Abstract
This paper explores relations between assemblages of carpological remains and vegetation in and around a small, shallow reservoir in southern Poland. The study was conducted from 2006 to 2008. Quantity and distribution of species in the reservoir were recorded annually during the growing season. In October 2008, 40 samples of surface sediment (top 2 cm) were collected along transects at 10 m intervals. Samples of 100 cm3 were prepared for analysis of plant macroremains. Assemblages of carpological remains generally reflect local vegetation well. In some cases, however, even analysis of numerous samples failed to fully capture the species composition or reflect plant ratios in the parent phytocenosis. Reasons for this include factors that affect seed production, transport and fossilization, which differ among species. Among the best-represented macroremains were plants of the rush phytocenosis. In analysed samples, macroremains of 68.8 % of extant rushes were identified. Sixty percent of submerged and floating-leaf taxa were found in carpological samples, whereas 26.7 % of the trees and bushes were represented in sediment deposits. Species composition of phytocenoses in the reservoir and in surrounding areas was best reflected by macroremains from the nearby reed bed. Numbers of diaspores of Mentha aquatica, Hippuris vulgaris and Carex reflected well their relative abundance in phytocenoses. Chara sp., Juncus inflexus and Eupatorium cannabinum were overrepresented, whereas Typha latifolia and Sparganium minimum were poorly represented in relation to contemporary plant cover. There were no diaspores of Phragmites australis, which dominates the contemporary reed bed. Besides the shape of a reservoir, the key factor influencing diaspore numbers is distribution of plant cover. In many cases, single diaspores (Potentilla erecta, Myosotis scorpioides, Lythrum salicaria, Scutellaria galericulata), or higher concentrations (Hippuris vulgaris, Mentha aquatica, Eleocharis palustris, Schoenoplectus tabernaemontani, Chara sp.) reflected well the location of parent vegetation. The findings indicate that carpological remains in sediments can be an important source of information about plants in and around lakes. They generally reflect well local vegetation and in some cases may be used to identify taxa that dominated in the past.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Vegetation growing in and around reservoirs influences the entire aquatic ecosystem. Plants provide habitat and are a main source of organic matter, trapping sediments and influencing the chemical composition of water (Barko and James 1998; Wetzel 2001). Furthermore, the distribution and composition of phytocenoses reflect habitat conditions in the reservoir and its surroundings. Plant macroremains, especially carpological remains, which survive well in lacustrine deposits, can thus be used to infer past changes in phytocenoses (Sayer et al. 1999; Odgaard and Rasmussen 2001) and the environment. Factors that can be inferred include changes in temperature (Kolstrup 1979; Isarin and Bohncke 1999), trophic state (Jackson and Charles 1987; Scheffer et al. 1993) and water level (Digerfeldt 1986; Hannon and Gaillard 1997; Dieffenbacher-Krall and Nurse 2005; Koff et al. 2005).
There exists great potential for interpreting plant macroremains (Dieffenbacher-Krall 2007), especially in cases of human modification of the environment (Rasmussen and Anderson 2005). Plant macroremains are rich sources of data for palaeoenvironmental reconstruction and supplement palynological analyses (Birks and Birks 2000; Tobolski 2000; Birks 2007). In lakes, macroremains usually reflect local vegetation (Birks 1980; Tobolski 2000) and can be identified to species level more often than can pollen grains (Birks 1980, 2007). This frequently allows for a more complete identification of species than with palynological analysis, enabling better paleoenvironmental reconstruction. Use of plant macroremains for reconstructing species composition, establishing spatial distribution of phytocenoses and determining dominant species, requires knowledge of complex relations between macrofossil assemblages and contemporary vegetation (Birks 2001; Dieffenbacher-Krall 2007). Such studies of these relations employ samples from the topmost layer of bottom deposits (Dieffenbacher-Krall 2007) and permit better interpretation of macrofossil records (Zhao et al. 2006). Studies of plant macrofossil taphonomy have concentrated on representation of terrestrial (Dunwiddie 1987; Wainman and Mathewes 1990) and aquatic taxa in bottom deposits (Birks 1973; Davis 1985; Dieffenbacher-Krall and Halteman 2000; Zhao et al. 2006; Koff and Vandel 2008). Results suggest that assemblages of macroremains generally reflect well the composition of contemporary vegetation. Whereas the dominant species can often be identified in historical studies, a full picture of composition of past phytocenoses is impossible to obtain. Information from surface sediments also enables researchers to plan paleolimnological research, and make decisions about lake selection, sampling sites and sample numbers (Dieffenbacher-Krall 2007).
This project was undertaken to determine how well assemblages of carpological macroremains found in the top 2 cm of sediment from a reservoir in southern Poland reflect contemporary vegetation. Another goal was to establish relations between contemporary phytocenoses and the distribution and amount of carpological macroremains, to assist paleolimnological interpretation. Most such research has been conducted on small, relatively shallow lakes (Zhao et al. 2006; Koff and Vandel 2008). The present study was carried out in a very shallow reservoir that, with the exception of a small area, is <1 m deep. The key factor controlling distribution of diaspores in the reservoir is the structure and spatial distribution of vegetation.
Materials and methods
Selection and characteristics of the reservoir
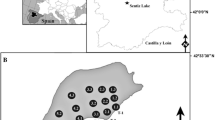
The water body selected for this study is a shallow reservoir in southern Poland (Fig. 1a). It formed from the damming of outflow from a small spring ca. 1900, which filled a former oxbow lake basin. The reservoir possesses submersed aquatic plants (10 spp.) and emergent reed vegetation (32 spp.). It contains large populations of Hippuris vulgaris and Sparganium minimum (Table 1), species that are now rare in Poland (Kłosowski and Kłosowski 2006), but are often well represented in late Pleistocene and Holocene deposits. The reservoir surface area is 2.45 ha. Maximum water depth, recorded in isolated holes, is 1.2 m (Fig. 1b) and the bottom is quite flat. Low, but stable flow from a spring maintains a constant water level. Except in the vicinity of the inflow and outflow, water flow is very slow, likely precluding diaspore transport. The reservoir is protected from strong winds and waves by high banks on the north shore and by high trees elsewhere. Wind is further attenuated by a reed belt. Shallow water and high transparency provide optimum conditions for vegetation development across the entire lake bottom. Moreover, these characteristics enable observation and collection of samples from a small boat. Because the reservoir bottom is muddy and access to open water is largely prevented by the wide reed belt, the water body is not subject to anthropogenic pressure, which might otherwise spread plants and their diaspores, as well as mix the topmost sediment layers. Nearby ruderal habitats and transportation routes may, however, introduce diaspores of exotic species to the local aquatic phytocenoses. Biota that might influence dispersal of plants and their diaspores include fish and plant-eating water birds such as Anas platyrhynchos L., Fulica atra L., Gallinula chloropus L. and Cygnus olor Gmelin.
Vegetation research (2006–2008)
Species composition, quantity and distribution were studied from 2006 to 2008. The reservoir was observed and sampled from a dinghy. Reed beds and submersed aquatic vegetation were analysed annually. Distributions of trees and bushes up to 50 m from shore were recorded graphically. To study submerged and floating-leaf taxa, 14 transects perpendicular to the shoreline were established every 10 m within the lake (Fig. 1b). Every 10 m along each transect a control point with a radius of 1 m was delimited. To identify small patches of Potamogeton natans, Persicaria amphibia and Chara sp., control points were placed every 5 m. Species composition was determined in the middle of each July using flora censuses along the transects. To record single examples of new species, spaces between control points were also sampled. Simultaneously, a general assessment of the total amount of individual species was conducted. This was accomplished using DAFOR, a five-level frequency scale (Palmer et al. 1992). In 2008, the Braun-Blanquet scale (Barbour et al. 1987) was used to assess percent cover for individual aquatic species at control points (Fig. 1b). Depth was measured along transects to enable creation of a bathymetric map. To record changes in the species composition of reed bed vegetation, the researchers travelled along the edge of reed vegetation, observing species in the vicinity of open water.
Collection of sediment samples and macrofossil analysis
In mid-July 2008, 40 samples of the topmost 2 cm of sediment were collected at the selected control points (Fig. 1b) using a modified “Kayak” sampler (Aaby and Digerfeldt 1986). According to previous studies (Dieffenbacher-Krall and Halteman 2000; Zhao et al. 2006) this layer should represent modern vegetation. Samples were collected to represent the shoreline zone, the central part of the reservoir and the small, deeper depression. The sample volume used for macrofossil analysis was 100 cm3. Fresh deposits were rinsed on a 0.2-mm-mesh sieve to separate diaspores. Carpological remains were identified using keys and atlases (Katz et al. 1965; Berggren 1969; Cappers et al. 2006). It was assumed that all seeds of Juncus came from Juncus inflexus, which grows in the belt of reeds, despite the fact that single Juncus articulatus L. grow in the vicinity, at a distance from the water.
Analysis of the relations between contemporary vegetation and macroremains
Plants were divided into three groups: submerged and floating-leaf taxa, reed bed plants, and trees and shrubs. Reed bed species included not only common taxa, but also several accompanying species. Submerged and floating-leaf taxa included Sparganium minimum and Hippuris vulgaris. These taxa outnumbered other totally submerged plants and were distributed throughout the entire reservoir.
Deposit samples were divided into two groups of 20 each. They represented the sediments of the shoreline zone near the reed bed and the central part of the lake basin (Fig. 1b). The shoreline samples were collected from points up to 5 m from the reed bed or from other larger vegetation patches. In most cases, however, the distance was <2 m. Sites with statistically significant concentrations of macroremains, with respect to species represented, were selected using the method developed by Doi (Runge 2006), which enables one to identify the dominant elements. The coefficient of similarity (Sørensen 1948) was calculated for species composition of macroremain assemblages and various groups of plants. In calculating the percentage of reed bed species in the macroremain assemblages, unidentified species of sedges and Carex rostrata perigynia were not included.
Results
Monitoring of the reservoir vegetation showed high stability of phytocenoses. This mainly refers to the reed bed habitat, where there was for the most part, no significant change in species composition for 3 years. Only Mentha aquatica and Eleocharis palustris displayed a significant increase in amount relative to 2006. Species composition in submerged and floating-leaf taxa underwent slight changes (Table 1). Generally, during the research period, there were nine species of vascular plants and Chara sp. Five taxa and Charales were recorded in the reservoir during all research seasons. A significant increase in numbers, in relation to 2006, was recorded for Hippuris vulgaris and Potamogeton natans. In the area surrounding the reservoir, there were 15 species of trees and bushes (Table 1). In 2008, the largest patches of vegetation had Hippuris vulgaris and Sparganium minimum. In places, they were accompanied by small phytocenoses of Potamogeton natans, Persicaria amphibia and Chara sp. Myriophyllum spicatum, Potamogeton crispus, Potamogeton pectinatus and Ceratophyllum demersum were represented by single specimens. Reed vegetation was mostly composed of Phragmites australis, Carex acutiformis, C. vulpina and C. rostrata. Smaller patches of vegetation in the reed bed habitats were composed of Typha latifolia, Schoenoplectus tabernaemontani, Eleocharis palustris, Acorus calamus, Glyceria maxima, Menyanthes trifoliata and Mentha aquatica. On the live and dead patches of Carex vulpina on the edge of the reed bed, single specimens of Myosotis scorpioides, Potentilla erecta, Lythrum salicaria, Myosoton aquaticum and Scutellaria galericulata grew; only Lycopus europaeus was more frequent. Trees in the vicinity of the reservoir were predominantly represented by Betula pendula and Alnus glutinosa. Among bushes, the most frequent were Sambucus nigra, Rubus sp. and species of Salix. The distributions of the patches of the selected plant species and their level of coverage in the area are represented in Fig. 2a, b.
Representation of the modern flora and vegetation in macroremain assemblages
A total of 1,366 carpological macroremains of vascular plants of 34 taxa and 1,150 Chara sp. oospores were identified in the samples. The richest samples in terms of quantity and species diversity of the macroremains came from the shoreline zone. They represent 76.5 % of the total recorded macroremains and 94.1 % of all represented taxa (Table 1). Figure 2 shows the distribution and quantity of diaspores of selected species in the samples.
Carpological remains associated with the reed vegetation were found in 97.5 % of samples, 63.7 % of which were collected close to the shore (Table 1; Fig. 2b). In total, 22 taxa were identified, which represent 68.8 % of contemporary reed bed species. The similarity index of the species composition of phytocenoses of contemporary reed vegetation and its representation in the macroremains is 0.81. Among the reed bed diaspores, Carex nuts (n = 642) were dominant and represented 59.9 % of the reed bed species. Of the Carex nuts, 336 came from Carex rostrata, which were found in 87.5 % of samples. Mentha aquatica, Juncus inflexus, Eupatorium cannabinum and Lycopus europaeus (Table 1) were also well represented. Seeds of Phragmites australis, which today is the main reed bed element, were not found. Likewise, Typha latifolia was poorly represented in relation to its contemporary coverage.
Diaspores of submerged and floating-leaf taxa were found in 82.5 % of samples, 90.2 % of which were recorded in the samples collected near the shoreline. In total, six taxa were identified (five species of vascular plants and Chara sp.), which represent 60 % of contemporary aquatic plants of the reservoir. The similarity index for the species composition of phytocenoses of contemporary submerged and floating-leaf taxa and their representation in the assemblages of macroremains was 0.75. Among the macroremains, Chara sp. oospores dominated. They were recorded in 70 % of samples throughout the reservoir. Their occurrence, however, was limited to the vicinity of parent vegetation patches. Among carpological remains of vascular plants, the most numerous were endocarps of Hippuris vulgaris, which were recorded in 55 % of the samples (Table 1). In relation to the contemporary coverage, Sparganium minimum was poorly represented.
Carpological remains of trees and bushes were found in 67.5 % of the samples, the majority of which (66.2 %) occurred in the shoreline samples. Only four taxa were identified among the samples, which represented 26.7 % of this plant group (Table 1). The similarity index for the contemporary representation of trees and bushes and their representation in the assemblages of macroremains is 0.42. Among trees and bushes, the fruits of Betula pendula (50.2 %) dominated; they were recorded in 62.5 % of the samples. Less frequent were Alnus glutinosa fruits (41.8 %), found in 50 % of the samples.
Discussion
Relations between contemporary vegetation and distribution of carpological macroremains
Knowledge of the complex mechanisms of diaspore dispersal in lake basins enables one to locate parent phytocenoses and use carpological macroremains of some species for palaeohydrological inference (Birks 1973; Dieffenbacher-Krall and Halteman 2000). It is generally accepted that the best sample collection site for macrofossil analysis is a shallow littoral location, near the lakeshore (Tobolski 2000; Dieffenbacher-Krall 2007). Previous studies of the relation between modern vegetation and assemblages of macroremains indicate samples collected in this zone include the largest number of carpological remains and highest diversity of species (Birks 1973; Dieffenbacher-Krall and Halteman 2000; Zhao et al. 2006; Koff and Vandel 2008). In this study, concentrations of macroremains in the zone near the reed bed, support earlier observations. This is a consequence of the fact that most plants found there produce floating diaspores. Among the taxa often represented in shoreline samples are Carex rostrata, C. acutiformis, C. vulpina, Menyanthes trifoliata, Lycopus europaeus, Persicaria amphibia, Rumex hydrolapathum, Sparganium erectum, Ranunculus lingua, Potamogeton natans, as well as Alnus glutinosa and Betula pendula. Such diaspores can be blown around the reservoir. Frequently, they are pushed toward the shore and sink. Their ability to float is limited only by the shape of the lake and distribution of emergent vegetation patches that trap them.
In small, shallow reservoirs, the influence of vegetation on dispersal of floating diaspores is large, because such lakes generally support lush plant growth. Such relations are well illustrated by the distribution of Potamogeton natans remains in the reservoir. All samples with statistically significant concentrations of endocarps of this species were located at the lakeshore, where contemporary P. natans phytocenoses are found (Fig. 2a). Movement of fruits from this species is probably inhibited by the patches of Hippuris vulgaris, with emergent shoots, in the central part of the lake. There is probably a similar influence on Menyanthes trifoliata, whose seeds can only drift along the open channel, free from emergent Hippuris vulgaris (Fig. 2a). This is supported by their highest concentration in sample 13. The influence of the distribution of the contemporary H. vulgaris phytocenoses on migration of drifting diaspores also applies to Glyceria maxima. Observations indicate that Glyceria maxima diaspores stick to emergent parts of plants and are trapped. This is probably why they were found at a maximum distance of 13 m from parent plants, mainly at the edge of the emergent vegetation belt. The highest concentration of Glyceria maxima diaspores was found in sample 39, collected in a small bottom depression. The large abundance might have resulted from seed redeposition. Concentration of carpological remains in depressions within overgrown parts of the bottom was observed for other, mainly small diaspores, of Eupatorium cannabinum, Juncus inflexus, Mentha aquatica and Betula pendula, but also the relatively larger and heavier nuts of Carex.
Translocation of diaspores along the bottom of small lakes, in areas where vegetation is lacking, and their concentration in holes, may be a consequence of even the slightest water movements caused by ducks, swans and even fish. In the case of Carex species, which grow along the entire lakeshore, determining the source of diaspores is impossible. Similar to other lakes (Wainman and Mathewes 1990), their transport is limited by emergent vegetation, and is reflected by the high concentrations of nuts in samples neighbouring the reed bed and in a patch of emerged Hippuris vulgaris. Similar to findings of other studies (Birks 1973; Dieffenbacher-Krall and Halteman 2000; Zhao et al. 2006), in this lake, patches of parent vegetation are best reflected by diaspores of Myriophyllum spicatum, Scutellaria galericulata, Myosotis scorpioides, Lythrum salicaria and Potentilla erecta, that float briefly and then sink. These taxa were recorded up to 3 m from parent plants on the reed bed edge and in Carex vulpina patches. Fruits of Eleocharis palustris and Schoenoplectus tabernaemontani were more dispersed. Although most of their diaspores were recorded up to 7 m from patches of parent vegetation in neighbouring open water, single diaspores were found at greater distances from source patches: E. palustris at 26 m and S. tabernaemontani at 23 m (Fig. 2a). Thus, the indicator value of their seeds should be carefully interpreted. It might be assumed that only high concentrations of their remains indicate the location of the parent phytocenoses. A similar conclusion was suggested by Dieffenbacher-Krall and Halteman (2000). In their study, the number of carpological remains of Eleocharis palustris in samples was higher close to the parent plants.
Results and field observations indicate that differences in dispersal of quickly sinking diaspores of Scutellaria galericulata, Myosotis scorpioides, Lythrum salicaria, Potentilla erecta, as well as E. palustris and S. tabernaemontani, depend on how overgrown the lake bottom is in the vicinity of parent vegetation patches. In bottom areas near patches of E. palustris and S. tabernaemontani, which lack vegetation, translocation by microcurrents of single fruits that sink outside the reed bed is promoted. Diaspores of Myosotis scorpioides, L. salicaria, S. galericulata and P. erecta, however, which sink onto an overgrown bottom, are effectively trapped. Although rapidly sinking fruits of Mentha aquatica were recorded in numerous places around the entire reservoir, most samples (71.5 %) of statistically significant concentration were located <10 m from fruiting specimens (Fig. 2b). Endocarps of Hippuris vulgaris were similarly dispersed, but as in the case of M. aquatica, samples with statistically significant concentrations were located within patches of contemporary vegetation or at a distance of <10 m from fruiting specimens. They reflected well the location of emergent forms of H. vulgaris, thus indicating lake shallowness.
Oospores of Chara sp. were also recorded in the entire lake, although their larger patches were found only in the northern section of the reservoir. Similar to findings in shallow Lake Veluwemeer (van den Berg 1999) or Green Plantation Pond (Zhao et al. 2006), highest concentrations in the sediment (>200 remains/100 cm3) were recorded only in places where algae grew at the time of sampling (Fig. 2a). This strengthens the claim that a high concentration of oospores is indicative of the location of the parent phytocenoses. Nevertheless, dispersal of oospores can result because of their small size and mass. They can be easily transported by slight water movements, especially in areas of shallow reservoirs devoid of vegetation. Oospores can also be transported by birds that eat algae (Proctor 1959).
It can be difficult to determine the source of diaspores of Alnus glutinosa and Betula pendula, growing around the lake. A comparison of the distribution of vegetation and A. glutinosa trees, and samples with statistically significant concentrations of diaspores of those species in the sediments, suggests that most remains come from trees that grow along the shore and even shade the lake. Fruits of A. glutinosa fly poorly and only disperse around the lake after falling in the water. They float well and their ability to translocate may only be limited by a reed bed or belt of emergent vegetation. Most samples with statistically significant fruit concentrations were located in the southwestern and western part of the reservoir, in the vicinity of patches of Alnus glutinosa, growing along the lakeshore. These samples were located on the inner side of patches of Hippuris vulgaris, which blocked diaspore dispersal. This indicates an effective barrier to fruit influx from trees growing on the northern and northeastern lakeshore by a wide (up to 85 m) belt of reeds in the northeast. Fruits of Betula pendula are transported much farther by the wind and similar to A. glutinosa, it is difficult to infer their origin. They may fall within the entire lake basin, including into patches of emergent vegetation, as indicated by the large number of samples containing carpological remains of this species (65 % of the total number) across the reservoir. Sampling distribution of statistically significant concentrations of macroremains also suggests that fruits of B. pendula drift easily on the water and become concentrated in the lakeshore zone, similar to findings in Estonian Lakes Juusa and Viitna (Koff and Vandel 2008). Presence of Sambucus nigra and Rubus sp. in macroremain assemblages, i.e. taxa growing in the immediate vicinity of the reservoir, may be explained by feeding birds that are capable of transporting fruits above the reed bed.
Relations between contemporary vegetation and its representation in the assemblages of carpological remains
Research on relations between assemblages of macroremains and contemporary vegetation (Birks 1973; Davis 1985; Dunwiddie 1987; Wainman and Mathewes 1990; Dieffenbacher-Krall and Halteman 2000; Davidson et al. 2005; Zhao et al. 2006; Koff and Vandel 2008), or seed banks (Kautsky 1990; Combroux et al. 2001) revealed that assemblages of carpological macroremains generally reflect well species composition of parent phytocenoses. Representation of contemporary vegetation in taphocenoses, however, varies among individual lakes.
In the study reservoir, reed vegetation was best represented (68.8 %) in the assemblages of macroremains, though diaspores of the dominant Phragmites australis were absent. Diaspores of some species may be under-represented if they were deposited deep in the reed bed where no samples were collected. High representation of reed bed taxa, with the exception of P. australis, is a consequence of high production and preservation of diaspores from taxa like Carex and Mentha aquatica (Table 1). Poorly represented species include those that do not produce seeds in the Polish climate, e.g. Acorus calamus (Kłosowski and Kłosowski 2006), or whose diaspores are too delicate to be preserved (e.g. Phalaroides arundinacea). A similar relation between the representation of reed vegetation in the assemblages of carpological remains and species composition of contemporary reed vegetation was found in sediments of Lakes Juusa and Viitna (Koff and Vandel 2008). They also found poor representation of reed vegetation (33 %) in the deposits. In the case of the studied reservoir, one reason for the lack of diaspores of abundant Phragmites australis may be their delicate structure. This leads to their rapid decomposition, favoured by high oxygen in the shallow water. Koff and Vandel (2008) also indicated poor representation of diaspores of P. australis in the sediments of one lake, though this species is presently common in the reed beds of both lakes. The study reservoir and the two Estonian lakes indicate that lack of carpological remains of P. australis cannot be used to indicate absence of this species. The role of this taxon in contemporary phytocenoses requires analyses of palynological and vegetative remains. The quantity of carpological remains of Carex and Mentha aquatica reflects well their important roles, after Phragmites australis, as reed bed elements. In terms of quantity, they are the best-represented reed bed plants in the macroremain assemblages (Table 1). Other relatively well-represented species include Schoenoplectus tabernaemontani, Glyceria maxima, Ranunculus lingua, Menyanthes trifoliata and Lycopus europaeus, which are not common today. In contrast, the large amount of seeds of Juncus inflexus does not correspond with the small contemporary population of this species, limited to single patches. This may be a consequence of the high production of seeds by Juncus (Birks 2007). A similar conclusion probably applies to the relatively high representation of diaspores of Eupatorium cannabinum. Their reed bed population is limited to a few dispersed specimens. To some extent, this also applies to Epilobium hirsutum and Rumex hydrolapathum. A reverse situation is recorded for Typha latifolia, which forms clustered patches deep inside the reed bed, but is represented by only two fruit fragments. As this species produces numerous flying diaspores, they may be blown above the surrounding vegetation. Such poor representation might also result from delicate fruits quickly decomposing. As observed in some reservoirs, however, ripe T. latifolia fruits glue together and get washed in by rain.
Most diaspores may have remained in the immediate vicinity of plants, where no samples were collected. Such a thesis is supported by the research in Estonian lakes (Koff and Vandel 2008) and Birks’s observations (1973). They suggest the fruits of T. latifolia are only recorded in marginal samples. Deposition in the vicinity of parent plants may also explain poor representation of Eleocharis palustris, in relation to contemporary cover. The same refers to the lack of diaspores of single specimens of Epilobium palustre deep inside the reed bed. In other lakes, its fruits were concentrated near the shore, in water up to 125 cm deep (Greatrex 1983), and Caltha palustris, Galium uliginosum, Oenanthe aquatica, Sagittaria sagittifolia, Scrophularia umbrosa and Scirpus sylvaticus were rare and dispersed in reed beds. Insignificant representation of diaspores of Alisma plantago − aquatica, Lythrum salicaria, Myosoton aquaticum, Potentilla erecta, Scutellaria galericulata, and Sparganium erectum is probably related to their contemporary poor representation. Considering the area presently taken up by patches of individual species of sedges in relation to other taxa, there are few C. acutiformis nuts (Table 1), although it takes up a larger area than C. rostrata or C. vulpina (Fig. 2b). This probably results from the fact that C. acutiformis is protected by a C. rostrata belt and thus has limited contact with open water (Fig. 2b). This causes their diaspores to be trapped within patches of parent vegetation. Thus, species composition of sedges in the macroremain assemblages beyond the reed bed reflects the proportions of individual species of the inner reed belt that are in contact with open water. The large number of diaspores of reed vegetation (27/sample on average) and species diversity in samples, is probably determined by contemporary species composition. An important factor is also the large amount of diaspores delivered beyond the reed bed, favoured by a long span where the reed bed is in contact with open water (Fig. 1b), and existence of C. vulpina patches that are habitat for species predominantly growing deep inside the reed bed zone.
Submerged and floating-leaf taxa were significantly more poorly reflected in assemblages of macroremains than reed vegetation (Table 1). This probably reflects the fact that they generally produce fewer diaspores and also display small amounts of their species in contemporary phytocenoses. Representation of submerged and floating-leaf taxa is similar to that reported by Koff and Vandel (2008) for Lakes Juusa and Viitna (50 %), but higher than in Groby Pool, where it was 40 % (Davidson et al. 2005), or in Green Plantation Pond where it was 16 % (Zhao et al. 2006). The reason for such discrepancies is the quantity of individual species and different species composition of phytocenoses. The samples do not contain Ceratophyllum demersum, Batrachium circinatum, Potamogeton crispus and P. pectinatus, and representation of diaspores of Myriophyllum spicatum and Persicaria amphibia is small, which may be linked to their tiny proportion in the contemporary phytocenoses.
Dominance of oospores of currently rare Chara sp. (Table 1) among the recorded macroremains is interpreted differently. Their over-representation, also noted by others (Kautsky 1990; Steinhardt and Selig 2007), is a consequence of their large production of oospores (Bonis and Grillas 2002). They create a rich bank in the deposits (van den Berg 1999). The share of endocarps of Hippuris vulgaris among diaspores of vascular plants correctly indicates domination of this species. With regard to the relatively small number of recorded diaspores, if compared to contemporary coverage, this is possibly the consequence of an exceptionally low number of fruiting specimens of Sparganium minimum. During the 2008 season, only 24 were observed. Low fruit production by phytocenoses of H. vulgaris in the studied reservoir is probably a consequence of the high proportion of vegetatively reproducing submerged forms of the taxon, which makes up about 65 % of the population.
Ability of this vegetative form to develop large phytocenoses (Barrat-Segretain and Bornette 2000) may be one of the reasons that Hippuris vulgaris is often represented by single endocarps in macroremain analyses (Birks 2000; Boyd 2007; Ayres et al. 2008). On rare occasions, 100 cm3 of sediment include them (Wohlfarth et al. 2006; van der Ham et al. 2008), and sporadically there are >10 (Bittmann 2007). Assuming that some species that coexist with H. vulgaris produce more diaspores that are preserved in deposits, the proportion of H. vulgaris in past phytocenoses may be underestimated. Such an assessment is not resolved by palynological analysis because this pollen is often poorly represented or missing (Wohlfarth et al. 2006).
Lake sediments often include numerous light diaspores of anemochorous trees. The best represented are Alnus glutinosa and Betula spp. (Tobolski 2000; Birks 2007). Heavier diaspores of other species growing nearby are rare. Similarly, in this reservoir, diverse tree and bush species composition in the vicinity is poorly reflected by assemblages of macroremains. Most tree and bush diaspores include Alnus glutinosa and Betula pendula (Table 1). Although A. glutinosa is represented by large numbers of trees that grow near the shore, sediments are dominated by lighter fruits of B. pendula, represented in the vegetation by only a few individuals. This apparent anomaly is probably caused by concentration of poorly flying A. glutinosa diaspores in a wide reed belt at the lake margin (Fig. 1b). Sediments lack light seeds from multiple species of Salix that live in the lake vicinity. This is difficult to explain because their numerous seeds were evenly distributed in other lakes (Koff and Vandel 2008). The probable reason for their absence in the reservoir deposits is rapid decomposition under high oxygen conditions. It is also possible that seeds of Salix, which are adapted for flying, fell onto the water surface and were blown deep inside the reed belt. Poor representation of remaining trees and bushes, especially heavy seed producers and terrestrial plants, may be attributed to the trapping of diaspores by reed vegetation. This is supported by research on Lakes Juusa and Viitna, where most diaspores of ruderal species were recorded in lakeshore samples (Koff and Vandel 2008).
Conclusions
The study showed that dispersion of diaspores in a very shallow reservoir was predominantly influenced by distribution of vegetation patches. Reed beds and emergent shoots of aquatic plants trap drifting seeds and fruits, limiting their transport and causing them to be concentrated along the edges or within patches of vegetation, even in the central part of the lake. Inadequate sampling of such a reservoir may decrease the probability of recording species whose ranges within the lake are limited. As a consequence of limited diaspore migration, however, assemblages of macroremains in such shallow lakes may reflect well the location of parent vegetation. In the case of the studied reservoir, the location of parent vegetation was best reflected by fast sinking diaspores of Myriophyllum spicatum, Lythrum salicaria, Myosotis scorpioides, Potentilla erecta and Scutellaria galericulata. In the case of the diaspores of Schoenoplectus tabernaemontani, Eleocharis palustris, Mentha aquatica and Hippuris vulgaris, a good indicator of the location of patches of parent vegetation was a statistically significant concentration of diaspores. Contemporary locations of algae (Chara sp.), similar to results found in other studies (van den Berg 1999; Zhao et al. 2006), were only indicated by significantly higher concentrations of oospores, which in this study amounted to >200/100 cm3. The distribution of fruits of Alnus glutinosa suggests they come from trees growing within 85 m of the water body. Diaspores of some plants were found in bottom areas devoid of vegetation and were concentrated in lake-bottom depressions. This phenomenon applied mostly to small, light diaspores of Juncus inflexus, Glyceria maxima, Eupatorium cannabinum, Mentha aquatica and Betula pendula. Transport may have been by water movements caused by ducks, swans or even fish. Species composition of phytocenoses in the reservoir and its vicinity was best reflected by macroremain assemblages from the nearby reed bed. Generally, 61.4 % of the extant species in and around the reservoir were represented in assemblages of macroremains. Among life-form groups, reed bed plants were best represented (total of 68.8 %), followed by submerged and floating-leaf taxa (total of 60 %), followed by trees and bushes (total of 26.7 %). Among the rushes, the share of fruits of Mentha aquatica and Carex in macroremains assemblages best reflected their role as principal elements, after the common reed (Phragmites australis), in contemporary phytocenoses. Domination of a specific species of Carex, however, could not be established by analysis of samples coming exclusively from beyond the reed bed zone. The number of carpological remains of individual species reflected accurately only the share of Carex species building the inner reed belt, neighbouring the open water. The numbers of diaspores of Schoenoplectus tabernaemontani, Ranunculus lingua, Menyanthes trifoliata and Lycopus europaeus corresponded relatively well with their representation in contemporary reed beds. However, Juncus inflexus and Eupatorium cannabinum, as well as Epilobium hirsutum and Rumex hydrolapathum to a lesser degree, were overrepresented. Carpological macroremains of reeds, the dominant form in rush vegetation, were missing. Moreover, relatively abundant representation of Typha latifolia and Eleocharis palustris in modern vegetation was not reflected in the number of recorded macroremains. In the group of submerged and floating-leaf taxa, contemporary domination of Hippuris vulgaris was well reflected by the number of diaspores. Sparganium minimum was poorly represented by macroremains, whereas Chara sp. remains were overrepresented.
Results of this study support the idea that carpological remains of plants can be important sources of information in paleolimnological investigations. They reflect local vegetation well and, in some cases can be used to determine the dominant taxa of the past. The data suggest, however, that even analysis of a large number of samples may not enable one to identify all members of the parent phytocenoses or determine relative abundances of the vegetation contributing macroremains. This is because individual species are influenced by many factors that ultimately control diaspore production, transportation and preservation. Thus, to obtain a complete picture of past phytocenoses, analysis of carpological remains should be complemented by analysis of pollen and preserved vegetative remains.
References
Aaby B, Digerfeldt G (1986) Sampling techniques for lakes and bogs. In: Berglund BE (ed) Handbook of holocen Paleoecology and Paleohydrology. John Wiley, Chichester, pp 181–194
Ayres KR, Sayer CD, Skeate ER, Perrow MR (2008) Palaeolimnology as a tool to inform shallow lake management: an example from upton great broad, Norfolk, UK. Biodivers Conserv 17:2153–2168
Barbour MG, Burk JH, Pitts WD (1987) Terrestrial plant ecology. Benjamin/Cummings, Menlo Park, p 634
Barko JW, James WF (1998) Effects of submerged aquatic macrophytes on nutrient dynamics, sedimentation, and resuspension. In: Jeppesen E, Søndergaard Ma, Søndergaard Mo, Christoffersen K (eds) The structuring role of submerged macrophytes in lakes. Ecological studies series 131. Springer, New York, pp 197–214
Barrat-Segretain MH, Bornette G (2000) Regeneration and colonization abilities of aquatic plant fragments: effect of disturbance seasonality. Hydrobiologia 421:31–39
Berggren G (1969) Atlas of seeds and small fruits of Northwest-European plant species, part 2 Cyperaceae. Swedisch Natural Science Research Council, Stockholm, p 107
Birks HH (1973) Modern macrofossil assemblages in lake sediments in Minnesota. In: Birks HJB, West RG (eds) Quaternary plant ecology. Blackwell Scientific Publications, Oxford, pp 173–189
Birks HH (1980) Plant macrofossils in quaternary lake sediments. Arch Hydrobiol 15:1–60
Birks HH (2000) Aquatic macrophyte vegetation development in Kråkenes lake, western Norway, during the late-glacial and early holocene. J Paleolimnol 23:7–19
Birks HH (2001) Plant macrofossils. In: Smol JP, Birks HJB, Last WM (eds) Tracking environmental change using lake sediments, terrestrial algal and siliceous indicators, vol 3. Kluwer, Dordecht, pp 49–74
Birks HH (2007) Plant macrofossil introduction. In: Elias SA (ed) Encyclopedia of quaternary science, vol 3. Elsevier, Amsterdam, pp 2266–2288
Birks HH, Birks HJB (2000) Future uses of pollen analysis must include plant macrofossils. J Biogeogr 27:31–35
Bittmann F (2007) Reconstruction of the Allerød vegetation of the neuwied basin, western Germany, and its surroundings at 12,900 cal b.p. Veg Hist Archaeobot 16:139–156
Bonis A, Grillas P (2002) Deposition, germination andspatio-temporal patterns of charophyte propagule banks: a review. Aquat Bot 72:235–248
Boyd M (2007) Early postglacial history of the southeastern assiniboine delta, glacial lake agassiz basin. J Paleolimnol 37:313–329
Cappers RTJ, Bekker RM, Jans JEA (2006) Digitale zadenatlas van Nederland (digital seed atlas of the Netherlands). Barkhuis Publishing & Groningen University Library, Groningen, p 502
Combroux I, Bornette G, Willby NJ, Amoros C (2001) Regenerative strategies of aquatic plants in disturbed habitats: the role of the propagule bank. Arch Hydrobiol 152:215–235
Davidson TA, Sayer CD, Bennion H, David C, Rose N, Wade PM (2005) A 250 year comparison of historical, macrofossil and pollen records of aquatic plants in a shallow lake. Freshw Biol 50:1671–1686
Davis FW (1985) Historical changes in submerged macrophyte communities of upper chesapeake bay. Ecology 66:981–993
Dieffenbacher-Krall AC (2007) Surface samples, taphonomy, representation. In: Elias SA (ed) Encyclopedia of quaternary science, vol 3. Elsevier, Amsterdam, pp 2367–2374
Dieffenbacher-Krall AC, Halteman WA (2000) The relationship of modern plant remains to water depth in alkaline lakes in New England, USA. J Paleolimnol 24:213–229
Dieffenbacher-Krall AC, Nurse AM (2005) Late-glacial and holocene record of lake levels of Mathews pond and whitehead lake. Northern Maine. USA. J Paleolimnol 34:283–309
Digerfeldt G (1986) Studies on past lake-level fluctuations. In: Berglund BE (ed) Handbook of Holocene Palaeoecology and Palaeohydrology. Wiley, Chichester, pp 127–143
Dunwiddie PW (1987) Macrofossil and pollen representation of coniferous trees in modern sediments from Washington. Ecology 68:1–11
Greatrex PA (1983) Interpretation of macrofossil assemblages from surface sampling of macroscopic plant remains in mire communities. J Ecol 71:773–791
Hannon GE, Gaillard M-J (1997) The plant macrofossil record of past lake-level changes. J Paleolimnol 18:15–28
Isarin RFB, Bohncke SJP (1999) Mean July temperatures during the younger dryas in Northwestern and Central Europe as inferred from climate indicator plant species. Quat Res 51:158–173
Jackson ST, Charles DF (1987) Aquatic macrophytes in Adirondack (New York) lakes: pattern of species composition in relation to environment. Can J Bot 66:1449–1460
Katz NJ, Katz SV, Kipiani MG (1965) Atlas i opredelitel’plodov i semyan, vstrechayushchikhsya v chetvertichnykh otlozheniyakh SSSR (Atlas and key to fruits and seeds occurring in the quaternary deposits of the soviet union). Nauka, Moscow, p 366
Kautsky L (1990) Seed and tuber banks of aquatic macrophytes in the Askö area, northern Baltic proper. Holarct Ecol 13:143–148
Kłosowski S, Kłosowski G (2006) Rośliny wodne i bagienne (Aquatic and marsh plants), Seria Flora Polski. MULTICO Oficyna Wydawnicza, Warszawa, p 333
Koff T, Vandel E (2008) Spatial distribution of macrofossil assemblages in surface sediments of two small lakes in Estonia. Est J Ecol 57:5–20
Koff T, Punning JM, Sarmaja-Korjonen K, Martma T (2005) Ecosystem response to early and late Holocene lake-level changes in Lake Juusa, southern Estonia. Pol J Ecol 53:553–570
Kolstrup E (1979) Herbs as July temperature indicators for parts of the Pleniglacial and the late–glacial in The Netherlands. Geol Mijnb 59:337–380
Odgaard B, Rasmussen P (2001) The occurrence of eggcocoons of the leech Piscicola geometra (L.) in recent sediments and their relationship with the remains of submerged macrophytes. Arch Hydrobiol 52:671–686
Palmer MA, Bell SL, Butterfield I (1992) A botanical classification of standing water in Britain: applications for conservation and monitoring. Aquat Conserv 2:125–143
Proctor VW (1959) Dispersal of fresh-water algae by migratory birds. Science 130:623–624
Rasmussen P, Anderson NJ (2005) Natural and anthropogenic forcing of aquatic macrophyte development in a shallow Danish lake during the last 7,000 years. J Biogeogr 32:1993–2005
Runge J (2006) Metody badań w geografii społeczno-ekonomicznej-elementy metodologii, wybrane narzędzia badawcze (Methods of research in socio-economic geography—the elements of methodology, selection of research tools). Publisher University of Silesia, Katowice, pp 327–332
Sayer C, Roberts N, Sadler J, David C, Wade PM (1999) Biodiversity changes in a shallow lake ecosystem: a multi-proxy palaeolimnological analysis. J Biogeogr 26:97–114
Scheffer M, Hosper SH, Mejer ML, Moss B, Jeppesen E (1993) Alternative equilibria in shallow lakes. Trends Ecol Evol 8:275–279
Sørensen TA (1948) A method of establishing groups of equal amplitude in plant sociology based on similarity of species content, and its application to analyses of the vegetation on Danish commons. Kongelige Danske Videnskabernes Selskabs Biologiske Skrifter Copenhagen 5:1–34
Steinhardt T, Selig U (2007) Estuarine Spatial distribution patterns and relationship between recent vegetation and diaspore bank of a brackish coastal lagoon. Estuar Coast Shelf Sci 74:205–214
Tobolski K (2000) Przewodnik do oznaczania torfów i osadów jeziornych (The Guide for the determination of peat and lake sediments). Vademecum Geobotanicum 2. PWN, Warszawa, p 508
van den Berg MS (1999) Charophyte colonization in shallow lakes: processes, ecological effects and implications for lake management. Thesis, Vrije Universiteit, Amsterdam, p 138
van der Ham RWJM, Kuijper WJ, Kortselius MJH, Van der Burgh J, Stone GN, Brewer JG (2008) Plant remains from the Kreftenheye formation (Eemian) at raalte, The Netherlands. Veg Hist Archaeobot 17:127–144
Wainman N, Mathewes RW (1990) Distribution of plant remains in surface sediments of Marion lake, southwestern British Columbia. Can J Bot 68:364–373
Wetzel RG (2001) Limnology, lake and river ecosystems. Academic Press, San Diego, p 1006
Wohlfarth B, Tarasov P, Bennike O, Lacourse T, Subetto D, Torssander P, Romanenko F (2006) Late glacial and Holocene palaeoenvironmental changes in the Rostov-Yaroslavl’ area, West Central Russia. J Paleolimol 35:543–569
Zhao Y, Sayer CD, Birks HH, Hughes M, Peglar SM (2006) Spatial representation of aquatic vegetation by macrofossils and pollen in a small and shallow lake. J Paleolimnol 35:335–350
Open Access
This article is distributed under the terms of the Creative Commons Attribution License which permits any use, distribution, and reproduction in any medium, provided the original author(s) and the source are credited.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 2.0 International License (https://creativecommons.org/licenses/by/2.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
About this article
Cite this article
Szymczyk, A. Relations between assemblages of carpological remains and modern vegetation in a shallow reservoir in southern Poland. J Paleolimnol 48, 503–516 (2012). https://doi.org/10.1007/s10933-012-9627-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10933-012-9627-0