today/home
Lemmens VE, Klaver YL, Verwaal VJ et al (2011) Predictors and survival of synchronous peritoneal carcinomatosis of colorectal origin: a population-based study. Int J Cancer 128(11):2717–2725. https://doi.org/10.1002/ijc.25596
Lurvink RJ, Bakkers C, Rijken A et al (2021) Increase in the incidence of synchronous and metachronous peritoneal metastases in patients with colorectal cancer: a nationwide study. Eur J Surg Oncol 47(5):1026–1033. https://doi.org/10.1016/j.ejso.2020.11.135
Thomassen I, van Gestel YR, Lemmens VE et al (2013) Incidence, prognosis, and treatment options for patients with synchronous peritoneal carcinomatosis and liver metastases from colorectal origin. Dis Colon Rectum 56(12):1373–1380. https://doi.org/10.1097/DCR.0b013e3182a62d9d
Bakkers C, Lurvink RJ, Rijken A et al (2021) Treatment strategies and prognosis of patients with synchronous or metachronous colorectal peritoneal metastases: a Population-Based study. Ann Surg Oncol 28(13):9073–9083. https://doi.org/10.1245/s10434-021-10190-z
Cortés-Guiral D, Hübner M, Alyami M et al (2021) Primary and metastatic peritoneal surface malignancies. Nat Rev Dis Primers 7(1):91. https://doi.org/10.1038/s41572-021-00326-6
Quénet F, Elias D, Roca L et al (2021) Cytoreductive surgery plus hyperthermic intraperitoneal chemotherapy versus cytoreductive surgery alone for colorectal peritoneal metastases (PRODIGE 7): a multicentre, randomised, open-label, phase 3 trial. Lancet Oncol 22(2):256–266. https://doi.org/10.1016/S1470-2045(20)30599-4
Amelung FJ, Consten ECJ, Siersema PD et al (2016) A Population-Based analysis of three treatment modalities for malignant obstruction of the proximal Colon: Acute Resection Versus Stent or Stoma as a bridge to surgery. Ann Surg Oncol 23(11):3660–3668. https://doi.org/10.1245/s10434-016-5247-7
’t Lam-Boer J, van der Geest LG, Verhoef C et al (2016) Palliative resection of the primary tumor is associated with improved overall survival in incurable stage IV colorectal cancer: a nationwide population-based propensity-score adjusted study in the Netherlands. Int J Cancer 139(9):2082–2094. https://doi.org/10.1002/ijc.30240
van Rooijen KL, Shi Q, Goey KKH et al (2018) Prognostic value of primary tumour resection in synchronous metastatic colorectal cancer: individual patient data analysis of first-line randomised trials from the ARCAD database. Eur J Cancer 91:99–106. https://doi.org/10.1016/j.ejca.2017.12.014
Alawadi Z, Phatak UR, Hu CY et al (2017) Comparative effectiveness of primary tumor resection in patients with stage IV colon cancer. Cancer 123(7):1124–1133. https://doi.org/10.1002/cncr.30230
Faron M, Pignon JP, Malka D et al (2015) Is primary tumour resection associated with survival improvement in patients with colorectal cancer and unresectable synchronous metastases? A pooled analysis of individual data from four randomised trials. Eur J Cancer 51(2):166–176. https://doi.org/10.1016/j.ejca.2014.10.023
Xu H, Xia Z, Jia X et al (2015) Primary tumor resection is Associated with Improved Survival in Stage IV Colorectal Cancer: an Instrumental Variable Analysis. Sci Rep 5:16516. https://doi.org/10.1038/srep16516
Maroney S, Chavez de Paz C, Reeves ME et al (2018) Benefit of Surgical Resection of the primary tumor in patients undergoing chemotherapy for stage IV Colorectal Cancer with Unresected Metastasis. J Gastrointest Surg 22(3):460–466. https://doi.org/10.1007/s11605-017-3617-5
Gulack BC, Nussbaum DP, Keenan JE et al (2016) Surgical Resection of the primary tumor in Stage IV Colorectal Cancer without Metastasectomy is Associated with Improved overall survival compared with Chemotherapy/Radiation Therapy alone. Dis Colon Rectum 59(4):299–305. https://doi.org/10.1097/DCR.0000000000000546
van der Kruijssen DEW, Brouwer NPM, van der Kuil AJS et al (2021) Interaction between primary tumor resection, primary Tumor Location, and Survival in Synchronous Metastatic Colorectal Cancer: a Population-Based study. Am J Clin Oncol 44(7):315–324. https://doi.org/10.1097/COC.0000000000000823
’t Lam - Boer J, Mol L, Verhoef C et al (2014) The CAIRO4 study: the role of surgery of the primary tumour with few or absent symptoms in patients with synchronous unresectable metastases of colorectal cancer–a randomized phase III study of the dutch Colorectal Cancer Group (DCCG). BMC Cancer 14(1):741. https://doi.org/10.1186/1471-2407-14-741
van der Kruijssen DEW, Elias SG, Vink GR et al (2021) Sixty-day mortality of patients with metastatic colorectal Cancer randomized to systemic treatment vs primary Tumor Resection followed by systemic treatment: the CAIRO4 phase 3 Randomized Clinical Trial. JAMA Surg 156(12):1093–1101. https://doi.org/10.1001/jamasurg.2021.4992
Kanemitsu Y, Shitara K, Mizusawa J et al (2021) Primary Tumor Resection Plus Chemotherapy Versus Chemotherapy alone for colorectal Cancer patients with asymptomatic, synchronous unresectable metastases (JCOG1007; iPACS): a Randomized Clinical Trial. J Clin Oncol 39(10):1098–1107. https://doi.org/10.1200/JCO.20.02447
Park EJ, Baek JH, Choi GS et al (2020) The role of primary Tumor Resection in Colorectal Cancer patients with asymptomatic, synchronous, unresectable metastasis: a Multicenter Randomized Controlled Trial. Cancers (Basel) 12(8):1–14. https://doi.org/10.3390/cancers12082306
Venderbosch S, de Wilt JH, Teerenstra S et al (2011) Prognostic value of resection of primary tumor in patients with stage IV colorectal cancer: retrospective analysis of two randomized studies and a review of the literature. Ann Surg Oncol 18(12):3252–3260. https://doi.org/10.1245/s10434-011-1951-5
van der Geest LGM, Lam-Boer J, Koopman M et al (2015) Nationwide trends in incidence, treatment and survival of colorectal cancer patients with synchronous metastases. Clin Exp Metastasis 32(5):457–465. https://doi.org/10.1007/s10585-015-9719-0
Franko J, Shi Q, Meyers JP et al (2016) Prognosis of patients with peritoneal metastatic colorectal cancer given systemic therapy: an analysis of individual patient data from prospective randomised trials from the analysis and research in cancers of the Digestive System (ARCAD) database. Lancet Oncol 17(12):1709–1719. https://doi.org/10.1016/S1470-2045(16)30500-9
Klaver YLB, Lemmens VEPP, Creemers GJ et al (2011) Population-based survival of patients with peritoneal carcinomatosis from colorectal origin in the era of increasing use of palliative chemotherapy. Ann Oncol 22(10):2250–2256. https://doi.org/10.1093/annonc/mdq762
Klaver YLB, Simkens LHJ, Lemmens VEPP et al (2012) Outcomes of colorectal cancer patients with peritoneal carcinomatosis treated with chemotherapy with and without targeted therapy. Eur J Surg Oncol 38(7):617–623. https://doi.org/10.1016/j.ejso.2012.03.008
Verhoef C, de Wilt JH, Burger JWA et al (2011) Surgery of the primary in stage IV colorectal cancer with unresectable metastases. Eur J Cancer 47(suppl 3):S61–S66. https://doi.org/10.1016/S0959-8049(11)70148-4
Konishi T, Rodriguez-Bigas MA (2022) Primary Tumor Resection in Colorectal Cancer with Unresectable Synchronous Metastasis: time to reconsider the role of the Surgeon. Ann Surg Oncol 29(1):1–3. https://doi.org/10.1245/s10434-021-10949-4
Hu CY, Bailey CE, You YN et al (2015) Time trend analysis of primary tumor resection for stage IV colorectal cancer: less surgery, improved survival. JAMA Surg 150(3):245–251. https://doi.org/10.1001/jamasurg.2014.2253
Rahbari NN, Lordick F, Fink C et al (2012) Resection of the primary tumour versus no resection prior to systemic therapy in patients with colon cancer and synchronous unresectable metastases (UICC stage IV): SYNCHRONOUS–a randomised controlled multicentre trial (ISRCTN30964555). BMC Cancer 12:142. https://doi.org/10.1186/1471-2407-12-142
Rahbari NN, Biondo S, Feiβt M et al (2022) Randomized clinical trial on resection of the primary tumor versus no resection prior to systemic therapy in patients with colon cancer and synchronous unresectable metastases. J Clin Oncol 40(suppl 17):LBA3507. https://doi.org/10.1200/JCO.2022.40.17_suppl.LBA3507
Benson AB, Venook AP, Al-Hawary MM et al (2018) NCCN Guidelines Insights: Colon cancer, Version 2.2018. J Natl Compr Canc Netw 16(4):359–369. https://doi.org/10.6004/jnccn.2018.0021
Marin D, Catalano C, Baski M et al (2010) 64-Section multi-detector row CT in the preoperative diagnosis of peritoneal carcinomatosis: correlation with histopathological findings. Abdom Imaging 35(6):694–700. https://doi.org/10.1007/s00261-008-9464-9
Dohan A, Hoeffel C, Soyer P et al (2017) Evaluation of the peritoneal carcinomatosis index with CT and MRI. Br J Surg 104(9):1244–1249. https://doi.org/10.1002/bjs.10527
Laoukili J, Constantinides A, Wassenaar ECE et al (2022) Peritoneal metastases from colorectal cancer belong to Consensus Molecular Subtype 4 and are sensitised to oxaliplatin by inhibiting reducing capacity. Br J Cancer 126(12):1824–1833. https://doi.org/10.1038/s41416-022-01742-5
van der Willik KD, Ruiter R, van Rooij FJA et al (2020) Ascertainment of cancer in longitudinal research: the concordance between the Rotterdam Study and the Netherlands Cancer Registry. Int J Cancer 147(3):633–640. https://doi.org/10.1002/ijc.32750
Acknowledgements
The authors thank the registration team of the Netherlands Comprehensive Cancer Organization (IKNL) for the collection of data for the Netherlands Cancer Registry.
Funding
No funding was received for conducting this study.
Author information
Authors and Affiliations
Contributions
Anouk Rijken: Conceptualization, Methodology, Software, Formal analysis, Investigation, Writing – Original Draft, Visualization. Vincent van de Vlasakker: Conceptualization, Methodology, Writing – Origin Draft, Visualization. Geert Simkens: Conceptualization, Methodology, Formal analysis, Writing – Review & Editing, Visualization, Supervision. Koen Rovers: Conceptualization, Methodology, Formal analysis, Writing – Review & Editing, Visualization, Supervision. Felice van Erning: Conceptualization, Methodology, Software, Formal analysis, Investigation, Resources, Writing – Review & Editing, Visualization, Supervision, Project administration. Miriam Koopman: Conceptualization, Methodology, Writing – Review & Editing, Visualization, Supervision. Cornelis Verhoef: Conceptualization, Methodology, Writing – Review & Editing, Visualization, Supervision. Johannes de Wilt: Conceptualization, Methodology, Writing – Review & Editing, Visualization, Supervision. Ignace de Hingh: Conceptualization, Methodology, Investigation, Resources, Writing – Review & Editing, Visualization, Supervision, Project administration. All authors reviewed and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
Dr. Koopman reported having an advisory role for Nordic Farma, Merck-Serono, Pierre Fabre, Servier, Institutional scientific grants from Bayer, Bristol Myers Squibb, Merck, Personal Genome Diagnostics (PGDx), Pierre Fabre, Roche, Sirtex, Servier. Dr. de Wilt reported an unrestricted research grant from Medtronic and Roche, outside the submitted work, paid to the institute. Dr. de Hingh reported an unrestricted research grant from RanD/QPS and Roche, outside the submitted work, paid to the institute. For the remaining authors none were declared.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Rijken, A., van de Vlasakker, V.C.J., Simkens, G.A. et al. Primary tumor resection or systemic treatment as palliative treatment for patients with isolated synchronous colorectal cancer peritoneal metastases in a nationwide cohort study. Clin Exp Metastasis 40, 289–298 (2023). https://doi.org/10.1007/s10585-023-10212-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10585-023-10212-y