Abstract
This study was conducted to evaluate the impact of human activities on the ecological health and food chain at the shores of four Nile Islands in Great Cairo including El-Qeratten, El-Waraq, El-Zamalek, and El-Manial. Water quality, bacteria, phytoplankton, benthic algae, zooplankton, macrobenthic invertebrates, and bony fishes were examined at each island shore over two seasons including the lowest and the highest flow (February and July 2020, respectively). The obtained results showed that the average values of most of the chemicals in Nile Island’s water were within the Egyptian water quality limits. Discriminant analysis of physicochemical factors revealed a similarity between El-Waraq and El-Qerateen and between El-Manial and El-Zamalek. El-Qeratten was the most polluted island. It has the highest total and fecal coliform bacteria count (3.155 and 3.050 Log MPN/100 mL, respectively). El-Zamalek shores have the highest phytoplankton (33,582 cells × 104 L−1) and zooplankton count (310,891 organisms × m−3) and phyto-biochemical values. Biochemical analysis of phytoplankton demonstrated the richness of the bulk by protein (> 85% of biomass), indicating that phytoplankton has a high nutritional value. Elevated zooplankton count was recorded at El-Zamalek, which coincided with the peak of phytoplankton abundance. Mollusca were the dominant macrobenthic invertebrates on most of the island’s shores. Bony fishes were represented by 27 species and two crustaceans. The levels of the metals in fish samples were compared with the food safety guideline endorsed by the World Health Organization and Food and Agriculture Organization (WHO/FAO) to evaluate the toxicity level.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Freshwater resources have an important place among other natural resources. Egypt is the Nile (Burn, 2018). The Nile River is the most significant source of freshwater in Egypt. The Nile flows through the very crowded Great Cairo and support water for domestic, agriculture, and industrial activities (Al-Afify & Abdel-Satar, 2020). The impact of agricultural runoff and industrial and urban wastes increases contamination (Othman et al., 2020). Polluted water not only affects human health directly when consumed but also indirectly affects when it is used to irrigate food crops (Hussein et al., 2021).
There is a lack of routine monitoring systems in Egypt for identifying and determining the various types of contaminants in the Nile River environment, which consists of various forms of persistent organic pollutant loads (Shalaby et al., 2018). Changes in the Nile River water quality are affected not only by water management measures through the Nile barrages, but also by water and land usage for agricultural, industrial, and recreational purposes. The pollutants can enter the Nile system by direct discharges or surface runoff (Abdel-Satar et al., 2017). After the construction of the Aswan High Dam, the flow regime of the Nile has changed to a dam-regulated flow. The discharge in the summer is double that in the winter, with a water-level difference of approximately 2.5 m (Mohamed, 2019). The increase in pollution caused by the low water level in the Nile has become Egypt’s major problem, particularly since the completion of the Grand Ethiopian Renaissance Dam (Abdel-Satar et al., 2017; Heggy et al., 2021).
Burn (2018) indicated that some life cycles of the Nile food chain become more difficult when the balance of nutrients changes, especially with the number of scavenger and decomposer organisms increasing due to greater mortality rates. The abundance and biodiversity of planktons and other living organisms respond quickly to changes in the aquatic environmental variables, their abundance, and biodiversity serve as an ecological indicator of the aquatic environment (Salem & Mageed, 2021). To maintain the high water quality in a river system, the identification of fecal contamination sources is crucial (Maes et al., 2022). Plankton, macrobenthic invertebrates, macrophytes, and fishes serve as a bioindicator for the environmental status in a given time (Zakaria et al., 2016, 2018; Farrag et al., 2019).
The Nile River has hundreds of islands from Aswan to the Mediterranean Sea. In recent years, the majority of Nile River Islands have undergone rapid changes in land use (Taha, 2014). The continuing trend of urbanization has resulted in significant increases in urban areas, reducing the amount of available green spaces. Consequently, there has been a change in the morphology of the river around the islands (Raslan & Salama, 2015). Many of them have been developed in the Great Cairo region because of the construction of the Aswan High Dam. Nile Islands are economically valuable as they have significant growth potential. Changes in the hydraulic characteristics and human interventions affect the processes of erosion and sedimentation on the islands (Sadek, 2013). Nile Islands are considered the real breathing area concerning their habitats and biological diversity in addition to their global interest. They currently represent one of the most important wintering grounds for water birds in Egypt that migrate through these islands (Bunbury, 2019).
The Great Cairo area has four large permanent islands: El-Qeratten, El-Waraq, El-Zamalek, and El-Manial. These islands were occupied by human activities such as cultivation, residential development, and industrialization (Bunbury, 2019). The literature indicates a paucity of information describing the freshwater biodiversity across the Nile Islands areas. In addition, there are very few studies on the health assessment of the Nile Islands. Moreover, there are no detailed studies on the Nile Islands in Egypt regarding their shore environment and biodiversity to improve their local environment. This study was conducted to evaluate the ecological health of four River Nile Islands in Great Cairo based on the water quality and biodiversity assessment. All the Nile Islands are important and in the assessment by the program of the National Institute of Oceanography and Fisheries, especially after the Grand Ethiopian Renaissance Dam work, where all Nile shores may be affected by the decrease in the water budget due to the Nile’s altered flow which will increase the existing water deficit for Egypt (Heggy, 2021).
Material and methods
Study area
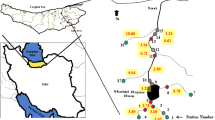
This study is focused on four River Nile Islands with three features, namely, agricultural activities only (El-Qeratten), agricultural activities and inhabitant residences (El-Waraq), and inhabitant residences only (El-Zamalek and El-Manial), as in Table 1 and Fig. 1. The survey program of the study was implemented between February (the lowest flow season) and July 2020 (the highest flow season). Six stations were examined which are located on surrounding islands.
Morphometric features and water quality
Water depth was measured using a portable echosounder device (Lowrance Sonar-X-25 and Navman, fish 4500). Water samples were collected from the subsurface for each station using a water sampler and then transferred into well-cleaned plastic bottles. Water temperature, electrical conductivity (EC), and pH were measured in the field using Con 500 conductivity/TDS/temperature meter and combined meter pH/EC/TDS/temperature (Mi 805). Transparency was measured using a Secchi disk (25 cm). Physical and chemical analyses were conducted according to APHA (2005). Total solid (TS) content was measured by evaporating a known volume of a well-mixed sample. Dissolved oxygen (DO) content was assayed using a modified Winkler method and calculated as a percentage. Biological oxygen demand (BOD5) was determined using the 5-day incubation method. Chemical oxygen demand (COD) was evaluated using the potassium dichromate method. Nitrate was measured as nitrite after cadmium reduction. Total phosphorus (TP) was estimated as reactive phosphate after persulfate digestion using the ascorbic acid–molybdate method. Sediment samples were collected from the four islands using the Ekman device from the top 20 cm layer of the bottom. Grain size analysis was performed using the dry sieving technique (Folk, 1980), and the sediment organic matter (OM) was estimated using the wet oxidation method (Loring & Rantala, 1992).
Biological methods
For microbiological analysis, water samples were aseptically collected in 200-mL sterile brown bottles, transported to the laboratory, and stored at 4 °C. Bacteriological analysis was completed within 48 h of sampling; total bacteria were enumerated on plate count agar medium at incubation temperatures of 22 °C and 37 °C. The most probable number (MPN) procedure was used to determine the total coliform (TC) and fecal coliform (FC) counts using lauryl tryptose broth (LTB) (35 °C ± 0.5 °C at 24 ± 2 h) for TC, EC broth (44.5 °C ± 0.2 °C at 24 ± 2 h) for FC, and azide dextrose broth for fecal streptococci (APHA, 2005). Total diazotrophs (TS) were enumerated using the surface-inoculated plate method on N-deficient combined carbon sources agar medium (Hanna et al., 2013). Samples for analyzing phytoplankton communities were collected using a 1.5-L Ruttner sampler, which was preserved immediately with 4% neutral formalin and then transferred into a glass cylinder, after which Lugol’s iodine solution was added (APHA, 2005). The phytoplankton count (cells L−1) was estimated using an inverted microscope. Epipelic diatom samples were collected using an Ekman grab sampler from the surface layer. Diatom sample preparation, diatom cleaning, and counting were conducted according to ANS (2002). For phyto-biochemical analysis, water samples were collected, sieved, and filtered through a 100-μm-sized mesh net. Then, 10 mL of the filtered water was refiltered using Whatman GF/F (0.7-μm pore diameter) fiber circles. Total protein content was determined by the Biuret method (Brown, 1984). Carbohydrate content was measured according to the phenol–sulfuric acid method (DuBois et al., 1956), and total lipid content was determined using the sulfo-phospho-vanillin procedure (Anschau et al., 2017). Elemental composition was determined as described by Vollenweider (Vollenweider, 2000). Zooplankton samples were collected from each site by filtering 30 L of surface water through a 55-µm mesh plankton net. The collected samples were preserved in plastic bottles containing 4% formalin. Zooplankton count was expressed as the number of organisms per cubic liter. Samples of macrobenthic invertebrates (MBI) were collected using an Ekman grab sampler. The collected samples were washed in a sieve of a mesh diameter of 500 μm and then preserved in a 7% neutral formalin solution. In the laboratory, the samples were washed again through a net with a mesh diameter of 0.5 mm and sorted according to genera or species, and each taxon was identified and counted. The population density was calculated and expressed as organisms per square meter. Submerged macrophytes were sampled using a grapnel to collect relative standing crop samples (five grapnel hauls per sampling area) (Ali et al., 2007). The presence of species was expressed as a percentage of sites with taxa. Fish samples were collected using commercial and experimental fishing gears and methods. Samples were maintained freshly in an icebox and transferred to the laboratory for taxonomic analysis to the species level according to Bishai and Khalil (1997) and FishBase (Froese & Pauly, 2019). For heavy metal (HM) analysis of fish muscles and gills, a small piece of tissue sample (0.5 ± 0.01 g) was digested using acid-washed digestive vessels of Teflon as described by Goldberg et al. (1983). The metal concentrations in the extracts were analyzed for total Fe, Mn, Zn, Cu, Pb, and Cd levels using GBC atomic absorption reader model Savant AA-AAS with GF 5000 graphite furnace. HM concentrations were determined on a ppm wet weight basis.
Analytical methods
Data were statistically analyzed to evaluate the characteristics of the different islands. Discriminant analysis (DA) was conducted for physicochemical factors for the different collection sites in the four islands. Bacteria, plankton, and bottom fauna were described and related to major environmental gradients using canonical correspondence analysis (CCA) using the XLSTAT 2016 software. The Mstat program (v.4, 1982) was used for the analysis of variance to investigate significant effects at p < 0.05.
Results and discussion
Aquatic food chains are affected by certain environmental contaminants. Pollution, eutrophication, and human activities are recognized as primary threats to freshwater biodiversity and river health (Abdel-Satar et al., 2017).
Physicochemical characteristics of water and sediments
The mean and range of the physicochemical characteristics of the four islands are presented in Table 2, showing the comparison of the islands’ water variables with Egyptian water quality limits (EWQL), which were specified for freshwater according to Law 48 of 1982 regarding the protection of the Nile River from pollution. In all islands, no spatial variations (p > 0.1) were recorded for all the examined variables. The water temperatures were low in February compared with those in July. In contrast, the pH values, conductivity, TDS, and transparency were high in February compared with those in July (Table 2). Overall, the islands were slightly basic with comparable mean pH values. The average DO values in all the examined islands were within the water quality limits endorsed by EWQL and showed significant temporal variations (p < 0.001), whereas the water samples collected in July showed the lowest DO percentage, but no significant differences were obtained (p > 0.05) in DO values. El-Qeratten had the lowest mean value for TP and suspended solids. The mean nitrate–N concentrations were the highest in El-Zamalek. The mean COD and BOD values decreased at El-Qeratten and El-Manial, whereas the BOD/COD ratio was > 0.6. Al-Afify et al. (2018) reported that nitrate and BOD values can be considered indicators of pollution in the Nile River. Nitrate and TP levels correlated significantly (r = 0.60, 0.82, 0.80, and 0.65; n = 12, p < 0.01, for El-Qeratten, El-Waraq, El-Zamalek, and El-Manial, respectively), indicating the common nutrient salt sources from industrial, domestic, and agricultural wastes as the main sources of N and P inputs into the Nile River water. This leads to biodiversity changes and disturbances in benthic algal, phytoplankton, and macroinvertebrate biomass inhabiting the Nile River (Abdel-Satar et al., 2017; Othman et al., 2020). The bottom sediments of El-Qeratten and El-Waraqwere are sandy mud, whereas those of El-Zamalek and El-Manial were sandy. Sediment OM content was the highest at El-Zamalek shores, followed by El-Qeratten, whereas El-Manial had the lowest content.
The results of the DA for the physicochemical variables in the four islands showed a similarity between El-Waraq and El-Qeratten and between El-Manial and El-Zamalek (Fig. 2). The first two islands were dominated by agricultural activities, while the other two had residences only.
Biodiversity
The number of phytoplankton, attached diatoms, zooplankton, MBI, macrophytes, and fish populations found in all samples followed a consistent pattern, with the mean richness in diversity for the four islands following the order: El-Qeratten > El-Zamalek > El-Waraq > El-Manial (Table 3). El-Qeratten showed more richness in diversity than the other islands’ biota, particularly in terms of phytoplankton and attached diatoms. Evaluation of the difference between the minimum and the maximum number of recorded species as well as the overall number of species at each island revealed the following order: El-Qeratten > El-Manial > El-Zamalek > El-Manial.
Changes in food chain
Bacteria
As shown in Table 4, the different bacterial counts significantly varied (p < 0.05) depending on the study islands. The differential temperature ratio test, in which the ratios of bacteria grown at 20 °C to those grown at 35 °C were less than 10 compared with the permissible standard of 10:1 (Directive, 1998), suggested that the island’s water was heavy pollution. Although the presence of pathogenic bacteria is not necessarily related to a high FC value, a high FC value implies poor water quality and increased health risk associated with the presence of pathogenic microorganisms (Farhadinejad et al., 2014). In general, the sampling site has an impact on the bacteriological quality of the water being examined. Land use and expanded animal–agriculture systems can have an impact on FC. The principal factors influencing bacterial pollution in agricultural streams are reported to be livestock and manure application as fertilizers (Pandey et al., 2014). Most households in the rural areas, such as in El-Qeratten, lack access to sewer systems, or the usage of individual septic tanks, resulting in untreated sewage being discharged into rivers, perhaps leading to fecal contamination (Al-Badaii & Shuhaimi-Othman, 2015; Othman et al., 2020). Associative nitrogen-fixing bacteria (diazotrophs) were found in significant population densities in the islands’ water, indicating the intrusion of agricultural drainage waters into the island’s Nile water. Diazotrophs play a vital role in the aquatic system, not only as N2 fixers but also they increase water productivity by releasing growth factors that are beneficial for flora and fauna (Othman et al., 2016). The diazotroph bacteria that are capable of growth on N-deficient combined carbon source medium were also detected in mid-stream of River Nile water where their values ranged between 102 and 104 (Ali et al., 2011). El-Qeratten had unusually high average levels of TC and FC bacteria when compared to other islands, while fecal streptococci were found in the lowest count. For monitoring microbiological water quality, FC counts are used as a guideline.
Phytoplankton
Phytoplankton abundance was represented by five classes: Bacillariophyceae, Chlorophyceae, Cyanobacteria, Dinophyceae, and Euglenophyceae, in order of their abundance, except in El-Zamalek where Cyanophyceae occupied the second predominant position. Cryptophyceae and Chrysophyceae were very rare classes in the islands. El-Qeratten showed the highest diversity of phytoplankton species, where 90 species were identified. The lowest species diversity was recorded at El-Manial (69 species). Phytoplankton abundance and species composition revealed slight variation between the four islands. El-Qeratten shores water had the lowest counts (16,112 cells × 104 L−1), while El-Zamalek shores had the highest counts (33,582 cells × 104 L−1), Table 6, with the highest levels of nitrate and BOD (103.27 µg L−1 and 5.85 mg L−1, respectively) recorded in its water. High nutrient concentrations as a limiting factor influenced phytoplankton distribution and growth, in addition to macrophyte density (Haroon & Hussian, 2017). Phytoplankton distribution in the islands was significantly affected by environmental variables. CCA (Fig. 3) indicated that diatoms were affected by COD, BOD, and TS, whereas the presence of blue-green algae was highly affected by pH, as they tend to grow better at a high pH. Through photosynthesis and respiration, there is a relationship between algal development and oxygen levels (DO, COD, and BOD) in the aquatic ecosystem (Ma et al., 2021).
Phyto-biochemical analysis revealed the richness of phytoplankton bulk by protein (> 85% of biomass), followed by carbohydrates and lipids (Table 5), indicating that phytoplankton has a high nutritional value. Protein contents indicated that phytoplankton is physiological healthy with high relative growth rates, and high protein content suggests that phytoplankton had no nitrogen depletion in the water body (Abd El-Hady et al., 2016). The highest phyto-biochemical contents were recorded at El-Zamalek (363.41 mg L−1). This elevation may be related to increasing nitrate levels (103.3 µg L−1) at the island (Fig. 4), wherein a strong correlation was observed between phytoplankton proteins and nitrate at El-Zamalek (r = 0.96). The N-sufficient condition can increase the cellular algal content of proteins where nitrogen represents a critical macronutrient that regulates metabolism and consequently the biochemical composition and growth of microalgae (Zarrinmehr et al., 2020). The increase in the phytoprotein contents at El-Zamalek was related to the highest phytoplankton counts (33,582 cells × 104 L−1), with Cyanophyceae and Bacillariophyceae representing the two largest groups (9583 and 15,833 cells × 104 L−1, respectively). Cyanophyceae have a high crude protein content (60–65%), which is the most component of cyanobacterial biomasses (Niccolai et al., 2019). Under climatic conditions, the carbon allocation pattern reveals a high proportion of proteins, which is required to support Bacillariophyceae’s rapid growth rate (Wagner et al., 2017).
Elemental ratios (C, N, H, and O) of phytoplankton provide insight into the mechanism by which nutrients in the water body are allocated within algal cells to support cellular functions, growth, and metabolism. The carbon contents of phytoplankton in the four investigated islands were > 50% of elemental compositions. The high incorporation of carbon into the phytoplankton biochemical structure was a characteristic pattern of the photosynthetic allocations of healthy phytoplankton biomass (Schulhof et al., 2019). The ideal C/N ratio, according to Redfield et al. (1963), is 6.6. However, phytoplankton progressed quickly, with the C/N ratio dropping to 3.1 along the study island’s shores with the increase in phytoprotein, indicating that the algae were not nitrogen-limited (Napiórkowska-Krzebietke et al., 2015). Protein represented the largest pool of cellular C and N in algal species under steady-state growth (Liefer et al., 2019; Islam et al., 2019).
Epipelic diatoms
The attached epipelic diatoms were represented by 10 orders with a total of 80 species. The most dominant order (> 50% of the total count) was Thalassiosirales, which had roughly comparable counts on all four islands, showing great cosmopolitanism (Table 6). El-Qeratten shores had the highest number of species, followed by El-Waraq, which is consistent with the high TP concentration. According to CCA (Fig. 5), diatoms highly correlated with TP and the sandy mud sediment texture. The species composition of benthic diatoms responds directly to nutrients, with total N or P concentrations increasing up to a point before leveling off. The region above which primary producers have not developed to adapt to nutrient concentration could be the point where the autotrophic state no longer varies. CCA findings revealed that the studied environmental variables can explain 40% of the diatom community variation. Fragilariales was substantially influenced and correlated with TP, whereas Thalassiosirales was near the zero point, demonstrating a cumulative effect of the different environmental variables. Fragilariales were highly affected and correlated with TP. The response of the same species to similar nutrient enrichment conditions can be different, suggesting that factors other than nutrients have a combined effect with the nutrient in determining species composition and other physical factors (Yang et al., 2015). According to the DA, the diatom data can characterize or separate El-Waraq and El-Manial in only a few points, whereas most points had the same pattern and could not separate the islands into distinct clusters. This shows that there are no major differences in the environment of the four examined islands when it comes to the community of attached diatoms. This could be due to some tolerant species cosmopolitan and the influence of other environmental conditions.
Canonical correspondence analysis (CCA). Ordination diagram of the attached diatoms orders; Achnanthales (Ach.), Aulacoseirales (Aul.), Bacillariales (Bci.), Cymbellales (Cym.), Eunotiales (Eun.), Fragilariales (Frg.), Naviculales (Nav.), Rhopalodiales (Rh.), Thalassiophysales (Thp.), and Thalassiosirales (Ths.) data together with physicochemical variables: pH, DO, COD, BOD, NO3−-N, TP, and TS
Zooplankton
Zooplankton organisms were represented by small rotifers (> 90% of total counts), copepods, and cladocerans. According to Sun et al. (2021) increased predation is critical for small-bodied zooplankton dominance in the aquatic system. The study islands’ major rotifer species are eutrophic indicator species (Palmer & Herat, 2021). As a result, the considerable dominance of rotifers over other zooplankton groups in the studied area, both in terms of abundance and species composition, provides evidence of the trophic status of the Nile River islands. Phytoplankton abundance has been reliably associated with zooplankton abundance, where the highest zooplankton count was recorded at El-Zamalek (310,891 organisms m−3), which coincided with the peak of phytoplankton abundance, Table 6. The variation in phytoplankton and zooplankton composition was consistent with the regulation of nutrients and top-down effects by fish (Makarewicz & Jones, 1990). Planktivore abundance increases with phytoplankton and zooplankton abundance, and these organisms selectively feed on larger zooplankton (Iglesias et al., 2011). CCA ordination of the zooplankton assemblage data (Fig. 6) demonstrated a broad continuum with TP, nitrate–N, and DO in the four islands, where El-Manial shores zooplankton assemblages were relatively distinct.
Macrobenthic invertebrates
A total of 34 species of MBI belonging to three major phyla, Arthropoda, Annelida, and Mollusca, were identified. Of these, there were 9 species of arthropods, 6 species of annelids, and 19 species of mollusks. Mollusca was the dominant group at all islands, except El-Waraq shores, where Annelida was the most dominant, indicating that the type of sediments at the islands has a specific role, where El-Waraq shores had the highest percentages of mud deposits, and Annelida preferred this sort of sediment (Abdel Gawad, 2019). El-Qeratten had the highest percentage of Mollusca, with 1478 organisms m−2, while El-Zamalek had the lowest number, with 739 organisms−2. When compared to El-Qeratten and El-Waraq, the overall number of Annelida species fell at El-Zamalek and El-Manial shores. Annelida showed positive correlations with pH and COD (r = 0.96 and 0.58, respectively) and negative correlations with DO and depth (r = − 0.35 and − 0.63, respectively). Arthropoda, especially the larvae of Chironomidae, showed significant positive correlations with BOD and DO (r = 0.96 and 0.6, respectively). Mollusca exhibited considerable positive correlations with transparency and Ca++ (r = 0.75 and 0.66, respectively). The results of CCA of MBI group data (Fig. 7) revealed a continuum between the distribution of MBI in the islands with TP, TS, COD, and DO, where the distribution of each species depends on the levels and types of such chemical variables.
Macrophytes
Five macrophyte species were recorded, demonstrating poor species diversity in the shores of the studied islands, especially at El-Zamalek and El-Manial. They were classified into three submerged and two floating macrophyte species belonging to five families and five genera (Table 7). Among these, dense populations of the submerged macrophyte species Myriophyllum spicatum were recorded at 83.33% of the total sampling sites, followed by those of Ceratophyllum demersum (41.66%). However, the other three species were very rare. Madsen (1998) considered M. spicatum as an invasive species, indicating the effect of environmental factors. In this study, it was found to have a prominent effect on the community composition of submerged macrophytes, as it competes aggressively to displace and reduce the diversity of the originally present aquatic plants. Similar findings were reported by Haroon and Hussian (2017) who reported the shading effect as well as the allelopathic substances released by M. spicatum in reducing the growth of other submerged macrophyte species. Furthermore, El-Ghani et al. (2011) indicated that high nitrate availability and sediment OM lead to increased invasion of the submerged macrophyte species M. spicatum.
Fish community
A total of 29 fish species (27 bony fish and 2 crustaceans) belonging to 17 families were recorded at the investigated islands (Table 8). The occurrence percentage showed that Cichlidae was the most dominant and diversified family (97.2%), followed by the families Clariidae (95.8%), Bagridae (93.8%), Poeciliidae (83.3%), Claroteidae (72.9), and Mormyridae (70.8%), and the other families constituted < 60% (Table 9). Elsaied et al. (2021) concluded that most inland African freshwater fishes belong to two broad phylogenetic orders, Cichliformes and Cyprinodontiformes, both significantly diversify in terms of fisheries and aquaculture. The family Cichlidae is the most prosperous, omnivorous, and their feeding habits slightly vary with fish size, sex, and season (Shalloof et al., 2020; Temesgen et al., 2022). The two crustaceans included Procambarus clarkii and Mediapotamon spp. El-Manial had the most abundant fish species.
Oreochromis niloticus as a bioindicator of pollution
Fish are at the top of the food chain and are one of the most significant biomonitors for heavy metal pollution assessment in the aquatic ecosystem. They can accumulate metals that are passed on to humans through fish consumption, resulting in acute or chronic diseases (Yacoub et al., 2021). O. niloticus is the main economic fish in the Nile, used as a bioindicator of water pollution. Fe, Mn, Cu, and Zn are essential metals but can be lethal at high concentrations, whereas Pb and Cd can be harmful to living organisms and the environment at low concentrations. The levels of the metals were compared with the food safety guideline endorsed by the World Health Organization/Food and Agriculture Organization (WHO/FAO) to evaluate the toxicity level. The bioaccumulation of heavy metals was lower in muscles than in the gills. The adsorption of metals onto the surface of gills, as the first target for pollutants in water, could also be an important influence on the total metal levels of the gill. The results indicated that Fe was the most accumulated element, followed by Zn and Cu, while Cd was the least accumulated one (Table 10). The rise in Fe accumulation in fish was higher than that of other metals, possibly because of the increase of total dissolved Fe levels in aquatic media (Abdel-Khalek et al., 2020). The concentrations of Cu, Pb, and Cd in both muscles and gills, and the Zn concentration in muscles were below the WHO/FAO FSG (FAO/WHO, 2010). The concentrations of heavy metals in fish depend on the feeding habit, size, age, lifestyle, and exposure duration to contaminants (Bastami et al., 2015).
Conclusion
Survey monitoring program was implemented during the lowest Nile flow season and the highest Nile flow season to maintain ecological health and food chain around some Nile islands. Discriminant analysis of physicochemical factors showed high similarity between El-Waraq and El-Qeratten and between El-Manial and El-Zamalek. High average levels of TC and FC bacteria were observed at El-Qeratten compared with those at other islands, whereas the highest phytoplankton counts and zooplankton were recorded at El-Zamalek. The highest number of species of the attached diatoms was recorded at El-Qeratten. The bioaccumulation of heavy metals was lower in the muscles of O. niloticus than that in the gills. El-Qeratten Island was the most polluted island in comparison with other studied islands. Regular evaluation surveys and further studies should be conducted to monitor the expected changes in water sources near highly populated areas, such as the Great Cairo.
Data availability
Data sharing is not applicable to this article as no datasets were generated or analyzed during the current study.
References
Abd El-Hady, H. H., Fathey, S. A., Ali, G. H., & Gabr, Y. G. (2016). Biochemical profile of phytoplankton and its nutritional aspects in some khors of Lake Nasser, Egypt. Egyptian Journal of Basic and Applied Sciences, 3(2), 187–193. https://doi.org/10.1016/j.ejbas.2016.03.002
Abdel-Khalek, A. A., Zayed, H. S., Elsayad, S. M., & Zaghloul, K. H. (2020). Assessment of metal pollution impacts on Tilapia zillii and Mugil cephalus inhabiting Qaroun and Wadi El-Rayan lakes, Egypt, using integrated biomarkers. Environmental Science and Pollution Research, 27(21), 26773–26785. https://doi.org/10.1007/s11356-020-09095-3
Abdel-Satar, A. M., Ali, M. H., & Goher, M. E. (2017). Indices of water quality and metal pollution of Nile River, Egypt. Egyptian Journal of Aquatic Research, 43(1), 21–29. https://doi.org/10.1016/j.ejar.2016.12.006
Abdel Gawad, S. S. (2019). Using benthic macroinvertebrates as indicators for assessment the water quality in River Nile, Egypt. Egyptian Journal of Basic and Applied Sciences, 6(1), 206–219. https://doi.org/10.1080/2314808X.2019.1700340
Al-Afify, A. D. G., & Abdel-Satar, A. M. (2020). Risk assessment of heavy metal pollution in water, sediment and plants in the Nile River in the Cairo region. Egypt. Oceanological and Hydrobiological Studies, 49(1), 1–12. https://doi.org/10.1515/ohs-2020-0001
Al-Afify, A. D. G., Othman, A. A., & Ramadan, M. F. (2018). Characterization of chemical and microbiological quality of Nile River surface water at Cairo (Egypt). Rendiconti Lincei, 29(3), 725–736. https://doi.org/10.1007/s12210-018-0721-8
Al-Badaii, F., & Shuhaimi-Othman, M. (2015). Water pollution and its impact on the prevalence of antibiotic-resistant E. coli and total coliform bacteria: A study of the Semenyih river, Peninsular Malaysia. Water Quality, Exposure and Health, 7(3), 319–330. https://doi.org/10.1007/s12403-014-0151-5
Ali, M. M., Mageed, A. A., & Heikal, M. (2007). Importance of aquatic macrophyte for invertebrate diversity in large subtropical reservoir. Limnologica, 37(2), 155–169. https://doi.org/10.1016/j.limno.2006.12.001
Ali, S. M., Sabae, S. Z., Fayez, M., Monib, M., & Hegazi, N. A. (2011). The influence of agro-industrial effluents on River Nile pollution. Journal of Advanced Research, 2(1), 85–95. https://doi.org/10.1016/J.JARE.2010.08.008
ANS. (2002). Protocols for the analysis of algal samples collected as part of the U.S. Geological Survey National Water-Quality Assessment Program, 02–06. https://water.usgs.gov/nawqa/protocols/algprotocol/index.html. Accessed 29 April 2021.
Anschau, A., Caruso, C. S., Kuhn, R. C., & Franco, T. T. (2017). Validation of the sulfo-phosphovanillin (SPV) method for the determination of lipid content in oleaginous microorganisms. Brazilian Journal of Chemical Engineering, 34(1), 19–27. https://doi.org/10.1590/0104-6632.20170341s20140222
APHA. (2005). American Public Health Association. Standard Methods for the Examination of Water and Wastewater, 18th ed. Washington. https://www.worldcat.org/title/standard-methods-for-the-examination-of-water-and-wastewater/oclc/156744115. Accessed 27 April 2021.
Bastami, K. D., Afkhami, M., Mohammadizadeh, M., Ehsanpour, M., Chambari, S., & Aghaei, S. (2015). Bioaccumulation and ecological risk assessment of heavy metals in the sediments and mullet Liza klunzingeri in the northern part of the persian gulf. Marine Pollution Bulletin, 94(1–2), 329–334. https://doi.org/10.1016/j.marpolbul.2015.01.019
Bishai, H. M., & Khalil, M. T. (1997). Freshwater fishes of Egypt: Egyptian Environmental Affairs Agency.
Brown, I. (1984). Analytical biochemistry. Biochemical Education, 12(4), 189. https://doi.org/10.1016/0307-4412(84)90148-1
Bunbury, J. (2019). Islands in the Nile. In The Nile and Ancient Egypt (pp. 77–92). Cambridge University Press. https://doi.org/10.1017/9780511997884.007
Burn, J. W. (2018). A river in drought: Consequences of a low Nile at the end of the old kingdom. Environment and Ecology Research, 6(5), 446–460. http://www.hrpub.org. https://doi.org/10.13189/eer.2018.060505
Directive, C. (1998). On the quality of water intended for human consumption. Official Journal of the European Communities, 330, 32–54.
DuBois, M., Gilles, K. A., Hamilton, J. K., Rebers, P. A., & Smith, F. (1956). Colorimetric method for determination of sugars and related substances. Analytical Chemistry, 28(3), 350–356. https://pubs.acs.org/doi/10.1021/ac60111a017. Accessed 27 April 2021.
El-Ghani, M. A., El-Fiky, A. M., Soliman, A., & Khattab, A. (2011). Environmental relationships of aquatic vegetation in the fresh water ecosystem of the Nile Delta, Egypt. African Journal of Ecology, 49(1), 103–118. https://doi.org/10.1111/j.1365-2028.2010.01237.x
Elsaied, H., Soliman, T., Abdelmageed, A. A., & Abu-Taleb, H. T. (2021). Applications and challenges of DNA barcoding and metabarcoding in African fisheries. The Egyptian Journal of Aquatic Research, 47(1), 1–12. https://doi.org/10.1016/J.EJAR.2021.02.003
FAO/WHO. (2010). Summary report of the seventy-third meeting of lECFA (p. 17). Joint FAO/WHO Expert Committee on Food Additives
Farrag, M. M., El-Naggar, H. A., Abou-Mahmoud, M. M., Alabssawy, A. N., Ahmed, H. O., Abo-Taleb, H. A., & Kostas, K. (2019). Marine biodiversity patterns off Alexandria area, southeastern Mediterranean Sea, Egypt. Environmental Monitoring and Assessment, 191, 367.
Farhadinejad, T., Khakzad, A., Jafari, M., Shoaee, Z., Khosrotehrani, K., Nobari, R., & Shahrokhi, V. (2014). The study of environmental effects of chemical fertilizers and domestic sewage on water quality of Taft region, Central Iran. Arabian Journal of Geosciences, 7(1), 221–229. https://doi.org/10.1007/s12517-012-0717-0
Folk, R. L. (1980). Petrology of sedimentary rocks. (p. 182). Hemphill Publishing Co., Austin, Texas. https://repositories.lib.utexas.edu/handle/2152/22930. Accessed 27 April 2021.
Froese, R., & Pauly, D. (2019). FishBase. World Wide Web Electronic Publication. https://www.fishbase.de/. Accessed 29 April 2021.
Goldberg, E. D., Koide, M., Hodge, V., Flegal, A. R., & Martin, J. (1983). U.S. Mussel Watch: 1977–1978 results on trace metals and radionuclides. Estuarine, Coastal and Shelf Science, 16(1), 69–93. https://doi.org/10.1016/0272-7714(83)90095-1
Hanna, A. L., Youssef, H. H., Amer, W. M., Monib, M., Fayez, M., & Hegazi, N. A. (2013). Diversity of bacteria nesting the plant cover of north Sinai deserts, Egypt. Journal of Advanced Research, 4(1), 13–26. https://doi.org/10.1016/j.jare.2011.11.003
Haroon, A. M., & Hussian, A. E. M. (2017). Ecological assessment of the macrophytes and phytoplankton in El-Rayah Al-Behery, River Nile, Egypt. Egyptian Journal of Aquatic Research, 43(3), 195–203. https://doi.org/10.1016/j.ejar.2017.08.002
Heggy, E., Sharkawy, Z., & Abotalib, A. Z. (2021). Egypt’s water budget deficit and suggested mitigation policies for the Grand Ethiopian Renaissance Dam filling scenarios. Environmental Research Letters, 16(7), 74022. https://doi.org/10.1088/1748-9326/AC0AC9
Hussein, A. M., Mahmoud, R. K., Sillanpää, M., & Abdel Wahed, M. S. M. (2021). Impacts alum DWTPs sludge discharge and changes in flow regime of the Nile River on the quality of surface water and cultivated soils in Fayoum watershed, Egypt. Science of the Total Environment, 766, 144333. https://doi.org/10.1016/J.SCITOTENV.2020.144333
Iglesias, C., Mazzeo, N., Meerhoff, M., Lacerot, G., Clemente, J. M., Scasso, F., et al. (2011). High predation is of key importance for dominance of small-bodied zooplankton in warm shallow lakes: Evidence from lakes, fish exclosures and surface sediments. Hydrobiologia, 667(1), 133–147. https://doi.org/10.1007/s10750-011-0645-0
Islam, M. J., Jang, C., Eum, J., Jung, S. M., Shin, M. S., Lee, Y., et al. (2019). C: N: P stoichiometry of particulate and dissolved organic matter in river waters and changes during decomposition. Journal of Ecology and Environment, 43(1), 4. https://doi.org/10.1186/s41610-018-0101-4
Liefer, J. D., Garg, A., Fyfe, M. H., Irwin, A. J., Benner, I., Brown, C. M., et al. (2019). The macromolecular basis of phytoplankton C:N:P under nitrogen starvation. Frontiers in Microbiology, 10(MAR), 763. https://doi.org/10.3389/fmicb.2019.00763
Loring, D. H., & Rantala, R. T. T. (1992). Manual for the geochemical analyses of marine sediments and suspended particulate matter. Earth Science Reviews, 32(4), 235–283. https://doi.org/10.1016/0012-8252(92)90001-A
Ma, Q., Chen, Y., Wang, F., & Li, H. (2021). Responses of primary productivity and phytoplankton community to the atmospheric nutrient deposition in the East China Sea. Atmosphere, 12(2), 210. https://doi.org/10.3390/atmos12020210
Madsen, J. D. (1998). Predicting invasion success of Eurasian watermilfoil. Journal of Aquatic Plant Management, 36, 28–32. https://www.cabi.org/isc/abstract/19982303267. Accessed 27 April 2021.
Maes, S., Odlare, M., & Jonsson, A. (2022). Fecal indicator organisms in northern oligotrophic rivers: An explorative study on Escherichia coli prevalence in a mountain region with intense tourism and reindeer herding. Environmental Monitoring and Assessment, 194–264.
Makarewicz, J. C., & Jones, H. D. (1990). Occurrence of Bythotrephes cederstroemi in Lake Ontario offshore waters. Journal of Great Lakes Research, 16(1), 143–147. https://doi.org/10.1016/S0380-1330(90)71407-4
Mohamed, N. N. (2019). Importance of Aswan High Dam to Egypt. In Handbook of Environmental Chemistry (vol. 79, pp. 53–72). Springer Verlag. https://doi.org/10.1007/698_2018_280
Napiórkowska-Krzebietke, A., Napiórkowska-Krzebietke, A., Hussian, A.-E. M., Toufeek, M. A. F., Abd El-Monem, A. M., & Morsi, H. H. (2015). Phytoplankton response to changes of physicochemical variables in Lake Nasser, Egypt. Journal of Elementology, (4/2015). https://doi.org/10.5601/jelem.2015.20.1.895
Niccolai, A., Chini Zittelli, G., Rodolfi, L., Biondi, N., & Tredici, M. R. (2019). Microalgae of interest as food source: Biochemical composition and digestibility. Algal Research, 42, 101617. https://doi.org/10.1016/j.algal.2019.101617
Othman, A. A., Al-Afify, A. D. G., Abdel-Satar, A. M., & Ramadan, M. F. (2020). Quality assessment of surface water using the Nile Chemical Pollution Index (NCPI) and microbiological pollution of the Rosetta Branch (Nile River, Egypt). African Journal of Aquatic Science. https://doi.org/10.2989/16085914.2020.1807898
Othman, A. A., Ali, S. M., & Abbas, M. T. (2016). Microbial water quality and diazotrophic bacteria community in lake Nasser Khors, Egypt. Research Journal of Environmental Toxicology, 10(2), 100–108. https://doi.org/10.3923/rjet.2016.100.108
Palmer, J., & Herat, S. (2021). Ecotoxicity of microplastic pollutants to marine organisms: A systematic review. Water, Air, & Soil Pollution, 232(5), 1–21.
Pandey, P. K., Kass, P. H., Soupir, M. L., Biswas, S., & Singh, V. P. (2014). Contamination of water resources by pathogenic bacteria. AMB Express, 4(1), 1–16. https://doi.org/10.1186/s13568-014-0051-x
Raslan, Y., & Salama, R. (2015). Development of Nile River islands between Old Aswan Dam and new Esna barrages. Water Science, 29(1), 77–92. https://doi.org/10.1016/j.wsj.2015.03.003
Redfield, A. C., Ketchum, B. H., & Richards, F. A. (1963). The influence of organisms on the composition of seawater. The Sea, 2, 26–77.
Sadek, N. (2013). Island development impacts on the Nile River morphology. Ain Shams Engineering Journal, 4(1), 25–41. https://doi.org/10.1016/j.asej.2012.06.006
Salem, T. A., & Mageed, A. A. A. (2021). Assessment of the impact of the Nile flood on food chain in Lake Nasser—Egypt, with Special Reference to Turbidity. Open Journal of Ecology, 11(1), 41–51. https://doi.org/10.4236/OJE.2021.111004
Schulhof, M. A., Shurin, J. B., Declerck, S. A. J., & Van de Waal, D. B. (2019). Phytoplankton growth and stoichiometric responses to warming, nutrient addition and grazing depend on lake productivity and cell size. Global Change Biology, 25(8), 2751–2762. https://doi.org/10.1111/gcb.14660
Shalaby, S. E. M., El-Saadany, S. S., Abo-Eyta, A. M., Abdel-Satar, A. M., Al-Afify, A. D. G., & Abd El-Gleel, W. M. M. (2018). Levels of pesticide residues in water, sediment, and fish samples collected from Nile River in Cairo, Egypt. Environmental Forensics, 19(4), 228–238. https://doi.org/10.1080/15275922.2018.1519735
Shalloof, K. A. S., El-Far, A. M., & Aly, W. (2020). Feeding habits and trophic levels of cichlid species in tropical reservoir, Lake Nasser, Egypt. Egyptian Journal of Aquatic Research, 46(2), 159–165. https://doi.org/10.1016/j.ejar.2020.04.001
Sun, D., Chen, Y., Feng, Y., Liu, Z., Peng, X., Cai, Y., et al. (2021). Seasonal variation in size diversity: Explaining the spatial mismatch between phytoplankton and mesozooplankton in fishing grounds of the East China Sea. Ecological Indicators, 131, 108201. https://doi.org/10.1016/J.ECOLIND.2021.108201
Taha, L. G. E. D. (2014). Assessment of urbanization encroachment over Al-Monib island using fuzzy post classification comparison and urbanization metrics. Egyptian Journal of Remote Sensing and Space Science, 17(2), 135–147. https://doi.org/10.1016/j.ejrs.2014.08.002
Temesgen, M., Getahun, A., Lemma, B., & Janssens, G. P. J. (2022). Food and feeding biology of Nile tilapia (Oreochromis niloticus) in Lake Langeno, Ethiopia. Sustainability, 14, 974. https://doi.org/10.3390/su14020974
Vollenweider, R. A. (2000). Concerning calculation methods and limitations of proxy-estimates of proteins, carbohydrates and lipids in crustacean zooplankton from CHN analyses. Journal of Limnology, 59(2), 170. https://doi.org/10.4081/jlimnol.2000.170
Wagner, H., Jakob, T., Fanesi, A., & Wilhelm, C. (2017). Towards an understanding of the molecular regulation of carbon allocation in diatoms: The interaction of energy and carbon allocation. Biological Sciences. Royal Society Publishing. https://doi.org/10.1098/rstb.2016.0410
WHO. (2006). Guidelines for drinking-water quality - World Health Organization - Google Books (3rd ed., vol. 1).
Yacoub, A. M., Mahmoud, S. A., & Abdel-Satar, A. M. (2021). Accumulation of heavy metals in tilapia fish species and related histopathological changes in muscles, gills and liver of Oreochromis niloticus occurring in the area of Qahr El-Bahr, Lake Al-Manzalah, Egypt. Oceanological and Hydrobiological Studies, 50(1), 1–15. https://doi.org/10.2478/OANDHS-2021-0001
Yang, Y., Cao, J. X., Pei, G. F., & Liu, G. X. (2015). Using benthic diatom assemblages to assess human impacts on streams across a rural to urban gradient. Environmental Science and Pollution Research, 22(22), 18093–18106. https://doi.org/10.1007/s11356-015-5026-1
Zakaria, H. Y., Hassan, A. M., El-Naggar, H. A., & Abou-Senna, F. M. (2016). Abundance, distribution, diversity and zoogeography of epipelagic copepods off the Egyptian coast (Mediterranean Sea). The Egyptian Journal of Aquatic Research, 42(4), 459–473.
Zakaria, H. Y., Hassan, A. M., El-Naggar, H. A., & Abou-Senna, F. M. (2018). Planktonic protozoan population in the Southeastern Mediterranean off Egypt. The Egyptian Journal of Aquatic Research, 44, 101–107.
Zarrinmehr, M. J., Farhadian, O., Heyrati, F. P., Keramat, J., Koutra, E., Kornaros, M., & Daneshvar, E. (2020). Effect of nitrogen concentration on the growth rate and biochemical composition of the microalga, Isochrysis galbana. Egyptian Journal of Aquatic Research, 46(2), 153–158. https://doi.org/10.1016/j.ejar.2019.11.003
Acknowledgements
This study was produced as a part of the program of the National Institute of Oceanography and Fisheries (NIOF) entitled “Ecological and Fisheries of River Nile.” Many thanks to Dr. Walid Aly for his help in the analysis of statistical data.
Funding
Open access funding provided by The Science, Technology & Innovation Funding Authority (STDF) in cooperation with The Egyptian Knowledge Bank (EKB).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Abdelmageed, A.A., Ellah, R.G.A., Abdel-Satar, A.M. et al. Evaluation of the ecological health and food chain on the shores of four River Nile Islands, Egypt. Environ Monit Assess 194, 309 (2022). https://doi.org/10.1007/s10661-022-09959-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10661-022-09959-w