Abstract
Hepatic fibrosis is one of the major worldwide health concerns which requires tremendous research due to the limited outcomes of the current therapies. The present study was designed to assess, for the first time, the potential therapeutic effect of rupatadine (RUP) in diethylnitrosamine (DEN)-induced liver fibrosis and to explore its possible mechanistic actions. For the induction of hepatic fibrosis, rats were treated with DEN (100 mg/kg, i.p.) once weekly for 6 consecutive weeks, and on the 6th week, RUP (4 mg/kg/day, p.o.) was administered for 4 weeks. Treatment with RUP ameliorated changes in body weights, liver indices, liver function enzymes, and histopathological alterations induced by DEN. Besides, RUP amended oxidative stress, which led to the inhibition of PAF/NF-κB p65-induced inflammation, and, subsequently, prevention of TGF-β1 elevation and HSCs activation as indicated by reduced α-SMA expression and collagen deposition. Moreover, RUP exerted significant anti-fibrotic and anti-angiogenic effects by suppressing Hh and HIF-1α/VEGF signaling pathways. Our results highlight, for the first time, a promising anti-fibrotic potential of RUP in rat liver. The molecular mechanisms underlying this effect involve the attenuation of PAF/NF-κB p65/TGF-β1 and Hh pathways and, subsequently, the pathological angiogenesis (HIF-1α/VEGF).
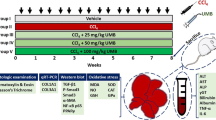
Graphical abstract

Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Liver fibrosis is a serious worldwide health problem that may progress to cirrhosis, hepatocellular carcinoma (HCC), and death. It is commonly caused by various factors like viral infections, malnutrition, alcohol abuse, biliary atresia, and hepatotoxins (Weber et al. 2016). Following chronic damage, hepatic stellate cells (HSCs) become stimulated and transdifferentiated into myofibroblast-like cells, secreting large amounts of extracellular matrix (ECM), which is linked to alpha-smooth muscle actin (α-SMA) and collagen production (Zhao et al. 2014; Cheng et al. 2018). This process impairs the material exchange between hepatocyte and hepatic sinusoidal blood (Wu et al. 2016).
Among the potent environmental hepatotoxins which induce liver fibrosis is diethylnitrosamine (DEN) (Jose et al. 1998). Liao et al. (2001) reported that DEN is found in numerous kinds of food, such as cheese, soybean, salted fish, and cured meat, in addition to alcoholic beverages. Experimentally, this model closely resembles human liver fibrosis in cellular alterations and histologic patterns (Jin et al. 2010). During its metabolic biotransformation, DEN generates reactive oxygen species (ROS), resulting in the activation of platelet-activating factor (PAF) and cellular injury (Travers 1999).
PAF is a potent lipid mediator of the inflammatory response and allergy (Borthakur et al. 2010) through binding to its G-protein-coupled receptor in hepatocytes (Karantonis et al. 2010), increasing the secretion of a variety of cytokines (Sugano et al. 2001), and leading to progression of hepatic inflammation that proceeds to fibrosis (Farombi et al. 2009). Growing evidence shows that nuclear factor-kappa B (NF-κB) stimulates fibrogenesis by releasing several growth factors like transforming growth factor-beta 1 (TGF-β1), the most potent fibrogenic stimulant for HSCs, which induces epithelial–mesenchymal transition and collagen deposition (Friedman 1999; Inagaki and Okazaki 2007).
Zhang et al. (2017) unveiled that hedgehog (Hh) signaling has been involved in the pathologic mechanisms of hepatic fibrosis. Aberrant activation of Hh signaling is reported in hepatic regeneration, vascular remodeling, fibrosis, and HCC (Omenetti et al. 2011; Zhang et al. 2017). Hypoxia is one of the main driving forces of angiogenesis where hypoxia-inducing factor-1alpha (HIF-1α), the downstream molecule of the Hh pathway, upregulates the expression of vascular endothelial growth factor (VEGF) (Feng et al. 2019). This factor plays an essential role in angiogenesis, HSCs activation, and, finally, fibrosis (Cai et al. 2021). Thus, the prevention of pathological angiogenesis can successfully inhibit the progression of liver fibrosis (Kantari-Mimoun et al. 2015).
Although studies on the pathophysiology of liver fibrosis have revealed significant progress, managing liver fibrosis is still a major health issue (Zhao et al. 2017), which requires continuous searches for new and effective therapeutic strategies (Tsochatzis et al. 2014). Rupatadine (RUP) is an approved second-generation antihistamine drug that demonstrates anti-inflammatory properties in addition to its ability to block both PAF and histaminic receptors (González-Núñez et al. 2016). Few studies have revealed its anti-fibrotic effect, where RUP ameliorated silica-induced pulmonary fibrosis (Lv et al. 2013) and isoprenaline-induced myocardial fibrosis experimentally (Ahmed et al. 2021). Most importantly, a recent study demonstrated its hepatoprotective effect against acute toxicity induced by 5-fluorouracil (5-FU) (Khalaf et al. 2022). Moreover, RUP inhibited ulcerative colitis in rats through modulation of PAF/IL-6/VEGF signaling (Ibrahim et al. 2022). Therefore, the goal of the present study was to assess the role of RUP in DEN-induced liver fibrosis and its actions on PAF/NF-κB p65/TGF-β1 and Hh/HIF-1α/VEGF signaling pathways.
Materials and methods
Animals
Eighteen adult male Wistar rats (8–10 weeks old), weighing 200–230 g, were purchased from the animal house of the National Research Center, Cairo, Egypt. The rats were left for one week to be accustomed at the animal house of the Faculty of Pharmacy, Cairo University, Egypt. The animals had unrestricted access to a standard diet and water throughout the experimental period. They were subjected to a controlled environment with a 12-h light/dark cycle, 23 ± 2 °C temperature, and relative humidity (60% ± 10%). The study was accepted by the Cairo University Faculty of Pharmacy Ethics Committee for Animal Experimentation [Permit Number: PT (3012)], which complies with the Guide for Care and Use of Laboratory Animals published by the US National Institutes of Health (NIH Publication, No. 85-23, revised 2011).
Chemicals and drugs
DEN was bought from Sigma-Aldrich (St. Louis, MO, USA) and diluted in normal saline, then administered to rats intraperitoneally (i.p.) using a 23-gauge needle. RUP was obtained from Mash Premiere (Badr, Egypt), dissolved in saline, and orally (p.o.) administered using a 16-gauge round-tipped gavage p.o. needle. Unless otherwise noted, all additional chemicals were obtained from Sigma-Aldrich (St. Louis, MO, USA).
Experimental design
Rats were randomly distributed into 3 groups (6 rats/group). The 1st group served as the normal group, which received saline intraperitoneally (once/week) for 6 weeks, followed by administration daily of oral saline for 4 successive weeks. Rats in the 2nd group received DEN (100 mg/kg, i.p) (MadanKumar et al. 2014; Essam et al. 2019) at a dose volume of 2.5 ml/kg (once/week) for 6 weeks, followed by oral administration of saline daily for 4 weeks. The 3rd group received DEN in the same regimen as group 2, followed by RUP (4 mg/kg/day, p.o.) (Lv et al. 2013; Ahmed et al. 2021), starting from the 6th to the 10th week. All animals were weighed at the start and end of the experiment. By the end of treatment, all rats were anesthetized using thiopental (50 mg/kg, i.p.). Blood samples were then obtained through the retro-orbital plexus using a sterile, non-heparinized capillary tube to separate the serum. Meanwhile, rats were sacrificed under anesthesia by cervical dislocation. Part of the liver tissues was homogenized, divided into aliquots, and frozen at − 80 °C to determine the biochemical parameters. The remaining parts were separated and sent off for histology and quantitative reverse transcription polymerase chain reaction (qRT-PCR) analyses.
Histopathological investigation
Determination of the degree and area of hepatic fibrosis
The liver sections (5 µm) were stained with hematoxylin and eosin (H&E) for investigation of the morphological fibrogenic scores in accordance with the Ishak scoring system after being fixed in 10% formalin and embedded in paraffin wax (Ishak et al. 1995). Additionally, sections were stained with Masson’s trichrome (Sigma-Aldrich, St. Louis, MO, USA) to reveal collagen fibers in hepatic tissues using image analysis software (ImageJ; Maryland, USA). A pathologist performed all histopathological changes in a blinded manner.
Immunohistochemical staining of NF-ĸB (p65) and α-SMA
Deparaffinized liver sections (5 μm) were serially dehydrated in ethanol, then treated with 5% hydrogen peroxide for 10 min. to deactivate the endogenous peroxidase. The slides were blocked for 2 h with 5% BSA in tris-buffered saline (TBS). The sections were immunostained with one of the subsequent primary antibodies: rabbit polyclonal anti-rat NF-κB p65 (Thermo Scientific, USA; Cat. #RB-9034-R7) or rabbit α-SMA polyclonal antibody to rat (ElabScience Biotechnology Inc, USA; Cat. #E-AB-34268) at a concentration of 1 μg/mL and incubated overnight at 4 °C. After rinsing slides with TBS, the sections were incubated in a solution of 0.02% diaminobenzidine containing 0.01% H2O2 for 10 min. The slides were examined using a light microscope (Olympus microscope BX-53, Crop, Tokyo, Japan) after counterstaining with hematoxylin, where the wall of blood vessels serves as an internal positive control for α-SMA (Bridle et al. 2009). Immunohistochemical quantification was carried out by assessing the percentage of the immunopositive area as indicated by the intense brown staining using image analysis software (ImageJ; Maryland, USA).
Biochemical assessments
Tests of liver function
Commercial kits (Biodiagnostics, Giza, Egypt) were utilized to measure the activities of aspartate aminotransferase (AST) and alanine aminotransferase (ALT) in the separated serum in accordance with the manufacturer’s instructions.
Enzyme-linked immunosorbent assay (ELISA) technique for MDA, SOD, PAF, TGF-β1, HIF-1α, and VEGF evaluation
The corresponding ELISA kits were used to assess MDA (LifeSpan Biosciences; WA; USA; Cat.# LS-F28018), SOD (MyBioSource; CA, USA; Cat.# MBS036924), PAF (MyBioSource; CA, USA; Cat.# MBS2024376), TGF-β1 (MyBioSource; CA, USA; Cat.# MBS011634), HIF-1α (Kamia Biomedical; Seattle, USA; Cat.# KT-17920) and VEGF (MyBioSource; CA, USA; Cat.# MBS8506132). All steps were carried out in accordance with the manufacturers’ instructions. For the determination of protein content, the method of Lowry et al. (1951) was performed.
Quantitative RT-PCR analysis of Hh molecules [patched-1 receptor (Ptch1), glioma-associated oncogene homolog2 (Gli2) and glioma-associated oncogene homolog1 (Gli1)]
In brief, SV total RNA isolation system (Promega, USA) was used to extract total RNA for Hh markers. The Reverse Transcription System (Promega, USA) was then used to reverse transcribe the RNA into complementary DNA in accordance with the manufacturer’s instructions. Next, qRT-PCR was performed using SYBR Green JumpStart Taq ReadyMix (Sigma-Aldrich, USA). The primer sequences utilized for the assay are listed in Table 1. Following qRT-PCR, the 2−∆∆CT formula was used to determine the relative expression of the target genes (Livak and Schmittgen 2001), where β-actin served as the housekeeping gene.
Statistical analysis
The mean ± standard error mean (SEM) was used to express the results. One-way analysis of variance (ANOVA) was then used, followed by Tukey’s post hoc multiple comparison test to interpret the data. For the histopathological scores’ evaluation, the nonparametric Kruskal–Wallis test (one-way ANOVA) and Dunn’s multiple comparison test were used to examine the results, which are expressed as median (min–max). All statistical tests and sketching of the graphs were carried out using the GraphPad Prism® software package, ver. 8 (GraphPad Software, Inc., USA). The level of significance for each statistical test was performed at p ˂ 0.05.
Results
RUP treatment amended DEN-induced changes in liver indices and liver function tests in rats
Administration of DEN led to a significant reduction in the final body weight and a significant increase in liver indices (liver weight/body weight %) compared to the normal rats. Co-treatment with RUP normalized body weight as well as liver indices (Table 2). Regarding the liver enzymes, serum ALT and AST activities were significantly elevated in the DEN group by approximately fourfold and threefold, respectively, with respect to the normal group. Conversely, rats treated with RUP unveiled a significant diminish in the serum ALT and AST activities by 57% and 55%, respectively, compared to the DEN group despite being still significantly higher than the normal values (Table 2).
RUP treatment curbed DEN-induced deterioration in the hepatic histology
According to Fig. 1A, the liver’s photomicrographs revealed the normal hepatic architecture of the central vein as well as surrounding hepatocytes in normal rats. Still, those present in the DEN group demonstrated hepatic nodules due to the formation of long fibrous connective tissue septa with infiltration of the portal area by mononuclear inflammatory cells infiltration and the presence of vacuolar degeneration in hepatocytes (Fig. 1B). On the contrary, RUP treatment revealed a marked amelioration in portal fibrosis by decreasing number of mononuclear inflammatory cells infiltration in a portal area with the disappearance of hepatic nodules and formation of short connective tissue septa (Fig. 1C).
RUP treatment improved the histopathological deterioration induced by DEN. A–D Histopathological analysis of rat liver sections using H&E staining (× 400). A Normal group exhibited normal histological structure liver. B DEN group revealed portal infiltration with mononuclear inflammatory cells (star), vacuolar degeneration in hepatocytes (arrowhead), and formation of long connective tissue septa led to nodular formation (arrow). C RUP group showed portal infiltration with low numbers of mononuclear inflammatory cells (black arrow) and the formation of short connective tissue septa (red arrow). These changes are scored in panel D, where each value represents the median of 4 experiments (min–max). Statistical analysis was carried out using the Kruskal–Wallis test (one-way ANOVA) followed by Dunn’s multiple comparison test, as compared to normal (*) and DEN (@) groups at p ˂ 0.05. DEN diethylnitrosamine, H&E hematoxylin and eosin, RUP rupatadine
RUP treatment attenuated DEN-induced oxidative stress
SOD activity was reduced by 81% in the DEN group; meanwhile, a significant elevation of the hepatic lipid peroxidation product (MDA) (about sixfold) was obvious compared to the normal group. RUP-treated rats showed about a threefold elevation of SOD activity together with a significant reduction of hepatic MDA content by 58%, compared to the DEN group (Table 3).
RUP treatment ameliorated DEN-induced hepatic inflammatory events
DEN exposure led to an upsurge of the inflammatory response (Fig. 2), as reflected by a marked increment of immunohistochemical NF-ĸB p65 expression (Fig. 2A–D) to 15% as well as a significant elevation of the hepatic PAF content to approximately threefold (Fig. 2E). On the other hand, co-treatment with RUP significantly attenuated these effects.
RUP treatment decreased the hepatic inflammation induced by DEN. A–C Immunohistochemical detection of NF-κB p65 (brown stain) (× 400). A Normal group showed no NF-κB expression. B DEN group revealed a significant increase in NF-κB p65 expression in nuclei and cytoplasm of hepatocytes (arrow). C RUP group demonstrated less NF-κB expression in nuclei of hepatocytes (arrow). D Quantitative image analysis of NF-κB p65 immunoreactivity (% area). E Effect of RUP on the hepatic PAF content as determined using a commercial ELISA kit. Values are represented as mean (n = 4 for histological examination and n = 6 for PAF estimation) ± SEM. Statistical analysis was done using one-way ANOVA followed by Tukey’s post hoc test. As compared with normal (*) and DEN (@) at p ˂ 0.05. DEN diethylnitrosamine, ELISA: enzyme-linked immunosorbent assay, NF-κB p65 nuclear factor-kappa B p65, PAF platelet-activating factor, RUP rupatadine
RUP treatment mitigated DEN-induced hepatic fibrogenesis
According to Fig. 3, which depicts the immunohistochemical detection of α-SMA, a significant amount of brown staining (11.22% of the immunopositive area) was revealed in the DEN group. On the other hand, RUP significantly decreased the positively stained area of α-SMA. Moreover, RUP’s anti-fibrotic effect was further proved by the histological visualization of collagen fibers using Masson’s trichrome stain, where the increase in collagen deposition induced by DEN was markedly alleviated by RUP as clarified in Fig. 3D–F. Additionally, ELISA analysis revealed a higher hepatic TGF-β1 content (about fourfold) in the DEN group than in the normal group. RUP treatment demonstrated a significant decrease in TGF-β1 content by 54%, compared with the DEN group Fig. 3I.
RUP treatment decreased the hepatic fibrosis induced by DEN. A–C Immunohistochemical detection of α-SMA (× 400) as an indicator for HSCs activation (brown color). A Normal group showed an absence of α-SMA expression in the hepatic parenchyma. B DEN group revealed extensive α-SMA expression in the portal area, indicating enormous numbers of activated HSCs (arrow). C RUP group clarified mild α-SMA expression in hepatic parenchyma (arrow). D–F Specimens stained with Masson’s trichrome to assess hepatic fibrosis (blue color). D Normal group showed a normal histological picture of the liver, E the DEN group showed severe portal fibrosis (arrowhead), and F the RUP group showed only short connective tissue septa between two portal areas (arrow). G Quantitative image analysis for α-SMA (% area). H Image analysis of % of fibrosis for Masson’s trichrome specimens. I Effect of RUP on the hepatic TGF-β1 content was determined using the relevant ELISA kit. Values are represented as mean (n = 4 for histological examination and n = 6 for TGF-β1 estimation) ± SEM. Statistical analysis was done using one-way ANOVA followed by Tukey’s post hoc test. As compared with normal (*) and DEN (@) at p ˂ 0.05. α-SMA alpha smooth muscle actin, DEN diethylnitrosamine, ELISA enzyme-linked immunosorbent assay, RUP rupatadine, TGF-β1 transforming growth factor-beta1
RUP treatment suppressed the extensive activation of Hh trajectory induced by DEN
As illustrated in Fig. 4, DEN turned on the Hh pathway, as revealed by the significant increase in Ptch1, Gli2, and Gli1 mRNA expressions to approximately nine-, six-, and five-fold, respectively, compared with the normal group. Strikingly, co-treatment with RUP markedly curtailed the genetic expressions of Ptch1, Gli2, and Gli1 by 57%, 57%, and 51%, respectively, compared with the DEN group, confirming the inhibitory effect of RUP on the Hh pathway.
RUP treatment blocked the Hh trajectory induced by DEN administration, as revealed by its effects on mRNA expression of A Ptch1, B Gli2, and C Gli1. Values are represented as mean (n = 6) ± SEM. Statistical analysis was done using one-way ANOVA followed by Tukey’s post hoc test. As compared with normal (*) and DEN (@) at p ˂ 0.05. DEN diethylnitrosamine, Gli1 glioma-associated oncogene homolog1, Gli2 glioma-associated oncogene homolog2, Ptch1 patched-1 receptor, RUP rupatadine
RUP treatment inhibited the activation of the HIF-1α/VEGF pathway
DEN-treated group markedly increased the hepatic contents of HIF-1α and VEGF by approximately fourfold (Fig. 5). Treatment with RUP signified its action on the pathological angiogenesis by diminishing HIF-1α and VEGF contents by 64% and 58%, respectively, compared to the DEN-treated group.
RUP treatment inhibited the activation of the HIF-1α/VEGF pathway induced by DEN administration, as revealed by its effects on hepatic contents of A HIF-1α and B VEGF. Values are represented as mean (n = 6) ± SEM. Statistical analysis was done using one-way ANOVA followed by Tukey’s post hoc test. As compared with normal (*) and DEN (@), at p ˂ 0.05. DEN diethylnitrosamine, HIF-1α hypoxia inducing factor-1alpha, RUP rupatadine, VEGF vascular endothelial growth factor
Discussion
Hepatic fibrosis is a complex universal health problem with significant morbidity and mortality rates worldwide due to its fast development into cirrhosis and HCC (Ogaly et al. 2022). Upon hepatic fibrosis, the dormant HSCs transdifferentiate into myofibroblast-like cells with loss of vitamin A lipid droplets, augmented α-SMA expression, and overproduction of ECM components such as collagens, proteoglycans, and glycoproteins (Perumal et al. 2017).
In the current study, the liver fibrosis model was induced by DEN. This model closely mimics the human liver fibrosis’s cellular alterations and histologic features (Jin et al. 2010). DEN-induced liver fibrosis causes loss and rupture of hepatocyte membrane functional integrity, which speeds up the leakage of liver enzymes (ALT and AST) into the bloodstream (Jin et al. 2019). These markers are usually considered direct sensitive biomarkers for evaluating hepatocellular damage (Moreira et al. 2015). Similarly, the histopathological findings of DEN-treated rats provided additional evidence of liver injury and demonstrated significant deviations from the normal patterns. These changes involved severe portal fibrosis with the nodular formation and high numbers of mononuclear inflammatory cell infiltration. Notably, these findings concur with earlier research (Essam et al. 2019).
Treatment with RUP exerted notable therapeutic effects against DEN-elicited fibrosis, as evidenced by significant suppression of ALT and AST activities. Histopathological examination of RUP showed a remarkable improvement and alleviation of portal fibrosis and mononuclear inflammatory cell infiltration compared to the DEN group. In the same context, Khalaf et al. (2022) showed that the RUP provided normal liver architecture and a significant decrease in ALT and AST activities in an experimental model of 5-FU-induced acute hepatotoxicity in rats. RUP could stabilize the membrane structure of hepatocytes, thereby preventing the cellular damage induced by DEN.
In the present study, body weight loss as well as increasing in liver weight index were observed in DEN-exposed rats during the experimentation period. Noticeably, the reduction in body weight could be due to the diminished appetite and decreased food intake caused by DEN (Farazi and DePinho 2006). The increase in liver weight index could also be related to the provoked inflammation in hepatotoxin-exposed animals with an accumulation of ECM and inflammatory cell infiltration (El-Mihi et al. 2017). Rodents treated with RUP preserved their body weights and showed marked suppression in liver indices compared to the DEN group. These results support the histopathological and biochemical results observed in the RUP group.
The present findings indicate that RUP has potent anti-oxidant and free radical scavenging activities, as evidenced by a significant decrease in the hepatic MDA content and an increase in SOD activity compared to the DEN group. It is well-recognized that oxidative stress increases collagen deposition, contributing to liver fibrosis development (Yang et al. 2019). Growing evidence has demonstrated that DEN is metabolized by cytochrome P450 2E1, elevating the production of ROS (Essam et al. 2019), enhancing lipid peroxidation, causing further damage and overwhelming the anti-oxidant enzymatic (such as SOD) and non-enzymatic defense systems. All these consequences contribute to cellular damage (Motawi et al. 2011). RUP previously alleviated ROS-induced hepatocyte and intestinal apoptosis (Mohamed and Mohammed 2021). Besides, the anti-oxidant activity of RUP was demonstrated in rats with acute pancreatitis and diabetic nephropathy as well as in allergic rhinitis in patients (Hafez et al. 2020; Kahveci et al. 2021; Mohamed et al. 2022). Based on these findings, the anti-oxidant activity of RUP could be postulated to prevent ROS-induced fibrosis.
In the current investigation, DEN intoxication disrupted the redox balance in favor of increased ROS, which increases the hepatic content of PAF, as demonstrated earlier by Hwa Choi et al. (2000) and Ajiboye et al. (2013). On the other hand, RUP significantly prevented PAF activation, mainly through the inhibition of oxidative stress. PAF is an upstream regulator and a proximal stimulator of NF-κB. The activation of PAF stimulates the activation and nuclear translocation of NF-κB p65, leading to vascular smooth muscle cell proliferation (Ibe et al. 2008; Ogbozor et al. 2015). Additionally, NF-κB increases the expression of various pro-inflammatory cytokines like tumor necrosis factor-alpha (TNF-α) that induce further stimulation of NF-κB level, provoking the inflammatory process (Sasaki and Iwai 2016). Pro-inflammatory mediators also increase phospholipase A2 synthesis, transforming lysophospholipids to PAF (Pfeilschifter et al. 1993; Tran et al. 2018). Importantly, the oxidative stress ameliorated by RUP could contribute mainly to preventing this inflammatory status by inhibiting NF-κB (Köhler et al. 2016; Herpers et al. 2016; Wang et al. 2018). In the same context, Mohamed and Mohammed (2021) showed that RUP suppressed PAF content and TNF-α/NF-κB p65 pathway in 5-FU-induced intestinal toxicity of rats. Moreover, RUP notably repressed macrophage and mast cell infiltration and inhibited expression of TNF-α and mast cell degranulation in a rat model of lung fibrosis (Lv et al. 2013). Similarly, our findings showed that RUP inhibited the hepatic PAF content, suppressed NF-κB p65 expression, and prevented the subsequent inflammatory cascade, providing evidence for the anti-inflammatory effect of RUP.
Besides upsurging the inflammation process, NF-κB plays a significant role in activating HSCs (Elsharkawy and Mann 2007; El-Agroudy et al. 2016) by releasing several growth factors like TGF-β1. TGF-β1 regulates the expression of α-SMA, a reliable marker of HSCs activation (Desmouliere et al. 1993; Abdel-Rahman et al. 2019). TGF-β1 also induces fibrosis by supporting the differentiation of inactive fibroblasts into matrix-secreting myofibroblasts (Vaughan et al. 2000; Lijnen et al. 2003) and increasing the synthesis of collagen (Epstein et al. 1994; Koh et al. 2015) in the ECM, exacerbating hepatic fibrosis (George et al. 2019). In this study, the hepatic content of TGF-β1 and α-SMA immunostaining were significantly perturbated in hepatic tissues of DEN-treated rats, which concurred with the results of earlier studies (Perumal et al. 2017; Cheng et al. 2018; Husain et al. 2018). These data were also confirmed by Masson’s trichrome staining, which revealed a massive deposition of collagen fibers in DEN-exposed rats.
Based on its obvious anti-oxidant and anti-inflammatory effects, RUP treatment significantly decreased the hepatic content of TGF-β1, inhibiting HSCs activation as shown by reduced α-SMA expression. Following the same pattern, the inhibition of ROS was previously shown to diminish TGF-β1-spurred α-SMA expression in rat hepatic fibroblast (Yao et al. 2012; Calleja et al. 2013). NF-κB inhibition also formerly abated the elevation of TGF-β1 and its mediated HSCs activation (Mao et al. 2015). The amelioration of these inflammatory and fibrogenic markers by RUP was confirmed by evident and marked amelioration of the degree of liver fibrosis as indicated by the significant reduction of α-SMA expression and collagen deposition. These results are consistent with previous research demonstrating the anti-fibrogenic potential of RUP in models of pulmonary and cardiac fibrosis (Lv et al. 2013; Ahmed et al. 2021).
Our findings revealed for the first time that RUP is a potent Hh cascade inhibitor, as shown by the marked reduction in expressions of Ptch1, Gli2, and Gli1. Hh pathway has been implicated in the progression of liver fibrosis (Sicklick et al. 2005; Guy et al. 2012; Yang et al. 2014). In normal liver tissues, this axis is inert where Hh ligands are absent (Omenetti et al. 2011). Meanwhile, in liver fibrosis, the activated HSCs induced by TGF-β1 are capable of producing Hh ligands that bind to their patched receptor (Choi et al. 2011; Feng et al. 2019), releasing smoothened transmembrane protein (SMO) which stimulates the activation and nuclear translocation of the Gli transcription factors (Jiayuan et al. 2020). Among these factors, Gli2 induces Gli1 activation, which serves again as a signal amplifier downstream of Gli2 (Kramann 2016), triggering the signaling of a plethora of target genes involved in myofibroblast formation such as α-SMA and TGF-β1. TGF-β1 is a potent inducer of Gli2 transcription (Syn et al. 2009; Choi et al. 2010) and Hh pathway (Jiayuan et al. 2020). In the same context, the current results reveal the correlation between the hepatic fibrosis prompted by DEN and the marked stimulation of Hh signaling, as clarified by the amplified gene expressions of Ptch1 as well as Gli1 and Gli2.
RUP principally attenuated Hh signaling by inhibiting TGF-β1 content, whereas the latter inhibition is known to turn off Hh signaling (Mansour et al. 2021). The inhibition of the Hh pathway downregulates the myofibroblast-related genes and causes the reversal of myofibroblasts to a quiescent phenotype, thus attenuating fibrosis (Pratap et al. 2012). GANT61, a Gli inhibitor, consistently proved an anti-fibrotic effect against CCl4-induced hepatic fibrosis in mice (Jiayuan et al. 2020). Inhibition of the Hh hub with vismodegib, a SMO antagonist, also stimulated the regression of both hepatic fibrosis and HCC (Philips et al. 2011).
Several studies highlighted the inhibition of hepatic angiogenesis as a novel target for treating hepatic fibrosis (Zhou et al. 2014; Feng et al. 2019). In the present study, the anti-angiogenic activities of RUP in the DEN model were confirmed by significant suppression of the measured HIF-1α/VEGF signaling which was markedly elevated in the DEN group. Noteworthy, the hypoxia in the fibrotic liver could be attributed to the sinusoids’ structural changes, including basement membrane deposition and loss of liver sinusoidal endothelial cells (LSECs) fenestrae. These changes lead to impaired oxygen diffusion from the sinusoids to the parenchyma (Rosmorduc and Housset 2010), where hypoxia initiates an intracellular pathway leading to the activation of the transcription factor HIF-1α. Additionally, several factors, such as ROS and inflammatory cytokines, could activate HIF-1α accumulation (Ushio-Fukai and Alexander 2004; Bonello et al. 2007; Moczydlowska et al. 2017). Noteworthy, in liver fibrosis, Gli1 has been clarified as a promoter for HIF-1α (Zhang et al. 2017; Yang et al. 2020). HIF-1α also binds to hypoxia-responsive elements found in the promoter of several target genes, including VEGF (Forsythe et al. 1996; Bozova and Elpek 2007), where VEGF directly stimulates LSECs and provokes angiogenesis together with activation of HSCs (Yin et al. 2013). HSCs also express several CXC chemokines, which can manipulate angiogenesis (Hickey and Simon 2006), supporting the positive correlation of angiogenesis with fibrogenesis (Feng et al. 2019). Similar to our RUP results, curcumin inhibited liver fibrosis in rats by suppressing HIF-1α/VEGF-induced angiogenesis (Zhang et al. 2014). As well, Ibrahim et al. (Ibrahim et al. 2022) reported that RUP downregulated VEGF content in the ulcerative colitis model via inhibition of PAF. PAF could stimulate angiogenesis by releasing NF-ĸB- dependent angiogenic factors such as VEGF (JH et al. 2001).
Conclusion
In summary, the present study may satisfy an unmet need in developing a therapeutic agent for the treatment of hepatic fibrosis. RUP is a safely prescribed drug that demonstrated a favorable anti-fibrotic effect in rat liver for the first time in the current study. The molecular mechanisms underlying this protective effect involve attenuating oxidative stress, inflammation, and fibrosis by inhibiting PAF/NF-κB p65/TGF-β1 axis. More importantly, RUP inhibited liver fibrosis not only through suppression of HSCs activation and ECM deposition but also through Hh pathway impediment as well as its anti-angiogenic effect via inhibition of HIF-1α/VEGF trajectory. Further experimental studies are required to confirm the anti-fibrotic potential of RUP in the liver and to elucidate other underlying mechanisms before starting any clinical trials.
Data availability statement
The data sets generated during and/or analyzed during the current study are available from the corresponding author upon reasonable request.
References
Abdel-Rahman N, Sharawy MH, Megahed N, El-Awady MS (2019) Vitamin D3 abates BDL-induced cholestasis and fibrosis in rats via regulating Hedgehog pathway. Toxicol Appl Pharmacol. https://doi.org/10.1016/J.TAAP.2019.114697
Ahmed LA, Mohamed AF, Abd El-Haleim EA, El-Tanbouly DM (2021) Boosting Akt pathway by rupatadine modulates Th17/Tregs balance for attenuation of isoproterenol-induced heart failure in rats. Front Pharmacol. https://doi.org/10.3389/FPHAR.2021.651150
Ajiboye TO, Komolafe YO, Oloyede HOB et al (2013) Diethylnitrosamine-induced redox imbalance in rat microsomes: protective role of polyphenolic-rich extract from Sorghum bicolor grains. J Basic Clin Physiol Pharmacol 24:41–49. https://doi.org/10.1515/JBCPP-2012-0031
Bonello S, Zähringer C, BelAiba RS et al (2007) Reactive oxygen species activate the HIF-1α promoter via a functional NFκB site. Arterioscler Thromb Vasc Biol 27:755–761. https://doi.org/10.1161/01.ATV.0000258979.92828.BC
Borthakur A, Bhattacharyya S, Alrefai WA et al (2010) Platelet-activating factor-induced NF-κB activation and IL-8 production in intestinal epithelial cells are Bcl10-dependent. Inflamm Bowel Dis 16:593–603. https://doi.org/10.1002/IBD.21092
Bozova S, Elpek GÖ (2007) Hypoxia-inducible factor-1α expression in experimental cirrhosis: correlation with vascular endothelial growth factor expression and angiogenesis. APMIS 115:795–801. https://doi.org/10.1111/J.1600-0463.2007.APM_610.X
Bridle KR, Popa C, Morgan ML et al (2009) Rapamycin inhibits hepatic fibrosis in rats by attenuating multiple profibrogenic pathways. Liver Transplant 15:1315–1324. https://doi.org/10.1002/LT.21804
Cai J, Hu M, Chen Z, Ling Z (2021) The roles and mechanisms of hypoxia in liver fibrosis. J Transl Med 19:1–13. https://doi.org/10.1186/S12967-021-02854-X/FIGURES/2
Calleja MA, Vieites JM, Montero-Meterdez T et al (2013) The antioxidant effect of β-caryophyllene protects rat liver from carbon tetrachloride-induced fibrosis by inhibiting hepatic stellate cell activation. Br J Nutr 109:394–401. https://doi.org/10.1017/S0007114512001298
Cheng Y, Zheng H, Wang B et al (2018) Sorafenib and fluvastatin synergistically alleviate hepatic fibrosis via inhibiting the TGFβ1/Smad3 pathway. Dig Liver Dis 50:381–388. https://doi.org/10.1016/J.DLD.2017.12.015
Choi SS, Witek RP, Yang L et al (2010) Activation of Rac1 promotes hedgehog-mediated acquisition of the myofibroblastic phenotype in rat and human hepatic stellate cells. Hepatology 52:278–290. https://doi.org/10.1002/HEP.23649
Choi SS, Omenetti A, Syn WK, Diehl AM (2011) The role of Hedgehog signaling in fibrogenic liver repair. Int J Biochem Cell Biol 43:238–244. https://doi.org/10.1016/J.BIOCEL.2010.10.015
Desmouliere A, Geinoz A, Gabbiani F, Gabbiani G (1993) Transforming growth factor-beta 1 induces alpha-smooth muscle actin expression in granulation tissue myofibroblasts and in quiescent and growing cultured fibroblasts. J Cell Biol 122:103–111. https://doi.org/10.1083/JCB.122.1.103
El-Agroudy NN, El-Naga RN, El-Razeq RA, El-Demerdash E (2016) Forskolin, a hedgehog signalling inhibitor, attenuates carbon tetrachloride-induced liver fibrosis in rats. Br J Pharmacol 173:3248–3260. https://doi.org/10.1111/BPH.13611
El-Mihi KA, Kenawy HI, El-Karef A et al (2017) Naringin attenuates thioacetamide-induced liver fibrosis in rats through modulation of the PI3K/Akt pathway. Life Sci 187:50–57. https://doi.org/10.1016/J.LFS.2017.08.019
Elsharkawy AM, Mann DA (2007) Nuclear factor-kappaB and the hepatic inflammation-fibrosis-cancer axis. Hepatology 46:590–597. https://doi.org/10.1002/HEP.21802
Epstein FH, Border WA, Noble NA (1994) Transforming growth factor beta in tissue fibrosis. N Engl J Med 331:1286–1292. https://doi.org/10.1056/NEJM199411103311907
Essam RM, Ahmed LA, Abdelsalam RM, El-Khatib AS (2019) Phosphodiestrase-1 and 4 inhibitors ameliorate liver fibrosis in rats: modulation of cAMP/CREB/TLR4 inflammatory and fibrogenic pathways. Life Sci 222:245–254. https://doi.org/10.1016/J.LFS.2019.03.014
Farazi PA, DePinho RA (2006) Hepatocellular carcinoma pathogenesis: from genes to environment. Nat Rev Cancer 6:674–687. https://doi.org/10.1038/NRC1934
Farombi EO, Shrotriya S, Surh YJ (2009) Kolaviron inhibits dimethyl nitrosamine-induced liver injury by suppressing COX-2 and iNOS expression via NF-κB and AP-1. Life Sci 84:149–155. https://doi.org/10.1016/J.LFS.2008.11.012
Feng J, Wang C, Liu T et al (2019) Procyanidin B2 inhibits the activation of hepatic stellate cells and angiogenesis via the Hedgehog pathway during liver fibrosis. J Cell Mol Med 23:6479–6493. https://doi.org/10.1111/JCMM.14543
Forsythe JA, Jiang BH, Iyer NV et al (1996) Activation of vascular endothelial growth factor gene transcription by hypoxia-inducible factor 1. Mol Cell Biol 16:4604–4613. https://doi.org/10.1128/MCB.16.9.4604
Friedman SL (1999) Cytokines and fibrogenesis. Semin Liver Dis 19:129–140. https://doi.org/10.1055/S-2007-1007105
George J, Tsuchishima M, Tsutsumi M (2019) Molecular mechanisms in the pathogenesis of N-nitrosodimethylamine induced hepatic fibrosis. Cell Death Dis. https://doi.org/10.1038/S41419-018-1272-8
González-Núñez V, Bachert C, Mullol J (2016) Rupatadine: global safety evaluation in allergic rhinitis and urticaria. Expert Opin Drug Saf 15:1439–1448. https://doi.org/10.1080/14740338.2016.1221399
Guy CD, Suzuki A, Zdanowicz M et al (2012) Hedgehog pathway activation parallels histologic severity of injury and fibrosis in human nonalcoholic fatty liver disease. Hepatology 55:1711–1721. https://doi.org/10.1002/HEP.25559
Hafez HM, Abdel-Hakeem EA, Hassanein H (2020) Rupatadine, a dual antagonist of histamine and platelet-activating factor (PAF), attenuates experimentally induced diabetic nephropathy in rats. Naunyn Schmiedebergs Arch Pharmacol 393:1487–1500. https://doi.org/10.1007/S00210-020-01856-8/FIGURES/6
Herpers B, Wink S, Fredriksson L et al (2016) Activation of the Nrf2 response by intrinsic hepatotoxic drugs correlates with suppression of NF-κB activation and sensitizes toward TNFα-induced cytotoxicity. Arch Toxicol 90:1163–1179. https://doi.org/10.1007/S00204-015-1536-3
Hickey MM, Simon MC (2006) Regulation of angiogenesis by hypoxia and hypoxia-inducible factors. Curr Top Dev Biol 76:217–257. https://doi.org/10.1016/S0070-2153(06)76007-0
Husain H, Latief U, Ahmad R (2018) Pomegranate action in curbing the incidence of liver injury triggered by diethylnitrosamine by declining oxidative stress via Nrf2 and NFκB regulation. Sci Rep 81(8):1–17. https://doi.org/10.1038/s41598-018-26611-1
Hwa Choi J, Jae Chung W, Han SJ et al (2000) Selective involvement of reactive oxygen intermediates in platelet-activating factor-mediated activation of NF-k B. Inflammation 24:385–398. https://doi.org/10.1023/A:1007068010645
Ibe BO, Abdallah MF, Portugal AM, Raj JU (2008) Platelet-activating factor stimulates ovine foetal pulmonary vascular smooth muscle cell proliferation: role of nuclear factor-kappa B and cyclin-dependent kinases. Cell Prolif 41:208–229. https://doi.org/10.1111/J.1365-2184.2008.00517.X
Ibrahim MA, Abdelmonaem AA, Abdel-Gaber SA et al (2022) Rupatadine ameliorated ulcerative colitis in rats via modulation of platelet-activatiweng factor/interleukin-6/vascular endothelial growth factor signalling pathway. J Pharm Pharmacol 74:537–546. https://doi.org/10.1093/JPP/RGAB170
Inagaki Y, Okazaki I (2007) Emerging insights into transforming growth factor β Smad signal in hepatic fibrogenesis. Gut 56:284–292. https://doi.org/10.1136/GUT.2005.088690
Ishak K, Baptista A, Bianchi L et al (1995) Histological grading and staging of chronic hepatitis. J Hepatol 22:696–699. https://doi.org/10.1016/0168-8278(95)80226-6
Jh C, Hm K, Jw K et al (2001) Platelet-activating factor-induced early activation of NF-kappa B plays a crucial role for organ clearance of Candida albicans. J Immunol. https://doi.org/10.4049/JIMMUNOL.166.8.5139
Jiayuan S, Junyan Y, Xiangzhen W et al (2020) Gant61 ameliorates CCl4-induced liver fibrosis by inhibition of Hedgehog signaling activity. Toxicol Appl Pharmacol 387:114853. https://doi.org/10.1016/J.TAAP.2019.114853
Jin N, Deng J, Chadashvili T et al (2010) Carbogen Gas-Challenge BOLD MR Imaging in a Rat Model of Diethylnitrosamine-induced Liver Fibrosis. Radiology 254:129. https://doi.org/10.1148/RADIOL.09090410
Jin X, Zhao TT, Shi D et al (2019) Protective role of fucoxanthin in diethylnitrosamine-induced hepatocarcinogenesis in experimental adult rats. Drug Dev Res 80:209–217. https://doi.org/10.1002/DDR.21451
Jose JK, Kuttan R, Bhattacharya RK (1998) Effect of Emblica officinalis extract on hepatocarcinogenesis and carcinogen metabolism. J Clin Biochem Nutr 25:31–39. https://doi.org/10.3164/JCBN.25.31
Kahveci OK, Kuzu S, Altıntaş M et al (2021) The effect of nasal steroid and antihistamine use on total oxidative stress and antioxidant status in the treatment of allergic rhinitis. Am J Rhinol Allergy 35:52–58. https://doi.org/10.1177/1945892420932768
Kantari-Mimoun C, Castells M, Klose R et al (2015) Resolution of liver fibrosis requires myeloid cell-driven sinusoidal angiogenesis. Hepatology 61:2042–2055. https://doi.org/10.1002/HEP.27635/SUPPINFO
Karantonis HC, Gribilas G, Stamoulis I et al (2010) Platelet-activating factor involvement in thioacetamide-induced experimental liver fibrosis and cirrhosis. Dig Dis Sci 55:276–284. https://doi.org/10.1007/S10620-009-0745-0
Khalaf HM, Hafez SMNA, Abdalla AM et al (2022) Role of Platelet-activating factor and HO-1 in mediating the protective effect of rupatadine against 5-fluorouracil-induced hepatotoxicity in rats. Environ Sci Pollut Res Int. https://doi.org/10.1007/S11356-022-18899-4
Koh RY, Lim CL, Uhal BD et al (2015) Inhibition of transforming growth factor-β via the activin receptor-like kinase-5 inhibitor attenuates pulmonary fibrosis. Mol Med Rep 11:3808–3813. https://doi.org/10.3892/MMR.2015.3193
Köhler UA, Böhm F, Rolfs F et al (2016) NF-κB/RelA and Nrf2 cooperate to maintain hepatocyte integrity and to prevent development of hepatocellular adenoma. J Hepatol 64:94–102. https://doi.org/10.1016/J.JHEP.2015.08.033
Kramann R (2016) Hedgehog Gli signalling in kidney fibrosis. Nephrol Dial Transplant 31:1989–1995. https://doi.org/10.1093/NDT/GFW102
Liao DJ, Blanck A, Eneroth P et al (2001) Diethylnitrosamine causes pituitary damage, disturbs hormone levels, and reduces sexual dimorphism of certain liver functions in the rat. Environ Health Perspect 109:943–947. https://doi.org/10.1289/EHP.01109943
Lijnen P, Petrov V, Rumilla K, Fagard R (2003) Transforming growth factor-beta 1 promotes contraction of collagen gel by cardiac fibroblasts through their differentiation into myofibroblasts. Methods Find Exp Clin Pharmacol 25:79–86. https://doi.org/10.1358/MF.2003.25.2.723680
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2-ΔΔCT method. Methods 25:402–408. https://doi.org/10.1006/meth.2001.1262
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275. https://doi.org/10.1016/0922-338X(96)89160-4
Lv X, Wang X, Li K et al (2013) Rupatadine protects against pulmonary fibrosis by attenuating PAF-mediated senescence in rodents. PLoS ONE 8:e68631. https://doi.org/10.1371/JOURNAL.PONE.0068631
Madankumar P, NaveenKumar P, Manikandan S et al (2014) Morin ameliorates chemically induced liver fibrosis in vivo and inhibits stellate cell proliferation in vitro by suppressing Wnt/β-catenin signaling. Toxicol Appl Pharmacol 277:210–220. https://doi.org/10.1016/J.TAAP.2014.03.008
Mansour SM, El-Abhar HS, Soubh AA (2021) MiR-200a inversely correlates with Hedgehog and TGF-β canonical/non-canonical trajectories to orchestrate the anti-fibrotic effect of Tadalafil in a bleomycin-induced pulmonary fibrosis model. Inflammopharmacology 29:167–182. https://doi.org/10.1007/S10787-020-00748-W
Mao Y, Zhang S, Yu F et al (2015) Ghrelin attenuates liver fibrosis through regulation of TGF-β1 expression and autophagy. Int J Mol Sci 16:21911–21930. https://doi.org/10.3390/IJMS160921911
Moczydlowska J, Miltyk W, Hermanowicz A et al (2017) HIF-1 α as a key factor in bile duct ligation-induced liver fibrosis in rats. J Investig Surg 30:41–46. https://doi.org/10.1080/08941939.2016.1183734
Mohamed MZ, Mohammed HH (2021) Rupatadine protects the intestinal mucosa from injury by 5-flurouracil via modulation of inflammation, apoptosis and intestinal permeability. https://doi.org/10.1080/01480545.2021.1997541
Mohamed MZ, Mohammed HH, Khalaf HM (2022) Therapeutic effect of rupatadine against l-arginine-induced acute pancreatitis in rats: role of inflammation. 100:176–183. https://doi.org/10.1139/CJPP-2021-0330
Moreira AJ, Ordoñez R, Cerski CT et al (2015) Melatonin activates endoplasmic reticulum stress and apoptosis in rats with diethylnitrosamine-induced hepatocarcinogenesis. PLoS ONE 10:e0144517. https://doi.org/10.1371/JOURNAL.PONE.0144517
Motawi TK, Hamed MA, Shabana MH et al (2011) Zingiber officinale acts as a nutraceutical agent against liver fibrosis. Nutr Metab 8:1–11. https://doi.org/10.1186/1743-7075-8-40/FIGURES/4
Ogaly HA, Aldulmani SAA, Al-Zahrani FAM, Abd-Elsalam RM (2022) D-carvone attenuates CCl 4-induced liver fibrosis in rats by inhibiting oxidative stress and TGF-ß 1/SMAD3 signaling pathway. Biology (Basel) 11:739. https://doi.org/10.3390/BIOLOGY11050739
Ogbozor UD, Opene M, Renteria LS et al (2015) Mechanism by which nuclear factor-kappa beta (NF-kB) regulates ovine fetal pulmonary vascular smooth muscle cell proliferation. Mol Genet Metab Rep 4:11–18. https://doi.org/10.1016/J.YMGMR.2015.05.003
Omenetti A, Choi S, Michelotti G, Diehl AM (2011) Hedgehog signaling in the liver. J Hepatol 54:366–373. https://doi.org/10.1016/J.JHEP.2010.10.003
Perumal NK, Perumal MK, Halagowder D, Sivasithamparam ND (2017) Morin attenuates diethylnitrosamine-induced rat liver fibrosis and hepatic stellate cell activation by co-ordinated regulation of Hippo/Yap and TGF-β1/Smad signaling. Biochimie 140:10–19. https://doi.org/10.1016/J.BIOCHI.2017.05.017
Pfeilschifter J, Schalkwijk C, Briner VA, Van den Bosch H (1993) Cytokine-stimulated secretion of group II phospholipase A2 by rat mesangial cells. Its contribution to arachidonic acid release and prostaglandin synthesis by cultured rat glomerular cells. J Clin Investig 92:2516–2523. https://doi.org/10.1172/JCI116860
Philips GM, Chan IS, Swiderska M et al (2011) Hedgehog signaling antagonist promotes regression of both liver fibrosis and hepatocellular carcinoma in a murine model of primary liver cancer. PLoS ONE. https://doi.org/10.1371/JOURNAL.PONE.0023943
Pratap A, Singh S, Mundra V et al (2012) Attenuation of early liver fibrosis by pharmacological inhibition of smoothened receptor signaling. J Drug Target 20:770–782. https://doi.org/10.3109/1061186X.2012.719900
Rosmorduc O, Housset C (2010) Hypoxia: a link between fibrogenesis, angiogenesis, and carcinogenesis in liver disease. Semin Liver Dis 30:258–270. https://doi.org/10.1055/S-0030-1255355
Sasaki Y, Iwai K (2016) Roles of the NF-κB pathway in B-lymphocyte biology. Curr Top Microbiol Immunol 393:177–209. https://doi.org/10.1007/82_2015_479
Sicklick JK, Li YX, Choi SS et al (2005) Role for Hedgehog signaling in hepatic stellate cell activation and viability. Lab Investig 8511(85):1368–1380. https://doi.org/10.1038/labinvest.3700349
Sugano T, Narahara H, Nasu K et al (2001) Effects of platelet-activating factor on cytokine production by human uterine cervical fibroblasts. Mol Hum Reprod 7:475–481. https://doi.org/10.1093/MOLEHR/7.5.475
Syn WK, Jung Y, Omenetti A et al (2009) Hedgehog-mediated epithelial-to-mesenchymal transition and fibrogenic repair in non-alcoholic fatty liver disease. Gastroenterology 137:1478. https://doi.org/10.1053/J.GASTRO.2009.06.051
Tran TV, Park SJ, Shin EJ et al (2018) Blockade of platelet-activating factor receptor attenuates abnormal behaviors induced by phencyclidine in mice through down-regulation of NF-κB. Brain Res Bull 137:71–78. https://doi.org/10.1016/J.BRAINRESBULL.2017.11.004
Travers JB (1999) Oxidative stress can activate the epidermal platelet-activating factor receptor. J Investig Dermatol 112:279–283. https://doi.org/10.1046/J.1523-1747.1999.00521.X
Tsochatzis EA, Crossan C, Longworth L et al (2014) Cost-effectiveness of noninvasive liver fibrosis tests for treatment decisions in patients with chronic hepatitis C. Hepatology 60:832–843. https://doi.org/10.1002/HEP.27296
Ushio-Fukai M, Alexander RW (2004) Reactive oxygen species as mediators of angiogenesis signaling role of NAD(P)H oxidase. Mol Cell Biochem 264:85–97. https://doi.org/10.1023/B:MCBI.0000044378.09409.b5
Vaughan MB, Howard EW, Tomasek JJ (2000) Transforming growth factor-beta1 promotes the morphological and functional differentiation of the myofibroblast. Exp Cell Res 257:180–189. https://doi.org/10.1006/EXCR.2000.4869
Wang R, Wang J, Song F et al (2018) Tanshinol ameliorates CCl 4-induced liver fibrosis in rats through the regulation of Nrf2/HO-1 and NF-κB/IκBα signaling pathway. Drug Des Dev Ther 12:1281–1292. https://doi.org/10.2147/DDDT.S159546
Weber SN, Bohner A, Dapito DH et al (2016) TLR4 deficiency protects against hepatic fibrosis and diethylnitrosamine-induced pre-carcinogenic liver injury in fibrotic liver. PLoS ONE 11:e0158819. https://doi.org/10.1371/JOURNAL.PONE.0158819
Wu K, Huang R, Wu H et al (2016) Collagen-binding vascular endothelial growth factor attenuates CCl4-induced liver fibrosis in mice. Mol Med Rep 14:4680–4686. https://doi.org/10.3892/MMR.2016.5826/HTML
Yang JJ, Tao H, Li J (2014) Hedgehog signaling pathway as key player in liver fibrosis: new insights and perspectives. Expert Opin Ther Targets 18:1011–1021. https://doi.org/10.1517/14728222.2014.927443
Yang HY, Kim KS, Lee YH et al (2019) Dendropanax morbifera ameliorates thioacetamide-induced hepatic fibrosis via TGF-β1/smads pathways. Int J Biol Sci 15:800. https://doi.org/10.7150/IJBS.30356
Yang X, Wang Z, Kai J et al (2020) Curcumol attenuates liver sinusoidal endothelial cell angiogenesis via regulating Glis-PROX1-HIF-1α in liver fibrosis. Cell Prolif 53:e12762. https://doi.org/10.1111/CPR.12762
Yao Q, Xu B, Wang J et al (2012) Inhibition by curcumin of multiple sites of the transforming growth factor-beta1 signalling pathway ameliorates the progression of liver fibrosis induced by carbon tetrachloride in rats. BMC Complement Altern Med 12:156. https://doi.org/10.1186/1472-6882-12-156
Yin C, Evason KJ, Asahina K, Stainier DYR (2013) Hepatic stellate cells in liver development, regeneration, and cancer. J Clin Investig 123:1902–1910. https://doi.org/10.1172/JCI66369
Zhang F, Zhang Z, Chen L et al (2014) Curcumin attenuates angiogenesis in liver fibrosis and inhibits angiogenic properties of hepatic stellate cells. J Cell Mol Med 18:1392–1406. https://doi.org/10.1111/JCMM.12286
Zhang F, Hao M, Jin H et al (2017) Canonical hedgehog signalling regulates hepatic stellate cell-mediated angiogenesis in liver fibrosis. Br J Pharmacol 174:409. https://doi.org/10.1111/BPH.13701
Zhao Y, Ma X, Wang J et al (2014) Paeoniflorin alleviates liver fibrosis by inhibiting HIF-1α through mTOR-dependent pathway. Fitoterapia 99:318–327. https://doi.org/10.1016/J.FITOTE.2014.10.009
Zhao S, Zhang Z, Yao Z et al (2017) Tetramethylpyrazine attenuates sinusoidal angiogenesis via inhibition of hedgehog signaling in liver fibrosis. IUBMB Life 69:115–127. https://doi.org/10.1002/IUB.1598
Zhou YN, Sun MY, Mu YP et al (2014) Xuefuzhuyu decoction inhibition of angiogenesis attenuates liver fibrosis induced by CCl4 in mice. J Ethnopharmacol 153:659–666. https://doi.org/10.1016/J.JEP.2014.03.019
Acknowledgements
The authors are grateful to Dr. Islam Elgohary, Animal Health Research Institute, Giza, Egypt, for his efforts in the histopathological investigations. The authors’ thanks are also extended to Dr. Laila Rashed (Cairo University, Faculty of Medicine) for conducting quantitative real-time PCR assessment.
Funding
Open access funding provided by The Science, Technology & Innovation Funding Authority (STDF) in cooperation with The Egyptian Knowledge Bank (EKB). This research did not receive any specific grant from public, commercial, or not-for-profit funding agencies.
Author information
Authors and Affiliations
Contributions
MD, LA developed the idea and designed the experimental approach. MD, AA performed the experiments. MD, AA, LA analyzed the data, prepared tables and figures, wrote, reviewed and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
None declared.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Didamoony, M.A., Atwa, A.M. & Ahmed, L.A. A novel mechanistic approach for the anti-fibrotic potential of rupatadine in rat liver via amendment of PAF/NF-ĸB p65/TGF-β1 and hedgehog/HIF-1α/VEGF trajectories. Inflammopharmacol 31, 845–858 (2023). https://doi.org/10.1007/s10787-023-01147-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10787-023-01147-7