Abstract
Protecting biodiversity of agricultural lowlands is one of the main topics of European nature conservation. Understanding the relationship and interaction between cultivated and other habitat types is necessary since arable lands cover a significant part of the landscape, but former studies rather focus on the remaining semi-natural sites. Orthoptera assemblages, which are sensitive indicators of habitat structure and conservation value, were studied in a traditional intensively used agricultural area (it has been cultivated for several decades or even 250 years) to assess how arable land and ruderal linear habitats together can preserve insect diversity. Twenty-nine Orthoptera species, including protected Gampsocleis glabra, Acrida ungarica and Celes variabilis were recorded. We identified four assemblages and their character species reflecting intensity of land use. Assemblages of agricultural sites differed from the assemblages of semi-natural habitats used as control group. Revealing their quantitative composition, ratios of lifeforms and ecotypes, character species, and use of Grasshopper Conservation Indexes (GCI) helped to identify habitat types that can effectively preserve natural insect diversity, even under long-term intensive land use regime.
Implications for conservation
Considering our findings, the role of linear ruderal habitats and less intensively cultivated arable lands covered by alfalfa and red clover in maintaining of biodiversity is higher than it was formerly assumed. Increasing ratio of these less intensive cultures (at least to 5%, optimally 8–10%) and its connection with each other and linear ruderal habitats may provide temporary habitats and corridors for metapopulations living in mosaic landscape. The local greening programs can help Orthoptera conservation as well through supporting the production of these cultures. Complex landscape- and crop rotation planning, that regard the spatial and temporal distribution, and ratio of agriculture landscape elements may become an effective tool for biodiversity conservation and forming sustainable farming systems.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The majority of the European grasslands have been transformed into secondary semi-natural and agricultural habitats due to long-term agricultural land use, thus one of the most important tasks of nature conservation is to protect the remaining biodiversity of these transformed agricultural landscapes (Sutherland 2004; Batáry et al. 2015). Maintaining and protecting these ecosystems is important not only for nature conservation, but for agriculture as well, because of the outstanding role of ecosystem-services it provides (Boetzl et al. 2021).
The percentage of agricultural lands is 57% in Hungary, containing 15% grasslands and 81% croplands (KSH 2021). In order to achieve efficient protection and sustainable use of these ecosystems, the natural and even conservation value of their elements and the way they function should be known.
Orthopterans are especially sensitive and widely used indicators of grassland structure and naturalness (Marshall 2010; Benton 2012; Cherrill 2015). In the temperate zone, their assemblages are relatively species-rich and play an important role in the food chain especially as herbivores (Gardiner et al. 2005). Composition of Orthoptera assemblages refer especially to the vegetation structure of their habitats. Since the species composition, dominance rank structure and changes of life form spectra of their assemblages sensitively and quickly follow changes of their habitats and they can used as indicators of grassland naturalness as well, they are widely used in grassland ecology and conservation planning (Noss 1990; Spellerberg 1991; Pearson 1994; Kisbenedek 1997; Báldi and Kisbenedek 1997; Batáry et al. 2007; Kenyeres 2011; Báldi et al. 2013; Torma et al. 2014; Kenyeres et al. 2020).
There are numerous studies on the Orthoptera fauna and assemblages living in natural and semi-natural habitat patches nested within agricultural fields. The role of these habitats in maintaining diversity is mostly known, and the effect of their vegetation structure, isolation and area and intensity and way of habitat use on diversity were also studied. Higher landscape diversity and lower intensity of habitat use can maintain larger species-richness. Ditch banks, burial mounds (kurgans) and grassy field margins preserve high species-richness and later can also buffer the negative effects of pesticide use (Gause 1930; Marshall et al. 2006; Torma et al. 2018; Löffler et al. 2019; Deák et al. 2020, 2021; Szanyi et al. 2021).
The natural and semi-natural habitat patches are spread in the agricultural matrix (e.g. 70% in Hungarian lowland; Berényi 2011) containing arable lands and different parts of crop production infrastructure (different kind of roads and channels etc.). In the most intensively used areas this matrix stands alone without remarkable patches of natural and semi-natural habitats. To understand of the role and relationship of the habitat types of this matrix is necessary for protection of both its own ecosystem and the diversity of nested natural habitat patches (Vandermeer and Perfecto 2007; Syiem et al. 2018). The Orthoptera assemblages of the cultivated fields are less known, despite their outstanding role in the functioning of the landscape’s ecosystem (Forman and Gordon 1981; Donald and Evans 2006; Melliger et al. 2017). The study of assemblages living in arable lands can reveal the role of different cultures and cropping systems in the maintenance of insect diversity and also provide useful data for the conservation of remaining natural habitat patches.
The Orthoptera assemblages of traditionally intensively used agricultural landscapes were studied in the Carpathian Lowland (East Hungary) using semi-natural habitats (pastures and hayfields) as control areas to reveal how main cultivars and other elements of the “agricultural matrix” preserve the original fauna. During a 3-year study, assemblages of different landscape elements and their quantitative character species were described and the effect of land use intensity as well as the cropping system on the species-richness and composition of Orthoptera assemblages was also evaluated. We aimed to designate habitat types, cultivars and cropping system that can preserve higher insect diversity and can be suggested for landscape engineers and farmers to forming a sustainable farming system.
Materials and methods
Study area
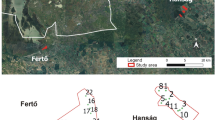
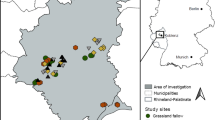
Studies were carried out in the northern part of the Carpathian Lowland, in the surroundings of Tiszaújváros, Sajószöged, Sajóörös and Kesznyéten in 2018–2020. The studied habitats have been used in the same way they are used nowadays for at least 80 years, but most arable lands have been regularly cultivated for 250 years based on military maps (https://maps.arcanum.com/hu/). In 2018, study sites were designated into three main habitat types: (1) semi-natural habitats: hayfields and pastures; (2) arable lands: red clover, alfalfa, sunflower, maize fields and stubble-fields; (3) linear ruderal habitats: country roadsides and channel banks, dirt roads and its grassy edges. In case of each habitat type 4–4 sampling sites (in total: 36 sites) were sampled each year of the three-year study. In 2019 and 2020 wheat field also appears in arable lands due to crop rotation (Table 1, Fig. 1).
Orthoptera sampling
Samplings were made by combined methods of sweep netting and direct search, which complement each other in Orthoptera studies and can provide comparable samples independently the vegetation structure (Nagy et al. 2007). Sweep net samples were taken by 200 sweeps with a net of 50 cm in diameter. The net was emptied after every 100 sweeps to protect sampled individuals. Sampling was done in calm and sunny weather considering the daily activity of the sampled species. The direct search was made by walking along parallel transects for 10 min at each sampling site and specimens were recorded based on both visual and acoustic detections along 1.5 m wide transects. Where the sweep net could not be used (e.g. in developed maize and sunflower cultures), a 30 min direct search was carried out. Samples were taken twice a year in each site and year in early June and late August. Data of the two samplings were summarized by sites.
Adults were separated from the samples and were identified at species level according to Harz (1957, 1960, 1975). The Orthoptera species file database was used (Cigliano et al. 2020) for nomenclature.
Data analysis
The Orthoptera fauna of the studied region was characterised by species-richness, relative frequency of species, life form and faunal types.
Principal Coordinate (PCoA) and cluster analysis with Bray–Curtis index were used to compare the sites based on the composition of Orthoptera assemblages. The Ward-Orlóci agglomeration method was applied for clustering (Podani 1997a). We used the relative frequency of species by sites for multivariate analyses in the SynTax 2000 software (Podani 1997b). Since orthopterans were not recorded in site 13 during the study, the data of 35 sites were involved in the analysis.
The sampling sites were categorized a priori into three main land use types (semi-natural, arable land, linear ruderal habitats) that also means different land use intensity, vegetation structure, degradation and disturbance. The correspondence between these a priori categories and the groups formed based on the multivariate analyses was studied. Considering crop rotation and changes in habitat use, intensity of land use of a given site was characterised by the mean intensity of habitat use in the three consecutive years. The intensity value (1–6) of the studied culture and/or habitat types depends on the vegetation structure, disturbance, using of pesticides and intensity of cultivation: pastures and hayfields = 1, country roadsides and dirt roadsides = 2, alfalfa and red clover fields = 3, stubble-fields = 4, wheat fields = 5, sunflower and maize fields = 6 (Table 1).
Assemblage types established by the multivariate analyses were characterised and compared based on their total species-richness, mean number of species, mean number of individuals, and mean relative frequencies of life and ecotype forms. To characterise and compare assemblages, Whittaker’s index (S/α; S = number of species, α = mean number of species) was also calculated for both assemblage types and the whole sample (Whittaker 1960). Species were grouped into life and ecotype forms according to Ingrisch and Koehler (1998). The mean intensity of land use of the sites grouped in the same type was also calculated and compared.
To evaluate the conservation value of the habitat types, a modified grasshopper conservation index (GCI’) was used based on Matenaar et al. (2015) and Szanyi et al. (2021). In the case of the original index, Matenaar et al. (2015) used three parameters: endemism, dispersal capacity and rarity, and each of them were grouped into three classes. The parameters were summed for each species and divided by nine (the maximum value) to obtain a GCI value between zero and one. Considering the distribution of the species living in the studied assemblages (there were no endemic species in the studied sites), a modified index was used, same as that of Szanyi et al (2021). Instead of endemism, the rarity of the species in European regions were used based on distribution data of Heller et al. (1998): distributed in all 12 European regions (= 1), distributed in 9–11 regions (= 2), distributed in 6–8 regions (= 3), distributed in 1–5 regions (= 4). In the case of dispersal capacity and rarity, the original method was followed but the parameters were grouped into four categories. Considering rarity, the categorization of Nagy and Rácz (2007) was followed. For dispersal capacity, the groups of well flying (= 1), poorly flying (= 2), wing-dimorphic (= 3) (also contains mesopterous mainly flightless species) and flightless (= 4) (contains wingless, macropterous and mesopterous flightless species) species were used. The local rarity was measured upon the spatial constancy (SC) of species in the studied 36 sites. A species was considered as common (= 1; SC > 0.6), frequent (= 2; SC = 0.3–059), low frequent (= 3; SC = 0.1–0.29) and rare (= 5; SC < 0.1). The three parameters were summed and divided by 12 (the maximum value) to obtain a GCI” value between zero and one. The GCI” values of the study sites were determined as a sum of the values of the species of the given site. Habitat types were characterised by the mean number of the GCI” values of sites. The modified standardized grasshopper conservation index (GCIn’) was also calculated for sites by dividing GCI’ by the number of species on the given site. While the GCI value belongs both to species number and value of the species, the GCIn is not influenced by species-richness (Matenaar et al. 2015).
Since our data did not fulfil the requirements of parametric tests, comparison of the assemblage types were made with Kruskall-Wallis nonparametric test and when it showed significant differences, pairwise comparisons were made with Mann-Whitey U test using Statistica 7 program package. The assumptions of the parametric tests were tested with Levene-test (homogeneity of variances) and Q-Q plots (normal distribution).
Correlation between intensity of land use and different parameters of assemblages (number of caught individuals, number of caught species and GCI’) was analysed with Pearson correlation analysis.
Quantitative character species (“indicator” species) of the assemblages were identified using the IndVal method (Dufrêne and Legendre 1997). The species were hierarchically classified according to their fidelity within the site groups. The IndVal (IV) value is highest (100) when the given species is present in all samples of the given group and is also exclusive for this group of samples. The program calculates the IV values of each species at each hierarchy level of the clustering. The indicator value is given by the maximum value. The significance of IV values was determined by randomisation (1000 iterations). Analysis was carried out with the IndVal program.
Results
During the investigation 29 Orthoptera species (11 Ensifera and 18 Caelifera) were caught. Most of the species belonged to the pratinicol life form (90.67%) while the ratio of geofilic species was remarkably lower (7.40%) (Tables 2 and 4).
The most common species are all widely distributed eurytipic species of the Carpathian lowlands. The potential agricultural pest Calliptamus italicus reached larger local abundance and density than at a regional scale (Arnóczkyné et al. 2020) and the regionally rare Chorthippus dichrous showed large local abundance and spatial constancy. Gampsocleis glabra and Celes variabilis are rare and protected, while Acrida ungarica is a protected member of the Hungarian fauna (Nagy and Rácz 2007b; Heller et al. 1998; Rácz 1998a) (Table 2).
The total number of caught Orthopterans was 2121, 21.5% of which were larvae. The total number of adults identified at species level was 1664. In the whole sample C. italicus (14.7%), P. parallelus (13.1%), C. dorsatus (12.7%), G. brunneus (11.6%), and O. rufipes (9.9%) were the five most abundant species. The relative frequency of C. oschei (7.0%) and R. roeselii (5.2%) also exceeded 5%.
During quantitative multivariate analysis of the Orthoptera assemblages, four site groups were identified along a gradient. The 1st PCoA axis represented a gradient from semi-natural (I) to intensively used arable lands (IV), while the 2nd PCoA axis represented a gradient between species-poor intensive arable lands (IV) and the group of moderately species-rich arable lands and roadsides (III) (Fig. 2.). The hierarchy of these groups is shown by the cluster analysis (Fig. 3.). Values of Whittaker’s index support the result of multivariate analysis. It showed the homogenous character of semi-natural and less intensively used agricultural habitats and large heterogeneity of assemblages living on intensively used arable lands (Table 4).
The correspondence between the a priori and cluster groups was total in case of semi-natural sites (I). The a priori group of linear ruderal habitats (e.g. roadsides) split between the two transitional group of agricultural habitats (II and III). These transitional groups also contained 6 and 5 arable lands respectively, while the remaining 9 intensively used arable lands formed a heterogeneous group (IV) at the end of the gradient (Table 3 and Fig. 2).
The species-richness of assemblages decreased from group of semi-natural sites to intensive arable lands following the gradient shown on the 1st axis of PCoA. The mean species richness and abundance of Orthoptera assemblages showed the same trend, and the semi-natural sites were the most species-rich ones. The species-richness of semi-natural and diverse agricultural habitats was significantly higher than that of intensive arable land, while less diverse agricultural habitats had intermediate values (Table 4).
The frequency of different life forms showed a similar pattern in case of all life forms excluding geophilic species, that showed the highest relative frequency in intensive arable lands. The difference was significant only in comparison with diverse agricultural lands, where the value was also significantly lower than in semi-natural sites. Parallelly the ratio of pratinicol species was lowest in intensive arable lands, but in this case, there were no significant differences. Regarding ecotypes the relative frequency of hygrophilous species continuously decrease from semi-natural habitats to more and more intensively used agricultural lands and between the intensively cultivated arable lands and semi natural sites it was significant difference. In the same time the ratios of moderately-xerophilous and xerophilous species were higher in more intensively used habitat types (Table 4).
The grasshopper conservation index (GCI’) showed the highest conservation value of Orthoptera assemblages inhabiting semi-natural sites and the value decreased along the gradient shown by the 1st axis of PCoA. Conservation value of intensive arable lands was significantly lower than that of semi-natural and diverse agricultural lands, while the less diverse agricultural lands showed an intermediate value. The mean standardized GCIn’ showed the same trend, but in this case only the semi-natural sites had significantly larger conservation value than intensive arable lands and the other two types showed intermediate values (Table 4).
The mean value of land use intensity showed significant negative correlation with the number of caught specimens (r = − 0.7429 p < 0.0001), species richness (r = − 0.767, p < 0.0001) and the value of the grasshopper conservation index (GCI’; r = − 0.7702, p < 0.0001) of the studied sites.
According to the results of the IndVal analysis, the most common species of the studied assemblages were Chorthippus brunneus, C. biguttulus and Omocestus haemorrhoidalis. The semi-natural and diverse agricultural habitats had ten mutual characteristic species (Pseudochorthippus parallelus, Chorthippus dorsatus, Omocestus rufipes, Roeseliana roeselii, Euchorthippus declivus, Conocephalus fuscus, Chorthippus dichrous, Mecostethus parapleurus, Ruspolia nitidula and Bicolorana bicolor), while the semi-natural sites alone were characterised by high fidelity and specificity of Chorthippus oschei, Gampsocleis glabra, Aiolopus thalassinus and Dociostaurus brevicollis. The diverse agricultural sites had no own character species. Contrarily, the less diverse agricultural habitats and intensive arable lands had only one mutual character species (Gryllus campestris). The less diverse agricultural sites could be characterised by Calliptamus italicus, and intensive arable lands had no own character species (Table 5.).
Discussion and conclusions
The Orthoptera assemblages of common semi-natural grasslands and agricultural habitats including fields of the most important crops (maize, sunflower, wheat, alfalfa, and red clover) and linear ruderal habitats of the Carpathian Lowland were described. The sampled 29 species represented significant part both the regional (43 species; Arnóczkyné and Nagy 2021) and Hungarian Orthoptera fauna (Nagy 2003). Samples taken in a less studied area provide data on three protected species of which Gampsocleis glabra and Acrida ungarica appeared also in agricultural sites and Celes variabilis inhabited only semi-natural hayfields. The potential pest Calliptamus italicus and widely distributed Chorthippus dichrous showed larger local, than regional abundances, that provides special character of the studied moderately species rich assemblages (9.2 species/site in average).
The most abundant and frequent species of the local fauna belonged to the grass living pratinicol life form represented 90.67% of the sampled material. The relative frequency value of geophilic species (7.4%) was similar with average value of assemblages lives in xeric open grassland of the wider surroundings (Rácz 1998b, 2001).
Using multivariate analysis, four types of assemblages characteristic to the studied habitat types were identified that corresponded rather with the fine scale differences in intensity of habitat use than the a priori categories of the studied habitats formed based on their naturalness. Szanyi et al. (2021) also proved strong effect of the intensity of the land use on composition of Orthoptera assemblages in the Nort-Eastern part of the Carpathian Lowland (West Ukraine). The correspondence between the a priori and cluster groups was univoque only in the case of semi-natural sites, while the arable lands and linear ruderal habitats separated along the 1st PCoA axis based on their species-richness and intensity of land use. The studied agricultural habitats have own characteristic assemblages that evolved under decades of habitat use and differ from the assemblages of semi-natural habitats of the same region. The relationship between insect assemblages of linear ruderal habitats and semi-natural sites is well studied (Ahmed et al, 2021; New et al. 2021), however the assemblages of intensively cultivated arable lands formerly were not take in consideration.
Most of the diversity and natural composition of the local assemblages represented by the studied semi-natural sites were preserved by the species-rich linear ruderal habitats (e.g. roadsides) and less intensively cultivated alfalfa and red clover fields. Although naturalness of these habitats measured with GCI values was lower than that of semi-natural sites, however comparing with intensively used arable lands (maize and sunflower fields) they had significantly higher natural value. The role of linear ruderal habitats (roadsides, ditch banks and other linear landscape elements) in preserving Orthoptera diversity is well studied (Berggren et al. 2001, 2002; Vadkerti and Szövényi 2005; Eriksson et al. 2013; Torma et al. 2018). Studies mostly focused on the wildlife of natural and semi-natural patches nested within intensively cultivated arable lands (Duelli and Obrist 2003; Rand et al. 2006; Braschler et al. 2009; Jauker et al. 2009; Marini et al. 2010; Badenhausser and Cordeau 2012; Bátory et al. 2016; Hall et al. 2022; Martínez-Núñez et al. 2022; Thorn et al. 2022). The distribution of sites a priori categorized as linear ruderal habitats (country roadsides, dirt roads) between cluster groups showed that the naturalness and diversity of these habitats and their assemblages may be affected by various factors e.g. the quality of neighbouring habitats, isolation and location, as it was found also in former studies (Torma et al. 2018; Rebrina et al. 2022).
Conversely, the importance of the cultivated areas is less known. Only the effect of the less intensive Fabaceae fields involved in the green direct payment scheme (“greening”) – part of the common agricultural policy of the European Union –, (e.g. alfalfa and red clover) were studied. Sites of greening programs are often designated in areas with originally higher natural value, thus the evaluation of their effect is quite difficult and often subject to debate (Kleijn and Sutherland 2003; Uthes and Matzdorf 2013; Batáry et al. 2015). Since our agricultural sites are not involved in greening programs and each showed average natural value at most, the positive effect revealed on Orthoptera assemblages was not biased by the above-mentioned effect.
The intensive land use in the sunflower, maize and wheat fields led to less diverse insect assemblages with lower species diversity and conservation value. Three-year monoculture of maize and/or sunflower resulted in especially low average species-richness (only 4.25 species/site) and in some cases total absence of orthopterans. Fumí et al. (2021) also blamed the change of land use for the decrease of Orthoptera diversity. The increasing intensity of land use causes decreasing diversity of lepidopterans as well (Habel et al. 2019), while Onga et al. (2022) draw the same consequence assessing results of 199 studies.
Beyond linear ruderal habitats, less intensively cultivated alfalfa and red-clover fields can also maintain relatively species-rich Orthoptera assemblages with high conservation value. The grass-like structure of stubble and wheat fields can also provide a suitable habitat, while the intensive and especially the monocultural sunflower and maize fields are not suitable for orthopterans.
The ratio and spatial pattern of the different landscape elements (considering the grown cultures as well) greatly affect the diversity, composition and naturalness of the Orthoptera assemblages. Since they are sensitive indicators, this effect may be assumed in the case of many other grass-living insect assemblages. This showed that the role of the agricultural landscape elements is higher than it was formerly assumed. Changes of the ratio and proper planning of the spatial distribution of these constant (roadsides etc.) and temporal (different cultures) landscape elements and considering them as a complex system can be an effective tool, as it was partly suggested by Sirami et al. (2019). The augmentation of the ratio of less intensive (3–4 year) cultures and maintenance of the linear ruderal habitats together may increase the abundance and diversity of orthopterans, since these cultures can serve as temporary habitats and corridors or stepping stones between remained natural and seminatural habitats. In this way an enough diverse agricultural landscape can maintain species rich Orthoptera assemblages, however it is unable to preserve stenotopic characteristic species of natural habitats, as it was formerly proven in case of birds as well (Syiem et al. 2018). Nevertheless, results prove that this strategy can be effective even in traditionally intensively used areas, such as the Hungarian Lowland where most of the species-richness and conservation value could be maintained over the last decades.
Code availability
During the analysis SynTax 2000, and IndVal 2.0 were used, no computer code was created.
References
Ahmed KSD, Volpato A, Day MF, Mulkeen CJ, O’Hanlon A, Carey J, Williams C, Ruas S, Moran J, Rotchés-Ribalta R, ÓhUallacháin D, Stout JC, Hodge S, White B, Gormally MJ (2021) Linear habitats across a range of farming intensities contribute differently to dipteran abundance and diversity. Insect Conserv Divers 14(3):335–347. https://doi.org/10.1111/icad.12455
Arnóczkyné Jakab D, Szanyi Sz, Nagy A (2020) Az olasz sáska (Caelifera: Calliptamus italicus Linnaeus, 1758) - Újra célkeresztben? Növényvédelem 56(9):405–411 (in Hungarian)
Arnóczkyné Jakab D, Nagy A (2021) Data on the Orthoptera fauna of characteristic agricultural landscape in the Carpathian Lowland. Acta Agraria Debreceniensis. 1:25–34. https://doi.org/10.34101/actaagrar/1/8495
Badenhausser I, Cordeau S (2012) Sown grassstrip – a stable habitat for grasshoppers (Orthop-tera: Acrididae) in dynamic agricultural landscapes. Agric Ecosyst Environ 159:105–111. https://doi.org/10.1016/j.agee.2012.06.017
Batáry P, Orci KM, Báldi A, Kleijn D, Kisbenedek T, Erdős S (2007) Effects of local and landscape scale and cattle grazing intensity on Orthoptera assemblages of the Hungarian Great Plain. Basic Appl Ecol 8:280–290. https://doi.org/10.1016/j.baae.2006.03.012
Batáry P, Dicks LV, Kleijn D, Sutherland WJ (2015) The role of agri-environment schemes in conservation and environmental management. Conserv Biol 29(4):1006–1016. https://doi.org/10.1111/cobi.12536
Báldi A, Batáry P, Kleijn D (2013) Effects of grazing and biogeographic regions on grassland biodiversity in Hungary, analysing assemblages of 1200 species. Agric Ecosyst Environ 166:28–34. https://doi.org/10.1016/j.agee.2012.03.005
Báldi A, Kisbenedek T (1997) Orthopteran assemblages as indicators of grassland naturalness in Hungary. Agric Ecosyst Environ 66:121–129. https://doi.org/10.1016/S0167-8809(97)00068-6
Bátori Z, Körmöczi L, Zalatnai M, Erdős L, Ódor P, Tölgyesi C, Margóczi K, Torma A, Gallé R, Cseh V, Török P (2016) River Dikes in agricultural landscapes: the importance of Secondary habitatsin maintaining landscape–scale diversity. Wetlands 36:251–264. https://doi.org/10.1007/s13157-016-0734-y
Benton T (2012) Grasshoppers and crickets. Harper Collins, London
Berényi I (2011) Mezőgazdaság. In: Kocsis, K., Schweitzer F. (Eds.), Magyarország térképekben. Magyar Tudományos Akadémia, Földrajztudományi Kutatóintézet, Budapest, pp 148–160. (in Hungarian)
Berggren Å, Carlson A, Kindvall O (2001) The effect of landscape composition on colonization success, growth rate and dispersal in introduced bush-crickets Metrioptera roeseli. Ecology 70:663–670. https://doi.org/10.1046/j.1365-2656.2001.00525.x
Berggren Å, Carlson A, Kindvall O (2002) Effect of corridors and habitat edges on dispersal behavior, movement rates, and movement angles in Roesel’s bush-cricket Metrioptera roeseli. Conserv Biol 16:1562–1569. https://doi.org/10.1046/j.1523-1739.2002.01203.x
Boetzl FA, Krauss J, Heinze J, Hoffmann H, Juffa J, König S, Krimmer E, Prante M, Martin EA, Holzschuh A, Steffan-Dewenter I (2021) A multitaxa assessment of the effectiveness of agri-environmental schemes for biodiversity management. PNAS 18(10):e2016038118. https://doi.org/10.1073/pnas.2016038118
Braschler B, Marini L, Thommen GH, Baur B (2009) Effects of small-scale grassland fragmen-tation and frequent mowing on population densityand species diversity of orthopterans: a long-term study. Ecol Entomol 34:321–329. https://doi.org/10.1111/j.1365-2311.2008.01080.x
Cherrill A (2015) Large-scale spatial patterns in species richness of Orthoptera in the Greater London Area, United Kingdom: relationships with land cover. Landsc Res 40:476–485. https://doi.org/10.1080/01426397.2014.902922
Cigliano MM, Braun H, Eades DC, Otte D (2020) Orthoptera species file. Version 5.0/5.0. http://Orthoptera.SpeciesFile.org. Accessed 28 February 2021
Deák B, Valkó O, Nagy DD, Török P, Torma A, Lőrinczi G, Kelemen A, Nagy A, Bede Á, Mizser Sz, Csathó AI, Tóthmérész B (2020) Habitat islands outside nature reserves, threatened biodiversity hotspots of grassland specialist plant and arthropod species. Biol Conserv. https://doi.org/10.1016/j.biocon.2019.108254
Deák B, Bede Á, Rádai Z, Tóthmérész B, Török P, Nagy DD, Torma A, Lőrinczi G, Nagy A, Mizser Sz, Kelemen A, Valkó O (2021) Different extinction debts among plants and arthropods after loss of grassland amount and connectivity. Biol Conserv. https://doi.org/10.1016/j.biocon.2021.109372
Donald PF, Evans AD (2006) Habitat connectivity and matrix restoration: the wider implications of agri-environment schemes. J Appl Ecol 43:209–218. https://doi.org/10.1111/j.1365-2664.2006.01146.x
Duelli P, Obrist MK (2003) Regional biodiversityin an agricultural landscape: the contribution ofseminatural habitat. Basic Appl Ecol 4:129–138. https://doi.org/10.1078/1439-1791-00140
Dufrêne M, Legendre P (1997) Species assemblages and indicator species: the need for a flexibile asymmetrical approach. Ecol Monogr 67(3):345–366. https://doi.org/10.1890/0012-9615(1997)067[0345:SAAIST]2.0.CO;2
Eriksson A, Low M, Berggren Å (2013) Influence of linear versus network corridors on the movement and dispersal of the bush-cricket Metrioptera roeseli (Orthoptera: Tettigoniidae) in an experimental landscape. Eur J Entomol 110(1): 81–86. https://doi.org/10.14411/eje.2013.010
Forman RTT, Gordon M (1981) Patches and structural components for landscape ecology. Bioscience 31:733–740. https://doi.org/10.2307/1308780
Fumy F, Kämpfer S, Fartmanna T (2021) Land-use intensity determines grassland Orthoptera assemblage composition across a moisture gradient. Agric Ecosyst Environ 315:107424. https://doi.org/10.1016/j.agee.2021.107424
Gardiner T, Hill J, Chesmore D (2005) Review of the methods frequently used to estimate the abundance of Orthoptera in grassland ecosystems. J Insect Conserv 9:151–173. https://doi.org/10.1007/s10841-005-2854-1
Gause GF (1930) Studies on the ecology of the Orthoptera. Ecology 11(2):307–325. https://doi.org/10.2307/1930266
Habel JC, Ulrich W, Biburger N, Seibold S, Schmitt T (2019) Agricultural intensification drives butterfly decline. Insect Conserv Divers 12(4):289–295. https://doi.org/10.1111/icad.12343
Hall MA, Nimmo DG, Bennett AF (2022) Birds and insects respond differently to combinations of semi-natural features in farm landscapes. J Appl Ecol. https://doi.org/10.1111/1365-2664.14266
Harz K (1957) Die Geradflügler Mitteleuropas. VEB Gustav Fischer Verlag, Jena. (in German)
Harz K (1960) Geradflügler oder Orthopteren (Blattodea, Mantodea, Sal-tatoria, Dermaptera). In: Dahl F (Eds.) Die tierwelt Deutschlands und der angrenzenden meeresteile nach ihren merkmalen und nach ihrer lebensweise. 46. Gustav Fischer, Teil Jena, p. 232. (in German)
Harz K (1975) Die Orthopteren Europas. Junk Publishers, The Hague (in German)
Heller KG, Korsunovskaya O, Ragge DR, Vadenina V, Willemse F, Zhantiev RD, Frantsevich L (1998) Check-List of European Orthoptera. Articulata 7:1–61
Ingrisch S, Koehler G (1998) Die Heuschrecken Mitteleuropas. Die Neue Brehm–Bücherei Bd. 629, Westarp Wissenschaften, Magdeburg
Jauker F, Diekötter T, Schwarzbach F, Wolters V (2009) Pollinator dispersal in an agricultural matrix: opposing responses of wild bees and hoverflies to landscape structure and distance from main habitat. Landsc Ecol 24(4):547–555. https://doi.org/10.1007/s10980-009-9331-2
Kenyeres Z (2011) Természetes és természetközeli gyepek egyenesszárnyú–együttesei (Orthoptera) a Bakonyvidéken. [Orthopteran assemblages (Orthoptera) of natural and semi-natural grasslands in the Bakony Region]. Természetvédelmi Közlemények. 17:42–56 (in Hungarian)
Kenyeres Z, Szabó Sz, Takács G, Szinetár Cs (2020) Orthoptera assemblages as indicators for the restoration of sand grassland networks. North-Western J Zool 16(1):7–14
Kisbenedek T (1997) Egyenesszárnyúak, Orthoptera. in: Forró, L. (Eds.), Nemzeti Biodiverzitás–monitorozó Renszer V., Rákok, szitakötők és egyenesszárnyúak. (National Biodiversity Monitoring System V., Crustacea, Odonata, Orthoptera) Magyar Természettudományi Múzeum, Budapest, pp 55–81. https://termeszetvedelem.hu/_user/downloads/biomon/V.%20R%C3%A1kok,%20szitak%C3%B6t%C5%91k%20%C3%A9s%20egyenessz%C3%A1rny%C3%BAak.pdf
Kleijn D, Sutherland WJ (2003) How effective are European agri-environment schemes in conserving and promoting biodiversity? J Appl Ecol 40:947–969. https://doi.org/10.1111/j.1365-2664.2003.00868.x
KSH 2021: Földhasználat művelési ágak és gazdaságcsoportok szerint (1990) (Land use according to cultivation and economic groups). https://www.ksh.hu/docs/hun/xstadat/xstadat_eves/i_omf001a.html. Accessed 11 May 2022
Löffler F, Poniatowski D, Fartmann T (2019) Orthoptera community shifts in response to land-use and climate change, Lessons from a long-term study across different grassland habitats. Biol Conserv 236:315–323. https://doi.org/10.1016/j.biocon.2019.05.058
Marini l, Bommarco R, Fontana P, Battisti A (2010) Disentangling effects of habitat diversity and area on orthopteran species with contrasting mobility. Biol Conserv 143(9):2164–2171. https://doi.org/10.1016/j.biocon.2010.05.029
Marshall EJP, West TM, Kleijn D (2006) Impacts of an agri-environment field margin prescription on the flora and fauna of arable farmland in different landscapes. Agric Ecosyst Environ 113:36–44. https://doi.org/10.1016/j.agee.2005.08.036
Marshall JA (2010) Grasshoppers, crickets and allied insects. In: Maclean N (ed) Silent Summer. Cambridge University Press, Cambridge, The state of wildlife in Britain and Ireland, pp 531–539
Martínez-Núñez C, Kleijn D, Ganuza C, Heupink D, Raemakers I, Vertommen W, Fijen TPM (2022) Temporal and spatial heterogeneity of semi-natural habitat, but not crop diversity, is correlated with landscape pollinator richness. J Appl Ecol 59(5):1258–1267. https://doi.org/10.1111/1365-2664.14137
Matenaar D, Bazelet C, Hochkirch A (2015) Simple tool for the evaluation of protected areas for conservation of grasshoppers. Biol Conserv 192:192–199. https://doi.org/10.1016/j.biocon.2015.09.023
Melliger RL, Rusterholz HP, Baur B (2017) Habitat- and matrix-related differences in species diversity and trait richness of vascular plants, Orthoptera and Lepidoptera in an urban landscape. Urban Ecosyst 20:1095–1107. https://doi.org/10.1007/s11252-017-0662-5
Nagy A, Rácz IA (2007) A hazai Orthoptera fauna 10 × 10 km–es UTM alapú adatbázisa. (10 × 10 km UTM-based database of the Hungarian Orthoptera fauna. In: Kövics G, Dávid I (Eds.) Tiszántúli Növényvédelmi Fórum előadások, Proceedings. Debreceni Egyetem, Debrecen, pp 189–198
Nagy A, Sólymos P, Rácz IA (2007) A test on the effectiveness and selectivity of three sampling methods frequently used in orthopterological field studies. Entomol Fennica 18(3):149–159. https://doi.org/10.33338/ef.84392
Nagy B (2003) A revised check-list of Orthoptera-species of Hungary supplemented by Hungarian names of grasshopper species. Folia Entomol Hungarica 64:85–94
New TR, Sands DPA, Taylor GS (2021) Roles of roadside vegetation in insect conservation in Australia. Austral Entomol 60(1):128–137. https://doi.org/10.1111/aen.12511
Noss RF (1990) Indicators for monitoring biodiversity: a hierarchical approach. Conserv Biol 4:355–364. https://doi.org/10.1111/j.1523-1739.1990.tb00309.x
Ogan S, Paulus C, Froehlich C, Renker C, Kolwelter C, Schendzielorz M, Danielczak A, Müller K, Eulering H, Hochkirch A (2022) Re-surveys reveal biotic homogenization of Orthoptera assemblages as a consequence of environmental change. Divers Distrib 28(9):1795–1809
Pearson DL (1994) Selecting indicator taxa for the quantitative assessment of biodiversity. Philos Trans R Soc B 345:75–79. https://doi.org/10.1098/rstb.1994.0088
Podani J (1997a) Bevezetés a többváltozós biológiai adatfeltárás rejtelmeibe. (An introduction to the mysteries of multivariate biological data analysis) Scientia. Budapest. p 412
Podani J (1997b) SYN–TAX 5.1: A new version for PC and Macintosh computers. Coenoses. 12:149–152
Rácz I (1998a) Biogeographical survey of the Orthoptera Funa in Central Part of the Carpathian Basin (Hungary): fauna types and community types. Articulata. 13(1):53–69
Rácz I (1998b) Life form spectra of Orthoptera fauna in alkaline grassland. Tiscia 31:35–39
Rácz I (2001) Egyenesszárnyú együttesek életforma-spektrumának változása a száraz és félszáraz gyepek struktúrájának függvényében. [Change in the life-form spectra of Orthoptera communities regarding tot he structure of dry and semi-dry grasslands in Hungary]. Állattani Közlemények 86:29–56 (in Hungarian)
Rand TA, Tylianakis JM, Tscharntke T (2006) Spillover edge effects: the dispersal of agriculturally subsidized insect natural enemies into adjacent natural habitats. Ecol Lett 9(5):603–614. https://doi.org/10.1111/j.1461-0248.2006.00911.x
Rebrina F, Reinhold K, Tvrtković N, Gulin V, Brigić A (2022) Vegetation Height as the Primary Driver of Functional Changes in Orthopteran Assemblages in a Roadside Habitat. InSects 3(7):572. https://doi.org/10.3390/insects13070572
Syiem BLN, Goswami VR, Vasudev D (2018) “In a tree by the brook, there’s a songbird who sings”: Woodlands in an agricultural matrix maintain functionality of a wintering bird community. PLoS ONE 13(8):e0201657. https://doi.org/10.1371/journal.pone.0201657
Sirami C, Gross N, Baillod AB, Bertrand C, Carrié R, Hass A, Henckel L, Miguet P, Vuillot C, Alignier A, Girard J, Batáry P, Clough Y, Violle C, Giralt D, Bota G, Badenhausser I, Lefebvre G, Gauffre B, Vialatte A, Calatayud F, Gil-Tena A, Tischendorf L, Mitchell S, Lindsay K, Georges R, Hilaire S, Recasens J, Solé-Senan XO, Robleño I, Bosch J, Barrientos JA, Ricarte A, Marcos-Garcia MÁ, Miñano J, Mathevet R, Gibon A, Baudry J, Balent G, Poulin B, Burel F, Tscharntke T, Bretagnolle V, Siriwardena G, Ouin A, Brotons L, Martin J-L, Fahrig L (2019) Increasing crop heterogeneity enhances multitrophic diversity across agricultural regions. PNAS 16:33. https://doi.org/10.1073/pnas.1906419116
Spellerberg IF (1991) Monitoring Ecological Change. Cambridge University Press, Cambridge, p 344
Sutherland WJ (2004) A blueprint for the countryside. Ibis 146:230–238. https://doi.org/10.1111/j.1474-919X.2004.00369.x
Szanyi Sz, Potish L, Rácz IA, Varga Z, Nagy A (2021) Effect of dramatic alteration of landscape structure on the Orthoptera assemblages of Transcarpathian lowland meadows (West Ukraine). J Insect Conserv 25:759–768. https://doi.org/10.1007/s10841-021-00341-8
Simon Thorn S, König S, Fischer-Leipold O, Gombert J, Griese J, Thein J (2022) Temperature preferences drive additive biotic homogenization of Orthoptera assemblages. Biol Lett 18:5. https://doi.org/10.1098/rsbl.2022.0055
Torma A, Gallé R, Bozsó M (2014) Effects of habitat and landscape characteristics on the arthropod assemblages (Araneae, Orthoptera, Heteroptera) of sand grassland remnants in Southern Hungary. Agric Ecosyst Environ 196:42–50. https://doi.org/10.1016/j.agee.2014.06.021
Torma A, Bozsó M, Gallé R (2018) Secondary habitats are important in biodiversity conservation: a case study on orthopterans along ditch banks. Anim Biodivers Conserv 41(1):97–108. https://doi.org/10.32800/abc.2018.41.0097
Uthes S, Matzdorf B (2013) Studies on agri-environmental measures: a survey of the literature. Environ Manag 51:251–266. https://doi.org/10.1007/s00267-012-9959-6
Vadkerti E, Szövényi G (2005) Habitat preference of four protected bush-cricket species (Orthoptera, Phaneropteridae, Isophya) in South Hungary. Biol Bratislava 60(5):545–549
Vandermeer J, Perfecto I (2007) The agricultural matrix and a future paradigm for conservation. Conserv Biol 21(1):274–277. https://doi.org/10.1111/j.1523-1739.2006.00582.x
Whittaker RH (1960) Vegetation of the Siskiyou Mountains. Oregon California Ecol Monogr 30(3):279–338. https://doi.org/10.2307/1943563
Funding
Open access funding provided by University of Debrecen. This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors. The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Contributions
DAJ and AN designed the experiment and analysed the data, all authors contributed to writing of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Research involving human participants and/or animals
This article does not contain any studies with human participants performed by any of the authors. All applicable international, national, and/or institutional guidelines for the care and use of animals were followed.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Arnóczkyné Jakab, D., Nagy, A. How can an intensively used agricultural landscape preserve diversity of Orthoptera assemblages?. J Insect Conserv 26, 947–958 (2022). https://doi.org/10.1007/s10841-022-00439-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10841-022-00439-7