Abstract
Seaweed aquaculture is receiving increasing attention for food and non-food applications in Europe, where it is still an emerging industry. The cultivation of seaweeds in the sea is attractive as it does not compete with agricultural crops for land and freshwater, whilst generating high yearly biomass yield. The selection of suitable cultivation sites in coastal waters is essential for the sustainable establishment and further development of seaweed aquaculture in Europe. Here, we investigate the effects of wave exposure and geographic location on growth and biofouling of kelp (Saccharina latissima), using a transplantation experiment along the Swedish west coast. Biofouling of kelp decreased with increased wave exposure, from 10 and 6% coverage at sheltered and moderately exposed locations, respectively, to 3% at exposed locations. Growth, measured as blade surface area, generally increased with decreased wave exposure, with approximately 40% less growth at exposed locations compared to sheltered or moderately exposed location. We identified that there is large spatial variation in growth and fouling of the seaweed biomass at the selected farm sites, with significant differences from the km-scale to the m-scale. In addition, exposure level affected the tissue composition, with a high carbon, but low nitrogen and water content at exposed locations compared to moderate and sheltered sites. Isotope signatures (i.e. δ13C and δ15N) also differed between exposure levels. Together, these results indicate that wave exposure is an important factor to consider in site selection for both yield as well as quality of the seaweed biomass for future kelp farms.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
There is a growing interest in seaweed cultivation for food and non-food applications (Holdt and Kraan 2011). Current biomass production for food applications is mainly based on agriculture on land; however, crop yield trends are predicted to be insufficient to reach the double global crop production needed by 2050 to meet FAO projections for food demands (Ray et al. 2013). Contrary to land-based agriculture, there is no need for arable land, fresh water, fertilizers and pesticides for the cultivation of seaweed. Accordingly, seaweed cultivation is often considered as the most environmentally friendly form of aquaculture (Folke et al. 1998), with a number of ecosystem and bioremediation services offered by the seaweeds (Hasselström et al. 2018). As a result, seaweed aquaculture is gaining interest and is predicted to expand into regions where it has thus far largely been underdeveloped, such as Europe and North America (FAO 2018). Recent studies have shown that European temperate coastal regions offer excellent environmental conditions for the cultivation of different kelp species, not least the sugar kelp, Saccharina latissima, which is ubiquitously found at the rocky shores of the North Atlantic (Sanderson et al. 2012; Marinho et al. 2015; Peteiro et al. 2016; Stévant et al. 2017).
The cultivation period of kelp species, such as S. latissima, is primarily dictated by seasonal changes in seawater temperatures and biofouling (Førde et al. 2015; Rolin et al. 2017). In nutrient-rich areas of most temperate coastal regions in the northern hemisphere, the cultivation season generally starts when seawater temperature gets below 15 °C in September–November and lasts until rapid increase in biofouling caused by rising seawater temperatures (Saunders and Metaxas 2007; Scheibling and Gagnon 2009; Park and Hwang 2012; Freitas et al. 2016) dictates the harvest in April–June. In European temperate regions, the most productive periods, i.e. with highest daily growth rates, are in early Autumn and Spring, when daylight is abundant (Broch et al. 2019). Accordingly, previous studies have shown that early deployment in Autumn and delayed harvest in Spring can provide substantial yield (Broch et al. 2019, Visch et al. unpublished data) at the same time as it circumvents the problem of biofouling that occurs later in the Spring.
Biofouling is one of the major challenges and constraints in the development and growth of the seaweed aquaculture sector (Getachew et al. 2015; Lüning and Mortensen 2015; Stévant et al. 2017; Bannister et al. 2019). Effects of biofouling include the loss of commercial value (Park and Hwang 2012), decrease in productivity due to breaking fronds (Dixon et al. 1981; Krumhansl et al. 2011), and reduced growth due to limited nutrient uptake (Hurd et al. 2000) and restricted light availability (Cancino et al. 1987). Biofouling is well documented in natural kelp populations (Christie et al. 2003; Christie et al. 2009), in farmed kelp species (Handa et al. 2013; Peteiro and Freire 2013b; Førde et al. 2015; Marinho et al. 2015; Rolin et al. 2017), as well as various other cultured seaweed species (Buschmann and Gómez 1993; Fletcher 1995; Vairappan 2006). The timing of recruitment of fouling species on farmed kelps has shown to follow a distinct seasonal pattern with an accumulation of species towards the end of the cultivation period (Sogn Andersen et al. 2011; Park and Hwang 2012; Førde et al. 2015; Rolin et al. 2017). This pattern is predictable across years (Walls et al. 2017) and typically coincides with an increase in water temperature (Sogn Andersen et al. 2011; Bruhn et al. 2016; Matsson et al. 2019). However, biofouling on seaweeds can also vary profoundly within a relatively small geographic range, unrelated to seasonal changes (Matsson et al. 2019). One important factor causing this variation can be hydrodynamic forces (strong currents and wave action) as previous studies have reported different biofouling cover on seaweeds in sheltered, semi-exposed and exposed localities (Peteiro and Freire 2013b; Bruhn et al. 2016; Mols-Mortensen et al. 2017; Matsson et al. 2019). This indicates that wave exposure and/or water current might be important to consider for site selection in order to reduce biofouling.
Efforts to reduce biofouling by cultivating seaweeds at different exposure and/or water current might affect seaweed growth (i.e. yield and morphology) and chemical content of farmed seaweeds (Hurd 2000; Hepburn et al. 2007). It is often considered that growth in natural kelp populations is higher in moderately exposed environments than wave-exposed or sheltered habitats (Leigh et al. 1987; Kregting et al. 2016). With respect to farmed kelp, Peteiro and Freire (2013a) showed that the biomass yield was higher at a moderately exposed location compared to a sheltered farm site, and Mols-Mortensen et al. (2017) found highest blade growth at a location with strong current compared to sheltered and wave-exposed sites. Furthermore, seaweeds grown in exposed water have higher carbon content due to increased carbon uptake and allocation to structural material (Kraemer and Chapman 1991b). Moreover, it is suggested that seaweed utilize different carbon sources (bicarbonate or carbon dioxide) depending on the wave exposure (Peterson and Fry 1987). Stable isotopes, such as δ13C and δ15N, can help determine the nutrient source (France 1995; Raven et al. 1995; Viana and Bode 2015). For example, tissue δ15N and nutrient concentrations decreased from more sheltered estuarine sites to more exposed marine waters, suggesting larger dominance of anthropogenic nitrogen sources in the estuary (Deutsch and Voss 2006; Raimonet et al. 2013; Viana and Bode 2015). Except for high exposure, selection of localities with different exposure will be limited to near shore coastal areas where most seaweed cultivation is placed today. Environmental conditions in near shore waters can vary extensively, especially in fjord systems or archipelagos that are often used for cultivation purposes, affecting seaweed growth and the yield of farmed seaweeds (Kerrison et al. 2015; Kim et al. 2015; Bruhn et al. 2016).
The Swedish west coast experiences a strong north to south salinity gradient created by the inflow of brackish water from the Baltic Sea into fully saline marine water from the North Sea. In addition, the outflow of river runoff water resulting in drops in salinity locally complicates the selection farm sites with favourable growth conditions. Thus, site selection for optimising yield and quality (fouling coverage and chemical composition) of farmed seaweeds also need to take spatial variation into account.
The aim of this study was to assess quantitative and qualitative effects of wave exposure and spatial variation on cultivated kelp (S. latissima). Quantitative effects were assessed by growth measurements and qualitative effects by measuring biofouling coverage and analysing tissue content of water, carbon and nitrogen.
Material and methods
Sample preparation
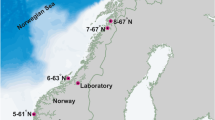
Non-fertile Saccharina latissima individuals were collected at the Swedish west coast (58° 83′ N, 10° 99′ S). Fertile sorus tissue was induced during a 10-week culturing period in 10 °C, short-day photoperiod and removal of the basal blade meristem (15 cm above the base) (Pang and Lüning 2004; Forbord et al. 2012). The spores were released in autoclaved seawater (ASW) with half strength Provasoli’s enriched seawater (PES) medium and the spore concentration was estimated using a haemocytometer. The spore solution was kept in aerated glass flask (5 L) at 10 °C with a light intensity of approximately 70 μmol photons m−2 s−1. Medium was changed weekly until the first sporophytes were visible after approximately 2 weeks. The developing gametophytes/sporophytes were concentrated using a 20-μm filter, rinsed with ASW and fragmented using a blender for 10 s. The resulting solution was diluted according to the number of released spores and sprayed onto a 1.2-mm diameter nylon cultivation line. Further development of seedlings was done under laboratory conditions: long-day photoperiod (16 h light), filtered (0.2 μm) half-strength PES medium at 10 °C and a light intensity of approximately 100 μmol photons m−2 s−1. Two meters of seeded long line, with small seedlings of up to 1 cm, were transplanted at the experimental localities (Fig. 1).
Geographical location and study design. The study area (a), the study design with the locations of the 15 experimental areas — each area exposure combination contains two lines (n = 90) — within three regions and three wave exposure classes (b). Asterisks denote the locations with temperature loggers
Experimental design and site description
The individual and interactive effects of geographic location and wave exposure on length, width, surface area and wet weight of transplanted S. latissima were examined in 3 regions with 5 areas per region (narea = 15). Each area contained three degrees of wave exposure, with two lines within each area wave exposure combination (nline = 90). From each line, 10 individual seaweed blades (nindividual = 900) were collected for measurements (Fig. 1 ). The three regions were defined as “North”, “Middle”, and “South”, with a distance of approximately 20 km between the nearest area from a different region. The lines were deployed approximately 20 m apart and contained 2 m seeded seaweed line that was anchored and buoyed to float so that the upper part of the line was submerged around 0.5 m below the water surface. The wave exposures were defined as follows: 500,000–800,000 m2s−1 (exposed), 100,000–200,000 m2s−1 (moderately exposed) and 10,000–30,000 m2s−1 (sheltered). Data for wave exposure came from the simplified wave model method SWM (Isæus 2004), and the chosen groups of wave exposure were subsets of the classes “Exposed”, “Moderately exposed” and “Sheltered” in the wave exposure index as suggested by Isæus (2004). The experiment was conducted over a period between January and June 2018 (147–172 days).
At the Swedish west coast, natural kelp (S. latissima) populations are present at the rocky shores from a depth of about 1 m. The tidal ranges are small (< 0.3 m), but differences between high and low water levels up to 2 m still occur due to changes in atmospheric pressure and wind (Johannesson 1989). The mean surface water (0–10 m deep) salinity during cultivation months of the last decade (2008–2018) at the experimental sites varied between 27.6 ± 3.5 psu (mean ± sd, n = 333), 28.3 ± 5.2 psu (mean ± sd, n = 588), and 25.7 ± 3.6 psu (mean ± sd, n = 485) in the northern, middle and southern experimental regions respectively (SMHI 2019). This is primarily influenced by to the inflow of saline marine water from the North Sea and low saline water from the Baltic Sea into the Skagerrak (Fig. 1a). Drops in salinities are locally due to nutrient-rich river run off, primarily in late spring/early summer. Historic nutrient concentrations in the seawater (i.e. total nitrogen, NO2 + NO3, NH4, total phosphorus and PO4) at the three experimental regions during the experimental period indicates relatively small variability between regions, but slightly higher concentrations of dissolved N and P the middle region compared to the northern and southern region (SMHI 2019; Table S1). In addition, the experimental regions consist primarily of rocky shores with many islands and skerries, creating variation of hydrodynamic forces on relatively small geographic scales without altering the chemistry of the seawater. Data from the STRÅNG model system (Landelius et al. 2001) show that the mean light irradiance during the experiment was 1320 ± 1159 Wh m−2 (mean ± sd, n = 7568), with an irradiance of 2799 ± 824 Wh m−2 (mean ± sd, n = 1408) in the final month of the experiment (Fig. S1). Seawater surface temperature (°C) was measured throughout the experimental period at selected areas within the experimental regions (Fig. 1) using Onset HOBO Pendant UA-002-64 loggers. The water temperature was < 15 °C until the middle of May, after that a sharp increase was noted up to approx. 20 °C (Fig. S2).
Growth and biofouling measurements
From each 2-m seeded seaweed line, 10 individual seaweed blades were haphazardly selected at 20-cm intervals starting at the water surface. Data was collected on wet weight (WW), length (L), width (W), surface area (SA), fouling coverage (F) and composition of fouling taxa. The size and fouling measurements (i.e. L, W, SA and F) were analysed using ImageJ software. The contribution of fouling species was related to total blade surface area, and biofouling species were grouped according to taxa.
Tissue composition
The same samples (n = 10) used for growth and fouling analysis were analysed for variation in water content (%), which was calculated by comparing tissue dry weight after freeze-drying with the tissue wet weight directly after harvest. The variation in chemical composition of total carbon, nitrogen, and C:N ratio of blade tissue related to wave exposure was analysed for a subset of seaweeds (n = 18) within the middle region. These samples were also analysed for δ13C and δ15N, in order to investigate the accessibility of carbon and the source of nitrogen (i.e. from a marine or terrestrial origin) that were assimilated by the seaweed in their respective exposure class. Freeze-dried blade samples were ground into a fine powder with a mortar, and samples of approximately 10 mg samples were analysed using an elemental analyser coupled to an isotope ratio mass spectrometer (SerCon GSL and 20–22 IR;MS; SerCon Ltd., UK).
Statistical analysis
All analyses were conducted using R software (R Core Team 2018). Effects on growth (WW, L, W, and SA), fouling coverage and water content of blade tissue for exposure (fixed factor, 3 levels), region (fixed factor, 3 levels), area within region (random factor, 5 levels, varying intercept) and line within area (random factor, 2 levels, varying intercept) were statistically analysed with mixed model analysis of variance (ANOVA) using the lmer function from the lme4 package (Bates et al. 2014). Means from duplicate samples of total carbon and nitrogen, C:N ratio, δ13C and δ15N were statistically analysed with ANOVA using the lm function (R Core Team 2018) using exposure (3 levels) as fixed factor. Differences in biofouling species composition were analysed using descriptive statistics, as the abundant double zero’s among samples restricted statistical testing.
Prior to all statistical analyses, data was graphically analysed and the best suited normalizing transformation was estimated using the bestNormalize package (Peterson 2017). Multiple comparisons of significant different fixed factors were performed with a Student-Newman-Keuls (SNK) post hoc test (α = 0.05), using the SNK.test function in the agricolae package (version 1.3-1). Marginal \( {R}_m^2 \) and the conditional \( {R}_c^2 \) were used to estimate the amount of variance explained by the fixed effects only and the combined fixed and random effects of the models, respectively (Nakagawa and Schielzeth 2013).
Results
Blade biofouling
The biofouling of the seaweed blades was interactively affected by exposure and region (Table 1). The overall pattern indicated less fouling with increasing exposure (Fig. 2b), with significantly lower fouling cover at exposed localities in each region and except for the north region also significantly lower fouling cover for the moderately exposed localities compared to the sheltered localities (Table 1; post hoc test §3). From a total of 729 blade samples, biofouling was absent in 79 individuals (i.e. 10.8%), of which 66 were found at exposed, 9 at moderate and 4 at sheltered locations. The mean biofouling coverage for the exposed, moderately exposed and sheltered localities was 2.8 ± 0.3% (mean ± se, n = 152), 6.2 ± 0.2% (mean ± se, n = 282) and 10.1 ± 0.6% (mean ± se, n = 295), respectively. The blade biofouling was also affected by region, with significantly higher fouling coverage for sheltered and moderately exposed localities at the southern region (16.6 ± 1.0% (mean ± se, n = 177)) compared to the northern (5.3 ± 0.3% (mean ± se, n = 200)) and the middle (3.7 ± 0.2% (mean ± se, n = 200)) region (Table 1; post hoc test §3). For exposed localities, the highest fouling coverage was in the northern region (4.3 ± 0.7% (mean ± se, n = 59)), followed by the middle region (2.0 ± 0.4% (mean ± se, n = 41)), and then by the southern region (1.6 ± 0.5% (mean ± se, n = 52)). Additionally, the fouling cover varied significantly among lines while variability was much lower among the area.
Fouling species
Four major biofouling taxa were observed to contribute to biofouling of the transplanted blade of S. latissima: bryozoans (Membranipora membranacea and Electra pilosa), hydroids (mainly Obelia geniculata), tube-building amphipods (mainly Jassa falcata) and tunicates (Ciona intestinalis). There was also a considerable amount of debris (denoted “undefined”) contributing to the total fouling cover. In terms of blade coverage (%), amphipods were most dominant (7.7 ± 0.6%), followed by bryozoans (4.7 ± 0.4%), hydroids (2.7 ± 0.3), undefined (1.7 ± 0.1) and tunicates (1.2 ± 0.1) (mean ± se) (Fig. 3). In addition, species composition varied between regions and exposure (Table S2). For example, tunicates were found to be most abundant in the northern and middle region at sheltered and moderately exposure while they were totally absent at the exposed locations. The fouling composition at exposed locations consisted predominately of bryozoans and hydroids, whilst biofouling of blades cultured at sheltered and moderately exposed locations in the northern and middle regions were found be more species diverse. Fouling in the southern region consisted mainly of tube-building amphipods, covering most of the blade.
Seaweed growth
Across all samples blade growth, expressed as surface area, increased with 85.5 ± 2.0 cm2 (mean ± se, n = 729) during the experimental period. Both exposure and region had a significant effect on the blade surface area (Table 1; Fig. 2a), as well as on all other measures of growth (i.e. wet weight, length and width; see Table S3 and Fig. S3). The highest growth was found at moderately exposed locations (95.64 ± 3.1 cm2 (mean ± se, n = 282)) and sheltered locations (91.4 ± 3.4 cm2 (mean ± se, n = 295)), while growth at exposed locations was significantly lower (55.3 ± 3.0 cm2 (mean ± se, n = 152)) (Table 1; post hoc test §1). This resembled a difference of approximately 40% in surface area (cm2) between both sheltered and moderately exposed locations compared to exposed locations. The regional difference consisted of lowest growth in the southern region (32.2 ± 1.1 cm2 (mean ± se, n = 229)), followed by the northern region (101.0 ± 3.2 cm2 (mean ± se, n = 259)) and the middle region (119.4 ± 2.7 cm2 (mean ± se, n = 241)) (Table 1; post hoc test §2). Additionally, the growth of blade surface area was found to vary significantly between areas and among lines (Table 1).
Tissue composition
The water content was significantly affected by exposure with decreasing levels when exposure increased (Table 1; post hoc test §4). In addition, region had a significant effect on the water content with highest levels in the middle region, followed by the northern region, while the lowest water content was observed at the southern region (Table 1; post hoc test §5). This pattern corresponded to the observed differences of growth in blade surface area (i.e. middle > north > south). The variability among areas was ten times higher than among lines, although the variance contribution for neither of the factors was statistically significant. With respect to the chemical content in the seaweed tissue (Table 2), we found a near significant effect of exposure on the carbon content in the tissue (p = 0.053) with a higher mean at more exposed locations compared to the moderate and sheltered sites (Fig. 4a). Tissue nitrogen content significantly differed between exposure, with a higher content at sheltered locations compared to exposed sites (Fig. 4b). Consequently, a significantly higher C:N ratio was found at exposed locations compared to moderate and sheltered sites (Table 2 and Fig. 4c). δ13C was also significantly different between exposures, with a lower value at exposed compared to moderate and sheltered sites (Table 2, Fig. 4d). Additionally, δ15N was significantly higher at sheltered compared to exposed and moderate locations (Table 2, Fig. 4e).
Discussion
In this study, cultivation of S. latissima was successful within all levels of exposure and tested regions along the northern part of the Swedish west coast. Wave exposure was found to affect biofouling, with less fouling at higher wave exposure. The exposure level also affected the tissue composition, with a high carbon but low nitrogen and water content at exposed locations compared to moderate and sheltered sites. In addition, there was relatively large spatial variation, from m-scale to tens of km-scale, in the growth of S. latissima. Overall, growth significantly increased with decreased wave exposure.
Biofouling has been identified previously as a key constraint for many seaweed farms worldwide (Fletcher 1995; Lüning and Mortensen 2015; Kim et al. 2017). Here, we found that biofouling occurred at all exposure levels, but that it was significantly lower at more exposed locations, which corroborates previous findings (Sogn Andersen et al. 2011; Peteiro and Freire 2013a). However, Matsson et al. (2019) found a reversed pattern, with a higher fouling coverage of the blade at more exposed sites compared to the inshore and fjord location in northern Norway, and Bruhn et al. (2016) found that fouling did not correspond to estimated degree of exposure within a Danish estuary. In addition, one of the most popular location for seaweed cultivation in Korea (Wando region) experiences significant problems with biofouling (Park and Hwang 2012), despite relatively high current velocities (approx. 50 cm s−1) compared to exposed locations where biofouling in European kelp farms has been examined (Peteiro and Freire 2013b; Førde et al. 2015; Mols-Mortensen et al. 2017; Matsson et al. 2019). Although we purposefully used single dropper lines in the present experiment that allowed us to explore various exposure classes at a greater geographic scale, the results may have been influenced by the size and density of the cultivation as wave exposure and/or current velocities are likely to alter within a large-scale farm and change during the cultivation period as the biomass increases (Shi et al. 2011).
Species abundance and biofouling taxa recorded in this study were both within the same range (between 3 and 32 species) and species composition compared to previous findings from other kelp farms (Kim et al. 2017; Rolin et al. 2017; Walls et al. 2017; Bannister et al. 2019; Matsson et al. 2019). Fouling taxa overlapped between region and exposure, but the species composition (i.e. the number of taxa) decreased with increased exposure. Similar to Matsson et al. (2019), hydroids and bryozoans were predominantly found at exposed locations. Biofouling of tunicates (C. intestinalis) was also observed at kelp farms in waters surrounding the Shetland Islands and Skagerrak (Sogn Andersen et al. 2011; Rolin et al. 2017); however, its presence was not related to the level of exposure. Here, we found that fouling of C. intestinalis was limited to sheltered and moderately exposed environments. The tube dwelling amphipod (J. falcata) was the dominant fouling species at low and intermediate exposure levels in the southern region. It is an important fouling organism that forms dense mats or “nests” made of debris that started at the culture line close to the base of the blade, from where it expanded towards the distal part. This pattern differed from the other fouling taxa found in the present study that mainly started at the distal part of the blade (Visch, pers. observation). The temperature during the experimental period in spring and early summer of 2016 was relatively warm in northern Europe (SMHI 2016a, 2016b) (Fig. S2), which may have affected the biofouling and its succession on the seaweed blades as it is known to coincide with increasing seawater temperature (Sogn Andersen et al. 2011; Bruhn et al. 2016; Matsson et al. 2019). Consequently, future predictability of timing of the harvest may be altered, as the frequency and duration of marine heat waves are expected to increase (Oliver et al. 2018).
Growth, expressed as blade surface area, was found to be lower at exposed locations compared to sheltered and moderately exposed locations. Previous studies investigating yield of kelp farms in relation to exposure have focused on different exposure types (e.g. sheltered, current velocity or wave exposure); therefore, results have been ambiguous and general conclusions have been difficult to draw. For example, high-exposed environments are where found to generate lower yields (Sanderson et al. 2012), or higher yields at wave-exposed and sheltered sites compared to current-exposed locations (Mols-Mortensen et al. 2017). Others, however, found a positive relationship between current velocity and yield (Peteiro and Freire 2011, 2013a), or no relationship between yield and degree of exposure (Bruhn et al. 2016; Matsson et al. 2019). In natural kelp populations, exposure has shown to affect productivity, with higher growth in moderately exposed environments compared to sheltered or wave-exposed habitats (Leigh et al. 1987; Kregting et al. 2016). The present study, showing less growth at higher wave exposure, provided a strong test of the effect of wave exposure that most likely were only minorly confounded with current exposure as it was conducted in waters with very weak tidal currents (tidal range < 0.3 m).
Morphological and biomechanical responses of seaweeds to different hydrodynamic regimes are well documented in natural communities, with generally narrow/thick blades in exposed habitats and wide/thin blades in sheltered habitats (Hurd et al. 1996; Hurd 2000; Koehl et al. 2008; Bekkby et al. 2014). The narrower blades found at kelp farms in exposed sites found in the present study, and in a study by Mols-Mortensen et al. (2017), substantiate this pattern. Furthermore, effects of wave exposure on tissue carbon content was previously found to be related to an increase in carbon uptake that is allocated to structural cell wall material (Kraemer and Chapman 1991a, b). It is hypothesised that differences in growth rate due to water motion may be related to trade-offs between blade growth and an increase in tissue strength (Kregting et al. 2016). This is corroborated by the chemical analysis and growth rates observed in the present study showing a higher carbon content in exposed locations, but a greater yield at moderately exposed cultivation sites. Based on the expected preferential uptake of 12C in seaweeds (Keeley and Sandquist 1992), the observed difference in δ13C could be explained by differences in accessibility of carbon at the different exposure levels, with less access of carbon in the sheltered localities. Overall, tissue nitrogen content was relatively low compared to previous findings for cultured kelp (Sanderson et al. 2012; Handa et al. 2013; Wang et al. 2014). This could be the result of a delayed harvest date (i.e. June) in the present study, compared to normal harvesting period in April/May in the region (Handa et al. 2013). Exposure also had a strong impact on the nitrogen content in the tissue, with elevated concentrations at sheltered locations. This could however be confounded by higher concentrations of dissolved inorganic nitrogen at sheltered sites unrelated to exposure level, as they were located closer to the shore where higher seawater nitrogen concentrations can be expected from an influx of nutrients from land. Although the dissolved nutrients where not directly measured, the elevated tissue δ15N at the sheltered sites suggest a larger dominance of anthropogenic nitrogen compared to more moderately and exposed locations (Deutsch and Voss 2006; Raimonet et al. 2013; Viana and Bode 2015).
Increasingly, there is the need to study and understand the potential effects of cultivation site on the quality and quantity of the biomass in seaweed aquaculture. In a recent study, between 475 and 544 km2 of sea area within the Swedish exclusive economic zone (EEZ) along the west coast was identified as suitable for seaweed cultivation, without conflicting with socio-economic activities (Thomas et al. 2019). Here, we identified effects of wave exposure on biofouling, growth and chemical content that will be informative for identifying new kelp farm locations within this area. A particular priority for consideration should be the large km-scale geographic variation in biomass yield and blade fouling. In conclusion, our findings together with previous studies can facilitate site selection in seaweed aquaculture, thereby improving the yield as well as the quality of farmed seaweed biomass.
References
Bannister J, Sievers M, Bush F, Bloecher N (2019) Biofouling in marine aquaculture: a review of recent research and developments. Biofouling 35:631–648
Bates D, Mächler M, Bolker B, Walker S (2014) Fitting linear mixed-effects models using lme4. arXiv preprint arXiv:14065823
Bekkby T, Rinde E, Gundersen H, Norderhaug KM, Gitmark JK, Christie H (2014) Length, strength and water flow: relative importance of wave and current exposure on morphology in kelp Laminaria hyperborea. Mar Ecol Prog Ser 506:61–70
Broch OJ, Alver MO, Bekkby T, Gundersen H, Forbord S, Handå A, Skjermo J, Hancke K (2019) The kelp cultivation potential in coastal and offshore regions of Norway. Front Mar Sci 5:529
Bruhn A, Tørring DB, Thomsen M, Canal-Vergés P, Nielsen MM, Rasmussen MB, Eybye KL, Larsen MM, Balsby TJS, Petersen JK (2016) Impact of environmental conditions on biomass yield, quality, and bio-mitigation capacity of Saccharina latissima. Aquacult Environ Interact 8:619–636
Buschmann AH, Gómez P (1993) Interaction mechanisms between Gracilaria chilensis (Rhodophyta) and epiphytes. Hydrobiologia 260:345–351
Cancino J, Mufioz J, Mufioz M, Orellana M (1987) Effects of the bryozoan Membranipora tuberculata (Bosc.) on the photosynthesis and growth of Gelidium rex Santelices et Abbott. J Exp Mar Biol Ecol 113:105–112
Christie H, Jørgensen NM, Norderhaug KM, Waage-Nielsen E (2003) Species distribution and habitat exploitation of fauna associated with kelp (Laminaria hyperborea) along the Norwegian coast. J Mar Biol Ass UK 83:687–699
Christie H, Norderhaug KM, Fredriksen S (2009) Macrophytes as habitat for fauna. Mar Ecol Prog Ser 396:221–233
Deutsch B, Voss M (2006) Anthropogenic nitrogen input traced by means of δ15N values in macroalgae: results from in-situ incubation experiments. Sci Total Environ 366:799–808
Dixon J, Schroeter SC, Kastendiek J (1981) Effects of the encrusting bryozoan, Membranipora membranacea, on the loss of blades and fronds by the giant kelp, Macrocystis pyrifera (Laminariales). J Phycol 17:341–345
FAO (2018) The State of World Fisheries and Aquaculture 2018 - Meeting the sustainable development goals. The State of World Fisheries and Aquaculture - SOFIA 2018. FAO, Rome
Fletcher RL (1995) Epiphytism and fouling in Gracilaria cultivation: an overview. J Appl Phycol 7:325–333
Folke C, Kautsky N, Berg H, Jansson Å, Troell M (1998) The ecological footprint concept for sustainable seafood production: a review. Ecol Appl 8:63–71
Forbord S, Skjermo J, Arff J, Handå A, Reitan K, Bjerregaard R, Lüning K (2012) Development of Saccharina latissima (Phaeophyceae) kelp hatcheries with year-round production of zoospores and juvenile sporophytes on culture ropes for kelp aquaculture. J Appl Phycol 24:393–399
Førde H, Forbord S, Handå A, Fossberg J, Arff J, Johnsen G, Reitan K (2015) Development of bryozoan fouling on cultivated kelp (Saccharina latissima) in Norway. J Appl Phycol 28:1225–1234
France R (1995) Source variability in δ15N of autotrophs as a potential aid in measuring allochthony in freshwaters. Ecography 18:318–320
Freitas JR, Morrondo JMS, Ugarte JC (2016) Saccharina latissima (Laminariales, Ochrophyta) farming in an industrial IMTA system in Galicia (Spain). J Appl Phycol 28:377–385
Getachew P, Kang J-Y, Choi J-S, Hong Y-K (2015) Does bryozoan colonization alter the biochemical composition of Saccharina japonica affecting food safety and quality? Bot Mar 58:267–274
Handa A, Forbord S, Wang X, Broch OJ, Dahle SW, Storseth TR, Reitan KI, Olsen Y, Skjermo J (2013) Seasonal- and depth-dependent growth of cultivated kelp (Saccharina latissima) in close proximity to salmon (Salmo salar) aquaculture in Norway. Aquaculture 414:191–201
Hasselström L, Visch W, Gröndahl F, Nylund GM, Pavia H (2018) The impact of seaweed cultivation on ecosystem services-a case study from the west coast of Sweden. Mar Pollut Bull 133:53–64
Hepburn CD, Holborow JD, Wing SR, Frew RD, Hurd CL (2007) Exposure to waves enhances the growth rate and nitrogen status of the giant kelp Macrocystis pyrifera. Mar Ecol Prog Ser 339:99–108
Holdt SL, Kraan S (2011) Bioactive compounds in seaweed: functional food applications and legislation. J Appl Phycol 23:543–597
Hurd CL (2000) Water motion, marine macroalgal physiology, and production. J Phycol 36:453–472
Hurd C, Harrison P, Druehl L (1996) Effect of seawater velocity on inorganic nitrogen uptake by morphologically distinct forms of Macrocystis integrifolia from wave-sheltered and exposed sites. Mar Biol 126:205–214
Hurd CL, Durante KM, Harrison PJ (2000) Influence of bryozoan colonization on the physiology of the kelp Macrocystis integrifolia (Laminariales, Phaeophyta) from nitrogen-rich and -poor sites in Barkley Sound, British Columbia, Canada. Phycologia 39:435–440
Isæus M (2004) Factors structuring Fucus communities at open and complex coastlines in the Baltic Sea. Doctoral Thesis, Stockholm University
Johannesson K (1989) The bare zone of Swedish rocky shores: why is it there? Oikos 1:77–86
Keeley JE, Sandquist D (1992) Carbon: freshwater plants. Plant Cell Environ 15:1021–1035
Kerrison PD, Stanley MS, Edwards MD, Black KD, Hughes AD (2015) The cultivation of European kelp for bioenergy: site and species selection. Biomass Bioenergy 80:229–242
Kim JK, Kraemer GP, Yarish C (2015) Use of sugar kelp aquaculture in Long Island sound and the Bronx River Estuary for nutrient extraction. Mar Ecol Prog Ser 531:155–166
Kim J-O, Kim W-S, Jeong H-N, Choi S-J, Seo J-S, Park M-A, Oh M-J (2017) A survey of epiphytic organisms in cultured kelp Saccharina japonica in Korea. Fish Aquat Sci 20:1
Koehl M, Silk WK, Liang H, Mahadevan L (2008) How kelp produce blade shapes suited to different flow regimes: a new wrinkle. Integr Comp Biol 48:834–851
Kraemer GP, Chapman DJ (1991a) Biomechanics and alginic acid composition during hydrodynamic adaptation by Egregia menziesii (Phaeophyta) juveniles. J Phycol 27:47–53
Kraemer GP, Chapman DJ (1991b) Effects of tensile force and nutrient availability on carbon uptake and cell wall synthesis in blades of juvenile Egregia menziesii (Turn.) Aresch.(Phaeophyta). J Exp Mar Biol Ecol 149:267–277
Kregting L, Blight AJ, Elsäßer B, Savidge G (2016) The influence of water motion on the growth rate of the kelp Laminaria digitata. J Exp Mar Biol Ecol 478:86–95
Krumhansl KA, Lee JM, Scheibling RE (2011) Grazing damage and encrustation by an invasive bryozoan reduce the ability of kelps to withstand breakage by waves. J Exp Mar Biol Ecol 407:12–18
Landelius T, Josefsson W, Persson T (2001) A system for modelling solar radiation parameters with mesoscale spatial resolution. SMHI Reports Meteorology and Climatology No 96. 52 p
Leigh EG, Paine RT, Quinn JF, Suchanek TH (1987) Wave energy and intertidal productivity. Proc Nat Acad Sci 84:1314–1318
Lüning K, Mortensen L (2015) European aquaculture of sugar kelp (Saccharina latissima) for food industries: iodine content and epiphytic animals as major problems. Bot Mar 58:449–455
Marinho GS, Holdt SL, Birkeland MJ, Angelidaki I (2015) Commercial cultivation and bioremediation potential of sugar kelp, Saccharina latissima, in Danish waters. J Appl Phycol 27:1963–1973
Matsson S, Christie H, Fieler R (2019) Variation in biomass and biofouling of kelp, Saccharina latissima, cultivated in the Arctic, Norway. Aquaculture 506:445–452
Mols-Mortensen A, Jacobsen C, Holdt SL (2017) Variation in growth, yield and protein concentration in Saccharina latissima (Laminariales, Phaeophyceae) cultivated with different wave and current exposures in the Faroe Islands. J Appl Phycol 29:2277–2286
Nakagawa S, Schielzeth H (2013) A general and simple method for obtaining R2 from generalized linear mixed-effects models. Methods Ecol Evol 4:133–142
Oliver EC, Donat MG, Burrows MT, Moore PJ, Smale DA, Alexander LV, Benthuysen JA, Feng M, Gupta AS, Hobday AJ (2018) Longer and more frequent marine heatwaves over the past century. Nat Commun 9:1324
Pang S, Lüning K (2004) Breaking seasonal limitation: year-round sporogenesis in the brown alga Laminaria saccharina by blocking the transport of putative sporulation inhibitors. Aquaculture 240:531–541
Park CS, Hwang EK (2012) Seasonality of epiphytic development of the hydroid Obelia geniculata on cultivated Saccharina japonica (Laminariaceae, Phaeophyta) in Korea. J Appl Phycol 24:433–439
Peteiro C, Freire Ó (2011) Effect of water motion on the cultivation of the commercial seaweed Undaria pinnatifida in a coastal bay of Galicia, Northwest Spain. Aquaculture 314:269–276
Peteiro C, Freire Ó (2013a) Biomass yield and morphological features of the seaweed Saccharina latissima cultivated at two different sites in a coastal bay in the Atlantic coast of Spain. J Appl Phycol 25:205–213
Peteiro C, Freire Ó (2013b) Epiphytism on blades of the edible kelps Undaria pinnatifida and Saccharina latissima farmed under different abiotic conditions. J World Aquacult Soc 44:706–715
Peteiro C, Sánchez N, Martínez B (2016) Mariculture of the Asian kelp Undaria pinnatifida and the native kelp Saccharina latissima along the Atlantic coast of Southern Europe: an overview. Algal Res 15:9–23
Peterson R (2017) Estimating normalization transformations with bestNormalize
Peterson BJ, Fry B (1987) Stable isotopes in ecosystem studies. Annu Rev Ecol Systemat 18:293–320
R Core Team (2018) R: a language and environment for statistical computing
Raimonet M, Guillou G, Mornet F, Richard P (2013) Macroalgae δ15N values in well-mixed estuaries: indicator of anthropogenic nitrogen input or macroalgae metabolism? Estuar Coast Shelf Sci 119:126–138
Raven JA, Walker DI, Johnston AM, Handley LL, Kübler JE (1995) Implications of 13C natural abundance measurements for photosynthetic performance by marine macrophytes in their natural environment. Mar Ecol Prog Ser 123:193–205
Ray DK, Mueller ND, West PC, Foley JA (2013) Yield trends are insufficient to double global crop production by 2050. PLoS One 8:e66428
Rolin C, Inkster R, Laing J, McEvoy L (2017) Regrowth and biofouling in two species of cultivated kelp in the Shetland Islands, UK. J Appl Phycol 29:2351–2361
Sanderson J, Dring M, Davidson K, Kelly M (2012) Culture, yield and bioremediation potential of Palmaria palmata (Linnaeus) Weber & Mohr and Saccharina latissima (Linnaeus) CE Lane, C. Mayes, Druehl & GW Saunders adjacent to fish farm cages in northwest Scotland. Aquaculture 354:128–135
Saunders M, Metaxas A (2007) Temperature explains settlement patterns of the introduced bryozoan Membranipora membranacea in Nova Scotia, Canada. Mar Ecol Prog Ser 344:95–106
Scheibling RE, Gagnon P (2009) Temperature-mediated outbreak dynamics of the invasive bryozoan Membranipora membranacea in Nova Scotian kelp beds. Mar Ecol Prog Ser 390:1–13
Shi J, Wei H, Zhao L, Yuan Y, Fang J, Zhang J (2011) A physical–biological coupled aquaculture model for a suspended aquaculture area of China. Aquaculture 318:412–424
SMHI (2016a) April 2016 - A warm spring changed to April weather (in Swedish). https://www.smhi.se/klimat/klimatet-da-och-nu/manadens-vader-och-vatten-sverige/manadens-vader-i-sverige/april-2016-varvarme-overgick-i-aprilvader-1.102944. Accessed 13 August 2019
SMHI (2016b) May 2016 - Warm with a dry start and a wet end (in Swedish). https://www.smhi.se/klimat/klimatet-da-och-nu/manadens-vader-och-vatten-sverige/manadens-vader-i-sverige/maj-2016-varm-med-torr-borjan-och-blott-slut-1.104093. Accessed 13 August 2019
SMHI (2019) Marina miljöövervakningsdata (Marine environmental data). https://www.smhi.se/klimatdata/oceanografi/havsmiljodata/marina-miljoovervakningsdata. Accessed 6 August 2019
Sogn Andersen G, Steen H, Christie H, Fredriksen S, Moy FE (2011) Seasonal patterns of sporophyte growth, fertility, fouling, and mortality of Saccharina latissima in Skagerrak, Norway: implications for forest recovery. J Mar Biol 2011:690375
Stévant P, Rebours C, Chapman A (2017) Seaweed aquaculture in Norway: recent industrial developments and future perspectives. Aquacult Int 25:1373–1390
Thomas J-BE, Ramos FS, Gröndahl F (2019) Identifying suitable sites for macroalgae cultivation on the Swedish west coast. Coast Manage 14:88–106
Vairappan CS (2006) Seasonal occurrences of epiphytic algae on the commercially cultivated red alga Kappaphycus alvarezii (Solieriaceae, Gigartinales, Rhodophyta). J Appl Phycol 18:611–617
Viana IG, Bode A (2015) Variability in δ15N of intertidal brown algae along a salinity gradient: differential impact of nitrogen sources. Sci Total Environ 512:167–176
Walls A, Edwards M, Firth L, Johnson M (2017) Successional changes of epibiont fouling communities of the cultivated kelp Alaria esculenta: predictability and influences. Aquacult Environ Interact 9:57–71
Wang X, Broch OJ, Forbord S, Handå A, Skjermo J, Reitan KI, Vadstein O, Olsen Y (2014) Assimilation of inorganic nutrients from salmon (Salmo salar) farming by the macroalgae (Saccharina latissima) in an exposed coastal environment: implications for integrated multi-trophic aquaculture. J Appl Phycol 26:1869–1878
Acknowledgements
Open access funding provided by University of Gothenburg. This work was associated with the Swedish Mariculture Research Center (SWEMARC), Center for Sea and Society, University of Gothenburg. The authors also wish to acknowledge the crew onboard R/V Nereus and Joel White for their help during deployment and retrieval of the seaweed lines.
Funding
This study is supported by The Swedish Foundation for Strategic Environmental Research MISTRA (grant no. 2013/75) and The Swedish Research Council Formas (grant no. 213-2013-92).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 1077 kb)
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Visch, W., Nylund, G.M. & Pavia, H. Growth and biofouling in kelp aquaculture (Saccharina latissima): the effect of location and wave exposure. J Appl Phycol 32, 3199–3209 (2020). https://doi.org/10.1007/s10811-020-02201-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10811-020-02201-5